Submitted:
07 November 2024
Posted:
08 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
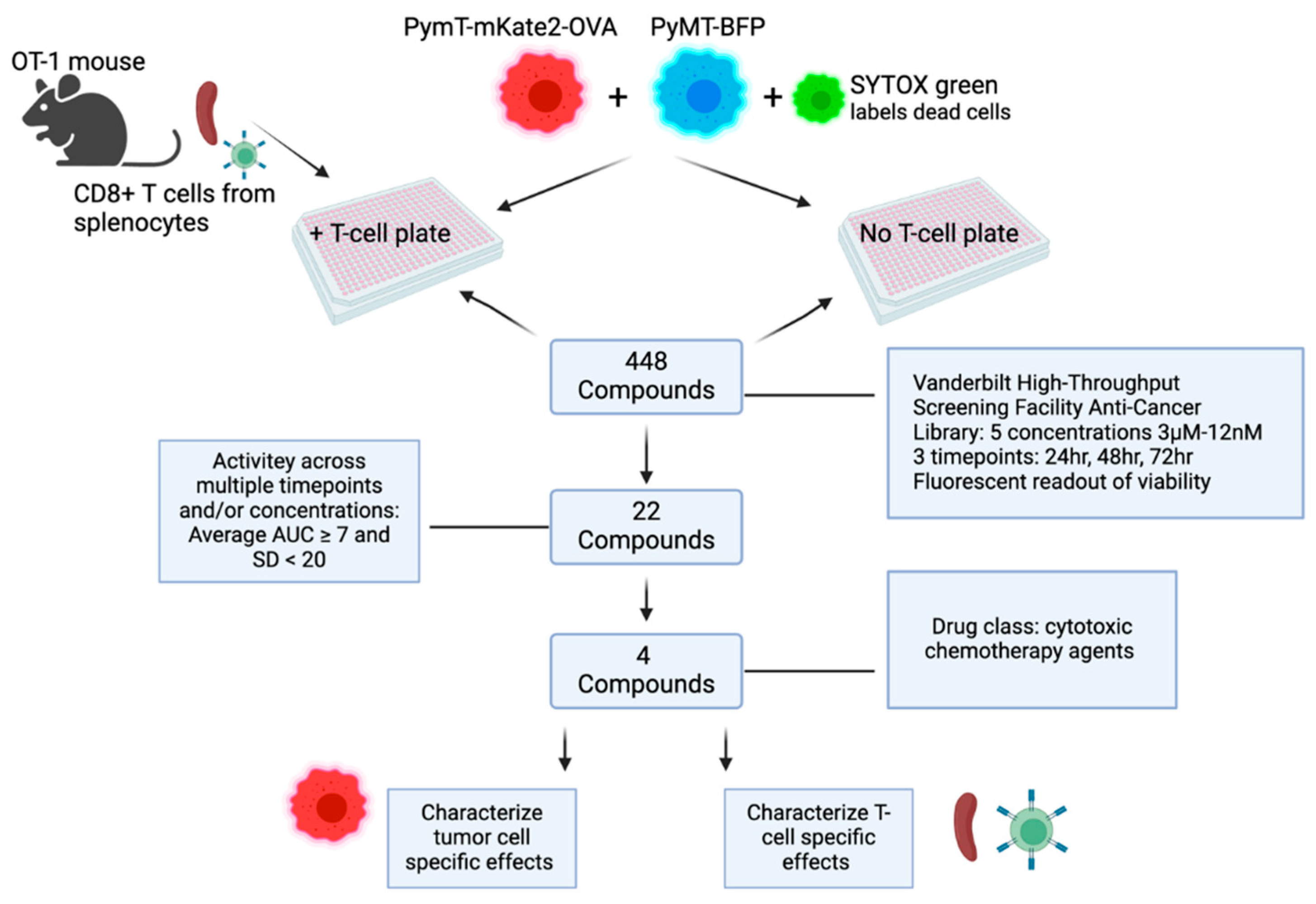
2. Materials and Methods
3. Results
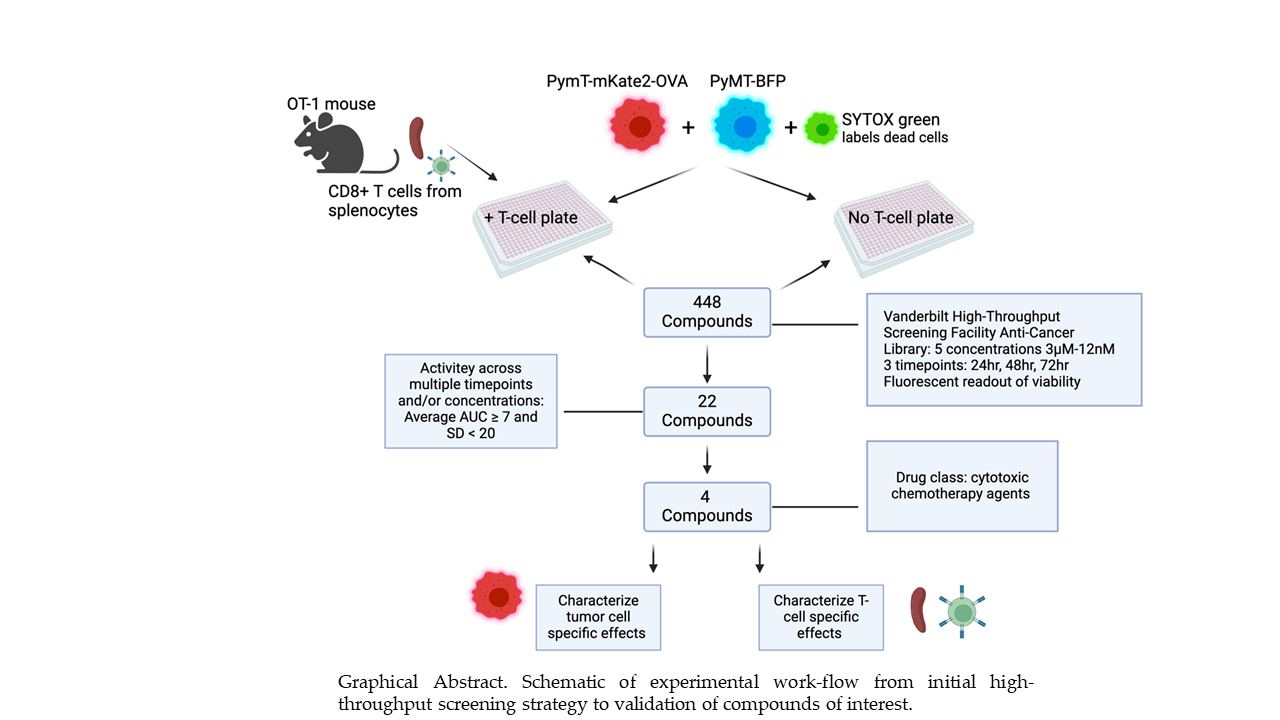
