Submitted:
24 October 2024
Posted:
25 October 2024
You are already at the latest version
Abstract
Solid propellants have significant applications in aerospace, and improving their combustion performance has become a hot research topic in recent years. Carbon black (CB) has been widely used in the field of catalytic pyrolysis due to its low cost, controllable morphology, and ease of modification. Herein, we first investigates the structure-activity relationship between the physicochemical properties of nine types of CB with a particle size range of 1-15 μm and a mass fraction of 5%, including oil absorption value, compression oil absorption value, and oxygen content, and the catalytic pyrolysis of ammonium perchlorate (AP), concluding that the function relationship between oil absorption value and high-temperature peak is Y1=1032.67-9.618x+0.038x2, the function relationship between compression oil absorption value and high-temperature peak is Y2=145.537+62.365ln(x), and the function relationship between oxygen content and high-temperature peak is Y3=e(6.085-0.02/x). Based on these, we further uses coal tar, which is abundant in sources, as the raw material to prepare five types of coal-based CB marked as CB1-CB5 at pyrolysis temperatures of 1000~1800 ℃. The relative error between the actual peak value and the predicted value of the CB/AP composite particles is less than 5%, proving that the peak temperature model of CB/AP composite particles can guide actual production. This study contributes to promoting CB application in improving the catalytic pyrolysis performance of ammonium perchlorate and enhancing the high added value utilization of coal-derived products.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
2.2. Model Selection
2.3. Calculation
2.4. CB Preparation Methods
