Submitted:
03 December 2024
Posted:
04 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
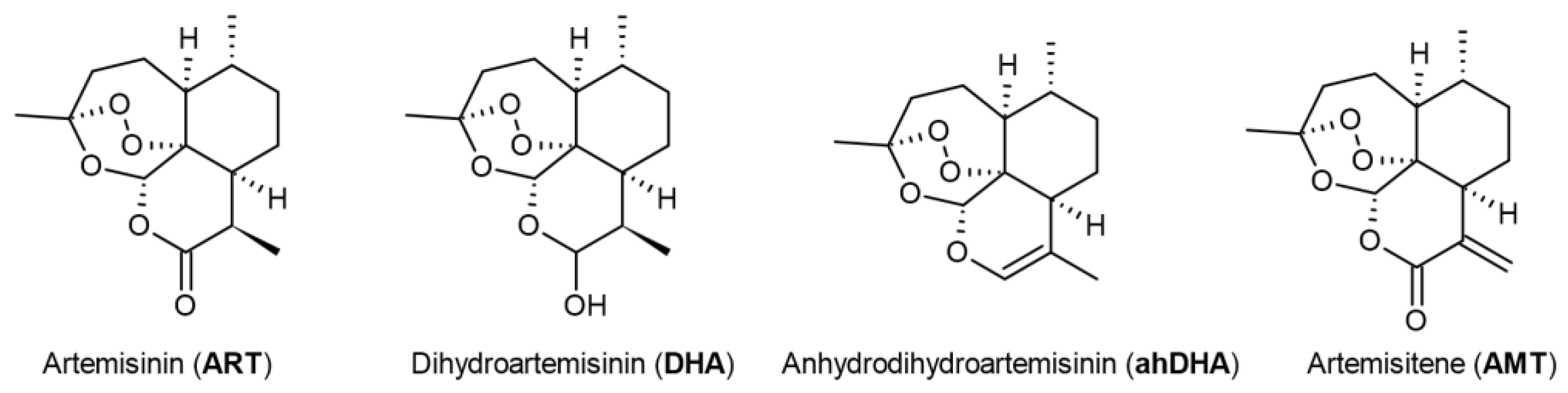
2.1. Preparation of Artemisinin Analogues with Modifications in the Lactone Cycle
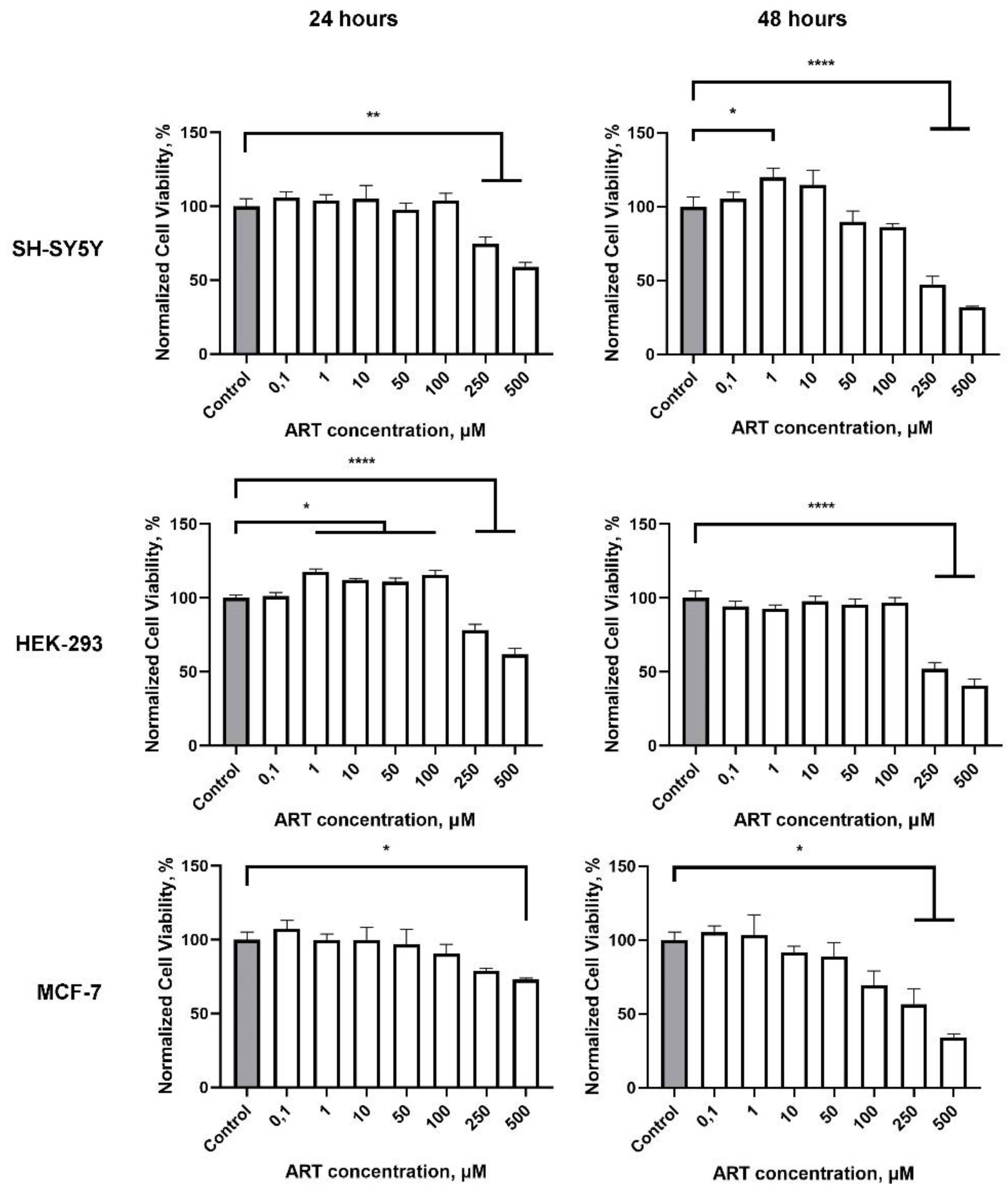
2.2. Artemisinin Increases Viability in SH-SY5Y and HEK-293 Cells, but Not in MCF-7 Cells
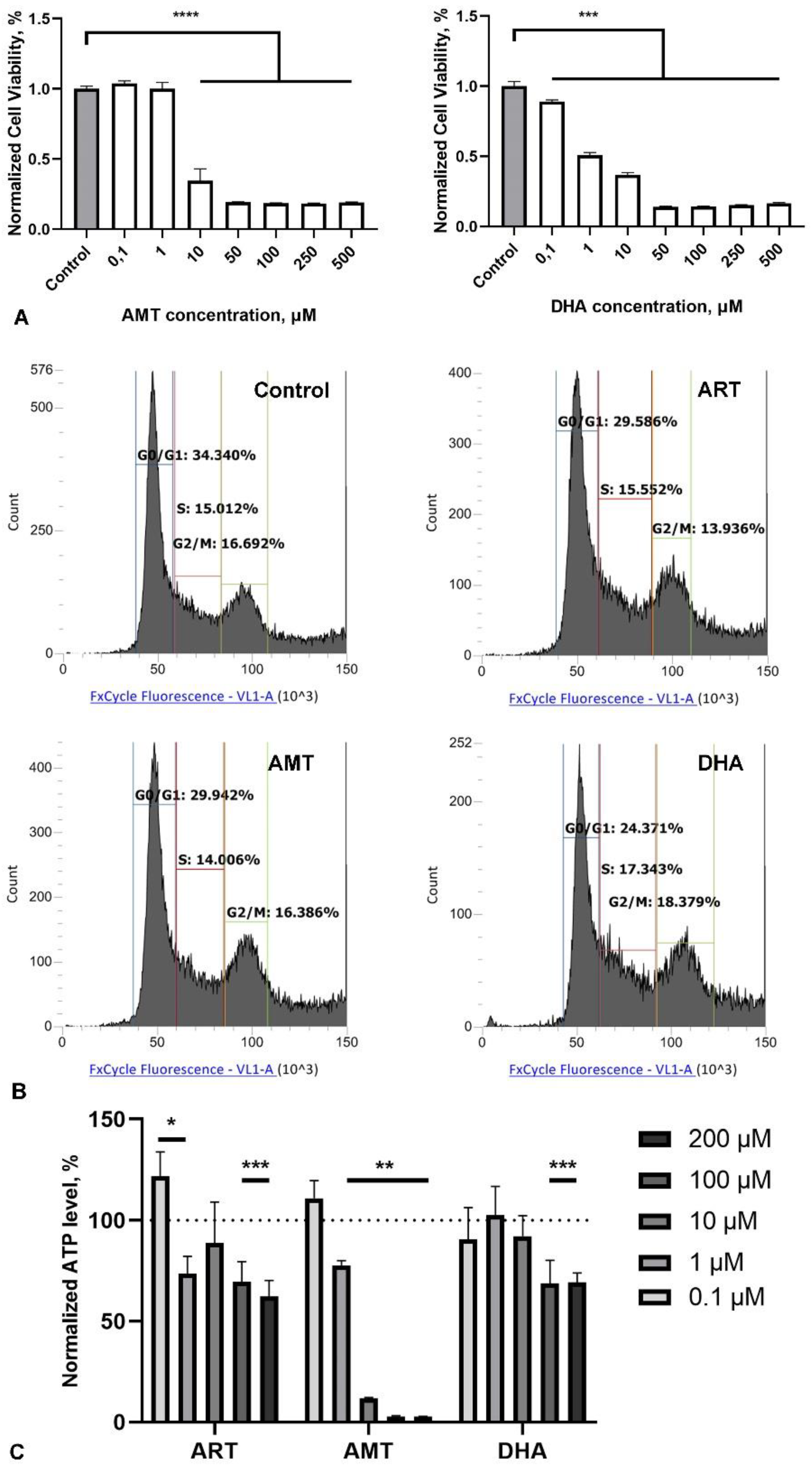
2.3. Artemisinin Does Not Affect the Cell Cycle, but Reduces ATP Levels
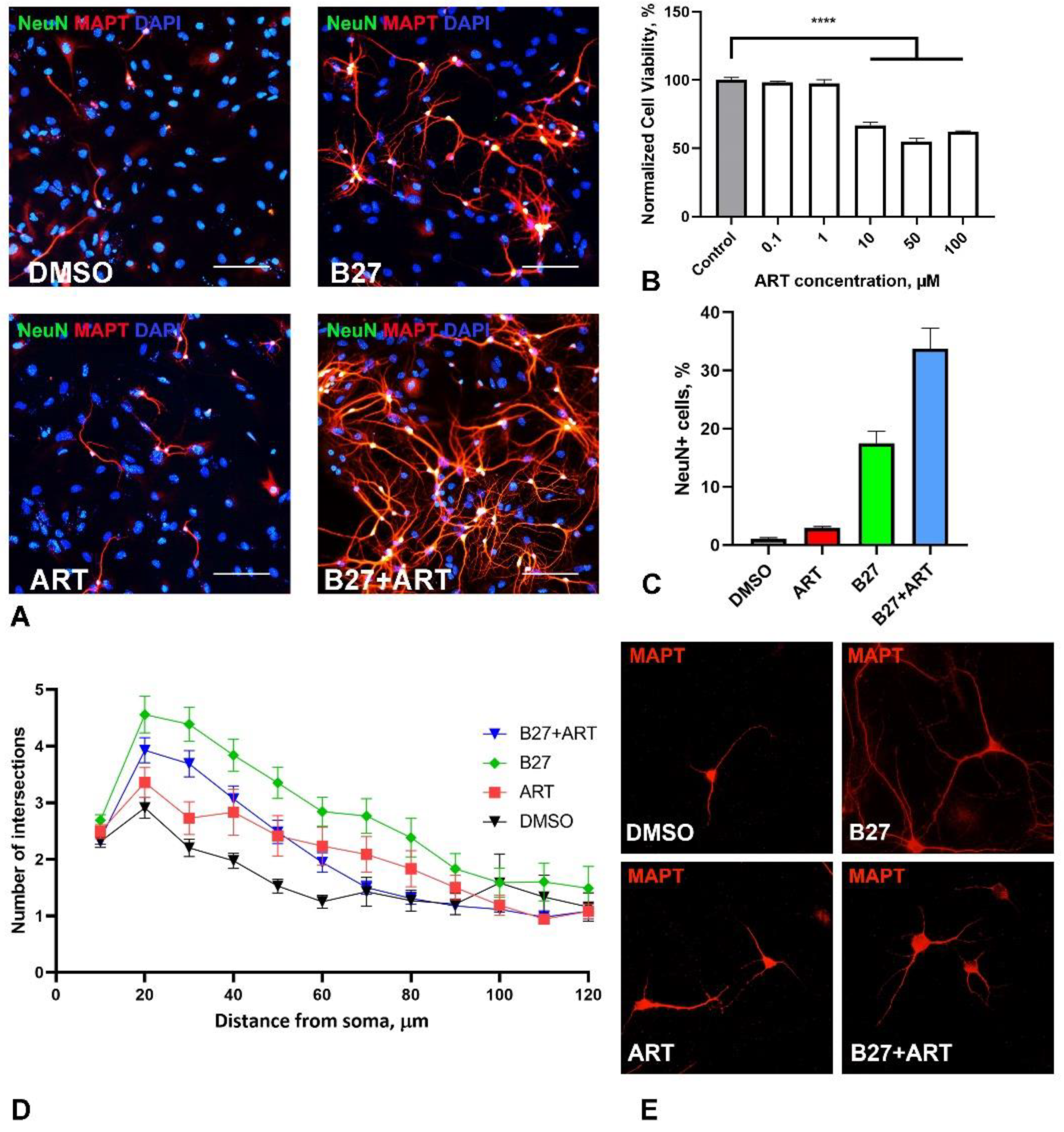
2.4. Artemisinin Increases Survival and Impact Branching of Neurons in Primary Hippocampal Culture
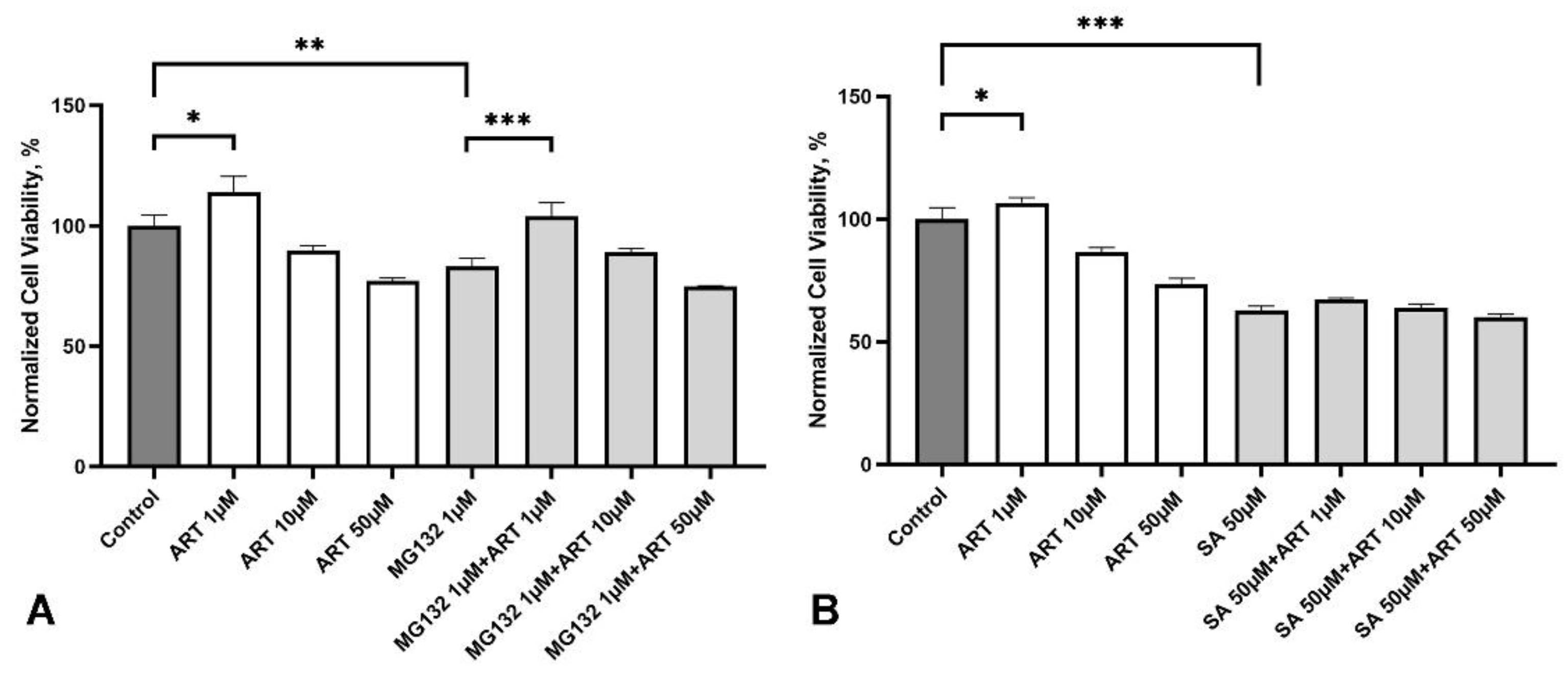
2.5. Artemisinin Protects SH-SY5Y Cells from Stress Induced by the Proteasome Inhibitor MG132
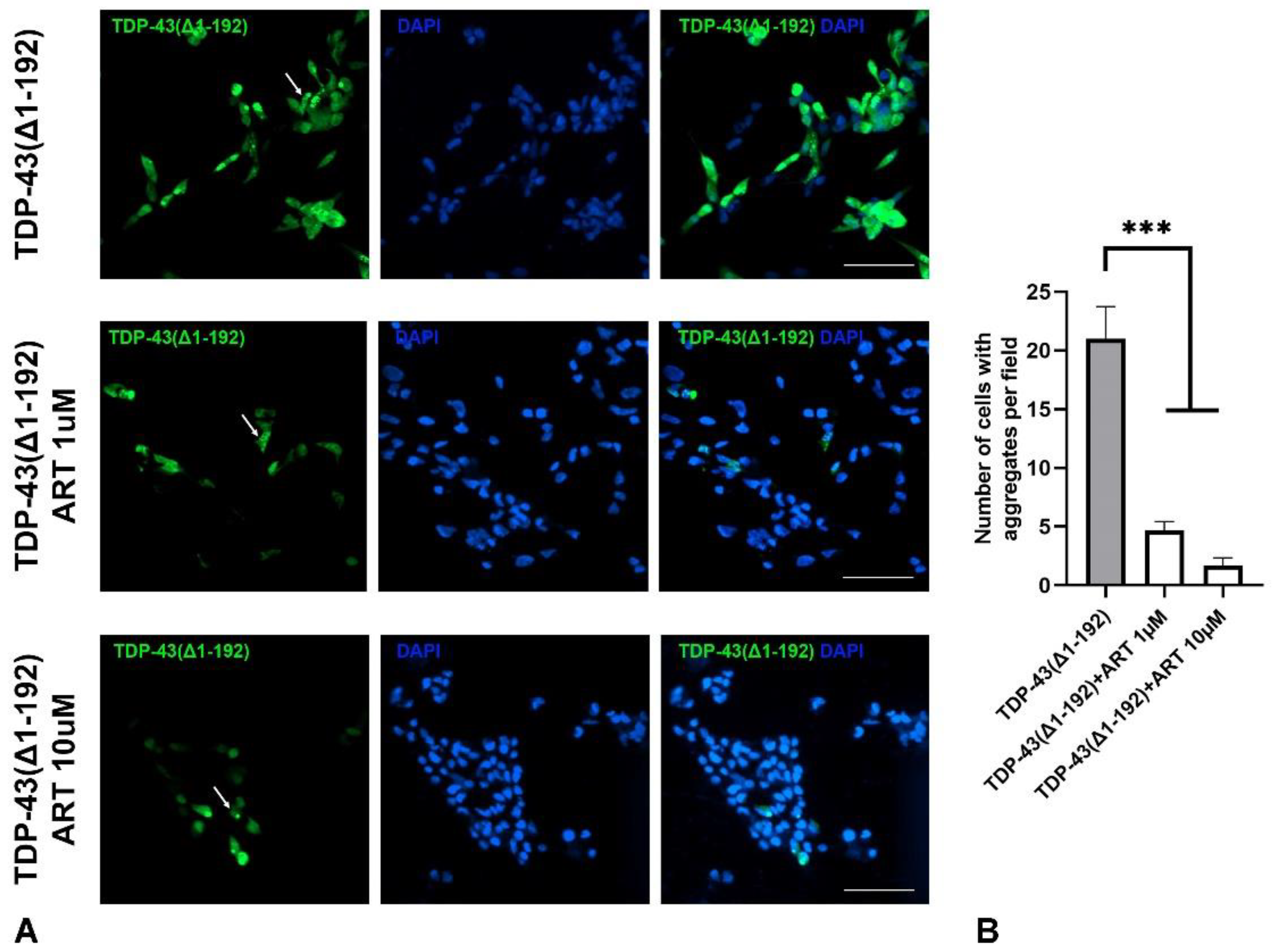
2.6. Artemisinin Inhibits Aggregation of Mutated TDP-43 in SH-SY5Y Cells
3. Discussion
4. Materials and Methods
4.1. Compounds
4.2. Stable Cell Lines, Cell Viability Assay
4.3. Cell Cycle Analysis
4.4. ATP Level Assessment
4.5. Primary Hippocampal Cultures
4.6. Immunocytochemical Staining
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tu, Y. The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nature medicine 2011, 17, 1217–1220. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Xu, J.; Zheng, W. Artemisinin protects PC12 cells against beta-amyloid-induced apoptosis through activation of the ERK1/2 signaling pathway. Redox biology 2017, 12, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Fang, J.; Li, S.; Gaur, U.; Xing, X.; Wang, H.; Zheng, W. Artemisinin Attenuated Hydrogen Peroxide (H(2)O(2))-Induced Oxidative Injury in SH-SY5Y and Hippocampal Neurons via the