3.1. Compound Screen of 448 Drugs Across Three Timepoints Identifies Four Lead Compounds
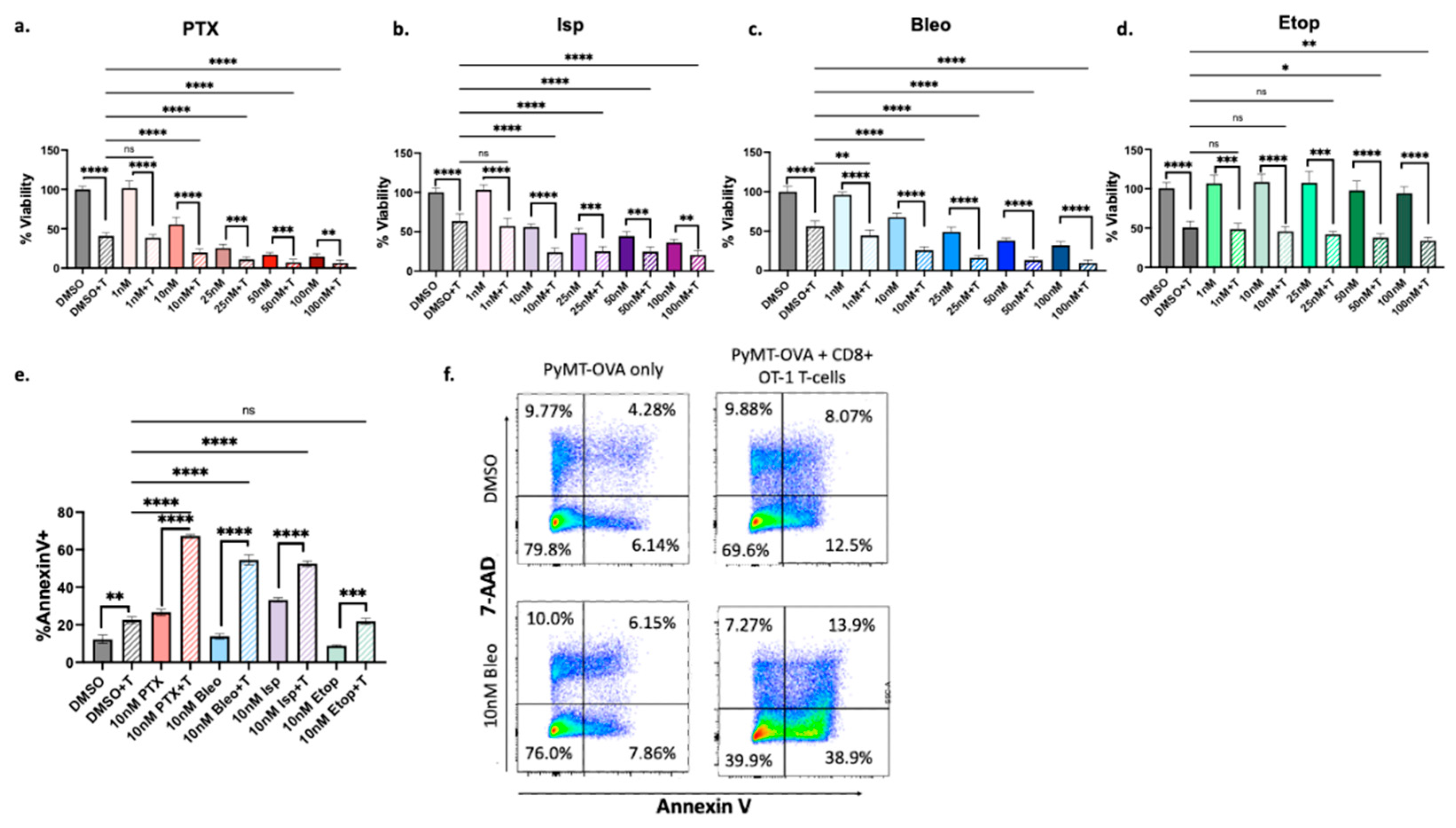
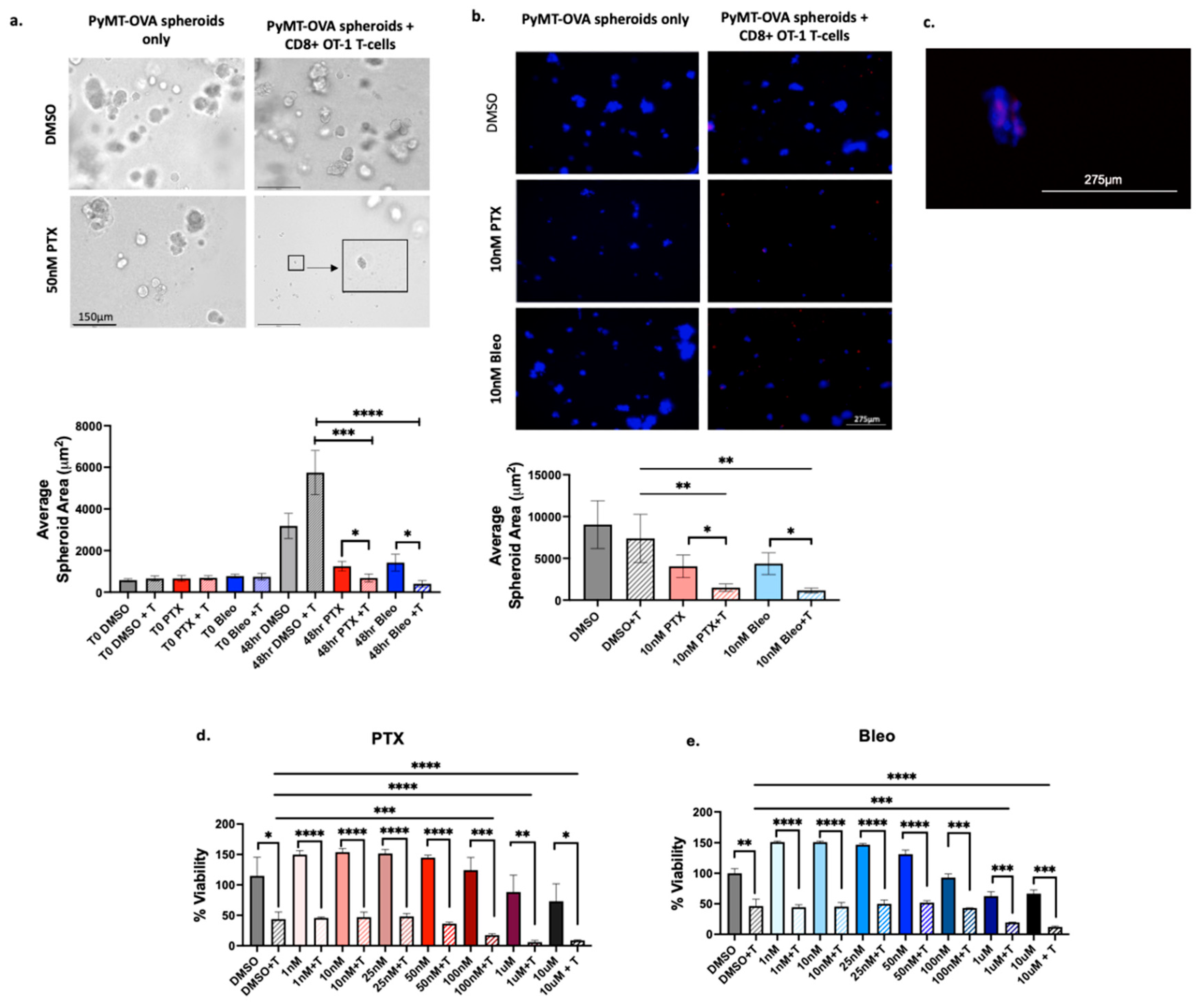
3.2. Confirmation of Increased T-Cell Mediated Cytotoxicity
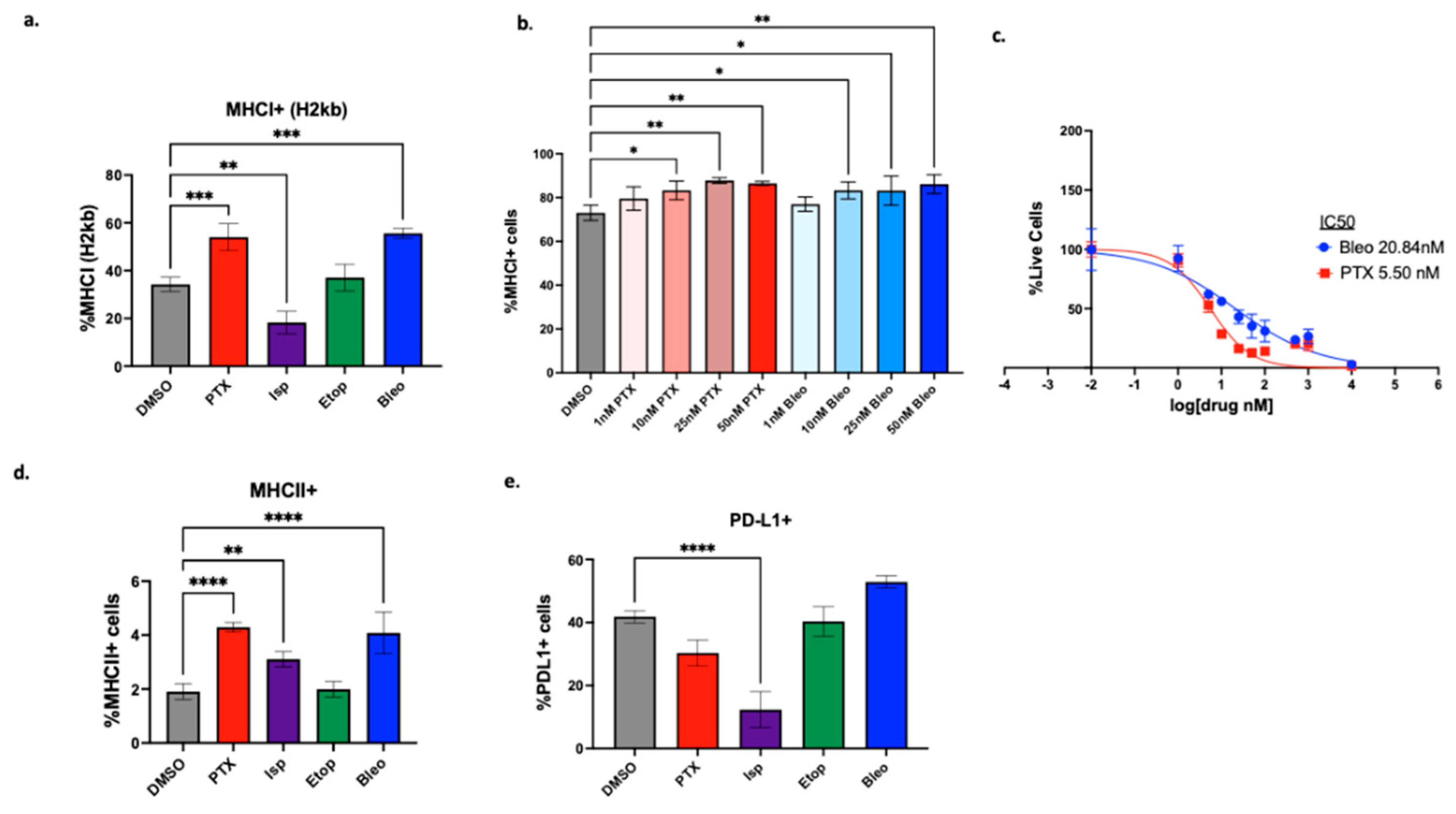
