Submitted:
25 November 2024
Posted:
26 November 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Mechanical Mixing Assisted Extraction (MMAE) of ß-Carotene
2.3. Homogenization Assisted Extraction (HAE) of ß-Carotene
2.4. Analysis of samples
2.5. Statistical Design of Experiments
3. Results and Discussion
3.1. Preparation of Deep Eutectic Solvents (DESs)
3.2. Baseline (Calibration Line) Study for ß-Carotene in Deep Eutectic Solvents (DESs)
3.3. Mechanical Mixing Assisted Extraction (MMAE) and Homogenization Assisted Extraction (HAE) of ß-Carotene
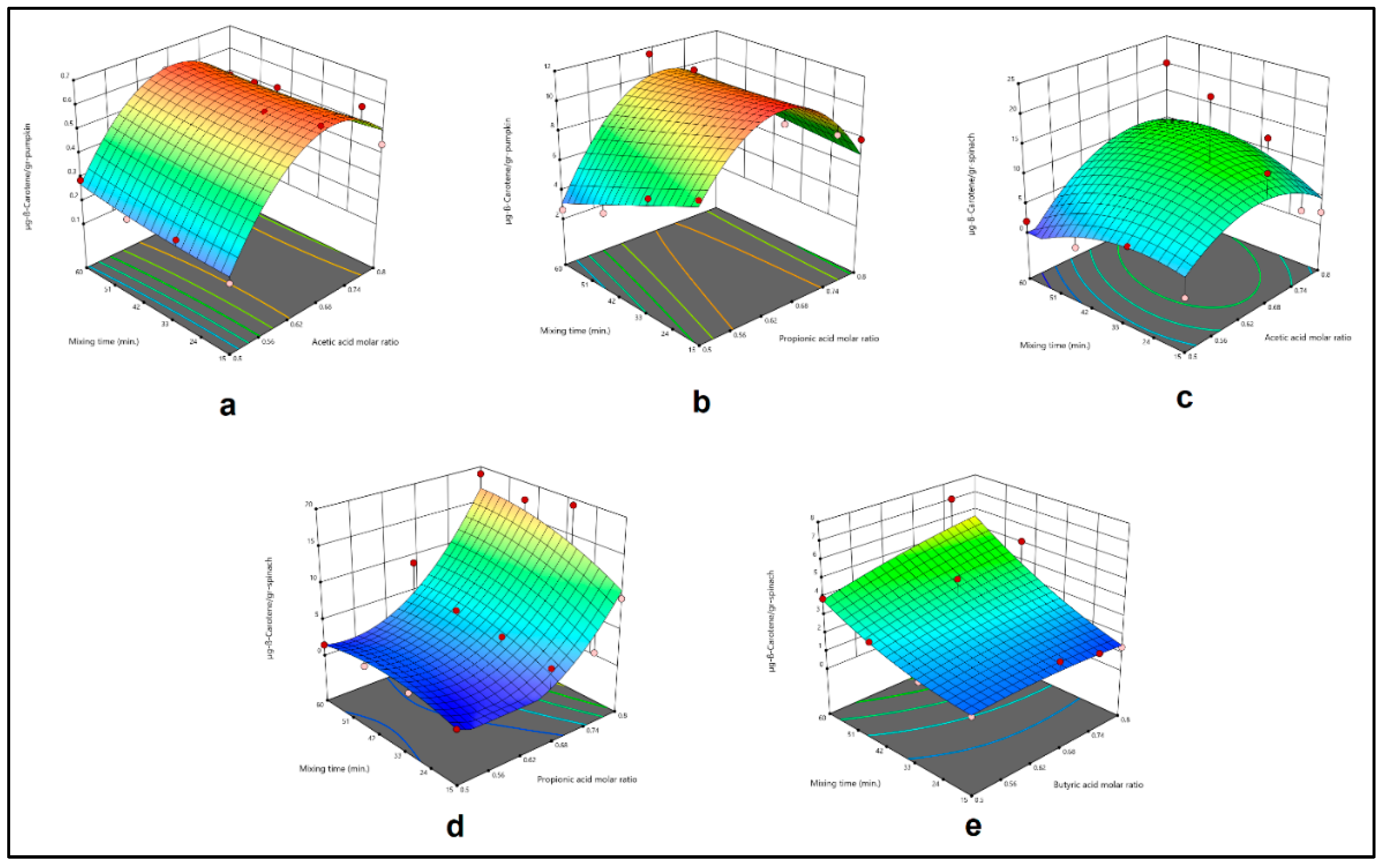
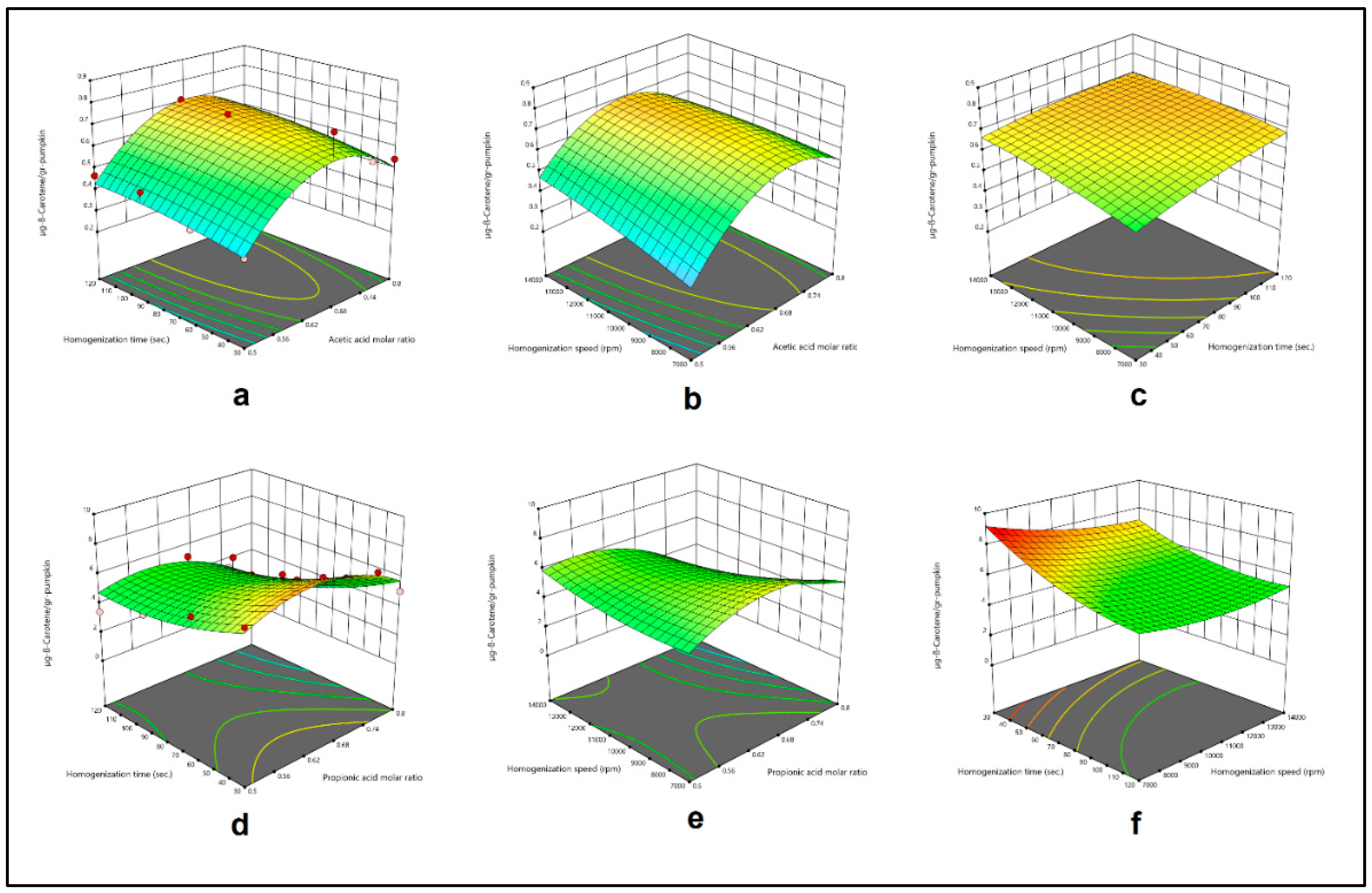
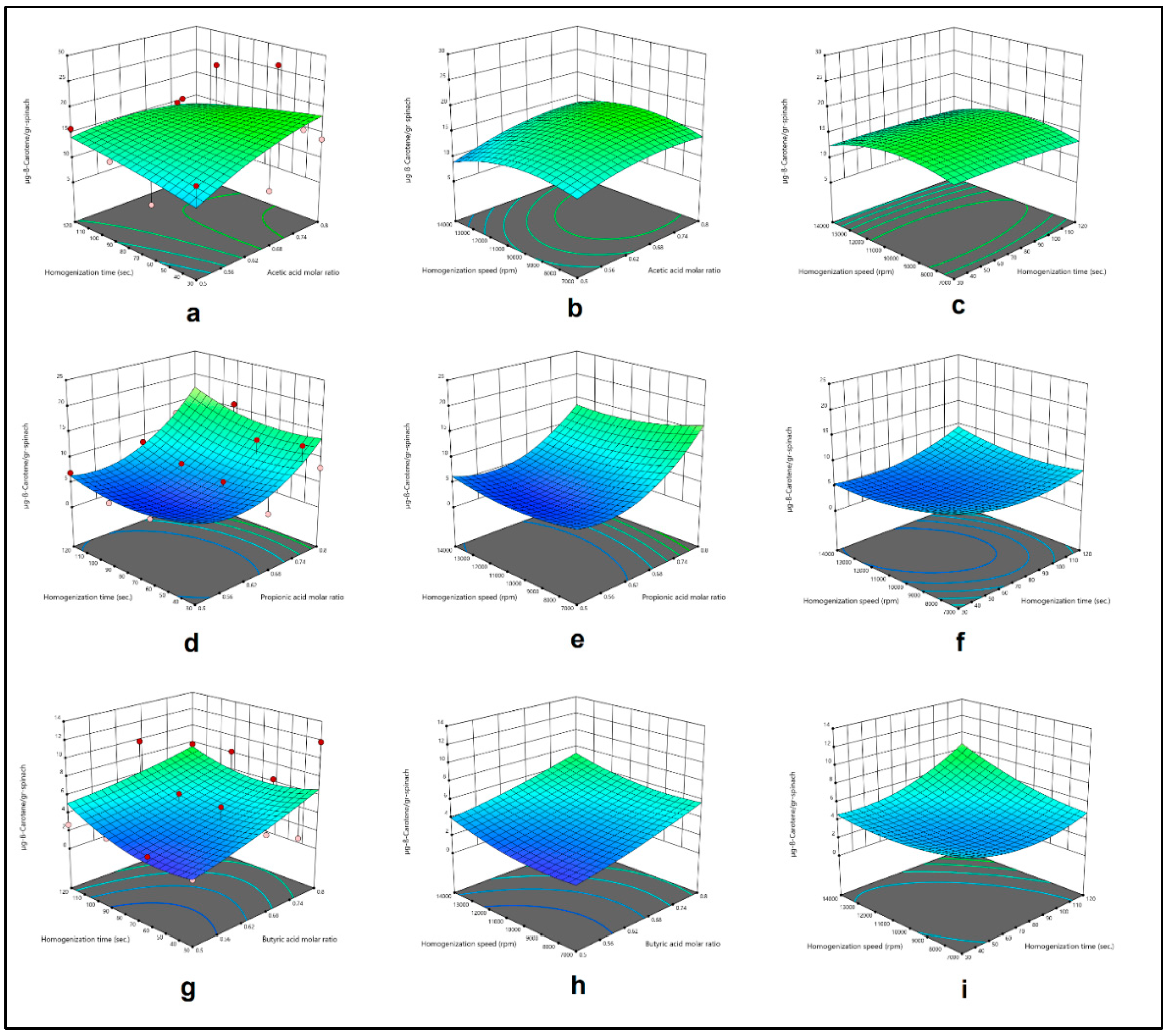
3.4. Modelling and Optimization Studies
3.5. Validation Study and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Şanal, İ.S.; Güvenç, A.; Salgın, U.; Mehmetoğlu, Ü.; Çalımlı, A. Recycling of apricot pomace by supercritical CO2 extraction. J. Supercrit. Fluids 2004, 32, 221–230. [Google Scholar] [CrossRef]
- Vega, P.J.; Balaban, M.O.; Sims, C.A.; O’Keefe, S.F.; Cornell, J.A. Supercritical carbon dioxide extraction efficiency for carotenes from carrots by RSM. J. Food Sci. 1996, 61, 757–759. [Google Scholar] [CrossRef]
- Ozturk, B.; Parkinson, C.; Gonzalez-Miquel, M. Extraction of polyphenolic antioxidants from orange peel waste using deep eutectic solvents. Sep. Purif. Technol. 2018, 206, 1–13. [Google Scholar] [CrossRef]
- α-Carotene, β-Carotene, β-Cryptoxanthin, Lycopene, Lutein, and Zeaxanthin. Available online: https://lpi.oregonstate.edu/mic/dietary-factors/phytochemicals/carotenoids#introduction (accessed on 11 November 2024).
- Lee, S.C.; Ristaino, J.B.; Heitman, J. Parallels in intercellular communication in oomycete and fungal pathogens of plants and humans. PLOS Pathog. 2012, 8, e1003028. [Google Scholar] [CrossRef]