Activation of AMPK Pathway. International journal of molecular sciences 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Li, W.; Lv, R.; Zhang, M.; Liu, W. Neuroprotective effects and mechanisms of action of artemisinin in retinal ganglion cells in a mouse model of traumatic optic neuropathy. Heliyon 2024, 10, e31378. [Google Scholar] [CrossRef]
- Niu, X.Y.; Ho, L.Y.; Ren, Z.H.; Song, Z.Y. Metabolic fate of Qinghaosu in rats; a new TLC densitometric method for its determination in biological material. European journal of drug metabolism and pharmacokinetics 1985, 10, 55–59. [Google Scholar] [CrossRef]
- Davis, T.M.; Binh, T.Q.; Ilett, K.F.; Batty, K.T.; Phuong, H.L.; Chiswell, G.M.; Phuong, V.D.; Agus, C. Penetration of dihydroartemisinin into cerebrospinal fluid after administration of intravenous artesunate in severe falciparum malaria. Antimicrobial agents and chemotherapy 2003, 47, 368–370. [Google Scholar] [CrossRef]
- Zheng, W.; Chong, C.M.; Wang, H.; Zhou, X.; Zhang, L.; Wang, R.; Meng, Q.; Lazarovici, P.; Fang, J. Artemisinin conferred ERK mediated neuroprotection to PC12 cells and cortical neurons exposed to sodium nitroprusside-induced oxidative insult. Free radical biology & medicine 2016, 97, 158–167. [Google Scholar] [CrossRef]
- Li, Q.; Li, S.; Fang, J.; Yang, C.; Zhao, X.; Wang, Q.; Zhou, W.; Zheng, W. Artemisinin Confers Neuroprotection against 6-OHDA-Induced Neuronal Injury In Vitro and In Vivo through Activation of the ERK1/2 Pathway. Molecules 2023, 28. [Google Scholar] [CrossRef]
- Lin, S.P.; Li, W.; Winters, A.; Liu, R.; Yang, S.H. Artemisinin Prevents Glutamate-Induced Neuronal Cell Death Via Akt Pathway Activation. Frontiers in cellular neuroscience 2018, 12, 108. [Google Scholar] [CrossRef]