2.5. Testing of the Catalytic Performance of CB
2.6. Characterizations
3. Results
3.1. Analysis of the Physicochemical Parameters of CB
3.1.1. Analysis of the Structural Parameters of CB
3.1.2. Analysis of the Combustion Catalytic Performance of CB
3.1.3. Analysis of CB Quality Index System
3.2. Analysis of CB Preparation Process
3.2.1. Determination of Coal Tar Properties
3.2.2. Analysis of the Structural Parameters of CB
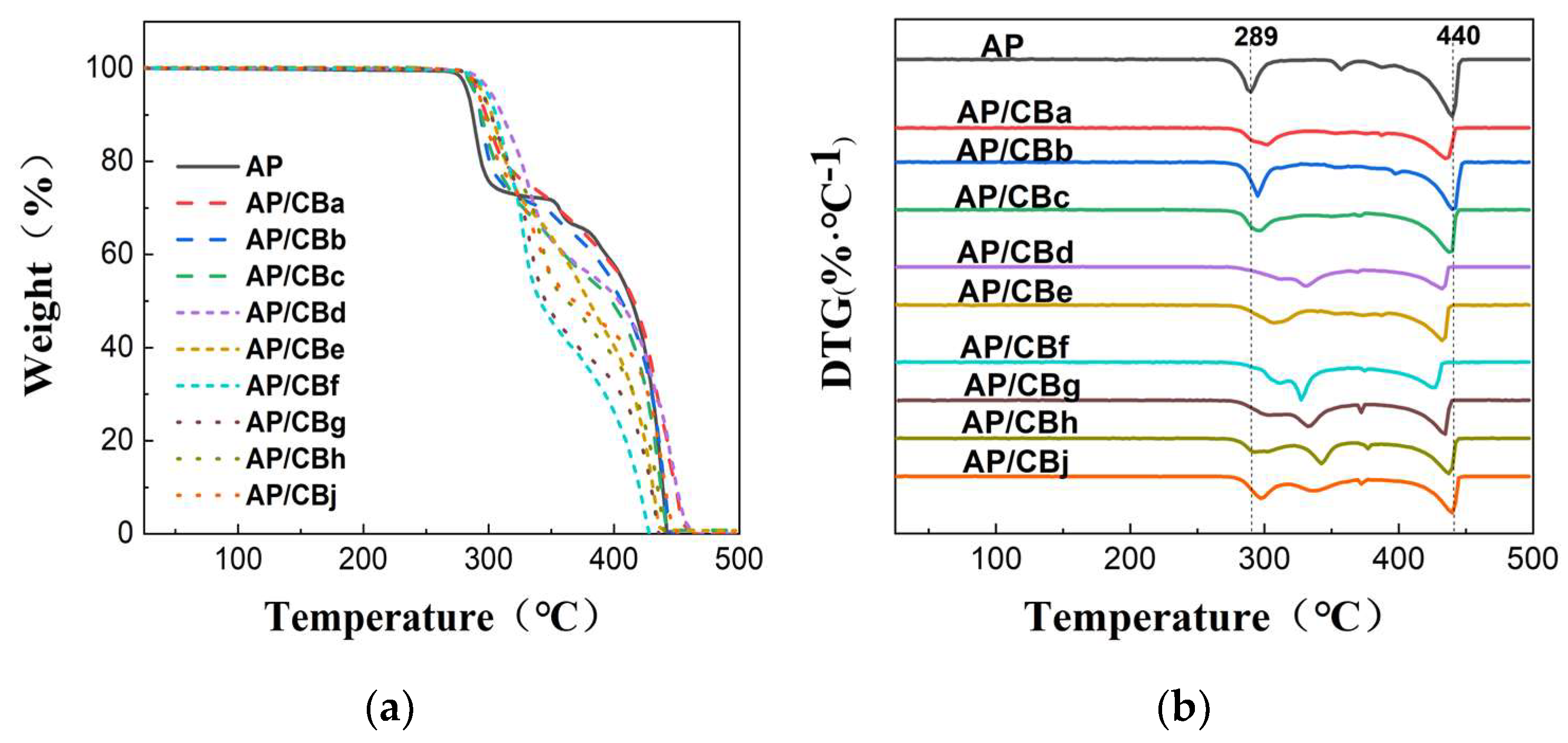
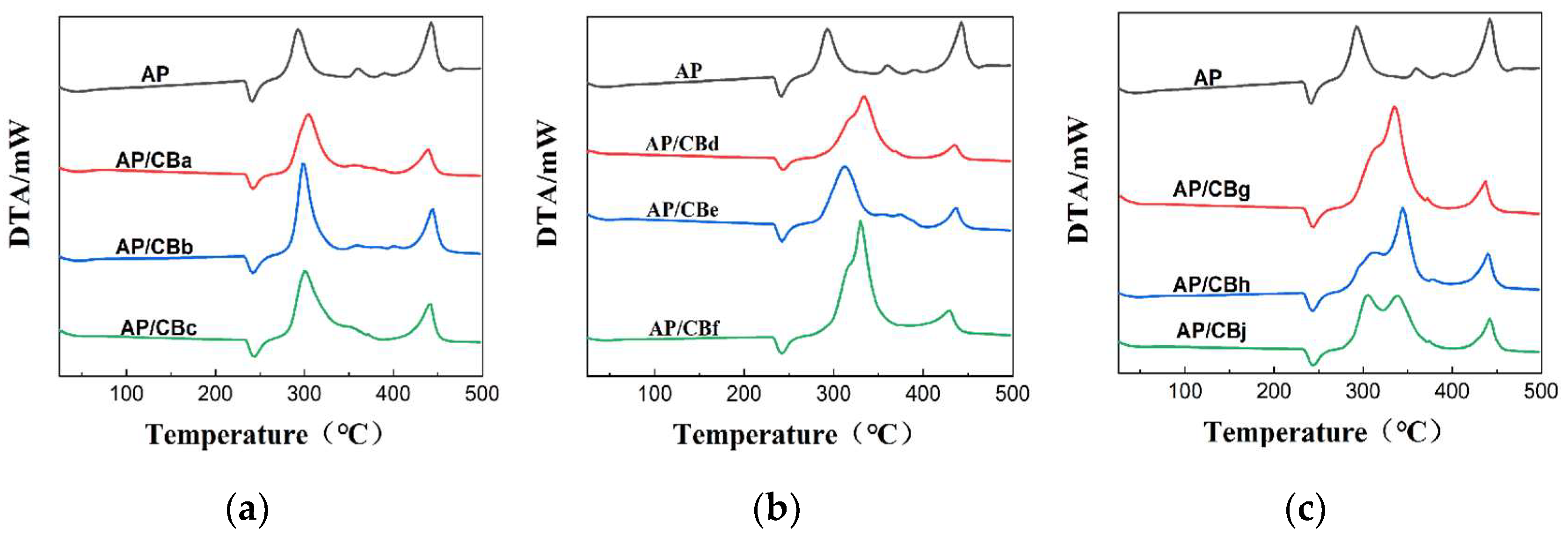
3.2.3. Analysis of the Thermal Catalytic Performance of CB
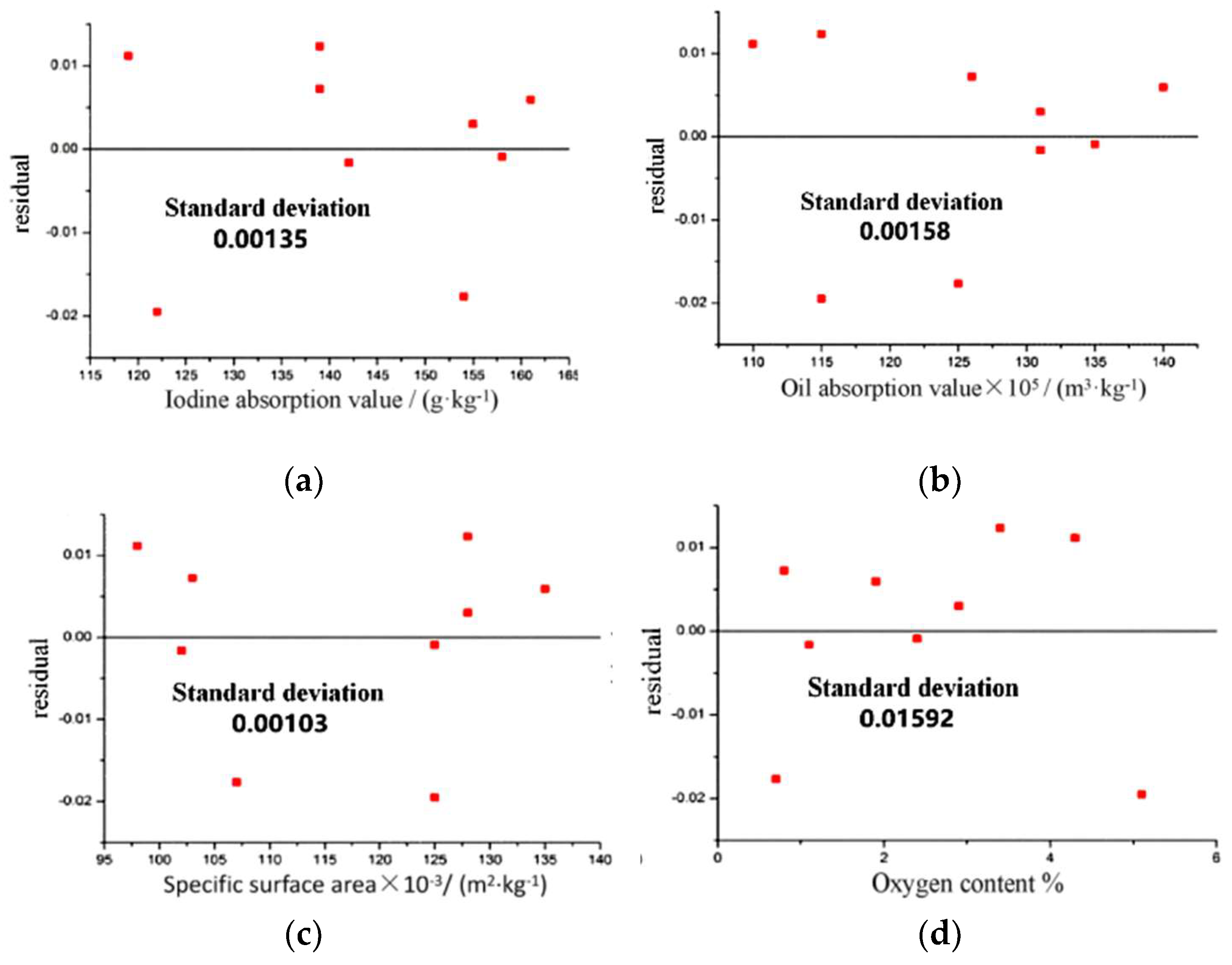
3.2.4. CB Preparation Process and Structural Parameter Regression Analysis
4. Discussion
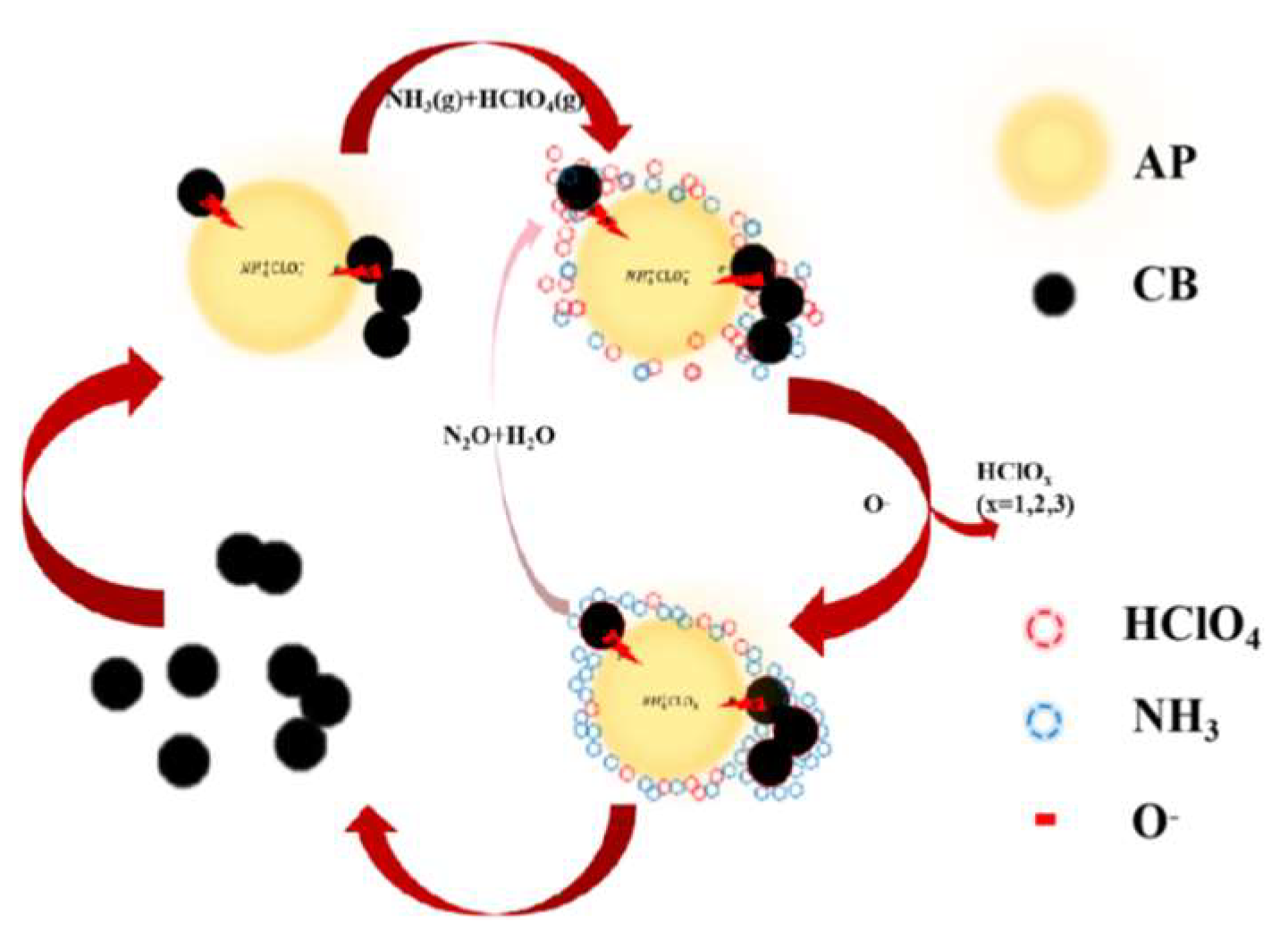
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chaturvedi, S.; Dave, P.N. Solid propellants: AP/HTPB composite propellants. Arab. J. Chem. 2019, 12, 2061-2068.
