Submitted:
25 November 2024
Posted:
27 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Key Genes, Genomic Regions, and Metabolic Pathways Associated with MacTel
2.1. Familial Occurrence of MacTel
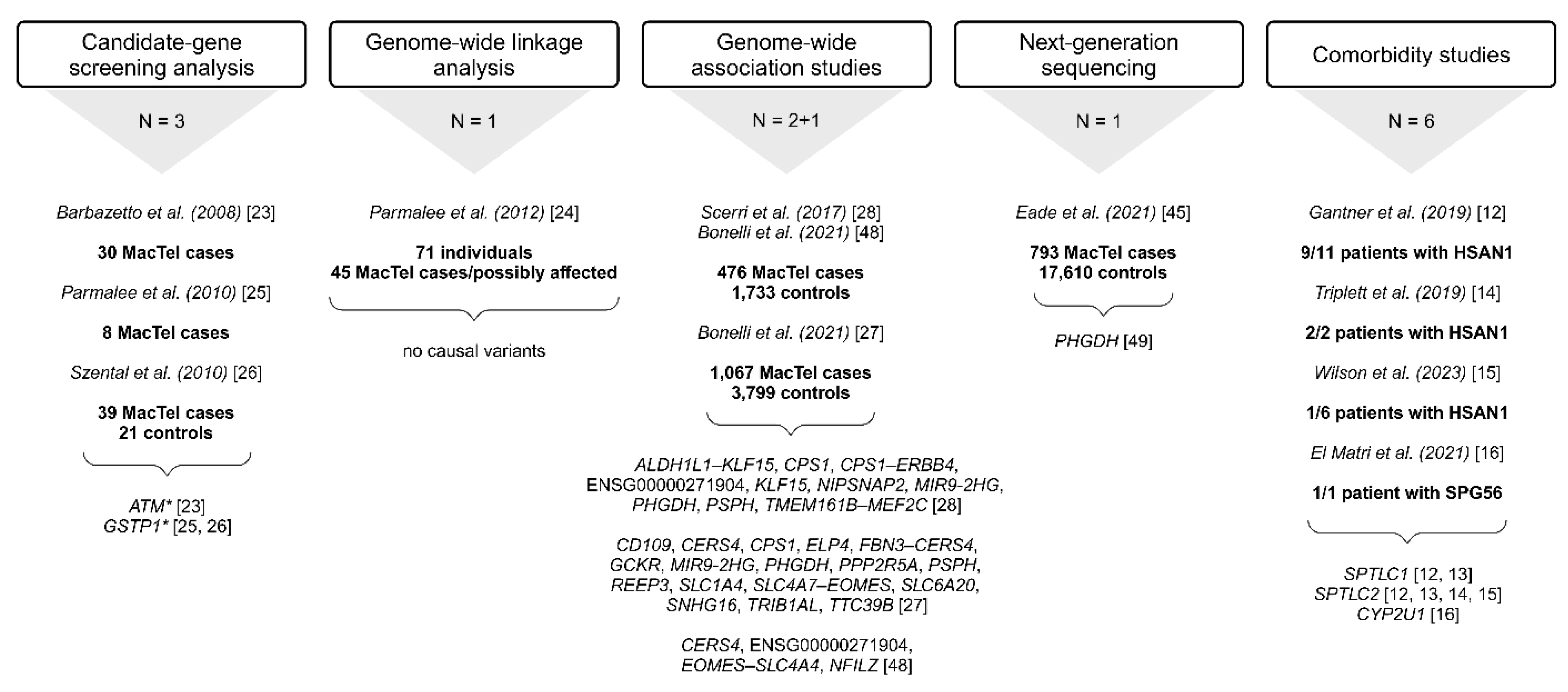
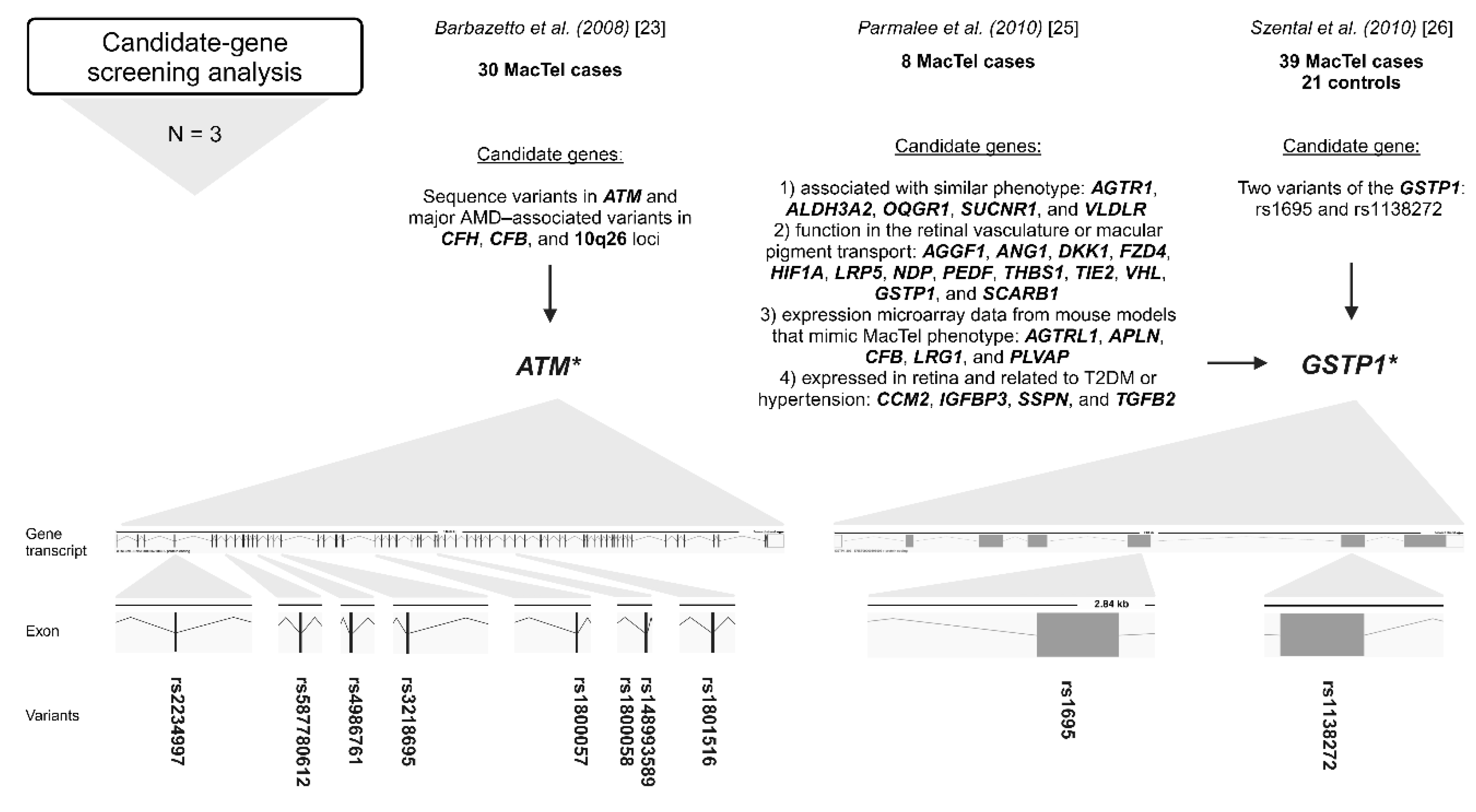
2.2. Candidate Gene-Screening Analysis
2.3. Genome-Wide Linkage Analysis
2.4. Genome-Wide Association Studies
2.5. Next-Generation Sequencing Studies
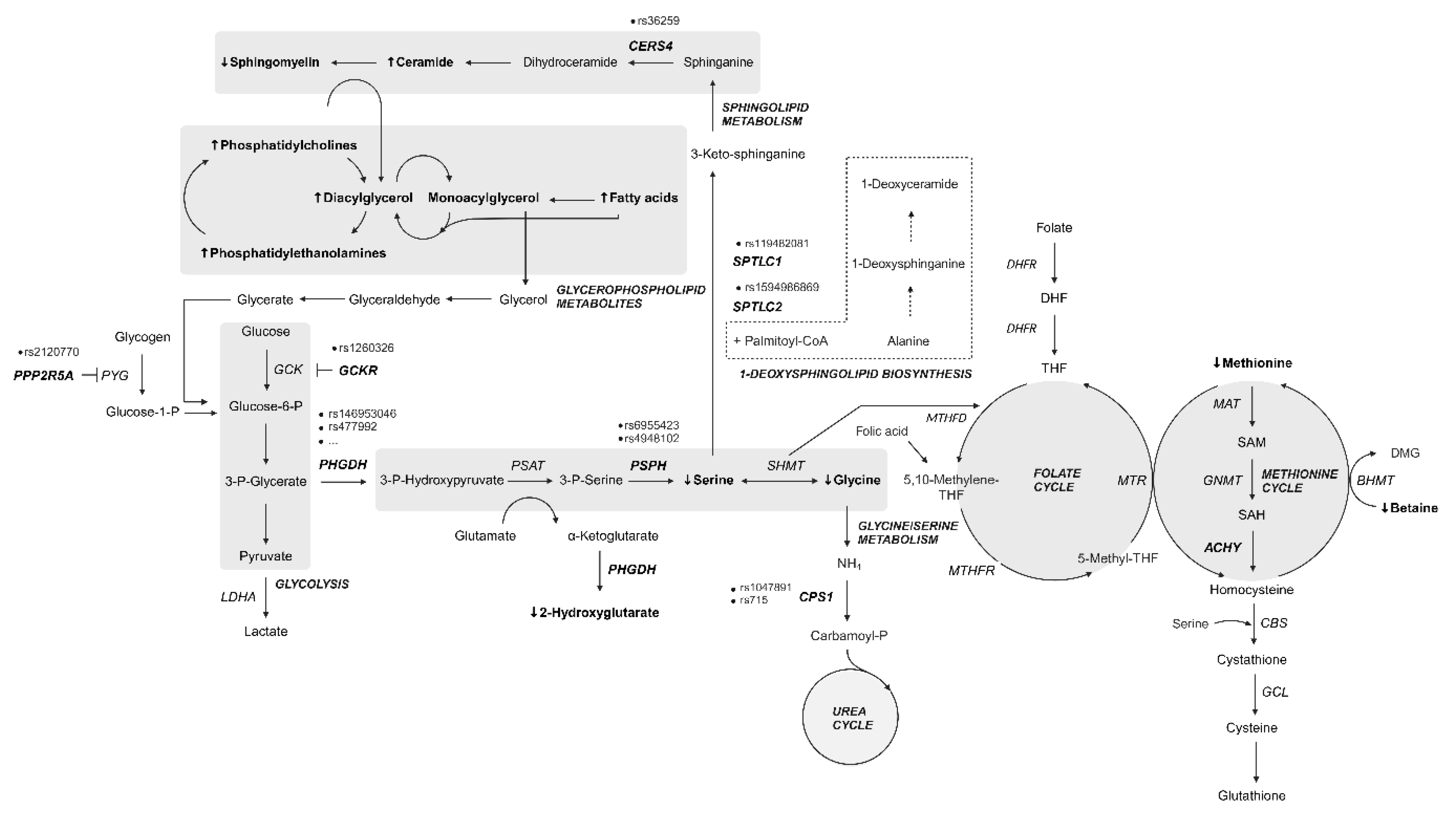
2.6. Implications of Risk-Associated Metabolic Pathways on MacTel Pathogenesis
2.7. Association of MacTel with Comorbidities
