Submitted:
01 November 2024
Posted:
01 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Small Non-Coding RNAs Can be Modified with Traditional Glycosylation
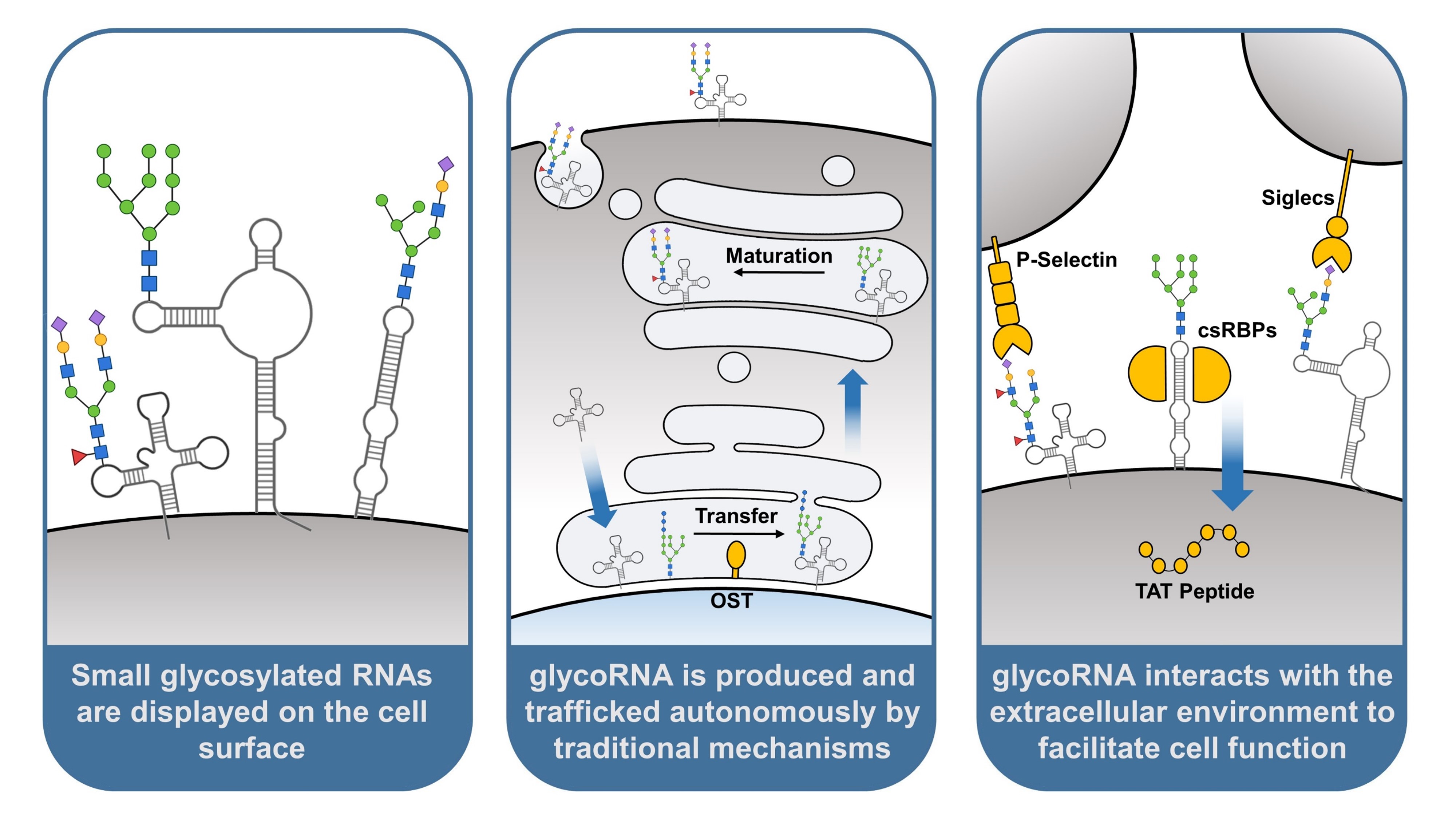
3. GlycoRNA Is Presented on the Mammalian Cell Surface
4. GlycoRNA Originates from Endogenous Glycosylation and Trafficking Mechanisms
5. GlycoRNA Selectively Interacts with Proteins to Facilitate Cell Functions
6. Qualities of GlycoRNA Expression
7. Key Challenges
8. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Crick, F. Central Dogma of Molecular Biology. Nature 1970, 227, 561–563. [Google Scholar] [CrossRef] [PubMed]
- Molecular Biology of the Cell; Alberts, B. , Ed.; 4th ed.; Garland Science: New York, 2002; ISBN 978-0-8153-3218-3. [Google Scholar]
- Flynn, R.A.; Pedram, K.; Malaker, S.A.; Batista, P.J.; Smith, B.A.H.; Johnson, A.G.; George, B.M.; Majzoub, K.; Villalta, P.W.; Carette, J.E.; et al. Small RNAs Are Modified with N-Glycans and Displayed on the Surface of Living Cells. Cell 2021, 184, 3109–3124. [Google Scholar] [CrossRef] [PubMed]
- Reily, C.; Stewart, T.J.; Renfrow, M.B.; Novak, J. Glycosylation in Health and Disease. Nat Rev Nephrol 2019, 15, 346–366. [Google Scholar] [CrossRef] [PubMed]
- Buck, C.A.; Horwitz, A.F. Integrin, a Transmembrane Glycoprotein Complex Mediating Cell-Substratum Adhesion. J Cell Sci Suppl 1987, 8, 231–250. [Google Scholar] [CrossRef]
- Crocker, P.R.; Paulson, J.C.; Varki, A. Siglecs and Their Roles in the Immune System. Nat Rev Immunol 2007, 7, 255–266. [Google Scholar] [CrossRef]
- Varki, A.; Lowe, J.B. Biological Roles of Glycans. In Essentials of Glycobiology; Varki, A., Cummings, R.D., Esko, J.D., Freeze, H.H., Stanley, P., Bertozzi, C.R., Hart, G.W., Etzler, M.E., Eds.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor (NY), 2009 ISBN 978-0-87969-770-9.
- Boccaletto, P.; Machnicka, M.A.; Purta, E.; Piątkowski, P.; Bagiński, B.; Wirecki, T.K.; de Crécy-Lagard, V.; Ross, R.; Limbach, P.A.; Kotter, A.; et al. MODOMICS: A Database of RNA Modification Pathways. 2017 Update. Nucleic Acids Research 2018, 46, D303–D307. [Google Scholar] [CrossRef]
- He, L.; Li, H.; Wu, A.; Peng, Y.; Shu, G.; Yin, G. Functions of N6-Methyladenosine and Its Role in Cancer. Mol Cancer 2019, 18, 176. [Google Scholar] [CrossRef]
