Introduction
Traumatic brain injury (TBI) is an injury that affects the overall functioning of the brain. The data suggest that experiencing TBI can have dangerous consequences on a patient's motor functioning and ability. This injury ranges from mild to severe and has a multitude of triggers. These triggers include sports injuries, falls, car accidents, and others. There were over 64,000 deaths arising out of complications due to traumatic brain injury in the United States in 2020 (Galgano et al., 2017). That adds up to almost 176 TBI-related deaths every day. The Centers for Disease Control report that between 2001 and 2010 there was an increase in the total combined rates of TBI-related emergency department visits, hospitalizations, and deaths. (Galgano et al., 2017). TBI is also largely prevalent in military operations. According to the US centers for disease control and prevention, the number of TBI cases in the United States increased significantly between 2000 and 2006, because of the return of large numbers of soldiers stationed overseas. The U.S. The Armed Forces Health Monitoring Centre reported that 375,230 US servicemen suffered from TBI from 2000 to 2016. According to a study done in 2007, TBI is among one of the leading causes of death and disability in the United States (Parikh et al., 2007), specifically from ages 1-44 and annually, about 2.5 million people suffer from a traumatic brain injury (Frieden et al., 2015). TBI can range from mild, moderate, to severe, and there are a number of negative implications and effects of this injury on a person, for instance difficulty with speech, movement, language or psychological effects such as anxiety or depression. Patients diagnosed with TBI must work towards recovery through rehabilitation, physical therapy, speech therapy, and so on. As part of the TRACK-TBI study, a multicenter study that aims to recruit participants at 11 US level I trauma centers. Nelson et al. (2019) recruited patients with mild TBI (mTBI) who were enrolled between February 26th 2014 and May 4th 2016 and patients with peripheral orthopedic trauma injury (OTC) who were recruited by August 8, 2018. These researchers used the GOSE score as a primary measure of the correlation between TBIs and a variety of aspects of daily functioning and is usually utilized as an outcome measure in studies of traumatic brain injury. A secondary measure was taken, which was recorded 12 months post injury and involved patient reports of generic TBI related symptoms such as range of psychological distress, general life satisfaction, and so on. The results indicated that at 12 months, the percentage of mTBI participants that reported more functional limitations was 53% vs 38% for OTCs. This lends to the fact that patients diagnosed with mTBI must continue to work towards recovery even after one year of being diagnosed. (Nelson et al., 2019). TBI can range from mild, moderate to severe. The clinical definition of mild TBI is focused on a loss of consciousness that lasts <30 minutes, and any variation of consciousness, or post-traumatic amnesia lasting <24 hours. On the other hand, moderate to severe TBI refers to the loss of consciousness lasting ≥ 30 minutes to prolonged coma and post-traumatic amnesia lasting ≥ 24 hours up to permanently (Carlson et al., 2009).
Studying Motor Function in TBI Patients
Aside from the psychological and cognitive after-effects of TBI, such as problems with attention, memory and executive functioning and/or sadness and grief (Arciniegas et al., 2002); motor function is another such aspect, one that encompasses the result of activating motor neurons. It is crucial to study motor function in a person with TBI particularly because motor neurons help us move, talk, breathe, swallow, etc., which is why without these neurons, we would find it hard to complete regular, basic tasks. The autonomic nervous system ; a part of the peripheral nervous system that manages involuntary physiological processes in the body, such as blood pressure, digestion, respiration, heart rate, etc…sends motor impulses to the visceral organs. This system works automatically, with no conscious effort, and the motor neurons of the autonomic nervous system are found in “autonomic ganglia.” These autonomic ganglia receive data from the cholinergic motor neurons in the brainstem/spinal cord (Vernino et al., 2009). With regards to motor impairment, there are two aspects that are considered. Gross and fine motor skills. Gross motor skills are skills that involve the utilization of our whole body. Any movement such as running, throwing a ball or climbing stairs is considered a gross motor skill. While fine motor skills are specifically targeted towards activities such as turning the page of a book, coloring, drawing, writing and far more. Motor skills encompass any sort of movement that is made using small muscles in our hands and wrists. Motor impairments remain one of the long term effects of TBI, and while there are multiple methods to recover from such injuries, it requires years and years of utmost dedication and consistent work. Loss of motor function ability particularly refers to a part of the body, typically a limb or limbs, experiencing lack of coordination or ataxia. The immediate effects of TBI however, (depending on severity) may range from complete paralysis, poor stamina, lack of muscle control, to weakened muscles. However, impairment primarily refers to the loss or limited function with muscle control and mobility, as well as overall movement.