- The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Eng. J. Med 1994, 330, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Slattery, M.L.; Beson, J.; Curtin, K.; Ma, K.-N.; Schaffer, D.; Potter, J.D. Carotenoids and colorectal cancer. Am. J. Clin. Nutr. 2000, 71, 575–582. [Google Scholar] [CrossRef]
- Kirsh, V.A.; Hayes, R.B.; Mayne, S.T.; Chatterjee, N.; Subar, A.F.; Dixon, L.B.; Albanes, D.; Andriole, G.L.; Urban, D.A.; Peters, U. Supplemental and dietary vitamin E, ß-Carotene, and vitamin C intakes and prostate cancer risk. J. Natl. Cancer Inst. 2006, 98, 245. [Google Scholar] [CrossRef]
- Stahl, W.; Krutmann, J. Systemische photoprotektion druch karotinoide. Hautarzt 2006, 57, 281–285. [Google Scholar] [CrossRef]
- Blot, W.J.; Li, J.-Y.; Taylor, P.R.; Guo, W.; Dawsey, S.; Wang, G.-Q.; Yang, C.S.; Zheng, S.-F.; Gail, M.; Li, G.-Y.; et al. Nutrition intervention trials in Linxian, China: Supplementation with specific vitamin/mineral combinations, cancer incidence, and desease-specific mortality in the general population. J. Natl. Cancer Inst. 1993, 85, 1483–1993. [Google Scholar] [CrossRef]
- Tapiero, H.; Townsend, D.M.; Tew, K.D. The role of carotenoids in the prevention of human pathologies. Biomed. Pharmacother. 2004, 58, 100–110. [Google Scholar] [CrossRef]
- Craft, N.E.; Soares, J.H., Jr. Relative Solubility, Stability, and absorptivity of lutein and β-Carotene in organic solvents. J. Agric. Food. Chem. 1992, 40, 431–434. [Google Scholar] [CrossRef]
- Huff, J. Benzene-induced cancers: Abridged history and occupational health impact. Int. J. Occup. Environ. Health. 2007, 13, 213–221. [Google Scholar] [CrossRef] [PubMed]
- ICH guideline Q3C (R9) on impurities: Guideline for residual solvents (EMA/CHMP/ICH/82260/2006). Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/ich-q3c-r9-guideline-impurities-guideline-residual-solvents-step-5_en.pdf, (accessed on 11th November 2024).
