1. Introduction
Meningiomas have been well described over centuries but received their formal name by the seminal work of Harvey Cushing in 1922 [
1]. Defined as extra-axial masses originating from arachnoidal cap cells of the leptomeninges [
2], these tumors grow within the brain's outer covering of the arachnoid mater and pia mater.
Meningiomas are the most common primary central nervous system (CNS) tumors, accounting for 15% of symptomatic intracranial neoplasms and 33% of asymptomatic intracranial neoplasms found on autopsy [
3,
4]. They have a strong predilection for women, with 65% found in women aged 40-50 years [
4].
Biopsy or resection is the sole method for confirming a definitive diagnosis; however, the typical radiologic appearance is often sufficient and the most common diagnostic technique [
1]. Magnetic resonance imaging (MRI) findings are usually consistent with a dural-based, well-circumscribed, homogeneously enhancing lesion [
1]. Radiographic classification distinguishes en plaque meningiomas, characterized by extensive dural thickening, from globose meningiomas, presenting as rounded extra-axial masses [
4].
The World Health Organization (WHO) acknowledges 15 histological subtypes of meningiomas, with the vast majority being WHO Grade 1 (benign). Despite their benign nature, encapsulated structure, and limited genetic aberrations, their anatomical locations may lead to serious and potentially lethal consequences [
5]. WHO Grade II and III meningiomas have higher recurrence rates and will require more aggressive treatment. Certain molecular factors also indicate malignant behavior including presence of telomerase reverse transcriptase (TERT) promoter mutation or homozygous deletions in the cell cycle regulator genes CDKN2A and/or CDKN2B [
6].
Meningiomas are typically slow-growing and non-infiltrative lesions with an insidious onset of symptoms, which are site dependent. While lacking a pathognomonic presentation, these neoplasms may manifest with common symptoms associated with CNS tumors, including headache due to increased intracranial pressure, focal neurological deficits, and seizures [
1]. Symptoms of meningioma are often with associated brain edema and mass effect, or direct compression of critical structures. Despite their high prevalence, clinical management varies significantly based on multifaceted considerations, including location, size, patient age, presentation, and pathology [
7].
Treatment options for intracranial meningiomas encompass surgical resection alone, surgery followed by adjuvant radiation therapy (RT), or primary RT [
8]. The treatment of choice, when safely attainable, is complete surgical resection (Simpson grade 1-3), including the dura of origin and infiltrated bone [
9]. However, inherent surgical risk may preclude complete resection, particularly for tumors infiltrating vital intracranial structures or within frail patient populations. Rates of recurrence are more frequent with subtotal resection and WHO grade II and III tumors, and undergoing multiple resections increases morbidity secondary to scarring and increased infiltration [
10]. After the 1980s when computerized tomography (CT) or magnetic resonance imaging (MRI) imaging were used to plan radiotherapy, the 5-year progression free survival rate for patients with benign meningiomas increased from 77% to 98% [
11]. In cases where complete surgical resection was not feasible, subtotal resection with adjuvant radiation can achieve results comparable to total resection [
11].
Radiotherapy (RT) can be delivered to intracranial meningiomas in different modalities including stereotactic radiosurgery (SRS), hypofractionated stereotactic radiotherapy (HSRT), and conventional radiotherapy. SRS utilizes a single irradiation fraction, while HSRT delivers 2-5 ablative doses of focused radiation. Conventional radiotherapy entails the delivery daily small individual doses of radiation over 5-6 weeks.
Historically, both SRS and FSRT were reserved for lesions that were recurrent, more malignant, or in patients who were poor candidates for surgical intervention [
9,
12]. With increased use of brain imaging and an increase in aging populations, there has been an increase in the diagnosis of asymptomatic meningiomas [
13,
14]. RT has become more commonplace as first-line therapy for select meningioma cases, particularly small meningiomas in challenging or high-risk anatomical locations [
15]. Treatment concepts involving a combination of surgery and SRS/FSRT are also increasingly used, but controversies remain regarding timing, type, and dosing therapy of various RT techniques [
16].
Population-based analysis reveals an increased incidence over recent years alongside decreasing tumor size at the time of diagnosis [
7]. A metanalysis on the natural history of meningioma growth showed that although half of the incidental meningiomas found will exhibit no growth, the remaining half exhibited either linear or exponential growth [
17]. Precise determination of the origin and anatomical considerations of meningiomas is fundamental for strategic surgical decision-making [
18]. Adjacent anatomical structures dictate the ease of surgical access, ability to achieve complete resection, and the risk of morbid tumor progression.
Over recent years, significant research focusing on RT for meningioma control has been shared among the literature. While prior investigations detailed outcomes in broader regions such as the skull base and parasellar regions, recent reports have accumulated detailing outcomes, nuances, and updated SRS treatment regimens for specific anatomical subregions; instead of a study investigating skull-based meningiomas broadly, more specific analysis of subregions such as the petroclival [
19,
20,
21], olfactory groove [
22,
23,
24], and foramen magnum [
25,
26,
27,
28] are now available.
In the subsequent sections, this review will discuss anatomical challenges inherent in specific locations and provide a synthesis of the latest evidence on radiosurgical intervention. We will then delve into the intricacies of radiosurgical methodology, dosimetric considerations, and future directions of radiotherapy.
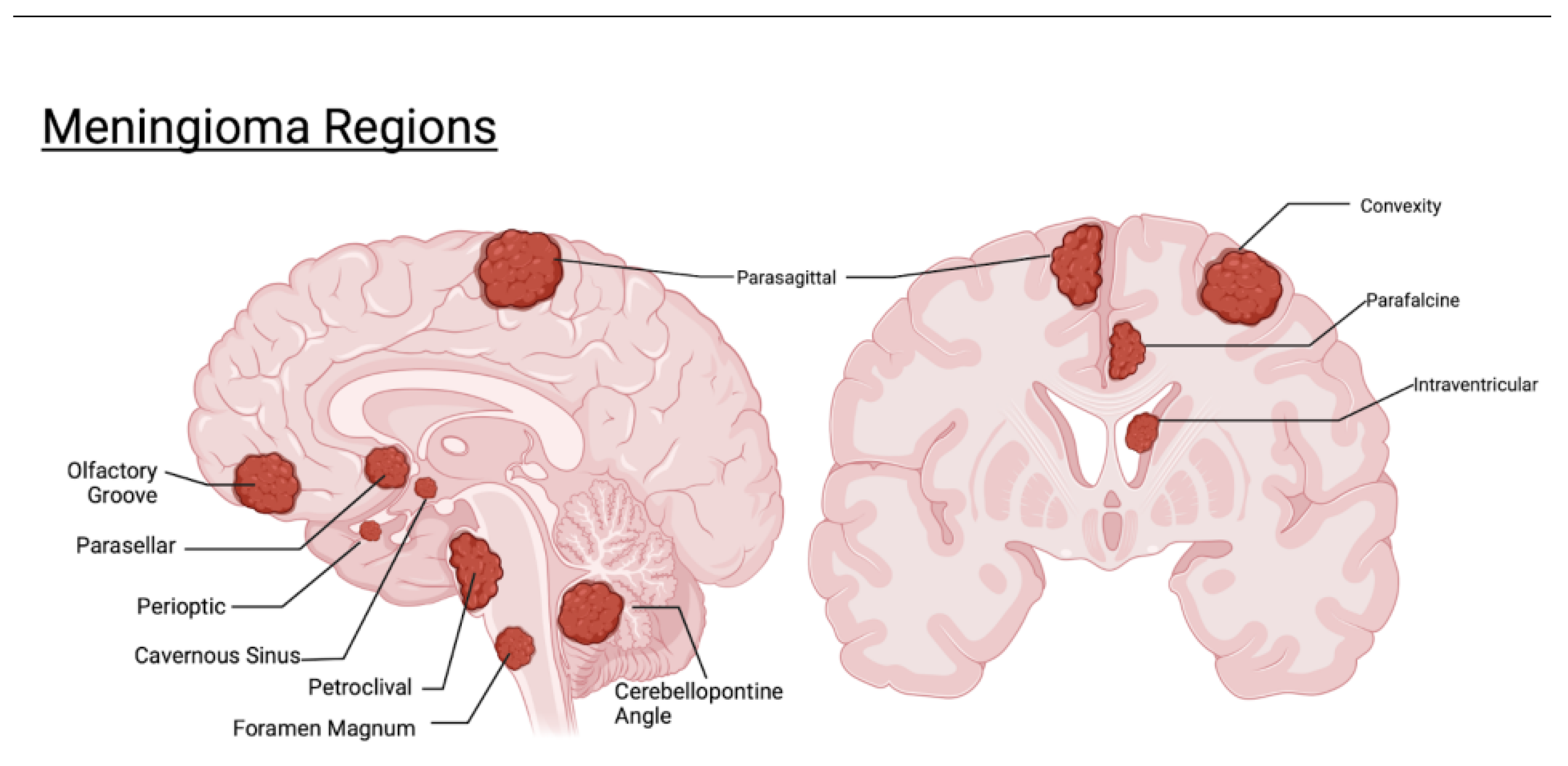
2. Classification by Location
Meningiomas are named based on their anatomical location. The most common sites reported are falcine, parasagittal, convexity, and sphenoid wing meningiomas [
4,
29]. Their presentation is entirely dependent on the adjacent neuronal and vascular structures affected by mass effect. Prior reviews have reported the most common presenting symptoms are headache (33.3–36.7%), focal cranial nerve deficit (28.8–31.3%), seizure (16.9–24.6%), cognitive change (14.4%), weakness (11.1%), vertigo/dizziness (9.8%), ataxia/gait change (6.3%), pain/sensory change (5.6%), proptosis (2.1%), syncope (1.0%), and asymptomatic (9.4%) [
30].
Although symptoms such as headache are common among locations, specific presenting symptoms based on location are detailed in
Table 1. With advances in modern imaging, meningiomas are being discovered earlier when they are smaller than ever before [
7]. Establishing a diagnosis at an earlier stage is associated with a lack of many symptoms often seen at different locations when the tumor is larger.
As further research becomes available investigating specific anatomical locations, results can be interpreted on a more specific basis than prior broad encompassing regions. For example, prior results have been posted on radiosurgery for “Skull base” meningiomas, but this truly encompasses subregions including the clivus, petrous bone, cavernous sinus, tentorium, sphenoid bone, sella turcica, olfactory groove, and optic nerve sheath [
9]. This paper reviews evidence for radiosurgery within these specific anatomical subregions, highlights regional challenges, and provides a depth of available outcome-based evidence on tumor control.
Figure 1 highlights regions covered within this review and their relation to one another.
3. Guidelines for Meningioma Treatment
There is no clear cut algorithm for meningioma management; the appropriate management strategy for must be determined on a case-by-case basis [
6]. Despite being one of the most common intracranial tumors, variations in management approaches exist between countries and surgical centers [
16]. Trends derived from the Surveillance, Epidemiology, and End Results (SEER) database of the National Cancer Institute in 2017 showed that 42.4% of patients with meningiomas underwent surgical treatment with or without radiotherapy, while 52.1% were managed with active surveillance [
7]. Of those managed surgically, 8.7% received RT at some point, with 95% receiving it after surgery. Only 5.6% of patients underwent radiation therapy alone [
7].
While some meningiomas may not increase in size, many do if watched long enough. Especially in young patient populations; it is statistically unlikely that a 20-year-old will have 50 years of no growth, so age is of utmost importance in treatment decision-making [
37]. Prior publications on natural history of meningioma growth have shown that exponential or linear growth is demonstrated in 63% [
38], 53%[
39], and overall growth in as high as 83.5% of tumors [
40]. Taking the potential for growth into consideration is particularly relevant when managing small asymptomatic meningiomas adjacent to vasculature or cranial nerves.
Recently, there has been increased investigation into using RT as primary monotherapy instead of conservative observation. An important study by Sheehan and colleagues compared SRS alone vs active surveillance in asymptomatic incidental meningiomas. Their analysis found that SRS affords superior radiologic tumor control compared to active surveillance without increasing the risk of neurological deficits [
41].
