Submitted:
04 November 2024
Posted:
05 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Location and Soil and Climatic Conditions
2.2. Experimental Design
2.3. Analytical Determinations
2.4. Statistical Analysis
3. Results
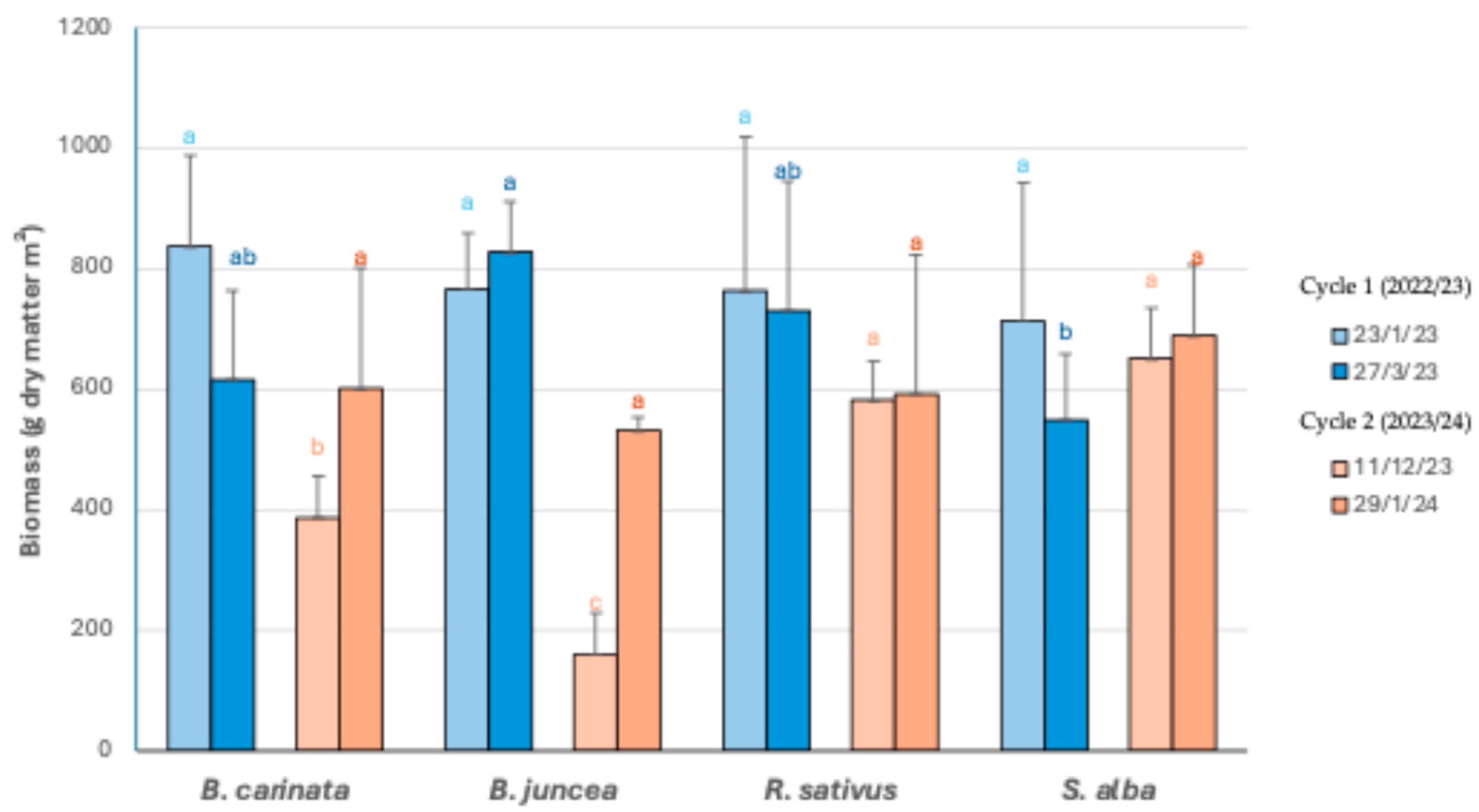
3.1. Agronomic Performance and Biomass Production
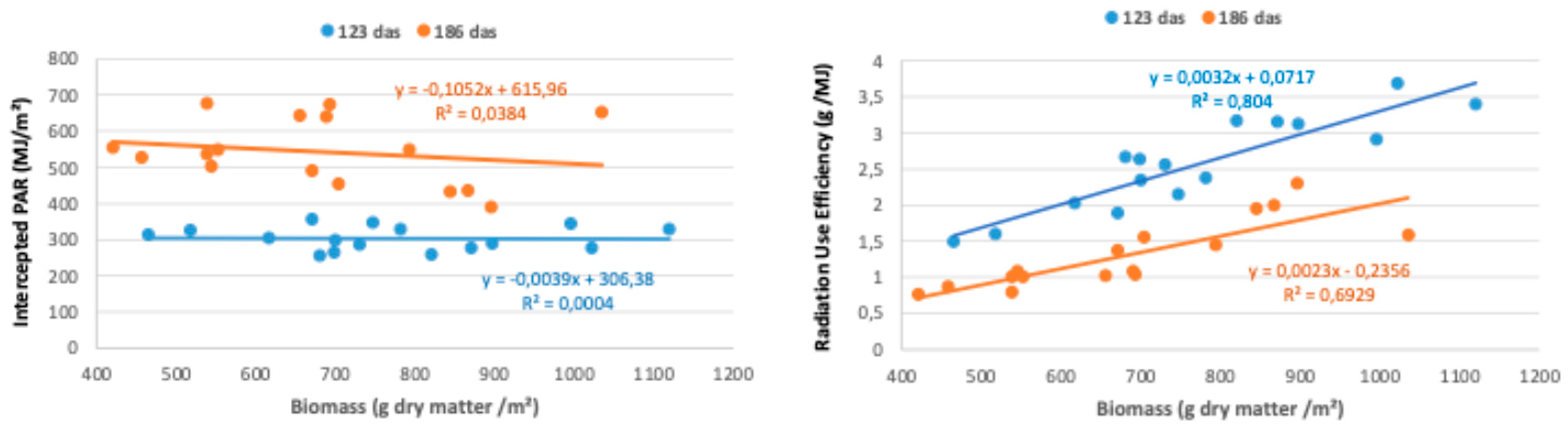
3.2. Radiation Interception and Radiation Use Efficiency
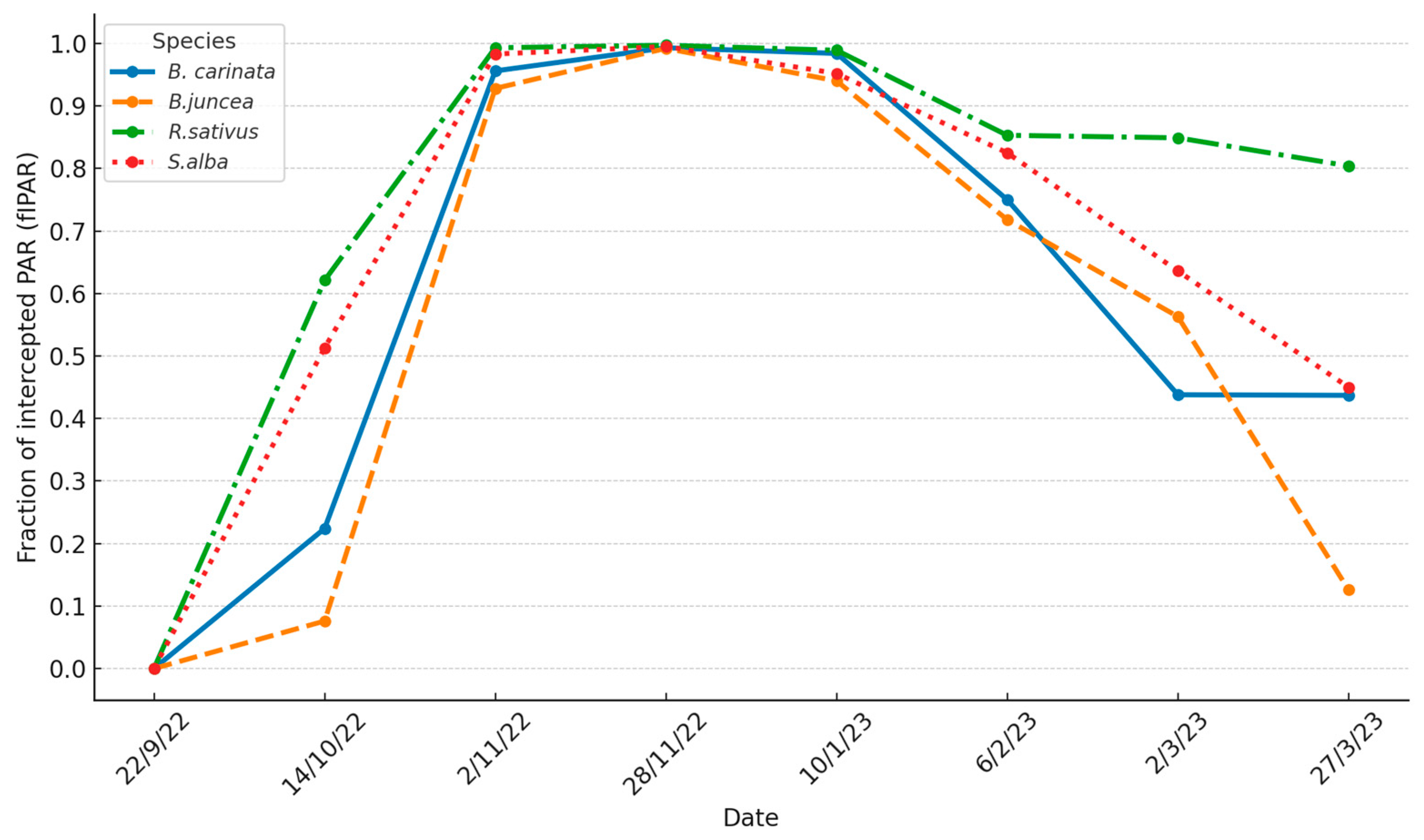
3.2.1. Fraction of Photosynthetically Active Radiation Intercepted by the Canopy (fIPAR)
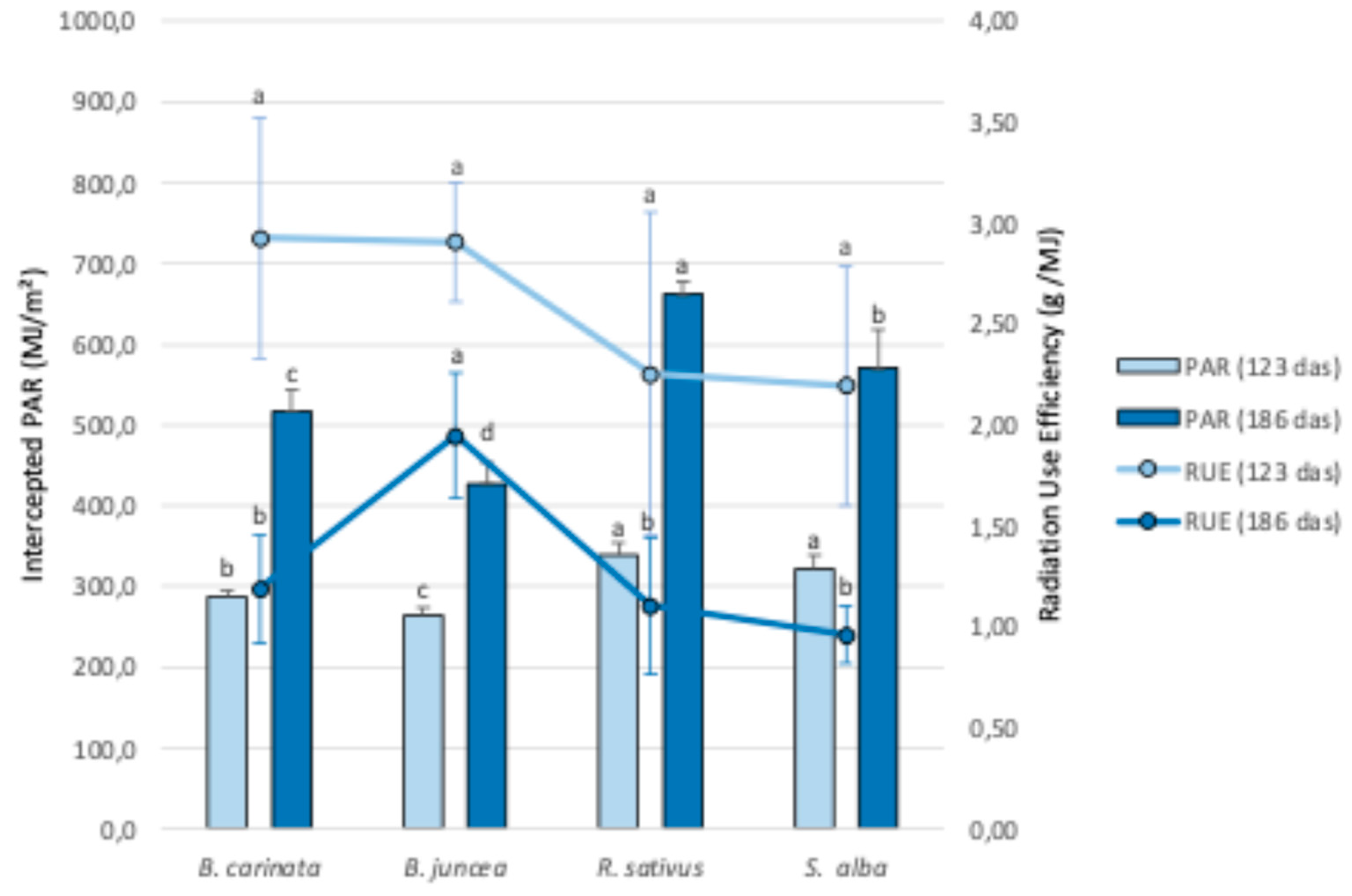
3.2.2. PAR Intercepted During the Cycle and Radiation Use Efficiency
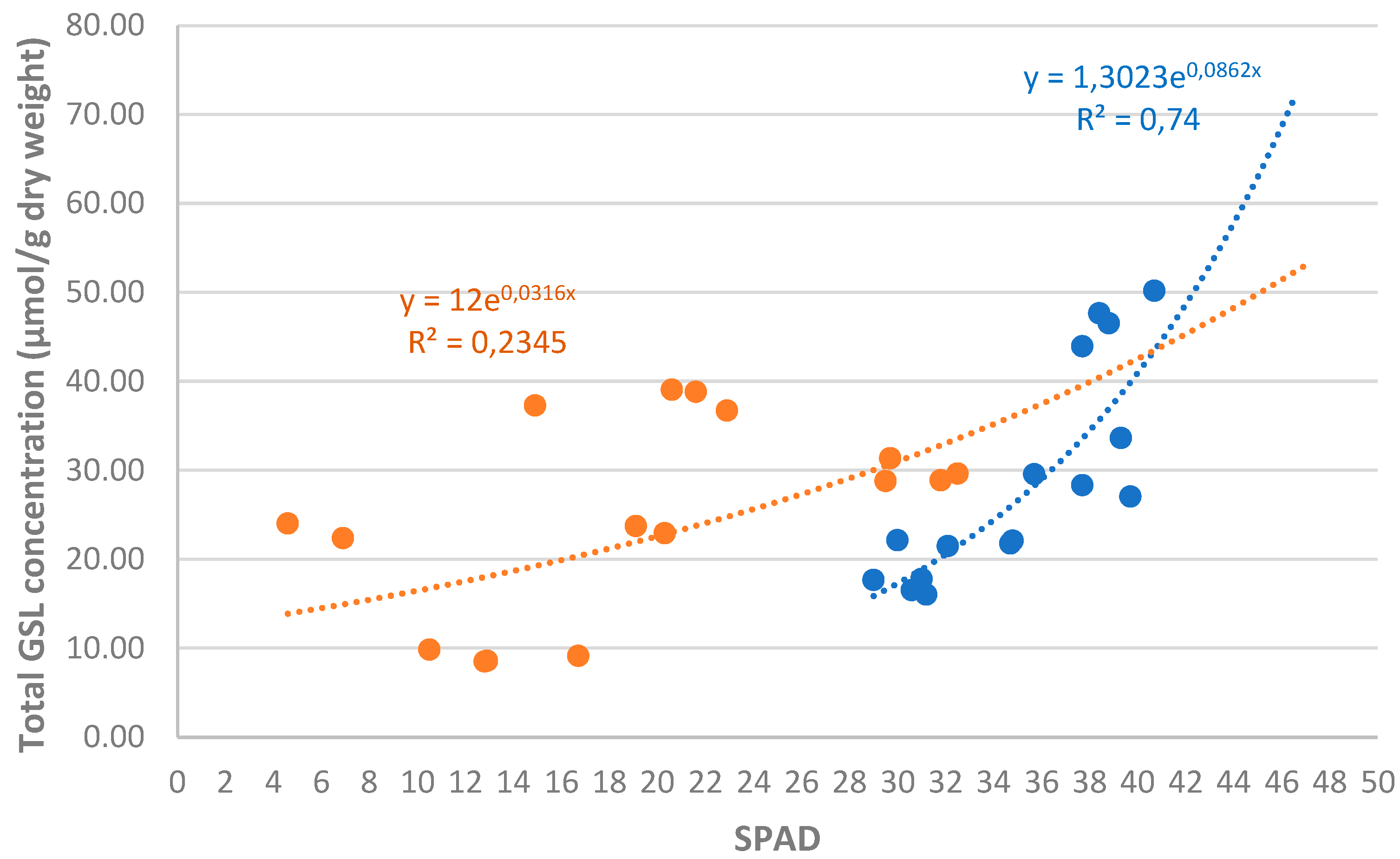
3.3. Chlorophyll Content Index
| Sampling date | 10-11-22 | 01-12-22 | 13-01-23 | 06-02-23 | 01-03-23 |
|---|---|---|---|---|---|
| B. carinata | 36,9 a1 | 37,2 a | 38,1 a | 40,5 a | 13,2 c |
| B. juncea | 37,3 a | 36,0 ab | 38,9 a | 37,4 ab | 20,0 b |
| R. sativus | 31,1 b | 31,2 c | 30,4 b | 33,0 b | 30,9 a |
| S. alba | 33,6 ab | 33,1 bc | 32,9 b | 25,2 c | 12,7 c |
| p-value | *2 | * | *** | *** | *** |