3.3. Dose-Dependent Effects of Lead Compounds on Antigen Presentation by Tumor Cells
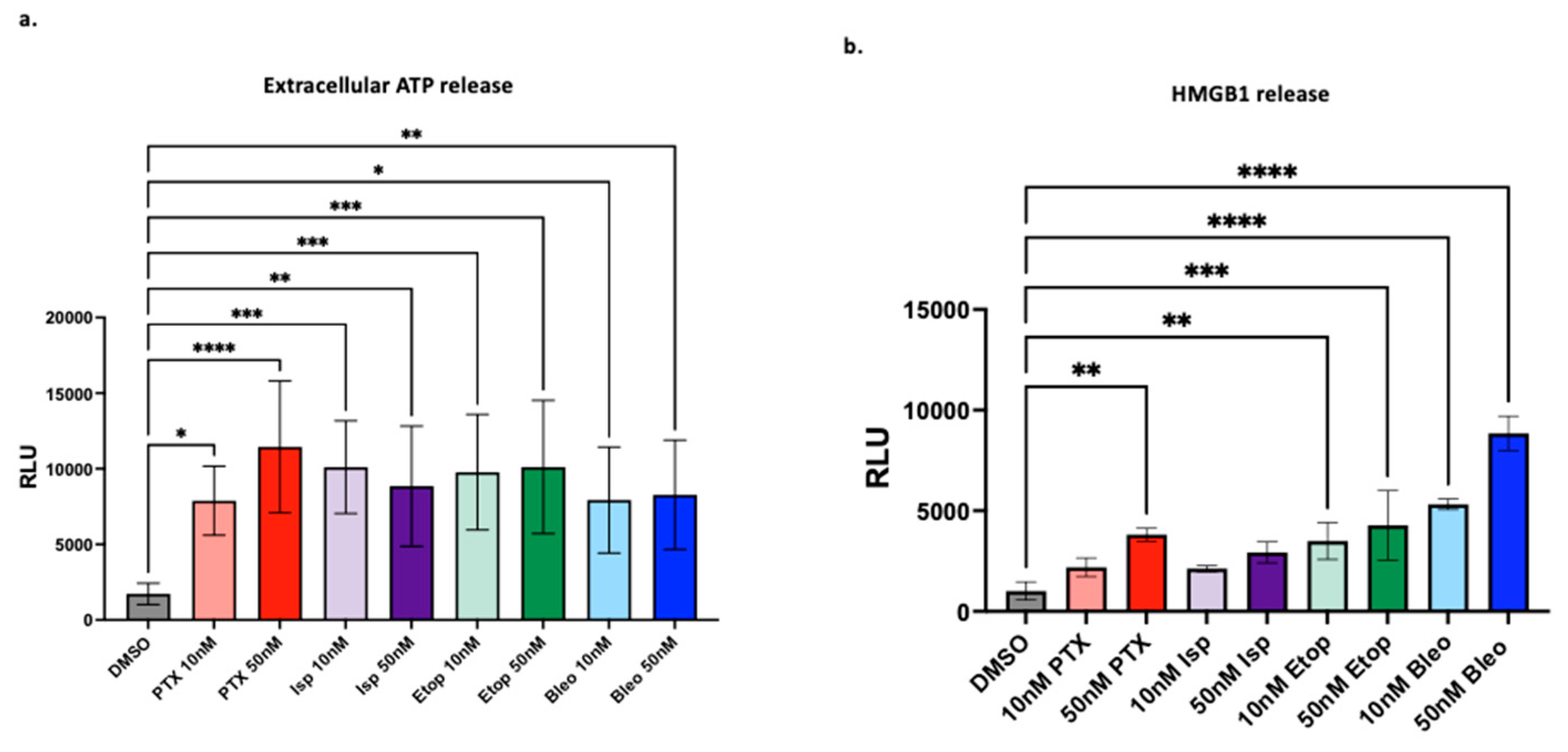
3.4. Lead Compounds Induce Markers of ICD
3.5. Dose-Dependent Effects of Lead Compounds on CD8+ T-Cell Activity