Updated 2021 European Association of Neuro-Oncology (EANO) guidelines state that observation is the first option for asymptomatic meningiomas. These guidelines also state that RT may be a complementary therapy or even an alternative approach to surgery in certain situations [
16].
4. Anatomical Locations and Updated Evidence
4.1. Parafalcine and Parasagittal
The falx cerebri is a sickle-shaped structure running within the longitudinal fissure between the two cerebral hemispheres. Anchored anterior to the crista galli and posterior to the occipital protuberance, it forms a sail-like divider of the two hemispheres [
42]. Meningiomas originating from the falx are termed parafalcine meningiomas [
43]. If the meningioma grows into at least one wall of the superior sagittal sinus, it is referred to as a parasagittal meningioma [
44].
Parafalcine and parasagittal meningiomas account for 30% of intracranial meningiomas [
45]. Microsurgery is the mainstay of meningiomas at this location, although there is a high recurrence rate and an increased risk of venous injury during surgical excision [
45]. Engulfment of the pericallosal arteries is another obstacle to radical excision, of which radiotherapy could be useful [
31]. With high rates of venous invasion, complete resection is not possible in the majority of cases [
45]. Optimal management remains controversial because both surgical and non-surgical options frequently fail to provide definitive treatment [
10].
A review by Pinzi and colleagues in 2019 found limited literature on SRT and RS for parafalcine and parasagittal meningiomas [
45]. Their search found only five available studies and only one study since 2015 [
41]. Their analysis showed that falcine tumors are more suitable for complete resection and that parasagittal meningiomas are less optimal for complete resection due to their adherence to the sagittal sinus [
45].
It has been reported previously that parasagittal and parafalcine meningiomas have a less optimal radiosurgical response compared with skull-based lesions [
41]. After review of the data, Pinzi and colleagues concluded that SRT and RS could be more widely applied for parafalcine and parasagittal meningiomas [
45]. They stated that radiation represents viable options for primary and adjuvant therapy for parasagittal and parafalcine meningiomas [
45].
Ding and colleagues published a retrospective review of 65 patients who were treated with a median prescription dose of 15 Gy for parasagittal and parafalcine meningiomas in one fraction. They found a tumor control rate of 85% and 70% at 3 and 5 years. When excluding tumors that had some form of treatment (resection, embolization, and radiotherapy), they found the control rate at both 3 and 5 years was 93% [
10]. This could signify that prior treatment negatively affected radiosurgical efficacy, but additional studies are warranted to investigate this. They additionally found that parasagittal location, no prior resection, and younger age were independent predictors of tumor progression-free survival.
Meningiomas located at the confluence of the falx and tentorium (CFT) represent <2% of intracranial meningiomas and are surgically challenging [
32]. Abdallah and colleagues published a paper reporting the largest series focused exclusively on outcomes of gamma knife radiosurgery for meningiomas arising from the confluence of the CFT in 2022 [
32]. Their paper detailed 20 CFT meningioma patients, 10 patients undergoing radiosurgery for progression of residual tumor following resection and 10 undergoing radiosurgery as primary management. The marginal and maximum doses delivered in this series were 13.0 Gy (IQR 12.1–14.8) and 26 Gy (IQR 25.0–29.5), respectively. All radiation was done with a single unfractionated session. The 1, 5, and 10-year local tumor control rates were 100% (N=20), 100% (N=10), and 83% (N=4), respectively [
32]. Tumor volume decreased in 11 patients and was stable in 8 patients. Their study concluded that radiosurgery is a safe and effective treatment for CFT meningiomas that can be utilized not only as adjuvant therapy but also for primary management [
32].
Studies reviewed suggest that radiosurgical intervention for parasagittal meningiomas may be more effective than parafalcine. There was good tumor control overall and acceptable rates of adverse events. More studies are warranted on this subgroup of meningiomas to determine optimal treatment.
4.2. Convexity
Convexity meningiomas are among the most common extra-axial tumors encountered in neurosurgery [
46]. They grow on the outer surface of the cerebrum directly adjacent to the flat bones of the skull (see
Figure 1). These tumors are relatively simple to approach and resect because both the mass and involved dura mater can be removed [
47], but they account for only about one-sixth of meningiomas [
1]. Craniotomy and removal of convexity meningiomas along with their dural base is the preferred treatment of choice for symptomatic patients [
47]. Despite easier surgical access, the optimal treatment for asymptomatic convexity meningiomas remains unclear, with options including active surveillance or SRS. Observation is the most commonly preferred initial treatment; however, 4.6-26.3% of these asymptomatic patients will develop symptoms and require treatment [
48]. The role of SRS for upfront treatment of asymptomatic convexity meningiomas has yet to be well defined [
48].
A study done by Pikis and colleagues in 2022 investigated data from patients with asymptomatic convexity meningiomas at 14 participating centers using a focused analysis from the International Multicenter Matched Cohort Analysis of Incidental Meningioma Progression During Active Surveillance (IMPASSE) study [
48]. Their study looked at two unmatched cohorts of patients with asymptomatic convexity meningiomas: one received upfront SRS (n=99), and one was managed conservatively (n=140). The mean marginal dose of radiation was 13.26 Gy. After propensity matching for age, they compared 98 patients in each cohort. Matched analysis showed that tumor control was 99% in SRS-treated patients and 69.4% in patients managed conservatively. Their results suggested that upfront SRS treatment of asymptomatic convexity meningiomas is associated with increased tumor control without an increased risk of new neurological deficits.
Another group of patients with Convexity meningiomas underwent retrospective review by Ruiz-Garcia et al. in 2021. They analyzed 18 patients with neurofibromatosis type 2 (NF2) who had convexity meningiomas. A total of 120 convexity meningiomas were treated with single fraction radiosurgical treatment; the median marginal dose was 12.0 Gy. Actuarial tumor control rates at 5, 10, and 15 years were 100% with only one radiation-induced adverse event [
49].
Overall, studies reviewed suggest there may be better control using SRS upfront vs. watching/waiting. There is a paucity of evidence available on SRS for convexity meningiomas. Symptomatic lesions are highly accessible for surgical intervention, but small asymptomatic incidental convexity meningiomas may benefit from SRS over watching and waiting. Additionally, patients with a high tumor lifetime burden such as those with NF2 may benefit from limiting invasive intervention to only when absolutely necessary. Fewer craniotomies may be relevant to preserving the quality of life in NF2 patients [
49].
4.3. Cavernous Sinus
The cavernous sinus is a bilateral structure located lateral to the sella turcica and extends from the superior orbital fissure anteriorly to the petrous part of the temporal bone posteriorly [
50]. Complete surgical resection of cavernous sinus (CS) meningiomas is quite difficult due to the numerous important regional structures and is not possible without significant patient risk [
51]. The cavernous sinus houses a segment of the internal carotid artery (ICA), the sympathetic plexus, cranial nerves III, IV, VI, and the first two branches of CN V. Additionally, a narrow surgical corridor and close relation with the optic chiasm further complicate open resection [
52]. Microsurgical operations may cause cranial nerve palsies, ICA injury or occlusion, and/or cerebrospinal fluid leak [
53].
Anatomical localization of vital structures, lack of arachnoid surfaces, and the tendency of CS meningiomas to invade nerve fascicles make surgical cure extraordinarily difficult to achieve [
52]. The degree of ICA involvement and vascular wall invasion is an important determinant of achieving complete resection [
52]. The available literature shows that the tumor growth control rate of CS meningiomas is >90%, but ongoing debate still exists regarding the optimal treatment; no class 1 evidence exists supporting the superiority of SRS alone, fractionated radiotherapy alone, or any combination of either with subtotal resection [
54].
Basak and colleagues sought to compare the efficacy of surgery and gamma-knife therapy with gamma-knife therapy alone for cavernous sinus meningiomas in 2023. This study uniquely compared two treatment methodologies at a single institution with nine having surgery and radiosurgery, and eleven with radiosurgery alone; a mean marginal dose of >13 Gy in a single fraction was used in both groups. Extraocular nerve palsy is the most common complication with tumor management and is an extremely disturbing detriment to patient quality of life [
52]. Their results showed that this complication was more likely with surgical excision and that recovery was more likely with GK treatment alone. Cranial nerves within the cavernous sinus seem to tolerate relatively higher doses than the optic apparatus [
9], but optic nerve complications also need to be taken into consideration considering its sensitivity to radiation. It has been suggested that keeping radiation to the optic nerve lower than 8Gy-10Gy reduces the risk of adverse radiation effects [
55].
After Basak and colleagues reviewed all available literature combined with their own cohort, they concluded that a distance of >3mm between the tumor and optical anatomical structures is required for safe and effective radiosurgical intervention. They additionally stated that tumors with a volume >10ml and less than 3mm margin with the optical apparatus would benefit from surgical volume reduction prior to SRS [
52].
A larger meta-analysis was performed by Sughrue and colleagues including 2,065 patients with CS meningiomas; 435 were treated with surgery alone, 71 with surgical subtotal resection followed by SRS, 1,300 with SRS alone, and 250 undergoing fractionated radiotherapy alone. Details on radiation parameters were not provided within the publication. The authors found that recurrence rates were better among SRS patients (3.2% [95% CI 1.9–4.5%]) compared with gross-total resection (11.8% [95% CI 7.4–16.1%]) or subtotal resection alone (11.1% [95% CI 6.6–15.7%]) (p < 0.01) [
54]. Additionally, cranial neuropathy rates were markedly higher in patients undergoing resection (59.6% [95% CI 50.3–67.5%]) than SRS alone (25.7% [95% CI 11.5–38.9%]) (p < 0.05) [
56].
An institutional retrospective review on CS meningiomas from 2003-2011 was performed by Zeiler et al. The maximum dosage to the cavernous sinus used was 30-40 Gy and <8 Gy for optic apparatus dosing. The average dose at the 50% isodose line was 13.5 Gy. Their study included 30 patients, 12 patients had previous surgical debulking, and only one patient was asymptomatic at the time of intervention. The average follow-up was 36.1 months with 26 total patients and found the tumor size decreased in 34.6%, was stable in 57.7%, and increased in 7.7% [
53]. The majority of complications were minor and transient.
A larger institutional retrospective review was done on 290 CS meningioma patients from 1987-2009 by Park et al. Gamma knife radiosurgery was performed with a median marginal dose of 13 Gy (range 10-20 Gy) in a single session. Maximum optic pathway dosage was ≤ 10 Gy. Their review had an especially long follow-up period providing insight on long-term control. Follow-up was done at 1, 5, 10, and 15-year appointments, and tumor control rates were 98%, 93%, 85%, and 85%, respectively. In addition to excellent long-term control, they found patients who underwent SRS without prior surgery or soon after microsurgery had a lower risk of treatment failure than patients undergoing SRS for progressive tumors [
51].
Another institutional retrospective review was done by Spiegelmann et al. using a Linear Accelerator to treat 102 patients from 1993-2007. Minimal marginal single session dose was 12-17.5 Gy and optic apparatus <10 Gy. The mean follow-up was 67 months and showed tumor control of 98% with 58% reducing volumes [
57]. Their data strongly supported the notion that early radiosurgery (<1 year from onset) without surgical intervention greatly increases the chance of improving CN deficits. Close to 60% of deficits improved or resolved. Authors regarded radiosurgery as the treatment of choice for CS meningiomas [
57]. These results were similar to the results from Kimball et al. where local tumor control was 100% at 5 years and 98% at 10 years [
58]. Authors found that RS offered greatly superior tumor control with much lower morbidity than surgical resection [
58].
Overall evidence above suggests that radiosurgery is an effective treatment for halting cavernous sinus meningioma progression with acceptable complication rates. Evidence suggests that SRS may decrease the incidence of cranial nerve palsies when compared with open surgical resection [
52,
56,
57] and that there are potential benefits to upfront SRS without prior surgical intervention [
51].
