Submitted:
06 November 2024
Posted:
07 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Antigen Preparation
2.2. Generation of Polyclonal Antibody
2.3. Animals
2.4. Western Blot
2.5. Brain Section Preparation and Immunostaining
2.6. Photographing
3. Results
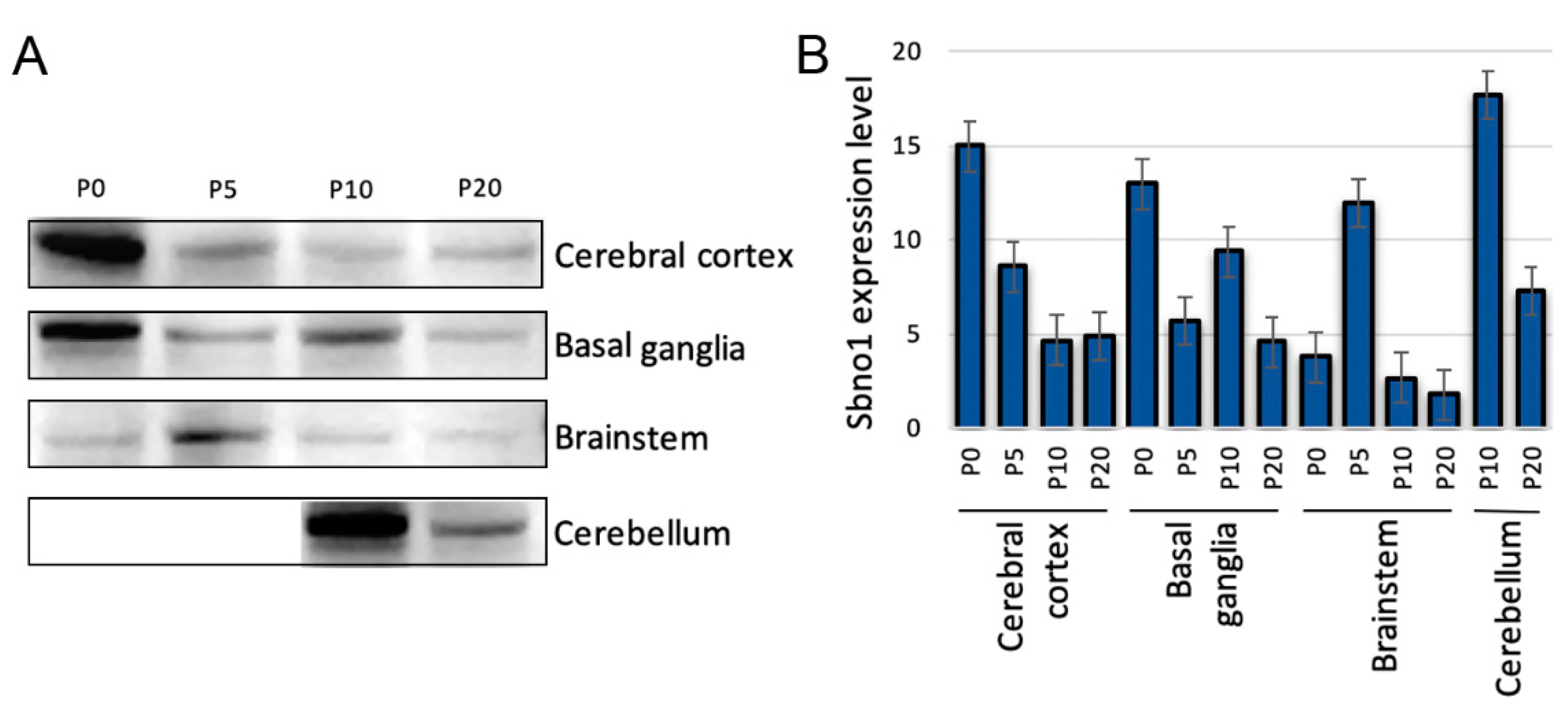
3.1. Quantitative Examination of Sbno1 Protein Level in the Different Brain Regions
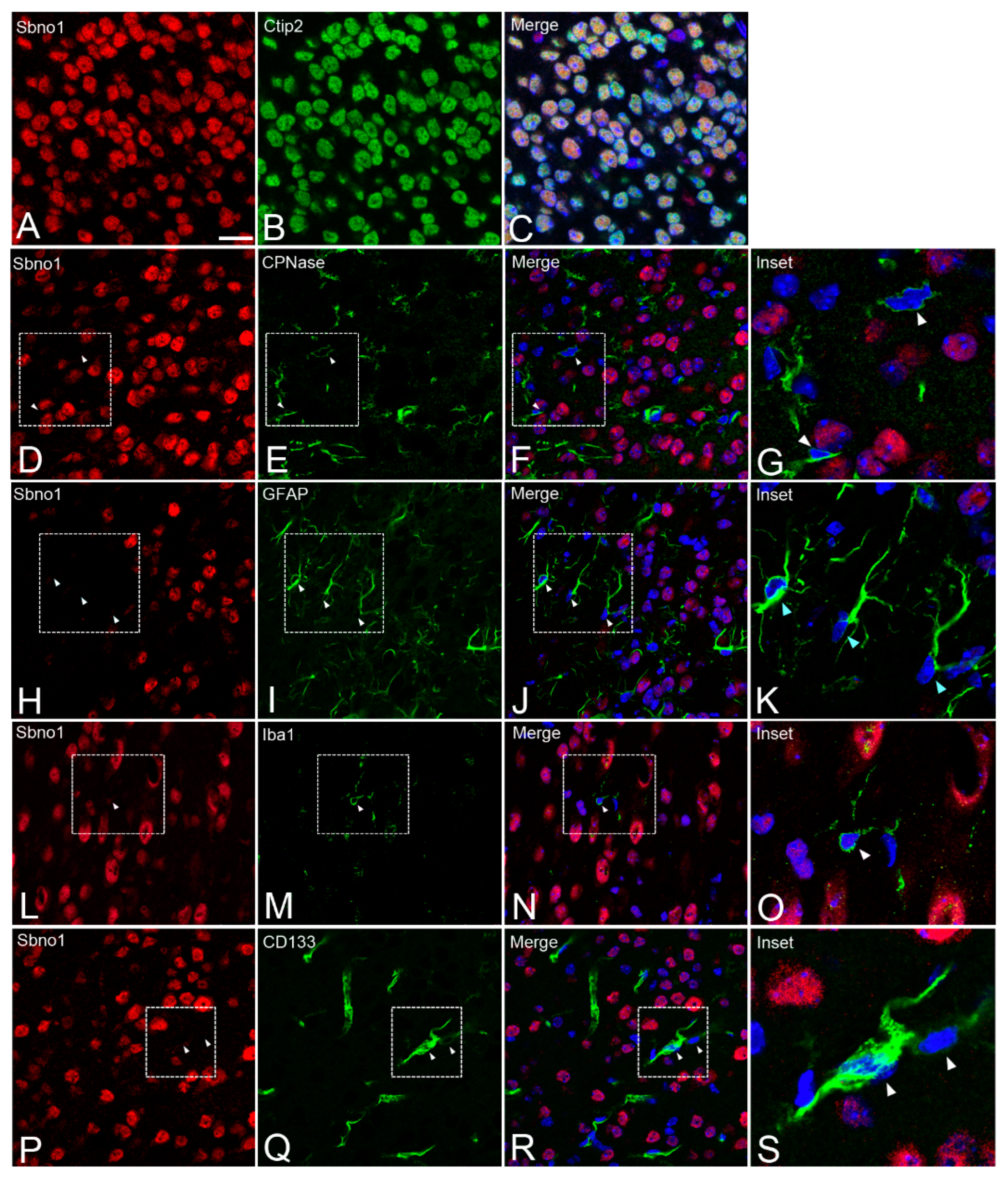
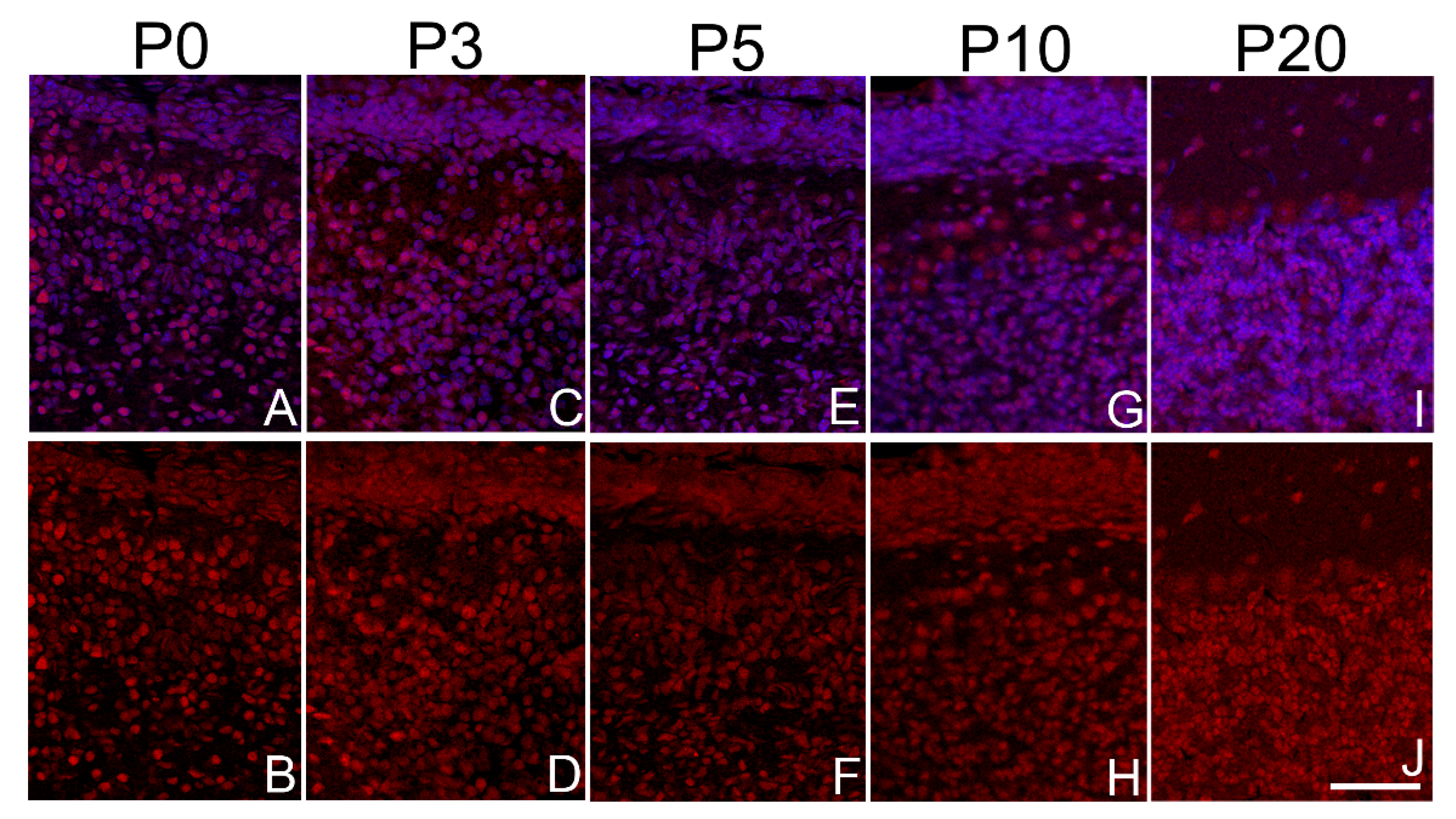
3.2. Sbno1 is Specifically Observed in Neurons in the Brain
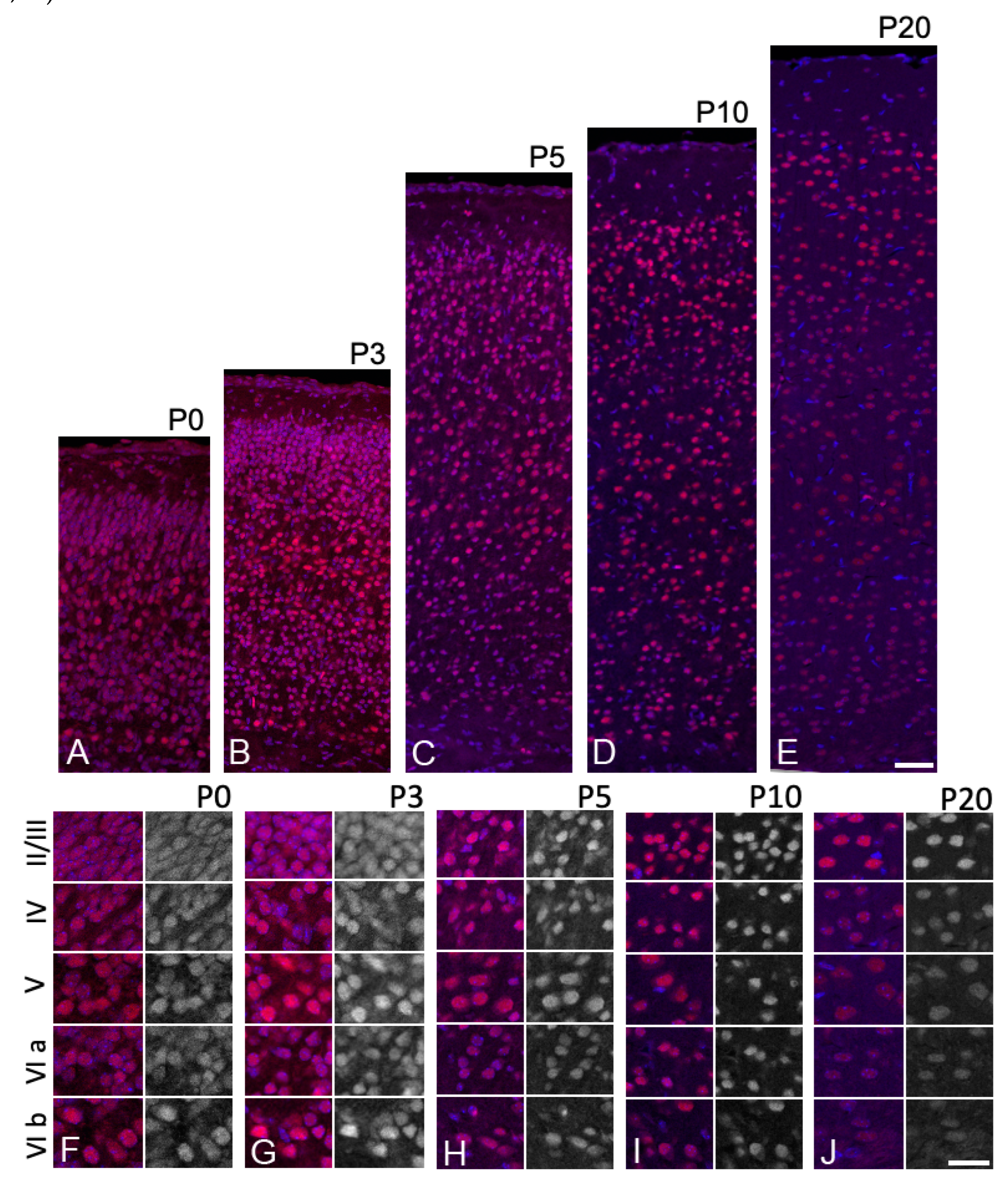
3.3. Sbno1 Expression in the Cerebral Cortex
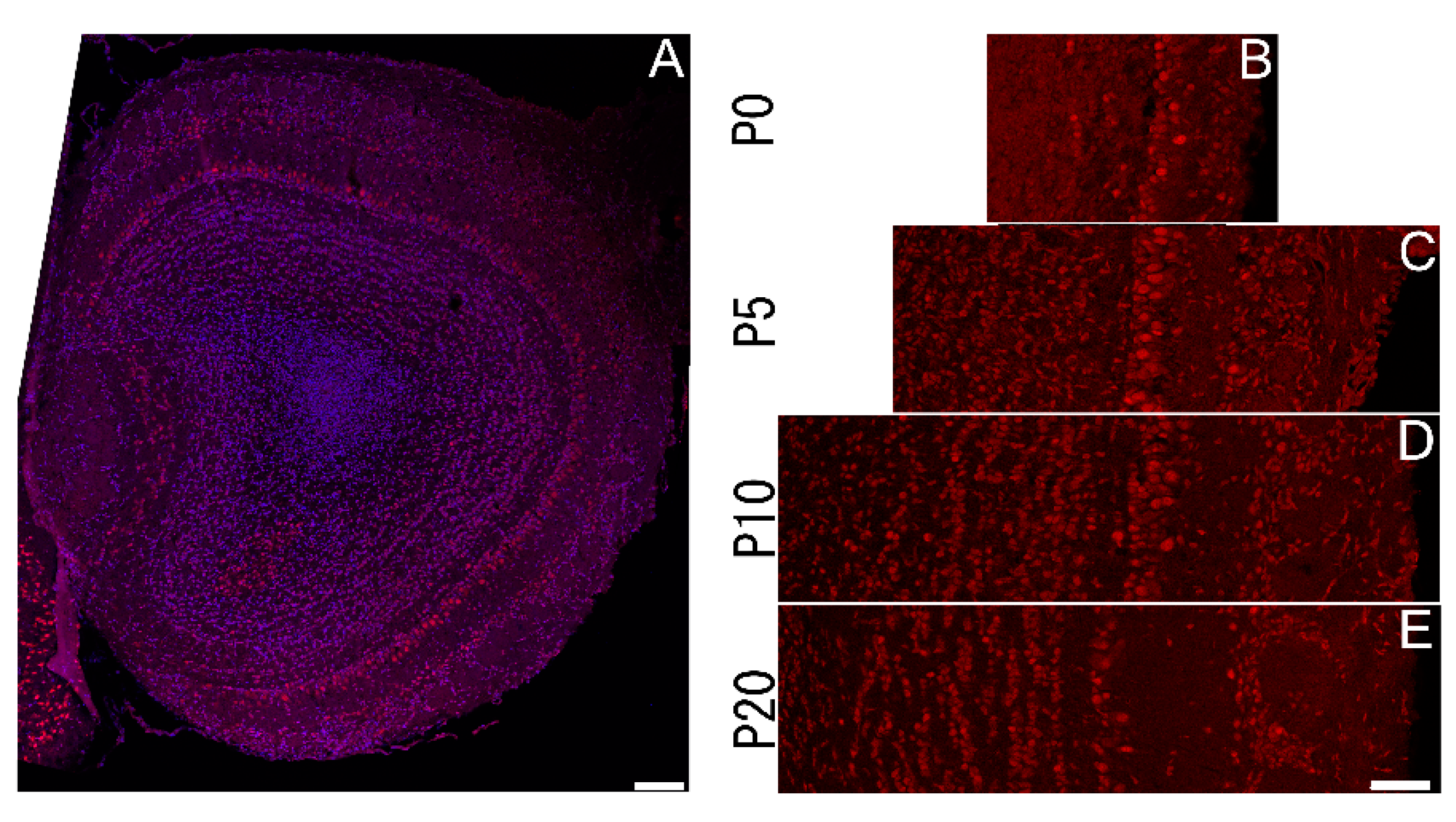
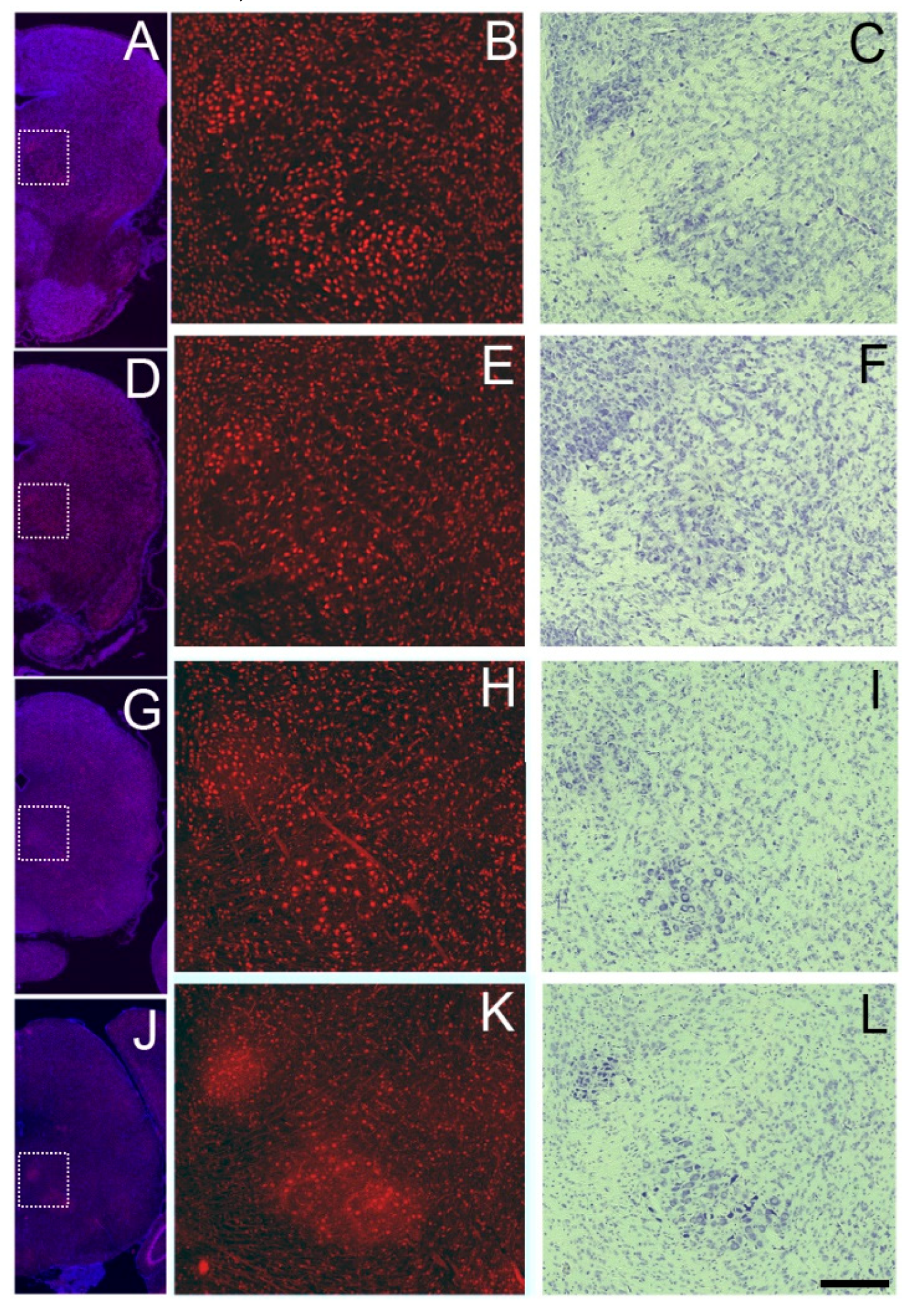
3.4. Sbno1 Expression in the Olfactory Bulb
3.5. Sbno1 Expression in the Cerebellum
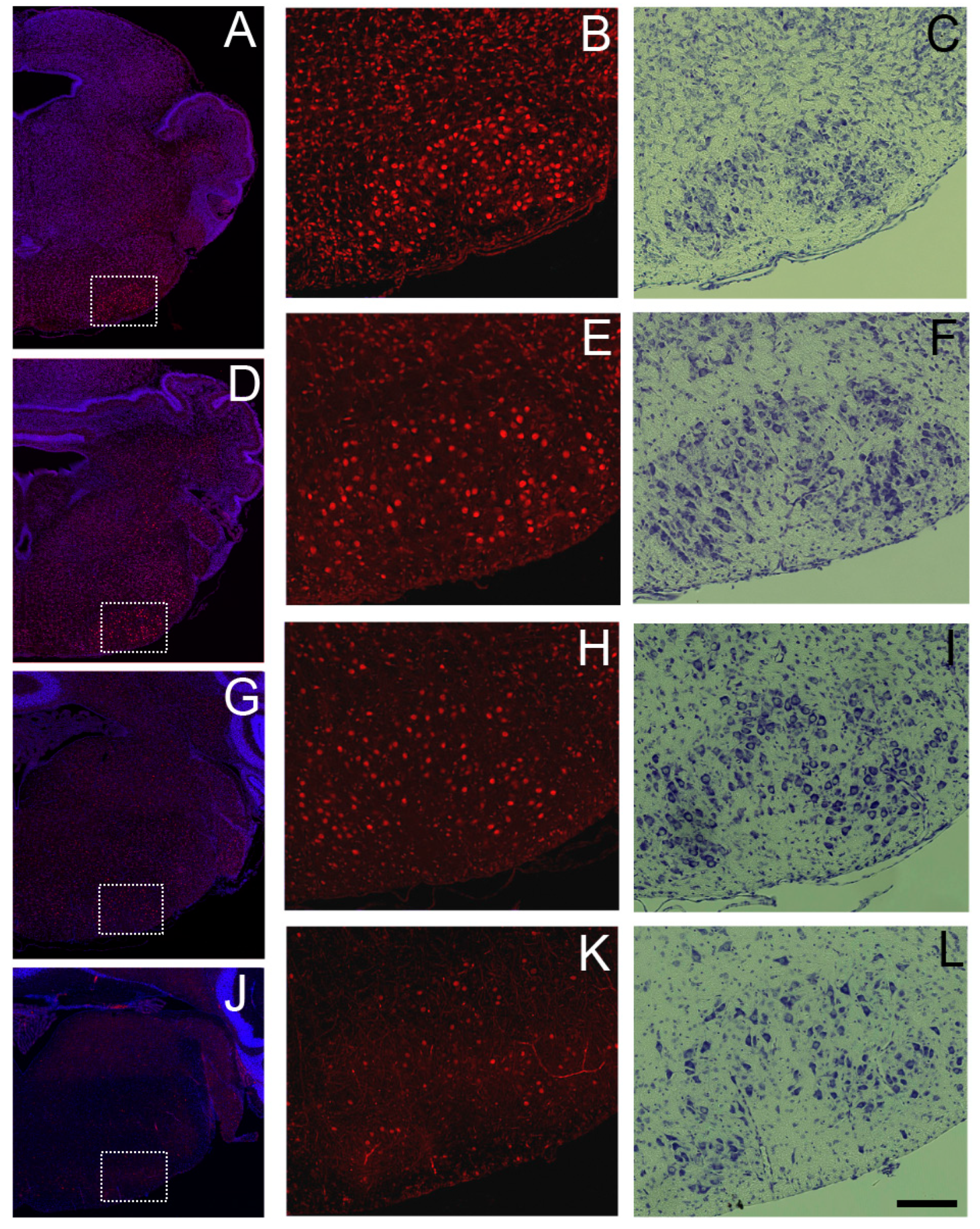
3.6. Sbno1 Expression in the Brainstem
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hilker, R.; et al. Heritability of Schizophrenia and Schizophrenia Spectrum Based on the Nationwide Danish Twin Register. Biol Psychiatry 2018, 83, 492–498. [Google Scholar] [CrossRef]
