Submitted:
26 November 2024
Posted:
27 November 2024
You are already at the latest version
Abstract
Keywords:
Introduction
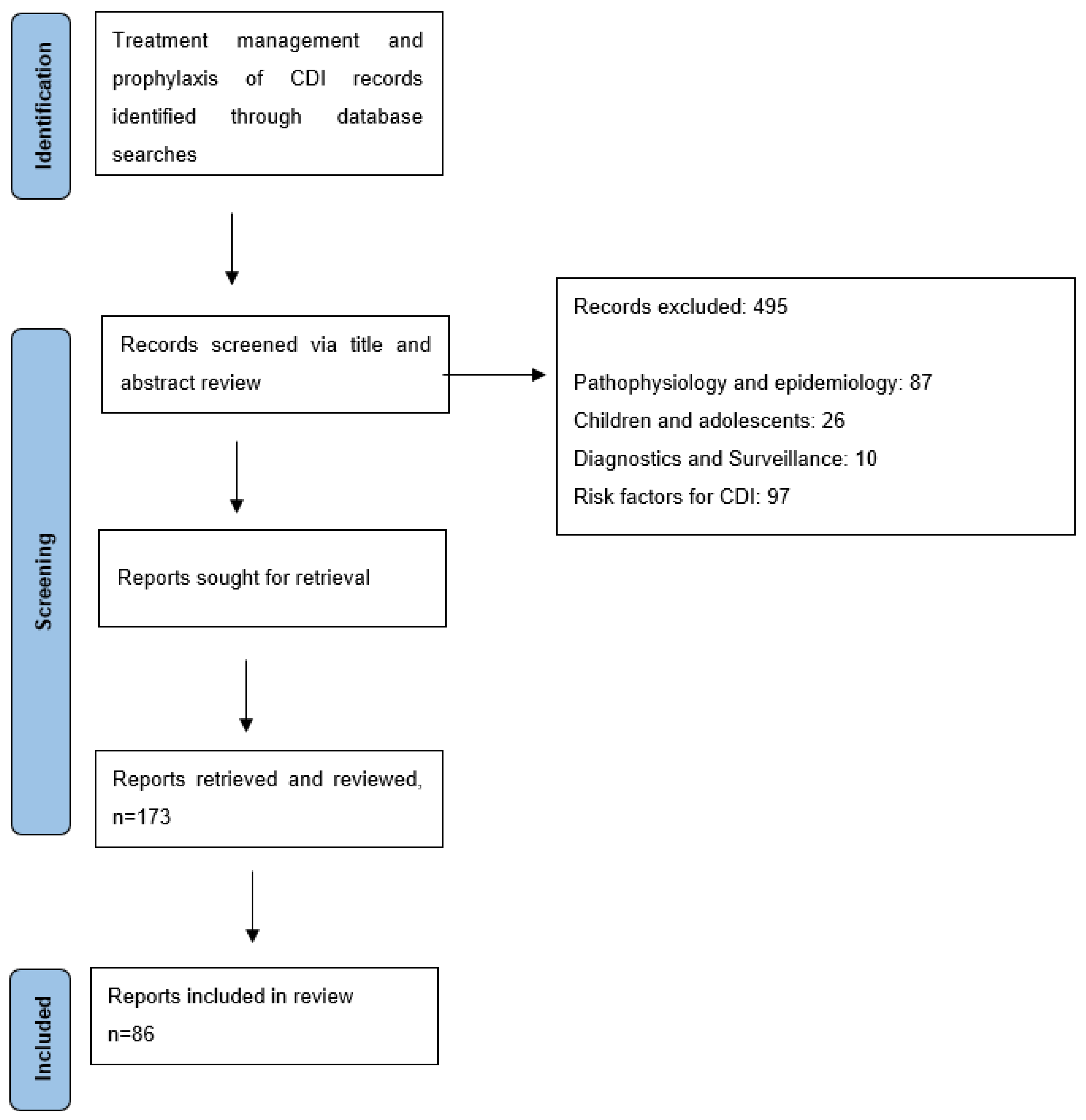
Materials & Methods
Results
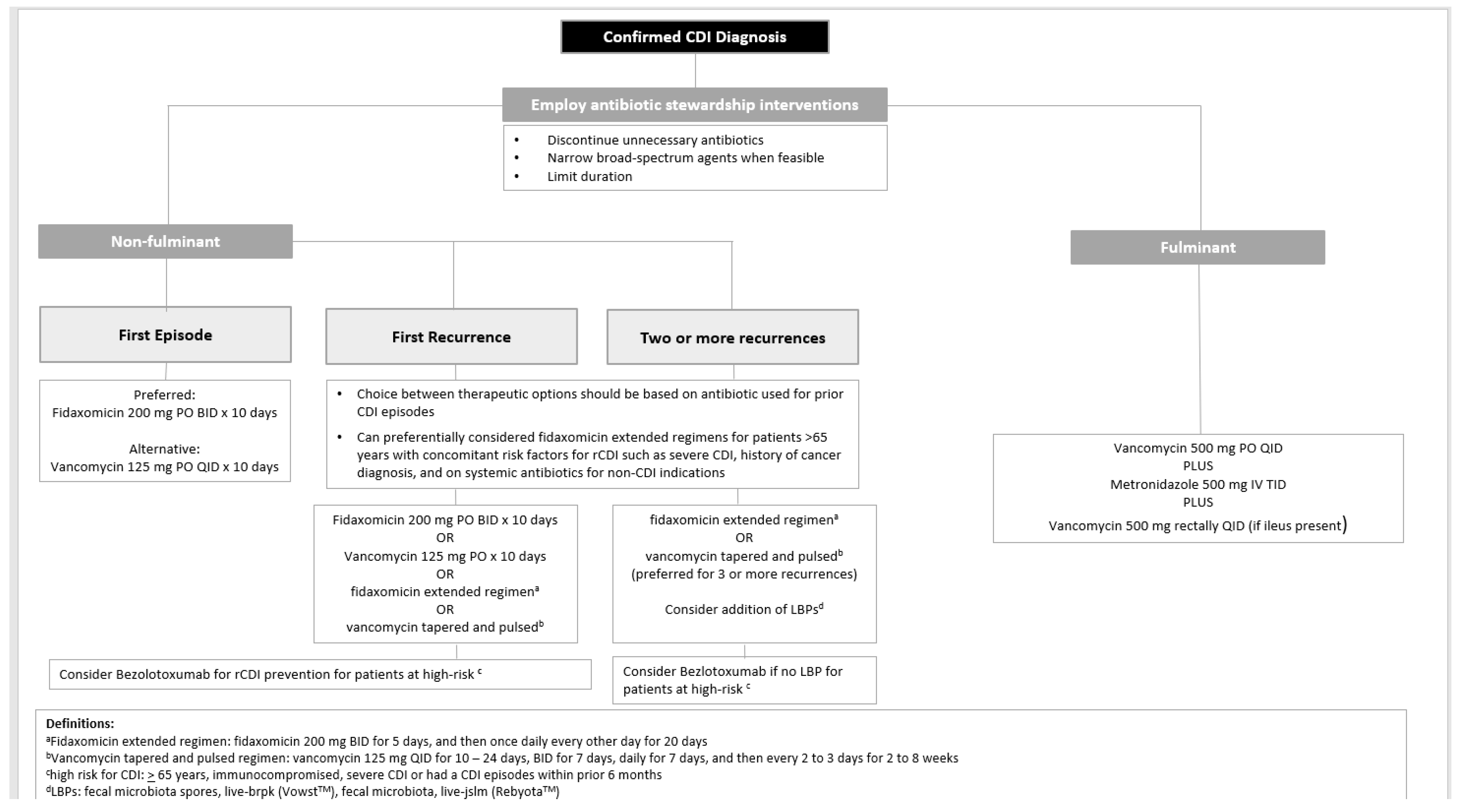
First Line Antimicrobials for the Treatment of CDI in Adults
Metronidazole
Vancomycin and Fidaxomicin
Alternative Antimicrobial Therapies
Rifaximin
Tigecycline
Adjunctive Treatment
Bezlotoxumab
Microbiota Restoration Therapies for the Prevention of CDI
Fecal Microbiota Transplantation (FMT)
Novel Live Biotherapeutic Products
Fecal Microbiota, Live-jslm (RebyotaTM)
Fecal Microbiota Spores, Live-brpk (VowstTM)
VE303
| Fecal microbiota, live-jslm (RebyotaTM) | Fecal microbiota spores, live-brpk (VowstTM) | |
|---|---|---|
| Route | Rectal enema | Oral capsule |
| Dose | Single dose of 150 mL rectally | 4 capsules orally once daily for 3 days |
| Antibiotic wash out period | Administered 24 to 72 hours after CDI antibiotic therapy discontinuation | First dose administered 24 to 72 hours after CDI antibiotic therapy discontinuation |
| Administration | Thaw by placing product in refrigerator (2-8°C) for 24 hours. Patient should empty their bladder and bowel, if possible, prior to administration Keep the patient in the left-side positive or knee-chest position for up to 15 minutes after administration to minimize cramping and expulsion |
