Submitted:
02 November 2024
Posted:
05 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
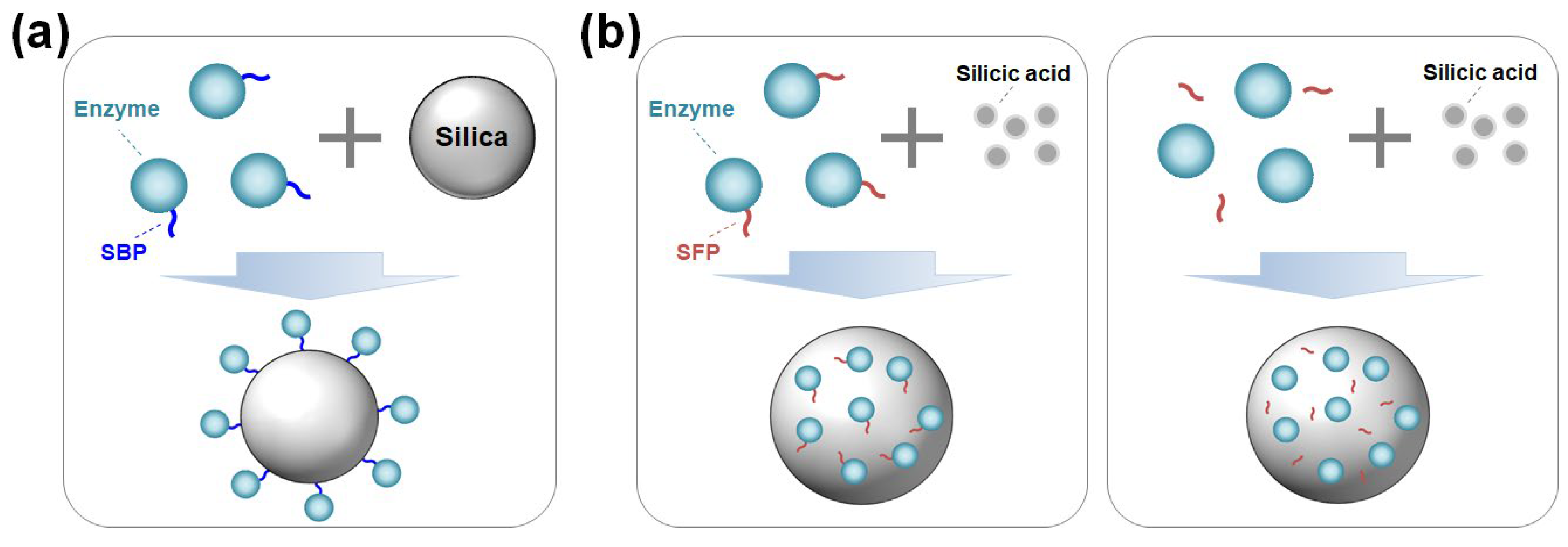
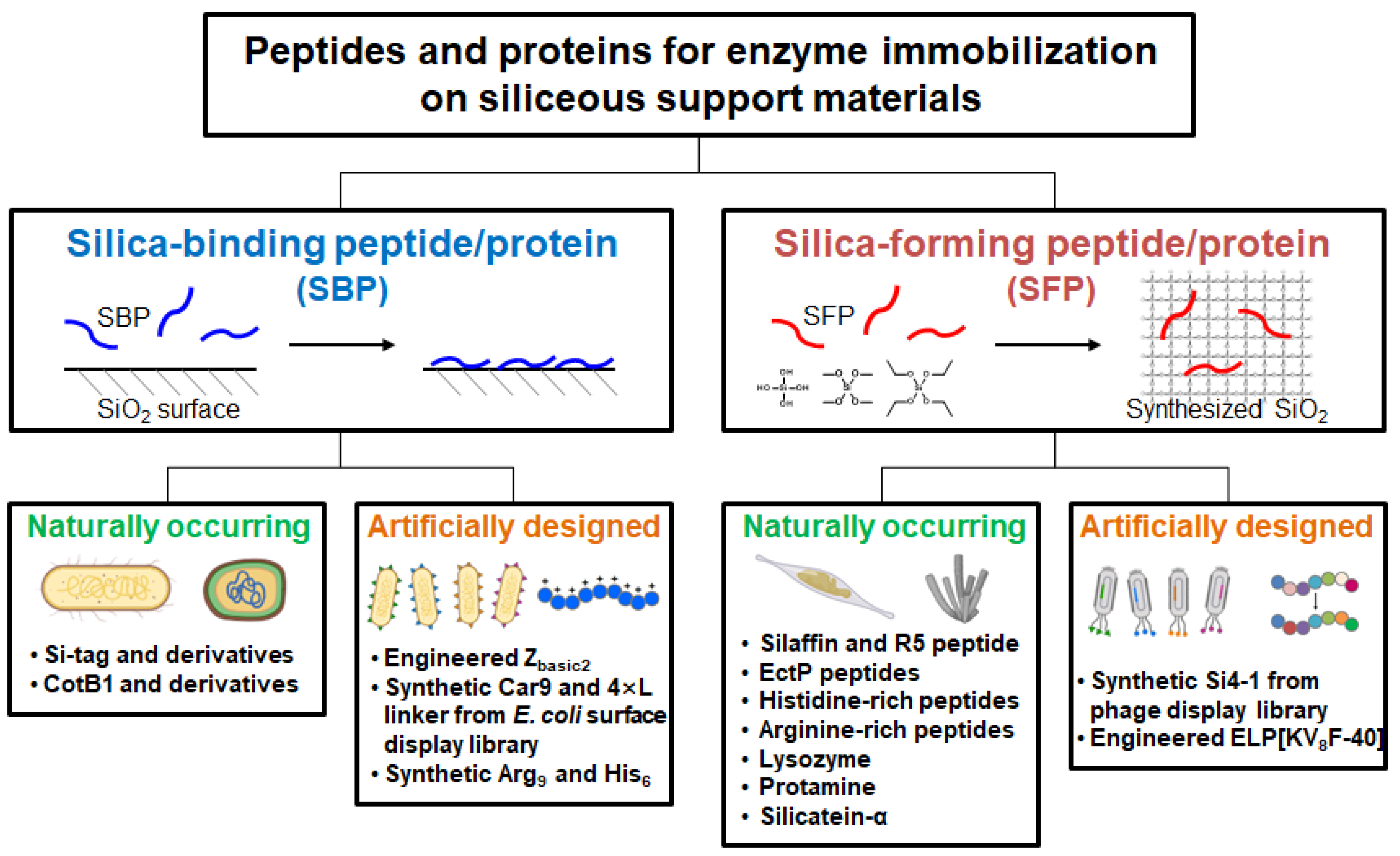
2. Silica-binding proteins/peptides (SBPs)
2.1. How SBPs work
2.2. Naturally occurring SBPs
2.2.1. Si-tag and its derivatives
2.2.2. CotB1 derivatives
2.3. Artificially engineered SBPs
2.3.1. Zbasic2
2.3.2. Car9 peptide
2.3.3. Poly(amino acid)
2.3.4. Linker peptide
3. Silica-forming proteins/peptides (SFPs)
3.1. How SFPs work
3.2. Naturally occurring SFPs
3.2.1. Silaffin-derived R5 peptide
3.2.2. EctP peptides
3.2.3. Histidine-rich Kpt peptide and glassin
3.2.4. Lysozyme
3.2.5. Protamine
3.2.6. Arginine-rich peptides
3.2.7. Silicatein
3.3. Artificially engineered SFPs
3.3.1. Synthetic peptides from phage display biopanning
3.3.2. Elastin-like polypeptide
4. Conclusions and outlook
Author Contributions
Funding
References
- Hartmann, M.; Kostrov, X. Immobilization of enzymes on porous silicas – benefits and challenges. Chem. Soc. Rev. 2013, 42, 6277–6289. [Google Scholar] [CrossRef] [PubMed]
- Jo, B.H.; Kim, C.S.; Jo, Y.K.; Cheong, H.; Cha, H.J. Recent developments and applications of bioinspired silicification. Korean J. Chem. Eng. 2016, 33, 1125–1133. [Google Scholar] [CrossRef]
- Costa, J.R.; Neto, T.; Pedrosa, S.S.; Sousa, S.C.; Azevedo-Silva, J.; Tavares-Valente, D.; Mendes, A.; Pintado, M.E.; Fernandes, J.C.; Oliveira, A.L.S.; et al. Biogenic silica microparticles as a new and sustainable cosmetic ingredient: Assessment of performance and quality parameters. Colloid Surf. B-Biointerfaces 2023, 226, 113305. [Google Scholar] [CrossRef]
- Abdelhamid, M.A.A.; Pack, S.P. Biomimetic and bioinspired silicifications: Recent advances for biomaterial design and applications. Acta Biomater. 2021, 120, 38–56. [Google Scholar] [CrossRef]
- Liu, C.; Steer, D.L.; Song, H.; He, L. Superior binding of proteins on a silica surface: Physical insight into the synergetic contribution of polyhistidine and a silica-binding peptide. J. Phys. Chem. Lett. 2022, 13, 1609–1616. [Google Scholar] [CrossRef]
- Nguyen, T.K.M.; Ki, M.R.; Son, R.G.; Kim, K.H.; Hong, J.; Pack, S.P. Synthesis of sub-50 nm bio-inspired silica particles using a C-terminal-modified ferritin template with a silica-forming peptide. J. Ind. Eng. Chem. 2021, 101, 262–269. [Google Scholar] [CrossRef]
- Ozaki, M.; Sakashita, S.; Hamada, Y.; Usui, K. Peptides for silica precipitation: Amino acid sequences for directing mineralization. Protein Pept. Lett. 2018, 25, 15–24. [Google Scholar] [CrossRef]