- Zhou, S.; Zhou, X.; Tang, G.; Guo, X.; Pang, A. Differences of thermal decomposition behaviors and combustion properties between CL-20-based propellants and HMX-based solid propellants. J. Therm. Anal. Calorim. 2019, 140, 2529-2540. [CrossRef]
- Yang, W.; Hu, R.; Zheng, L.; Yan, G.; Yan, W. Fabrication and investigation of 3D-printed gun propellants. Mater. Des. 2020, 192, 108761. [CrossRef]
- Assovskiy, I.G.; Kuznetsov, G.P.; Melik-Gaikazov, G.V. Energetic water compositions as rocket propellants. Acta Astronautica. 2021, 181, 643-648. [CrossRef]
- Padwal, M.B.; Castaneda, D.A.; Natan, B. Hypergolic combustion of boron based propellants. Proc. Combust. Inst. 2021, 38, 6703-6711. [CrossRef]
- Gallier, S.; Ferrand, A.; Plaud, M. Three-dimensional simulations of ignition of composite solid propellants. Combust. Flame. 2016, 173, 2-15. [CrossRef]
- Buchheiser, S.; Kistner, F.; Rhein, F.; Nirschl, H. Spray flame synthesis and multiscale characterization of carbon black–silica hetero-aggregates. Nanomaterials. 2023, 13(12), 1893. [CrossRef]
- Liang, T.X.; Yang, X.J.; Liu, Bo.; Song, R.D.; Xiao, F.; Yang, Y.J.; Wang, D.; Dong, M.Y.; Ren, J.; Xu, B.B.; Algadi, H.; Yang, Y.Y. Ammonium perchlorate@graphene oxide/Cu-MOF composites for efficiently catalyzing the thermal decomposition of ammonium perchlorate. Adv. Compos. Hybrid Mater. 2023, 6, 67. [CrossRef]
- Kakavas-Papaniaros, P. A. On the distribution of particles in propellant solids. Acta. Mech. 2019, 231, 863-875. [CrossRef]
- Dolgoborodov, A.Y.; Streletskii, A.N.; Shevchenko, A.A.; Vorobieva, G.A.; Val’yano, G.E. Thermal decomposition of mechanoactivated ammonium perchlorate. Thermochim. Acta. 2018, 669, 60-65. [CrossRef]
- Eslami, A.; Juibari, N.M.; Hosseini, S.G. Fabrication of ammonium perchlorate/copper-chromium oxides core-shell nanocomposites for catalytic thermal decomposition of ammonium perchlorate. Mater. Chem. Phys. 2016, 181, 12-20. [CrossRef]
- Elbasuney, S.; Yehia, M. Thermal decomposition of ammonium perchlorate catalyzed with CuO nanoparticles. Def. Technol. 2019, 15, 868-874. [CrossRef]
- Lei, G.; Zhong, Y.; Xu, Y.; Yang, F.; Bai, J.; Li, Z.; Zhang, J.; Zhang, T. New energetic complexes as catalysts for ammonium perchlorate thermal decomposition. Chin. J. Chem. 2021, 39, 1193-1198. [CrossRef]
- Zhang, J.H.; Jin, B.; Hao, W.J.; Song, Y.L.; Hou, C.J.; Huang, T.; Peng, R.F. Catalytic thermal decomposition of ammonium perchlorate by a series of lanthanide EMOFs. J. Rare Earths. 2022, 41, 516-522. [CrossRef]
- Sun, Y.; Han, Z.Y.; Du, Z.M.; Li, Z.Y.; Cong, X.M. Preparation and performance of environmental friendly sulphur-free propellant for fireworks. Appl. Therm. Eng. 2017, 126, 987-996. [CrossRef]
- Gao, F.; Li, S.F. Approach to the catalytic mechanism of fullerene in propellants. J. Energ. Mater. 2003, 21(1), 33-41.