- Hillmeier, M.; Wagner, M.; Ensfelder, T.; Korytiakova, E.; Thumbs, P.; Müller, M.; Carell, T. Synthesis and Structure Elucidation of the Human tRNA Nucleoside Mannosyl-Queuosine. Nat Commun 2021, 12, 7123. [Google Scholar] [CrossRef]
- Agard, N.J.; Prescher, J.A.; Bertozzi, C.R. A Strain-Promoted [3 + 2] Azide−Alkyne Cycloaddition for Covalent Modification of Biomolecules in Living Systems. J. Am. Chem. Soc. 2004, 126, 15046–15047. [Google Scholar] [CrossRef]
- Zhang, N.; Tang, W.; Torres, L.; Wang, X.; Ajaj, Y.; Zhu, L.; Luan, Y.; Zhou, H.; Wang, Y.; Zhang, D.; et al. Cell Surface RNAs Control Neutrophil Recruitment. Cell 2024, 187, 846–860. [Google Scholar] [CrossRef]
- Ming Bi; Zirui Zhang; Tao Wang; Hongwei Liang; Zhixin Tian A Draft of Human N-Glycans of glycoRNA. bioRxiv 2023, 2023.09.18.558371. [CrossRef]
- Trimble, R.B.; Tarentino, A.L. Identification of Distinct Endoglycosidase (Endo) Activities in Flavobacterium Meningosepticum: Endo F1, Endo F2, and Endo F3. Endo F1 and Endo H Hydrolyze Only High Mannose and Hybrid Glycans. Journal of Biological Chemistry 1991, 266, 1646–1651. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Chai, P.; Till, N.A.; Hemberger, H.; Lebedenko, C.G.; Porat, J.; Watkins, C.P.; Caldwell, R.M.; George, B.M.; Perr, J.; et al. The Modified RNA Base acp3U Is an Attachment Site for N-Glycans in glycoRNA. Cell 2024, S0092867424008389. [Google Scholar] [CrossRef] [PubMed]
- Takakura, M.; Ishiguro, K.; Akichika, S.; Miyauchi, K.; Suzuki, T. Biogenesis and Functions of Aminocarboxypropyluridine in tRNA. Nat Commun 2019, 10, 5542. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Fan, X.; Zaleta-Rivera, K.; Nguyen, T.C.; Zhou, J.; Luo, Y.; Gao, J.; Fang, R.H.; Yan, Z.; Chen, Z.B.; et al. Natural Display of Nuclear-Encoded RNA on the Cell Surface and Its Impact on Cell Interaction. Genome Biol 2020, 21, 225. [Google Scholar] [CrossRef]
- Lv, J.; Liu, Y.; Tang, J.; Xiao, H.; Hu, R.; Wang, G.; Niu, D.; Shao, P.-L.; Yang, J.; Jin, Z.; et al. A Novel Cell Membrane-Associated RNA Extraction Method and Its Application in the Discovery of Breast Cancer Markers. Anal Chem 2023, 95, 11706–11713. [Google Scholar] [CrossRef]