Impairment to motor skills are normally evident in patients with moderate to severe TBI. Motor recovery after severe TBI normally occurs during the time period that is considered “short term.” Primarily, problems with balance, coordination and movement are evident. While physiotherapy and rehabilitation aims to help the patient recover and restore motor impairments, it is difficult to ensure the patient is fully and completely recovered and back to their original, post-injury state. Neurological damage, both internally and externally, has the ability to cause immense damage to muscles. Muscles become stiff and particular movements become hard to synchronize. However, several studies conducted reveal how early residual neuromotor dysfunction may affect functional outcome after severe TBI. According to Englander and colleagues, patients who had motor strength below 3/5 on the MMT and moderate to severe incoordination upon admission to rehabilitation were more likely to require physical assistance for mobility upon discharge from rehabilitation and at one year following injury (Walker, 2007). Aside from movement, speech is a fine motor skill that is also affected as a result of brain injury. Dysarthria for instance is the inability to control the muscles used in speech. It refers to the abnormality of the neurological system that results in facial paralysis or weakening of the tongue or throat muscles. The speech of a patient, post-TBI may be slurred, slow and difficult to understand, however it’s crucial to study and analyze the level of impairment of dysfunction to the various fine motor skills that aid us in basic tasks. In a recent meta analysis of 11 studies conducted, researchers found motor deficits in school-aged children born prematurely after the 1990s and it was shown to range from 19.0% to 40.5%. (Wang et al., 2019). fNIRS are also useful in analyzing and detecting cognitive function and impairment. Aside from motor function, fNIRS can be extremely useful in studying cognitive functions in our brain. In order for cognitive processes to function effectively, it is vital that our brain is provided with the right amount of oxygen and blood. In order to study the effects of hemodynamics; or how the blood flows through the arteries and veins, and cerebral oxygenation a neuroimaging scan is required. While other scans like the MRI are far more useful in studying cognition due to its ability to accurately image brain activity; the fNIRS has its own advantages such as its capability to quantify any alterations in in the concentration of the oxygenated (oxyHb) and deoxygenated (doxyHb) hemoglobin in a non-invasive manner. However, the advantages of fNIRS are far more useful in testing overall motor impairment
What Is fNIRS?
Most commonly, Computed Tomography (CT) scans and Magnetic Resonance Imaging (MRI) scans are used to detect TBIs. Over the past few years, the MRI imaging technique has obtained extensive interest primarily because it allows for high spatial resolution mapping of the brain. This is primarily because such scans produce a detailed structural image of the brain, making it easier to detect internal and external damage in the brain. The fNIRS was first discovered by F.F. Jöbsis in 1977 (Karim et al., 2012). who was able to observe the ability of red light to penetrate through a 4-mm-thick bone of a beefsteak while he was holding it against visible light (Pinti et al., 2020). This indicated the idea that red light and NIR (near infrared) light with longer wavelengths had the capability to travel through our scalp and skull in order to reach the underlying tissues. The fNIRS machine takes advantage of the transparent nature of the skin and bones to NIR light. The fNIRS works in the following way. Measurements are carried out by transmitting NIR light onto the scalp. Before reaching the brain, the NIR light travels through a multitude of different layers like the scalp skin, cerebrospinal fluid, etc…each with its distinct optical properties. The interaction of the NIR light with human tissue is complicated since the tissue has different physical properties in different directions (direction-dependent) and is inhomogeneous through the different layers. Although, this can be simplified since the NIR light is attenuated by absorption and scattering.