- Yan, J.; Ma, H.; Lai, X.; Wu, J.; Liu, A.; Huang, J.; Sun, W.; Shen, M.; Zhang, Y. Artemisinin attenuated oxidative stress and apoptosis by inhibiting autophagy in MPP(+)-treated SH-SY5Y cells. Journal of biological research 2021, 28, 6. [Google Scholar] [CrossRef]
- Lim, H.S.; Park, G. Artemisinin protects dopaminergic neurons against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced neurotoxicity in a mouse model of Parkinson's disease. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 2024, 170, 115972. [Google Scholar] [CrossRef]
- Shi, J.Q.; Zhang, C.C.; Sun, X.L.; Cheng, X.X.; Wang, J.B.; Zhang, Y.D.; Xu, J.; Zou, H.Q. Antimalarial drug artemisinin extenuates amyloidogenesis and neuroinflammation in APPswe/PS1dE9 transgenic mice via inhibition of nuclear factor-kappaB and NLRP3 inflammasome activation. CNS neuroscience & therapeutics 2013, 19, 262–268. [Google Scholar] [CrossRef]
- Zhao, X.; Li, S.; Gaur, U.; Zheng, W. Artemisinin Improved Neuronal Functions in Alzheimer's Disease Animal Model 3xtg Mice and Neuronal Cells via Stimulating the ERK/CREB Signaling Pathway. Aging and disease 2020, 11, 801–819. [Google Scholar] [CrossRef] [PubMed]
- Kiss, E.; Kins, S.; Gorgas, K.; Venczel Szakacs, K.H.; Kirsch, J.; Kuhse, J. Another Use for a Proven Drug: Experimental Evidence for the Potential of Artemisinin and Its Derivatives to Treat Alzheimer's Disease. International journal of molecular sciences 2024, 25. [Google Scholar] [CrossRef]
- Hetz, C.; Saxena, S. ER stress and the unfolded protein response in neurodegeneration. Nature reviews. Neurology 2017, 13, 477–491. [Google Scholar] [CrossRef]
