Introduction
The diagnostic procedure for fallopian tube tumors commences with clinical assessment (patient interview), followed by physical examination (external and pelvic) and ultrasonography. The results of these tests will determine the presence or absence of fallopian tube and/or ovarian tumors. Moreover, to a certain extent, these tests allow for the determination of whether the tumor is benign or malignant. The tumor is benign in most cases when ultrasonography reveals a cystic tumor (sac-like structure). A large swollen ovary is called an ovarian tumor. Ovarian tumors are divided into the following two categories: cystic ovarian tumors (ovarian cysts), which are fluid-filled sac-like structures, and solid ovarian tumors, which are growing hard lumps. Approximately 80%–90% of ovarian tumors are ovarian cysts that collect fluid. Ovarian cysts are classified into the following four types: serous cystadenomas, mucinous cystadenomas, mature cystic teratomas, and chocolate cysts [
1].
However, malignant or borderline malignant tumors are suspected in cases wherein a mixture of solid (lump tissue) and cystic parts is noted or when the entire tumor is observed to be solid [
2]. Magnetic resonance imaging (MRI) or tumor marker measurement is performed when more detailed information is deemed necessary. To avoid unnecessary follow-up treatments (e.g., chemotherapy) following surgical procedures, the doctor in charge must comprehensively determine from the results of these tests whether the patient’s tumor is benign, malignant, or borderline malignant. However, as the accuracy of these tests is limited, a definitive diagnosis is ultimately made on the basis of the results of a surgical histopathological examination using the tumor tissue removed by surgical treatment.
Ultrasonography of a 68-year-old woman with no subjective symptoms revealed a right adnexal tumor measuring 97 × 61 mm consisting of a collection of microcysts. Cervical cancer screening results showed a diagnosis of negative for intraepithelial lesion or malignancy (NILM) [
3]. However, at the age of 51, she developed breast cancer and underwent hormone therapy; therefore, our medical staff suspected that she might have developed high-grade serous