TBIs result in major motor impairment, which is hard to test when a patient is required to lie still and display zero movement. Contrastingly, the temporal resolution of an MRI is far less accurate than that of an EEG (electroencephalogram) for instance. The reason for the temporal resolution Is because the MRI machine is restricted by its hemodynamic response time. The hemodynamic response is a homeostatic process that brings the nutrients used by biological tissues by working to adjust blood flow to areas of focal activity. Normally, the BOLD response signal, which is the Blood-Oxygen-Level-Dependent signal (detected in MRI), reflects changes in deoxyhemoglobin driven by localized changes in brain blood flow and blood oxygenation has a response width of ~3s. This is far slower than the basic neural processes, hence temporal resolution is extremely blurred. The MRI machine produces a scan that displays images of various layers of the brain. Using these images, detection of any sort of motor or cognitive impairment is possible; hence allowing for patients with TBI or any other brain injury to understand lesions or impairments in the brain. With regards to primarily motor impairment, there are two aspects that are considered. Gross and fine motor skills. Gross motor skills are skills that involve the utilization of our whole body. Any movement such as running, throwing a ball or climbing stairs is considered a gross motor skill. While fine motor skills are specifically targeted towards activities such as turning the page of a book, coloring, drawing, writing and far more. Motor skills encompass any sort of movement that is made using small muscles in our hands and wrists. The MRI machine works to produce scans that allow for detection of any impairment to a motor skill or function. However, while it can be used to study fine motor skills, there are limitations such as any sort of large movement that prevents the study of gross motor skills.
Functional near infrared spectroscopy (fNIRS) is a newly developed neuroimaging technique. It is a non-invasive, non-ionizing imaging method that utilizes near infrared light. This light is shined on cortical brain tissue right after passing through the skull and skin. This tissue absorbs light at various speeds. While blood with a high oxygen concentration absorbs mostly wavelengths above 790 nm, blood with low oxygen content absorbs primarily wavelengths below 790 nm. The near-infrared light (650–1,000 nm) is relatively transparent to tissue, making it simpler for fNIRS to identify variations in oxygenated (HbO2) and deoxygenated (HHb) hemoglobin. Through the observation of hemodynamic changes, fNIRS can indirectly detect functional activity. While MRI is able to identify variations in HHb, fNIRS can distinguish between HbO2 and HHb, offering more information about hemodynamics and oxygenation. Although fNIRS has relatively high temporal and spatial resolution, MRIs and EEG scans have higher spatial and temporal resolution, respectively. (Karim et al., 2012). The temporal sampling resolution of the fNIRS is 0.01 second, which is faster than that typically observed with fMRI (Wilcox & Biondi, 2015). The spatial resolution of the fNIRS is around 2 to 3 centimeters which is lower than the MRI but far higher than the EEG (Sakai, 2022). fNIRS has the ability to image the brain at reasonably high spatial and temporal resolution that allows the imaging of a particular structure. This is done using optical intensity measurements that calculate changes in brain hemoglobin concentration.