- Naik, R.R.; Brott, L.L.; Clarson, S.J.; Stone, M.O. Silica-precipitating peptides isolated from a combinatorial phage display peptide library. J. Nanosci. Nanotechnol. 2002, 2, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.X.; Chen, J.L.; Li, E.; Hu, C.G.; Luo, S.Z.; He, C.Z. Ultrahigh adhesion force between silica-binding peptide SB7 and glass substrate studied by single-molecule force spectroscopy and molecular dynamic simulation. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Puddu, V.; Perry, C.C. Peptide adsorption on silica nanoparticles: Evidence of hydrophobic interactions. ACS nano 2012, 6, 6356–6363. [Google Scholar] [CrossRef]
- Patwardhan, S.V.; Emami, F.S.; Berry, R.J.; Jones, S.E.; Naik, R.R.; Deschaume, O.; Heinz, H.; Perry, C.C. Chemistry of aqueous silica nanoparticle surfaces and the mechanism of selective peptide adsorption. J. Am. Chem. Soc. 2012, 134, 6244–6256. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, K.; Nomura, K.; Hata, Y.; Nishimura, T.; Asami, Y.; Kuroda, A. The Si-tag for immobilizing proteins on a silica surface. Biotechnol. Bioeng. 2007, 96, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Joo, K.I.; Jo, B.H.; Cha, H.J. Stability-controllable self-immobilization of carbonic anhydrase fused with a silica-binding tag onto diatom biosilica for enzymatic CO2 capture and utilization. ACS Appl. Mater. Interfaces 2020, 12, 27055–27063. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Kuroda, A. Why does the silica-binding protein "Si-tag" bind strongly to silica surfaces? Implications of conformational adaptation of the intrinsically disordered polypeptide to solid surfaces. Colloid Surf. B-Biointerfaces 2011, 86, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Zurier, H.S.; Goddard, J.M. Directed immobilization of PETase on mesoporous silica enables sustained depolymerase activity in synthetic wastewater conditions. ACS Appl. Bio Mater. 2022, 5, 4981–4992. [Google Scholar] [CrossRef]
- Gu, B.; Park, T.J.; Ahn, J.H.; Huang, X.J.; Lee, S.Y.; Choi, Y.K. Nanogap field-effect transistor biosensors for electrical detection of avian influenza. Small 2009, 5, 2407–2412. [Google Scholar] [CrossRef]
- Motomura, K.; Ikeda, T.; Matsuyama, S.; Abdelhamid, M.A.; Tanaka, T.; Ishida, T.; Hirota, R.; Kuroda, A. The C-terminal zwitterionic sequence of CotB1 is essential for biosilicification of the Bacillus cereus spore coat. J. Bacteriol. 2016, 198, 276–282. [Google Scholar] [CrossRef]
- Abdelhamid, M.A.A.; Motomura, K.; Ikeda, T.; Ishida, T.; Hirota, R.; Kuroda, A. Affinity purification of recombinant proteins using a novel silica-binding peptide as a fusion tag. Appl. Microbiol. Biotechnol. 2014, 98, 5677–5684. [Google Scholar] [CrossRef]
- Abdelhamid, M.A.; Ikeda, T.; Motomura, K.; Tanaka, T.; Ishida, T.; Hirota, R.; Kuroda, A. Application of volcanic ash particles for protein affinity purification with a minimized silica-binding tag. J. Biosci. Bioeng. 2016, 122, 633–638. [Google Scholar] [CrossRef]