- Pan, P.Y.; et al. Genetic and environmental contributions to co-occurring physical health conditions in autism spectrum condition and attention-deficit/hyperactivity disorder. Mol Autism. 2023, 14, 17. [Google Scholar] [CrossRef]
- Girard, S.L.; et al. Increased exonic de novo mutation rate in individuals with schizophrenia. Nat Genet. 2011, 43, 860–863. [Google Scholar] [CrossRef]
- Ripke, S.; et al. Biological insights from 108 schizophrenia-associated genetic loci. Nature 2014, 511, 421–427. [Google Scholar] [CrossRef]
- Reay, W.R.; Cairns, M.J. Pairwise common variant meta-analyses of schizophrenia with other psychiatric disorders reveals shared and distinct gene and gene-set associations. Transl Psychiatry. 2020, 10, 134. [Google Scholar] [CrossRef]
- de Onis, M.; Blössner, M. The World Health Organization Global Database on Child Growth and Malnutrition: methodology and applications. Int J Epidemiol. 2003, 32, 518–526. [Google Scholar] [CrossRef]
- Taal, H.R.; et al. Common variants at 12q15 and 12q24 are associated with infant head circumference. Nat Genet. 2012, 44, 532–538. [Google Scholar] [CrossRef]
- Bulayeva, K.; et al. Genomic structural variants are linked with intellectual disability. J Neural Transm. 2015, 122, 1289–301. [Google Scholar] [CrossRef]
- Thyme, S.B.; et al. Phenotypic Landscape of Schizophrenia-Associated Genes Defines Candidates and Their Shared Functions. Cell, 2019, 177, 478–491. [Google Scholar] [CrossRef]
- Coyle-Thompson, C.A.; Banerjee, U. The strawberry notch gene functions with Notch in common developmental pathways. Development 1993, 119, 377–395. [Google Scholar] [CrossRef]
- Majumdar, A.; Nagaraj, R.; Banerjee, U. strawberry notch encodes a conserved nuclear protein that functions downstream of Notch and regulates gene expression along the developing wing margin of Drosophila. Genes Dev 1997, 11, 1341–1353. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, L.; Nagaraj, R.; Zipursky, S.L.; Banerjee, U. An EGFR/Ebi/Sno pathway promotes delta expression by inactivating Su(H)/SMRTER repression during inductive notch signaling. Cell 2002, 110, 625–637. [Google Scholar] [CrossRef] [PubMed]
- Takano, A.; Zochi, R.; Hibi, M.; Terashima, T.; Katsuyama, Y. Expression of strawberry notch family genes during zebrafish embryogenesis. Dev Dyn. 2010, 239, 1789–1796. [Google Scholar] [CrossRef] [PubMed]