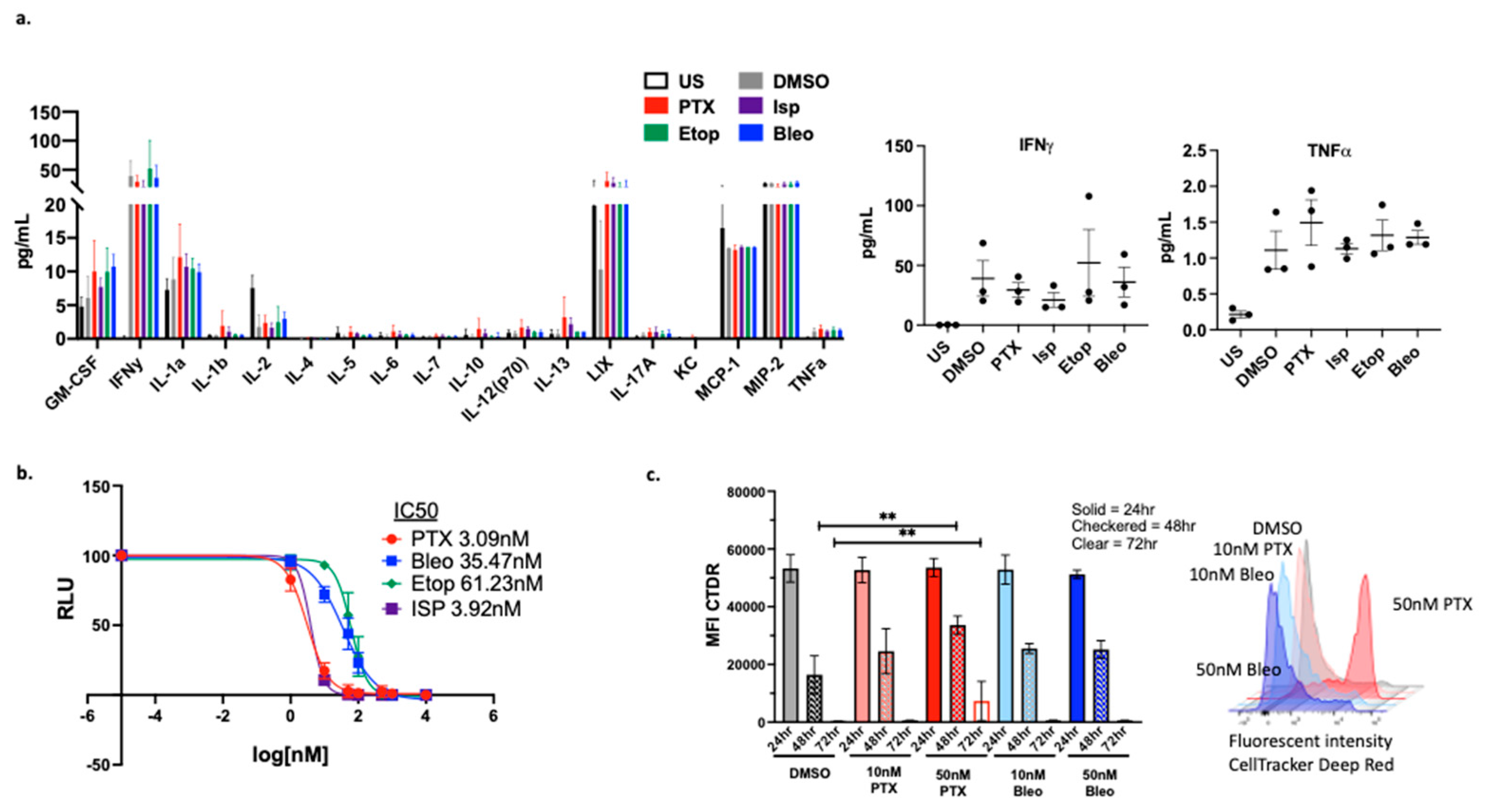
3.6. Validation in Additional Models
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- American Cancer Society Breast Cancer Facts & Figures 2022-2024; Atlanta , 2022.