carcinoma. Furthermore, ultrasonography revealed two left adnexal tumors measuring 8 × 8 and 10 × 9 mm. Conversely, contrast MRI did not reveal any solid areas within the mass, and no findings suggestive of a malignant tumor were observed. However, the tumor was surgically removed owing to its large size. Based on the results of the surgical histopathological examination using the removed tumor tissue, the tumor was diagnosed as endosalpingiosis [
4]. Endosalpingiosis is not a malignant tumor; therefore, no additional treatment, including chemotherapy, is necessary. Tumors that develop in the bilateral gynecological appendages are not necessarily malignant tumors. To avoid unnecessary additional treatments (e.g., chemotherapy) following surgical treatment, the differential diagnosis between endosalpingiosis and ovarian borderline (low-grade) tumors or malignant tumor dissemination is essential.
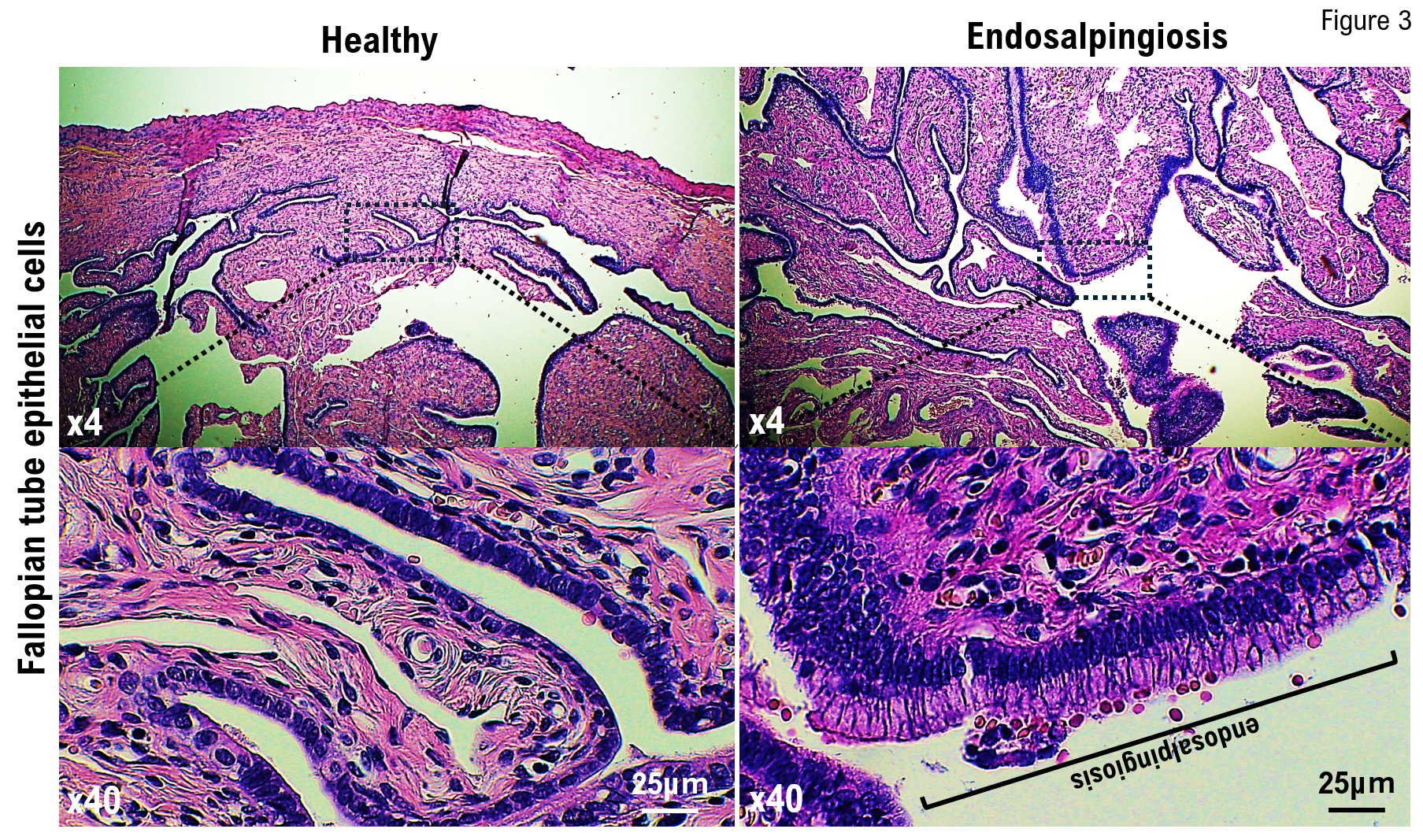
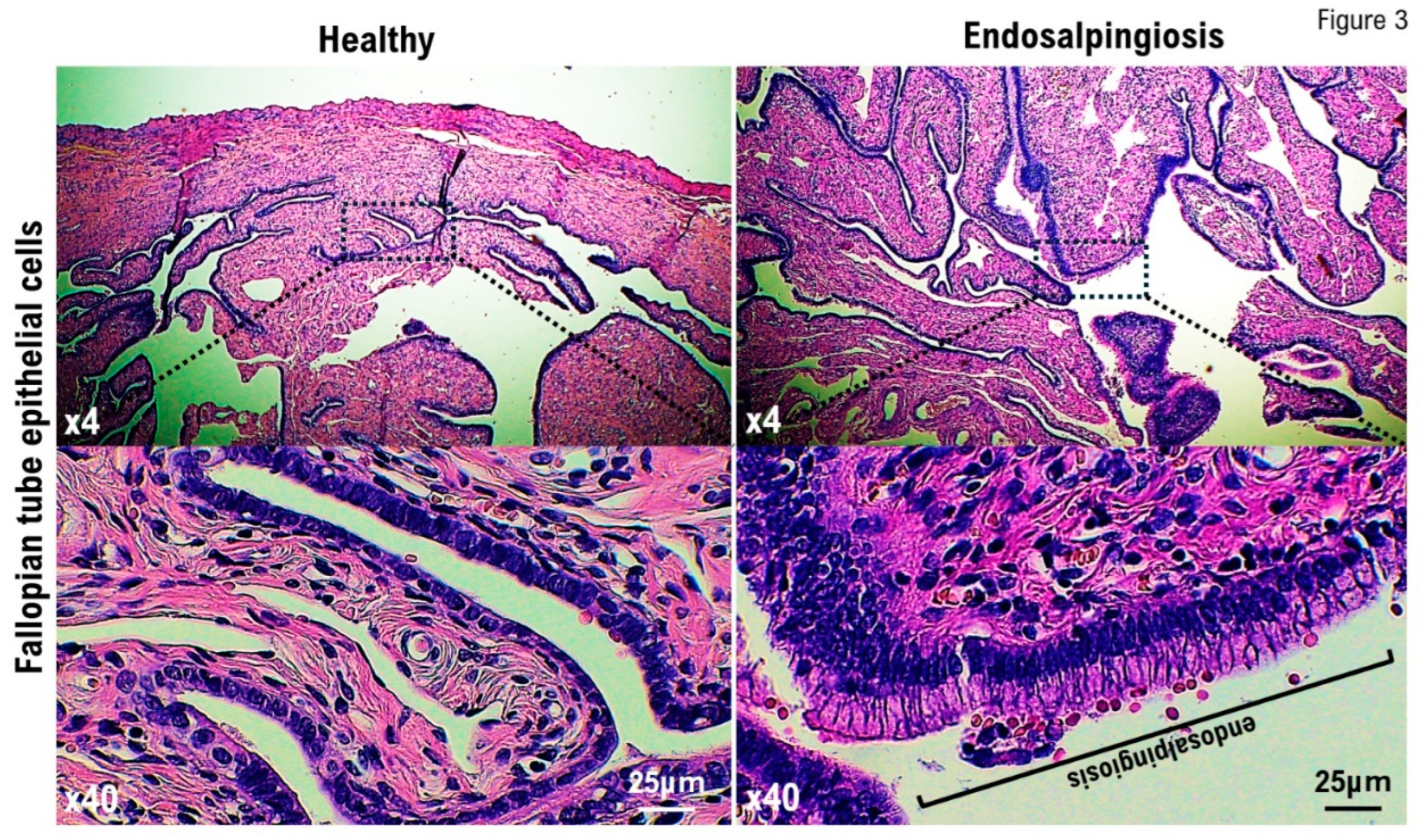
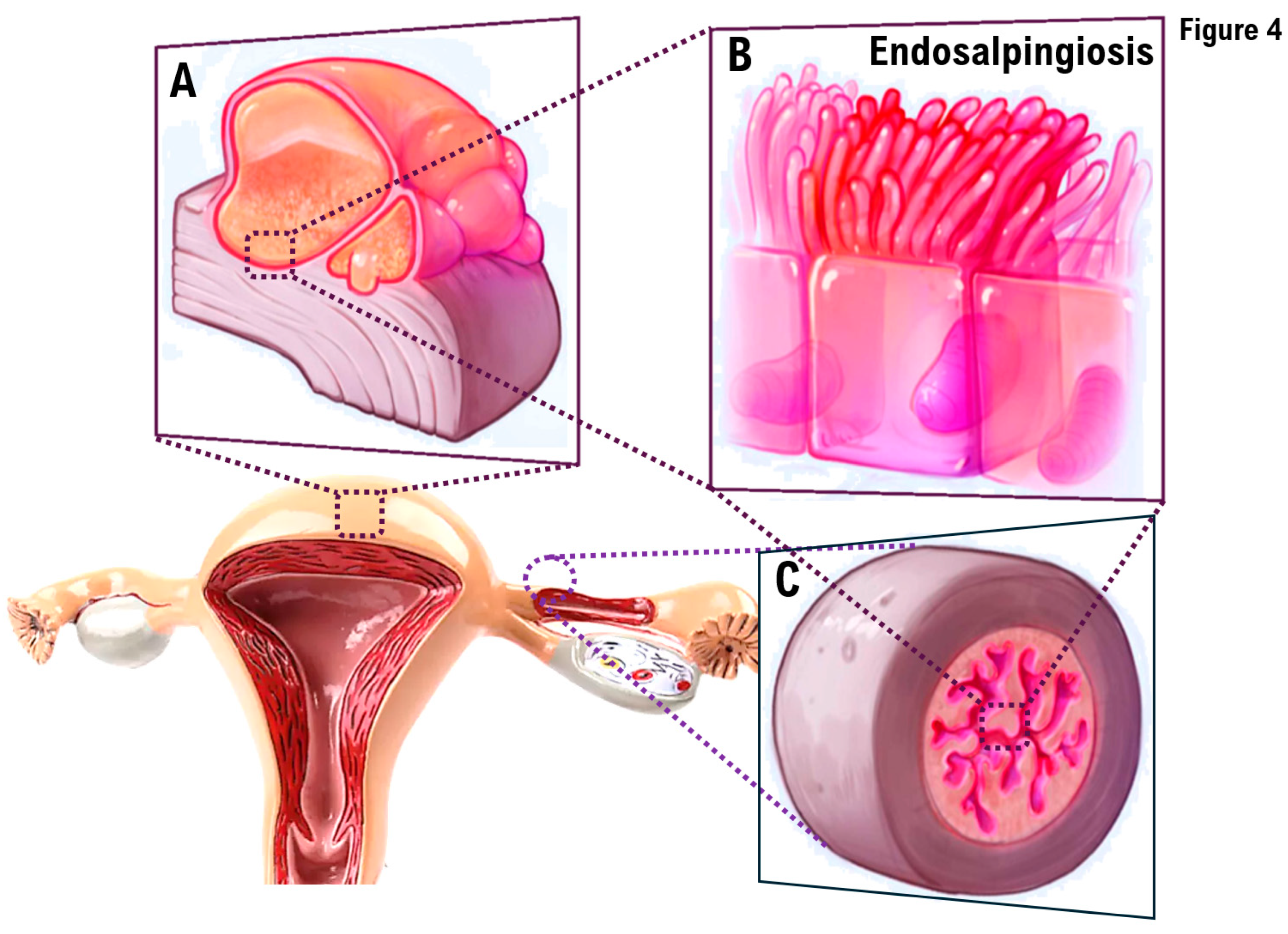
Endosalpingiosis is the presence of glands lined by benign tubal-type epithelium outside the fallopian tube [
4,
5].