- Erkhembaatar, M.; et al. Involvement of strawberry notch homologue 1 in neurite outgrowth of cortical neurons. Dev Growth Differ 2022, 64, 379–394. [Google Scholar] [CrossRef]
- Paxinos, G.; Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates. 2012, United States of America: Academic press.
- Sekine, A.; et al. Prominin-1/CD133 expression as potential tissue-resident vascular endothelial progenitor cells in the pulmonary circulation. Am J Physiol Lung Cell Mol Physiol. 2016, 310, L1130–L1142. [Google Scholar] [CrossRef]
- Nimchinsky, E.A.; et al. Differential vulnerability of oculomotor, facial, and hypoglossal nuclei in G86R superoxide dismutase transgenic mice. J Comp Neurol. 2000, 416, 112–125. [Google Scholar] [CrossRef]
- Abdelhaleem, M.; Maltais, L.; Wain, H. The human DDX and DHX gene families of putative RNA helicases. Genomics 2003, 81, 618–622. [Google Scholar] [CrossRef]
- Fuller-Pace, F.V. DExD/H box RNA helicases: multifunctional proteins with important roles in transcriptional regulation. Nucleic Acids Res 2006, 34, 4206–4215. [Google Scholar] [CrossRef]
- Givogri, M.I.; Costa, R.M.; Schonmann, V.; Silva, A.J.; Campagnoni, A.T.; Bongarzone, E.R. Central nervous system myelination in mice with deficient expression of Notch1 receptor. J Neurosci Res 2002, 67, 309–320. [Google Scholar] [CrossRef]
- Woodhoo, A.; et al. Notch controls embryonic Schwann cell differentiation, postnatal myelination and adult plasticity. Nat Neurosci 2009, 2009. 12, 839–847. [Google Scholar] [CrossRef]
- Arzate, D.M.; et al. Dll1 haploinsufficiency causes brain abnormalities with functional relevance. Front Neurosci. 2022, 16, 951418. [Google Scholar] [CrossRef] [PubMed]
- L. Redmond, L.; Oh, S.R.; Hicks, C.; Weinmaster, G.; Ghosh, A. Nuclear Notch1 signaling and the regulation of dendritic development. Nat Neurosci 2000, 3, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Bonini, S.A.; et al. Nuclear factor κB-dependent neurite remodeling is mediated by Notch pathway. J Neurosci. 2011, 31, 11697–705. [Google Scholar] [CrossRef] [PubMed]
- Sestan, N.; Artavanis-Tsakonas, S.; Rakic, P. Contact-dependent inhibition of cortical neurite growth mediated by notch signaling. Science 1999, 286, 741–746. [Google Scholar] [CrossRef]
