Submitted:
05 November 2024
Posted:
06 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Isolation of Peripheral Blood Neutrophils
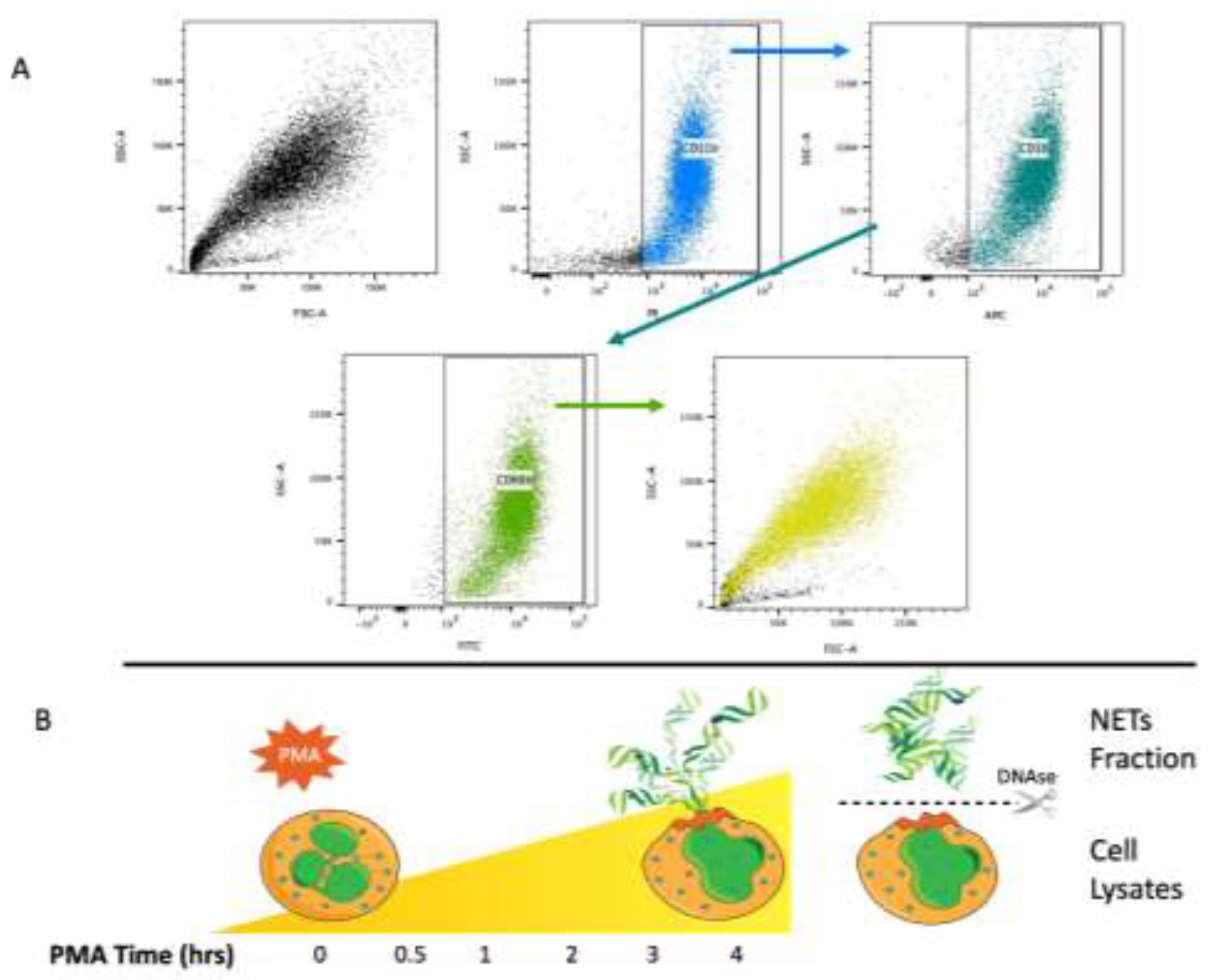
2.3. Flow Cytometry for Neutrophil Quality Assessment
2.4. Neutrophil Cell Culture
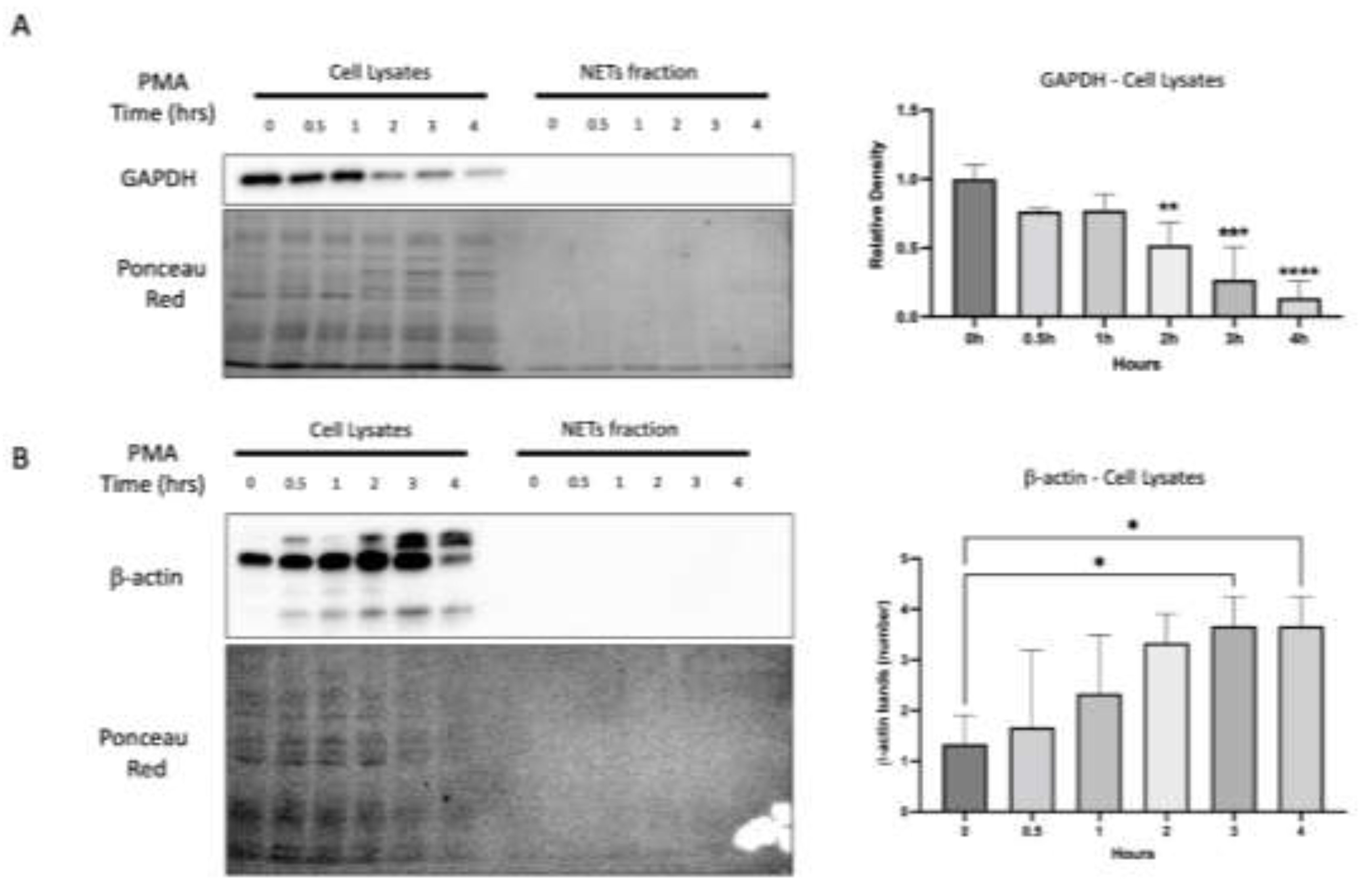
2.5. Western Blot Assay
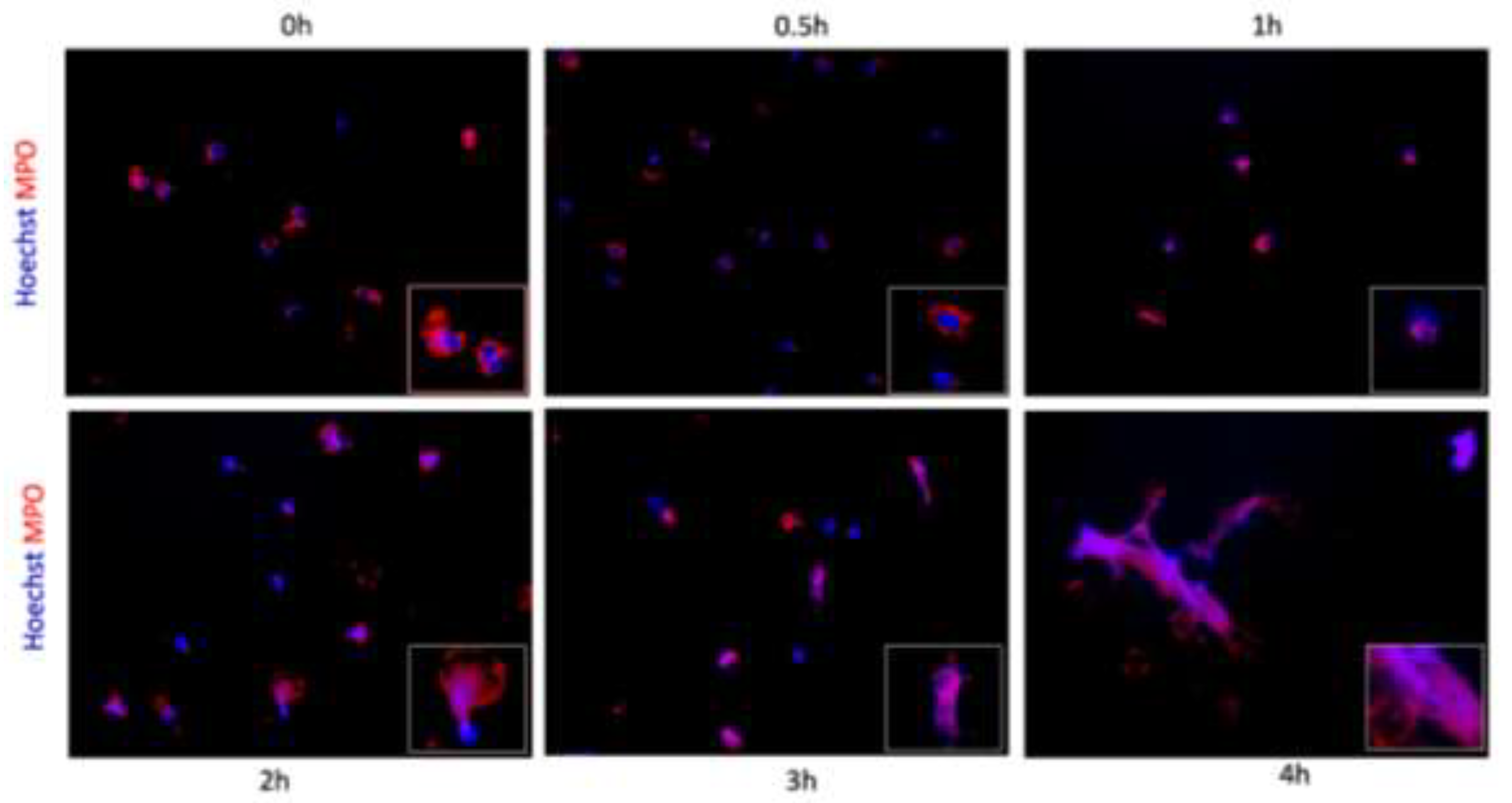
2.6. Immunofluorescence
2.7. Statistical Analysis
3. Results
3.1. Sample Processing
3.2. NETs Release Verification
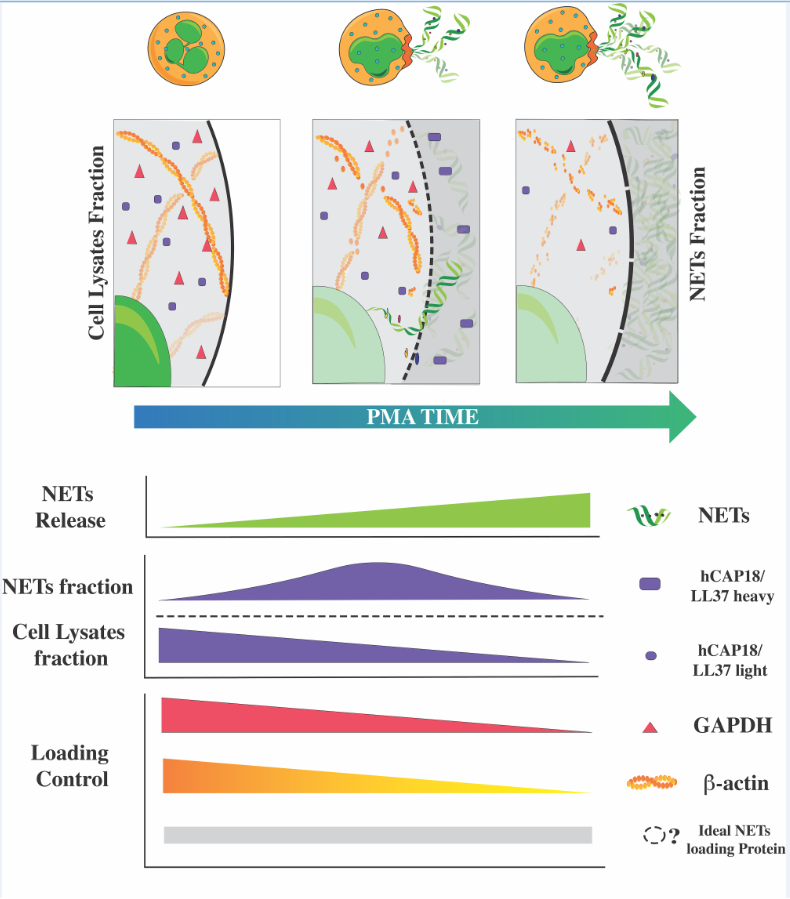
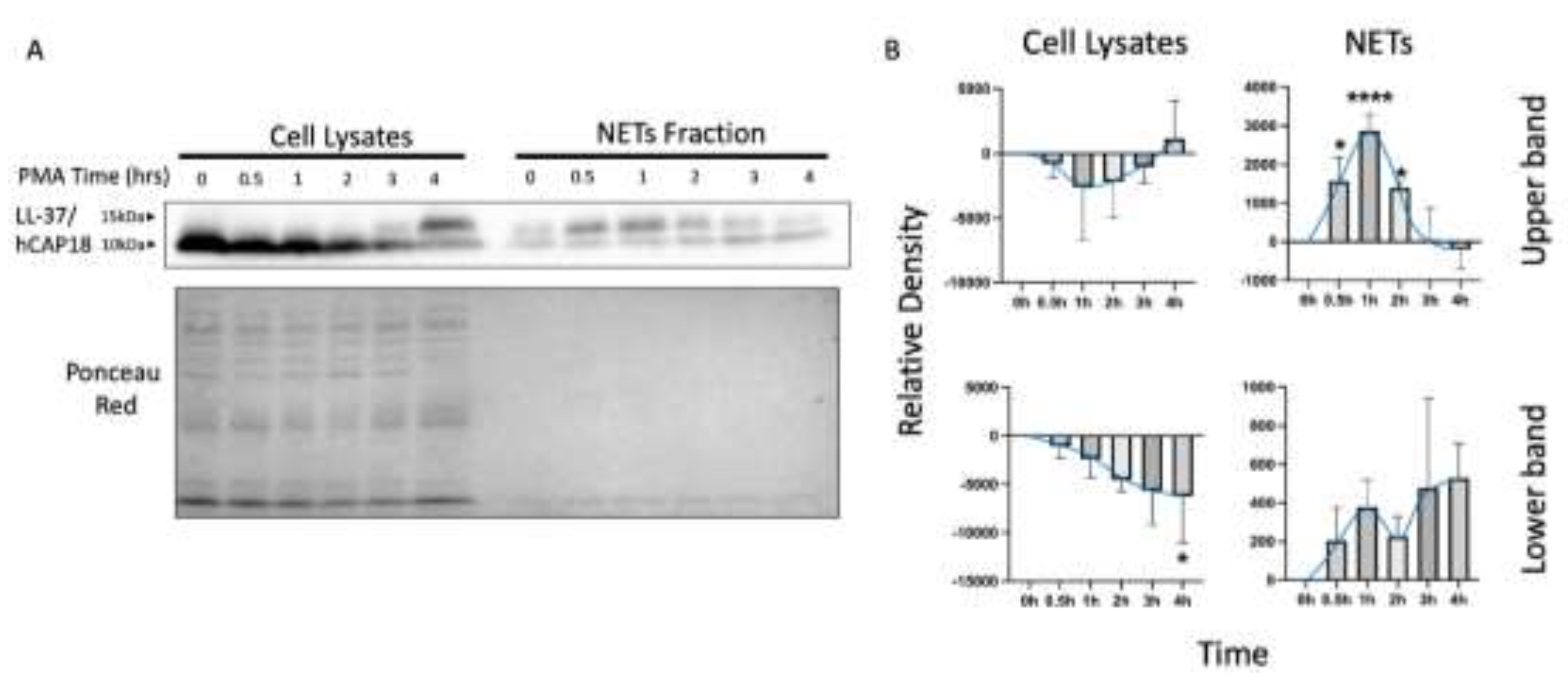
3.3. Loading Control and NETs-Associated Protein (NAPs) Assessment
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mayadas, T.N.; Cullere, X.; Lowell, C.A. The Multifaceted Functions of Neutrophils. Annu Rev Pathol 2014, 9, 181–218. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil Extracellular Traps Kill Bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Kolaczkowska, E.; Kubes, P. Neutrophil Recruitment and Function in Health and Inflammation. Nat Rev Immunol 2013, 13, 159–175. [Google Scholar] [CrossRef]
- Scieszka, D.; Lin, Y.-H.; Li, W.; Choudhury, S.; Yu, Y.; Freire, M. NETome: A Model to Decode the Human Genome and Proteome of Neutrophil Extracellular Traps. Sci Data 2022, 9, 702. [Google Scholar] [CrossRef]
- Tan, C.; Aziz, M.; Wang, P. The Vitals of NETs. J Leukoc Biol 2021, 110, 797–808. [Google Scholar] [CrossRef]