- United Nations Environment Programme. Food Waste Index Report 2024. Think Eat Save: Tracking Progress to Halve Global Food Waste; Knowledge Repository - UNEP. UNEP.: Nairobi, Kenya, 2024; p. 12. [Google Scholar]
- Oh my gourd! 1.3B pounds of pumpkins reach landfills every year. Available online: https://www.wastedive.com/news/oh-my-gourd-13b-pounds-of-pumpkins-reach-landfills-every-year/408341/, (accessed on 8th November 2024).
- Lin, H.; Black, M.J.; Walsh, L.; Giordano, F.S.; Borrion, A. Life cycle assessment of baby leaf spinach: Reduction of waste through interventions in growing treatments and packaging. J. Clean. Prod. 2024, 449, 141723. [Google Scholar] [CrossRef]
- Frankowska, A.; Jeswani, H.K.; Azapagic, A. Environmental impacts of vegetables consumption in the UK, Science of the Total Environment. Sci. Total Environ. 2019, 682, 80–105. [Google Scholar] [CrossRef]
- Sebdani, M.M.; Abbasi, H. Green extraction of carotenoids from pumpkin with ultrasound-assisted method; optimization using response surface methodology. Microchem. J. 2023, 193, 109092. [Google Scholar] [CrossRef]
- Stupar, A.; Šeregelj, V.; Ribeiro, B.D.; Pezo, L.; Cvetanović, A.; Mišan, A.; Marrucho, I. Recovery of β-carotene from pumpkin using switchable natural deep eutectic solvents. Ultrason. Sonochem. 2021, 76, 105638. [Google Scholar] [CrossRef]
- Shi, X.; Wu, H.; Shi, J.; Xue, S.J.; Wang, D.; Wang, W.; Cheng, A.; Gong, Z.; Chen, X.; Wang, C. Effect of modifier on the composition and antioxidant activity of carotenoid extracts from pumpkin (Cucurbita maxima) by supercritical CO2. LWT 2013, 51, 433–440. [Google Scholar] [CrossRef]
- The United States Pharmacopoeia Beta Carotene monograph. Available online: https://www.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/beta_carotene.pdf (accessed on 12th November 2024).