Localization: peritoneum, pelvic or abdominal organs, skin, nerves, and lymph nodes.
Clinical features: It is usually an incidental finding.
Epidemiology: It occurs in approximately 12.5% of women, with a wide age range.
Materials and Methods
MRI
To determine the presence, size, and location of the patient’s mass, contrast-enhanced MRI was performed to localize the patient’s mass using MRI equipment (Vantage Centurian: Vantage Galan 3T MRT-3020, Canon Medical Systems, Inc., Ohtawara, Tochigi, Japan).
Laparoscopic Surgery
Pyloric gastrectomy and reconstruction of the remnant stomach and duodenum were performed by laparoscopic surgery using a laparoscope (ENDOEYE FLEX 3D, Olympus Corporation, Shinjuku, Tokyo, Japan) and a surgical device (HICURA, Olympus Corporation, Shinjuku, Tokyo, Japan) for surgically treating the gastric corpus area where the gastric tumor was detected.
Histopathological Examination
A surgical pathologist performed a histopathological analysis of the sections from the formalin-fixed paraffin-embedded resected tissue to assess the gross and histopathological characteristics of the resected specimens.
Using the standard procedure, hematoxylin and eosin staining analyses were performed.
Case
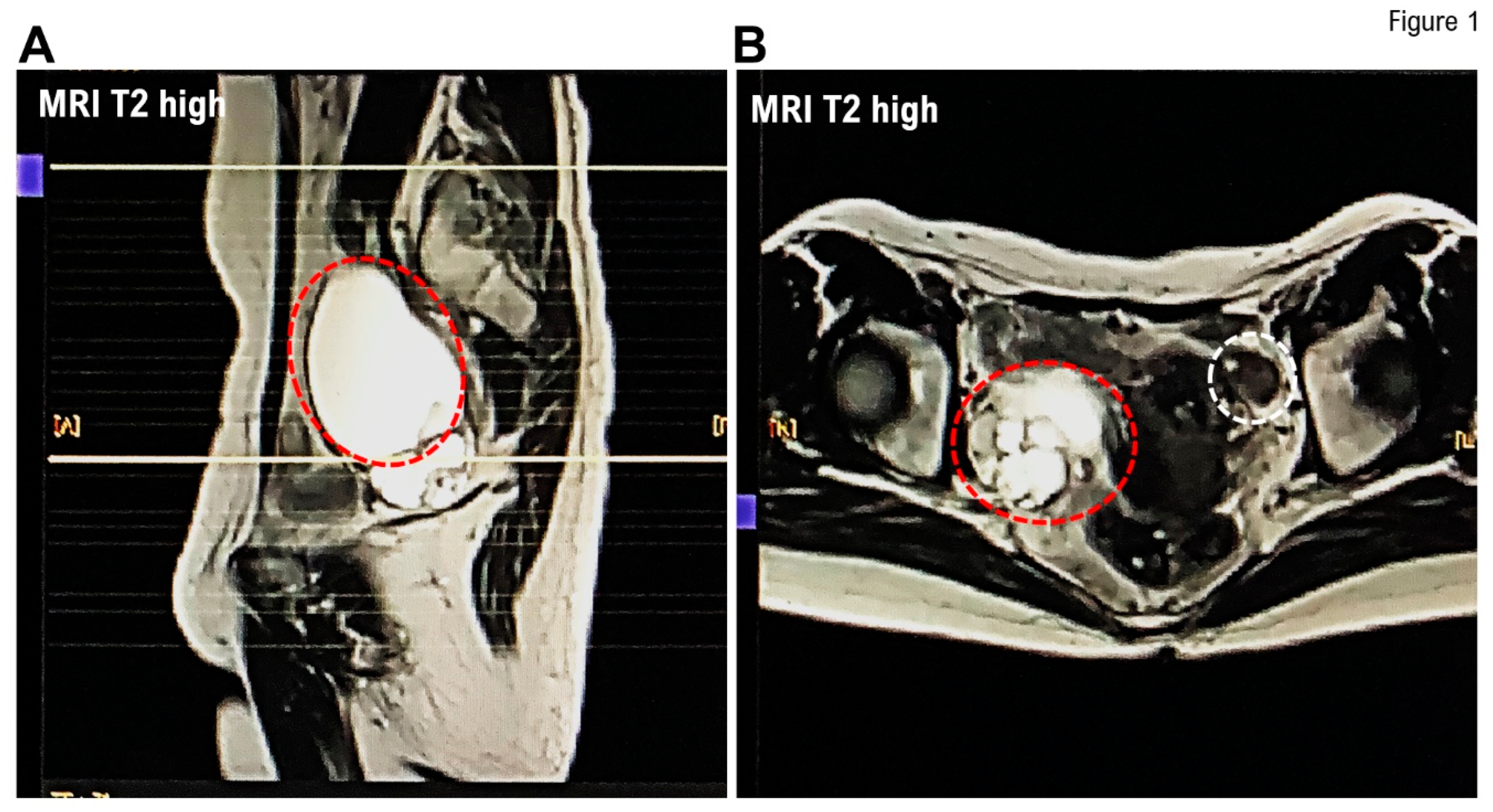