- Bruschi, M.; Petretto, A.; Santucci, L.; Vaglio, A.; Pratesi, F.; Migliorini, P.; Bertelli, R.; Lavarello, C.; Bartolucci, M.; Candiano, G.; et al. Neutrophil Extracellular Traps Protein Composition Is Specific for Patients with Lupus Nephritis and Includes Methyl-Oxidized Aenolase (Methionine Sulfoxide 93). Sci Rep 2019, 9, 7934. [Google Scholar] [CrossRef] [PubMed]
- Petretto, A.; Bruschi, M.; Pratesi, F.; Croia, C.; Candiano, G.; Ghiggeri, G.; Migliorini, P. Neutrophil Extracellular Traps (NET) Induced by Different Stimuli: A Comparative Proteomic Analysis. PLoS One 2019, 14, e0218946. [Google Scholar] [CrossRef]
- Frangou, E.; Vassilopoulos, D.; Boletis, J.; Boumpas, D.T. An Emerging Role of Neutrophils and NETosis in Chronic Inflammation and Fibrosis in Systemic Lupus Erythematosus (SLE) and ANCA-Associated Vasculitides (AAV): Implications for the Pathogenesis and Treatment. Autoimmun Rev 2019, 18, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Arampatzioglou, A.; Papazoglou, D.; Konstantinidis, T.; Chrysanthopoulou, A.; Mitsios, A.; Angelidou, I.; Maroulakou, I.; Ritis, K.; Skendros, P. Clarithromycin Enhances the Antibacterial Activity and Wound Healing Capacity in Type 2 Diabetes Mellitus by Increasing LL-37 Load on Neutrophil Extracellular Traps. Front Immunol 2018, 9, 2064. [Google Scholar] [CrossRef]