Prior to the first dose, patient should drink 296 mL (10 oz) of magnesium citrate (or polyethylene glycol for patients with renal dysfunction) on the day before and at least 8 h prior to taking the first dose Should be administered before the first meal on an empty stomach |
| Storage | Ultracold freezer (-60° to -90°C) -OR- Refrigerator (2° to 8°C) for up to 5 d (including thaw time) **do not freeze after thawing** |
Original packaging at 2° to 25° C **do not freeze** |
Other Preventative Strategies
Antimicrobial Stewardship
Probiotics
C. difficile Vaccine
Antibiotic Prophylaxis
Conclusion and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Snyder, m. Further Studies on Bacillus Dificilis (Jall and O'Toole). The Journal of Infectious Diseases 1937, 60, 223-231.
- Lessa, F.C.; Mu, Y.; Bamberg, W.M.; Beldavs, Z.G.; Dumyati, G.K.; Dunn, J.R.; Farley, M.M.; Holzbauer, S.M.; Meek, J.I.; Phipps, E.C., et al. Burden of Clostridium difficile infection in the United States. N Engl J Med 2015, 372, 825-834. [CrossRef]
- Prevention, C.f.D.C.a. Antibitoic Resistance Threats in the United States 2019. US Department of Health and Human Services, Centers for Diseae Control and Prevention 2019.