- Son, A.I.; et al. Dual Role of Rbpj in the Maintenance of Neural Progenitor Cells and Neuronal Migration in Cortical Development. Cereb Cortex. 2020, 30, 6444–6457. [Google Scholar] [CrossRef]
- Turetsky, B.I.; Hahn, C.G.; Borgmann-Winter, K.; Moberg, P.J. Scents and nonsense: olfactory dysfunction in schizophrenia. Schizophr Bull. 2009, 35, 1117–1131. [Google Scholar] [CrossRef]
- Sheng, M.; Greenberg, M.E. The regulation and function of c-fos and other immediate early genes in the nervous system. Neuron 1990, 4, 477–485. [Google Scholar] [CrossRef]
- Pollina, E.A.; et al. A NPAS4-NuA4 complex couples synaptic activity to DNA repair. Nature 2023, 614, 732–741. [Google Scholar] [CrossRef]
- H. Lu, H.; Davis, A.J. Human RecQ Helicases in DNA Double-Strand Break Repair. Front Cell Dev Biol, 2021, 9, 640755. [Google Scholar] [CrossRef]
- Dai, J.; et al. The Role of SF1 and SF2 Helicases in Biotechnological Applications. Appl Biochem Biotechnol. [CrossRef]
- Zolzaya, S.; Narumoto, A.; Katsuyama, Y. Genomic variation in neurons. Dev Growth Differ 2024, 66, 35–42. [Google Scholar] [CrossRef]