- Muller, M.M.; Nedielkov, R.; Arndt, K.M. Strategies for enzymatic inactivation of the veterinary antibiotic florfenicol. Antibiotics 2022, 11, 443. [Google Scholar] [CrossRef]
- Nilsson, B.; Moks, T.; Jansson, B.; Abrahmsen, L.; Elmblad, A.; Holmgren, E.; Henrichson, C.; Jones, T.A.; Uhlen, M. A synthetic IgG-binding domain based on staphylococcal protein A. Protein eng. 1987, 1, 107–113. [Google Scholar] [CrossRef]
- Hedhammar, M.; Hober, S. Zbasic—A novel purification tag for efficient protein recovery. J. Chromatogr. A 2007, 1161, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Bolivar, J.M.; Nidetzky, B. Positively charged mini-protein Zbasic2 as a highly efficient silica binding module: Opportunities for enzyme immobilization on unmodified silica supports. Langmuir 2012, 28, 10040–10049. [Google Scholar] [CrossRef] [PubMed]
- Bolivar, J.M.; Gascon, V.; Marquez-Alvarez, C.; Blanco, R.M.; Nidetzky, B. Oriented coimmobilization of oxidase and catalase on tailor-made ordered mesoporous silica. Langmuir 2017, 33, 5065–5076. [Google Scholar] [CrossRef]
- Coyle, B.L.; Baneyx, F. A cleavable silica-binding affinity tag for rapid and inexpensive protein purification. Biotechnol. Bioeng. 2014, 111, 2019–2026. [Google Scholar] [CrossRef]
- Soto-Rodriguez, J.; Coyle, B.L.; Samuelson, A.; Aravagiri, K.; Baneyx, F. Affinity purification of Car9-tagged proteins on silica matrices: Optimization of a rapid and inexpensive protein purification technology. Protein Expr. Purif. 2017, 135, 70–77. [Google Scholar] [CrossRef]
- Xu, M.; Bailey, M.J.; Look, J.; Baneyx, F. Affinity purification of Car9-tagged proteins on silica-derivatized spin columns and 96-well plates. Protein Expr. Purif. 2020, 170, 105608. [Google Scholar] [CrossRef]
- Wender, P.A.; Mitchell, D.J.; Pattabiraman, K.; Pelkey, E.T.; Steinman, L.; Rothbard, J.B. The design, synthesis, and evaluation of molecules that enable or enhance cellular uptake: Peptoid molecular transporters. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 13003–13008. [Google Scholar] [CrossRef] [PubMed]
- S. M., F.; R.T., R. Polyarginine as a multifunctional fusion tag. Protein sci. 2005, 14, 1538–1544. [Google Scholar] [CrossRef]
- Young, C.L.; Britton, Z.T.; Robinson, A.S. Recombinant protein expression and purification: A comprehensive review of affinity tags and microbial applications. Biotechnol. J. 2012, 7, 620–634. [Google Scholar] [CrossRef]
- Eteshola, E.; Brillson, L.J.; Lee, S.C. Selection and characteristics of peptides that bind thermally grown silicon dioxide films. Biomol. Eng. 2005, 22, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Freitas, A.I.; Domingues, L.; Aguiar, T.Q. Bare silica as an alternative matrix for affinity purification/immobilization of His-tagged proteins. Sep. Purif. Technol. 2022, 286, 120448. [Google Scholar] [CrossRef]