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Clemons TE, Gillies MC, Chew EY, Bird AC, Peto T, Figueroa MJ; et al. Baseline characteristics of participants in the natural history study of macular telangiectasia (MacTel) MacTel Project Report No. 2. Ophthalmic epidemiology. 2010;17(1):66-73. [CrossRef]
- Gass JD, Blodi BA. Idiopathic juxtafoveolar retinal telangiectasis. Update of classification and follow-up study. Ophthalmology. 1993;100(10):1536-46. [CrossRef]
- Charbel Issa P, Gillies MC, Chew EY, Bird AC, Heeren TF, Peto T; et al. Macular telangiectasia type 2. Progress in retinal and eye research. 2013;34:49-77. [CrossRef]
- Balaskas K, Leung I, Sallo FB, Clemons TE, Bird AC, Peto T. Associations between autofluorescence abnormalities and visual acuity in idiopathic macular telangiectasia type 2: MacTel project report number 5. Retina (Philadelphia, Pa). 2014;34(8):1630-6. [CrossRef]
- Clemons TE, Gillies MC, Chew EY, Bird AC, Peto T, Figueroa M; et al. The National Eye Institute Visual Function Questionnaire in the Macular Telangiectasia (MacTel) Project. Investigative ophthalmology & visual science. 2008;49(10):4340-6.