In April 2024, a 68-year-old woman visited a local hospital for a health checkup. At that time, her attending physician emphasized that she had developed ovarian enlargement. Ultrasonography revealed a right adnexal tumor containing a collection of small cysts measuring 97 × 61 mm and two left adnexal tumors containing a collection of small cysts measuring 8 × 8 and 10 × 9 mm. Contrast-enhanced T1low or T2high MRI revealed a homogeneous tumor mass in the left adnexa and a heterogeneous tumor mass in the right adnexa (
Figure 1). Contrast-enhanced MRI did not reveal any solid part within the mass; therefore, no findings suggestive of a malignant tumor development were noted.
Her doctor urgently referred her to our medical group for further examination and treatment. Contrast MRI revealed a multilocular cystic mass measuring approximately 70 mm in the short axis in the right ovary, with some stained glass patterns (
Figure 1). A homogeneous cluster of tumors was observed in her right ovary; however, no solid area was noted. Contrast-enhanced MRI revealed multiple small cysts and mild induration in the Douglas pouch and a mobile cystic mass that was believed to be a right adnexal mass on the cranial side of the uterine body. Furthermore, contrast-enhanced MRI revealed two small cysts in the left ovary; however, the cysts were not in contact with each other and were not diagnosed as neoplasm. In the uterine corpus, small uterine leiomyomas were observed; however, no endometrial hyperplasia was noted (
Figure 1). Therefore, her physician recommended surgical treatment, including laparoscopic total hemodialysis, bilateral salpingo-oophorectomy (BSO), and partial omentectomy.