However, the advantage of using the fNIRS is that while the accuracy levels of the scan (ranges from 700 to 1000 nm, in attempt to accurately measure concentration changes in hemoglobin that is present in the cortex) may not be as high as other techniques, it has the ability to display temporal resolution more accurately than an MRI and better spatial resolution than an EEG (Wilcox & Biondi, 2015). This is beneficial particularly since no other method is a good enough compromise between the two. In comparison to other neuroimaging methods, it is quiet, portable, and reasonably resistant to movement artifacts. When comparing the fNIRS to an fMRI, they each have their respective merits and demerits. The fMRI is non-invasive, repeatable and widely available. It allows for lateral surface and depth measurement of the brain and displays advanced spatial resolution. Similarly, the fNIRS Is also non-invasive. However, its advantages lie more in its ability to be less restrictive towards motion or movement. It is also portable and inexpensive; as opposed to an fMRI which is expensive. Limitations of an fMRI however, include its restrictions when it comes to motion, the noise, triggering claustrophobic patients and the requirement for a particular position. Contrastingly, the fNIRS is limited in terms of the lack of anatomical information it can provide, the superficial scalp signals, and its limitations to the frontal regions and surface analysis of the brain. (Scarapicchia et al., 2017) However, the fNIRS unique ability to stay resistant to movement artifacts, aids in allowing the subject or patient to be scanned whilst moving or conducting a task, hence making it easier to test and analyze motor function. Although, we must comprehend the correspondence between the scalp region where a fNIRS measurement is made and its underlying cortical surface where the source signal is located in order to fully comprehend the spatial resolution through the use of fNIRS. In an effort to combine the benefits of fNIRS with other technologies: The hybrid wearable high-density fNIRS-EEG instrumentation (efNIRS), together with eye-tracking, is being developed by a team of researchers. (Ferreri et al., 2014). They hope to achieve long-term continuous monitoring of regular and abnormal brain activity while moving, perceiving, and interacting with others in the present.
Overall, these benefits make NIRS a significant new tool for the study of motor processes and are pertinent for psychological and neurological research. A study on the clinical application of fNIRS concluded that fNIRS does not directly aid in diagnosis; rather, it is an aid that analyzes the brain's functional activities and infers abnormalities of brain functionality indirectly. (Rahman et al., 2020). The main advantage of the imaging technique in comparison to others is its ability to allow the subject to move and be resistant to movement artifacts. Thus far, near infrared spectroscopy has been primarily used to study cognitive function. However we can infer that it could be a helpful tool to study motor ability because of the primary advantage of using this technique, which is primarily that a subject has the capability to move or display any kind of movement while being scanned.
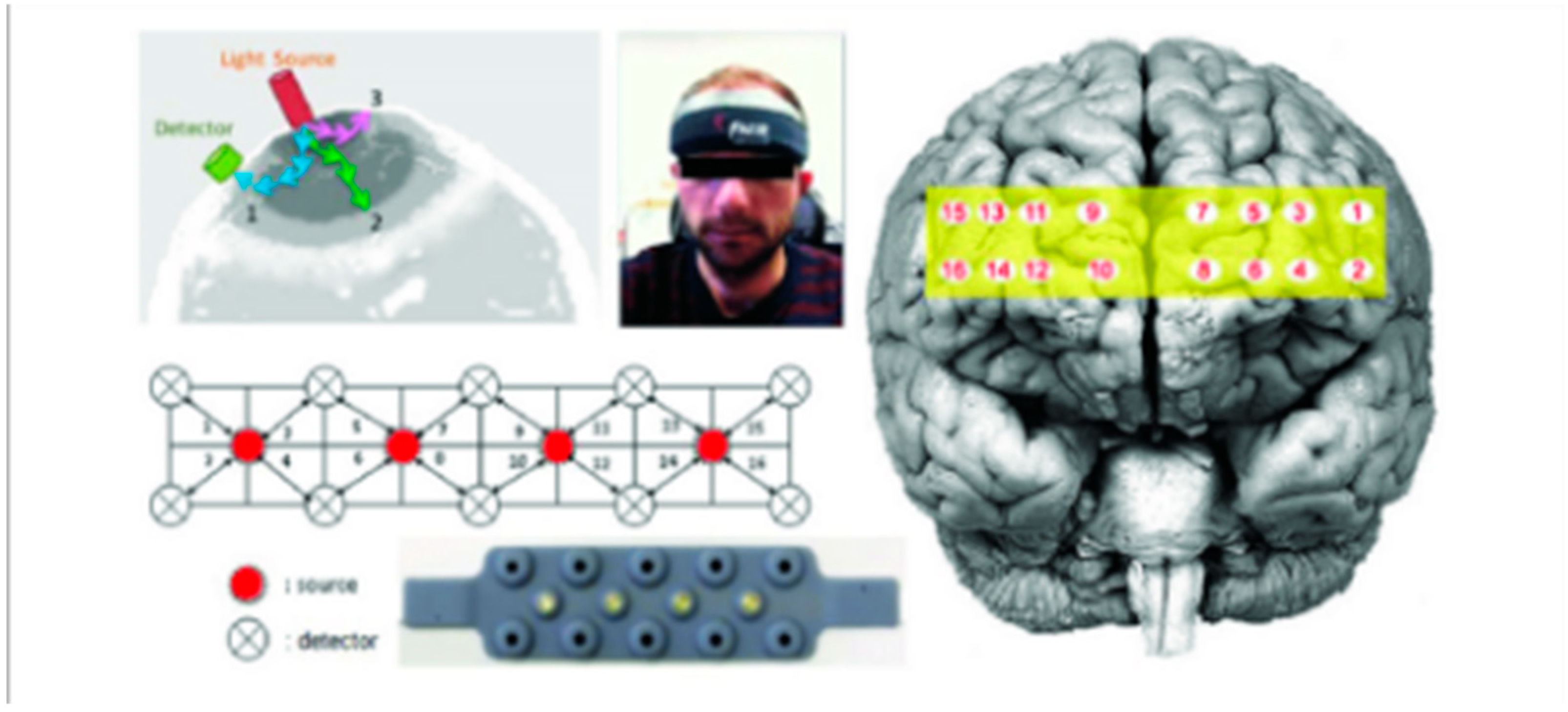
Figure 1.
The functional near-infrared spectroscopy (fNIRS) probe with four light sources and 10 detectors, and the corresponding 16 optodes mapped over the prefrontal cortex (PFC).
Figure 1.
The functional near-infrared spectroscopy (fNIRS) probe with four light sources and 10 detectors, and the corresponding 16 optodes mapped over the prefrontal cortex (PFC).
The Application of fNIRS in Studying Motor Function