- Clemons TE, Gillies MC, Chew EY, Bird AC, Peto T, Wang JJ; et al. Medical characteristics of patients with macular telangiectasia type 2 (MacTel Type 2) MacTel project report no. 3. Ophthalmic epidemiology. 2013;20(2):109-13.
- Heeren TFC, Chew EY, Clemons T, Fruttiger M, Balaskas K, Schwartz R; et al. Macular Telangiectasia Type 2: Visual Acuity, Disease End Stage, and the MacTel Area: MacTel Project Report Number 8. Ophthalmology. 2020;127(11):1539-48.
- Peto T, Heeren TFC, Clemons TE, Sallo FB, Leung I, Chew EY; et al. CORRELATION OF CLINICAL AND STRUCTURAL PROGRESSION WITH VISUAL ACUITY LOSS IN MACULAR TELANGIECTASIA TYPE 2: MacTel Project Report No. 6-The MacTel Research Group. Retina (Philadelphia, Pa). 2018;38 Suppl 1(Suppl 1):S8-s13.
- Vujosevic S, Heeren TFC, Florea D, Leung I, Pauleikhoff D, Sallo F; et al. SCOTOMA CHARACTERISTICS IN MACULAR TELANGIECTASIA TYPE 2: MacTel Project Report No. 7-The MacTel Research Group. Retina (Philadelphia, Pa). 2018;38 Suppl 1:S14-s9.
- Zhang Q, Wang RK, Chen CL, Legarreta AD, Durbin MK, An L; et al. Swept Source Optical Coherence Tomography Angiography of Neovascular Macular Telangiectasia Type 2. Retina (Philadelphia, Pa). 2015;35(11):2285-99.
- Gillies MC, Zhu M, Chew E, Barthelmes D, Hughes E, Ali H; et al. Familial asymptomatic macular telangiectasia type 2. Ophthalmology. 2009;116(12):2422-9.
- Gantner ML, Eade K, Wallace M, Handzlik MK, Fallon R, Trombley J; et al. Serine and Lipid Metabolism in Macular Disease and Peripheral Neuropathy. The New England journal of medicine. 2019;381(15):1422-33.
- Rodrigues FG, Pipis M, Heeren TFC, Fruttiger M, Gantner M, Vermeirsch S; et al. Description of a patient cohort with Hereditary Sensory Neuropathy type 1 without retinal disease Macular Telangiectasia type 2 - implications for retinal screening in HSN1. Journal of the peripheral nervous system : JPNS. 2022;27(3):215-24.
- Triplett J, Nicholson G, Sue C, Hornemann T, Yiannikas C. Hereditary sensory and autonomic neuropathy type IC accompanied by upper motor neuron abnormalities and type II juxtafoveal retinal telangiectasias. Journal of the peripheral nervous system : JPNS. 2019;24(2):224-9.
- Wilson LMQ, Saba S, Li J, Prasov L, Miller JML. Specific Deoxyceramide Species Correlate with Expression of Macular Telangiectasia Type 2 (MacTel2) in a SPTLC2 Carrier HSAN1 Family. Genes. 2023;14(4).
- El Matri K, Falfoul Y, Habibi I, Chebil A, Schorderet D, El Matri L. Macular Dystrophy with Bilateral Macular Telangiectasia Related to the CYP2U1 Pathogenic Variant Assessed with Multimodal Imaging Including OCT-Angiography. Genes. 2021;12(11).