- Moradi, M.; Fazlzadehdavil, M.; Pirsaheb, M.; Mansouri, Y.; Khosravi, T.; Sharafi, K. Response surface methodology (RSM) and its application for optimization of ammonium ions removal from aqueous solutions by pumice as a natural and low cost adsorbent. Arch. Environ. Prot. 2016, 42, 33–43. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef]
- Bhat, B.; Vaid, S.; Habib, B.; Bajaj, B.K. Design of experiments for enhanced production of bioactive exopolysaccharides from indigenous probiotic lactic acid bacteria. Indian J. Biochem. Biophys. 2020, 57, 539–551. [Google Scholar]
- Florindo, C.; McIntosh, A.J.S.; Welton, T.; Branco, L.C.; Marrucho, I.M. A closer look into deep eutectic solvents: Exploring intermolecular interactions using solvatochromic probes. Phys. Chem. Chem. Phys. 2018, 20, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Acetic acid (glacial) 100% SDS. Available online: https://www.merckmillipore.com/DE/en/product/msds/MDA_CHEM-100056 (accessed on 13th November 2024).
- Propionic acid for synthesis SDS. Available online: https://www.merckmillipore.com/DE/en/product/msds/MDA_CHEM-800605?Origin=PDP (accessed on 13th November 2024).
- Kwan, Y.H.; Tung, Y.K.; Kochhar, J.S.; Li, H.; Poh, A.L.; Kang, L. 6-Esstial Monographs. In Handbook of Cosmeceutical Excipients and their Safeties, 1st ed.; Woodhead Publishing: Kidlington, UK, 2014; p. 105. [Google Scholar]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2023, 70–71. [Google Scholar]
| Hydrogen bond acceptor (HBA) | Hydrogen bond donor (HBD) | Molar Ratio (HBA:HBD) |
Abbreviations |
| L-Menthol | Acetic acid | 1:1 | M1ACA1 |
| L-Menthol | Acetic acid | 1:2 | M1ACA2 |
| L-Menthol | Acetic acid | 1:3 | M1ACA3 |
| L-Menthol | Acetic acid | 1:4 | M1ACA4 |
| L-Menthol | Propionic acid | 1:1 | M1PRA1 |
| L-Menthol | Propionic acid | 1:2 | M1PRA2 |
| L-Menthol | Propionic acid | 1:3 | M1PRA3 |
| L-Menthol | Propionic acid | 1:4 | M1PRA4 |
| L-Menthol | Butyric acid | 1:1 | M1BTA1 |
| L-Menthol | Butyric acid | 1:2 | M1BTA2 |
| L-Menthol | Butyric acid | 1:3 | M1BTA3 |
| L-Menthol | Butyric acid | 1:4 | M1BTA4 |
| DES | Mixing Time (min.) | μg-β-Carotene /gr-Pumpkin 1 |
μg-β-Carotene /gr-Spinach 1 |
| M1ACA1 | 15 | 0.196 ± 0.043 | 0.714 ± 0.025 |
| M1ACA1 | 30 | 0.254 ± 0.011 | 5.093 ± 0.032 |
| M1ACA1 | 45 | 0.229 ± 0.012 | 1.083 ± 0.016 |
| M1ACA1 | 60 | 0.290 ± 0.026 | 1.959 ± 0.031 |
| M1ACA2 | 15 | 0.637 ± 0.011 | 13.867 ± 0.069 |
| M1ACA2 | 30 | 0.603 ± 0.009 | 23.238 ± 0.075 |
| M1ACA2 | 45 | 0.566 ± 0.006 | 9.348 ± 0.108 |
| M1ACA2 | 60 | 0.610 ± 0.004 | 6.186 ± 0.026 |
| M1ACA3 | 15 | 0.639 ± 0.020 | 4.447 ± 0.029 |
| M1ACA3 | 30 | 0.558 ± 0.012 | 10.994 ± 0.029 |
| M1ACA3 | 45 | 0.572 ± 0.010 | 6.863 ± 0.040 |
| M1ACA3 | 60 | 0.561 ± 0.003 | 5.058 ± 0.028 |
| M1ACA4 | 15 | 0.442 ± 0.008 | 2.202 ± 0.039 |
| M1ACA4 | 30 | 0.483 ± 0.010 | 12.089 ± 0.035 |
| M1ACA4 | 45 | 0.510 ± 0.007 | 7.820 ± 0.069 |
| M1ACA4 | 60 | 0.486 ± 0.009 | 20.168 ± 0.044 |
| M1PRA1 | 15 | 7.873 ± 0.023 | 0.866 ± 0.019 |
| M1PRA1 | 30 | 6.452 ± 0.040 | 1.803 ± 0.009 |
| M1PRA1 | 45 | 3.960 ± 0.019 | 1.869 ± 0.009 |
| M1PRA1 | 60 | 2.631 ± 0.014 | 1.476 ± 0.005 |
| M1PRA2 | 15 | 10.816 ± 0.044 | 3.567 ± 0.020 |