- Pilsczek, F.H.; Salina, D.; Poon, K.K.H.; Fahey, C.; Yipp, B.G.; Sibley, C.D.; Robbins, S.M.; Green, F.H.Y.; Surette, M.G.; Sugai, M.; et al. A Novel Mechanism of Rapid Nuclear Neutrophil Extracellular Trap Formation in Response to Staphylococcus Aureus. J Immunol 2010, 185, 7413–7425. [Google Scholar] [CrossRef]
- Sha, L.-L.; Wang, H.; Wang, C.; Peng, H.-Y.; Chen, M.; Zhao, M.-H. Autophagy Is Induced by Anti-Neutrophil Cytoplasmic Abs and Promotes Neutrophil Extracellular Traps Formation. Innate Immun 2016, 22, 658–665. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Sha, L.-L.; Ma, T.-T.; Zhang, L.-X.; Chen, M.; Zhao, M.-H. Circulating Level of Neutrophil Extracellular Traps Is Not a Useful Biomarker for Assessing Disease Activity in Antineutrophil Cytoplasmic Antibody-Associated Vasculitis. PLoS One 2016, 11, e0148197. [Google Scholar] [CrossRef] [PubMed]
- Rycyk-Bojarzynska, A.; Kasztelan-Szczerbinska, B.; Cichoz-Lach, H.; Surdacka, A.; Rolinski, J. Neutrophil PAD4 Expression and Its Pivotal Role in Assessment of Alcohol-Related Liver Disease. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Monti, M.; Iommelli, F.; De Rosa, V.; Carriero, M.V.; Miceli, R.; Camerlingo, R.; Di Minno, G.; Del Vecchio, S. Integrin-Dependent Cell Adhesion to Neutrophil Extracellular Traps through Engagement of Fibronectin in Neutrophil-like Cells. PLoS One 2017, 12, e0171362. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yang, Y.; Gan, T.; Li, Y.; Hu, F.; Hao, N.; Yuan, B.; Chen, Y.; Zhang, M. Lung Cancer Cells Release High Mobility Group Box 1 and Promote the Formation of Neutrophil Extracellular Traps. Oncol Lett 2019, 18, 181–188. [Google Scholar] [CrossRef]
- Watanabe-Kusunoki, K.; Abe, N.; Nakazawa, D.; Karino, K.; Hattanda, F.; Fujieda, Y.; Nishio, S.; Yasuda, S.; Ishizu, A.; Atsumi, T. A Case Report Dysregulated Neutrophil Extracellular Traps in a Patient with Propylthiouracil-Induced Anti-Neutrophil Cytoplasmic Antibody-Associated Vasculitis. Medicine 2019, 98, e15328. [Google Scholar] [CrossRef]
- Domingo-Gonzalez, R.; Martínez-Colón, G.J.; Smith, A.J.; Smith, C.K.; Ballinger, M.N.; Xia, M.; Murray, S.; Kaplan, M.J.; Yanik, G.A.; Moore, B.B. Inhibition of Neutrophil Extracellular Trap Formation after Stem Cell Transplant by Prostaglandin E2. Am J Respir Crit Care Med 2016, 193, 186–197. [Google Scholar] [CrossRef]
- Kahlenberg, J.M.; Carmona-Rivera, C.; Smith, C.K.; Kaplan, M.J. Neutrophil Extracellular Trap-Associated Protein Activation of the NLRP3 Inflammasome Is Enhanced in Lupus Macrophages. J Immunol 2013, 190, 1217–1226. [Google Scholar] [CrossRef]
- McInturff, A.M.; Cody, M.J.; Elliott, E.A.; Glenn, J.W.; Rowley, J.W.; Rondina, M.T.; Yost, C.C. Mammalian Target of Rapamycin Regulates Neutrophil Extracellular Trap Formation via Induction of Hypoxia-Inducible Factor 1 α. Blood 2012, 120, 3118–3125. [Google Scholar] [CrossRef]