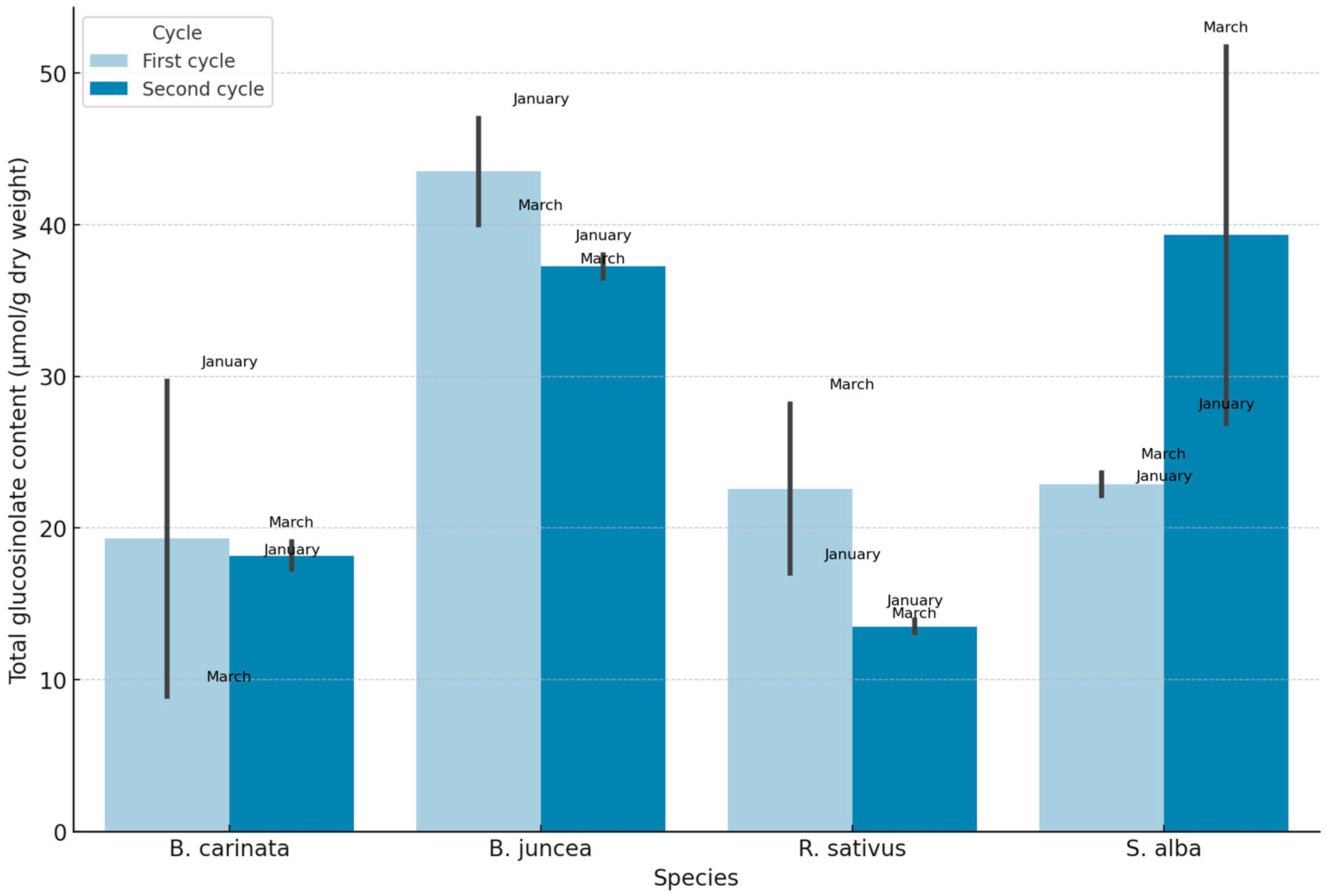
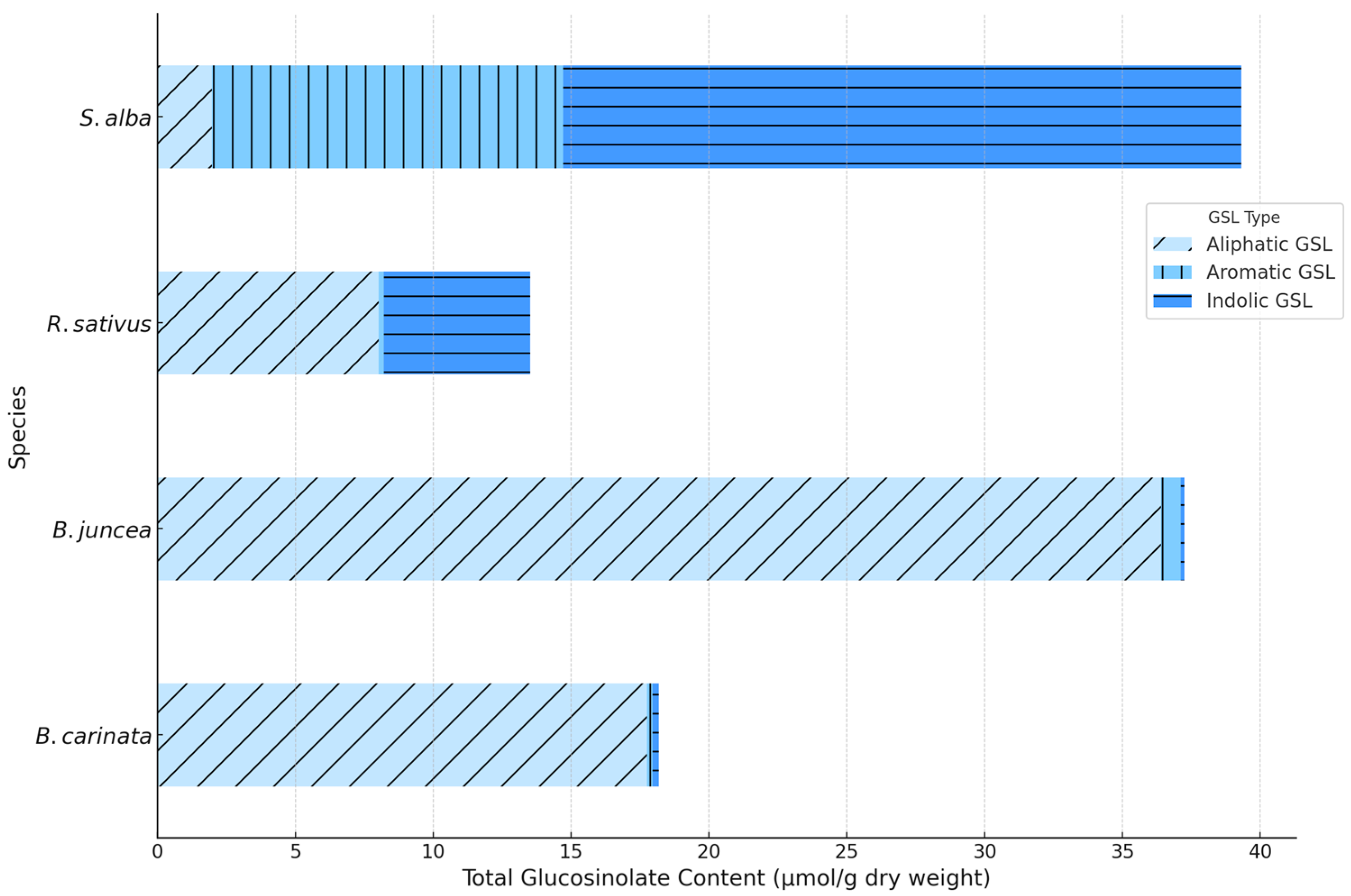
3.4. Glucosinolate Profiles and Isothiocyanate Potential
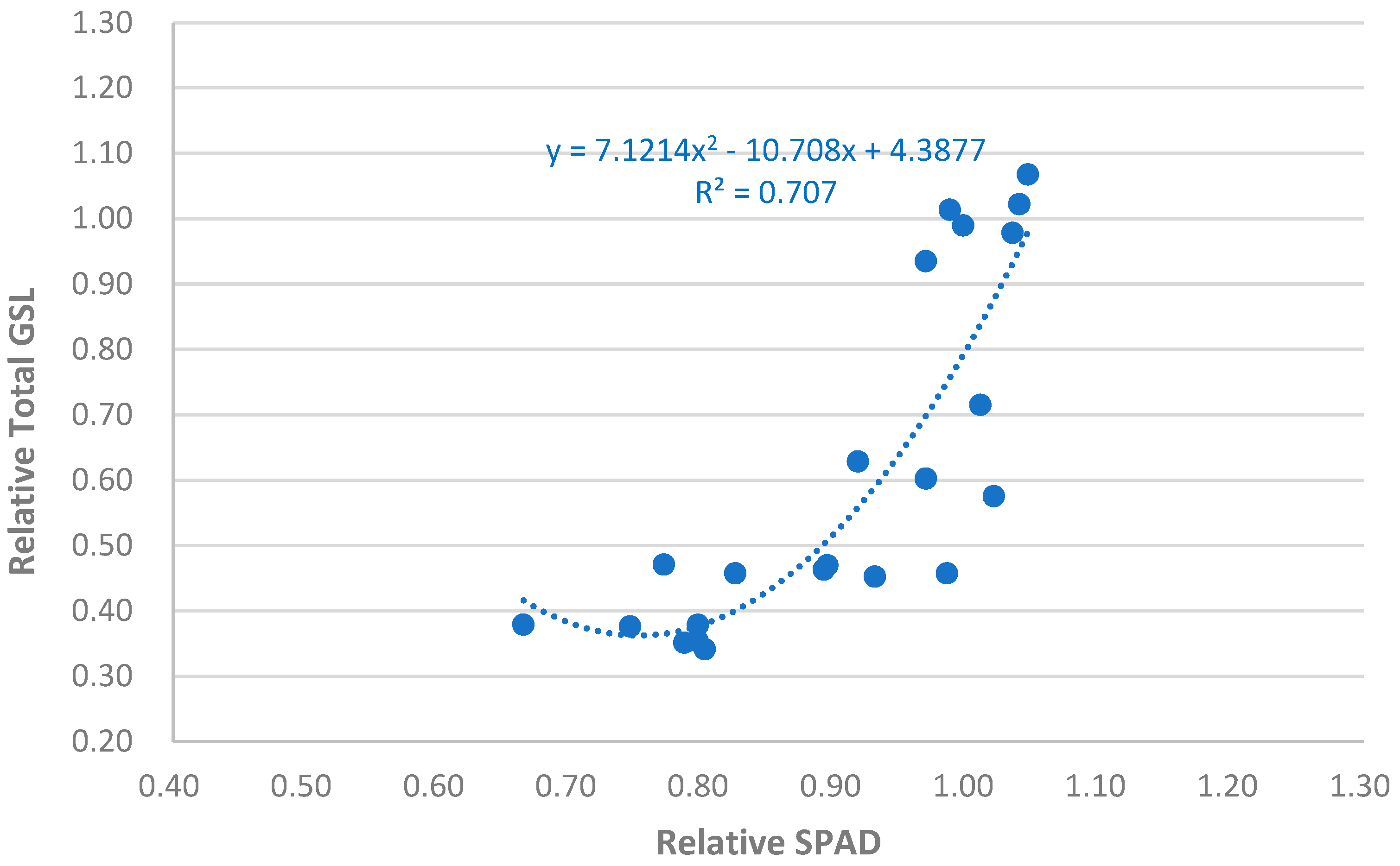
3.5. Correlation Analysis of Measured Variables
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Billen, G.; Aguilera, E.; Einarsson, R.; Garnier, J.; Gingrich, S.; Grizzetti, B.; Lassaletta, L.; Le Noë, J.; Sanz-Cobena, A. Beyond the Farm to Fork Strategy: Methodology for Designing a European Agro-Ecological Future. Science of the Total Environment 2024, 908, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Brennan, R. J. B.; Glaze-Corcoran, S.; Wick, R.; Hashemi, M. Biofumigation: An Alternative Strategy for the Control of Plant Parasitic Nematodes. Journal of Integrative Agriculture. Chinese Academy of Agricultural Sciences July 1, 2020, pp 1680–1690. [CrossRef]
- dos Santos, C. A.; de Souza, A. C.; Ferreira, M. G. Biofumigation with Species of the Brassicaceae Family: A Review. Ciencia Rural 2021, 51(1), 1–17. [Google Scholar] [CrossRef]