| M1PRA2 | 30 | 10.585 ± 0.045 | 4.508 ± 0.026 |
| M1PRA2 | 45 | 11.542 ± 0.034 | 5.044 ± 0.027 |
| M1PRA2 | 60 | 11.566 ± 0.044 | 8.986 ± 0.033 |
| M1PRA3 | 15 | 8.796 ± 0.051 | 3.256 ± 0.011 |
| M1PRA3 | 30 | 8.234 ± 0.042 | 5.889 ± 0.019 |
| M1PRA3 | 45 | 9.264 ± 0.023 | 6.944 ± 0.017 |
| M1PRA3 | 60 | 8.939 ± 0.072 | 5.915 ± 0.023 |
| M1PRA4 | 15 | 7.917 ± 0.041 | 9.312 ± 0.049 |
| M1PRA4 | 30 | 8.063 ± 0.031 | 19.156 ± 0.122 |
| M1PRA4 | 45 | 7.757 ± 0.047 | 17.580 ± 0.043 |
| M1PRA4 | 60 | 7.465 ± 0.043 | 18.990 ± 0.057 |
| M1BTA1 | 15 | 1.552 ± 0.045 | |
| M1BTA1 | 30 | 1.944 ± 0.039 | |
| M1BTA1 | 45 | 2.774 ± 0.031 | |
| M1BTA1 | 60 | 3.925 ± 0.024 | |
| M1BTA2 | 15 | 2.193 ± 0.040 | |
| M1BTA2 | 30 | 1.718 ± 0.044 | |
| M1BTA2 | 45 | 4.321 ± 0.036 | |
| M1BTA2 | 60 | 4.142 ± 0.029 | |
| M1BTA3 | 15 | 1.593 ± 0.032 | |
| M1BTA3 | 30 | 2.280 ± 0.026 | |
| M1BTA3 | 45 | 2.720 ± 0.021 | |
| M1BTA3 | 60 | 7.036 ± 0.032 | |
| M1BTA4 | 15 | 1.319 ± 0.024 | |
| M1BTA4 | 30 | 2.226 ± 0.028 | |
| M1BTA4 | 45 | 5.109 ± 0.029 | |
| M1BTA4 | 60 | 4.468 ± 0.017 |
| DES | Homogenization Time (sec.) | Homogenization Speed (rpm) | μg-β-Carotene/gr-Pumpkin 1 | μg-β-Carotene/gr-Spinach 1 |
| M1ACA1 | 30 | 7000 | 0.287 ± 0.002 | 8.830 ± 0.027 |
| M1ACA1 | 60 | 7000 | 0.295 ± 0.002 | 13.459 ± 0.084 |
| M1ACA1 | 90 | 7000 | 0.241 ± 0.005 | 12.948 ± 0.056 |
| M1ACA1 | 120 | 7000 | 0.291 ± 0.003 | 11.703 ± 0.012 |
| M1ACA1 | 30 | 10500 | 0.359 ± 0.003 | 14.116 ± 0.054 |
| M1ACA1 | 60 | 10500 | 0.390 ± 0.002 | 7.225 ± 0.021 |
| M1ACA1 | 90 | 10500 | 0.471 ± 0.005 | 12.260 ± 0.047 |
| M1ACA1 | 120 | 10500 | 0.467 ± 0.004 | 15.822 ± 0.044 |
| M1ACA1 | 30 | 14000 | 0.464 ± 0.002 | 7.731 ± 0.078 |
| M1ACA1 | 60 | 14000 | 0.449 ± 0.002 | 7.511 ± 0.070 |
| M1ACA1 | 90 | 14000 | 0.441 ± 0.003 | 7.239 ± 0.020 |
| M1ACA1 | 120 | 14000 | 0.506 ± 0.002 | 9.139 ± 0.106 |
| M1ACA2 | 30 | 7000 | 0.553 ± 0.039 | 19.093 ± 0.074 |
| M1ACA2 | 60 | 7000 | 0.666 ± 0.019 | 13.158 ± 0.067 |
| M1ACA2 | 90 | 7000 | 0.637 ± 0.028 | 9.728 ± 0.056 |
| M1ACA2 | 120 | 7000 | 0.811 ± 0.032 | 12.469 ± 0.056 |
| M1ACA2 | 30 | 10500 | 0.762 ± 0.044 | 7.622 ± 0.014 |
| M1ACA2 | 60 | 10500 | 0.652 ± 0.041 | 15.598 ± 0.053 |
| M1ACA2 | 90 | 10500 | 0.707 ± 0.008 | 20.129 ± 0.041 |
| M1ACA2 | 120 | 10500 | 0.711 ± 0.002 | 14.714 ± 0.029 |
| M1ACA2 | 30 | 14000 | 0.562 ± 0.038 | 18.474 ± 0.017 |
| M1ACA2 | 60 | 14000 | 0.632 ± 0.020 | 9.334 ± 0.016 |
| M1ACA2 | 90 | 14000 | 0.771 ± 0.006 | 11.925 ± 0.081 |
| M1ACA2 | 120 | 14000 | 0.670 ± 0.006 | 17.460 ± 0.107 |
| M1ACA3 | 30 | 7000 | 0.434 ± 0.003 | 6.767 ± 0.027 |
| M1ACA3 | 60 | 7000 | 0.639 ± 0.005 | 8.553 ± 0.031 |
| M1ACA3 | 90 | 7000 | 0.651 ± 0.054 | 16.257 ± 0.048 |
| M1ACA3 | 120 | 7000 | 0.640 ± 0.000 | 15.457 ± 0.098 |
| M1ACA3 | 30 | 10500 | 0.571 ± 0.002 | 16.982 ± 0.024 |
| M1ACA3 | 60 | 10500 | 0.563 ± 0.010 | 15.152 ± 0.015 |
| M1ACA3 | 90 | 10500 | 0.645 ± 0.003 | 24.930 ± 0.057 |
| M1ACA3 | 120 | 10500 | 0.588 ± 0.002 | 14.775 ± 0.042 |
| M1ACA3 | 30 | 14000 | 0.606 ± 0.018 | 13.767 ± 0.053 |
| M1ACA3 | 60 | 14000 | 0.611 ± 0.002 | 11.293 ± 0.028 |
| M1ACA3 | 90 | 14000 | 0.689 ± 0.000 | 15.814 ± 0.039 |
| M1ACA3 | 120 | 14000 | 0.644 ± 0.000 | 13.564 ± 0.027 |