- Deshpande, A.; Pasupuleti, V.; Thota, P.; Pant, C.; Rolston, D.D.; Hernandez, A.V.; Donskey, C.J.; Fraser, T.G. Risk factors for recurrent Clostridium difficile infection: a systematic review and meta-analysis. Infect Control Hosp Epidemiol 2015, 36, 452-460. [CrossRef]
- Czepiel, J.; Drozdz, M.; Pituch, H.; Kuijper, E.J.; Perucki, W.; Mielimonka, A.; Goldman, S.; Wultanska, D.; Garlicki, A.; Biesiada, G. Clostridium difficile infection: review. Eur J Clin Microbiol Infect Dis 2019, 38, 1211-1221. [CrossRef]
- Guery, B.; Menichetti, F.; Anttila, V.J.; Adomakoh, N.; Aguado, J.M.; Bisnauthsing, K.; Georgopali, A.; Goldenberg, S.D.; Karas, A.; Kazeem, G., et al. Extended-pulsed fidaxomicin versus vancomycin for Clostridium difficile infection in patients 60 years and older (EXTEND): a randomised, controlled, open-label, phase 3b/4 trial. Lancet Infect Dis 2018, 18, 296-307. [CrossRef]
- Juo, Y.Y.; Sanaiha, Y.; Jabaji, Z.; Benharash, P. Trends in Diverting Loop Ileostomy vs Total Abdominal Colectomy as Surgical Management for Clostridium difficile Colitis. JAMA Surg 2019, 154, 899-906. [CrossRef]
- Kwon, J.H.; Olsen, M.A.; Dubberke, E.R. The morbidity, mortality, and costs associated with Clostridium difficile infection. Infect Dis Clin North Am 2015, 29, 123-134. [CrossRef]
- Appaneal, H.J.; Caffrey, A.R.; Beganovic, M.; Avramovic, S.; LaPlante, K.L. Predictors of Mortality Among a National Cohort of Veterans With Recurrent Clostridium difficile Infection. Open Forum Infect Dis 2018, 5, ofy175. [CrossRef]
- Zimlichman, E.; Henderson, D.; Tamir, O.; Franz, C.; Song, P.; Yamin, C.K.; Keohane, C.; Denham, C.R.; Bates, D.W. Health care-associated infections: a meta-analysis of costs and financial impact on the US health care system. JAMA Intern Med 2013, 173, 2039-2046. [CrossRef]
- Teasley, D.G.; Gerding, D.N.; Olson, M.M.; Peterson, L.R.; Gebhard, R.L.; Schwartz, M.J.; Lee, J.T., Jr. Prospective randomised trial of metronidazole versus vancomycin for Clostridium-difficile-associated diarrhoea and colitis. Lancet 1983, 2, 1043-1046. [CrossRef]
- Wenisch, C.; Parschalk, B.; Hasenhundl, M.; Hirschl, A.M.; Graninger, W. Comparison of vancomycin, teicoplanin, metronidazole, and fusidic acid for the treatment of Clostridium difficile-associated diarrhea. Clin Infect Dis 1996, 22, 813-818. [CrossRef]
- Belmares, J.; Gerding, D.N.; Parada, J.P.; Miskevics, S.; Weaver, F.; Johnson, S. Outcome of metronidazole therapy for Clostridium difficile disease and correlation with a scoring system. J Infect 2007, 55, 495-501. [CrossRef]
- Brazier, J.S.; Raybould, R.; Patel, B.; Duckworth, G.; Pearson, A.; Charlett, A.; Duerden, B.I.; Network, H.P.A.R.M. Distribution and antimicrobial susceptibility patterns of Clostridium difficile PCR ribotypes in English hospitals, 2007-08. Euro Surveill 2008, 13. [CrossRef]
- Peng, Z.; Jin, D.; Kim, H.B.; Stratton, C.W.; Wu, B.; Tang, Y.W.; Sun, X. Update on Antimicrobial Resistance in Clostridium difficile: Resistance Mechanisms and Antimicrobial Susceptibility Testing. J Clin Microbiol 2017, 55, 1998-2008. [CrossRef]
- Zar, F.A.; Bakkanagari, S.R.; Moorthi, K.M.; Davis, M.B. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis 2007, 45, 302-307. [CrossRef]
- Cohen, S.H.; Gerding, D.N.; Johnson, S.; Kelly, C.P.; Loo, V.G.; McDonald, L.C.; Pepin, J.; Wilcox, M.H.; Society for Healthcare Epidemiology of, A.; Infectious Diseases Society of, A. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA). Infect Control Hosp Epidemiol 2010, 31, 431-455. [CrossRef]