fNIRS are particularly useful in studying motor impairment (Guérin et al., 2021) Unlike other scans such as an fNIRS, EEG, PET or CT, an fNIRS does not require one to stay still during the scanning process. As mentioned previously, there are a multitude of use cases for fNIRS. Particularly in studying pediatric populations who are often unable to be scanned effectively and for studying gross motor function in adults.
Figure 3.
Functional near infrared spectroscopy (fNIRS) in an experimental setting. The subject is seated comfortably in a chair with a high back. In order to fit the size and shape of the head, the lighting and collecting optical fiber bundles are combined into a flexible probe holder that is fastened to the head using a Velcro brand fastener (Quaresima & Ferrari, 2019).
Figure 3.
Functional near infrared spectroscopy (fNIRS) in an experimental setting. The subject is seated comfortably in a chair with a high back. In order to fit the size and shape of the head, the lighting and collecting optical fiber bundles are combined into a flexible probe holder that is fastened to the head using a Velcro brand fastener (Quaresima & Ferrari, 2019).
The usability of this scan is highly effective because of the convenience of its design and structure. fNIRS is useful in studying how cortical sensorimotor function affects motor behavior. On days 3-57 of sedated sleep, researchers examined passive knee movements on healthy term newborns and preterm infants (GA: 24-35 weeks; median, 29.6 weeks). All newborns' principal sensorimotor areas showed a rise in oxyhemoglobin (HbO) and total hemoglobin (tHb), while local deoxyhemoglobin (HHb) reduced in 6 out of 7 newborns in response to contralateral knee movement. The largest rates of TBI typically occur in children (0–4 years old), teenagers, and young adults (15–24 y) (Galgano et al., 2017) and since it's challenging to get a younger subject to stay motionless, especially an infant or toddler, fNIRS is an effective tool to employ with younger patients. In a study conducted, fNIRS was used to record activity from the prefrontal cortex of crawling infants during active motion during which they were required to crawl to a parent (Weibley et al., 2021). Additionally, two cognitive activities that required executive function and passive attention to a dancing cat puppet were used to measure the prefrontal activity. This leads us to understanding the overall benefit of the fNIRS technique while compared to others. It is helpful when studying motor, cognitive and executive function in a patient for them to be able to move, which is possible with an fNIRS. A study was conducted by (Kurz et al., 2014) for which, the aim of the investigation was to conclude whether there are discrepancies in cortical activation of children with spastic diplegic cerebral palsy (CP) and regularly developing children during gait. This study utilized the fNIRS tool to measure the concentration of oxyHb present in the sensorimotor cortices while children with and without cerebral palsy walked on a treadmill. The differences in activation of cortical networks used in the control of gait were quantified by keeping track of the change in the concentration of oxyHb. The fNIRS technique was vital in carrying out this experiment as the movement of the participants was critical. During any fNIRS experiment, patients wear a cap that contains a series of photon emitters and detectors, and this cap is situated on the scalp near a brain region of interest. With this study particularly, the fNIRS had an advantage over other imaging methods since it is less vulnerable to head movements, does not require a confined testing environment, and it can be used to evaluate cortical activity during motor tasks such as walking which was crucial for this investigation. The fNIRS technique has emerged as a valuable tool within