| M1ACA4 | 30 | 7000 | 0.449 ± 0.012 | 22.831 ± 0.056 |
| M1ACA4 | 60 | 7000 | 0.548 ± 0.020 | 20.749 ± 0.062 |
| M1ACA4 | 90 | 7000 | 0.629 ± 0.029 | 12.540 ± 0.014 |
| M1ACA4 | 120 | 7000 | 0.579 ± 0.005 | 7.834 ± 0.041 |
| M1ACA4 | 30 | 10500 | 0.545 ± 0.004 | 13.751 ± 0.063 |
| M1ACA4 | 60 | 10500 | 0.534 ± 0.008 | 26.006 ± 0.092 |
| M1ACA4 | 90 | 10500 | 0.572 ± 0.005 | 11.626 ± 0.014 |
| M1ACA4 | 120 | 10500 | 0.554 ± 0.011 | 9.568 ± 0.038 |
| M1ACA4 | 30 | 14000 | 0.581 ± 0.003 | 18.883 ± 0.055 |
| M1ACA4 | 60 | 14000 | 0.525 ± 0.017 | 14.477 ± 0.080 |
| M1ACA4 | 90 | 14000 | 0.581 ± 0.050 | 7.578 ± 0.053 |
| M1ACA4 | 120 | 14000 | 0.543 ± 0.006 | 7.993 ± 0.069 |
| M1PRA1 | 30 | 7000 | 7.675 ± 0.030 | 6.081 ± 0.039 |
| M1PRA1 | 60 | 7000 | 5.030 ± 0.023 | 7.450 ± 0.020 |
| M1PRA1 | 90 | 7000 | 3.769 ± 0.013 | 5.234 ± 0.014 |
| M1PRA1 | 120 | 7000 | 4.698 ± 0.023 | 8.447 ± 0.034 |
| M1PRA1 | 30 | 10500 | 6.896 ± 0.013 | 11.252 ± 0.027 |
| M1PRA1 | 60 | 10500 | 6.088 ± 0.021 | 4.241 ± 0.019 |
| M1PRA1 | 90 | 10500 | 4.712 ± 0.019 | 4.010 ± 0.019 |
| M1PRA1 | 120 | 10500 | 3.456 ± 0.014 | 6.955 ± 0.013 |
| M1PRA1 | 30 | 14000 | 6.120 ± 0.024 | 8.539 ± 0.020 |
| M1PRA1 | 60 | 14000 | 7.147 ± 0.013 | 3.704 ± 0.020 |
| M1PRA1 | 90 | 14000 | 6.487 ± 0.023 | 6.080 ± 0.012 |
| M1PRA1 | 120 | 14000 | 5.340 ± 0.020 | 8.139 ± 0.028 |
| M1PRA2 | 30 | 7000 | 8.762 ± 0.029 | 4.731 ± 0.082 |
| M1PRA2 | 60 | 7000 | 6.586 ± 0.037 | 5.323 ± 0.013 |
| M1PRA2 | 90 | 7000 | 8.579 ± 0.043 | 6.669 ± 0.021 |
| M1PRA2 | 120 | 7000 | 6.472 ± 0.039 | 11.232 ± 0.013 |
| M1PRA2 | 30 | 10500 | 7.316 ± 0.045 | 2.958 ± 0.033 |
| M1PRA2 | 60 | 10500 | 6.561 ± 0.038 | 6.132 ± 0.011 |
| M1PRA2 | 90 | 10500 | 6.411 ± 0.043 | 6.950 ± 0.058 |
| M1PRA2 | 120 | 10500 | 5.201 ± 0.032 | 8.706 ± 0.013 |
| M1PRA2 | 30 | 14000 | 6.935 ± 0.040 | 8.774 ± 0.032 |
| M1PRA2 | 60 | 14000 | 5.880 ± 0.031 | 10.494 ± 0.053 |
| M1PRA2 | 90 | 14000 | 4.935 ± 0.039 | 4.587 ± 0.048 |
| M1PRA2 | 120 | 14000 | 6.839 ± 0.017 | 7.195 ± 0.035 |
| M1PRA3 | 30 | 7000 | 8.596 ± 0.021 | 14.763 ± 0.034 |
| M1PRA3 | 60 | 7000 | 5.541 ± 0.045 | 10.024 ± 0.044 |
| M1PRA3 | 90 | 7000 | 3.802 ± 0.043 | 17.005 ± 0.031 |
| M1PRA3 | 120 | 7000 | 4.888 ± 0.039 | 14.631 ± 0.048 |
| M1PRA3 | 30 | 10500 | 6.981 ± 0.041 | 13.676 ± 0.052 |
| M1PRA3 | 60 | 10500 | 5.321 ± 0.039 | 12.177 ± 0.034 |
| M1PRA3 | 90 | 10500 | 1.803 ± 0.033 | 9.143 ± 0.035 |
| M1PRA3 | 120 | 10500 | 3.367 ± 0.039 | 12.853 ± 0.035 |
| M1PRA3 | 30 | 14000 | 5.253 ± 0.041 | 10.178 ± 0.065 |
| M1PRA3 | 60 | 14000 | 2.010 ± 0.032 | 5.838 ± 0.052 |
| M1PRA3 | 90 | 14000 | 0.631 ± 0.038 | 11.197 ± 0.045 |
| M1PRA3 | 120 | 14000 | 3.596 ± 0.043 | 13.982 ± 0.049 |
| M1PRA4 | 30 | 7000 | 8.898 ± 0.031 | 24.598 ± 0.126 |
| M1PRA4 | 60 | 7000 | 6.158 ± 0.027 | 19.838 ± 0.113 |
| M1PRA4 | 90 | 7000 | 7.714 ± 0.031 | 15.597 ± 0.069 |
| M1PRA4 | 120 | 7000 | 1.773 ± 0.045 | 12.177 ± 0.118 |
| M1PRA4 | 30 | 10500 | 5.064 ± 0.028 | 7.972 ± 0.091 |
| M1PRA4 | 60 | 10500 | 4.638 ± 0.037 | 10.282 ± 0.065 |
| M1PRA4 | 90 | 10500 | 3.145 ± 0.030 | 15.900 ± 0.056 |
| M1PRA4 | 120 | 10500 | 2.184 ± 0.025 | 16.399 ± 0.057 |
| M1PRA4 | 30 | 14000 | 5.653 ± 0.026 | 8.387 ± 0.078 |