- Ren, H.; Liu, Y.Y.; Jiao, Q.J.; Fu, X.F.; Yang, T.T. Preparation of nanocomposite PbO·CuO/CNTs via microemulsion process and its catalysis on thermal decomposition of RDX. J. Phys. Chem. Solids. 2010, 71, 149-152. [CrossRef]
- Sizov, V.A.; Denisyuk, A.P.; Demidova, L.A. Various carbon materials action on the burning rate modifiers of low-calorie double-base propellant. Combust. Sci. Technol. 2021, 195, 2327-2339. [CrossRef]
- Aprianti, N.; Kismanto, Agus.; Supriatna, N.K.; Yarsono, S.; Nainggolan, L.M.T.; Purawiardi, R.L.; Fariza, O.; Ermada, F.J.; Zuldian, P.; Raksodewanto, A.A.; Alamsyah, R. Prospect and challenges of producing carbon black from oil palm biomass: A review. Bioresource Technology Reports. 2023, 23, 101587. [CrossRef]
- Okoye, C.O.; Zhu, M.M.; Jones, L.; Zhang, J.; Zhang, Z.Z.; Zhang, D.K.; An investigation into the preparation of carbon black by partial oxidation of spent tyre pyrolysis oil. Waste Manag. 2022, 137, 110-120. [CrossRef]
- Yantaboot, K.; Amornsakchai, T. Effect of preparation methods and carbon black distribution on mechanical properties of short pineapple leaf fiber-carbon black reinforced natural rubber hybrid composites. Polym. Test. 2017, 61, 223-228. [CrossRef]
- Albright, T.; Hobeck, J. Investigating the electromechanical properties of carbon black-based conductive polymer composites via Stochastic modeling. Nanomaterials. 2023, 13(10), 1641. [CrossRef]
- Albright, T.; Hobeck, J. Characterization of carbon-black-based nanocomposite mixtures of varying dispersion for improving stochastic model fidelity. Nanomaterials. 2023, 13(5), 916. [CrossRef]
- Ma, T.; Zhao, X.; Cao, Y.; Wu, Y.; Zhou, Y.; Chen, H. L-histidine-functionalized carbon black pigment with zwitterionic property: Preparation, characterization and application. Dyes Pigments. 2020, 173, 107992. [CrossRef]
- Tang, R.; Yang, W.; Wang, H.; Zhou, J.; Zhang, Z.; Wu, S. Preparation of fly-ash-modified bamboo-shell carbon black and its mercury removal performance in simulated flue gases. Energy Fuels. 2016, 30, 4191-4196. [CrossRef]
- Liang, L.; Meng, S.; Tao, J.; Li, J.; Wang, X.; Guan, J.; Fu, H.; Dai, C.; Liu, H. Preparation and application of modified carbon black nanofluid as a novel flooding system in ultralow permeability reservoirs. J. Mol. Liq. 2023, 383, 122099. [CrossRef]
- Teng, Z.; Zhang, Z.; Li, X. Preparation of Pt catalysts supported on polyaniline modified carbon black and electrocatalytic methanol oxidation. Synth. Met. 2023, 293, 117256. [CrossRef]
- Lin, C.; Chung, D. D. L. Effect of carbon black structure on the effectiveness of carbon black thermal interface pastes. Carbon. 2007, 45, 2922-2931. [CrossRef]
- Gerspacher, M.; O’Farrell, C.P.; Nikiel, L.; Yang, H.H. Furnace carbon black characterization: continuing caga. Rubber Chem. Technol. 1996, 69(3), 569-576.
- Lockwood, F.C.; Niekerk, J.E.V.; Parametric study of a carbon black oil furnace. Combust. Flame. 1995, 103, 76-90. [CrossRef]
- Asokan, V.; Kosinski, P.; Skodvin, T.; Myrseth, V. Characterization of carbon black modified by maleic acid. Front. Mater. Sci. 2013, 7, 302-307. [CrossRef]
- Borah, D.; Satokawa, S.; Kato, S.; Kojima, T. Characterization of chemically modified carbon black for sorption application. Appl. Surf. Sci. 2008, 254, 3049-3056. [CrossRef]
- Heidarian, J.; Hassan, A. Characterization and comparison of fluoroelastomer unfilled, filled with carbon nanotube (unmodified, acid or base surface modified) and carbon black using TGA-GCMS. J. Elastom. Plast. 2021, 53, 861-885. [CrossRef]
- Dai, S.Y.; Ao, G.Y.; Kim, M.S. Properties of carbon black/SBR rubber composites filled by surface modified carbon blacks. Carbon Lett. 2007, 8, 115-119. [CrossRef]
- Li, R.; Guo, Q.; Shi, Z.; Pei, J. Effects of conductive carbon black on PZT/PVDF composites. Ferroelectrics. 2018, 526, 176-186. [CrossRef]
- Andrade-Guel, M.; Reyes-Rodríguez, P.Y.; Cabello-Alvarado, C.J.; Cadenas-Pliego, G.; Ávila-Orta, C.A. Influence of modified carbon black on nylon 6 nonwoven fabric and performance as adsorbent material. Nanomaterials. 2022, 12(23), 4247. [CrossRef]
- Prioglio, G.; Naddeo, S.; Giese, U.; Barbera, V.; Galimberti, M. Bio-based pyrrole compounds containing sulfur atoms as coupling agents of carbon black with unsaturated elastomers. Nanomaterials. 2023, 13(20), 2761. [CrossRef]
- Klyne, R.A.; Simpson, B.D.; Studebaker, M.L. A comparison of methods for determining surface areas of carbon black. Rubber Chem. Technol. 1973, 46(1), 192-203. [CrossRef]
- Mercer, H.N.; Boyer, A.H.; Bhusky, P.L.; Deviney, M.L. The third dimension of carbon black structure. Rubber Chem. Technol. 1976, 49(4), 1068-1075. [CrossRef]
- Voet, A.; Aboytes, P. Measurement of chain structure in carbon blacks. Rubber Chem. Technol. 1970, 43(6), 1359-1366. [CrossRef]
- Rodriguez-Fernandez, H.; Dasappa, S.; Sabado, K. D.; Camacho, J. Production of carbon black in turbulent spray flames of coal tar distillates. Appl. Sci. 2021, 11(21), 10001. [CrossRef]
- Lin, C.G.; Chung, D.D.L. Effect of carbon black structure on the effectiveness of carbon black thermal interface pastes. Carbon. 2007, 45, 2922-2931. [CrossRef]
- Goeringer, S.; Tacconi, N.R.D.; Chenthamarakshan, C.R.; Rajeshwar, K.; Wampler, W.A. Redox characterization of furnace carbon black surfaces. Carbon. 2001, 39, 515-522. [CrossRef]
- Ishola, F.A.; Inegbenebor, A.O.; Oyawale, F.A. Thermal modelling for a pilot scale pyrolytic furnace for production of carbon black. J. Phys. Conf. Ser. 2019, 1378, 032089. [CrossRef]
- Ouyang, C.; Ni, B.; Sun, Z.; Zhuang, J.; Xiao, H.; Wang, X. Boosting the ORR performance of modified carbon black via C-O bonds. Chem. Sci. 2019, 10, 2118-2123.