- Yannuzzi LA, Bardal AM, Freund KB, Chen KJ, Eandi CM, Blodi B. Idiopathic macular telangiectasia. Archives of ophthalmology (Chicago, Ill : 1960). 2006;124(4):450-60.
- Chew EY, Peto T, Clemons TE, Sallo FB, Pauleikhoff D, Leung I; et al. Macular Telangiectasia Type 2: A Classification System Using MultiModal Imaging MacTel Project Report Number 10. Ophthalmology science. 2023;3(2):100261.
- Toygar O, Guess MG, Youssef DS, Miller DM. Long-term Ootcomes of Intravitreal Bevacizumab Therapy for Subretinal Neovascularization Secondary to Idiopathic Macular Telangiectasia Type 2. Retina (Philadelphia, Pa). 2016;36(11):2150-7.
- Aung KZ, Wickremasinghe SS, Makeyeva G, Robman L, Guymer RH. The prevalence estimates of macular telangiectasia type 2: The Melbourne Collaborative Cohort Study. Retina (Philadelphia, Pa). 2010;30(3):473-8.
- Klein R, Blodi BA, Meuer SM, Myers CE, Chew EY, Klein BE. The prevalence of macular telangiectasia type 2 in the Beaver Dam eye study. American journal of ophthalmology. 2010;150(1):55-62.e2.
- Ronquillo CC, Wegner K, Calvo CM, Bernstein PS. Genetic Penetrance of Macular Telangiectasia Type 2. JAMA ophthalmology. 2018;136(10):1158-63.
- Barbazetto IA, Room M, Yannuzzi NA, Barile GR, Merriam JE, Bardal AM; et al. ATM gene variants in patients with idiopathic perifoveal telangiectasia. Investigative ophthalmology & visual science. 2008;49(9):3806-11.
- Parmalee NL, Schubert C, Figueroa M, Bird AC, Peto T, Gillies MC; et al. Identification of a potential susceptibility locus for macular telangiectasia type 2. PLoS ONE. 2012;7(8):e24268.
- Parmalee NL, Schubert C, Merriam JE, Allikmets K, Bird AC, Gillies MC; et al. Analysis of candidate genes for macular telangiectasia type 2. Molecular vision. 2010;16:2718-26.
- Szental JA, Baird PN, Richardson AJ, Islam FM, Scholl HP, Charbel Issa P; et al. Analysis of glutathione S-transferase Pi isoform (GSTP1) single-nucleotide polymorphisms and macular telangiectasia type 2. International ophthalmology. 2010;30(6):645-50.
- Bonelli R, Jackson VE, Prasad A, Munro JE, Farashi S, Heeren TFC; et al. Identification of genetic factors influencing metabolic dysregulation and retinal support for MacTel, a retinal disorder. Communications biology. 2021;4(1):274.
- Scerri TS, Quaglieri A, Cai C, Zernant J, Matsunami N, Baird L; et al. Genome-wide analyses identify common variants associated with macular telangiectasia type 2. Nature genetics. 2017;49(4):559-67.
- Bonelli R, Woods SM, Ansell BRE, Heeren TFC, Egan CA, Khan KN; et al. Systemic lipid dysregulation is a risk factor for macular neurodegenerative disease. Scientific reports. 2020;10(1):12165.
- Seal RL, Braschi B, Gray K, Jones TEM, Tweedie S, Haim-Vilmovsky L; et al. Genenames.org: The HGNC resources in 2023. Nucleic acids research. 2023;51(D1):D1003-d9.
- Sayers EW, Bolton EE, Brister JR, Canese K, Chan J, Comeau DC; et al. Database resources of the national center for biotechnology information. Nucleic acids research. 2022;50(D1):D20-d6.
- Karczewski KJ, Francioli LC, Tiao G, Cummings BB, Alföldi J, Wang Q; et al. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature. 2020;581(7809):434-43.
- Harrison PW, Amode MR, Austine-Orimoloye O, Azov Andrey G, Barba M, Barnes I; et al. Ensembl 2024. Nucleic acids research. 2023;52(D1):D891-D9.