| M1PRA4 | 60 | 14000 | 3.880 ± 0.034 | 14.157 ± 0.115 |
| M1PRA4 | 90 | 14000 | 2.029 ± 0.040 | 13.669 ± 0.100 |
| M1PRA4 | 120 | 14000 | 1.318 ± 0.025 | 24.910 ± 0.071 |
| M1BTA1 | 30 | 7000 | 2.563 ± 0.033 | |
| M1BTA1 | 60 | 7000 | 3.240 ± 0.032 | |
| M1BTA1 | 90 | 7000 | 3.129 ± 0.033 | |
| M1BTA1 | 120 | 7000 | 4.082 ± 0.035 | |
| M1BTA1 | 30 | 10500 | 2.244 ± 0.023 | |
| M1BTA1 | 60 | 10500 | 2.768 ± 0.032 | |
| M1BTA1 | 90 | 10500 | 2.889 ± 0.039 | |
| M1BTA1 | 120 | 10500 | 2.701 ± 0.021 | |
| M1BTA1 | 30 | 14000 | 2.979 ± 0.022 | |
| M1BTA1 | 60 | 14000 | 4.285 ± 0.029 | |
| M1BTA1 | 90 | 14000 | 3.862 ± 0.050 | |
| M1BTA1 | 120 | 14000 | 6.460 ± 0.038 | |
| M1BTA2 | 30 | 7000 | 4.171 ± 0.046 | |
| M1BTA2 | 60 | 7000 | 4.609 ± 0.038 | |
| M1BTA2 | 90 | 7000 | 4.188 ± 0.027 | |
| M1BTA2 | 120 | 7000 | 6.461 ± 0.026 | |
| M1BTA2 | 30 | 10500 | 3.844 ± 0.042 | |
| M1BTA2 | 60 | 10500 | 5.263 ± 0.042 | |
| M1BTA2 | 90 | 10500 | 5.191 ± 0.031 | |
| M1BTA2 | 120 | 10500 | 9.820 ± 0.077 | |
| M1BTA2 | 30 | 14000 | 5.457 ± 0.045 | |
| M1BTA2 | 60 | 14000 | 7.097 ± 0.050 | |
| M1BTA2 | 90 | 14000 | 6.782 ± 0.034 | |
| M1BTA2 | 120 | 14000 | 13.221 ± 0.044 | |
| M1BTA3 | 30 | 7000 | 5.708 ± 0.045 | |
| M1BTA3 | 60 | 7000 | 4.283 ± 0.020 | |
| M1BTA3 | 90 | 7000 | 3.736 ± 0.032 | |
| M1BTA3 | 120 | 7000 | 3.884 ± 0.020 | |
| M1BTA3 | 30 | 10500 | 2.020 ± 0.038 | |
| M1BTA3 | 60 | 10500 | 2.917 ± 0.021 | |
| M1BTA3 | 90 | 10500 | 3.239 ± 0.030 | |
| M1BTA3 | 120 | 10500 | 3.332 ± 0.027 | |
| M1BTA3 | 30 | 14000 | 5.543 ± 0.037 | |
| M1BTA3 | 60 | 14000 | 4.595 ± 0.029 | |
| M1BTA3 | 90 | 14000 | 2.968 ± 0.033 | |
| M1BTA3 | 120 | 14000 | 8.684 ± 0.027 | |
| M1BTA4 | 30 | 7000 | 8.584 ± 0.028 | |
| M1BTA4 | 60 | 7000 | 6.231 ± 0.037 | |
| M1BTA4 | 90 | 7000 | 9.738 ± 0.049 | |
| M1BTA4 | 120 | 7000 | 4.637 ± 0.037 | |
| M1BTA4 | 30 | 10500 | 11.840 ± 0.043 | |
| M1BTA4 | 60 | 10500 | 6.343 ± 0.024 | |
| M1BTA4 | 90 | 10500 | 8.197 ± 0.039 | |
| M1BTA4 | 120 | 10500 | 7.814 ± 0.046 | |
| M1BTA4 | 30 | 14000 | 8.211 ± 0.025 | |
| M1BTA4 | 60 | 14000 | 5.023 ± 0.033 | |
| M1BTA4 | 90 | 14000 | 7.401 ± 0.046 | |
| M1BTA4 | 120 | 14000 | 12.534 ± 0.023 |
| Extraction setup | Independent Variables | Dependent Variable | Model Equation | R2 |
| Sample: Pumkin Method: MMAE & Menthol:Acetic acid DESs |
X1:Acetic acid molar ratio X2:Mixing time (min.) |
μg-β-Carotene/ gr-Pumpkin |
-4.45150+14.57986X1+0.001912X2-0.005262X1X2 -10.44080X12+0.000024X22 |
0.9642 |
| Sample: Pumkin Method: MMAE & Menthol:Propionic acid DESs |
X1:Propionic acid molar ratio X2:Mixing time (min.) |
μg-β-Carotene/ gr-Pumpkin |
-63.76949+241.35123X1-0.301289X2+0.401802X1X2 -191.69872X12+0.000041X22 |
0.9006 |
| Sample: Spinach Method: MMAE & Menthol:Acetic acid DESs |
X1:Acetic acid molar ratio X2:Mixing time (min.) |
μg-β-Carotene/ gr-Spinach |
-87.29436+291.71772X1-0.080113X2+0.815406X1X2 -232.20477X12-0.006091X22 |
0.3941 |
| Sample: Spinach Method: MMAE & Menthol:Propionic acid DESs |
X1:Propionic acid molar ratio X2:Mixing time (min.) |
μg-β-Carotene/ gr-Spinach |
81.41602-286.71718X1+0.011693X2 +0.437977X1X2+239.97280 X12-0.002896X22 |
0.7835 |
| Sample: Spinach Method: MMAE & Menthol:Butyric acid DESs |