4.4. Parasellar
Parasellar meningiomas represent 15% of all meningiomas [
18]. These meningiomas encompass a subset of skull base meningiomas in or around the sella turcica. This is an anatomically complex region forming the junction of important neurovascular structures [
33]. It is difficult to accomplish a complete resection of these tumors safely without injury to adjacent vital structures including the internal carotid artery, and optic, oculomotor, and trigeminal nerves [
18,
59]. Considering the surgical risk, goals of treatment have shifted from oncological cure to local tumor control. Additionally, some form of intervention may be warranted considering a systematic review of parasellar meningiomas showed new or progressive symptoms 61% of the time [
60]. The cavernous sinus, Meckel’s cave, petroclival region, optic canal, and the anterior clinoid process are considered part of the parasellar region [
18]; these specific subregions within the parasellar region will be discussed individually in subsequent sections.
A study performed by Matoušek and colleagues investigated a combination of endoscopic transnasal optic nerve decompression (ETOND) followed by SRS for parasellar meningiomas. Their study included 12 patients, and 9 of the patients underwent fractionated radiosurgery, with most receiving 5 fractions totaling 30 Gy of radiation. Visual acuity was noted in 10 of 14 eyes (71.4%) in 8 of 12 patients (66.7%). ETOND prior to SRS appeared to improve rates of visual function and reduce rates of SRS complications.
Hu and colleagues sought to investigate if prior surgery affected treatment outcomes of patients with parasellar meningiomas. Their study investigated 93 patients, 45 of whom had surgery prior to gamma knife surgery. The median marginal dose was 12 Gy for both groups, in a single-session. They found that patients were more likely to have improvement of preexisting symptoms without prior surgery (P = 0.009) and more likely to have stable symptoms with prior surgery (P=0.012) [
61].
Cohen-Inbar and colleagues investigated the influence of SRS treatment parameters and timing on parasellar meningiomas with long-term volumetric follow-up evaluation. A mean marginal single session dose of 14 Gy (12-70 Gy) was given to 189 patients achieving tumor control in 91.5% of patients [
59]. Results showed a significant difference in control rates between patients receiving <16 Gy compared to >16 Gy. At the 15-year follow-up, the lesser group had a progression-free survival rate of 79.4%, compared to 95.7% in those that received >16 Gy [
59]. They additionally found tumor volume at initial SRS being >14cm
3 was predictive for failed tumor control and early follow-up volumetric analysis of size change predicted long-term volume changes.
Williams and colleagues performed a retrospective review of 138 patients with parasellar meningiomas treated from 1989 to 2006. Notably, 84 out of 138 patients had undergone previous resection. The mean marginal dose used was 13.7 Gy (range 4.8–30 Gy). There was a low incidence of neurological defects and acceptable rates of tumor control within this cohort. Progression-free survival at 5 and 10 years was 95.4% and 69%, respectively [
62]. Multivariate analysis showed that younger age at the time of intervention was a predictor of effective tumor control.
A larger 10-center study was conducted by Sheehan et al. from 1988 to 2011. Authors performed a retrospective analysis of 763 patients with sellar and parasellar meningiomas treated with Gamma Knife radiosurgery. Patients received a mean marginal dose of 13.2 Gy in a single session. Results showed a 90.2% overall tumor control rate and tumor progression-free survival rates at 3, 5, 8, and 10 years were 98%, 95%, 88%, and 82%, respectively. Contrary to other studies that showed prior surgical resection was a predictor of progression, this study found that prior surgery was favorable for tumor control. They also found smaller tumors and a higher marginal dose favored tumor control.
It has been proposed for parasellar meningiomas that a distance of 5 mm between the meningioma and the optic nerve is considered safe in single-shot RS [
55], and the majority of studies limited radiation to <8 Gy. The overarching theme with parasellar meningiomas is radiosurgery is an effective form of intervention with low complication rates. There was mixed evidence on the impact of previous surgical interventions in patients treated with SRS. One study suggested patients are more likely to have symptomatic improvement from SRS when they don’t undergo a surgical procedure prior [
61]; another study found prior surgery was favorable for tumor control [
60]. Intervening in younger patients was also a predictor of increased tumor control [
62].
More evidence is warranted on larger patient populations to determine the effects of prior surgery and other factors on SRS outcomes for parasellar meningiomas. Overall, SRS for parasellar meningiomas proved to be an effective upfront and adjuvant treatment with adequate rates of tumor control and minimal neurological complications.
4.5. Perioptic
Perioptic meningiomas are defined as those less than 3 mm from the optic apparatus [
63]. Small asymptomatic lesions may be handled with serial imaging, although even minor growth can lead to visual deterioration or complete blindness [
63]. An increasing number of practitioners are supporting radiotherapy for optic nerve sheath meningiomas, considering this to produce better visual outcomes [
64]. Still, surgical decision-making has been constrained because of heterogeneous evidence in the literature [
64]. Surgery is not an option with acceptable outcomes for many tumors within the optic nerve sheath since the tumor and optic nerve may share the same blood supply [
65]; this makes SRS a good choice. When using radiation, a balance between stopping tumor progression and preventing radiation-induced optic neuropathy (RION) is challenging, as both may lead to compromised vision. Specific locations of tumors such as those of the clinoid process, cavernous sinus, and parasellar region may cause compression of the optic apparatus, but these will be discussed in independent sections when possible.
A systematic review done in 2018 by Hénaux and colleagues reported visual outcomes for the surgical management of meningiomas compressing the optic nerve [
64]. Their review consisted of 317 patient cases of meningiomas compressing the optic nerve from 2004-2012. Residual tumor was described in 61 patients, and 23 received postoperative radiotherapy. Researchers investigated how location-specific compression along the course of the optic nerve should influence operative decision-making. Their analysis revealed differences in visual outcome associated with meningiomas in three anatomical subdivisions (intraorbital, optic canal, and intracranial space). Postoperative visual improvement was found to be 50% with meningiomas of the intracranial segment, 31% with optic canal involvement, and 11% with intraorbital location [
64].
Considering outcomes of intraorbital lesions, authors suggested avoiding surgical resection of optic nerve sheath meningiomas and considering radiotherapy as a lower risk alternative. Authors additionally cautioned that mechanical strains of the orbital canal may inhibit radiotherapy effectiveness; they suggested opening the orbital canal over the entire length of the optic nerve offers the best conditions for visual recovery. Radiation therapy may then be proposed while observing for further development of the remnant [
64].
Peters and colleagues performed an extensive systematic review and meta-analysis in 2023 on both single fraction SRS and hypofractionated radiosurgery. The majority of analyzed studies focused on hypofractionated radiosurgery (4/6) and the remaining single fraction SRS. In total, 865 patients were analyzed with 427 receiving hypofractionated and 438 single fraction. Tumor control rate was near equivalent between modalities with 95.1% for single fraction and 95.6% for hypofractionated [
63]. The most used hypofractionation regiment within the review was 25 Gy in 5 fractions. Visual stability was slightly better in the hypofractionated group (90.4% single fraction, 95.6% hypofractionated). The single fraction showed better visual improvement (29.4% vs 22.7%), but also higher rates of visual decline (9.6% vs 5.1%). Authors concluded SRS was effective and safe, with both hypofractionated and single fraction SRS considered viable options.
One of the largest studies conducted was a retrospective analysis of 438 perioptic meningioma patients from 12 institutions, done by Bunevicius et al. The majority of patients (92.5%) underwent single fraction SRS with a median marginal dose of 12 Gy. Median maximal dose to the optic apparatus was 8.5 Gy. Their series found that single-session SRS demonstrated superior tumor control when compared to 2-5 fractions, although the fraction group was small, limiting generalizability of the findings. The study showed that 5 and 10-year progression-free survival was 96% and 89%, respectively [
66]. A dose ≥ 10 Gy to the optic apparatus and tumor progression were independent predictors of post-SRS visual decline.
Another large multicenter retrospective analysis was done by Asuzu et al. Their study included 328 patients at 11 institutions who all had meningiomas in direct contact with the optic apparatus. More specifically, there were 107 patients with tuberculum meningiomas, 126 with clinoidal meningiomas, and 105 with cavernous sinus meningiomas. The majority (64.6%) of patients underwent SRS as the initial treatment modality. Results showed a tumor control rate of 91%, and no difference in visual outcomes between patients whose tumors contacted the optic nerves compared with chiasm or anterior optic tract contact [
67]. Authors also found that duration of symptoms predicted failure to respond to SRS, and it was unrelated to lesion size. Based on these findings, authors suggested earlier intervention could improve the odds of tumor response and that for patients with symptomatic compressive optic neuropathy, pre-SRS decompressive surgery should be considered. Within their study 93% of patients had single session SRS, the rest fractionated therapy. Their results found no association between fractionation, visual dicline, nor tumor response [
67].
Chen et al. retrospectively analyzed 60 patients with perioptic tumors from 2007 to 2020. Of the 60 tumors, 53 were meningiomas and 7 schwannomas. Patients received hypofractionated radiosurgery with 6-7 Gy three times daily for three consecutive days (mean optic apparatus dose 6.05 Gy). Observed tumor control rates at 1, 3, 5, 8, and 13-year follow-up were 98.3%, 93.4%, 90.6%, 88.4%, and 88.4% [
68]. The risk of radiation-induced optic neuropathy was higher in patients with pre-existing optic nerve compression, concurrent chemotherapy, and prior radiotherapy. Based on their follow-up results, authors concluded that if tumor growth is noted within 6 months, further close monitoring is required, and if enlargement continues >2 years, further intervention may be required. Overall hypofractionated dosage over three days provided adequate control without significant side effects.
Wei and colleagues performed a retrospective analysis of 7 patients with recurrent optic nerve sheath meningiomas following gross surgical resection. The median maximal radiation to the optic nerve was 6.5 Gy (1.9-8.1 Gy). Their results showed that salvage SRS resulted in tumor control and visual preservation in 5/7 patients [
69].
Su et al. performed volume-staged GKS using 2-3 fractions. In the first stage, a higher median marginal dose of 13.5 Gy was applied to the basal portion of the tumor. The second stage involved a smaller 9 Gy isodose to the superior portion proximal to the optic apparatus. Authors referred to this as snowman-shape volumetric treatment. All four patients demonstrated a 34-46% reduction in tumor volume and improvement of neurological defects to various degrees [
70].
El-Shehaby and colleagues performed a retrospective analysis of 175 patients with large (≥10 cm
3) perioptic meningiomas. Patients received single-session SRS with a median prescription dose of 12 Gy. The tumor control rate was 92%, and 97% had better or stable visual outcomes [
71].
When treating primary optic nerve sheath meningiomas radiation has achieved superior results in stabilizing or improving vision compared with surgery or observation [
72]. An additional technology is being used to treat primary optic nerve sheath meningiomas, intensity modulated radiotherapy (IMRT). IMRT is an advanced form of three-dimensional conformal radiation therapy that has been investigated for meningioma treatment since the mid 1990s [
73]. Using IMRT allows for an optimized dose distribution with sparring of surrounding normal tissues. Prospective studies comparing IMRT with SRS/fSRT are warranted and IMRT is not widely available at this time [
72].
Reviewed studies suggest that both fractionated and single-session radiosurgical intervention offer high control and acceptable complication rates for perioptic meningiomas. This holds true for both upfront and salvage SRS for post-surgical progression. Considering the duration of symptoms predicted SRS response failure regardless of tumor size [
67], earlier intervention may be of benefit. Fractionated therapy has been shown to have a smaller risk of radiation-induced optic neuropathy (0%-6.7%) when compared to single session therapy which approaches 13% [
74]. Additional studies are warranted to determine optimal treatment timing and fractionation dosage.