- Kirkegaard, J. A.; Gardner, P. A.; Desmarchelier, J. M.; Angus, J. F. Biofumigation - Using Brassica Species to Control Pests and Diseases in Horticulture and Agriculture. In 9th Australian Research Assembly on Brassicas; Wratten, N., Mailer, R. J., Eds.; Agricultural Research Institute,: Wagga Wagga, 1993; pp. 77–82. [Google Scholar]
- Dutta, T. K.; Khan, M. R.; Phani, V. Plant-Parasitic Nematode Management via Biofumigation Using Brassica and Non-Brassica Plants: Current Status and Future Prospects. Current Plant Biology. Elsevier B.V. January 1, 2019, pp 17–32. [CrossRef]
- Karavina, C.; Mandumbu, R. Biofumigation for Crop Protection: Potential for Adoption in Zimbabwe. J Anim Plant Sci 2012, 14(3), 1996–2005. [Google Scholar]
- Lazzeri, L.; Malaguti, L.; Cinti, S.; Ugolini, L.; De Nicola, G. R.; Bagatta, M.; Casadei, N.; D’Avino, L.; Matteo, R.; Patalano, G. The Brassicaceae Biofumigation System for Plant Cultivation and Defence. An Italian Twenty-Year Experience of Study and Application. Acta Hortic 2013, 1005, 375–382. [Google Scholar] [CrossRef]