- Barr, F.D.; Ochsenbauer, C.; Wira, C.R.; Rodriguez-Garcia, M. Neutrophil Extracellular Traps Prevent HIV Infection in the Female Genital Tract. Mucosal Immunol 2018, 11, 1420–1428. [Google Scholar] [CrossRef]
- Neumann, A.; Völlger, L.; Berends, E.T.M.; Molhoek, E.M.; Stapels, D.A.C.; Midon, M.; Friães, A.; Pingoud, A.; Rooijakkers, S.H.M.; Gallo, R.L.; et al. Novel Role of the Antimicrobial Peptide LL-37 in the Protection of Neutrophil Extracellular Traps against Degradation by Bacterial Nucleases. J Innate Immun 2014, 6, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Zhang, S.; Zhao, Y.; Liu, Y.; Yan, B. Neutrophil Extracellular Traps May Contribute to Interstitial Lung Disease Associated with Anti-MDA5 Autoantibody Positive Dermatomyositis. Clin Rheumatol 2018, 37, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Lande, R.; Ganguly, D.; Facchinetti, V.; Frasca, L.; Conrad, C.; Gregorio, J.; Meller, S.; Chamilos, G.; Sebasigari, R.; Riccieri, V.; et al. Neutrophils Activate Plasmacytoid Dendritic Cells by Releasing Self-DNA-Peptide Complexes in Systemic Lupus Erythematosus. Sci Transl Med 2011, 3, 73ra19. [Google Scholar] [CrossRef] [PubMed]