- Johnson, S.; Louie, T.J.; Gerding, D.N.; Cornely, O.A.; Chasan-Taber, S.; Fitts, D.; Gelone, S.P.; Broom, C.; Davidson, D.M.; Polymer Alternative for, C.D.I.T.i. Vancomycin, metronidazole, or tolevamer for Clostridium difficile infection: results from two multinational, randomized, controlled trials. Clin Infect Dis 2014, 59, 345-354. [CrossRef]
- Stevens, V.W.; Nelson, R.E.; Schwab-Daugherty, E.M.; Khader, K.; Jones, M.M.; Brown, K.A.; Greene, T.; Croft, L.D.; Neuhauser, M.; Glassman, P., et al. Comparative Effectiveness of Vancomycin and Metronidazole for the Prevention of Recurrence and Death in Patients With Clostridium difficile Infection. JAMA Intern Med 2017, 177, 546-553. [CrossRef]
- Thorpe, C.M.; Kane, A.V.; Chang, J.; Tai, A.; Vickers, R.J.; Snydman, D.R. Enhanced preservation of the human intestinal microbiota by ridinilazole, a novel Clostridium difficile-targeting antibacterial, compared to vancomycin. PLoS One 2018, 13, e0199810. [CrossRef]
- Gonzales, M.; Pepin, J.; Frost, E.H.; Carrier, J.C.; Sirard, S.; Fortier, L.C.; Valiquette, L. Faecal pharmacokinetics of orally administered vancomycin in patients with suspected Clostridium difficile infection. BMC Infect Dis 2010, 10, 363. [CrossRef]
- Thabit, A.K.; Nicolau, D.P. Impact of vancomycin faecal concentrations on clinical and microbiological outcomes in Clostridium difficile infection. Int J Antimicrob Agents 2015, 46, 205-208. [CrossRef]
- Thorpe, C.M.; McDermott, L.A.; Tran, M.K.; Chang, J.; Jenkins, S.G.; Goldstein, E.J.C.; Patel, R.; Forbes, B.A.; Johnson, S.; Gerding, D.N., et al. U.S.-Based National Surveillance for Fidaxomicin Susceptibility of Clostridioides difficile-Associated Diarrheal Isolates from 2013 to 2016. Antimicrob Agents Chemother 2019, 63. [CrossRef]
- Fernandez, A.; Anand, G.; Friedenberg, F. Factors associated with failure of metronidazole in Clostridium difficile-associated disease. J Clin Gastroenterol 2004, 38, 414-418. [CrossRef]
- McDonald, L.C.; Gerding, D.N.; Johnson, S.; Bakken, J.S.; Carroll, K.C.; Coffin, S.E.; Dubberke, E.R.; Garey, K.W.; Gould, C.V.; Kelly, C., et al. Clinical Practice Guidelines for Clostridium difficile Infection in Adults and Children: 2017 Update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis 2018, 66, e1-e48. [CrossRef]
- Louie, T.J.; Miller, M.A.; Mullane, K.M.; Weiss, K.; Lentnek, A.; Golan, Y.; Gorbach, S.; Sears, P.; Shue, Y.K.; Group, O.P.T.C.S. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med 2011, 364, 422-431. [CrossRef]
- Cornely, O.A.; Crook, D.W.; Esposito, R.; Poirier, A.; Somero, M.S.; Weiss, K.; Sears, P.; Gorbach, S.; Group, O.P.T.C.S. Fidaxomicin versus vancomycin for infection with Clostridium difficile in Europe, Canada, and the USA: a double-blind, non-inferiority, randomised controlled trial. Lancet Infect Dis 2012, 12, 281-289. [CrossRef]
- Okumura, H.; Fukushima, A.; Taieb, V.; Shoji, S.; English, M. Fidaxomicin compared with vancomycin and metronidazole for the treatment of Clostridioides (Clostridium) difficile infection: A network meta-analysis. J Infect Chemother 2020, 26, 43-50. [CrossRef]
- Mikamo, H.; Tateda, K.; Yanagihara, K.; Kusachi, S.; Takesue, Y.; Miki, T.; Oizumi, Y.; Gamo, K.; Hashimoto, A.; Toyoshima, J., et al. Efficacy and safety of fidaxomicin for the treatment of Clostridioides (Clostridium) difficile infection in a randomized, double-blind, comparative Phase III study in Japan. J Infect Chemother 2018, 24, 744-752. [CrossRef]
- Venugopal, A.A.; Johnson, S. Fidaxomicin: a novel macrocyclic antibiotic approved for treatment of Clostridium difficile infection. Clin Infect Dis 2012, 54, 568-574. [CrossRef]
