Submitted:
13 November 2024
Posted:
15 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Antimicrobial Effect of Onion Extracts on Salmonella Survival and Growth
2.1.1. Salmonella Strains and Inoculum Preparation
2.1.2. Onion extract preparation
2.1.3. Microtiter Broth Dilution Method
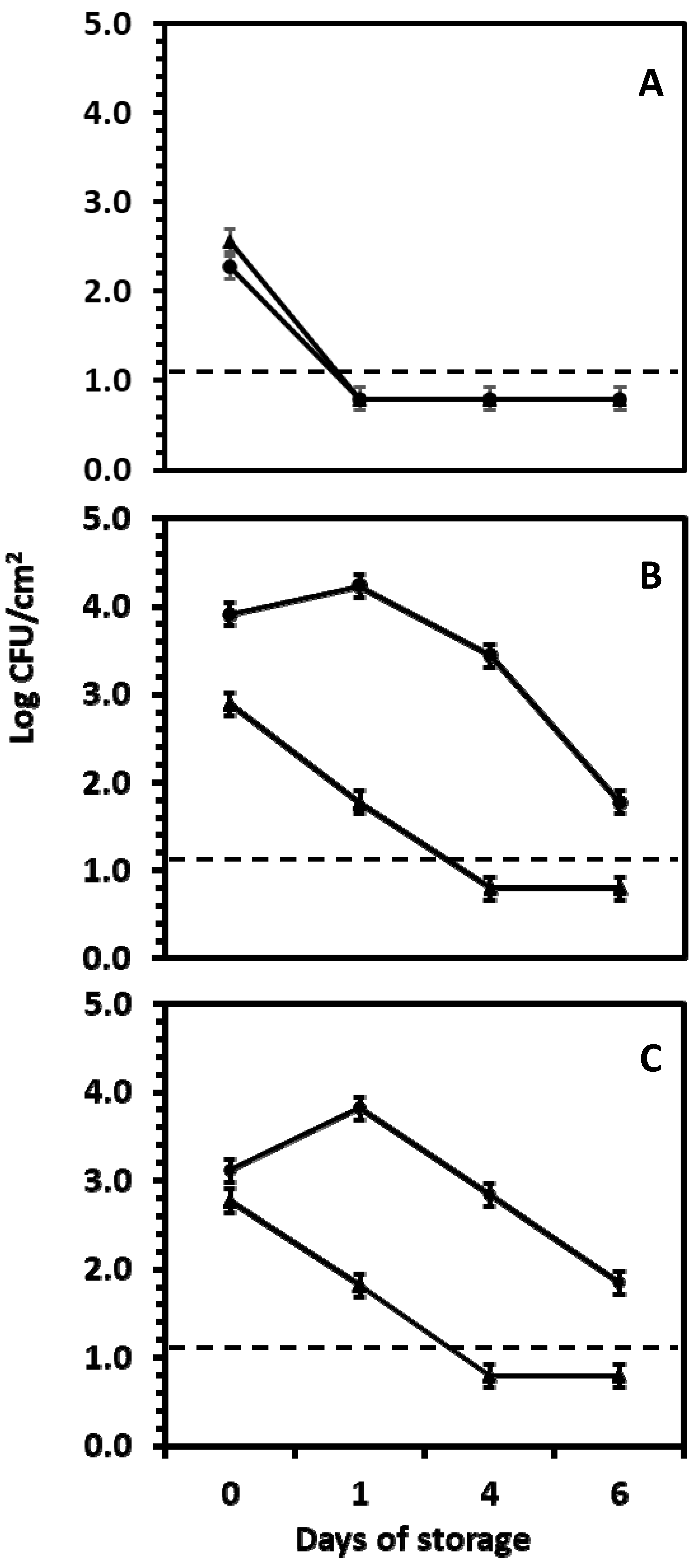
2.2. Fate of Salmonella serotype Newport on Red, White and Yellow Onion Varieties
2.2.1. Procurement of Onion Bulbs
2.2.2. Salmonella strains and inoculum preparation
2.2.3. Bulb Onion Inoculation
2.2.4. Sample Collection and Salmonella Enumeration
2.3. Measurement of Water Activity
4. Results and Discussion
4.1. Antimicrobial Effect of Onion Extracts on Salmonella Survival and Growth.
4.1.1. Effect of Various Exposed Lights During Onion Growth on the Inhibition of Salmonella by Extracts of red, white and yellow Onion Varieties
4.1.2. Impact of various onion genotypes and their layers on Salmonella growth