- Hutton WL, Snyder WB, Fuller D, Vaiser A. Focal parafoveal retinal telangiectasis. Archives of ophthalmology (Chicago, Ill : 1960). 1978;96(8):1362-7.
- Putteman A, Toussaint D, Graff E, Verougstraete C. [Idiopathic familial juxtafoveolar retinal telangiectasias]. Bulletin de la Societe belge d’ophtalmologie. 1984;209:81-90.
- Isaacs TW, McAllister IL. Familial idiopathic juxtafoveolar retinal telangiectasis. Eye (London, England). 1996;10 ( Pt 5):639-42.
- Oh KT, Park DW. Bilateral juxtafoveal telangiectasis in a family. Retina (Philadelphia, Pa). 1999;19(3):246-7.
- Delaere L, Spielberg L, Leys AM. Vertical transmission of macular telangiectasia type 2. Retinal cases & brief reports. 2012;6(3):253-7.
- Menchini U, Virgili G, Bandello F, Malara C, Rapizzi E, Lanzetta P. Bilateral juxtafoveolar telangiectasis in monozygotic twins. American journal of ophthalmology. 2000;129(3):401-3.
- Siddiqui N, Fekrat S. Group 2A idiopathic juxtafoveolar retinal telangiectasia in monozygotic twins. American journal of ophthalmology. 2005;139(3):568-70.
- Hannan SR, Madhusudhana KC, Rennie C, Lotery AJ. Idiopathic juxtafoveolar retinal telangiectasis in monozygotic twins. The British journal of ophthalmology. 2007;91(12):1729-30.
- Lin H, Wu H, Li H, Song A, Yin W. The essential role of GSTP1 I105V polymorphism in the prediction of CDNB metabolism and toxicity: In silico and in vitro insights. Toxicology in vitro : An international journal published in association with BIBRA. 2023;90:105601.
- Chen YC, Tzeng CH, Chen PM, Lin JK, Lin TC, Chen WS; et al. Influence of GSTP1 I105V polymorphism on cumulative neuropathy and outcome of FOLFOX-4 treatment in Asian patients with colorectal carcinoma. Cancer science. 2010;101(2):530-5.
- Watson MA, Stewart RK, Smith GB, Massey TE, Bell DA. Human glutathione S-transferase P1 polymorphisms: Relationship to lung tissue enzyme activity and population frequency distribution. Carcinogenesis. 1998;19(2):275-80.
- Pan WW, Wubben TJ, Besirli CG. Photoreceptor metabolic reprogramming: Current understanding and therapeutic implications. Communications biology. 2021;4(1):245.
- Karolchik D, Hinrichs AS, Furey TS, Roskin KM, Sugnet CW, Haussler D; et al. The UCSC Table Browser data retrieval tool. Nucleic acids research. 2004;32(Database issue):D493-6.
- Nassar LR, Barber GP, Benet-Pagès A, Casper J, Clawson H, Diekhans M; et al. The UCSC Genome Browser database: 2023 update. Nucleic acids research. 2023;51(D1):D1188-d95.
- Bonelli R, Ansell BRE, Lotta L, Scerri T, Clemons TE, Leung I; et al. Genetic disruption of serine biosynthesis is a key driver of macular telangiectasia type 2 aetiology and progression. Genome medicine. 2021;13(1):39.
- Eade K, Gantner ML, Hostyk JA, Nagasaki T, Giles S, Fallon R; et al. Serine biosynthesis defect due to haploinsufficiency of PHGDH causes retinal disease. Nature metabolism. 2021;3(3):366-77.
- Baumann B, Sterling J, Song Y, Song D, Fruttiger M, Gillies M; et al. Conditional Müller Cell Ablation Leads to Retinal Iron Accumulation. Investigative ophthalmology & visual science. 2017;58(10):4223-34.
- Byrne LC, Khalid F, Lee T, Zin EA, Greenberg KP, Visel M; et al. AAV-mediated, optogenetic ablation of Müller Glia leads to structural and functional changes in the mouse retina. PLoS ONE. 2013;8(9):e76075.