- Kelly, C.R.; Fischer, M.; Allegretti, J.R.; LaPlante, K.; Stewart, D.B.; Limketkai, B.N.; Stollman, N.H. ACG Clinical Guidelines: Prevention, Diagnosis, and Treatment of Clostridioides difficile Infections. Am J Gastroenterol 2021, 116, 1124-1147. [CrossRef]
- Cornely, O.A.; Miller, M.A.; Louie, T.J.; Crook, D.W.; Gorbach, S.L. Treatment of first recurrence of Clostridium difficile infection: fidaxomicin versus vancomycin. Clin Infect Dis 2012, 55 Suppl 2, S154-161. [CrossRef]
- Hota, S.S.; Sales, V.; Tomlinson, G.; Salpeter, M.J.; McGeer, A.; Coburn, B.; Guttman, D.S.; Low, D.E.; Poutanen, S.M. Oral Vancomycin Followed by Fecal Transplantation Versus Tapering Oral Vancomycin Treatment for Recurrent Clostridium difficile Infection: An Open-Label, Randomized Controlled Trial. Clin Infect Dis 2017, 64, 265-271. [CrossRef]
- Khanna, S. My Treatment Approach to Clostridioides difficile Infection. Mayo Clinic Proceedings 2021, 10.1016. [CrossRef]
- Fekety, R.; Silva, J.; Kauffman, C.; Buggy, B.; Deery, H.G. Treatment of antibiotic-associated Clostridium difficile colitis with oral vancomycin: comparison of two dosage regimens. Am J Med 1989, 86, 15-19. [CrossRef]
- Lam, S.W.; Bass, S.N.; Neuner, E.A.; Bauer, S.R. Effect of vancomycin dose on treatment outcomes in severe Clostridium difficile infection. Int J Antimicrob Agents 2013, 42, 553-558. [CrossRef]
- Ng, Q.X.; Loke, W.; Foo, N.X.; Mo, Y.; Yeo, W.S.; Soh, A.Y.S. A systematic review of the use of rifaximin for Clostridium difficile infections. Anaerobe 2019, 55, 35-39. [CrossRef]
- Garey, K.W.; Ghantoji, S.S.; Shah, D.N.; Habib, M.; Arora, V.; Jiang, Z.D.; DuPont, H.L. A randomized, double-blind, placebo-controlled pilot study to assess the ability of rifaximin to prevent recurrent diarrhoea in patients with Clostridium difficile infection. J Antimicrob Chemother 2011, 66, 2850-2855. [CrossRef]
- Finegold, S.M.; Molitoris, D.; Vaisanen, M.L. Study of the in vitro activities of rifaximin and comparator agents against 536 anaerobic intestinal bacteria from the perspective of potential utility in pathology involving bowel flora. Antimicrob Agents Chemother 2009, 53, 281-286. [CrossRef]
- Kechagias, K.S.; Chorepsima, S.; Triarides, N.A.; Falagas, M.E. Tigecycline for the treatment of patients with Clostridium difficile infection: an update of the clinical evidence. Eur J Clin Microbiol Infect Dis 2020, 39, 1053-1058. [CrossRef]
- Larson, K.C.; Belliveau, P.P.; Spooner, L.M. Tigecycline for the treatment of severe Clostridium difficile infection. Ann Pharmacother 2011, 45, 1005-1010. [CrossRef]
- Manea, E.; Sojo-Dorado, J.; Jipa, R.E.; Benea, S.N.; Rodriguez-Bano, J.; Hristea, A. The role of tigecycline in the management of Clostridium difficile infection: a retrospective cohort study. Clin Microbiol Infect 2018, 24, 180-184. [CrossRef]
- Herpers, B.L.; Vlaminckx, B.; Burkhardt, O.; Blom, H.; Biemond-Moeniralam, H.S.; Hornef, M.; Welte, T.; Kuijper, E.J. Intravenous tigecycline as adjunctive or alternative therapy for severe refractory Clostridium difficile infection. Clin Infect Dis 2009, 48, 1732-1735. [CrossRef]
- Gergely Szabo, B.; Kadar, B.; Szidonia Lenart, K.; Dezsenyi, B.; Kunovszki, P.; Fried, K.; Kamotsay, K.; Nikolova, R.; Prinz, G. Use of intravenous tigecycline in patients with severe Clostridium difficile infection: a retrospective observational cohort study. Clin Microbiol Infect 2016, 22, 990-995. [CrossRef]
- Bezlotoxumab [package insert]. Merck: 2016.