- Cortes, J.; Cescon, D.W.; Rugo, H.S.; Nowecki, Z.; Im, S.-A.; Yusof, M.M.; Gallardo, C.; Lipatov, O.; Barrios, C.H.; Holgado, E.; et al. Pembrolizumab plus Chemotherapy versus Placebo plus Chemotherapy for Previously Untreated Locally Recurrent Inoperable or Metastatic Triple-Negative Breast Cancer (KEYNOTE-355): A Randomised, Placebo-Controlled, Double-Blind, Phase 3 Clinical Trial. Lancet 2020, 396, 1817–1828. [Google Scholar] [CrossRef] [PubMed]
- Gradishar, W.J.; Tjulandin, S.; Davidson, N.; Shaw, H.; Desai, N.; Bhar, P.; Hawkins, M.; O’Shaughnessy, J. Phase III Trial of Nanoparticle Albumin-Bound Paclitaxel Compared with Polyethylated Castor Oil-Based Paclitaxel in Women with Breast Cancer. J Clin Oncol 2005, 23, 7794–7803. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Humeau, J.; Buqué, A.; Zitvogel, L.; Kroemer, G. Immunostimulation with Chemotherapy in the Era of Immune Checkpoint Inhibitors. Nat Rev Clin Oncol 2020, 17, 725–741. [Google Scholar] [CrossRef] [PubMed]
- Axelrod, M.L.; Nixon, M.J.; Gonzalez-Ericsson, P.I.; Bergman, R.E.; Pilkinton, M.A.; McDonnell, W.J.; Sanchez, V.; Opalenik, S.R.; Loi, S.; Zhou, J.; et al. Changes in Peripheral and Local Tumor Immunity after Neoadjuvant Chemotherapy Reshape Clinical Outcomes in Patients with Breast Cancer. Clin Cancer Res 2021, 26, 5668–5681. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.L.; Huppert, L.A.; Rugo, H.S. Role of Immunotherapy in Breast Cancer. JCO Oncol Pract 2023, 19, 167–179. [Google Scholar] [CrossRef]
- Purcell, J.W.; Davis, J.; Reddy, M.; Martin, S.; Samayoa, K.; Vo, H.; Thomsen, K.; Bean, P.; Kuo, W.L.; Ziyad, S.; et al. Activity of the Kinesin Spindle Protein Inhibitor Ispinesib (SB-715992) in Models of Breast Cancer. Clin Cancer Res 2010, 16, 566–576. [Google Scholar] [CrossRef]
- Tan, M.H.; De, S.; Bebek, G.; Orloff, M.S.; Wesolowski, R.; Downs-Kelly, E.; Budd, G.T.; Stark, G.R.; Eng, C. Specific Kinesin Expression Profiles Associated with Taxane Resistance in Basal-like Breast Cancer. Breast Cancer Res Treat 2012, 131, 849–858. [Google Scholar] [CrossRef]
- Powell, R.T.; Redwood, A.; Liu, X.; Guo, L.; Cai, S.; Zhou, X.; Tu, Y.; Zhang, X.; Qi, Y.; Jiang, Y.; et al. Pharmacologic Profiling of Patient-Derived Xenograft Models of Primary Treatment-Naïve Triple-Negative Breast Cancer. Sci Rep 2020, 10. [Google Scholar] [CrossRef]
- Bailly, C. Etoposide: A Rider on the Cytokine Storm. Cytokine 2023, 168. [Google Scholar] [CrossRef]
- Montecucco, A.; Zanetta, F.; Biamonti, G. Molecular Mechanisms of Etoposide. EXCLI J 2015, 14, 95–108. [Google Scholar] [CrossRef]
- Pacheco, J.M. Immunotherapy for Extensive Stage Small Cell Lung Cancer. J Thorac Dis 2020, 12, 6212–6224. [Google Scholar] [CrossRef] [PubMed]
- Giannone, G.; Milani, A.; Ghisoni, E.; Genta, S.; Mittica, G.; Montemurro, F.; Valabrega, G. Oral Etoposide in Heavily Pre-Treated Metastatic Breast Cancer: A Retrospective Series. Breast 2018, 38, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Cabel, L.; Carton, M.; Cheaib, B.; Pierga, J.Y.; Dalenc, F.; Mailliez, A.; Levy, C.; Jacot, W.; Debled, M.; Leheurteur, M.; et al. Oral Etoposide in Heavily Pre-Treated Metastatic Breast Cancer: Results from the ESME Cohort and Comparison with Other Chemotherapy Regimens. Breast Cancer Res Treat 2019, 173, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Mignot, G.; Bugaut, H.; Ghiringhelli, F. Immune Ambivalence: The Schizophrenic Bleomycin. Oncoimmunology 2013, 2. [Google Scholar] [CrossRef] [PubMed]
- Bugaut, H.; Bruchard, M.; Berger, H.; Derangère, V.; Odoul, L.; Euvrard, R.; Ladoire, S.; Chalmin, F.; Végran, F.; Rébé, C.; et al. Bleomycin Exerts Ambivalent Antitumor Immune Effect by Triggering Both Immunogenic Cell Death and Proliferation of Regulatory T Cells. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Schiff, P.B.; Fant, J.; Horwitz, S.B. Promotion of Microtubule Assembly in Vitro by Taxol. Nature 1979, 277, 665–667. [Google Scholar] [CrossRef]