- Wu, E.; Guo, X.; Teng, X.; Zhang, R.; Li, F.; Cui, Y.; Zhang, D.; Liu, Q.; Luo, J.; Wang, J.; et al. Discovery of Plasma Membrane-Associated RNAs through APEX-Seq. Cell Biochem Biophys 2021, 79, 905–917. [Google Scholar] [CrossRef]
- Block, K.F.; Puerta-Fernandez, E.; Wallace, J.G.; Breaker, R.R. Association of OLE RNA with Bacterial Membranes via an RNA-Protein Interaction. Mol Microbiol 2011, 79, 21–34. [Google Scholar] [CrossRef]
- Weiss, L.; Mayhew, E. The Presence of Ribonucleic Acid within the Peripheral Zones of Two Types of Mammalian Cell. Journal Cellular Physiology 1966, 68, 345–359. [Google Scholar] [CrossRef]
- Laktionov, P.P.; Tamkovich, S.N.; Rykova, E.Y.; Bryzgunova, O.E.; Starikov, A.V.; Kuznetsova, N.P.; Vlassov, V.V. Cell-Surface-Bound Nucleic Acids: Free and Cell-Surface-Bound Nucleic Acids in Blood of Healthy Donors and Breast Cancer Patients. Ann N Y Acad Sci 2004, 1022, 221–227. [Google Scholar] [CrossRef]
- Morozkin, E.S.; Laktionov, P.P.; Rykova, E.Y.; Vlassov, V.V. Extracellular Nucleic Acids in Cultures of Long-Term Cultivated Eukaryotic Cells. Ann N Y Acad Sci 2004, 1022, 244–249. [Google Scholar] [CrossRef]
- Ma, Y.; Guo, W.; Mou, Q.; Shao, X.; Lyu, M.; Garcia, V.; Kong, L.; Lewis, W.; Ward, C.; Yang, Z.; et al. Spatial Imaging of glycoRNA in Single Cells with ARPLA. Nat Biotechnol 2023. [Google Scholar] [CrossRef] [PubMed]
- Sezgin, E.; Levental, I.; Mayor, S.; Eggeling, C. The Mystery of Membrane Organization: Composition, Regulation and Roles of Lipid Rafts. Nat Rev Mol Cell Biol 2017, 18, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Chai, P.; Lebedenko, C.G.; Flynn, R.A. RNA Crossing Membranes: Systems and Mechanisms Contextualizing Extracellular RNA and Cell Surface GlycoRNAs. Annu Rev Genomics Hum Genet 2023, 24, 85–107. [Google Scholar] [CrossRef] [PubMed]
- Dou, D.R.; Zhao, Y.; Belk, J.A.; Zhao, Y.; Casey, K.M.; Chen, D.C.; Li, R.; Yu, B.; Srinivasan, S.; Abe, B.T.; et al. Xist Ribonucleoproteins Promote Female Sex-Biased Autoimmunity. Cell 2024, 187, 733–749. [Google Scholar] [CrossRef]
- Tinazzi, E.; Patuzzo, G.; Lunardi, C. Autoantigens and Autoantibodies in the Pathogenesis of Sjögren’s Syndrome. In Sjogren’s Syndrome; Elsevier, 2016; pp. 141–156 ISBN 978-0-12-803604-4.