4.6. Petroclival
The petroclival meningioma (PCM) accounts for 11.42% of all posterior fossa meningiomas and is located in a particularly difficult area [
9]. PCMs typically arise from the upper two-thirds of the clivus, directly adjacent to the brainstem, basilar artery, perforating arteries, and multiple cranial nerves [
75]. Pure petroclival meningiomas are not considered to belong to the parasellar region, although cavernous sinus meningiomas commonly extend to the parasellar region [
18]. Extending cavernous sinus meningiomas involving the petroclival region should be named caverno-petroclival or spheno-cavernous-petroclival [
18]. This section will be addressing pure petroclival meningiomas.
With the proximity of vital neurological and vascular structures, PCMs are one of the most challenging skull base tumors to treat [
19]. Microsurgical resection has morbidity ranging from 28% to 76% with incomplete resection in most patients [
21]. Due to a lack of published literature, the use of SRS as treatment for petroclival meningiomas remains relatively undetermined [
19]. Similar to other anatomical locations, the optimal management of asymptomatic PCMs remains controversial [
20].
A systematic review of petroclival meningioma patients undergoing SRS was conducted by Wijaya and colleagues in 2022 [
19]. Their study was the first to determine the safety and efficacy of SRS treatment of PCMs and consisted of synthesized evidence from ten studies including 719 patients. Their results showed an overall tumor control rate at a mean of 98.8% (85%-100%) and a low number of complications following SRS [
19]. Although authors suggest a significant selection bias likely exists between primary and adjuvant therapy that may impact results.
Another systematic review in 2022 was conducted by Alamer et al. They reviewed seven articles involving 722 cases of petroclival meningiomas. Their review found that a mean marginal tumor dose of 13.5 Gy resulted in 5 and 10-year progression-free survival of 91–100% and 69.6–89.9%, respectively [
21]. Authors compared outcomes between primary (61.9%) and adjuvant radiosurgery (38.1%) and found that primary SRS reported higher rates of tumor control (94.3% vs. 88.2%) and fewer SRS-related complications (3.7% vs. 10.3%) [
21]. Authors concluded primary SRS was effective for smaller lesions without symptoms related to mass effect.
Mantziaris and colleagues performed a retrospective, international, multicenter study on asymptomatic PCMs using data from the IMPASSE study. They found that upfront SRS for asymptomatic, petroclival region meningiomas achieved local tumor control in all 79 patients with <5% experiencing new neurological deficits [
20]. Authors concluded SRS could be considered at the diagnosis of PCMs and recommended if any growth is noted during active surveillance [
20].
Overall reviewed studies provide evidence that radiosurgery provides excellent long-term control and minimal complications for surgically challenging petroclival meningiomas. SRS has roles in both upfront primary intervention and adjuvant treatment for petroclival meningiomas. We were unable to find any data available on the efficacy of fractionated dosing for PCMs.
4.7. Clinoidal
Clinoidal meningiomas comprise less than 10% of supratentorial meningiomas [
76] and represent a challenge for neurosurgeons. The clinoidal area is among the areas with the highest neurosurgical complexity, with resection associated with significant morbidity and mortality [
34]. Group I clinoidal meningiomas grow from the inferior portion of the clinoidal process with frequent ICA attachment, making resection exceedingly difficult. Group II arises from the superior and lateral face of the anterior clinoid process; preservation of the arachnoid layer and the carotid cistern facilitate tumor dissection. Group III arises from the optic canal from the medial side of the anterior clinoid process [
18,
34]. These tumors are likely to adhere to vessels such as the middle cerebral artery, anterior cerebral artery, and internal carotid artery [
34,
76]. Rates of subtotal resection and subsequent progression are high (41% and 38% respectively), making SRS a valuable option [
76].
Prior to a study done by Bunevicius et al., the majority of published studies have not specifically considered outcomes of clinoidal meningiomas [
76]. Their multi-institutional study included 270 patients treated with SRS for clinoidal meningiomas. The majority of patients underwent single-session treatment, with only 15 having hypofractionated sessions (2-5). Hypofractionated SRS is considered when the growths are directly abutting optic structures and/or large tumors [
76]. The median prescription dose was 12 Gy with a maximum of 8.5 Gy to the optic apparatus. Results showed overall tumor control in 93% of patients without significant difference between those treated with single fraction vs 2-5 fractions of radiation [
76]. Visual decline was associated with a dosage ≥ 10 Gy to the optic apparatus and the presence of visual impairment prior to SRS.
There were two studies that investigated anterior clinoid process (ACP) meningiomas specifically [
77,
78]. Prior to this publication, ACP meningiomas were included in studies for sphenoid meningiomas instead of being given their own individual analysis.
Demiral and colleagues published on 19 patients who all underwent hypofractionated radiotherapy for ACP meningiomas. Volumetric modulated arc therapy was performed with a 6MV linear accelerator. All patients were given 25 Gy total dosage in 5 fractions over 5 days. Results showed local control at 1 and 3 years was 100% and 89.4% [
77]. This was the first study to investigate fractionated radiosurgery on ACP meningiomas.
Akyoldas et al. performed a retrospective analysis on 61 patients receiving GKRS from 2008-2016. A median marginal dose of 12 Gy was administered and the majority received radiosurgical intervention as primary treatment (80%). Their results showed 100% tumor control and 61% having tumor regression [
78]. Visual impairment was the only preoperative deficit in this cohort, and 55% of those patients improved. This was the first study to investigate single-fraction radiosurgery on ACP meningiomas.
There was a paucity of evidence looking directly at clinoidal meningiomas prior to the noted studies. Collectively, the studies described above demonstrated that radiation for clinoidal meningiomas provides excellent tumor control with acceptable complication rates. Both single fraction and fractionated intervention were effective on clinoidal meningiomas.
4.8. Intraventricular
Intraventricular meningiomas (IVMs) are rare, accounting for only 0.3-5% of all meningiomas [
35]. The ventricular system comprises a series of cerebrospinal fluid (CSF)-filled cavities deep within the brain that cushion the surrounding gray and white matter. Due to their deep locations, IVMs are among the most challenging tumors to surgically manage, with significant treatment-associated morbidity [
35,
79]. The surgical approach requires traversing the cerebral cortex, avoiding critical nerve tracts adjacent to the tumor and along the trajectory, and controlling homeostasis deep inside the brain [
35]. IVMs may be closely associated with vital structures such as the thalamus, hypothalamus, basal ganglia, internal capsule, and brainstem [
80]. They are most commonly located in the lateral ventricle, followed by the third and fourth ventricles [
79]. Due to their rarity, there remains a paucity of evidence available on the use of SRS to treat IVMs.
Kim et al. published the first study of SRS for IVMs in 2009 with their case series of 9 patients. Using a median marginal dose of 16 Gy (range 14.0-22.5), they achieved tumor control in 7 out of 9 patients within the study. The authors concluded that SRS should be considered an option for small intraventricular tumors with minimal symptoms and can serve as an alternative to repeat surgery for residual or remnant tumors [
81].
Christ et al. published a pooled analysis of all prior studies in 2022 in addition to their case series of patients undergoing primary SRS for IVMs. Their addition was the largest case series of those reviewed, with 33 patients included. Prior studies published had between 2 and 19 patients comparatively [
79,
80,
81,
82,
83]. They used a mean marginal dose of 13.9 Gy to treat mostly lateral ventricular IVMs (97%). Results covered a long-term median follow-up of 7.9 years. Analysis of their case series showed 66% of patients either had a partial or minor response, and the rest had stable disease [
84]. The pooled analysis also demonstrated local control in 91% of all patients.
Daza-Ovalle and colleagues performed a literature review alongside their case series in 2022, updating the field on IVM SRS results. In Daza-Ovalle’s recent case series, symptom control was achieved in 18 out of 19 patients after single-session GKRS. The progression-free survival was 95% at five years and 85% at ten years [
79], results comparable to a Simpson Grade 1 resection. Authors concluded that early SRS intervention should be considered for primary management of small to medium IVMs and adjuvant for residual IVMs.
In 2023, Umekawa and colleagues published their analysis of institutional data over thirty years. Their retrospective observational study included 11 patients with 12 IVMs treated with SRS. A median marginal dose of 16 Gy was given (range 9-18). Results demonstrated tumor control of 100% (follow-up period 52 months) with 55% decreasing in size [
35]. Notably, their cohort included patients with NF2 related IVMs and demonstrated efficacy regardless of NF2 mutation status.
There were three additional smaller studies done by Nundkumar (n=2), Mindermann (n=5), and Samanci (n=6) between 2013 and 2020. The mean marginal dose ranged from 12-16 Gy, and tumor control was 100% for all of the studies [
80,
82,
83].
Numerous studies were available for IVMs; however, due to their rarity, all were small in patient size. Overall, the studies reviewed demonstrated excellent tumor control with an acceptable complication rate for IVMs. Compared to other anatomical sites reviewed, there were fewer total cases available to draw data from, and more research in larger studies is warranted. No studies mentioned the use of fractionated radiation for IVM treatment.
4.9. Olfactory Groove
Olfactory groove meningiomas comprise 10% of intracranial meningiomas; collectively, they describe tumors arising from midline anterior cranial fossa structures such as the crista galli, cribriform plate, olfactory groove, and planum sphenoidale [
24]. Anosmia after olfactory groove meningioma resection can cause significant disability. Rates of olfactory preservation are between 50% and 95% with surgical resection [
85]. Radiosurgical intervention is a less invasive option that may promote olfactory preservation.
A study by Gande and colleagues in 2014 was the first to investigate subjective olfactory function after gamma knife SRS. They performed a retrospective analysis of 41 patients who underwent SRS for anterior fossa cranial tumors with more than 6 months of follow-up available. Radiosurgery was performed as primary management for 22 patients and as adjuvant therapy for the remaining 19 patients. Two patients underwent prior fractionated external beam radiation therapy. The mean marginal dose was 13 Gy, and the cumulative progression-free tumor control rates were 97% at 1 year and 95% at 2, 10, and 20 years [
24]. Post-SRS, 27 (66%) patients reported intact olfaction, nine (22%) reported partial deficits, and five (12%) patients had no improvement from complete anosmia [
24].
A large multi-institutional study was conducted by Bunevicius and colleagues in 2021 using pooled results from 20 institutions participating in the Radiosurgery Research Foundation. They found 278 patients with more than 6 months of follow-up who underwent SRS with a median dose of 12 Gy. Forty-three percent of patients had partial or marginal response, 54% had stable disease, and 3% experienced progression [
22]. After the SRS, olfaction remained stable, improved, or deteriorated in 90%, 8%, and 2% of patients, respectively [
22].
Another single-center retrospective cohort study was performed by Liu et al. They reviewed 13 patients treated with single-session SRS (n=5), hypofractionated stereotactic radiotherapy (HSRT) (n=6), and fractionated stereotactic radiotherapy (FSRT) (n=2). SRS was one dose of 10 Gy, HSRT was 25 Gy in 5 fractions, and FSRT was 54 Gy in 30 fractions. The median prescribed dose was 14.8 Gy for SRS, 27.3 Gy for HSRT, and 50.2 Gy for FSRT. Overall median maximal dose was 32.27 Gy. They achieved 100% regional control rate in 12 out of 13 patients, and 6 out of 12 had reduced tumor volume [
86]. Every patient included in the study had stable vision and olfactory function at the time of the last follow-up (median follow-up 48 months). Authors highlighted the advantages of using fractionated regimens for cranial nerve sparing when tumors are spatially associated with the optic apparatus or >10cm
3. Zaorsky et al. reviewed 13 patients treated with 52.8 Gy over 25 fractions or 16.1 Gy in a single fraction. Authors concluded that both FSRT and SRS have a higher likelihood of preserving or improving cranial nerve function and decreasing morbidity compared with operative intervention [
87].
Radiosurgical intervention for olfactory groove meningiomas showed comparable preservation of olfaction with surgical intervention and acceptable control rates among all studies reviewed. Fractionated and unfractionated radiation are both viable options that may offer better preservation or improvement of cranial nerve function.