- Zhao, G.; Liang, L.H.; Lv, D.F.; Ji, W.J.; You, Q.; Dai, C.L. A novel nanofluid of modified carbon black nanoparticles for enhanced oil recovery in low permeability reservoirs. Pet. Sci. 2023, 20, 1598-1607. [CrossRef]
- Ono, K.; Yanaka, M.; Tanaka, S.; Saito, Y.; Aoki, H.; Fukuda, O.; Aoki, T.; Yamaguchi, T. Influence of furnace temperature and residence time on configurations of carbon black. Chem. Eng. J. 2012, 200-202, 541-548. [CrossRef]
- Ling, Q.; Wang, Z.H.; Liang, H.W.; Wu, R.; Lei, Zhao.; Zhao, Z.G.; Ke, Q.P.; Xie, R.L.; Liu, X.C.; Zhang, N.; Cui, P. Fluorinated graphene and its catalytic performance for AP pyrolysis: the promotion of F doping. Fuller. Nanotub. Carbon Nanostruct. 2023, 31(9), 888-896.
- Wang, C.A.; Xu, J.; Dai, J.; Wang, Y.; Shen, Y.; Zhang, Z.; Shen, R.; Ye, Y. Probing the reaction mechanism of Al/CuO nanocomposites doped with ammonium perchlorate. Nanotechnology. 2020, 31, 255401. [CrossRef]
- Bernigaud, P.; Davidenko, D.; Catoire, L. A revised model of ammonium perchlorate combustion with detailed kinetics. Cobust. Flame. 2023, 255, 112891. [CrossRef]

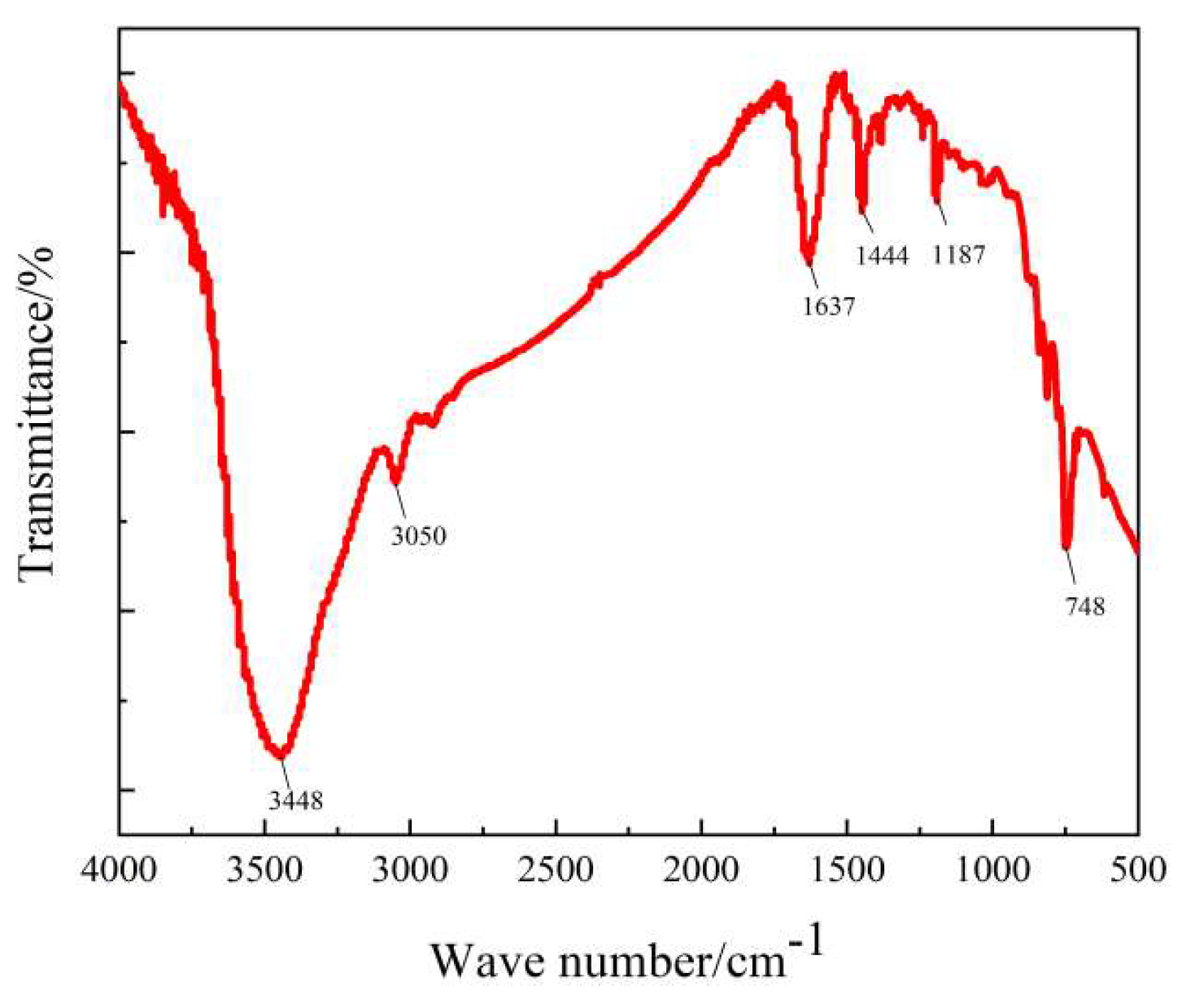
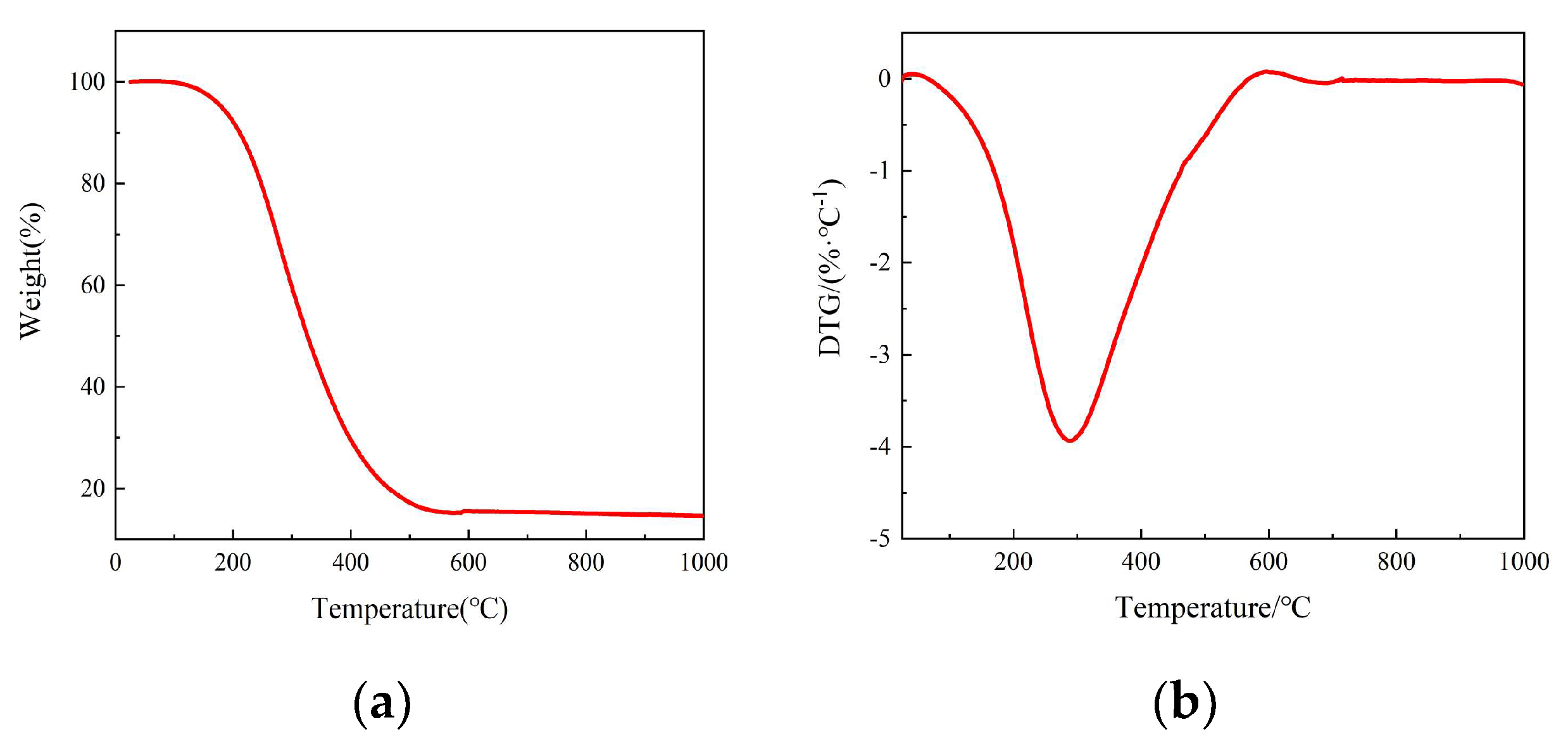
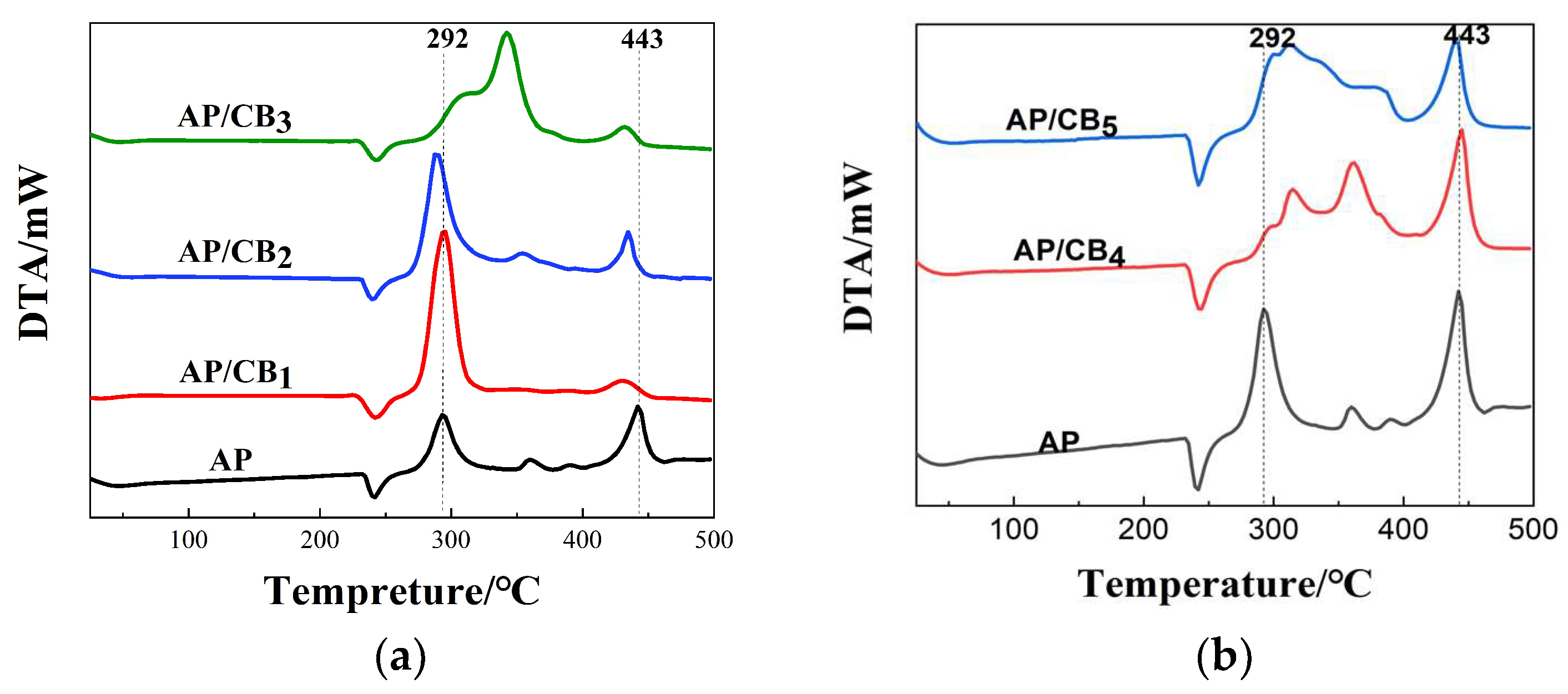