- Sunna, A.; Chi, F.; Bergquist, P.L. A linker peptide with high affinity towards silica-containing materials. New biotech. 2013, 30, 485–492. [Google Scholar] [CrossRef]
- Nygaard, S.; Wendelbo, R.; Brown, S. Surface-specific zeolite-binding proteins. Adv. Mater. 2002, 14, 1853–1856. [Google Scholar] [CrossRef]
- Bansal, R.; Elgundi, Z.; Care, A.; S, C.G.; M, S.L.; Rodger, A.; Sunna, A. Elucidating the binding mechanism of a novel silica-binding peptide. Biomolecules 2019, 10, 4. [Google Scholar] [CrossRef]
- Lu, S.; Zou, K.; Guo, B.; Pei, J.; Wang, Z.; Xiao, W.; Zhao, L. One-step purification and immobilization of thermostable β-glucosidase on Na-Y zeolite based on the linker and its application in the efficient production of baohuoside I from icariin. Bioorganic chem. 2022, 121, 105690. [Google Scholar] [CrossRef]
- Care, A.; Petroll, K.; Gibson, E.S.Y.; Bergquist, P.L.; Sunna, A. Solid-binding peptides for immobilisation of thermostable enzymes to hydrolyse biomass polysaccharides. Biotechnol. Biofuels 2017, 10, 29. [Google Scholar] [CrossRef]
- Lechner, C.C.; Becker, C.F.W. A sequence-function analysis of the silica precipitating silaffin R5 peptide. J. Pept. Sci. 2014, 20, 152–158. [Google Scholar] [CrossRef]
- Lechner, C.C.; Becker, C.F.W. Silaffins in silica biomineralization and biomimetic silica precipitation. Mar. Drugs 2015, 13, 5297–5333. [Google Scholar] [CrossRef]
- Kuno, T.; Nonoyama, T.; Hirao, K.; Kato, K. Influence of the charge relay effect on the silanol condensation reaction as a model for silica biomineralization. Langmuir 2011, 27, 13154–13158. [Google Scholar] [CrossRef]
- Shimizu, K.; Cha, J.; Stucky, G.D.; Morse, D.E. Silicatein α: Cathepsin L-like protein in sponge biosilica. Proc. Natl. Acad. Sci. U. S. A. 1998, 95, 6234–6238. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.N.; Stucky, G.D.; Morse, D.E.; Deming, T.J. Biomimetic synthesis of ordered silica structures mediated by block copolypeptides. Nature 2000, 403, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Schröder, H.C.; Wiens, M.; Schlossmacher, U.; Brandt, D.; Müller, W.E.G. Silicatein-mediated polycondensation of orthosilicic acid: Modeling of a catalytic mechanism involving ring formation. Silicon 2012, 4, 33–38. [Google Scholar] [CrossRef]
- Cha, J.N.; Shimizu, K.; Zhou, Y.; Christiansen, S.C.; Chmelka, B.F.; Stucky, G.D.; Morse, D.E. Silicatein filaments and subunits from a marine sponge direct the polymerization of silica and silicones in vitro. Proc. Natl. Acad. Sci. USA 1999, 96, 361–365. [Google Scholar] [CrossRef]
- Coradin, T.; Durupthy, O.; Livage, J. Interactions of amino-containing peptides with sodium silicate and colloidal silica: A biomimetic approach of silicification. Langmuir 2002, 18, 2331–2336. [Google Scholar] [CrossRef]