- Wilcox, M.H.; Gerding, D.N.; Poxton, I.R.; Kelly, C.; Nathan, R.; Birch, T.; Cornely, O.A.; Rahav, G.; Bouza, E.; Lee, C., et al. Bezlotoxumab for Prevention of Recurrent Clostridium difficile Infection. N Engl J Med 2017, 376, 305-317. [CrossRef]
- Gerding, D.N.; Kelly, C.P.; Rahav, G.; Lee, C.; Dubberke, E.R.; Kumar, P.N.; Yacyshyn, B.; Kao, D.; Eves, K.; Ellison, M.C., et al. Bezlotoxumab for Prevention of Recurrent Clostridium difficile Infection in Patients at Increased Risk for Recurrence. Clin Infect Dis 2018, 67, 649-656. [CrossRef]
- Birch, T.; Golan, Y.; Rizzardini, G.; Jensen, E.; Gabryelski, L.; Guris, D.; Dorr, M.B. Efficacy of bezlotoxumab based on timing of administration relative to start of antibacterial therapy for Clostridium difficile infection. J Antimicrob Chemother 2018, 73, 2524-2528. [CrossRef]
- Wang, J.W.; Kuo, C.H.; Kuo, F.C.; Wang, Y.K.; Hsu, W.H.; Yu, F.J.; Hu, H.M.; Hsu, P.I.; Wang, J.Y.; Wu, D.C. Fecal microbiota transplantation: Review and update. J Formos Med Assoc 2019, 118 Suppl 1, S23-S31. [CrossRef]
- van Nood, E.; Vrieze, A.; Nieuwdorp, M.; Fuentes, S.; Zoetendal, E.G.; de Vos, W.M.; Visser, C.E.; Kuijper, E.J.; Bartelsman, J.F.; Tijssen, J.G., et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med 2013, 368, 407-415. [CrossRef]
- Minkoff, N.Z.; Aslam, S.; Medina, M.; Tanner-Smith, E.E.; Zackular, J.P.; Acra, S.; Nicholson, M.R.; Imdad, A. Fecal microbiota transplantation for the treatment of recurrent Clostridioides difficile (Clostridium difficile). Cochrane Database Syst Rev 2023, 4, CD013871. [CrossRef]
- Porcari, S.; Benech, N.; Valles-Colomer, M.; Segata, N.; Gasbarrini, A.; Cammarota, G.; Sokol, H.; Ianiro, G. Key determinants of success in fecal microbiota transplantation: From microbiome to clinic. Cell Host Microbe 2023, 31, 712-733. [CrossRef]
- Yadegar, A.; Pakpoor, S.; Ibrahim, F.F.; Nabavi-Rad, A.; Cook, L.; Walter, J.; Seekatz, A.M.; Wong, K.; Monaghan, T.M.; Kao, D. Beneficial effects of fecal microbiota transplantation in recurrent Clostridioides difficile infection. Cell Host Microbe 2023, 31, 695-711. [CrossRef]
- Carlson, P.E., Jr. Regulatory Considerations for Fecal Microbiota Transplantation Products. Cell Host Microbe 2020, 27, 173-175. [CrossRef]
- Lavoie, T.; Appaneal, H.J.; LaPlante, K.L. Advancements in Novel Live Biotherapeutic Products for Clostridioides difficile Infection Prevention. Clin Infect Dis 2023, 77, S447-S454. [CrossRef]
- Fecal Microbiota for Transplantation: Safety Alert - Risk fo Serious Adverse Events Likely Due to Transmission of Pathogenic Organisms. US Food and Drug Administration: 2020.
- Hunt, A.; Drwiega, E.; Wang, Y.; Danziger, L. A review of fecal microbiota, live-jslm for the prevention of recurrent Clostridioides difficile infection. Am J Health Syst Pharm 2024, 81, e402-e411. [CrossRef]
- fecal microbiota, live-jslm (Rebyota) [package insert]. Ferring Pharmaceuticals.
- Feuerstadt, P.; Crawford, C.V.; Tan, X.; Pokhilko, V.; Bancke, L.; Ng, S.; Guthmueller, B.; Bidell, M.R.; Tillotson, G.; Johnson, S., et al. Fecal Microbiota, Live-jslm for the Prevention of Recurrent Clostridioides difficile Infection : Subgroup Analysis of PUNCH CD2 and PUNCH CD3. J Clin Gastroenterol 2024, 58, 818-824. [CrossRef]
- Khanna, S.; Assi, M.; Lee, C.; Yoho, D.; Louie, T.; Knapple, W.; Aguilar, H.; Garcia-Diaz, J.; Wang, G.P.; Berry, S.M., et al. Efficacy and Safety of RBX2660 in PUNCH CD3, a Phase III, Randomized, Double-Blind, Placebo-Controlled Trial with a Bayesian Primary Analysis for the Prevention of Recurrent Clostridioides difficile Infection. Drugs 2022, 82, 1527-1538. [CrossRef]
- Orenstein, R.; Dubberke, E.; Hardi, R.; Ray, A.; Mullane, K.; Pardi, D.S.; Ramesh, M.S.; Investigators, P.C. Safety and Durability of RBX2660 (Microbiota Suspension) for Recurrent Clostridium difficile Infection: Results of the PUNCH CD Study. Clin Infect Dis 2016, 62, 596-602. [CrossRef]
- Dubberke, E.R.; Orenstein, R.; Khanna, S.; Guthmueller, B.; Lee, C. Final Results from a Phase 2b Randomized, Placebo-Controlled Clinical Trial of RBX2660: A Microbiota-Based Drug for the Prevention of Recurrent Clostridioides difficile Infection. Infect Dis Ther 2023, 12, 703-709. [CrossRef]
- Feuerstadt, P.; Chopra, T.; Knapple, W.; Van Hise, N.W.; Dubberke, E.R.; Baggott, B.; Guthmueller, B.; Bancke, L.; Gamborg, M.; Steiner, T.S., et al. PUNCH CD3-OLS: a phase 3 prospective observational cohort study to evaluate the safety and efficacy of fecal microbiota, live-jslm (REBYOTA) in adults with recurrent Clostridioides difficile infection. Clin Infect Dis 2024. [CrossRef]
- Blair, H.A. SER-109 (VOWST()): A Review in the Prevention of Recurrent Clostridioides difficile Infection. Drugs 2024, 84, 329-336. [CrossRef]
- Feuerstadt, P.; Louie, T.J.; Lashner, B.; Wang, E.E.L.; Diao, L.; Bryant, J.A.; Sims, M.; Kraft, C.S.; Cohen, S.H.; Berenson, C.S., et al. SER-109, an Oral Microbiome Therapy for Recurrent Clostridioides difficile Infection. N Engl J Med 2022, 386, 220-229. [CrossRef]
- Fecal microbiota spors, live-brpk (Vowst) [package insert]. Seres Therapeutics, Inc: 2023.