| Project | AP | AP/CB | ||||||||
| a a | b b | c c | d d | e e | f f | g g | h h | j j | ||
| D50 | - | 5.93 | 2.87 | 0.23 | 4.45 | 3.32 | 2.05 | 1.81 | 1.6 | 2.38 |
| R | - | 0.93 | 0.91 | 0.95 | 0.97 | 0.98 | 0.98 | 0.96 | 0.99 | 0.99 |
| Typical absorption peak /℃ | 241 | 241 | 243 | 243 | 243 | 242 | 241 | 243 | 243 | 243 |
| Low-temperature decomposition peak /℃ |
293 | 302 | 295 | 296 | 331 | 307 | 327 | 333 | 342 | 299 |
| High-temperature decomposition peak /℃ |
443 | 434 | 439 | 436 | 431 | 431 | 425 | 434 | 437 | 439 |
| Typical absorption peak variation value /℃ |
- | 0 | 2 | 2 | 2 | 1 | 0 | 2 | 2 | 2 |
| Low-temperature decomposition peak variation value /℃ |
- | 9 | 54 | 55 | 90 | 66 | 86 | 92 | 101 | 58 |
| High-temperature decomposition peak variation value /℃ |
- | -9 | -4 | -7 | -12 | -12 | -18 | -9 | -6 | -4 |
| Project | Pyrolysis temperatures | |||||
| Reference | 1000 ℃ CB1 |
1200 ℃ CB2 |
1400 ℃ CB3 |
1600 ℃ CB4 |
1800 ℃ CB5 |
|
| Iodine absorption value /(g·kg-1) |
90 | 112 | 119 | 139 | 145 | 102 |
| Oil absorption value × 105 /(m3·kg-1) |
110 | 105 | 115 | 131 | 121 | 140 |
| Compression oil absorption value × 105 /(m3·kg-1) |
101 | 92 | 80 | 104 | 100 | 106 |