- Kroger, N.; Deutzmann, R.; Sumper, M. Polycationic peptides from diatom biosilica that direct silica nanosphere formation. Science 1999, 286, 1129–1132. [Google Scholar] [CrossRef]
- Patwardhan, S.V.; Clarson, S.J.; Perry, C.C. On the role(s) of additives in bioinspired silicification. Chem. Commun. 2005, 1113–1121. [Google Scholar] [CrossRef]
- Sumper, M.; Kröger, N. Silica formation in diatoms: The function of long-chain polyamines and silaffins. J. Mater. Chem. 2004, 14, 2059–2065. [Google Scholar] [CrossRef]
- Kroger, N.; Lorenz, S.; Brunner, E.; Sumper, M. Self-assembly of highly phosphorylated silaffins and their function in biosilica morphogenesis. Science 2002, 298, 584–586. [Google Scholar] [CrossRef]
- Kozak, F.; Brandis, D.; Potzl, C.; Epasto, L.M.; Reichinger, D.; Obrist, D.; Peterlik, H.; Polyansky, A.; Zagrovic, B.; Daus, F.; et al. An atomistic view on the mechanism of diatom peptide-guided biomimetic silica formation. Adv. Sci. 2024, 11, e2401239. [Google Scholar] [CrossRef]
- Senior, L.; Crump, M.P.; Williams, C.; Booth, P.J.; Mann, S.; Perriman, A.W.; Curnow, P. Structure and function of the silicifying peptide R5. J. Mat. Chem. B 2015, 3, 2607–2614. [Google Scholar] [CrossRef]
- Luckarift, H.R.; Spain, J.C.; Naik, R.R.; Stone, M.O. Enzyme immobilization in a biomimetic silica support. Nat. Biotechnol. 2004, 22, 211–213. [Google Scholar] [CrossRef] [PubMed]
- Betancor, L.; Luckarift, H.R. Bioinspired enzyme encapsulation for biocatalysis. Trends Biotechnol. 2008, 26, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Jo, B.H.; Seo, J.H.; Yang, Y.J.; Baek, K.; Choi, Y.S.; Pack, S.P.; Oh, S.H.; Cha, H.J. Bioinspired silica nanocomposite with autoencapsulated carbonic anhydrase as a robust biocatalyst for CO2 sequestration. ACS Catal. 2014, 4, 4332–4340. [Google Scholar] [CrossRef]
- Lim, G.T.; Jo, B.H. Cation-assisted stabilization of carbonic anhydrase one-step in situ loaded in diatom-inspired silica nanospheres for potential applications in CO2 capture and utilization. Sep. Purif. Technol. 2024, 333, 125926. [Google Scholar] [CrossRef]
- Lee, A.S.; Kim, K.R.; Kim, J.H.; Jo, B.H.; Song, Y.H.; Seo, J.H.; Heo, H.R.; Kim, C.S. Bioinspired synthesis of micelle-templated ultrathin silica-layered mesoporous nanoparticles with enhanced mass transfer and stability for biocatalysis. Chem. Eng. J. 2023, 473, 145108. [Google Scholar] [CrossRef]
- Tao, H.C.; Lee, S.C.; Moeller, A.; Roy, R.S.; Siu, F.Y.; Zimmermann, J.; Stevens, R.C.; Potter, C.S.; Carragher, B.; Zhang, Q.H. Engineered nanostructured β-sheet peptides protect membrane proteins. Nat. Methods 2013, 10, 759–761. [Google Scholar] [CrossRef]