- Harada, Y.; Ohkawa, Y.; Kizuka, Y.; Taniguchi, N. Oligosaccharyltransferase: A Gatekeeper of Health and Tumor Progression. Int J Mol Sci 2019, 20, 6074. [Google Scholar] [CrossRef]
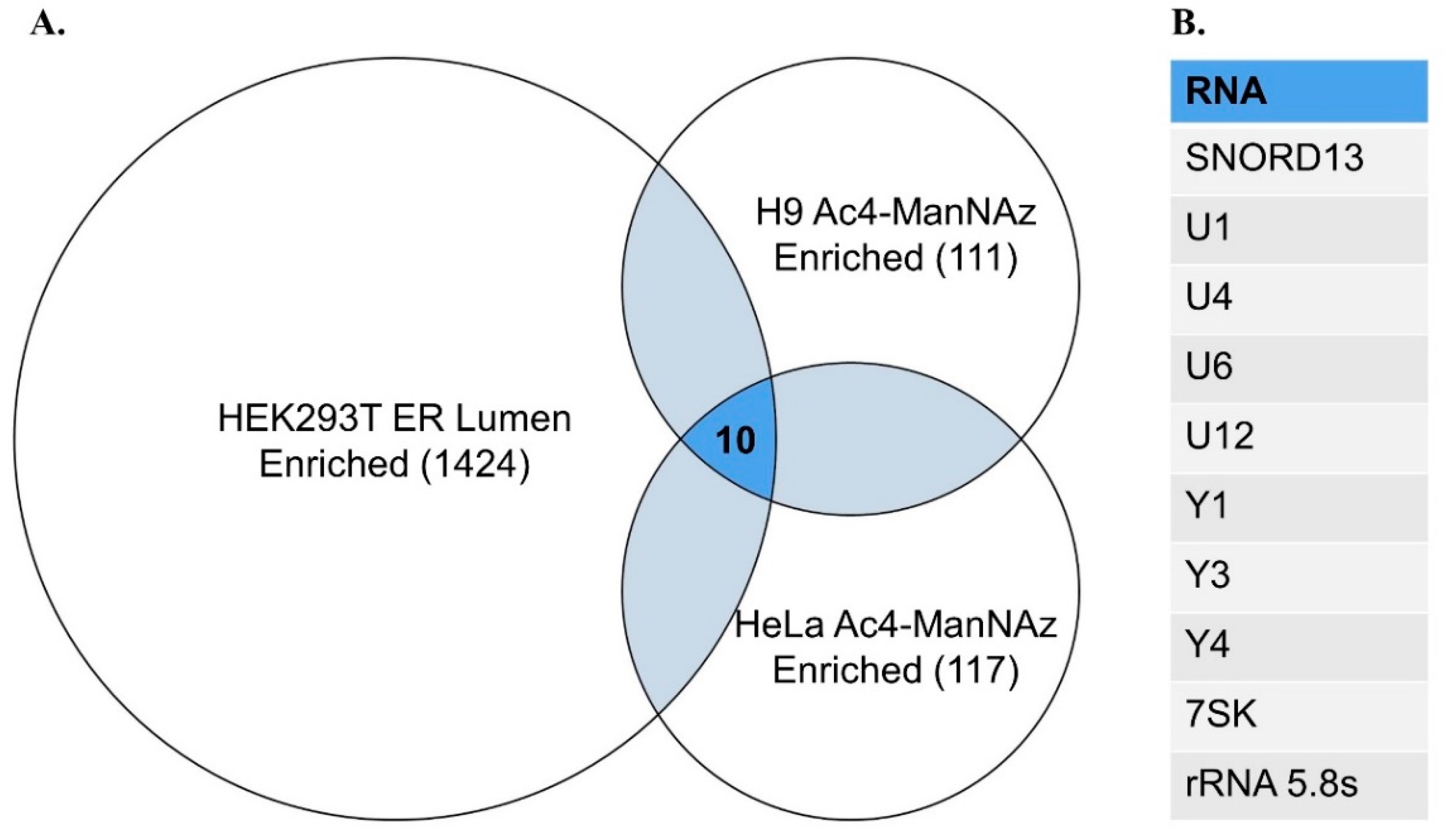
- Ren, Z.; Li, R.; Zhou, X.; Chen, Y.; Fang, Y.; Zou, P. Enzyme-Mediated Proximity Labeling Identifies Small RNAs in the Endoplasmic Reticulum Lumen. Biochemistry 2023, 62, 1844–1848. [Google Scholar] [CrossRef]
- Qian, D.; Cong, Y.; Wang, R.; Chen, Q.; Yan, C.; Gong, D. Structural Insight into the Human SID1 Transmembrane Family Member 2 Reveals Its Lipid Hydrolytic Activity. Nat Commun 2023, 14, 3568. [Google Scholar] [CrossRef]
- Feinberg, E.H.; Hunter, C.P. Transport of dsRNA into Cells by the Transmembrane Protein SID-1. Science 2003, 301, 1545–1547. [Google Scholar] [CrossRef]
- Nguyen, T.A.; Smith, B.R.C.; Elgass, K.D.; Creed, S.J.; Cheung, S.; Tate, M.D.; Belz, G.T.; Wicks, I.P.; Masters, S.L.; Pang, K.C. SIDT1 Localizes to Endolysosomes and Mediates Double-Stranded RNA Transport into the Cytoplasm. The Journal of Immunology 2019, 202, 3483–3492. [Google Scholar] [CrossRef]
- Stalder, D.; Gershlick, D.C. Direct Trafficking Pathways from the Golgi Apparatus to the Plasma Membrane. Seminars in Cell & Developmental Biology 2020, 107, 112–125. [Google Scholar] [CrossRef]
- Hong, W. SNAREs and Traffic. Biochim Biophys Acta 2005, 1744, 120–144. [Google Scholar] [CrossRef] [PubMed]
- Stewart, N.; Daly, J.; Drummond-Guy, O.; Krishnamoorthy, V.; Stark, J.C.; Riley, N.M.; Williams, K.C.; Bertozzi, C.R.; Wisnovsky, S. The Glycoimmune Checkpoint Receptor Siglec-7 Interacts with T-Cell Ligands and Regulates T-Cell Activation. Journal of Biological Chemistry 2024, 300, 105579. [Google Scholar] [CrossRef] [PubMed]
- Rosen, S.D.; Bertozzi, C.R. The Selectins and Their Ligands. Current Opinion in Cell Biology 1994, 6, 663–673. [Google Scholar] [CrossRef]
- McEver, R.P.; Zhu, C. Rolling Cell Adhesion. Annu Rev Cell Dev Biol 2010, 26, 363–396. [Google Scholar] [CrossRef]
- Koczera, P.; Martin, L.; Marx, G.; Schuerholz, T. The Ribonuclease A Superfamily in Humans: Canonical RNases as the Buttress of Innate Immunity. Int J Mol Sci 2016, 17, 1278. [Google Scholar] [CrossRef]