Histological Findings
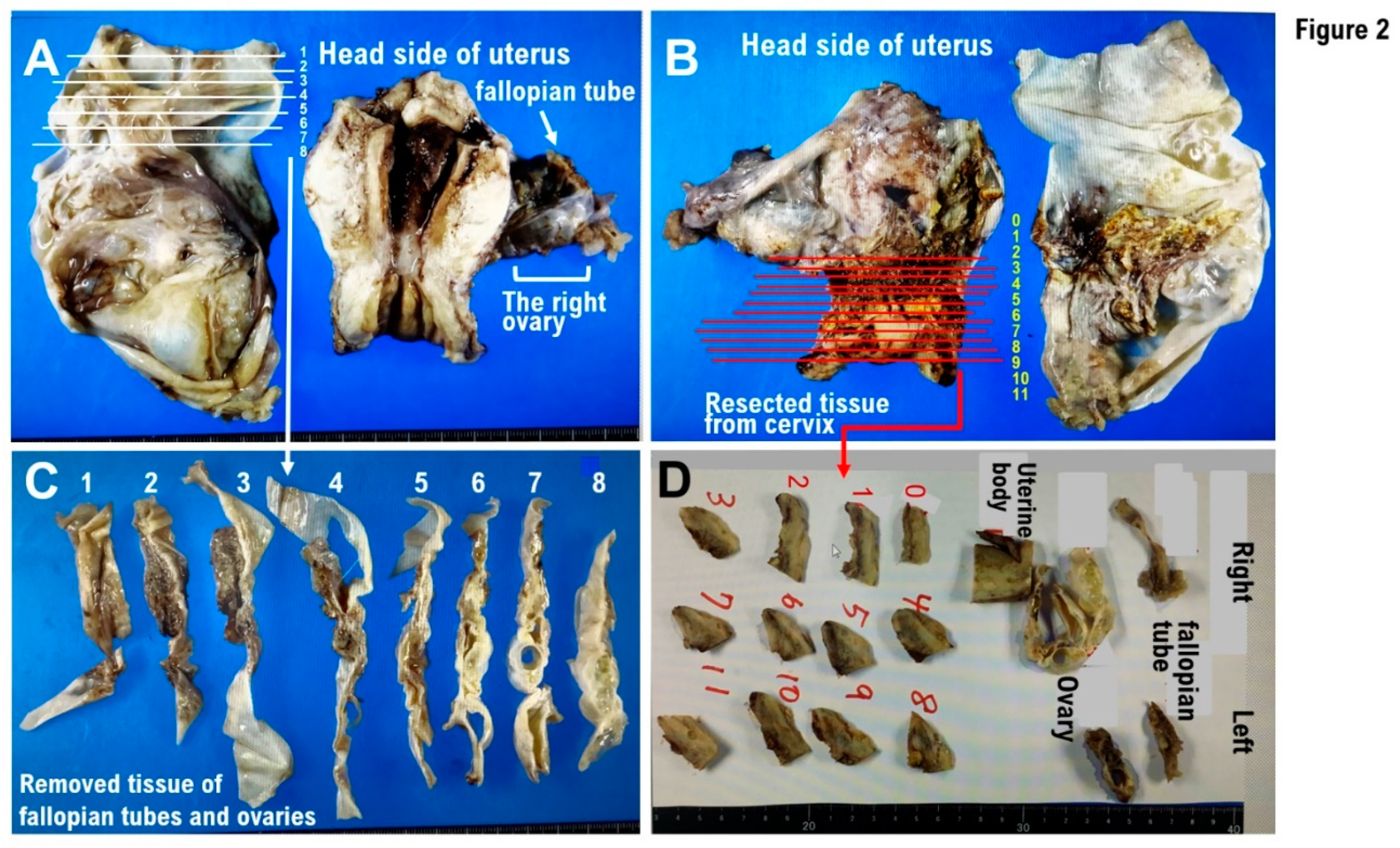
No evidence of mucinous cystadenomas or malignant or borderline malignant lesions was noted in the bilateral ovarian tissues. Leiomyomas, endometrial polyps, ectopic fallopian tube epithelium, and adenomyosis were noted in the uterine body tissue. No squamous intraepithelial lesion (SIL) or malignant findings were observed in the cervical tissue. Ectopic fallopian tube and calcified tissues were observed in the peritoneum of Douglas fossa (
Figure 2). Histological examinations revealed no malignant findings.
Histopathological examination
Clinical diagnosis: ovarian tumor and cervical dysplasia
Clinical course:
Suspected right ovarian mucinous adenoma (benign~border)
left ovarian cyst
History of cervical intraepithelial neoplasia (CIN) (details unknown, 2024/4: NILM). Laparoscopic total hysterectomy and BSO were performed owing to bilateral ovarian tumors and a history of CIN. The right ovary measured 9 cm in size and was multilocular.
Pathology findings
The specimens included total hysterectomy tissue specimen, bilateral adnexal specimen, and Douglas fossa peritoneal resection specimen. The ovaries and fallopian tubes were divided into eight sections, and all tissue blocks were used as specimens (
Figure 2A,C). In the right ovary, a multilocular cystic lesion containing mucus was noted (
Figure 2C) and no solid areas were observed. The tissue containing the lesion site was used as a specimen. The cervix was divided into 12 sections, and all tissue blocks were used as specimens (
Figure 2B,D). A 12-mm cyst was observed in the left ovarian tissue. The uterine corpus had a leiomyomatous nodule and an endometrial polyp.