- Baba, K.; Dekimoto, H.; Muraoka, D.; Agata, K.; Terashima, T.; Katsuyama, Y. A mouse homologue of Strawberry Notch is transcriptionally regulated by Reelin signal. Biochem Biophys Res Commun 2006, 350, 842–849. [Google Scholar] [CrossRef]
| Region | Stage | ||||
| P0 | P3 | P5 | P10 | P20 | |
| Olfactory bulb | |||||
| Granule cell layer | + | + | + | + | + |
| Internal plexiform layer | + | + | + | + | + |
| Mitral cell layer | +++ | +++ | +++ | +++ | +++ |
| External plexiform layer | + | + | + | + | + |
| Glomerular layer | + | + | ++ | ++ | ++ |
| Cerebral cortex | |||||
| Layer I | ± | ± | ± | ± | ± |
| Layer II/III | ++ | ++ | ++ | +++ | +++ |
| Layer IV | ++ | ++ | ++ | ++ | + |
| Layer V | +++ | +++ | ++ | ++ | + |
| Layer VI a | ++ | ++ | ++ | ++ | + |
| Layer VI b | +++ | +++ | + | + | + |
| Hippocampus | |||||
| Dentate gyrus | |||||
| Molecular layer | ± | ± | ± | ± | ± |
| Granule cell layer | + | + | + | + | ++ |
| Hilus | ++ | ++ | ++ | ++ | + |
| CA3 | |||||
| S. oriens | ± | ± | ± | ± | ± |
| S. pyramidale | +++ | +++ | +++ | +++ | +++ |
| S. radiatum | ± | ± | ± | ± | ± |
| CA1 | |||||
| S. oriens | ± | ± | ± | ± | ± |
| S. pyramidale | ++ | ++ | ++ | ++ | ++ |