- Lakschevitz, F.S.; Hassanpour, S.; Rubin, A.; Fine, N.; Sun, C.; Glogauer, M. Identification of Neutrophil Surface Marker Changes in Health and Inflammation Using High-Throughput Screening Flow Cytometry. Exp Cell Res 2016, 342, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Sirover, M.A. Subcellular Dynamics of Multifunctional Protein Regulation: Mechanisms of GAPDH Intracellular Translocation. J Cell Biochem 2012, 113, 2193–2200. [Google Scholar] [CrossRef]
- Li, Y.; Hook, J.S.; Ding, Q.; Xiao, X.; Chung, S.S.; Mettlen, M.; Xu, L.; Moreland, J.G.; Agathocleous, M. Neutrophil Metabolomics in Severe COVID-19 Reveal GAPDH as a Suppressor of Neutrophil Extracellular Trap Formation. Nat Commun 2023, 14, 2610. [Google Scholar] [CrossRef]
- Metzler, K.D.; Goosmann, C.; Lubojemska, A.; Zychlinsky, A.; Papayannopoulos, V. A Myeloperoxidase-Containing Complex Regulates Neutrophil Elastase Release and Actin Dynamics during NETosis. Cell Rep 2014, 8, 883–896. [Google Scholar] [CrossRef]
- Thiam, H.R.; Wong, S.L.; Qiu, R.; Kittisopikul, M.; Vahabikashi, A.; Goldman, A.E.; Goldman, R.D.; Wagner, D.D.; Waterman, C.M. NETosis Proceeds by Cytoskeleton and Endomembrane Disassembly and PAD4-Mediated Chromatin Decondensation and Nuclear Envelope Rupture. Proc Natl Acad Sci U S A 2020, 117, 7326–7337. [Google Scholar] [CrossRef]
- Murakami, M.; Ohtake, T.; Dorschner, R.A.; Gallo, R.L. Cathelicidin Antimicrobial Peptides Are Expressed in Salivary Glands and Saliva. J Dent Res 2002, 81, 845–850. [Google Scholar] [CrossRef]