- Ntalli, N.; Caboni, P. A Review of Isothiocyanates Biofumigation Activity on Plant Parasitic Nematodes. Phytochemistry Reviews 2017, 16(5), 827–834. [Google Scholar] [CrossRef]
- Wei, F.; Passey, T.; Xu, X. Effects of Individual and Combined Use of Bio-Fumigation-Derived Products on the Viability of Verticillium Dahliae Microsclerotia in Soil. Crop Protection 2016, 79, 170–176. [Google Scholar] [CrossRef]
- Goswami, B.; Pariyar, B. Biofumigation-a Sustainable Alternative to Chemical Control of Soil Borne Pathogens: A Review. International Journal of Advanced Multidisciplinary Research and Studies 2024, 4(1), 134–138. [Google Scholar]
- Gamliel, A.; van Bruggen, A. H. C. Maintaining Soil Health for Crop Production in Organic Greenhouses. Sci Hortic 2016, 208, 120–130. [Google Scholar] [CrossRef]
- Céspedes, C. L.; Avila, J. G.; Martínez, A.; Serrato, B.; Calderón-Mugica, J. C.; Salgado-Garciglia, R. Antifungal and Antibacterial Activities of Mexican Tarragon (Tagetes Lucida). J Agric Food Chem 2006, 54(10), 3521–3527. [Google Scholar] [CrossRef]