- Bialas, F.; Becker, C.F.W. Biomimetic silica encapsulation of lipid nanodiscs and β-sheet-stabilized diacylglycerol kinase. Bioconjugate Chem. 2021, 32, 1742–1752. [Google Scholar] [CrossRef]
- Yeo, K.B.; Ki, M.R.; Park, K.S.; Pack, S.P. Novel silica-forming peptides derived from Ectocarpus siliculosus. Process Biochem. 2017, 58, 193–198. [Google Scholar] [CrossRef]
- Thi, K.M.N.; Ki, M.R.; Son, R.G.; Kim, K.H.; Hong, J.; Pack, S.P. A dual-functional peptide, Kpt from Ruegeria pomeroyi DSS-3 for protein purification and silica precipitation. Biochem. Eng. J. 2020, 163, 107726. [Google Scholar]
- Shimizu, K.; Amano, T.; Bari, M.R.; Weaver, J.C.; Arima, J.; Mori, N. Glassin, a histidine-rich protein from the siliceous skeletal system of the marine sponge Euplectella, directs silica polycondensation. Proc. Natl. Acad. Sci. USA 2015, 112, 11449–11454. [Google Scholar] [CrossRef] [PubMed]
- Luckarift, H.R.; Dickerson, M.B.; Sandhage, K.H.; Spain, J.C. Rapid, room-temperature synthesis of antibacterial bionanocomposites of lysozyme with amorphous silica or titania. Small 2006, 2, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Bruno, F.; Gigli, L.; Ferraro, G.; Cavallo, A.; Michaelis, V.K.; Goobes, G.; Fratini, E.; Ravera, E. Lysozyme is sterically trapped within the silica cage in bioinspired silica–lysozyme composites: A multi-technique understanding of elusive protein–material interactions. Langmuir 2022, 38, 8030–8037. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.B.; Luckarift, H.R.; Urban, V.S.; O'Neill, H.; Johnson, G.R. Protein localization in silica nanospheres derived via biomimetic mineralization. Adv. Funct. Mater. 2010, 20, 3031–3038. [Google Scholar] [CrossRef]
- Ruseska, I.; Fresacher, K.; Petschacher, C.; Zimmer, A. Use of protamine in nanopharmaceuticals—A review. Nanomaterials 2021, 11, 1508. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, H.; Li, J.; Li, L.; Jiang, Y.; Jiang, Y.; Jiang, Z. Protamine-templated biomimetic hybrid capsules: Efficient and stable carrier for enzyme encapsulation. Chem. Mater. 2008, 20, 1041–1048. [Google Scholar] [CrossRef]
- Haase, N.R.; Shian, S.; Sandhage, K.H.; Kroger, N. Biocatalytic nanoscale coatings through biomimetic layer-by-layer mineralization. Adv. Funct. Mater. 2011, 21, 4243–4251. [Google Scholar] [CrossRef]
- Abdelhamid, M.A.A.; Yeo, K.B.; Ki, M.R.; Pack, S.P. Self-encapsulation and controlled release of recombinant proteins using novel silica-forming peptides as fusion linkers. Int. J. Biol. Macromol. 2019, 125, 1175–1183. [Google Scholar] [CrossRef]
- Min, K.H.; Yeo, K.B.; Ki, M.R.; Jun, S.H.; Pack, S.P. Novel silica forming peptide, RSGH, from Equus caballus: Its unique biosilica formation under acidic conditions. Biochem. Eng. J. 2020, 153, 107389. [Google Scholar] [CrossRef]