- Geitani, R.; Moubareck, C.A.; Costes, F.; Marti, L.; Dupuis, G.; Sarkis, D.K.; Touqui, L. Bactericidal Effects and Stability of LL-37 and CAMA in the Presence of Human Lung Epithelial Cells. Microbes Infect 2022, 24, 104928. [Google Scholar] [CrossRef]
- Sollberger, G.; Choidas, A.; Burn, G.L.; Habenberger, P.; Di Lucrezia, R.; Kordes, S.; Menninger, S.; Eickhoff, J.; Nussbaumer, P.; Klebl, B.; et al. Gasdermin D Plays a Vital Role in the Generation of Neutrophil Extracellular Traps. Sci Immunol 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, Y.; Chen, Z.; Yuan, Y.; Su, Q.; Ye, K.; Chen, C.; Li, G.; Song, Y.; Chen, H.; et al. GSDMD-Dependent Neutrophil Extracellular Traps Promote Macrophage-to-Myofibroblast Transition and Renal Fibrosis in Obstructive Nephropathy. Cell Death Dis 2022, 13, 693. [Google Scholar] [CrossRef] [PubMed]
- Santa Cruz Garcia, A.B.; Schnur, K.P.; Malik, A.B.; Mo, G.C.H. Gasdermin D Pores Are Dynamically Regulated by Local Phosphoinositide Circuitry. Nat Commun 2022, 13, 52. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Zhang, Z.; Magupalli, V.G.; Pablo, J.L.; Dong, Y.; Vora, S.M.; Wang, L.; Fu, T.-M.; Jacobson, M.P.; Greka, A.; et al. Gasdermin D Pore Structure Reveals Preferential Release of Mature Interleukin-1. Nature 2021, 593, 607–611. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