- Barros, A. F.; Campos, V. P.; da Silva, J. C. P.; Pedroso, M. P.; Medeiros, F. H. V.; Pozza, E. A.; Reale, A. L. Nematicidal Activity of Volatile Organic Compounds Emitted by Brassica Juncea, Azadirachta Indica, Canavalia Ensiformis, Mucuna Pruriens and Cajanus Cajan against Meloidogyne Incognita. Applied Soil Ecology 2014, 80, 34–43. [Google Scholar] [CrossRef]
- Arnault, I.; Fleurance, C.; Vey, F.; Fretay, G. Du; Auger, J. Use of Alliaceae Residues to Control Soil-Borne Pathogens. Ind Crops Prod 2013, 49, 265–272. [Google Scholar] [CrossRef]
- Elhakeem, A.; van der Werf, W.; Bastiaans, L. Radiation Interception and Radiation Use Efficiency in Mixtures of Winter Cover Crops. Field Crops Res 2021, 264, 1–9. [Google Scholar] [CrossRef]
- Kalaji, H. M.; Dąbrowski, P.; Cetner, M. D.; Samborska, I. A.; Łukasik, I.; Brestic, M.; Zivcak, M.; Tomasz, H.; Mojski, J.; Kociel, H.; Panchal, B. M. A Comparison between Different Chlorophyll Content Meters under Nutrient Deficiency Conditions. J Plant Nutr 2017, 40(7), 1024–1034. [Google Scholar] [CrossRef]
- Morris, E. K.; Fletcher, R.; Veresoglou, S. D. Effective Methods of Biofumigation: A Meta-Analysis. Plant Soil 2020, 446(1–2), 379–392. [Google Scholar] [CrossRef]
- Matthiessen, J.; Kirkegaard, J. Biofumigation and Enhanced Biodegradation: Opportunity and Challenge in Soilborne Pest and Disease Management. CRC Crit Rev Plant Sci 2006, 25(3), 235–265. [Google Scholar] [CrossRef]
- Hanschen, F. S.; Winkelmann, T. Biofumigation for Fighting Replant Disease-a Review. Agronomy 2020, 10(3), 1–16. [Google Scholar] [CrossRef]
- Kirkegaard, J. Biofumigation for Plant Disease Control – from the Fundamentals to the Farming System. In Disease Control in Crops: Biological and Environmentally Friendly Approaches; Walters, D., Ed.; Wiley-Blackwell,: Oxford, 2009; pp. 172–195. [Google Scholar]
- Fahey, J. W.; Zalcmann, A. T.; Talalay, P. The Chemical Diversity and Distribution of Glucosinolates and Isothiocyanates among Plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Clarkson, J.; Michel, V.; Neilson, R. Biofumigation for the Control of Soil-Borne Diseases. Soil borne disease focus group 2015. https://ec.europa.eu/eip/agriculture/sites/default/files/9_eip_sbd_mp_biofumigation_final_0.pdf (accessed 2024-11-01).