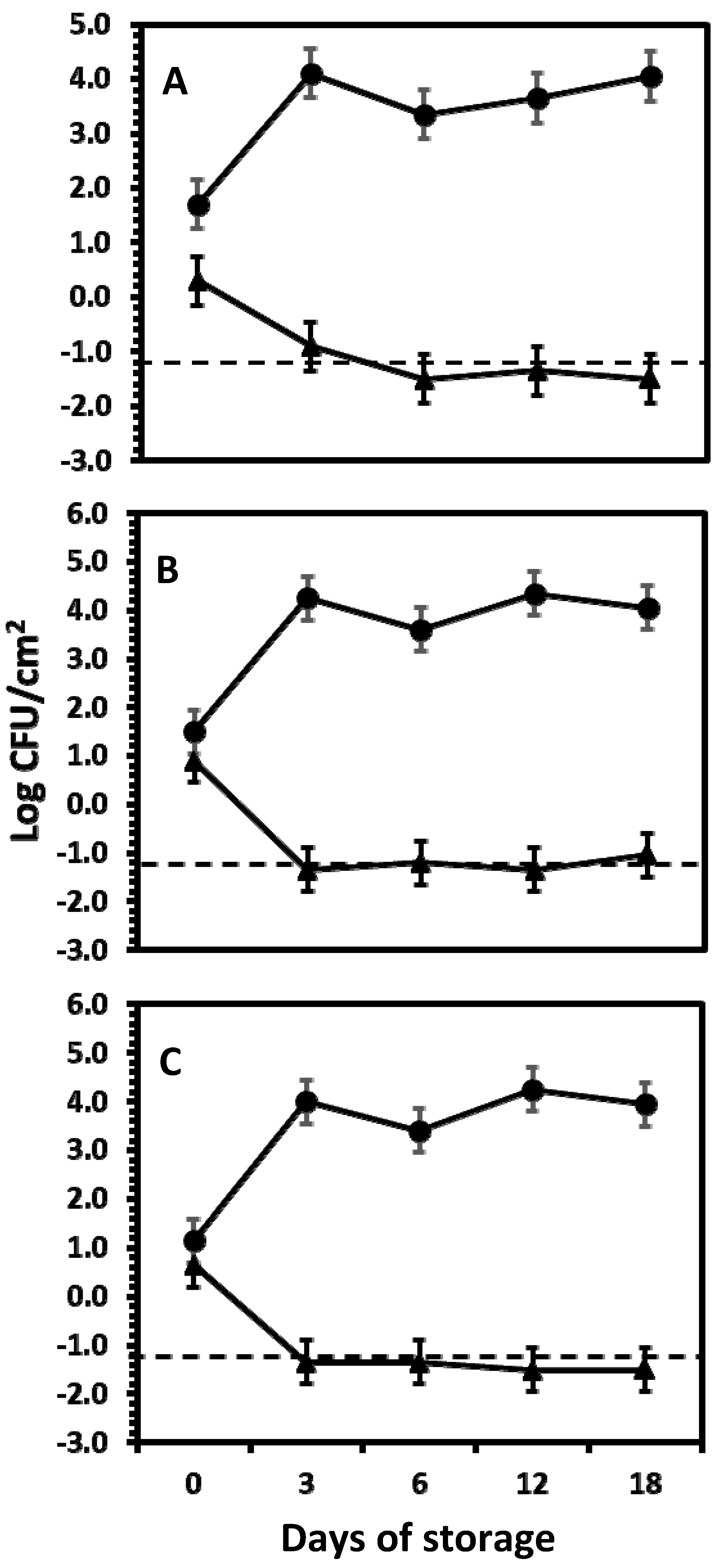
4.1.3. Effect of Salmonella Inoculum Internalization
| Layer | |||||
|---|---|---|---|---|---|
| Variety | Outer papery | 1st after outer | 2nd after outer | 3rd after outer | 4th after outer |
| Red | 0.509 ± 0.032 Aa | 0.764 ± 0.023 C | 0.836 ± 0.023 G | 0.897 ± 0.024 E | 0.961 ± 0.011 Fb |
| White | 0.583 ± 0.003 B | 0.904 ± 0.042 E | 0.909 ± 0.001 E | 0.928 ± 0.003 E | 0.997 ± 0.003 G |
| Yellow | 0.525 ± 0.011 A | 0.863 ± 0.027 D | 0.895 ± 0.017 E | 0.921 ± 0.015 E | 0.972 ± 0.001 F |
|
a Water activity (aw) values are means ± standard error. b Within columns and rows, means with different letters are significantly different (P < 0.05). | |||||
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brummell, D.A. and Toivonen, P.M. A. Postharvest Physiology of Vegetables, in Handbook of Vegetables and Vegetable Processing, 2018, pp. 223–245.