- Palmieri, C.; Linden, H.; Birrell, S.N.; Wheelwright, S.; Lim, E.; Schwartzberg, L.S.; Dwyer, A.R.; Hickey, T.E.; Rugo, H.S.; Cobb, P.; et al. Activity and Safety of Enobosarm, a Novel, Oral, Selective Androgen Receptor Modulator, in Androgen Receptor-Positive, Oestrogen Receptor-Positive, and HER2-Negative Advanced Breast Cancer (Study G200802): A Randomised, Open-Label, Multicentre, Multinational, Parallel Design, Phase 2 Trial. Lancet Oncol 2024, 25, 317–325. [Google Scholar] [CrossRef]
- Robarge, K.D.; Lee, W.; Eigenbrot, C.; Ultsch, M.; Wiesmann, C.; Heald, R.; Price, S.; Hewitt, J.; Jackson, P.; Savy, P.; et al. Structure Based Design of Novel 6,5 Heterobicyclic Mitogen-Activated Protein Kinase Kinase (MEK) Inhibitors Leading to the Discovery of Imidazo[1,5-a] Pyrazine G-479. Bioorg Med Chem Lett 2014, 24, 4714–4723. [Google Scholar] [CrossRef]
- Di Masi, A.; Leboffe, L.; De Marinis, E.; Pagano, F.; Cicconi, L.; Rochette-Egly, C.; Lo-Coco, F.; Ascenzi, P.; Nervi, C. Retinoic Acid Receptors: From Molecular Mechanisms to Cancer Therapy. Mol Aspects Med 2015, 41, 1–115. [Google Scholar] [CrossRef]
- Dawson, M.A.; Curry, J.E.; Barber, K.; Beer, P.A.; Graham, B.; Lyons, J.F.; Richardson, C.J.; Scott, M.A.; Smyth, T.; Squires, M.S.; et al. AT9283, a Potent Inhibitor of the Aurora Kinases and Jak2, Has Therapeutic Potential in Myeloproliferative Disorders: Research Paper. Br J Haematol 2010, 150, 46–57. [Google Scholar] [CrossRef]
- Bryan, C.; Rice, C.; Hoffman, H.; Harkisheimer, M.; Sweeney, M.; Skordalakes, E. Structural Basis of Telomerase Inhibition by the Highly Specific BIBR1532. Structure 2015, 23, 1934–1942. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.J.; Tran-Dubé, M.; Shen, H.; Nambu, M.; Kung, P.P.; Pairish, M.; Jia, L.; Meng, J.; Funk, L.; Botrous, I.; et al. Structure Based Drug Design of Crizotinib (PF-02341066), a Potent and Selective Dual Inhibitor of Mesenchymal-Epithelial Transition Factor (c-MET) Kinase and Anaplastic Lymphoma Kinase (ALK). J Med Chem 2011, 54, 6342–6363. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Shakalya, K.; Stejskal, A.; Goldman, A.; Beeck, S.; Cooke, L.; Mahadevan, D. NSC348884, a Nucleophosmin Inhibitor Disrupts Oligomer Formation and Induces Apoptosis in Human Cancer Cells. Oncogene 2008, 27, 4210–4220. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Guan, X.; Yan, J. Two-Pore Channel Blockade by Phosphoinositide Kinase Inhibitors YM201636 and PI-103 Determined by a Histidine Residue near Pore-Entrance. Commun Biol 2022, 5. [Google Scholar] [CrossRef]
- Hilgard, P.; Kampherm, E.; Nolan, L.; Pohl, J.; Reissmann, T. Investigation into the Immunological Effects of Miltefosine, a New Anticancer Agent under Development. J Cancer Res Clin Oncol 1991, 117, 403–408. [Google Scholar] [CrossRef]
- Montecucco, A.; Zanetta, F.; Biamonti, G. Molecular Mechanisms of Etoposide. EXCLI J 2015, 14, 95–108. [Google Scholar] [CrossRef]
- Rivera-Rivera, Y.; Marina, M.; Jusino, S.; Lee, M.; Velázquez, J.V.; Chardón-Colón, C.; Vargas, G.; Padmanabhan, J.; Chellappan, S.P.; Saavedra, H.I. The Nek2 Centrosome-Mitotic Kinase Contributes to the Mesenchymal State, Cell Invasion, and Migration of Triple-Negative Breast Cancer Cells. Sci Rep 2021, 11, 9016. [Google Scholar] [CrossRef]
- Zhang, C. qi; Bai, X.; Wan, Y.; Li, H.; Sun, H. Efficacy, Safety, and Retention Rate of Extended-Release Divalproex versus Conventional Delayed-Release Divalproex: A Protocol for Systematic Review and Meta-Analysis. Medicine 2021, 100. [Google Scholar] [CrossRef]