4.10. Cerebellopontine
Cerebellopontine angle (CPA) meningiomas comprise 1% of all intracranial meningiomas [
88], but they are the second most common tumor located at the CPA after vestibular schwannomas [
36]. These tumors arise from the petrous face of the temporal bone and are categorized into anterior, middle, or posterior based on their relationship with the internal auditory meatus [
89]. This area is compactly filled with neurovascular elements, including cranial nerves IV to XII, vertebral, and basilar arteries [
90]. Meningiomas are the minority of tumors at the CPA, with vestibular schwannomas being the most common tumor in this area. When compared directly, it was observed that hearing preservation was better for CPA meningiomas when compared to vestibular schwannomas [
91]. There has been research showing increased post-surgical facial paralysis in tumors located anterior (60%) or below (50%) the internal auditory canal (IAC) compared with those located posterior or superior (15%). Considering the anatomical risk, radiosurgical intervention is a viable option to avoid surgical morbidity.
Gendreau and colleagues performed a systematic review and meta-analysis analyzing surgical outcomes and radiation dosages. Their review found 6 studies including 406 patients who underwent radiosurgery for CPA meningiomas. With a median marginal dose of 12-15 Gy, they found minimal cranial nerve complications while having an overall tumor control rate of 95.6% [
36]. Postprocedural tumor regression was associated with a median prescription dose of >13 Gy.
Another retrospective descriptive analysis was performed by Garcia et al. on 80 patients with CPA meningiomas from 2001-2014. SRS was the primary treatment in the majority of the cases (83.7%). All patients received single-dose treatment, and the median coverage dose was 14 Gy. After an extensive 12-year follow-up, tumor control rates were found to be 95% [
92]. Radiation dose to the brainstem was related to the symptomatic deterioration in four patients.
Park and colleagues investigated 74 patients with CPA meningiomas from 1990-2010. Patients underwent single-session SRS with a median prescription dose of 13 Gy. Tumor extension into the IAC was related to preexisting hearing loss (p=0.01). The median radiation dose to the cochlea was 3.2 Gy in patients with unchanged hearing and 2.5 Gy in patients with improved hearing. Control rates were found to be 97% [
93], although the average follow-up time of 40 months was comparatively shorter than Garcia’s study. Their results additionally showed that patients with trigeminal neuralgia related to CPA tumors are more likely to experience pain worsening over long-term follow-up.
Another large retrospective review was performed by Jahanbakhshi and colleagues on 93 CPA meningioma patients. Patients received a median marginal dose of 13.6 Gy. Progression-free survival was seen in 96% of patients [
90]. Their median tumor volume was 6 cm
3, twice as large as Park’s 3 cm
3 median tumor volume and Garcia’s average tumor volume of 3.12 cm
3. Their results demonstrated adequate tumor control rates and minimal adverse radiation events even with tumors of larger size. They additionally found that male sex was associated with lower progression-free survival and tumor volume ≥ 8.5cc was associated with worse symptomatic outcomes.
Overall, there was a larger number of patients to draw conclusions from for CPA meningiomas compared with other anatomical regions. Studies reviewed indicate that radiation therapy is effective at long-term control with acceptable complication rates. Tumors located anterior or below the IAC, with a higher risk of facial paralysis, may benefit from radiotherapy. A median dose of >13 Gy is recommended for tumor regression, and radiation to the brainstem should be avoided at all costs.
4.11. Foramen Magnum
Foramen magnum meningiomas (FMM) represent only 2-3% of intracranial meningiomas; hence, treatment outcomes and optimal management paradigms remain poorly defined [
28]. Surgical intervention is extremely challenging for neurosurgeons due to their intimate proximity to critical structures, including the brainstem, lower cranial nerves, medulla, vertebral artery, and its branches [
26]. Anterior and anterolateral FMMs carry the highest surgical risk and are the most common location for FMMs [
28]. Considering the highly eloquent location of FMMs, radiation may be a viable alternative for primary management in addition to handling residual/recurrent tumors.
Karras and colleagues performed the first systematic review of the literature on FMMs in 2022, contributing their own institutional knowledge alongside. Their review found 9 patients from the authors' institution and 165 patients accrued from four other prior publications. SRS was utilized as the primary therapy in 63.6% of patients and as salvage (21.8%) or adjuvant (14.5%) therapy for the remaining patients [
25]. Clinical stability and local control at last follow-up were achieved in 98.8% and 97.0% of patients, respectively [
25]. Researchers concluded SRS may be a viable primary option for symptomatic lesions without significant brainstem compression and for higher-risk surgical patients. They stated larger tumors (>35 mm in diameter) should undergo maximal safe resection, and SRS strongly considered in cases of subtotal resection [
25].
Akyoldas and colleagues performed the largest from a single institution and longest follow-up period to date on FMM in 2021. Researchers performed a retrospective analysis of 37 FMM patients undergoing gamma knife radiosurgery, 12 of which had prior microsurgical resection. Their results showed GKRS provided excellent tumor control with a rate of 97.3% during a median radiological follow-up of 7 years [
26]. Results from their cohort demonstrated that GKRS is an effective and safe treatment for patients with both primary or recurrent/residual FMM [
26].
Recent publications suggest despite historic underutilization of SRS for FMMs, it is recently gaining traction and recognition as an important treatment option [
25]. Both the systematic review by Karras and institutional analysis with long-term follow-up by Akyoldas suggest radiation is a viable option for long-term control.
5. Discussion
The gold standard for meningioma treatment remains complete surgical removal of the tumor and adjacent dura/bone involved. Advances in neurosurgical technique and technology permit better gross total resections than ever. However, as radiation therapy technology has advanced, one has to balance the ability to achieve a total resection against the associated morbidity of the particular procedure. Alongside advances in operative technique, our understanding of radiation efficacy and application builds with additional publications. For meningiomas in both high and low-risk locations, the role radiation plays in management is continually evolving. It is increasingly clear that a “hybrid” approach using resection followed by RT can significantly reduce morbidity in certain high-risk situations. Adaptive hybrid surgery analysis (AHSA) is an intraoperative tool used for automatic assessment of tumor properties. The software provides immediate assessment of adjuvant radiation coverage options for single fraction and hypofractionated radiation based on an IMRT algorithm [
94].
5.1. Observation, Radiation, or Surgery
The course of treatment remains dependent on individual patient characteristics, including age, life expectancy, location-specific risk, and tumor characteristics. For lesions that are symptomatic, grade II or III, or in dangerous locations, observation is an unreasonable strategy, and intervention of some form is recommended [
6]. Gross total resection remains the gold standard for symptomatic accessible lesions in patients who can tolerate surgery. When discussing management for small asymptomatic lesions, there is less clarity on the optimal management. A meta-analysis of 27 studies was performed to formulate practice guidelines for non-cavernous sinus benign intracranial meningiomas in 2020 [
95]. Analysis showed although the current literature lacks level I and II evidence supporting SRS for benign intracranial meningiomas, the large quantatiy of level III studies justifies recommending SRS as an effective primary treatment option for grade I meningiomas (recommendation level II) [
95].
Incidentally discovered meningiomas now account for 30% of newly diagnosed intracranial meningiomas [
96]. Conventional wisdom has favored observation over radiosurgical intervention with small and asymptomatic meningiomas. This approach is favorable for elderly patients, those with significant comorbidities, and poor performance status [
65]. A recent meta-analysis done on 2,130 patients with incidentally discovered meningiomas showed initial management strategy at diagnosis was active monitoring (50.7%), surgery (27.3%), and stereotactic radiosurgery (22.0%) [
96].
The watch-and-scan paradigm is being challenged as further studies suggest better tumor control and fewer side effects associated with radiosurgical intervention [
97]. Upfront SRS may be a better option for younger patients where tumor progression will inevitably require active treatment [
65]. Additionally, some studies have suggested that earlier intervention, at younger ages, is favorable for progression-free survival [
10]. As further research continues to amass favoring SRS for long-term control, we may see guidelines change in the future of meningioma management.
Although younger patients are good candidates for upfront SRS intervention, certain patient populations may be at higher risks of developing iatrogenic complications. One of the paradoxical risks of radiation treatment of meningiomas is radiation-induced meningiomas growing as a result of treatment. There is limited data on the treatment of radiation-induced meningiomas, but they are more often atypical and highly proliferative [
65].
Recent studies have uncovered gene mutations that may contribute to the development of radiation-induced meningiomas. The presence of a mutation in Cycline D1, p16, 1p, and 22q were more commonly associated with radiation-induced meningiomas [
6]. As we further our understanding of pre-existing risk factors for radiation-induced meningioma development, it may be used to suggest surgical intervention or observation over radiation. This is especially true in younger patients or those who are likely to have multiple tumors (NF2 patients).
Advances in predicting an individual tumor’s growth trajectory would aid in choosing between SRS and observation. Currently, tumor size ≥3 cm, peritumoral edema, young age (<60 years old), lack of calcifications, and lesion hyperintensity on T2-weighted MRI were found to be significantly related to symptomatic progression [
65]. A risk calculator developed to determine progression risk based on tumor location, molecular markers, radiogenomics, and individual patient comorbidities would be of high clinical utility. There are studies investigating MRI-based machine-learning tools for diagnosis, prognostication, and automatic segmentation of meningiomas; these tools are in the early stage of research but may serve as radiomic-based predictive models of aggressiveness in the future [
98].
5.2. Radiation Dose and Technique
Radiation dosages should be optimized to reduce postprocedural morbidity while also achieving high rates of tumor control [
36]. Commonly used dose regimens for WHO grade I, II, and III meningiomas treated with single-fraction SRS are 12–16 Gy, 16–20 Gy, and 18–24 Gy, respectively [
63]. Although, in patient’s with recurrent WHO Grade II meningiomas the optimal modality of radiation is not well-established [
99]. The most common radiation doses, techniques, and planning used in studies reviewed within prior sections are reported in
Table 2 for comparison. In general SRS and HSRT are not regarded as appropriate treatment for WHO grade III meningiomas; larger resection margins are typically needed to avoid recurrance. Both WHO Grade II and III lesions typically require a combination of maximum surgical resection and radiation [
100].
Radiosensitive structures such as the optic apparatus, brainstem, cochlear nerve, and other cranial nerves should be taken into consideration. Damage to the optic apparatus may cause radiation-induced optic neuropathy, which may manifest months or years later with painless visual loss, changes in color vision, and pupillary abnormalities [
63]. Risk of developing clinically significant radiation optic neuropathy was found to be 1.1% for those who received ≤ 12 Gy [
101]. Using fractionated therapy doses as high as 54 Gy total, delivereed in 6-30 fractions with 1.8-4 Gy per fraction, has demonstrated minimal visual deterioration (1.5% of patients) [
74].
Fractionating the radiation intends to decrease the risk of radiation damage to normal tissues and allow for increased dose delivery to larger tumor volumes. The fractionation decreases risk of complications but does not decrease the overall rdation exposure. Conventionally fractionated radiation takes advantage of differing cell repair potentials between healthy tissue and tumor, allowing healthy tissue to repair between spread-out sessions [
102]. Hypofractionated and single-session SRS require increased conformity and dose gradient steepness to avoid damage to radiosensitive tissues [
102]. Typically for grade I meningioma the recommended fractionated dose is 50.4 Gy – 54 Gy in 1.8 Gy per fraction.
For region-specific treatments discussed within this review, there was more evidence available for single-fraction radiosurgery than HSRT . Current guidelines support the use of conventionally fractionated radiotherapy over SRS for WHO grade II and III meningiomas; for WHO grade I, either SRS or conventionally fractionated radiotherapy is a suggested option [
16]. Other guidelines suggest fractionated radiation is beneficial in tumors > 7.5 cc and <3–5 mm from radiosensitive structures [
102].
A systematic review and meta-analysis including 1,736 meningioma patients compared single-session SRS, hypofractionated radiation, and conventionally-fractionated radiation. Their results showed both were safe options, but fractionation had superior radiographic control and a lower incidence of symptomatic decline and edema [
103].
Another publication compared conventionally fractionated with hypofractionated radiation among 341 patients with skull base meningiomas. Pooled multicenter analysis demonstrated no difference in control rates between fractionation patterns and comparable rates to SRS [
102].