- Eade KT, Ansell BRE, Giles S, Fallon R, Harkins-Perry S, Nagasaki T; et al. iPSC-derived retinal pigmented epithelial cells from patients with macular telangiectasia show decreased mitochondrial function. The Journal of clinical investigation. 2023;133(9).
- Hua J, Guerin KI, Chen J, Michán S, Stahl A, Krah NM; et al. Resveratrol inhibits pathologic retinal neovascularization in Vldlr(-/-) mice. Investigative ophthalmology & visual science. 2011;52(5):2809-16.
- Thomas ED, Timms AE, Giles S, Harkins-Perry S, Lyu P, Hoang T; et al. Cell-specific cis-regulatory elements and mechanisms of non-coding genetic disease in human retina and retinal organoids. Developmental cell. 2022;57(6):820-36.e6.
- Zhao M, Andrieu-Soler C, Kowalczuk L, Paz Cortés M, Berdugo M, Dernigoghossian M; et al. A new CRB1 rat mutation links Müller glial cells to retinal telangiectasia. The Journal of neuroscience : The official journal of the Society for Neuroscience. 2015;35(15):6093-106.
- Pauleikhoff L, Wingert V, Grünert SC, Lange C, Hannibal L, Bucher F. Methylation-associated Pathways in Macular Telangiectasia Type 2 and Ophthalmologic Findings in Patients with Genetic Methylation Disorders. Retina (Philadelphia, Pa). 2024;44(6):1052-62.
- Lotta LA, Pietzner M, Stewart ID, Wittemans LBL, Li C, Bonelli R; et al. A cross-platform approach identifies genetic regulators of human metabolism and health. Nature genetics. 2021;53(1):54-64.
- Len AC, Powner MB, Zhu L, Hageman GS, Song X, Fruttiger M; et al. Pilot application of iTRAQ to the retinal disease Macular Telangiectasia. Journal of proteome research. 2012;11(2):537-53.
- Holeček, M. Serine Metabolism in Health and Disease and as a Conditionally Essential Amino Acid. Nutrients. 2022;14(9).
- Pan S, Fan M, Liu Z, Li X, Wang H. Serine, glycine and one-carbon metabolism in cancer (Review). International journal of oncology. 2021;58(2):158-70.
- Shunxi W, Xiaoxue Y, Guanbin S, Li Y, Junyu J, Wanqian L. Serine Metabolic Reprogramming in Tumorigenesis, Tumor Immunity, and Clinical Treatment. Advances in nutrition (Bethesda, Md). 2023;14(5):1050-66.
- Zhou X, Tian C, Cao Y, Zhao M, Wang K. The role of serine metabolism in lung cancer: From oncogenesis to tumor treatment. Frontiers in genetics. 2022;13:1084609.
- Brodzka S, Baszyński J, Rektor K, Hołderna-Bona K, Stanek E, Kurhaluk N; et al. The Role of Glutathione in Age-Related Macular Degeneration (AMD). International journal of molecular sciences. 2024;25(8).
- Cordes T, Kuna RS, McGregor GH, Khare SV, Gengatharan J, Muthusamy T; et al. 1-Deoxysphingolipid synthesis compromises anchorage-independent growth and plasma membrane endocytosis in cancer cells. Journal of lipid research. 2022;63(10):100281.
- Bejaoui K, Uchida Y, Yasuda S, Ho M, Nishijima M, Brown RH, Jr.; et al. Hereditary sensory neuropathy type 1 mutations confer dominant negative effects on serine palmitoyltransferase, critical for sphingolipid synthesis. The Journal of clinical investigation. 2002;110(9):1301-8.
- Rosarda JD, Giles S, Harkins-Perry S, Mills EA, Friedlander M, Wiseman RL; et al. Imbalanced unfolded protein response signaling contributes to 1-deoxysphingolipid retinal toxicity. Nature communications. 2023;14(1):4119.
- Handzlik MK, Metallo CM. Sources and Sinks of Serine in Nutrition, Health, and Disease. Annual review of nutrition. 2023;43:123-51.
- Bode H, Bourquin F, Suriyanarayanan S, Wei Y, Alecu I, Othman A; et al. HSAN1 mutations in serine palmitoyltransferase reveal a close structure-function-phenotype relationship. Human molecular genetics. 2016;25(5):853-65.