- Lindholm, D.; Wootz, H.; Korhonen, L. ER stress and neurodegenerative diseases. Cell death and differentiation 2006, 13, 385–392. [Google Scholar] [CrossRef]
- Chen, G.; Wei, T.; Ju, F.; Li, H. Protein quality control and aggregation in the endoplasmic reticulum: From basic to bedside. Frontiers in cell and developmental biology 2023, 11, 1156152. [Google Scholar] [CrossRef]
- Brossi, A.; Venugopalan, B.; Dominguez Gerpe, L.; Yeh, H.J.; Flippen-Anderson, J.L.; Buchs, P.; Luo, X.D.; Milhous, W.; Peters, W. Arteether, a new antimalarial drug: synthesis and antimalarial properties. Journal of medicinal chemistry 1988, 31, 645–650. [Google Scholar] [CrossRef]
- Zuma, N.H.; Smit, F.J.; de Kock, C.; Combrinck, J.; Smith, P.J.; N'Da, D.D. Synthesis and biological evaluation of a series of non-hemiacetal ester derivatives of artemisinin. European journal of medicinal chemistry 2016, 122, 635–646. [Google Scholar] [CrossRef]
- Semakov, A.V.; Anikina, L.V.; Klochkov, S.G. Synthesis and Cytotoxic Activity of the Products of Addition of Thiophenol to Sesquiterpene Lactones. Russian Journal of Bioorganic Chemistry 2021, 47, 906–917. [Google Scholar] [CrossRef]
- El-Feraly, F.S.; Ayalp, A.; Al-Yahya, M.A.; McPhail, D.R.; McPhail, A.T. Conversion of Artemisinin to Artemisitene. Journal of Natural Products 1990, 53, 66–71. [Google Scholar] [CrossRef]
- Cervera, L.; Gutierrez, S.; Godia, F.; Segura, M.M. Optimization of HEK 293 cell growth by addition of non-animal derived components using design of experiments. BMC proceedings 2011, 5 Suppl 8, P126. [Google Scholar] [CrossRef]