| Nitrogen adsorption specific surface area × 10-3 /(m2·kg-1) |
104 | 78 | 77 | 110 | 140 | 97 |
| Φ value | 0.33 | 0.34 | 0.32 | 0.30 | 0.31 | 0.28 |
| O content (%) | - | 0.7 | 1.2 | 1.6 | 3.2 | 2.4 |
| Project | AP | AP/CB | ||||
| 1 | 2 | 3 | 4 | 5 | ||
| Typical absorption peak /℃ | 241 | 241 | 242 | 241 | 243 | 242 |
| Low-temperature decomposition peak /℃ |
293 | 295 | 288 | 341 | 313,361 | 311,384 |
| High-temperature decomposition peak /℃ |
443 | 429 | 433 | 433 | 439 | 435 |
| Typical absorption peak variation value /℃ |
- | 0 | 1 | 0 | 2 | 1 |
| Low-temperature decomposition peak variation value /℃ |
- | 2 | -5 | 48 | 20,68 | 18,91 |
| High-temperature decomposition peak variation value /℃ |
- | -14 | -10 | -10 | -4 | -8 |
| CB | Predictive value | Actual value | Relative error value 1 |
Relative error value 2 |
Relative error value 3 |
||
| 1 | 441.73 | 427.53 | 426.85 | 429 | 2.97% | 0.34% | 0.50% |
| 2 | 429.15 | 418.82 | 431.96 | 433 | 0.89% | 3.27% | 0.24% |
| 3 | 424.83 | 435.18 | 433.76 | 433 | 1.89% | 0.50% | 0.18% |
| 4 | 425.25 | 432.73 | 436.48 | 439 | 3.13% | 1.43% | 0.57% |
| 5 | 430.95 | 436.37 | 435.57 | 435 | 0.93% | 0.32% | 0.13% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