- McGovern, B.H.; Ford, C.B.; Henn, M.R.; Pardi, D.S.; Khanna, S.; Hohmann, E.L.; O'Brien, E.J.; Desjardins, C.A.; Bernardo, P.; Wortman, J.R., et al. SER-109, an Investigational Microbiome Drug to Reduce Recurrence After Clostridioides difficile Infection: Lessons Learned From a Phase 2 Trial. Clin Infect Dis 2021, 72, 2132-2140. [CrossRef]
- Cohen, S.H.; Louie, T.J.; McGovern, B.H. Extended Follow-up of Microbiome Therapeutic SER-109 for Recurrent Clostridioides difficile Infection-Reply. JAMA 2023, 329, 1033-1034. [CrossRef]
- Berenson, C.S.; Lashner, B.; Korman, L.Y.; Hohmann, E.; Deshpande, A.; Louie, T.J.; Sims, M.; Pardi, D.; Kraft, C.S.; Wang, E.E.L., et al. Prevalence of Comorbid Factors in Patients With Recurrent Clostridioides difficile Infection in ECOSPOR III, a Randomized Trial of an Oral Microbiota-Based Therapeutic. Clin Infect Dis 2023, 77, 1504-1510. [CrossRef]
- Sims, M.D.; Khanna, S.; Feuerstadt, P.; Louie, T.J.; Kelly, C.R.; Huang, E.S.; Hohmann, E.L.; Wang, E.E.L.; Oneto, C.; Cohen, S.H., et al. Safety and Tolerability of SER-109 as an Investigational Microbiome Therapeutic in Adults With Recurrent Clostridioides difficile Infection: A Phase 3, Open-Label, Single-Arm Trial. JAMA Netw Open 2023, 6, e2255758. [CrossRef]
- Louie, T.; Golan, Y.; Khanna, S.; Bobilev, D.; Erpelding, N.; Fratazzi, C.; Carini, M.; Menon, R.; Ruisi, M.; Norman, J.M., et al. VE303, a Defined Bacterial Consortium, for Prevention of Recurrent Clostridioides difficile Infection: A Randomized Clinical Trial. JAMA 2023, 329, 1356-1366. [CrossRef]
- Magill, S.S.; Edwards, J.R.; Beldavs, Z.G.; Dumyati, G.; Janelle, S.J.; Kainer, M.A.; Lynfield, R.; Nadle, J.; Neuhauser, M.M.; Ray, S.M., et al. Prevalence of antimicrobial use in US acute care hospitals, May-September 2011. JAMA 2014, 312, 1438-1446. [CrossRef]
- Centers for Disease Control and Prevention. 2024. Emerging Infections Programs, healthcare-Associated Infections-Community Interface Surveillance Report, Clostridioides difficile infection (CDI). 2022.
- Webb, B.J.; Subramanian, A.; Lopansri, B.; Goodman, B.; Jones, P.B.; Ferraro, J.; Stenehjem, E.; Brown, S.M. Antibiotic Exposure and Risk for Hospital-Associated Clostridioides difficile Infection. Antimicrob Agents Chemother 2020, 64. [CrossRef]
- Miller, A.C.; Arakkal, A.T.; Sewell, D.K.; Segre, A.M.; Tholany, J.; Polgreen, P.M.; Group, C.D.C.M.-H. Comparison of Different Antibiotics and the Risk for Community-Associated Clostridioides difficile Infection: A Case-Control Study. Open Forum Infect Dis 2023, 10, ofad413. [CrossRef]
- Goldenberg, J.Z.; Yap, C.; Lytvyn, L.; Lo, C.K.; Beardsley, J.; Mertz, D.; Johnston, B.C. Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children. Cochrane Database Syst Rev 2017, 12, CD006095. [CrossRef]
- Goldenberg, J.Z.; Mertz, D.; Johnston, B.C. Probiotics to Prevent Clostridium difficile Infection in Patients Receiving Antibiotics. JAMA 2018, 320, 499-500. [CrossRef]
- Heil, E.L.; Harris, A.D.; Brown, C.; Seung, H.; Thom, K.A.; von Rosenvinge, E.; Sorongon, S.; Pineles, L.; Goodman, K.E.; Leekha, S. A Multicenter Evaluation of Probiotic Use for the Primary Prevention of Clostridioides difficile Infection. Clin Infect Dis 2021, 73, 1330-1337. [CrossRef]
- McFarland, L.V. Probiotics for the Primary and Secondary Prevention of C. difficile Infections: A Meta-analysis and Systematic Review. Antibiotics (Basel) 2015, 4, 160-178. [CrossRef]
- Donskey, C.J.; Dubberke, E.R.; Klein, N.P.; Liles, E.G.; Szymkowiak, K.; Wilcox, M.H.; Lawrence, J.; Bouguermouh, S.; Zhang, H.; Koury, K., et al. CLOVER: A Phase 3 Randomized Trial Investigating the Efficacy and Safety of a Detoxified Toxin A/B Vaccine in Adults 50 Years and Older at Increased Risk of Clostridioides difficile Infection. Clin Infect Dis 2024. [CrossRef]