- Nile, S.H.; Nile, A.S.; Keum, Y.S.; Sharma, K. Utilization of quercetin and quercetin glycosides from onion (Allium cepa L.) solid waste as an antioxidant, urease, and xanthine oxidase inhibitors. Food Chem. 2017, 235, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Majid, I.; Hussain, S.; Nanda, V. Impact of sprouting on the degradation kinetics of color and vitamin C of onion powder packaged in different packaging materials. JFPP 2018, 43, e138. [Google Scholar] [CrossRef]
- Hollman, P.C. H.; Arts, I.C. W. Flavonols, flavones and flavanols – nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1081–1093. [Google Scholar] [CrossRef]
- Islam, M.; Doyle, M.P.; Phatak, S.C.; Millner, P.; Jiang, X. Survival of Escherichia coli O157:H7 in soil and on carrots and onions grown in fields treated with contaminated manure composts or irrigation water. Food Microbiol. 2005, 63–70. [Google Scholar] [CrossRef]
- Emch, A.W.; Waite-Cusic, J.G. Conventional curing practices reduce generic Escherichia coli and Salmonella spp. on dry bulb onions produced with contaminated irrigation water. Food Microiol. 2016, 41–47. [Google Scholar] [CrossRef]
- Machado-Moreira, B.; Richards, K.; Brennan, F.; Abram, F.; Burgess, C.M. Microbial Contamination of Fresh Produce: What, Where, and How? Compr. Rev. Food Sci.Food Saf. 18, 1727–1750. [CrossRef]
- Kianian, F.; Marefati, N.; Boskabady, M.; Ghasemi, S.Z.; Boskabady, M.H. Pharmacological properties of Allium cepa, preclinical and clinical evidences; A review. Iran J. Pharm. Res. 2021, 107–134. [Google Scholar] [CrossRef]
- Prakash, D.; Singh, B.N.; Upadhyay, Antioxidant and free radical scavenging activities of phenols from onion (Allium cepa), Food Chemistry, vol. 102, no. 4, pp. 1389–1393, 2007/01/01/ 2007. [CrossRef]
- Sagar, N.A.; Pareek, S.; Gonzalez-Aguilar, G.A. Quantification of flavonoids, total phenols and antioxidant properties of onion skin: a comparative study of fifteen Indian cultivars. J. Food Sci. Technol., 2020, 57, 2423–2432.ko. [Google Scholar] [CrossRef]
- Lombard, K.; Peffley, E.; Geoffriau, E. ; Thompson, L; Herring, A. Quercetin in onion (Allium cepa L.) after heat-treatment simulating home preparation. J. Food Compos. Anal. 2005, 18, 571–581. [Google Scholar] [CrossRef]
- Jang, M.; Asnin, L.; Nile, S.H.; Keum, Y.S.; Kim, H.Y.; Park, S.W. Ultrasound-assisted extraction of quercetin from onion solid wastes. Int. J. Food Sci. Tech. 2013, 48, 246–252. [Google Scholar] [CrossRef]