Ovary and fallopian tube, bilateral, salpingo-oophorectomy:
- -
mucinous cystadenoma (bilateral ovaries)
Uterine corpus, hysterectomy:
- -
leiomyoma, endometrial polyp, endosalpingiosis, and adenomyosis
Uterine cervix, hysterectomy:
Peritoneum of Douglas fossa, excision:
Discussion
In recent years, as an individualized medicine, cancer treatment using antitumor agents selected on the basis of cancer gene panel testing results has been actively implemented in several countries for patients with advanced or metastatic cancer [
6]. Individuals with pathogenic variants (PVs) (pathogenic mutations and/or gene amplifications) in the
germline breast cancer susceptibility gene I (BRCA1) gene or
gBRCA2 gene are prone to developing early onset breast cancer in their 30s [
7,
8]. Those with
gBRCA1 PVs and/or
gBRCA2 PVs are prone to developing high-grade serous ovarian cancer within 10 years of the first breast cancer onset. Therefore, BRACAnalysis diagnostic system testing (Myriad Genetics, Inc., Salt Lake City, UT, USA) was performed on the patient, and the results showed no
gBRCA1 PVs and/or
gBRCA2 PVs. Hormone treatment administered for breast cancer sparing can increase the incidence of gynecological malignancies, including ovarian and/or fallopian tube cancers [
9]. The patient was undergoing hormone treatment for breast cancer sparing. Therefore, we suspected the development of fallopian tube and ovarian cancers induced by hormone treatment for breast cancer preservation. Ultimately, following a histopathological examination, her ovarian tumor was diagnosed as endosalpingiosis.
The etiology of endosalpingiosis is believed to be due to the coelomic metaplasia theory, in which tissues derived from the Mullerian duct invade and proliferate within the coelomic epithelium during development, and the transplantation theory, in which shed tissues reflux into the peritoneal cavity [
10,
11]. Surgical treatments, including tubal ligation and salpingectomy, as well as chronic pelvic inflammatory diseases, including salpingitis, are believed to be triggers for endosalpingitis. Furthermore, although the clinical significance of endosalpingiosis remains unclear, endosalpingiosis frequently occurs in cases of low-grade serous ovarian cancer and borderline tumors [
12]. It is believed that the fallopian tube epithelium is the most likely source of high-grade serous ovarian cancer [
13]. However, as the mechanism of development of low-grade serous ovarian cancer remains unknown, research into the relationship between endosalpingiosis and serous ovarian cancer has been ongoing based on the abovementioned reports. In the present case, our medical staff employed a 4K camera to demonstrate that the white lesion approximately 3 mm in size with a 1-mm transparent cyst formation in the center, located in the postmenopausal Douglas cavity peritoneum, was endosalpingiosis.
Conclusions
Tumors arising in the bilateral gynecological appendages are not necessarily malignant. The differential diagnosis between endosalpingiosis and ovarian borderline (low-grade) tumors or malignant tumor dissemination is significant to avoid unnecessary additional treatments, including chemotherapy, following surgical treatment.
Author Contributions
All authors had full access to the data in the study and take responsibility for the integrity of the data and accuracy of the data analysis. Y.M. Y.A. and K.A. performed the recommendation operations (TLH, BSO, pOM). Conceptualization, T.H. and I.K.; writing—original draft, T.H. and I.K.; writing—review & editing, I.K.; visualization, T.H. and I.K.; supervision, T.H. and I.K.; funding acquisition, T.H. and I.K. All authors have read and agreed to the published version of the manuscript.
Funding
This clinical research was performed with research funding from the following: Japan Society for Promoting Science for T.H. (Grant No. 19K09840), START-program Japan Science and Technology Agency for T.H. (Grant No. STSC20001), and the National Hospital Organization Multicenter clinical study for T.H. (Grant No. 2019-Cancer in general-02), and The Japan Agency for Medical Research and Development (AMED) (Grant No. 22ym0126802j0001), Tokyo, Japan.
Institutional Review Board Statement
This study was reviewed and approved by the Central Ethics Review Board of the National Hospital Organization Headquarters in Japan (Tokyo, Japan) on 8 November 2019, and Kyoto University School of Medicine (Kyoto, Japan) on 17 August 2019, with approval codes NHO H31-02 and M192. The completion numbers for the authors are AP0000151756, AP0000151757, AP0000151769, and AP000351128. As this research was considered clinical research, consent to participate was required. After briefing regarding the clinical study and approval of the research contents, the participants signed an informed consent form.