- Feles, S.; Overath, C.; Reichardt, S.; Diegeler, S.; Schmitz, C.; Kronenberg, J.; Baumstark-Khan, C.; Hemmersbach, R.; Hellweg, C.E.; Liemersdorf, C. Streamlining Culture Conditions for the Neuroblastoma Cell Line SH-SY5Y: A Prerequisite for Functional Studies. Methods and protocols 2022, 5. [Google Scholar] [CrossRef] [PubMed]
- Brewer, G.J.; Torricelli, J.R.; Evege, E.K.; Price, P.J. Optimized survival of hippocampal neurons in B27-supplemented Neurobasal, a new serum-free medium combination. Journal of neuroscience research 1993, 35, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.T.; Kuo, P.H.; Chiang, C.H.; Liang, J.R.; Chen, Y.R.; Wang, S.; Shen, J.C.; Yuan, H.S. The truncated C-terminal RNA recognition motif of TDP-43 protein plays a key role in forming proteinaceous aggregates. The Journal of biological chemistry 2013, 288, 9049–9057. [Google Scholar] [CrossRef]
- He, B.; Soderlund, D.M. Human embryonic kidney (HEK293) cells express endogenous voltage-gated sodium currents and Na v 1.7 sodium channels. Neuroscience letters 2010, 469, 268–272. [Google Scholar] [CrossRef]
- Shaw, G.; Morse, S.; Ararat, M.; Graham, F.L. Preferential transformation of human neuronal cells by human adenoviruses and the origin of HEK 293 cells. FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2002, 16, 869–871. [Google Scholar] [CrossRef]
- Madhusudana, S.N.; Sundaramoorthy, S.; Ullas, P.T. Utility of human embryonic kidney cell line HEK-293 for rapid isolation of fixed and street rabies viruses: comparison with Neuro-2a and BHK-21 cell lines. International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases 2010, 14, e1067–1071. [Google Scholar] [CrossRef]