- Ioku, K.; Aoyama, Y.; Tokuno, A.; Terao, J.; Nakatani, N.; Takei, Y. Various cooking methods and the flavonoid content in onion. J. Nutr. Sci. Vitaminol. 2001, 47, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Islek, M.; Nilufer-Erdil, D.; Knuthsen, P. Changes in flavonoids of sliced and fried yellow onions (Allium cepa L. var. zittauer) during storage at different atmospheric, temperature, and light conditions. J. Food Proc. Preserv 2015, 39, 357–368. [Google Scholar] [CrossRef]
- Benett, S.D.; Sodha, S.V.; Ayers, T.L.; Lynch, M.F.; Gould, L.H.; Tauxe, R.V. Produce-associated foodborne disease outbreaks, USA, 1998–2013. Epidemiol. Infect. 2018, 146, 1397–1406. [Google Scholar] [CrossRef] [PubMed]
- Cueva, C.; Moreno-Arribas, M.V.; Martín-Álvarez, P.J.; Bills, G.; Vicente, M.F.; Basilio, A.; Rivas, C.L.; Requena, T.; Rodríguez, J.M.; Bartolomé, B. Antimicrobial activity of phenolic acids against commensal, probiotic and pathogenic bacteria. Res. Microbiol. 2010, 161, 372–382. [Google Scholar] [CrossRef]
- Lee, O.-H.; Lee, B.-Y. Antioxidant and antimicrobial activities of individual and combined phenolics in Olea europaea leaf extract. Bioresour. Technol. 2010, 101, 3751–3754. [Google Scholar] [CrossRef]
- Salaheen, S.; Jaiswal, E.; Joo, J.; Peng, M.; Ho, R.; Oconnor, D.; Adlerz, K.; Aranda-Espinoza, J.H.; A.; Sexton, R.; McKenna, C.; Cornell, J.; Sandoval, A.L.; Wang, H.; Bell, R.L.; Christan Stager, C.; Zamora Nava, M.C.; Lara de la Cruz, J.L.; Sánchez Córdova, L.I.; Regalado Galván, P.; Arias Ortiz, J.; Flowers, S.; Grisamone, A.; Gieraltowski , L.; Bazaco, M.; Viazis, S. Multistate outbreak of Salmonella Oranienburg infections linked to bulb onions imported from Mexico – United States, 2021. Food Cont. 2024, 160, 110325. [CrossRef]
- Sandquist, E.L. Effect of Simulated Storage and Distribution on Listeria innocua Growth in Non-traditional Salad Ingredients. M. Sc. Thesis, California Polytechnic State University, San Luis Obispo, CA, January 2021.
- McCormic, Z.D.; Patel, K.; Higa, J.; Bancroft, J.; Donovan, D.; Edwards, L.; Cheng, J.; Adcock, B.; Bond, C.; Pereira, E.; Doyle, M.; Wise, M.E.; Gieraltowski, L.; Brandenburg, J.M.; Leeper, M.; Kim, B.; Arroyo, F.; Lui, J.; Schwensohn, C.; Gollarza, L. Bi-national outbreak of Salmonella Newport infections linked to onions: the United States experience. Epidemiol. Infect. 2022, 150, 1–8. [Google Scholar] [CrossRef]