X1:Butyric acid molar ratio X2:Mixing time (min.) |
μg-β-Carotene/ gr-Spinach |
0.019209+7.15429X1-0.089691X2+0.147515X1X2 -7.71617X12+0.000871X22 |
0.7688 |
| Sample: Pumkin Method: HAE & Menthol:Acetic acid DESs |
X1:Acetic acid molar ratio X2:Homogenization time (sec.) X3:Homogenization speed (rpm) |
μg-β-Carotene/ gr-Pumpkin |
-4.30138+12.54252X1+0.003089X2+0.000103X3 +0.000543X1X2-0.000081X1X3-1.29643*10-7X2X3 -8.63510X12-8.37963*10-6X22-1.42092*10-9X32 |
0.8418 |
| Sample: Pumkin Method: HAE & Menthol:Propionic acid DESs |
X1:Propionic acid molar ratio X2:Homogenization time (sec.) X3:Homogenization speed (rpm) |
μg-β-Carotene/ gr-Pumpkin |
-24.31624+109.68892X1-0.063963X2+0.000120X3 -0.068021X1X2-0.001957X1X3+2.65619*10-6X2X3 -69.07260X12+0.000334X22+3.75153*10-8X32 |
0.7052 |
| Sample: Spinach Method: HAE & Menthol:Acetic acid DESs |
X1:Acetic acid molar ratio X2:Homogenization time (sec.) X3:Homogenization speed (rpm) |
μg-β-Carotene/ gr-Spinach |
-46.56104+104.57014X1+0.229675X2+0.003205X3 -0.333622X1X2+0.000977X1X3+2.93333*10-7X2X3 -59.45170X12-0.000142X22-1.93862*10-7X32 |
0.2725 |
| Sample: Spinach Method: HAE & Menthol:Propionic acid DESs |
X1:Propionic acid molar ratio X2:Homogenization time (sec.) X3:Homogenization speed (rpm) |
μg-β-Carotene/ gr-Spinach |
88.81933-218.95677X1-0.304985X2-0.002093X3 +0.146208X1X2-0.001599X1X3+7.80619*10-6X2X3 +195.82812X12+0.000968X22+1.13311*10-7X32 |
0.6540 |
| Sample: Spinach Method: HAE & Menthol:Butyric acid DESs |
X1:Butyric acid molar ratio X2:Homogenization time (sec.) X3:Homogenization speed (rpm) |
μg-β-Carotene/ gr-Spinach |
9.20900+1.75614X1-0.106600X2-0.001464X3 -0.073053X1X2-0.000142X1X3+7.09500*10-6X2X3 +12.70142X12+0.000667X22+5.99541*10-8X32 |
0.4718 |
| Extraction method | DESs (HBA:HBD) | Optimum extraction conditions | μg-β-Carotene/gr-Pumpkin | |
| Predicted | Actual 1 | |||
| MMAE | Menthol:Acetic acid | HBD Molar Ratio: 0.6831 Mixing Time: 60.0 min. |
0.621 | 0.610 |
| Menthol:Propionic acid | HBD Molar Ratio: 0.6452 Mixing Time: 15.0 min. |
11.528 | 10.816 | |
| HAE | Menthol:Acetic acid | HBD Molar Ratio: 0.6757 Homogenization Time: 116.3 sec. Homogenization Speed: 11623 rpm |
0.712 | 0.711 |
| Menthol:Propionic acid | HBD Molar Ratio: 0.6198 Homogenization Time: 30.0 sec. Homogenization Speed: 7061 rpm |
8.966 | 8.762 | |
| Extraction method | DESs (HBA:HBD) | Optimum extraction conditions | μg-β-Carotene/gr-spinach | |
| Predicted | Actual 1 | |||
| MMAE | Menthol:Acetic acid | HBD Molar Ratio: 0.6987 Mixing Time: 40.2 min. |
13.010 | 9.348 |
| Menthol:Propionic acid | HBD Molar Ratio: 0.8000 Mixing Time: 60.0 min. |
16.924 | 18.990 | |
| Menthol:Butyric acid | HBD Molar Ratio: 0.8000 Mixing Time: 60.0 min. |
5.640 | 4.468 | |
| HAE | Menthol:Acetic acid | HBD Molar Ratio: 0.8000 Homogenization Time: 30.0 sec. Homogenization Speed: 10311 rpm |
18.390 | 13.751 |
| Menthol:Propionic acid | HBD Molar Ratio: 0.8000 Homogenization Time: 120.0 sec. Homogenization Speed: 7000 rpm |
18.870 | 12.177 | |
| Menthol:Butyric acid | HBD Molar Ratio: 0.8000 Homogenization Time: 120.0sec. Homogenization Speed: 14000 rpm |
10.131 | 12.534 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