Informed Consent Statement
A multi-center retrospective observational clinical study of subjects who underwent cancer genomic medicine at a cancer medical facility in Kyoto, Japan. This study was reviewed and approved by the Central Ethics Review Board of the National Hospital Organization Headquarter in Japan (Tokyo, Japan) on 18 November 2020, and Kyoto University School of Medicine (Kyoto, Japan) on 24 August 2022, with approval codes NHO R4-04 and M237. All participants agreed to take part in the present study. We have obtained Informed Consent Statements from people participating in clinical studies.
Data Availability Statement
Data available on request due to restrictions eg privacy or ethical The data presented in this study are available on request from the corresponding author.
Acknowledgments
We thank all medical staff for clinical research at Kyoto University Hospital and the National Hospital Organization Kyoto Medical Center.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Matalliotakis M, Matalliotaki C, Tsakiridis I, Dagklis T, Michos G, Romanos A, Krithinakis K, Kalogiannidis IA. Co-existence of Ovarian Teratomas With Other Gynecological Tumors. Cureus. 2024 Apr 11;16(4):e58068. [CrossRef]
- Chui MH, Song Q, Zhu J, Jiao Y, Wang B, Wang Y, Wang TL, Vang R, Shih IM. Early Genetic Divergence of High-Grade Carcinomas Originating from Low-Grade Serous Ovarian Neoplasms. Mod Pathol. 2024 Oct 9;38(1):100629. [CrossRef]
- Cui X, Yang D, Zhang J, Zhao Y, Cui Z, Wang C, Qiao Y. Clinical value of optical coherence tomography in the early diagnosis of cervical cancer and precancerous lesions: a cross-sectional study. Front Oncol. 2024 Jul 29;14:1423128. [CrossRef]
- Shapiro S, Abdulhak A, Lenczowski J, Sayeed S, Mochel M.J Cutaneous endosalpingiosis arising from C-section scar: A case report with review of literature. Cutan Pathol. 2023 Apr;50(4):310-315. [CrossRef]
- Chui MH, Shih IM. Oncogenic BRAF and KRAS mutations in endosalpingiosis. J Pathol. 2020 Feb;250(2):148-158. [CrossRef]
- Grellety T, Lucchesi C, Hostein I, Auzanneau C, Khalifa E, Soubeyran I, Italiano A. High-depth sequencing of paired primary and metastatic tumours: Implications for personalised medicine. Eur J Cancer. 2017 Oct;84:250-256. [CrossRef]
- Zhang Z, Ye S, Bernhardt SM, Nelson HD, Velie EM, Borges VF, Woodward ER, Evans DGR, Schedin PJ. Postpartum Breast Cancer and Survival in Women With Germline BRCA Pathogenic Variants. JAMA Netw Open. 2024 Apr 1;7(4):e247421. [CrossRef]
- Yerushalmi R, Pomerantz A, Lewin R, Paluch-Shimon S, Soussan-Gutman L, Baehner FL, Voet H, Bareket-Samish A, Kedar I, Goldberg Y, Peretz-Yablonski T, Kadouri L. BRCA1/2 mutation carriers vs the general breast cancer population (N = 799,986): 21-gene assay-based molecular characterization. Breast Cancer Res Treat. 2024 Jul;206(1):67-76. [CrossRef]
- Kuo DY, Jones J, Fields AL, Runowicz CD, Goldberg GL. Endometrioid adenocarcinoma of the ovary and long-term tamoxifen therapy: a coincidence or a cause for concern? Eur J Gynaecol Oncol. 1997;18(6):457-60.
- Yığıt S, Dere Y, Yetımalar H, Etıt D. Tumor-like cystic endosalpingiosis in the myometrium: a case report and a review of the literature. Turk Patoloji Derg. 2014;30(2):145-8. [CrossRef]
- Shield P. Peritoneal washing cytology. Cytopathology. 2004 Jun;15(3):131-41. [CrossRef]
- Sunde J, Wasickanin M, Katz TA, Wickersham EL, Steed DOE, Simper N. Prevalence of endosalpingiosis and other benign gynecologic lesions. PLOS ONE, 2020;13;15:e0232487 .
- Lisio MA, Fu L, Goyeneche A, Gao ZH, Telleria C. High-Grade Serous Ovarian Cancer: Basic Sciences, Clinical and Therapeutic Standpoints. Int. J. Mol. Sci. 2019;20:952..
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).