- Mitchell, M.R.; Kirchner, M.; Schneider, B.; McClure, M.; Neil, K.P.; Madad, A.; Jemaneh, T.; Tijerina, M.; Nolte, K.; Wellman, A.; Neises, D.; Pightling, A.; Angela Swinford, A.; Piontkowski, A.; Sexton, R.; McKenna, C.; Cornell, J.; Sandoval, A.L.; Wang, H.; Bell, R.L.; Christan Stager, C.; Zamora Nava, M.C.; Lara de la Cruz, J.L.; Sánchez Córdova, L.I.; Regalado Galván, P.; Arias Ortiz, J.; Flowers, S.; Grisamone, A.; Gieraltowski, L.; Bazaco, M.; Viazis, S. Multistate outbreak of Salmonella Oranienburg infections linked to bulb onions imported from Mexico – United States, 2021. Food Cont. 2024, 160, 110325. [Google Scholar] [CrossRef]
- Commichaux, S. , Rand, H., Javkar, K., Molloy, E.K., Pettengill, J.B., Pightling, A., Hoffmann, M., Pop, M., Jayeola, V., Foley, S., and Luo, Y. Assessment of plasmids for relating the 2020 Salmonella enterica serovar Newport onion outbreak to farms implicated by the outbreak investigation. BMC Genomics 2023, 24, 165. [Google Scholar] [CrossRef]
- Zhang, X.; Bian, Z.; Yuan, X. ; Chen, X, Lu, C. A review on the effects of light-emitting diode (LED) light on the nutrients of sprouts and microgreens. Trends Food Sci. Technol. 2020, 99, 203–216. [Google Scholar] [CrossRef]
- Sharma, K.; Mahato, N.; Lee, Y.R. Systematic study on active compounds as antibacterial and antibiofilm agent in aging onions. J. Food Drug Anal. 2018, 26, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Brandt, A.L.; Castillo, A.; Harris, K.B.; Keeton, J.T.; Hardin, M.D.; Taylor, T.M. Inhibition of Listeria monocytogenes by food antimicrobials applied singly and in combination. J. Food Sci. 2010, 75, M557–M563. [Google Scholar] [CrossRef] [PubMed]
- Branen, J.K.; Davidson, P.M. Enhancement of nisin, lysozyme, and monolaurin antimicrobial activities by ethylenediaminetetraacetic acid and lactoferrin. Int. J. Food Microbiol. 2004, 63–74. [Google Scholar] [CrossRef] [PubMed]
- USDA Food Safety and Inspection Service. Isolation and identification of Salmonella from meat, poultry, pasteurized egg, Siluriformes (fish) products and carcass and environmental sponges. https://www.fsis.usda.gov/sites/default/files/media_file/documents/MLG-4.14.pdf (accessed on 12 March 2024).
- Trammell K., W.; Peterson, C.E. Quantitative differences in the flavonol content of yellow onion, AIIium cepa L.1. J. Amer. Soc. Hort. Sci. 1976, 101, 205–207. [Google Scholar] [CrossRef]
- Roso, R.; Nunes, U.R.; Müller, C.A.; Paranhos, J.T.; Lopes, S.J.; Dornelles, S.H. B.; Bertagnolli, C.M.; Huth, C.; Forte, C.T.; Menegaes, J.F. Light quality and dormancy overcoming in seed germination of Echium plantagineum L. (Boraginaceae). Braz. J. Biol. 2021, 81, 650–656. [Google Scholar] [CrossRef]