- Ahmed-Laloui, H.; Zaak, H.; Rahmani, A.; Kashi, I.; Chemat, S.; Miara, M.D.; Cherb, N.; Derdour, M. Assessment of artemisinin and antioxidant activities of three wild Artemisia species of Algeria. Natural product research 2022, 36, 6344–6352. [Google Scholar] [CrossRef]
- Chebbac, K.; Benziane Ouaritini, Z.; El Moussaoui, A.; Chalkha, M.; Lafraxo, S.; Bin Jardan, Y.A.; Nafidi, H.A.; Bourhia, M.; Guemmouh, R. Antimicrobial and Antioxidant Properties of Chemically Analyzed Essential Oil of Artemisia annua L. (Asteraceae) Native to Mediterranean Area. Life 2023, 13. [Google Scholar] [CrossRef]
- Ferreira, J.F.S.; Zheljazkov, V.D.; Gonzalez, J.M. Artemisinin concentration and antioxidant capacity of Artemisia annua distillation byproduct. Industrial Crops and Products 2013, 41, 294–298. [Google Scholar] [CrossRef]
- Rodriguez-Gabriel, M.A.; Russell, P. Distinct signaling pathways respond to arsenite and reactive oxygen species in Schizosaccharomyces pombe. Eukaryotic cell 2005, 4, 1396–1402. [Google Scholar] [CrossRef] [PubMed]
- Nontprasert, A.; Pukrittayakamee, S.; Nosten-Bertrand, M.; Vanijanonta, S.; White, N.J. Studies of the neurotoxicity of oral artemisinin derivatives in mice. The American journal of tropical medicine and hygiene 2000, 62, 409–412. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Shan, L.; Tong, P.; Efferth, T. Cardiotoxicity and Cardioprotection by Artesunate in Larval Zebrafish. Dose-response : a publication of International Hormesis Society 2020, 18, 1559325819897180. [Google Scholar] [CrossRef] [PubMed]