- Handzlik MK, Gengatharan JM, Frizzi KE, McGregor GH, Martino C, Rahman G; et al. Insulin-regulated serine and lipid metabolism drive peripheral neuropathy. Nature. 2023;614(7946):118-24.
- Holeček, M. Role of Impaired Glycolysis in Perturbations of Amino Acid Metabolism in Diabetes Mellitus. International journal of molecular sciences. 2023;24(2).


| Gass and Blodi (1993) [2] | Yannuzzi et al. (2006) [17] | Chew et al. (2023) [18] |
|---|---|---|
| Group 1 1A: Visible and exudative IJRT 1B: Visible, exudative, and focal IJRT |
||
| Group 2 2A: Occult and nonexudative IJRT Stage 1: Diffuse hyperfluorescence Stage 2: Reduced parafoveolar retinal Stage 2: transparency Stage 3: Dilated right-angled venules Stage 4: Intraretinal pigment clumping Stage 5: Vascular membranes |
Non-proliferative perifoveal telangiectasia Proliferative perifoveal telangiectasia |
Grade 0: No EZ Break/No Pigmentation/No OCT HR Grade 1: Noncentral EZ Break/No pigment/No OCT HR Grade 2: Central EZ break/No pigment/No OCT HR Grade 3: Noncentral pigment/No, non-central, or central EZ/No Grade 3: OCT HR Grade 4: OCT HR/EZ break (either central or noncentral)/No Grade 4: pigment Grade 5: Central pigment/no exudative neovascularization/EZ Grade 5: present or not gradable Grade 6: Neovascularization (exudative) ± central pigment |
| 2B: Juvenile occult familial IJRT | ||
| Group 3 3A: Occlusive IJRT 3B: Occlusive IJRT with CNS vasculopathy |
| Affected individuals (age (years)) | Associated primary condition | Reference |
|---|---|---|
| Sisters (461 and 562) | 1Blurring of vision; 2Slightly distorted vision after a car accident | [34] |
| Several family members | / | [35] |
| Brothers (651 and /) | 1T2DM, mild nonproliferative DR, systemic hypertension, asteroid hyalosis, pigment epithelial hyperplasia, and mild macular edema | [6] |
| 3/92 patients were siblings the disease is present in 2/89 families |
14/92 patients had hypertension, other accompanying diseases were polycythemia vera, coronary artery disease, and borderline diabetes in one case each, two had coronary artery disease, and one had renal insufficiency associated with Alport’s disease | [2] |
| Sisters (491 and 482) | 1Reduced vision; 2Blurring of distance vision | [36] |
| Daughter (291) of the affected father (582) | 1T2DM, decreased vision; 2Macular edema | [37] |
| Monozygotic twins, sisters (641,2) | 1,2Vision loss | [39] |
| Monozygotic twins, sisters (681/2) | 1Bilateral metamorphopsia; 2T2DM, amblyopia | [40] |
| Monozygotic twins, sisters (631/2) | 1Metamorphopsia, subretinal NV; 2Metamorphopsia | [41] |
| Daughters (41 and 45) of affected mother (681) Brother (61) of the affected sister (742) Monozygotic twins, sisters (563) Monozygotic twins, brothers (564,5) |
1Blurring of vision; 2Blurring of vision, T2DM; 3Bilateral blurred vision, phototherapeutic keratectomy; 4Decrease of vision, developed T2DM; 5T2DM | [11] |
| Son (451) of the affected mother (792) Son (623) of the affected mother (764) Sister (655) and brother (786) |
1Reading difficulties. metamorphopsia, T2DM, mild DR, underwent coronary bypass at age of 40; 2Loss of vision, maculopathy of unknown origin, T2DM, DR; 3Visual loss, obesity, arterial hypertension, phlebothrombotic event at age of 57, hyperhomocysteinemia, antiphospholipid syndrome, T2DM; 4T2DM, reading difficulties, maculopathy in the absence of DR; 5Metamorphopsia, T2DM; 6T2DM | [38] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