- Ko, E.Y.; Nile, S.H.; Sharma, K.; Li, G.H.; Park, S.W. Effect of different exposed lights on quercetin and quercetin glucoside content in onion (Allium cepa L.). Saudi J. Biol. Sci. 2015, 22, 398–403. [Google Scholar] [CrossRef]
- Kołton, A. ; Długosz-GrochowskaO.; Wojciechowska, R.; Czaja, M. Biosynthesis Regulation of Folates and Phenols in Plants. Sci. Hortic. 2022, 291, 110561. [Google Scholar] [CrossRef]
- Grace, S.C.; Logan, B.A. Energy dissipation and radical scavenging by the plant phenylpropanoid pathway. Phil. Trans. R. Soc. Lond. 2000, 355, 1499–1510. [Google Scholar] [CrossRef]
- Sagar, N.A.; Pareek, S.; Benkeblia, N.; Xiao, J. Onion (Allium cepa L.) bioactives: chemistry, pharmacotherapeutic functions, and industrial applications. Food Front. 2022, 3, 380–412. [Google Scholar] [CrossRef]
- Górniak, I. , Bartoszewski, R. & Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Quecan, B.X. V.; et al. Effect of quercetin rich onion extracts on bacterial quorum sensing. Front. Microbiol. 2019, 10, 867. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, K. Flavonols and flavones in food plants: a review. Int.. Food Sci. Technol. 1976, 11, 433–448. [Google Scholar] [CrossRef]
- Haytowitz, D.B.; Bhagwat, S.; Holden, J.M. Sources of variability in the flavonoid content of foods. Procedia Food Sci. 2013, 2, 46–51. [Google Scholar] [CrossRef]
- Quecan, B.X. V.; Santos, J.T. C.; Rivera, M.L. C.; Hassimotto, N.M. A.; Almeida, F.A.; Pinto, U.M. Effect of quercetin rich onion extracts on bacterial quorum sensing. Front. Microbiol. 2019, 10, 867. [Google Scholar] [CrossRef]
- Cushnie, T.P.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef]
- Wu, D.; Kong, Y.; Han, C.; Chen, J.; Hu, L.; Jiang, H.; Xu Shen, X. d-Alanine:d-alanine ligase as a new target for the flavonoids quercetin and apigenin. Int. J. Antimicrob. Agents 2008, 32, 421–426. [Google Scholar] [CrossRef]
- Brandl, M. Plant lesions promote the rapid multiplication of Escherichia coli O157: H7 on postharvest lettuce. Appl. Environ. Microbiol. 2008, 74, 5285–5289. [Google Scholar] [CrossRef]
- Semenov, A.M.; Kuprianov, A.A.; Van Bruggen, A.H. Transfer of enteric pathogens to successive habitats as part of microbial cycles. Microb. Ecol. 2010, 60, 239–249. [Google Scholar] [CrossRef]
- Lim, J.A.; Lee, D.H.; Heu, S. The interaction of human enteric pathogens with plants. Plant Pathol. J. 2014, 30, 109–116. [Google Scholar] [CrossRef]