- Tuli, H.S.; Rath, P.; Chauhan, A.; Ramniwas, S.; Vashishth, K.; Varol, M.; Jaswal, V.S.; Haque, S.; Sak, K. Phloretin, as a Potent Anticancer Compound: From Chemistry to Cellular Interactions. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Krop, I.; Demuth, T.; Guthrie, T.; Wen, P.Y.; Mason, W.P.; Chinnaiyan, P.; Butowski, N.; Groves, M.D.; Kesari, S.; Freedman, S.J.; et al. Phase I Pharmacologic and Pharmacodynamic Study of the Gamma Secretase (Notch) Inhibitor MK-0752 in Adult Patients with Advanced Solid Tumors. J Clin Oncol 2012, 30, 2307–2313. [Google Scholar] [CrossRef]
- Garton, A.J.; Crew, A.P.A.; Franklin, M.; Cooke, A.R.; Wynne, G.M.; Castaldo, L.; Kahler, J.; Winski, S.L.; Franks, A.; Brown, E.N.; et al. OSI-930: A Novel Selective Inhibitor of Kit and Kinase Insert Domain Receptor Tyrosine Kinases with Antitumor Activity in Mouse Xenograft Models. Cancer Res 2006, 66, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Becker, B.N.; Hoffmann, F.M.; Mertz, J.E. Complete Reversal of Epithelial to Mesenchymal Transition Requires Inhibition of Both ZEB Expression and the Rho Pathway. BMC Cell Biol 2009, 10. [Google Scholar] [CrossRef]
- Borisa, A.C.; Bhatt, H.G. A Comprehensive Review on Aurora Kinase: Small Molecule Inhibitors and Clinical Trial Studies. Eur J Med Chem 2017, 140, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, T.; Takeuchi, M.; Kinoyama, I.; Minematsu, T.; Shirasuna, K.; Matsuhisa, A.; Kita, A.; Tominaga, F.; Yamanaka, K.; Kudoh, M.; et al. YM155, a Novel Small-Molecule Survivin Suppressant, Induces Regression of Established Human Hormone-Refractory Prostate Tumor Xenografts. Cancer Res 2007, 67, 8014–8021. [Google Scholar] [CrossRef] [PubMed]
- Ohnuma, T.; Holland, J.F.; Masuda, H.; Waligunda, J.A.; Goldberg, G.A. Microbiological Assay of Bleomycin: Inactivation, Tissue Distribution, and Clearance. Cancer 1974, 33, 1230–1238. [Google Scholar] [CrossRef]
- Wang, X.Q.; Danenberg, E.; Huang, C.S.; Egle, D.; Callari, M.; Bermejo, B.; Dugo, M.; Zamagni, C.; Thill, M.; Anton, A.; et al. Spatial Predictors of Immunotherapy Response in Triple-Negative Breast Cancer. Nature 2023, 621, 868–876. [Google Scholar] [CrossRef]
- Rodriguez-Barbosa, J.I.; Azuma, M.; Zelinskyy, G.; Perez-Simon, J.A.; del Rio, M.L. Critical Role of PD-L1 Expression on Non-Tumor Cells Rather than on Tumor Cells for Effective Anti-PD-L1 Immunotherapy in a Transplantable Mouse Hematopoietic Tumor Model. Cancer Immunology, Immunotherapy 2020, 69, 1001–1014. [Google Scholar] [CrossRef]
- Fucikova, J.; Kepp, O.; Kasikova, L.; Petroni, G.; Yamazaki, T.; Liu, P.; Zhao, L.; Spisek, R.; Kroemer, G.; Galluzzi, L. Detection of Immunogenic Cell Death and Its Relevance for Cancer Therapy. Cell Death Dis 2020, 11. [Google Scholar] [CrossRef]
- Vilgelm, A.E.; Johnson, C.A.; Prasad, N.; Yang, J.; Chen, S.C.; Ayers, G.D.; Pawlikowski, J.S.; Raman, D.; Sosman, J.A.; Kelley, M.; et al. Connecting the Dots: Therapy-Induced Senescence and a Tumor-Suppressive Immune Microenvironment. J Natl Cancer Inst 2016, 108. [Google Scholar] [CrossRef]
- Yan, C.; Saleh, N.; Yang, J.; Nebhan, C.A.; Vilgelm, A.E.; Reddy, E.P.; Roland, J.T.; Johnson, D.B.; Chen, S.C.; Shattuck-Brandt, R.L.; et al. Novel Induction of CD40 Expression by Tumor Cells with RAS/RAF/PI3K Pathway Inhibition Augments Response to Checkpoint Blockade. Mol Cancer 2021, 20. [Google Scholar] [CrossRef]
- Ljunggren, H.-G.; Karre, K. In Search of the “Missing Self”: MHC Molecules and NK Cell Recognition. Immunol Today 1990, 11, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.S.; Zhang, W.; Wang, X.; Jiang, P.; Traugh, N.; Li, Z.; Meyer, C.; Stewig, B.; Xie, Y.; Bu, X.; et al. Therapeutically Increasing MHC-I Expression Potentiates Immune Checkpoint Blockade. Cancer Discov 2021, 11, 1524–1541. [Google Scholar] [CrossRef] [PubMed]