- Min, K.H.; Shin, J.W.; Ki, M.R.; Kim, S.H.; Kim, K.H.; Pack, S.P. Bio-inspired formation of silica particles using the silica-forming peptides found by silica-binding motif sequence, RRSSGGRR. Process Biochem. 2021, 111, 262–269. [Google Scholar] [CrossRef]
- Vigil, T.N.; Rowson, M.J.C.; Frost, A.J.; Berger, B.W. Understanding the relationships between solubility, stability, and activity of silicatein. Mater. Adv. 2023, 4, 662–668. [Google Scholar] [CrossRef]
- Godigamuwa, K.; Nakashima, K.; Tsujitani, S.; Naota, R.; Maulidin, I.; Kawasaki, S. Interfacial biosilica coating of chitosan gel using fusion silicatein to fabricate robust hybrid material for biomolecular applications. J. Mat. Chem. B 2023, 11, 1654–1658. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.K.; Zawadzka, A.M.; Deobald, L.A.; Crawford, R.L.; Paszczynski, A.J. Novel method for immobilization of enzymes to magnetic nanoparticles. J. Nanopart. Res. 2008, 10, 1009–1025. [Google Scholar] [CrossRef]
- Cohen, R.; Lata, J.P.; Lee, Y.; Hernández, J.C.C.; Nishimura, N.; Schaffer, C.B.; Mukai, C.; Nelson, J.L.; Brangman, S.A.; Agrawal, Y.; et al. Use of tethered enzymes as a platform technology for rapid analyte detection. PLoS ONE 2015, 10, e0142326. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Zhang, N.; Huang, Y.W.; Li, S.Y.; Zhang, G.M. Simple and efficient enzymatic procedure for p-coumaric acid synthesis: Complete bioconversion and biocatalyst recycling under alkaline condition. Biochem. Eng. J. 2022, 188, 108693. [Google Scholar] [CrossRef]
- Wu, X.; Fraser, K.; Zha, J.; Dordick, J.S. Flexible peptide linkers enhance the antimicrobial activity of surface-immobilized bacteriolytic enzymes. ACS Appl. Mater. Interfaces 2018, 10, 36746–36756. [Google Scholar] [CrossRef] [PubMed]
- Meyer, D.E.; Chilkoti, A. Purification of recombinant proteins by fusion with thermally-responsive polypeptides. Nat. Biotechnol. 1999, 17, 1112–1115. [Google Scholar] [CrossRef]
- Han, W.; Chilkoti, A.; Lopez, G.P. Self-assembled hybrid elastin-like polypeptide/silica nanoparticles enable triggered drug release. Nanoscale 2017, 9, 6178–6186. [Google Scholar] [CrossRef]
- Han, W.; MacEwan, S.R.; Chilkoti, A.; López, G.P. Bio-inspired synthesis of hybrid silica nanoparticles templated from elastin-like polypeptide micelles. Nanoscale 2015, 7, 12038–12044. [Google Scholar] [CrossRef]
- Lin, Y.; Jin, W.; Qiu, Y.; Zhang, G. Programmable stimuli-responsive polypeptides for biomimetic synthesis of silica nanocomposites and enzyme self-immobilization. Int. J. Biol. Macromol. 2019, 134, 1156–1169. [Google Scholar] [CrossRef]
- Yuan, H.; Liu, G.Z.; Chen, Y.X.; Yi, Z.W.; Jin, W.H.; Zhang, G.Y. A versatile tag for simple preparation of cutinase towards enhanced biodegradation of polyethylene terephthalate. Int. J. Biol. Macromol. 2023, 225, 149–161. [Google Scholar] [CrossRef]