| S. radiatum | ± | ± | ± | ± | ± |
| Piriform cortex | |||||
| Layer I | + | + | + | + | + |
| Layer II | ++ | ++ | ++ | ++ | ++ |
| Layer III | +++ | +++ | +++ | +++ | +++ |
| Basal ganglia | ++ | + | + | + | + |
| Thalamic nuclei | + | + | + | + | + |
| Superior colliculus | + | + | + | + | + |
| Red nucleus | ++++ | ++++ | ++++ | ++++ | ++++ |
| Inferior colliculus | + | + | + | + | + |
| Cerebellum | |||||
| External granular layer | + | + | + | + | |
| Molecular layer | ++ | ||||
| Purkinje cell layer | + | + | + | ++ | ++ |
| Internal granular layer | + | ++ | ++ | +++ | +++ |
| Deep cerebellar nucleus | ++ | ++ | |||
| Pontine nucleus | + | + | + | + | + |
| Inferior olivery nucleus | ++ | ++ | ++ | ++ | ++ |
| Dorsal cochlear nucleus | + | + | + | + | + |
| Vestibular nucleus | + | + | + | + | + |
| Motor nuclei of brainstem | |||||
| Oculomotor nucleus | ++++ | ++++ | ++++ | ++++ | ++++ |
| Facial nucleus | +++ | ++++ | ++++ | +++ | + |
| Trigeminal motor nucleus | ++++ | ++++ | ++++ | ++++ | ++ |
| Hypoglossal nucleus | +++ | ++++ | ++++ | ++++ | ++++ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