- Agerbirk, N.; Olsen, C. E. Glucosinolate Structures in Evolution. Phytochemistry 2012, 77, 16–45. [Google Scholar] [CrossRef]
- Gimsing, A. L.; Kirkegaard, J. A. Glucosinolates and Biofumigation: Fate of Glucosinolates and Their Hydrolysis Products in Soil. Phytochemistry Reviews 2009, 8(1), 299–310. [Google Scholar] [CrossRef]
- Rosa, E. A. S.; Heaney, R. K.; Fenwick, G. R.; Portas, C. A. M. Glucosinolate in Crop Plants. In Horticultural Reviews; Janick, J., Ed.; Wiley, 1997; Vol. 19, pp 99–215.
- Björkman, M.; Klingen, I.; Birch, A. N. E.; Bones, A. M.; Bruce, T. J. A.; Johansen, T. J.; Meadow, R.; Mølmann, J.; Seljåsen, R.; Smart, L. E.; Stewart, D. Phytochemicals of Brassicaceae in Plant Protection and Human Health - Influences of Climate, Environment and Agronomic Practice. Phytochemistry 2011, 72(7), 538–556. [Google Scholar] [CrossRef]
- Bellostas, N.; Sørensen, J. C.; Sørensen, H. Profiling Glucosinolates in Vegetative and Reproductive Tissues of Four Brassica Species of the U-Triangle for Their Biofumigation Potential. J Sci Food Agric 2007, 87(8), 1586–1594. [Google Scholar] [CrossRef]
- Fourie, H.; Ahuja, P.; Lammers, J.; Daneel, M. Brassicacea-Based Management Strategies as an Alternative to Combat Nematode Pests: A Synopsis. Crop Protection 2016, 80, 21–41. [Google Scholar] [CrossRef]
- Sarwar, M.; Kirkegaard, J. A. Biofumigation Potential of Brassicas II. Effect of Environment and Ontogeny on Glucosinolate Production and Implications for Screening. Plant Soil 1998, 201, 91–101. [Google Scholar] [CrossRef]
- Brown, P. D.; Morra, M. J.; McCaffrey, J. P.; Auld, D. L.; Williams III, L. Allelochemicals Produced during Glucosinolate Degradation in Soil. J Chem Ecol 1991, 17, 2021–2034. [Google Scholar] [CrossRef] [PubMed]
- Neubauer, C.; Heitmann, B.; Müller, C. Biofumigation Potential of Brassicaceae Cultivars to Verticillium Dahliae. Eur J Plant Pathol 2014, 140(2), 341–352. [Google Scholar] [CrossRef]
- Borek, V.; Elberson, L. R.; McCaffrey, J. P.; Morra, M. J. Toxicity of Isothiocyanates Produced by Glucosinolates in Brassicaceae Species to Black Vine Weevil Eggs. J Agric Food Chem 1998, 46, 5318–5323. [Google Scholar] [CrossRef]
- Morra, M. J.; Kirkegaard, J. A. Isothiocyanate Release from Soil-Incorporated Brassica Tissues. Soil Biol Biochem 2002, 34, 1683–1690. [Google Scholar] [CrossRef]
- Bellostas, N.; Sørensen, J. C.; Sørensen, H. Qualitative and Quantitative Evaluation of Glucosinolates in Cruciferous Plants during Their Life Cycles. Agroindustria 2004, 3(3), 5–10. [Google Scholar]
- Kirkegaard, J. A.; Sarwar, M. Biofumigation Potential of Brassicas I. Variation in Glucosinolate Profiles of Diverse Field-Grown Brassicas. Plant Soil 1998, 201, 71–89. [Google Scholar] [CrossRef]
- Lake, L.; Sadras, V. O. Associations between Yield, Intercepted Radiation and Radiation Use Efficiency in Chickpea. In 18th Australian Society of Agronomy Conference; RD Armstrong and L Hafner, Ed.; Australian Society of Agronomy: Ballarat, 2017; pp. 24–28. [Google Scholar]
- Wang, S.; Wang, E.; Wang, F.; Tang, L. Phenological Development and Grain Yield of Canola as Affected by Sowing Date and Climate Variation in the Yangtze River Basin of China. Crop Pasture Sci 2012, 63(5), 478–488. [Google Scholar] [CrossRef]
- Manevski, K.; Lærke, P. E.; Jiao, X.; Santhome, S.; Jørgensen, U. Biomass Productivity and Radiation Utilisation of Innovative Cropping Systems for Biorefinery. Agric For Meteorol 2017, 233, 250–264. [Google Scholar] [CrossRef]
- Morrison, M. J.; Stewart, D. W. Radiation-Use Efficiency in Summer Rape. Agron J 1995, 87(6), 1139–1142. [Google Scholar] [CrossRef]
- Larkin, R. P.; Griffin, T. S. Control of Soilborne Potato Diseases Using Brassica Green Manures. Crop Protection 2007, 26(7), 1067–1077. [Google Scholar] [CrossRef]
- Winde, I.; Wittstock, U. Insect Herbivore Counteradaptations to the Plant Glucosinolate-Myrosinase System. Phytochemistry 2011, 72(13), 1566–1575. [Google Scholar] [CrossRef] [PubMed]
- Ashiq, S.; Edwards, S.; Watson, A.; Blundell, E.; Back, M. Antifungal Effect of Brassica Tissues on the Mycotoxigenic Cereal Pathogen Fusarium Graminearum. Antibiotics 2022, 11(9), 1–13. [Google Scholar] [CrossRef]