- Bridgford, J.L.; Xie, S.C.; Cobbold, S.A.; Pasaje, C.F.A.; Herrmann, S.; Yang, T.; Gillett, D.L.; Dick, L.R.; Ralph, S.A.; Dogovski, C.; et al. Artemisinin kills malaria parasites by damaging proteins and inhibiting the proteasome. Nature communications 2018, 9, 3801. [Google Scholar] [CrossRef]
- Dogovski, C.; Xie, S.C.; Burgio, G.; Bridgford, J.; Mok, S.; McCaw, J.M.; Chotivanich, K.; Kenny, S.; Gnadig, N.; Straimer, J.; et al. Targeting the cell stress response of Plasmodium falciparum to overcome artemisinin resistance. PLoS biology 2015, 13, e1002132. [Google Scholar] [CrossRef]
- Mok, S.; Ashley, E.A.; Ferreira, P.E.; Zhu, L.; Lin, Z.; Yeo, T.; Chotivanich, K.; Imwong, M.; Pukrittayakamee, S.; Dhorda, M.; et al. Drug resistance. Population transcriptomics of human malaria parasites reveals the mechanism of artemisinin resistance. Science 2015, 347, 431–435. [Google Scholar] [CrossRef]
- Kannan, D.; Joshi, N.; Gupta, S.; Pati, S.; Bhattacharjee, S.; Langsley, G.; Singh, S. Cytoprotective autophagy as a pro-survival strategy in ART-resistant malaria parasites. Cell death discovery 2023, 9, 160. [Google Scholar] [CrossRef]
- Kiss, E.; Kins, S.; Zoller, Y.; Schilling, S.; Gorgas, K.; Gross, D.; Schlicksupp, A.; Rosner, R.; Kirsch, J.; Kuhse, J. Artesunate restores the levels of inhibitory synapse proteins and reduces amyloid-beta and C-terminal fragments (CTFs) of the amyloid precursor protein in an AD-mouse model. Molecular and cellular neurosciences 2021, 113, 103624. [Google Scholar] [CrossRef]
- Kisler, K.; Sagare, A.P.; Lazic, D.; Bazzi, S.; Lawson, E.; Hsu, C.J.; Wang, Y.; Ramanathan, A.; Nelson, A.R.; Zhao, Z.; et al. Anti-malaria drug artesunate prevents development of amyloid-beta pathology in mice by upregulating PICALM at the blood-brain barrier. Molecular neurodegeneration 2023, 18, 7. [Google Scholar] [CrossRef]