- Belo, T., Du Toit, L.J., and Lahue, G.T. Reducing the risk of onion bacterial diseases: A review of cultural management strategies. Agronomy Journal 2023, 115, 459–473.
- Gorshkov, V.; Tsers, I. Plant susceptible responses: the underestimated side of plant-pathogen interactions. Biol. Rev. Camb. Philos. Soc. 2022, 97, 45–66. [Google Scholar] [CrossRef] [PubMed]
- Škerget, M.; Majhenič, L.; Bezjak, M.; Knez, Ž. Antioxidant, radical scavenging and antimicrobial activities of red onion (Allium cepa L) skin and edible part extracts. CABEQ 2009, 23, 435–444. Available:https://hrcak.srce.hr/en/45385.
- Masfria, G.H.; Mierza, V. Analysis chemical compounds and antimicrobial activity of red onion (Allium cepa L. ) bulb skin extract. RASAYAN J. Chem. 2019, 12, 1002–1010. [Google Scholar] [CrossRef]
- Kumar, M.; Barbhai, M.D.; Hasan, M.; Punia, S.; Dhumal, S.; Radha; Rais, N.; Chandran, D.; Pandiselvam, R.; Kothakota, A.; Tomar, M.; Satankar, V.; Senapathy, M.; Anitha, T.; Dey, A.; Sayed, A.A. S.; Gadallah , F.M.; Amarowicz, R.; Mekhemar, M. Onion (Allium cepa L.) peels: A review on bioactive compounds and biomedical activities. Biomed. Pharmacother. 2022, 146, 112498. [CrossRef]
- Lieberman, V.M.; Zhao, I.Y.; Schaffner, D.W.; Danyluk, M.D.; Harris, L.J. Survival or growth of inoculated Escherichia coli O157:H7 and Salmonella on yellow onions (Allium cepa) under conditions simulating food service and consumer handling and storage. J. Food Prot. 2015, 78, 42–50. [Google Scholar] [CrossRef]
- Lieberman, V.M.; Harris, L.J. Fate of inoculated Listeria monocytogenes on yellow onions (Allium cepa) under conditions simulating food service and consumer handling and storage. Food Cont. 2019, 96, 375–382. [Google Scholar] [CrossRef]
- Khiari, Z.; Makris, D.P. Stability and transformation of major flavonols in onion (Allium cepa) solid wastes. J. Food Sci. Technol. 2012, 49, 489–494. [Google Scholar] [CrossRef]
- Santiago, B.; Arias Calvo, A.; Gullón, B.; Feijoo, G.; Moreira, M.T.; González-García, S. Production of flavonol quercetin and fructooligosaccharides from onion (Allium cepa L.) waste: An environmental life cycle approach. Chem. Eng. J. 2020, 392, 123772. [Google Scholar] [CrossRef]
- Mardani, N.; Jahadi, M.; Sadeghian, M.; Keighobadi, K.; Khosravi-Darani, K. Antimicrobial activities, phenolic and flavonoid contents, antioxidant and DNA protection of the internal and outer layers of Allium cepa L. from Iran." NFS Journal 2023, 31, 93–101. [Google Scholar] [CrossRef]