- Ríos, P.; Obregón, S.; González, M.; de Haro, A.; Sánchez, M. E. Screening Brassicaceous Plants as Biofumigants for Management of Phytophthora Cinnamomi Oak Disease. For Pathol 2016, 46(6), 652–659. [Google Scholar] [CrossRef]
- Lazzeri, L.; Malaguti, L.; Cinti, S.; Ugolini, L.; De Nicola, G. R.; Bagatta, M.; Casadei, N.; D’Avino, L.; Matteo, R.; Patalano, G. The Brassicaceae Biofumigation System for Plant Cultivation and Defence. An Italian Twenty-Year Experience of Study and Application. Acta Hortic 2013, 1005, 375–382. [Google Scholar] [CrossRef]
- Wang, S.; Wang, E.; Wang, F.; Tang, L. Phenological Development and Grain Yield of Canola as Affected by Sowing Date and Climate Variation in the Yangtze River Basin of China. Crop Pasture Sci 2012, 63(5), 478–488. [Google Scholar] [CrossRef]

| Type | Chemical name | Main Hydrolysis Product |
|---|---|---|
| Trivial name | ||
| Aliphatic | ||
| Epiprogoitrin | 2(S)-Hydroxy-3-butenylglucosinolate | Oxazolidine-2-thiones |
| Glucoalyssin | 5-Methylsulphinylpentyl | Isothiocyanate |
| Glucobrassicanapin | 4-Pentenyl | Isothiocyanate |
| Glucoerucin | 4-Methylthiobutyl | Isothiocyanate (Erucin) |
| Glucoiberverin | 3-Methylthiopropyl | Isothiocyanate |
| Gluconapin | 3-Butenyl | Isothiocyanate |
| Glucoraphanin | 4-Methylsulphinylbutyl | Isothiocyanate (Sulforaphane) |
| Glucoraphasatin | 4-Methylsulfanyl-3-butenyl | Isothiocyanate |
| Napoleiferin | 5-Allyl-1,3-oxazolidin-2-thione | Isothiocyanate |
| Progoitrin | 2-Hydroxy-3-butenyl | Oxazolidine-2-thiones |
| Sinigrin | 2-Propenyl | Isothiocyanate (Allyl) |
| Aromatic | ||
| Gluconasturtiin | 2-Phenylethyl | Isothiocyanate (Phenethyl) |
| Glucotropaeolin | Benzyl | Isothiocyanate (Benzyl) |
| Sinalbin | 4-hydroxybenzyl glucosinolate; | Nitrile |
| Indolyl | ||
| 4-Hydroxyglucobrassicin | 4-Hydroxy-3-indolylmethyl | Thiocyanate |
| Glucobrassicin | 3-Indolylmethyl | Indolyl-3-carbinol |
| Parameter | Measurement dates 2022/23 (das *) | Measurement dates 2023/24 (das) | Equipment | Method/procedure |
|---|---|---|---|---|
| Fraction of intercepted PAR (fIPAR) | 22, 41, 67, 110, 123, 137, 161, 186 | LI-191-R Quantum line sensor | fIPAR = [Incident PAR - Transmitted PAR] / Incident PAR | |
| Transmitted PAR: average value of 3 measurements per elementary plot | ||||
| SPAD | 49, 70, 114, 138, 160, 174, 186. | 122 | SPAD-502® chlorophyll meter (Konica Minolta Inc., Japan) | Average value of 15 measurements (one leaf from 15 plants) per elementary plot and measurement date |
| Biomass produced: total, root, and aerial part | 123 ,186 | 73 ,122 | Precision balances and drying oven | Sampled area per elementary plot: 0.5 m² (1 m x 0.5 m) |
| Glucosinolate concentration and types in the aerial part | 123, 186 | 120, 190 | Lyophilizer (Lyoquest 55 plus-Telstar), IKA A11 basic analytical mill (IKA-Werke GmbH & Co. KG) | Leaf or root samples were frozen shortly after collection at -80 °C. The samples were lyophilized and maintained in a drying oven at 80 ºC for 24 h for myrosinase inactivation, then ground. |
| Growing cycle | 1st cycle (2022-23) | 2nd cycle (2023/24) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling date | 23-01-23 (123 das) | 27-03-23 (186 das) | 11-12-23 (73 das) | 29-01-24 (122 das) | ||||||||
| Specie | ADB1 | RDB | TDB | ADB | RDB | TDB | ADB | RDB | TDB | ADB | RDB1 | TDB |
| B. carinata | 763,9 | 73,1 | 837 | 576,4 ab | 39,9 c | 616,3 ab | 315,0 b | 73,1 c | 388,1 b | 515,6 | 85,3 b | 600,9 |
| B. juncea | 642,5 | 124,8 | 767,3 | 735,3 a | 92,1 ab | 827,5 a | 130,1 c | 31,3 d | 161,5 c | 383,8 | 148,4 b | 532,2 |
| R. sativus | 641,6 | 122 | 763,6 | 598,9 ab | 131,4 a | 730,3 ab | 380,4 b | 202,0 a | 582,4 a | 354,1 | 238,5 a | 592,6 |
| S. alba | 647,2 | 67,4 | 714,7 | 493,5 b | 56,5 bc | 550,1 b | 507,3 a | 143,4 b | 650,7 a | 605 | 83,7 b | 688,7 |
| p value | ns2 | ns | ns | * | ** | * | *** | *** | *** | n.s. | ** | n.s. |
| Sources of variation |
ADB1 | RDB | TDB |
|---|---|---|---|
| B. carinata | 639,8 | 79,2 b | 719,0 |
| B. juncea | 513,1 | 136,6 ab | 649,7 |
| R. sativus | 497,8 | 180,2 a | 678,1 |
| S. alba | 626,1 | 75,6 b | 701,7 |
| Species | ns2 | * | ns |
| Cycle | * | n.s. | * |
| First | 569,2 a3 | 117,9 | 687,1 a |
| Second | 464,6 b | 139,0 | 603,6 b |
| Species x cycle | ns | ns | ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