- Jonathan Perr; Andreas Langen; Karim Almahayni; Gianluca Nestola; Peiyuan Chai; Charlotta G. Lebedenko; Regan Volk; Reese M. Caldwell; Malte Spiekermann; Helena Hemberger; et al. RNA Binding Proteins and glycoRNAs Form Domains on the Cell Surface for Cell Penetrating Peptide Entry. bioRxiv 2023, 2023.09.04.556039. [CrossRef]
- Bottens, R.A.; Yamada, T. Cell-Penetrating Peptides (CPPs) as Therapeutic and Diagnostic Agents for Cancer. Cancers (Basel) 2022, 14, 5546. [Google Scholar] [CrossRef]
- Trabulo, S.; Cardoso, A.L.; Mano, M.; De Lima, M.C.P. Cell-Penetrating Peptides-Mechanisms of Cellular Uptake and Generation of Delivery Systems. Pharmaceuticals (Basel) 2010, 3, 961–993. [Google Scholar] [CrossRef]
- Lipfert, J.; Doniach, S.; Das, R.; Herschlag, D. Understanding Nucleic Acid-Ion Interactions. Annu Rev Biochem 2014, 83, 813–841. [Google Scholar] [CrossRef]
- Frankel, A.D.; Pabo, C.O. Cellular Uptake of the Tat Protein from Human Immunodeficiency Virus. Cell 1988, 55, 1189–1193. [Google Scholar] [CrossRef]
- Lowe, R.; Shirley, N.; Bleackley, M.; Dolan, S.; Shafee, T. Transcriptomics Technologies. PLoS Comput Biol 2017, 13, e1005457. [Google Scholar] [CrossRef]
- Hemberger, H.; Chai, P.; Lebedenko, C.G.; Caldwell, R.M.; George, B.M.; Flynn, R.A. Rapid and Sensitive Detection of Native glycoRNAs 2023.
- Yeini, E.; Ofek, P.; Pozzi, S.; Albeck, N.; Ben-Shushan, D.; Tiram, G.; Golan, S.; Kleiner, R.; Sheinin, R.; Israeli Dangoor, S.; et al. P-Selectin Axis Plays a Key Role in Microglia Immunophenotype and Glioblastoma Progression. Nat Commun 2021, 12, 1912. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Reis, C.A. Glycosylation in Cancer: Mechanisms and Clinical Implications. Nat Rev Cancer 2015, 15, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, E.; Macauley, M.S. Hypersialylation in Cancer: Modulation of Inflammation and Therapeutic Opportunities. Cancers (Basel) 2018, 10, 207. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.C. Mucins: Structure, Function, and Role in Pulmonary Diseases. Am J Physiol 1992, 263, L413–L429. [Google Scholar] [CrossRef]