- Liu, G.Z.; Yuan, H.; Chen, Y.X.; Mao, L.; Yang, C.; Zhang, R.F.; Zhang, G.Y. Magnetic silica-coated cutinase immobilized via ELPs biomimetic mineralization for efficient nano-PET degradation. Int. J. Biol. Macromol. 2024, 279, 135414. [Google Scholar] [CrossRef] [PubMed]
| Name | Amino acid sequence | Length | pIa | Reference |
|---|---|---|---|---|
| Naturally occurring SBP | ||||
| Si-tag | MAVVKCKPTSPGRRHVVKVVNPELHKGKPFAPLLEKNSKSGGRNNNGRITTRHIGGGHKQAYRIVDFKRNKDGIPAVVERLEYDPNRSANIALVLYKDGERRYILAPKGLKAGDQIQSGVDAAIKPGNTLPMRNIPVGSTVHNVEMKPGKGGQLARSAGTYVQIVARDGAYVTLRLRSGEMRKVEADCRATLGEVGNAEHMLRVLGKAGAARWRGVRPTVRGTAMNPVDHPHGGGEGRNFGKHPVTPWGVQTKGKKTRSNKRTDKFIVRRRSK | 273 | 10.9 | [12] |
| L2NC | MAVVKCKPTSPGRRHVVKVVNPELHKGKPFAPLLEKNSKSGGRNNNGRITTRHIGGGHKQRVLGKAGAARWRGVRPTVRGTAMNPVDHPHGGGEGRNFGKHPVTPWGVQTKGKKTRSNKRTDKFIVRRRSK | 131 | 12.1 | [12,13] |
| CotB1p | SGRARAQRQSSRGR | 14 | 12.6 | [18] |
| SB7 | RQSSRGR | 7 | 12.3 | [19] |
| Artificially engineered SBP | ||||
| Zbasic2 | VDNKFNKERRRARREIRHLPNLNREQRRAFIRSLRDDPSQSANLLAEAKKLNDAQPK | 57 | 11.4 | [23] |
| Car9 | DSARGFKKPGKR | 9 | 11.1 | [25] |
| Arg9 | RRRRRRRRR | 9 | 12.9 | [29] |
| His6 | HHHHHH | 6 | 7.2 | [5,32] |
| Linker peptide | VKTQATSREEPPRLPSKHRPGVKTQATSREEPPRLPSKHRPGVKTQATSREEPPRLPSKHRPGVKTQATSREEPPRLPSKHRPGVKTQTAS | 91 | 11.6 | [33] |
| Name | Amino acid sequence | Length | pIa | Reference |
|---|---|---|---|---|
| Naturally occurring SFP | ||||
| R5 | SSKKSGSYSGSKGSKRRIL | 19 | 11.2 | [46] |
| EctP1 | SSRSSSHRRHDHHDHRRGS | 19 | 11.8 | [59] |
| EctP2 | SSKKSGERHHRSA | 13 | 11.0 | [59] |
| Kpt | KPTHHHHHHDG | 11 | 7.2 | [60] |
| Lysozyme | KVFGRCELAAAMKRHGLDNYRGYSLGNWVCAAKFESNFNTQATNRNTDGSTDYGILQINSRWWCNDGRTPGSRNLCNIPCSALLSSDITASVNCAKKIVSDGNGMNAWVAWRNRCKGTDVQAWIRGCRL | 129 | 9.3 | [62] |
| Protamine | MPRRRRSSSRPVRRRRRPRVSRRRRRRGGRRRR | 33 | 13.3 | [66] |
| Salp1 | CGRRRGGRGGRGRGGCGRRR | 20 | 12.3 | [68] |
| RSGH | RRSGHSHEGRRRRSGHSHEGRR | 22 | 12.2 | [69] |
| Wa-RSG | RRSSGGSRKRDDKPRGDRRSSGG | 23 | 11.9 | [70] |
| Silicatein-α | AYPETVDWRTKGAVTGIKSQGDCGASYAFSAMGALEGINALATGKLTYLSEQNIIDCSVPYGNHGCKGGNMYVAFLYVVANEGVDDGGSYPFRGKQSSCTYQEQYRGASMSGSVQINSGSESDLEAAVANVGPVAVAIDGESNAFRFYYSGVYDSSRCSSSSLNHAMVITGYGISNNQEYWLAKNSWGENWGELGYVKMARNKYNQCGIASDASYPTL | 218 | 4.9 | [41] |
| Artificially engineered SFP | ||||
| Si4-1 | MSPHPHPRHHHT | 12 | 9.6 | [8] |
| ELP[KV8F-40] | VPGKGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGFGVPGKGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGFGVPGKGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGFGVPGKGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGVGVPGFG | 200 | 10.5 | [80] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