the realm of rehabilitation, as extensively explored in the research conducted by (Rea et al., 2014). This study aimed to delve into the intricate relationship between fNIRS signals and the preparatory processes governing lower limb motor functions. This investigation not only sought to comprehend the linkages between these signals and motor planning but also aimed to establish a robust framework for their consistent measurement and classification. Such insights were deemed as pivotal for harnessing the full potential of fNIRS in the context of rehabilitating lower limb movements. In this study, the fNIRS was used to test specific hemodynamic changes associated with left and right hip movement preparation. The discerning outcomes of this exploration unveiled fNIRS as an adept tool capable of capturing the subtleties of brain activity associated with single-trial motor preparation in individuals recovering from stroke. This revelation marked a significant advancement, especially considering that while various modalities such as EEG and fMRI have been explored for rehabilitation applications, only a handful have concentrated their efforts on fNIRS. Building upon the collective findings from this study and the array of earlier research endeavors, it becomes increasingly evident that the integration of fNIRS technology holds promise in the realm of gait rehabilitation. The positive outcomes and meaningful insights garnered from studies utilizing the machine, provide compelling evidence for the efficacy of fNIRS devices as a part of the rehabilitation process. This study particularly focused on the potential for fNIRS to play a transformative role in refining and enhancing the rehabilitation process for individuals aiming to restore optimal lower limb movement capabilities.
The Future of fNIRS
Ultimately, the fNIRS technique is a highly effective tool that has a multitude of use cases. From measuring brain activity, hemodynamic responses to neural activity and quantifying changes in oxygenated and deoxygenated hemoglobin…future research directions will warrant immense investigation with this technique. As the fNIRS technology continues to develop, its unique advantages will prove to be even more useful when studying cortical activation in the brain. In spite of the demerits of this technology such as its limited depth of penetration, fairly low spatial resolution (in comparison to other techniques) and a few others, the application of fNIRS in various fields of medicine, scientific research, neuroimaging, rehabilitation and more, provide a strong foundation for the usefulness of the machine and its future applications. Researchers (Quaresima & Ferrari, 2019) mention the vast potential of the fNIRS and its vital and distinct role in fields of interactive neuroscience, cortical activation in sport performance and even neurofeedback training. The benefits and disadvantages of fNIRS have been covered in vast detail among several review articles. Although ultimately, the fNIRS primary advantage lies in its ability to be easily experimented with. The machine is tolerant to movement artifacts, is silent and permits continuous measurements over an extended period of time. Reviews of the benefits and drawbacks of fNIRS have been extensively documented, but its primary advantage lies in its ease of experimentation. It is resistant to movement artifacts, is silent, and enables continuous measurements over extended periods. With ongoing advancements and innovative applications, the fNIRS technology will continue to make significant contributions to the understanding of brain function and its practical applications across various domains. The future of fNIRS technology holds great promise, offering exciting opportunities for researchers and practitioners alike.