- Christensen, S.; Bouguermouh, S.; Ilangovan, K.; Pride, M.W.; Webber, C.; Lockhart, S.P.; Shah, R.; Kitchin, N.; Lamberth, E.; Zhang, H., et al. A phase 3 study evaluating the lot consistency, immunogenicity, safety, and tolerability of a Clostridioides difficile vaccine in healthy adults 65 to 85 years of age. Vaccine 2023, 41, 7548-7559. [CrossRef]
- Razim, A.; Gorska, S.; Gamian, A. Non-Toxin-Based Clostridioides difficile Vaccination Approaches. Pathogens 2023, 12. [CrossRef]
- Carlson, T.J.; Gonzales-Luna, A.J. Utilizing antibiotics to prevent Clostridioides difficile infection: does exposure to a risk factor decrease risk? A systematic review. J Antimicrob Chemother 2020, 75, 2735-2742. [CrossRef]
- Johnson, S.W.; Brown, S.V.; Priest, D.H. Effectiveness of Oral Vancomycin for Prevention of Healthcare Facility-Onset Clostridioides difficile Infection in Targeted Patients During Systemic Antibiotic Exposure. Clin Infect Dis 2020, 71, 1133-1139. [CrossRef]
- Mullane, K.M.; Winston, D.J.; Nooka, A.; Morris, M.I.; Stiff, P.; Dugan, M.J.; Holland, H.; Gregg, K.; Adachi, J.A.; Pergam, S.A., et al. A Randomized, Placebo-controlled Trial of Fidaxomicin for Prophylaxis of Clostridium difficile-associated Diarrhea in Adults Undergoing Hematopoietic Stem Cell Transplantation. Clin Infect Dis 2019, 68, 196-203. [CrossRef]
- Reigadas, E.; van Prehn, J.; Falcone, M.; Fitzpatrick, F.; Vehreschild, M.; Kuijper, E.J.; Bouza, E.; European Society of Clinical, M.; Infectious Diseases Study Group on Clostridioides, d.; Study Group for, H., et al. How to: prophylactic interventions for prevention of Clostridioides difficile infection. Clin Microbiol Infect 2021, 27, 1777-1783. [CrossRef]
| IDSA/SHEA | ACG | |
|---|---|---|
| Initial CDI episode | Fidaxomicin 200 mg BID for 10 days (preferred) -OR- Vancomycin 125 mg QID for 10 days -OR- Metronidazole 500 mg TID for 10-14 days (non-severe CDI only and if vancomycin or fidaxomicin is unavailable) Adjunctive: bezlotoxumab 10 mg/kg IV oncea |
Non-Severe Vancomycin 125 mg QID for 10 days -OR- Fidaxomicin 200 mg BID for 10 days -OR- Metronidazole 500 mg TID for 10 days (low risk patientsc) Severe Vancomycin 125 mg QID for 10 days -OR- Fidaxomicin 200 mg BID for 10 days -OR- FMTd Adjunctive: bezlotoxumab 10mg/kg IV onceb |
| First recurrence | Fidaxomicin 200 mg BID for 10 days (preferred) -OR- Fidaxomicin 200 mg BID for 5 days, and then once daily every other day for 20 days (preferred) -OR- Vancomycin tapered and pulsed (eg. 125 mg QID for 10-14 days, BID for 7 days, daily for 7 days, and then every 2 to 3 days for 2 to 8 weeks) -OR- Vancomycin 125 mg QID for 10 days (if metronidazole was used for primary infection) Adjunctive: bezlotoxumab 10 mg/kg IV oncea |
Vancomycin tapered and pulsed -OR- Fidaxomicin 200 mg BID for 10 days (unless fidaxomicin was used for primary infection) Adjunctive: bezlotoxumab 10mg/kg IV onceb |
| Second or subsequent recurrence | Fidaxomicin 200 mg BID for 10 days -OR- Fidaxomicin 200 mg BID for 5 days, and then once daily every other day for 20 days -OR- Vancomycin tapered and pulsed (eg. 125 mg QID for 10-14 days, BID for 7 days, daily for 7 days, and then every 2 to 3 days for 2 to 8 weeks) -OR- Vancomycin 125 mg QID for 10 days followed by rifaximin 400 mg TID for 20 days -OR- FMT Adjunctive: bezlotoxumab 10 mg/kg IV oncea |
FMT Adjunctive: bezlotoxumab 10mg/kg IV once |
| Fulminant CDI | Vancomycin 500 QID -PLUS- Metronidazole IV 500 mg TID -PLUS- Vancomycin rectal 500 mg in 100 mL 0.9% sodium chloride QID (if ileus present) |
Vancomycin 500 QID for the first 48-72 hours, followed by 125 mg QID -PLUS- Metronidazole IV 500 mg TID -PLUS- Vancomycin rectal 500 mg in 100 mL 0.9% sodium chloride QID (if ileus present) -OR- FMTd |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