- Solari, J.I.G.; Filippi-Chiela, E.; Pilar, E.S.; Nunes, V.; Gonzalez, E.A.; Figueiró, F.; Andrade, C.F.; Klamt, F. Damage-Associated Molecular Patterns (DAMPs) Related to Immunogenic Cell Death Are Differentially Triggered by Clinically Relevant Chemotherapeutics in Lung Adenocarcinoma Cells. BMC Cancer 2020, 20. [Google Scholar] [CrossRef] [PubMed]
- Hodge, J.W.; Garnett, C.T.; Farsaci, B.; Palena, C.; Tsang, K.Y.; Ferrone, S.; Gameiro, S.R. Chemotherapy-Induced Immunogenic Modulation of Tumor Cells Enhances Killing by Cytotoxic T Lymphocytes and Is Distinct from Immunogenic Cell Death. Int J Cancer 2013, 133, 624–636. [Google Scholar] [CrossRef]
- Haugnes, H.S.; Oldenburg, J.; Bremnes, R.M. Pulmonary and Cardiovascular Toxicity in Long-Term Testicular Cancer Survivors. Urologic Oncology: Seminars and Original Investigations 2015, 33, 399–406. [Google Scholar] [CrossRef]
- Yin, Q.; Wu, L.; Han, L.; Zheng, X.; Tong, R.; Li, L.; Bai, L.; Bian, Y. Immune-Related Adverse Events of Immune Checkpoint Inhibitors: A Review. Front Immunol 2023, 14. [Google Scholar] [CrossRef]
- Shaikh, S.; Younis, M.; Yingying, S.; Tanziela, T.; Yuan, L. Bleomycin Loaded Exosomes Enhanced Antitumor Therapeutic Efficacy and Reduced Toxicity. Life Sci 2023, 330. [Google Scholar] [CrossRef]
- Yuba, E.; Osaki, T.; Ono, M.; Park, S.; Harada, A.; Yamashita, M.; Azuma, K.; Tsuka, T.; Ito, N.; Imagawa, T.; et al. Bleomycin-Loaded PH-Sensitive Polymer-Lipid-Incorporated Liposomes for Cancer Chemotherapy. Polymers (Basel) 2018, 10. [Google Scholar] [CrossRef]
- Bullock, K.K.; Shattuck-Brandt, R.; Scalise, C.; Luo, W.; Chen, S.C.; Saleh, N.; Gonzalez-Ericsson, P.I.; Garcia, G.; Sanders, M.E.; Ayers, G.D.; et al. Endogenous PAKT Activity Is Associated with Response to AKT Inhibition Alone and in Combination with Immune Checkpoint Inhibition in Murine Models of TNBC. Cancer Lett 2024, 586, 216681. [Google Scholar] [CrossRef]
- Athanassiadou, P. : Athanassiades, P.; Petrakakou, E.; Zerva, C.; Mavrikakis, M.; Immunocytochemical detection of P-glycoprotein in the management of malignant effusions. J Clin Res Clin Oncol, 1997; 123, 456–490. [Google Scholar] [CrossRef]
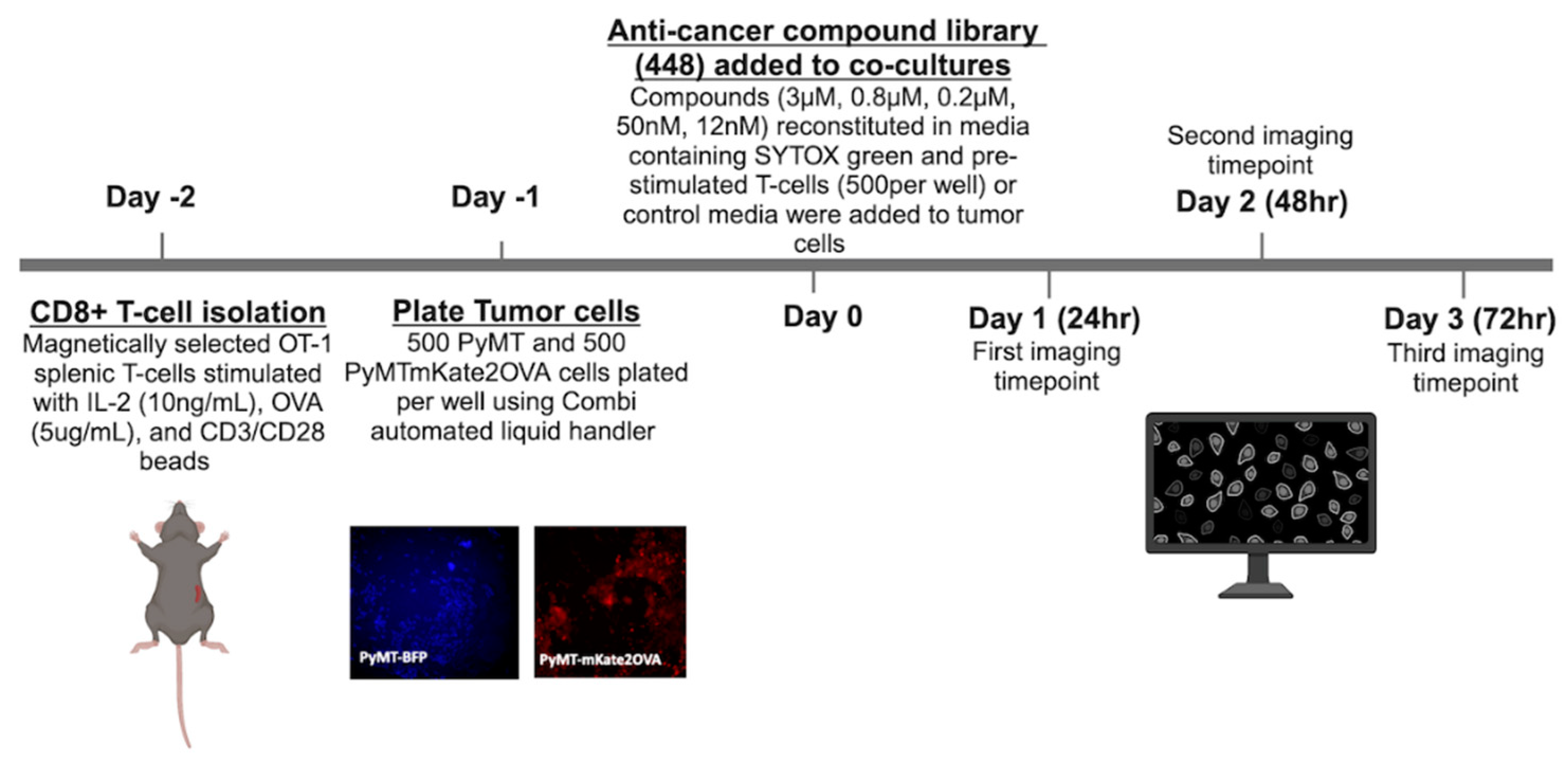
| Compound | AUC | SD | Drug Class | Ref |
|---|---|---|---|---|
| Paclitaxel | 6.99 | 1.11 | Taxane chemotherapy | [17] |
| Ispinesib (Sb-715992) | 7.06 | 3.84 | Kinesin spindle protein inhibitor | [7] |
| MK-2866 (GTx-024) | 7.25 | 3.88 | Nonsteroidal selective androgen receptor modulator | [18] |
| GDC-0623 | 7.31 | 4.09 | MEK1 inhibitor | [19] |
| Tretinoin (Aberela) | 7.33 | 4.7 | Vitamin A derivative | [20] |
| AT9283 | 7.40 | 4.24 | Aurora kinase A/B and Jak2 inhibitor | [21] |
| BIBR1532 | 7.49 | 1.88 | Telomerase inhibitor | [22] |
| Crizotinib (PF-02341066) | 7.76 | 0.87 | c-MET and ALK inhibitor | [23] |
| NSC348884 | 7.79 | 3.85 | Nucleophosphmin inhibitor | [24] |
| YM201636 | 7.85 | 1.83 | PIKfyve inhibitor | [25] |
| Miltefosine | 8.03 | 4.04 | Alkylphosphocholine | [26] |
| Etoposide (VP-16) | 8.04 | 8.08 | Topoisomerase II inhibitor | [27] |
| INH6 | 8.23 | 3.81 | Nek2/Hec1 inhibitor | [28] |
| Divalproex sodium | 8.39 | 6.07 | Anti-epileptic1 | [29] |
| Isoretinoin | 8.72 | 9.08 | Vitamin A derivative | [20] |
| Phloretin | 9.05 | 7.15 | Natural dihydrochalcone | [30] |
| MK-0752 | 9.06 | 9.79 | γ-secretase inhibitor | [31] |
| OSI-930 | 10.99 | 3.85 | c-Kit inhibitor | [32] |
| Y-27632 2HCI | 11.00 | 12.19 | ROCK1/2 inhibitor | [33] |
| CYC116 | 11.60 | 10.85 | Aurora Kinase A/B inhibitor | [34] |
| YM155 (Sepantronium Bromide) | 12.25 | 10.78 | Survivin inhibitor | [35] |
| Bleomycin sulfate | 12.39 | 14.99 | glycopeptide antibiotic | [36] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