- Kim, S.; Kim, G.-H. Quantification of quercetin in different parts of onion and its DPPH radical scavenging and antibacterial activity. Food Sci. Biotechnol. 2006, 15, 39–43. [Google Scholar]
- Mark, G.L.; Gitaitis, R.D.; Lorbeer, J.W. Bacterial Diseases of Onion. In Allium Crop Science: Recent Advances; Rabinowitch, H.D., Currah, L., Eds.; CABI Publishing: New York, NY, USA, 2002; pp. 267–292. [Google Scholar]
- Van Linden, V.; Scheerlinck, N.; Desmet, M.; De Baerdemaeker, J. Factors that affect tomato bruise development as a result of mechanical impact. Postharvest Biol. Technol. 2006, 42, 260–270. [Google Scholar] [CrossRef]
- Aliasgarian, S.; Ghassemzadeh, S.H. R.; Moghaddam, H.R. M.; Ghaffari, H. Mechanical damage of strawberry during harvest and postharvest operations. World Appl. Sci. J. 2013, 22, 969–974. [Google Scholar] [CrossRef]
- Tokarskyy, O.; De, J.; Fatica, M.K.; Brecht, J.; Schneider, K. R. Survival of Escherichia coli O157:H7 and Salmonella on bruised and unbruised tomatoes from three ripeness stages at two temperatures. J. Food Prot. 2018, 81, 2028–2033. [Google Scholar] [CrossRef] [PubMed]
- Wells J., M.; Butterfield, J. E. Incidence of Salmonella on fresh fruits and vegetables affected by fungal rots or physical injury. Plant Dis. 1999, 83, 722–726. [Google Scholar] [CrossRef] [PubMed]
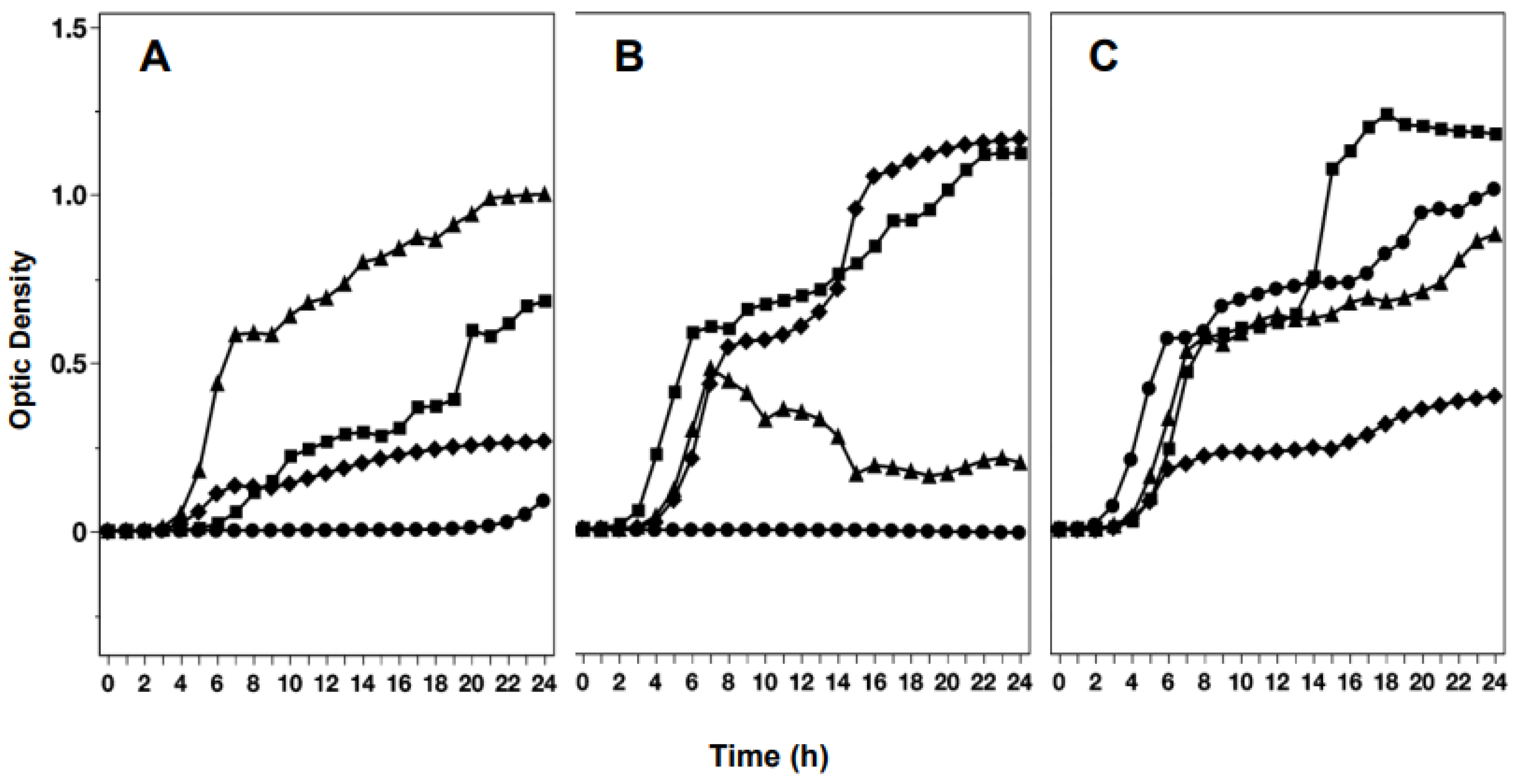
| Mean ∆ODa ± STDEV by location of scales used for extracts | ||
|---|---|---|
| Genotype | Outer | Inner |
| 25X | 0.62 ± 0.11 ABb | 0.97 ± 0.35 AB |
| 218Y | 0.85 ± 0.38 AB | 0.67 ± 0.02 AB |
| 231Y | 0.60 ± 0.02 AB | 0.70 ± 0.22 AB |
| 541X | 0.76 ± 0.20 AB | 0.68 ± 0.04 AB |
| 542 | 0.93 ± 0.38 AB | 0.33 ± 0.47 AB |
| 544X | 0.82 ± 0.01 AB | 0.80 ± 0.01 AB |
| 545X | 0.88 ± 0.43 A | 1.14 ± 0.03 AB |
| 1102 | 0.83 ± 0.37 AB | 1.08 ± 0.02 A |
| 1104 | -0.0025 ± 0.01 B | 0.26 ± 0.36 AB |
| 1196 | 0.64 ± 0.03 AB | 0.92 ± 0.34 AB |
| 1197 | 0.87 ± 0.35 AB | 0.58 ± 0.06 AB |
| YELLOW H6 | 0.67 ± 0.03 A | 1.17 ± 0.03 AB |
|
a ∆OD represents the change in OD reading at λ 630 for each extract and is calculated by subtracting the OD at zero time from the OD observed after 24 h incubation period. A ∆OD of ≤ 0.05 would be considered as no Salmonella growth. b Means followed by the same letter are not significantly different (P ≥ 0.05). Each mean represents the OD values obtained from 9 analytical values. | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