- Cummings, R.D. The Repertoire of Glycan Determinants in the Human Glycome. Mol. BioSyst. 2009, 5, 1087. [Google Scholar] [CrossRef]
- Sterner, E.; Flanagan, N.; Gildersleeve, J.C. Perspectives on Anti-Glycan Antibodies Gleaned from Development of a Community Resource Database. ACS Chem. Biol. 2016, 11, 1773–1783. [Google Scholar] [CrossRef]
- Zhu, G.; Zhang, H.; Jacobson, O.; Wang, Z.; Chen, H.; Yang, X.; Niu, G.; Chen, X. Combinatorial Screening of DNA Aptamers for Molecular Imaging of HER2 in Cancer. Bioconjugate Chem. 2017, 28, 1068–1075. [Google Scholar] [CrossRef]
- Dard-Dascot, C.; Naquin, D.; d’Aubenton-Carafa, Y.; Alix, K.; Thermes, C.; Van Dijk, E. Systematic Comparison of Small RNA Library Preparation Protocols for Next-Generation Sequencing. BMC Genomics 2018, 19, 118. [Google Scholar] [CrossRef]
- Liao, X.; Zhu, W.; Zhou, J.; Li, H.; Xu, X.; Zhang, B.; Gao, X. Repetitive DNA Sequence Detection and Its Role in the Human Genome. Commun Biol 2023, 6, 954. [Google Scholar] [CrossRef]
- Maguire, S.; Lohman, G.J.S.; Guan, S. A Low-Bias and Sensitive Small RNA Library Preparation Method Using Randomized Splint Ligation. Nucleic Acids Res 2020, 48, e80. [Google Scholar] [CrossRef]
- Zayakin, P. sRNAflow: A Tool for the Analysis of Small RNA-Seq Data. ncRNA 2024, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Berko, D.; Carmi, Y.; Cafri, G.; Ben-Zaken, S.; Sheikhet, H.M.; Tzehoval, E.; Eisenbach, L.; Margalit, A.; Gross, G. Membrane-Anchored Β2-Microglobulin Stabilizes a Highly Receptive State of MHC Class I Molecules. The Journal of Immunology 2005, 174, 2116–2123. [Google Scholar] [CrossRef] [PubMed]
- Khvorova, A.; Kwak, Y.G.; Tamkun, M.; Majerfeld, I.; Yarus, M. RNAs That Bind and Change the Permeability of Phospholipid Membranes. Proc Natl Acad Sci U S A 1999, 96, 10649–10654. [Google Scholar] [CrossRef] [PubMed]
- Supplitt, S.; Karpinski, P.; Sasiadek, M.; Laczmanska, I. Current Achievements and Applications of Transcriptomics in Personalized Cancer Medicine. Int J Mol Sci 2021, 22, 1422. [Google Scholar] [CrossRef]