Table 2.
This table shows the radiation parameters reportedly used among anatomical regions. Any region with reported fractionated dosing was also included and denoted N/A.
Table 2.
This table shows the radiation parameters reportedly used among anatomical regions. Any region with reported fractionated dosing was also included and denoted N/A.
| Anatomical Location |
Mean Marginal Dose (Single Session) |
Mean Marginal Dose (Fractionated) |
| Parafalcine / Parasagittal |
14-18 Gy [45] |
N/A |
| Convexity |
13.26 Gy [48] |
25 Gy (5 fractions) or 50.4 Gy (28 fractions) [104] |
| Cavernous Sinus |
12-14 Gy [55] |
51.2 Gy (15.5 sessions) [105] |
| Parasellar |
12-14 Gy [59,61] |
25-30 Gy (5-6 sessions) [33] |
| Perioptic |
10-12 Gy [63] |
25 Gy (5 sessions) [63] |
| Petroclival |
15 Gy [19] |
N/A |
| Clinoidal |
12 Gy [76,78] |
25 Gy (5 sessions) [77] |
| Intraventricular |
12-16 Gy [79] |
N/A |
| Olfactory Groove |
12 Gy [22] |
25 Gy (5 sessions) [86] |
| Cerebellopontine Angle |
12-15 Gy [36] |
N/A |
| Foramen Magnum |
12-14 Gy [25] |
20 Gy (5 sessions) [25] |
5.3. Predicting Response to Radiation
Understanding which meningiomas are responsive to radiation enables informed surgical decision-making. WHO Grade I meningiomas demonstrate a much better response to radiation than Grade II or III meningiomas [
106]. The only method currently available for a definitive diagnosis of WHO grade is a tissue biopsy, and the question of which patients warrant an upfront biopsy has yet to be answered [
16,
54]. Sughrue et al. suggested that patients with immunosuppression, nasopharyngeal or infratemporal fossa extension, rapid symptom onset, cavernous sinus syndrome, or other synchronous lesions are indications for a tissue diagnosis [
54].
The standard postoperative treatment for intermediate risk meningiomas (WHO Grade II or recurrent Grade I) is conventionally fractionated radiotherapy irrespective of resection extent [
107]. Radiotherapy inluding remaining enhancing tumor and an additional volumetric expansion of 10mm accounts for any microscopic subclincal disease extension [
107]. For high risk meningiomas (Grade III, recurrent Grade II) trials have investigated radiation expansion up to 1-2cm margins [
108].
Trial NRG-BN003 (NCT 03180268) suggests that completely resected Grade II meningiomas may be watched without adjuvant radiation [
109]. In a prospective observational study of patients with Grade II meningiomas who underwent gross total resection, 2/12 patients were fond to have progression. Both were succesfully salvaged with focal fractionated radiosurgery after recurrence.
Variability of oncologic outcomes exists within each grade; even Grade I meningiomas have certain subsets that will recur [
110]. Postoperative radiation practice patterns for subtotally resected WHO Grade I or totally resected Grade II meningiomas also remain varied among providers [
111]. Prior research has suggested use of recursive partitioning analysis (RPA) to select optimal candidates for SRS therapy in patients with Grade II meningiomas [
112]. Analysis using RPA of 230 patients with Grade II meningiomas at 12 institutions demonstrated that patients age ≤ 50 years old, with up to one prior resection, and no prior radiation therapy are within a good-prognostic group and are likely appropriate candidates for adjuvant SRS [
112].
The WHO grades meningiomas based on results from tissue sample histology and tumor features. In the 2016 updated WHO classification, tumors with a mitotic count greater than four or brain invasion are classified as WHO Grade II, or an atypical meningioma [
113]. The update in 2021 included molecular factors that significantly shorten survival and increase recurrence, such as meningiomas with a telomerase reverse transcriptase (TERT) promoter mutation or homozygous deletions in the cell cycle regulator genes CDKN2A and/or CDKN2B [
6]. Presence of either TERT or CDK2A/2B will earn a Grade III classification. Future research is warranted to develop clinical biomarkers for both progression and prognostication of meningioma behavior.
5.4. Timing of Radiation Intervention
Radiation can be delivered as monotherapy, adjuvant therapy following resection, or after recurrence. The timing of SRS may have an impact on tumor control. The large multicenter retrospective analysis conducted by Asuzu et al. suggested that when upfront SRS is used, earlier intervention from diagnosis could improve the odds of tumor response [
67]. Another previous study of 238 WHO Grade I meningiomas investigated the timing of post-resection SRS on symptom control. Authors found excellent long-term control and concluded that earlier radiosurgery was associated with superior symptomatic improvement (p = 0.007) [
114]. Large prospective multicenter studies are warranted to determine the optimal timing for SRS intervention.
5.5. Future Treatment Horizons
Considering meningiomas reside outside of the BBB, they are accessible to systemic therapies and circulating immune cells [
110,
115]. Currently, systemic therapy is regarded as having minimal role in the management of meningiomas. There has been evidence suggesting links between lymphocyte infiltration and mutational burden in meningiomas; authors have suggested a role for adjuvant immunotherapy in subsets of amenable tumors that cannot be grossly resected [
110]. There is strong evidence suggesting PD-L1 correlates with WHO grade and possible tumor recurrence [
115]. There are ongoing clinical trials investigating PD-1 checkpoint inhibitors (NCT02648997, NCT03173950, NCT03604978, NCT03279692, NCT03016091, NCT04659811) and PD-L1 inhibitors (NCT03267836). There are also a wide variety of molecular mutations being targeted for systemic therapy [
30]. Classic cytotoxic agents, somatostatin analogs, and anti-hormone therapies have shown limited efficacy thus far; tyrosine kinase inhibitors and monoclonal antibodies targeting angiogenesis have shown promise in small phase II trials [
116].
6. Conclusions
Meningioma management is individualized based on patient comorbidities, tumor location/characteristics, symptomatic burden, and patient age. The gold standard of care remains gross total resection, including bone and dura. SRS publications have demonstrated an expanded utility in meningioma management. In addition to SRS’s established role for surgically inaccessible, recurrent, and highly selected Grade II meningiomas, the use of SRS as upfront management for small asymptomatic meningiomas is increasingly investigated. As evidence becomes more regionally specific, we are better enabled to make informed surgical decisions. For all sub-regions reported, radiosurgical intervention resulted in high tumor control rates and acceptable adverse radiation events. Depending on anatomical location, SRS may be a key tool to complement open surgical intervention or an alternative option as monotherapy.
Author Contributions
Conceptualization, M.G., B.T., and D.B.; resources, M.G., B.T., and D.B.; writing—original draft preparation, M.G.; writing—review and editing, M.G., B.T., D.B., and S.L; supervision, D.B.; All authors have read and agreed to the published version of the manuscript.
Funding
Funding: The work was supported by Donna and Kenneth R. Peak, the Kenneth R. Peak Foundation, the John S. Dunn Foundation, the Taub Foundation, the Blanche Green Fund of the Pauline Sterne Wolff Memorial Foundation, The Methodist Hospital Foundation, the Veralan Foundation and donations in honor of Will McKone. The John S. Dunn Foundation also supports the Distinguished Professorship of MS.
Acknowledgments
We honor contributions made in honor of our patients. We are grateful for the many patients who we have been privileged to care for who have provided us with knowledge, experience, and insight into the diagnosis and treatment of meningiomas.
Conflicts of Interest
Simon S Lo, Elekta AB, member of ICON Gamma Knife Expert Group and research support; Kuni Foundation, research funding, Hutchinson Center as Lead Academic Participating Site; UG1 CA 233328; Japanese Society for Radiation Oncology, travel expenses; American College of Radiology, Councilor on behalf of American Radium Society and Chair of CARROS Nominating Committee; Radiosurgery Society, Board of Directors and National Medical Director of the Distinction in Practice in Stereotactic Radiotherapy Accreditation Program. Other authors declare no conflicts of interest.
References
- Buerki RA, Horbinski CM, Kruser T, Horowitz PM, James CD, Lukas RV. An overview of meningiomas. Future Oncol. 2018;14(21):2161-2177. [CrossRef]
- Bhat AR, Wani MA, Kirmani AR, Ramzan AU. Histological-subtypes and anatomical location correlated in meningeal brain tumors (meningiomas). J Neurosci Rural Pract. 2014;5(3):244-249. [CrossRef]
- Ostrom QT, Francis SS, Barnholtz-Sloan JS. Epidemiology of Brain and Other CNS Tumors. Curr Neurol Neurosci Rep. 2021;21(12):68. [CrossRef]
- Singh V, Bluestone A, Chen D, Shah A, Linden C, Bangiyev L. Meningiomas: Variations and Clinical Presentations.
- Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. [CrossRef]
- Yarabarla V, Mylarapu A, Han TJ, McGovern SL, Raza SM, Beckham TH. Intracranial meningiomas: an update of the 2021 World Health Organization classifications and review of management with a focus on radiation therapy. Front Oncol. 2023;13:1137849. [CrossRef]
- Agarwal V, McCutcheon BA, Hughes JD, et al. Trends in Management of Intracranial Meningiomas: Analysis of 49,921 Cases from Modern Cohort. World Neurosurg. 2017;106:145-151. [CrossRef]
- Pinzi V, Biagioli E, Roberto A, et al. Radiosurgery for intracranial meningiomas: A systematic review and meta-analysis. Crit Rev Oncol Hematol. 2017;113:122-134. [CrossRef]
- McGregor JM, Sarkar A. Stereotactic Radiosurgery and Stereotactic Radiotherapy in the Treatment of Skull Base Meningiomas. Otolaryngol Clin North Am. 2009;42(4):677-688. [CrossRef]
- Ding D, Xu Z, McNeill IT, Yen CP, Sheehan JP. Radiosurgery for parasagittal and parafalcine meningiomas: Clinical article. J Neurosurg. 2013;119(4):871-877. [CrossRef]
- Goldsmith BJ, Wara WM, Wilson CB, Larson DA. Postoperative irradiation for subtotally resected meningiomas: A retrospective analysis of 140 patients treated from 1967 to 1990. J Neurosurg. 1994;80(2):195-201. [CrossRef]
- Wu A, Garcia MA, Magill ST, et al. Presenting Symptoms and Prognostic Factors for Symptomatic Outcomes Following Resection of Meningioma. World Neurosurg. 2018;111:e149-e159. [CrossRef]
- Sheehan JP, Williams BJ, Yen CP. Stereotactic radiosurgery for WHO grade I meningiomas. J Neurooncol. 2010;99(3):407-416. [CrossRef]
- Ruge MI, Tutunji J, Rueß D, et al. Stereotactic radiosurgery for treating meningiomas eligible for complete resection. Radiat Oncol. 2021;16(1):22. [CrossRef]
- Tuleasca C, Ruess D, Ruge M, Régis J. Stereotactic Therapies for Meningiomas. Adv Exp Med Biol. 2023;1416:107-119. [CrossRef]
- Goldbrunner R, Stavrinou P, Jenkinson MD, et al. EANO guideline on the diagnosis and management of meningiomas. Neuro-Oncol. 2021;23(11):1821-1834. [CrossRef]
- NAKASU S, NAKASU Y. Natural History of Meningiomas: Review with Meta-analyses. Neurol Med Chir (Tokyo). 2020;60(3):109-120. [CrossRef]
- Graillon T, Regis J, Barlier A, Brue T, Dufour H, Buchfelder M. Parasellar Meningiomas. Neuroendocrinology. 2020;110(9-10):780-796. [CrossRef]
- Wijaya JH, Arjuna YYE, July J. The role of stereotactic radiosurgery in the management of petroclival meningioma: a systematic review. J Neurooncol. 2022;159(1):33-42. [CrossRef]
- Mantziaris G, Pikis S, Bunevicius A, et al. Stereotactic radiosurgery for asymptomatic petroclival region meningiomas: a focused analysis from the IMPASSE study. Acta Neurochir (Wien). 2022;164(1):273-279. [CrossRef]
- Bin Alamer O, Palmisciano P, Mallela AN, et al. Stereotactic radiosurgery in the management of petroclival meningiomas: a systematic review and meta-analysis of treatment outcomes of primary and adjuvant radiosurgery. J Neurooncol. 2022;157(2):207-219. [CrossRef]
- Bunevicius A, Ahn J, Fribance S, et al. Stereotactic Radiosurgery for Olfactory Groove Meningiomas: An International, Multicenter Study. Neurosurgery. 2021;89(5):784-791. [CrossRef]
- Karaaslan B, Celtici E, Bulduk EB, et al. Stereotactic radiosurgery after subtotal resection of critically located grade i meningioma: a single-center experience and review of literature. Turk Neurosurg. Published online 2020. [CrossRef]
- Gande A, Kano H, Bowden G, et al. Gamma Knife radiosurgery of olfactory groove meningiomas provides a method to preserve subjective olfactory function. J Neurooncol. 2014;116(3):577-583. [CrossRef]
- Karras CL, Texakalidis P, Nie JZ, et al. Outcomes following stereotactic radiosurgery for foramen magnum meningiomas: a single-center experience and systematic review of the literature. Neurosurg Focus. 2022;53(5):E6. [CrossRef]
- Akyoldaş G, Samancı Y, Yılmaz M, Şengöz M, Peker S. Long-term results of gamma knife radiosurgery for foramen magnum meningiomas. Neurosurg Rev. 2021;44(5):2667-2673. [CrossRef]
- Malone J, Gaviolli E, Doody J, Sinclair J, Malone S. Unresectable Foramen Magnum Meningioma Treated with CyberKnife Robotic Stereotactic Radiosurgery. Cureus. Published online June 2, 2020. [CrossRef]
- Mehta GU, Zenonos G, Patibandla MR, et al. Outcomes of stereotactic radiosurgery for foramen magnum meningiomas: an international multicenter study. J Neurosurg. 2018;129(2):383-389. [CrossRef]
- Sun C, Dou Z, Wu J, et al. The Preferred Locations of Meningioma According to Different Biological Characteristics Based on Voxel-Wise Analysis. Front Oncol. 2020;10:1412. [CrossRef]
- Ogasawara C, Philbrick BD, Adamson DC. Meningioma: A Review of Epidemiology, Pathology, Diagnosis, Treatment, and Future Directions. Biomedicines. 2021;9(3):319. [CrossRef]
- Casali C, Del Bene M, DiMeco F. Falcine meningiomas. In: Handbook of Clinical Neurology. Vol 170. Elsevier; 2020:101-106. [CrossRef]
- Abdallah HM, Mallela AN, Wei Z, Abou-Al-Shaar H, Niranjan A, Lunsford LD. Gamma Knife radiosurgery for meningiomas of the confluence of the falx and tentorium. J Neurooncol. 2023;161(2):225-233. [CrossRef]
- Matoušek P, Cvek J, Čábalová L, et al. Does Endoscopic Transnasal Optic Nerve Decompression Followed by Radiosurgery Improve Outcomes in the Treatment of Parasellar Meningiomas? Medicina (Mex). 2022;58(8):1137. [CrossRef]
- Valencia-Ramos C, Arroyo Del Castillo JG, Aragon JF, et al. Prognostic Factors in the Surgery of Clinoidal Meningiomas. Cureus. Published online June 8, 2023. [CrossRef]
- Umekawa M, Shinya Y, Hasegawa H, Katano A, Saito N. Primary Stereotactic Radiosurgery Provides Favorable Tumor Control for Intraventricular Meningioma: A Retrospective Analysis. J Clin Med. 2023;12(3):1068. [CrossRef]
- Gendreau JL, Sheaffer K, Macdonald N, et al. Stereotactic radiosurgery for cerebellopontine meningiomas: a systematic review and meta-analysis. Br J Neurosurg. 2023;37(2):199-205. [CrossRef]
- Glenn CA, Tullos HJ, Sughrue ME. Natural history of intracranial meningiomas. In: Handbook of Clinical Neurology. Vol 169. Elsevier; 2020:205-227. [CrossRef]
- Hashimoto N, Rabo CS, Okita Y, et al. Slower growth of skull base meningiomas compared with non-skull base meningiomas based on volumetric and biological studies. J Neurosurg. 2012;116(3):574-580. [CrossRef]
- Lee EJ, Kim JH, Park ES, et al. A novel weighted scoring system for estimating the risk of rapid growth in untreated intracranial meningiomas. J Neurosurg. 2017;127(5):971-980. [CrossRef]
- Delgado-López PD, Montalvo-Afonso A, Martín-Alonso J, et al. Volumetric growth rate of incidental asymptomatic meningiomas: a single-center prospective cohort study. Acta Neurochir (Wien). 2021;163(6):1665-1675. [CrossRef]
- Sheehan J, Pikis S, Islim AI, et al. An international multicenter matched cohort analysis of incidental meningioma progression during active surveillance or after stereotactic radiosurgery: the IMPASSE study. Neuro-Oncol. 2022;24(1):116-124. [CrossRef]
- Bair MM, Munakomi S. Neuroanatomy, Falx Cerebri. In: StatPearls. StatPearls Publishing; 2024. Accessed February 6, 2024. http://www.ncbi.nlm.nih.gov/books/NBK545304/.
- Kong X, Gong S, Lee IT, Yang Y. Microsurgical treatment of parafalcine meningiomas: a retrospective study of 126 cases. OncoTargets Ther. 2018;11:5279-5285. [CrossRef]
- Mathiesen T. Chapter 9 - Parasagittal meningiomas. In: McDermott MW, ed. Handbook of Clinical Neurology. Vol 170. Meningiomas, Part II. Elsevier; 2020:93-100. [CrossRef]
- Pinzi V, Fariselli L, Marchetti M, Scorsetti M, Navarria P. Stereotactic Radiotherapy for Parasagittal and Parafalcine Meningiomas: Patient Selection and Special Considerations. Cancer Manag Res. 2019;Volume 11:10051-10060. [CrossRef]
- Sarris C, Sanai N. Convexity meningioma resection in the modern neurosurgical era. In: Handbook of Clinical Neurology. Vol 170. Elsevier; 2020:87-92. [CrossRef]
- Kondziolka D, Madhok R, Lunsford LD, et al. Stereotactic radiosurgery for convexity meningiomas: Clinical article. J Neurosurg. 2009;111(3):458-463. [CrossRef]
- Pikis S, Mantziaris G, Islim AI, et al. Stereotactic radiosurgery versus active surveillance for incidental, convexity meningiomas: a matched cohort analysis from the IMPASSE study. J Neurooncol. 2022;157(1):121-128. [CrossRef]
- Ruiz-Garcia H, Trifiletti DM, Mohammed N, et al. Convexity Meningiomas in Patients with Neurofibromatosis Type 2: Long-Term Outcomes After Gamma Knife Radiosurgery. World Neurosurg. 2021;146:e678-e684. [CrossRef]
- Ngnitewe Massa R, Minutello K, Mesfin FB. Neuroanatomy, Cavernous Sinus. In: StatPearls. StatPearls Publishing; 2024. Accessed February 6, 2024. http://www.ncbi.nlm.nih.gov/books/NBK459244/.
- Park KJ, Kano H, Iyer A, et al. Gamma Knife stereotactic radiosurgery for cavernous sinus meningioma: long-term follow-up in 200 patients. J Neurosurg. 2019;130(6):1799-1808. [CrossRef]
- Basak A, Ozbek M. Single-Fraction Gamma-Knife Radiosurgery with or without Previous Surgery for Cavernous Sinus Meningiomas: A Single-Center Experience and Systematic Literature Review. Niger J Clin Pract. 2023;26(5):545-551. [CrossRef]
- Zeiler FA, McDonald PJ, Kaufmann AM, et al. Gamma Knife Radiosurgery of Cavernous Sinus Meningiomas: An Institutional Review. Can J Neurol Sci J Can Sci Neurol. 2012;39(6):757-762. [CrossRef]
- Sughrue ME, Rutkowski MJ, Aranda D, Barani IJ, McDermott MW, Parsa AT. Treatment decision making based on the published natural history and growth rate of small meningiomas: A review and meta-analysis. J Neurosurg. 2010;113(5):1036-1042. [CrossRef]
- Fariselli L, Biroli A, Signorelli A, Broggi M, Marchetti M, Biroli F. The cavernous sinus meningiomas’ dilemma: Surgery or stereotactic radiosurgery? Rep Pract Oncol Radiother. 2016;21(4):379-385. [CrossRef]
- Sughrue ME, Rutkowski MJ, Aranda D, Barani IJ, McDermott MW, Parsa AT. Factors affecting outcome following treatment of patients with cavernous sinus meningiomas: Clinical article. J Neurosurg. 2010;113(5):1087-1092. [CrossRef]
- Spiegelmann R, Cohen ZR, Nissim O, Alezra D, Pfeffer R. Cavernous sinus meningiomas: a large LINAC radiosurgery series. J Neurooncol. 2010;98(2):195-202. [CrossRef]
- Kimball MM, Friedman WA, Foote KD, Bova FJ, Chi YY. Linear Accelerator Radiosurgery for Cavernous Sinus Meningiomas. Stereotact Funct Neurosurg. 2009;87(2):120-127. [CrossRef]
- Cohen-Inbar O, Tata A, Moosa S, Lee C chia, Sheehan JP. Stereotactic radiosurgery in the treatment of parasellar meningiomas: long-term volumetric evaluation. J Neurosurg. 2018;128(2):362-372. [CrossRef]
- Sheehan JP, Starke RM, Kano H, et al. Gamma Knife radiosurgery for sellar and parasellar meningiomas: a multicenter study: Clinical article. J Neurosurg. 2014;120(6):1268-1277. [CrossRef]
- Hu Y jia, Xie Y bing, Zhang L feng, Ding C, Chen J. Comparison of clinical outcomes in patients who underwent Gamma Knife radiosurgery for parasellar meningiomas with or without prior surgery. BMC Neurol. 2020;20(1):153. [CrossRef]
- Williams BJ, Yen CP, Starke RM, et al. Gamma Knife surgery for parasellar meningiomas: long-term results including complications, predictive factors, and progression-free survival: Clinical article. J Neurosurg. 2011;114(6):1571-1577. [CrossRef]