Summary
TBI is one of the leading causes for death and disability in the US, (Brain Trauma Foundation, n.d.), resulting in impairment to motor ability and function as well as significant economic impact which is why understanding and advancing our techniques for investigating the underlying brain dysfunction is vital, particularly since older methods like MRI limit our ability to study motor function. Functional near infrared spectroscopy is a helpful neuro-imaging technique that can aid in understanding and analyzing motor impairment. In recent years, there has been a growing use of the functional near-infrared spectroscopy amongst other brain imaging techniques. This increase in the machines’ popularity is primarily because of the variety of unique aspects and advantages that it offers compared to other techniques. fNIRS is resistant to movement artifacts, extremely portable, and fitting for all participant populations including geriatric patients and newborns. (Pinti et al., 2020). Detecting or diagnosing a traumatic brain injury can be done using a multitude of techniques, however using an fNIRS is one such imaging technology that can aid particularly in analyzing motor ability in a patient.
References
-
Brain Trauma Foundation. (n.d.). Retrieved 2 December 2022, from https://www.braintrauma.org/faq.
- Ferreri, L., Bigand, E., Perrey, S., & Bugaïska, A. (2014). The promise of Near-Infrared Spectroscopy (NIRS) for psychological research: A brief review. L’Année psychologique, 114(3), 537–569. https://doi.org/10.3917/anpsy.143.0537 Galgano, M., Toshkezi, G., Qiu, X., Russell, T., Chin, L., & Zhao, L.-R. (2017). Traumatic Brain Injury. Cell Transplantation, 26(7), 1118–1130. [CrossRef]
- Guérin, S. M. R., Vincent, M. A., Karageorghis, C. I., & Delevoye-Turrell, Y. N. (2021). Effects of Motor Tempo on Frontal Brain Activity: An fNIRS Study. NeuroImage, 230, 117597. https://doi.org/10.1016/j.neuroimage.2020.117597 Karim, H., Schmidt, B., Dart, D., Beluk, N., & Huppert, T. (2012). Functional Near-infrared Spectroscopy (fNIRS) of Brain Function During Active Balancing Using a Video Game System. Gait & Posture, 35(3), 367–372. [CrossRef]
- Parikh, S., Koch, M., & Narayan, R. K. (2007). Traumatic Brain Injury. International Anesthesiology Clinics, 45(3), 119–135. [CrossRef]
- Quaresima, V., & Ferrari, M. (2019). Functional Near-Infrared Spectroscopy (fNIRS) for Assessing Cerebral Cortex Function During Human Behavior in Natural/Social Situations: A Concise Review. Organizational Research Methods, 22(1), 46–68. [CrossRef]
- Rahman, Md. A., Siddik, A. B., Ghosh, T. K., Khanam, F., & Ahmad, M. (2020). A Narrative Review on Clinical Applications of fNIRS. Journal of Digital Imaging, 33(5), 1167–1184. [CrossRef]
-
Traumatic Brain Injury Long-Term Effects, Symptoms, and Treatment. (n.d.). Retrieved 4 December 2022, from https://www.cognitivefxusa.com/blog/traumatic-brain-injury-long-term-effects-and-tr eatment.
- Walker, W. C. (2007). Motor impairment after severe traumatic brain injury: A longitudinal multicenter study. The Journal of Rehabilitation Research and Development, 44(7), 975–982. [CrossRef]
- Wang, Q., Zhu, G.-P., Yi, L., Cui, X.-X., Wang, H., Wei, R.-Y., & Hu, B.-L. (2019). A Review of Functional Near-Infrared Spectroscopy Studies of Motor and Cognitive Function in Preterm Infants. Neuroscience Bulletin, 36(3), 321–329. [CrossRef]
- Weibley, H., Di Filippo, M., Liu, X., Lazenby, L., Goscha, J., Ferreira, A., Muscalu, L., &.
- Rader, N. (2021). FNIRS Monitoring of Infant Prefrontal Cortex During Crawling and an Executive Functioning Task. Frontiers in Behavioral Neuroscience, 15. https://www.frontiersin.org/articles/10.3389/fnbeh.2021.675366.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).