- Xiao, M.; Xiang, W.; Chen, Y.; Peng, N.; Du, X.; Lu, S.; Zuo, Y.; Li, B.; Hu, Y.; Li, X. DHA Ameliorates Cognitive Ability, Reduces Amyloid Deposition, and Nerve Fiber Production in Alzheimer's Disease. Frontiers in nutrition 2022, 9, 852433. [Google Scholar] [CrossRef] [PubMed]
- de Boer, E.M.J.; Orie, V.K.; Williams, T.; Baker, M.R.; De Oliveira, H.M.; Polvikoski, T.; Silsby, M.; Menon, P.; van den Bos, M.; Halliday, G.M.; et al. TDP-43 proteinopathies: a new wave of neurodegenerative diseases. Journal of neurology, neurosurgery, and psychiatry 2020, 92, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Jo, M.; Lee, S.; Jeon, Y.M.; Kim, S.; Kwon, Y.; Kim, H.J. The role of TDP-43 propagation in neurodegenerative diseases: integrating insights from clinical and experimental studies. Experimental & molecular medicine 2020, 52, 1652–1662. [Google Scholar] [CrossRef]
- Kukharsky, M.S.; Quintiero, A.; Matsumoto, T.; Matsukawa, K.; An, H.; Hashimoto, T.; Iwatsubo, T.; Buchman, V.L.; Shelkovnikova, T.A. Calcium-responsive transactivator (CREST) protein shares a set of structural and functional traits with other proteins associated with amyotrophic lateral sclerosis. Molecular neurodegeneration 2015, 10, 20. [Google Scholar] [CrossRef]
- Kukharsky, M.S.; Ninkina, N.N.; An, H.; Telezhkin, V.; Wei, W.; Meritens, C.R.; Cooper-Knock, J.; Nakagawa, S.; Hirose, T.; Buchman, V.L.; et al. Long non-coding RNA Neat1 regulates adaptive behavioural response to stress in mice. Translational psychiatry 2020, 10, 171. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