- Peters DR, Asher A, Conti A, et al. Single fraction and hypofractionated radiosurgery for perioptic meningiomas—tumor control and visual outcomes: a systematic review and meta-analysis. Neurosurg Rev. 2023;46(1):287. [CrossRef]
- Hénaux PL, Bretonnier M, Le Reste PJ, Morandi X. Modern Management of Meningiomas Compressing the Optic Nerve: A Systematic Review. World Neurosurg. 2018;118:e677-e686. [CrossRef]
- Bailo M, Gagliardi F, Boari N, Castellano A, Spina A, Mortini P. The Role of Surgery in Meningiomas. Curr Treat Options Neurol. 2019;21(10):51. [CrossRef]
- Bunevicius A, Anand RK, Suleiman M, et al. Stereotactic Radiosurgery for Perioptic Meningiomas: An International, Multicenter Study. Neurosurgery. 2021;88(4):828-837. [CrossRef]
- Asuzu DT, Bunevicius A, Kormath Anand R, et al. Clinical and radiologic outcomes after stereotactic radiosurgery for meningiomas in direct contact with the optic apparatus: an international multicenter study. J Neurosurg. 2022;136(4):1070-1076. [CrossRef]
- Chen HY, Chuang CC, Chen HC, et al. Clinical outcomes of fractionated stereotactic radiosurgery in treating perioptic meningiomas and schwannomas: A single-institutional experience. J Clin Neurosci. 2020;81:409-415. [CrossRef]
- Wei Z, Taori S, Song S, Deng H, Niranjan A, Lunsford LD. Does Adjuvant Gamma Knife Stereotactic Radiosurgery Have a Role in Treating Optic Nerve Sheath Meningiomas? J Neuro-Ophthalmol Off J North Am Neuro-Ophthalmol Soc. Published online July 5, 2023. [CrossRef]
- Su CF, Liu DW, Lee CC, Chiu TL. Volume-staged gamma knife surgery for the treatment of large skull base meningioma surrounding the optical apparatus: A snowman-shape design. J Chin Med Assoc. 2017;80(11):697-704. [CrossRef]
- El-Shehaby AMN, Reda WA, Abdel Karim KM, Nabeel AM, Emad Eldin RM, Tawadros SR. Single-session stereotactic radiosurgery for large perioptic meningiomas. J Neurosurg. 2023;139(3):780-789. [CrossRef]
- Hinojosa J, Hernandez JO, Teh BS, Paulino AC, Butler EB. Intensity Modulated Radiotherapy for Optic Nerve Sheath Meningioma. In: Jeremic B, Pitz S, eds. Primary Optic Nerve Sheath Meningioma. Medical Radiology. Springer Berlin Heidelberg; 2008:95-103. [CrossRef]
- Uy NW, Woo SY, Teh BS, et al. Intensity-modulated radiation therapy (IMRT) for meningioma. Int J Radiat Oncol Biol Phys. 2002;53(5):1265-1270. [CrossRef]
- Barber SM, Teh BS, Baskin DS. Fractionated Stereotactic Radiotherapy for Pituitary Adenomas: Single-Center Experience in 75 Consecutive Patients. Neurosurgery. 2016;79(3):406-417. [CrossRef]
- Nicosia L, Di Pietro S, Catapano M, et al. Petroclival meningiomas: radiological features essential for surgeons. ecancermedicalscience. 2019;13. [CrossRef]
- Bunevicius A, Pikis S, Anand RK, et al. Stereotactic radiosurgery for clinoid meningiomas: a multi-institutional study. Acta Neurochir (Wien). 2021;163(10):2861-2869. [CrossRef]
- Demiral S, Dincoglan F, Sager O, et al. Hypofractionated stereotactic radiotherapy (HFSRT) for who grade I anterior clinoid meningiomas (ACM). Jpn J Radiol. 2016;34(11):730-737. [CrossRef]
- Akyoldaş G, Hergünsel ÖB, Yılmaz M, Şengöz M, Peker S. Gamma Knife Radiosurgery for Anterior Clinoid Process Meningiomas: A Series of 61 Consecutive Patients. World Neurosurg. 2020;133:e529-e534. [CrossRef]
- Daza-Ovalle A, Bin-Alamer O, Flickinger J, Niranjan A, Lunsford LD. Outcomes after gamma knife radiosurgery for intraventricular meningiomas. J Neurooncol. 2022;160(1):23-31. [CrossRef]
- Samanci Y, Oktug D, Yilmaz M, Sengoz M, Peker S. Efficacy of gamma knife radiosurgery in the treatment of intraventricular meningiomas. J Clin Neurosci. 2020;80:38-42. [CrossRef]
- Kim IY, Kondziolka D, Niranjan A, Flickinger JC, Lunsford LD. Gamma knife radiosurgery for intraventricular meningiomas. Acta Neurochir (Wien). 2009;151(5):447-452. [CrossRef]
- Nundkumar N, Guthikonda M, Mittal S. Peritumoral edema following Gamma Knife radiosurgery as the primary treatment for intraventricular meningiomas. J Clin Neurosci. 2013;20(4):616-618. [CrossRef]
- Mindermann T, Heckl S, Mack A. High incidence of transient perifocal edema following upfront radiosurgery for intraventricular meningiomas. Acta Neurochir (Wien). 2020;162(9):2177-2182. [CrossRef]
- Christ SM, Farhadi DS, Junzhao S, et al. Efficacy and Safety of Primary Stereotactic Radiosurgery in Patients with Intraventricular Meningiomas. Adv Radiat Oncol. 2023;8(1):101098. [CrossRef]
- Candy NG, Hinder D, Jukes AK, Wormald PJ, Psaltis AJ. Olfaction preservation in olfactory groove meningiomas: a systematic review. Neurosurg Rev. 2023;46(1):186. [CrossRef]
- Liu J, Rojas R, Lam FC, Mirza FA, Mahadevan A, Kasper EM. Indications, feasibility, safety, and efficacy of CyberKnife radiotherapy for the treatment of olfactory groove meningiomas: a single institutional retrospective series. Radiat Oncol. 2020;15(1):63. [CrossRef]
- Zaorsky N, Shi W, Gunn V, Werner-Wasik M, Farrell C, Andrews DW. Fractionated Stereotactic Radiation Therapy and Stereotactic Radiosurgery for the Treatment of Olfactory Groove Meningiomas: Observations of 13 Patients with or Without Prior Surgery. Int J Radiat Oncol Biol Phys. 2012;84(3):S277. [CrossRef]
- Granick MS, Martuza RL, Parker SW, Ojemann RG, Montgomery WW. Cerebellopontine angle meningiomas: clinical manifestations and diagnosis. Ann Otol Rhinol Laryngol. 1985;94(1 Pt 1):34-38. [CrossRef]
- Ali MS, Magill ST, McDermott MW. Petrous face meningiomas. In: Handbook of Clinical Neurology. Vol 170. Elsevier; 2020:157-165. [CrossRef]
- Jahanbakhshi A, Azar M, Kazemi F, Jalessi M, Chanideh I, Amini E. Gamma Knife stereotactic radiosurgery for cerebellopontine angle meningioma. Clin Neurol Neurosurg. 2019;187:105557. [CrossRef]
- El-Shehaby AMN, Reda WA, Abdel Karim KM, Nabeel AM, Emad Eldin RM, Tawadros SR. Hearing preservation after Gamma Knife radiosurgery for cerebellopontine angle meningiomas. J Neurosurg. 2018;129(Suppl1):38-46. [CrossRef]
- Ortiz García IM, Jorques Infante AM, Cordero Tous N, Almansa López J, Expósito Hernández J, Olivares Granados G. Cerebellopontine angle meningiomas: LINAC stereotactic radiosurgery treatment. Neurocir Engl Ed. 2023;34(6):283-291. [CrossRef]
- Park SH, Kano H, Niranjan A, Flickinger JC, Lunsford LD. Stereotactic radiosurgery for cerebellopontine angle meningiomas: Clinical article. J Neurosurg. 2014;120(3):708-715. [CrossRef]
- Kienzler JC, Fandino J. Adaptive Hybrid Surgery Experiences in Benign Skull Base Tumors. Brain Sci. 2022;12(10):1326. [CrossRef]
- Marchetti M, Sahgal A, De Salles AAF, et al. Stereotactic Radiosurgery for Intracranial Noncavernous Sinus Benign Meningioma: International Stereotactic Radiosurgery Society Systematic Review, Meta-Analysis and Practice Guideline. Neurosurgery. 2020;87(5):879-890. [CrossRef]
- Islim AI, Mohan M, Moon RDC, et al. Incidental intracranial meningiomas: a systematic review and meta-analysis of prognostic factors and outcomes. J Neurooncol. 2019;142(2):211-221. [CrossRef]
- Pikis S, Mantziaris G, Samanci Y, et al. Stereotactic Radiosurgery for Incidentally Discovered Cavernous Sinus Meningiomas: A Multi-institutional Study. World Neurosurg. 2022;158:e675-e680. [CrossRef]
- Neromyliotis E, Kalamatianos T, Paschalis A, et al. Machine Learning in Meningioma MRI: Past to Present. A Narrative Review. J Magn Reson Imaging. 2022;55(1):48-60. [CrossRef]
- Momin AA, Shao J, Soni P, et al. Outcomes of salvage radiation for recurrent world health organization grade II meningiomas: a retrospective cohort study. J Neurooncol. 2021;152(2):373-382. [CrossRef]
- Walcott BP, Nahed BV, Brastianos PK, Loeffler JS. Radiation Treatment for WHO Grade II and III Meningiomas. Front Oncol. 2013;3:227. [CrossRef]
- Stafford SL, Pollock BE, Leavitt JA, et al. A study on the radiation tolerance of the optic nerves and chiasm after stereotactic radiosurgery. Int J Radiat Oncol Biol Phys. 2003;55(5):1177-1181. [CrossRef]
- Alfredo C, Carolin S, Güliz A, et al. Normofractionated stereotactic radiotherapy versus CyberKnife-based hypofractionation in skull base meningioma: a German and Italian pooled cohort analysis. Radiat Oncol Lond Engl. 2019;14:201. [CrossRef]
- Fatima N, Meola A, Pollom EL, Soltys SG, Chang SD. Stereotactic radiosurgery versus stereotactic radiotherapy in the management of intracranial meningiomas: a systematic review and meta-analysis. Neurosurg Focus. 2019;46(6):E2. [CrossRef]
- Girvigian MR, Chen JCT, Rahimian J, Miller MJ, Tome M. COMPARISON OF EARLY COMPLICATIONS FOR PATIENTS WITH CONVEXITY AND PARASAGITTAL MENINGIOMAS TREATED WITH EITHER STEREOTACTIC RADIOSURGERY OR FRACTIONATED STEREOTACTIC RADIOTHERAPY. Neurosurgery. 2008;62(5):A19. [CrossRef]
- Leroy HA, Tuleasca C, Reyns N, Levivier M. Radiosurgery and fractionated radiotherapy for cavernous sinus meningioma: a systematic review and meta-analysis. Acta Neurochir (Wien). 2018;160(12):2367-2378. [CrossRef]
- Mansouri A, Guha D, Klironomos G, Larjani S, Zadeh G, Kondziolka D. Stereotactic Radiosurgery for Intracranial Meningiomas: Current Concepts and Future Perspectives. Neurosurgery. 2015;76(4):362-371. [CrossRef]
- Rogers L, Zhang P, Vogelbaum MA, et al. Intermediate-risk meningioma: Initial outcomes from NRG Oncology RTOG 0539. J Neurosurg. 2018;129(1):35-47. [CrossRef]
- Rogers CL, Won M, Vogelbaum MA, et al. High-risk Meningioma: Initial Outcomes from NRG Oncology/RTOG 0539. Int J Radiat Oncol Biol Phys. 2020;106(4):790-799. [CrossRef]
- Knisely JPS, Haghdel A, Chang SJ, et al. Progression-Free Survival in Patients with WHO-2 Meningioma Undergoing Active Surveillance Based on DOTATATE PET Evidence of Gross Total Resection: Prospective Observational Study. Int J Radiat Oncol. 2023;117(2):e118-e119. [CrossRef]
- Rutland JW, Dullea JT, Shrivastava RK. Future directions for immunotherapy in meningioma treatment. Oncotarget. 2021;12(22):2300-2301. [CrossRef]
- Chao S, Rogers L. External beam radiation therapy for meningioma. In: Handbook of Clinical Neurology. Vol 170. Elsevier; 2020:259-278. [CrossRef]
- Kowalchuk RO, Shepard MJ, Sheehan K, et al. Treatment of WHO Grade 2 Meningiomas with Stereotactic Radiosurgery: Identification of an Optimal Group for SRS Using RPA. Int J Radiat Oncol. 2021;110(3):804-814. [CrossRef]
- Louis DN, Perry A, Reifenberger G, et al. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol (Berl). 2016;131(6):803-820. [CrossRef]
- Bowden G, Faramand A, Mallella A, et al. Does the Timing of Radiosurgery after Grade 1 Meningioma Resection Affect Long-Term Outcomes? Stereotact Funct Neurosurg. 2021;99(6):506-511. [CrossRef]
- Karimi S, Mansouri S, Nassiri F, et al. Clinical significance of checkpoint regulator “Programmed death ligand-1 (PD-L1)” expression in meningioma: review of the current status. J Neurooncol. 2021;151(3):443-449. [CrossRef]
- Mair MJ, Berghoff AS, Brastianos PK, Preusser M. Emerging systemic treatment options in meningioma. J Neurooncol. 2023;161(2):245-258. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).