- Saferding, V.; Blüml, S. Innate Immunity as the Trigger of Systemic Autoimmune Diseases. Journal of Autoimmunity 2020, 110, 102382. [Google Scholar] [CrossRef]
| Cell Line | Species | Tissue | Pathology | Culture Type |
|---|---|---|---|---|
| HeLa | Human | Cervical | Cervical cancer (adenocarcinoma) | Adherent |
| H9 | Human | Embryonic T lymphocytes | Lymphoma | Suspension |
| K562 | Human | Bone marrow | Chronic myelogenous leukemia | Suspension |
| GM12878 | Human | PBMC | - | Suspension |
| T-ALL 4188 | Mouse | T lymphocytes | Acute lymphoblastic leukemia | Suspension |
| CHO | Chinese hamster | Ovarian | - | Adherent |
| 293T | Human | Embryonic kidney | - | Adherent |
| Primary mouse liver | Mouse | Liver | - | - |
| Primary mouse spleen | Mouse | Spleen | - | - |
| PANC-1 | Human | Pancreas | Epithelioid carcinoma | Adherent |
| Sh-SY5Y | Human | Bone marrow | Metastatic neuroblastoma | Adherent and suspension |
| MCF-10A | Human | Breast | Breast cancer (non-malignant) | Adherent |
| MCF-7 | Human | Breast | Breast cancer (malignant, adenocarcinoma) | Adherent and suspension |
| MDA-MB-231 | Human | Breast | Breast cancer (metastatic, adenocarcinoma) | Adherent |
| HL-60 | Human | PBMC | Acute promyelocytic leukemia | Suspension |
| THP-1 | Human | PBMC, monocyte | Acute monocytic leukemia | Suspension |
| HOXB8-immortalized primary bone marrow progenitors, differentiated into neutrophils | Mouse | Bone marrow | Differentiated into neutrophils | Suspension |
| HOXB8-immortalized primary bone marrow progenitors | Mouse | Bone marrow | - | Suspension |
| Primary bone marrow derived mouse neutrophils | Mouse | Bone marrow | - | Suspension |
| A549 | Human | Lung | Pulmonary carcinoma | Adherent |
| OCI-AML3 | Human | PBMC | Acute myeloid leukemia | Suspension |
| HUH7 | Human | Liver | Hepatocyte derived carcinoma | Adherent |
| HAP1 | Human | Bone marrow | Chronic myelogenous leukemia | Adherent |
| Primary CD19+ B cells | Human | PBMC | - | Suspension |
| Primary CD3+ T cells | Human | PBMC | - | Suspension |
| Primary CD14+ monocytes | Human | PBMC | - | Suspension |
| Primary CD16+ neutrophils | Human | PBMC | - | Suspension |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




