Submitted:
28 November 2024
Posted:
29 November 2024
You are already at the latest version
Abstract
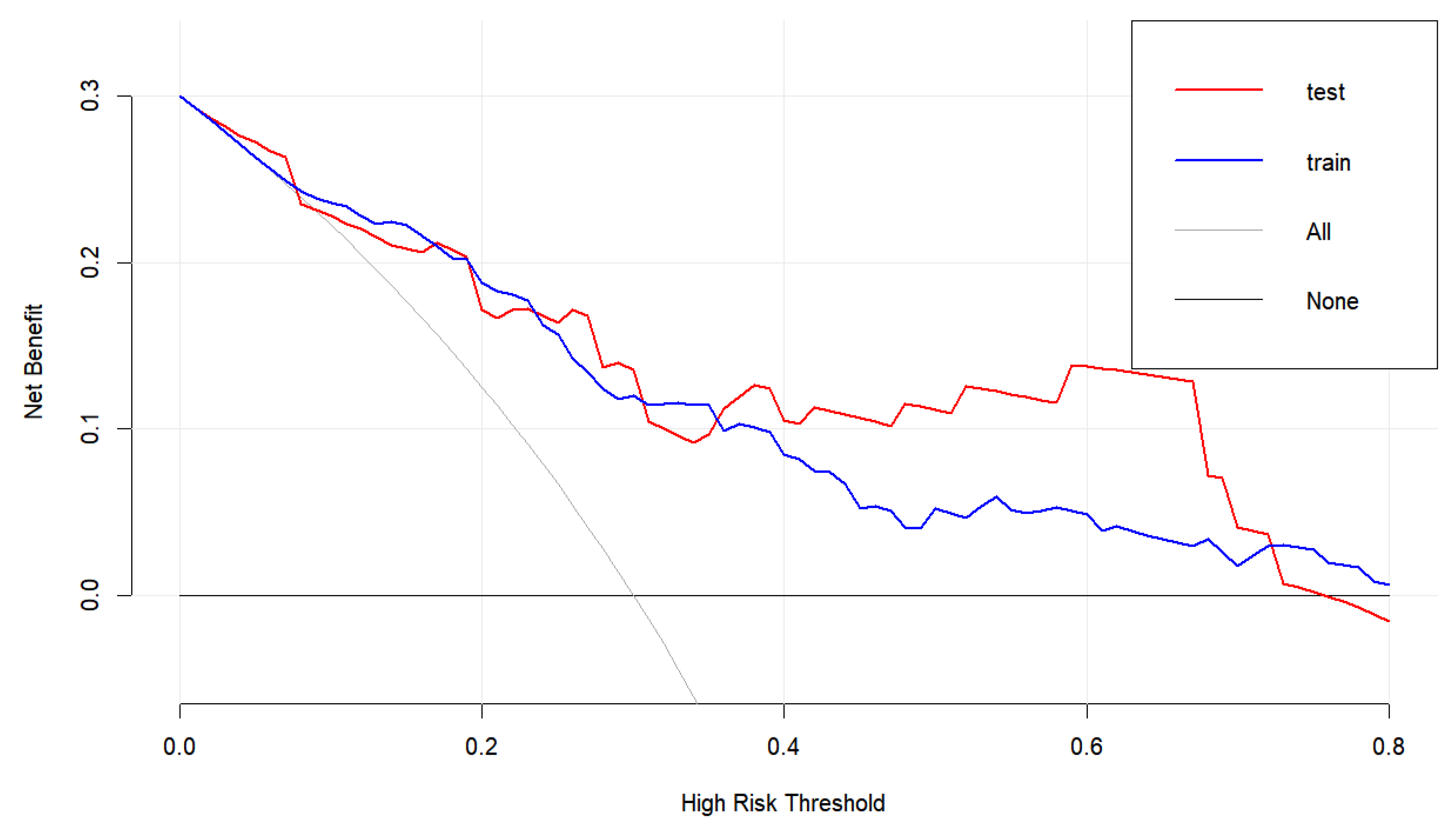
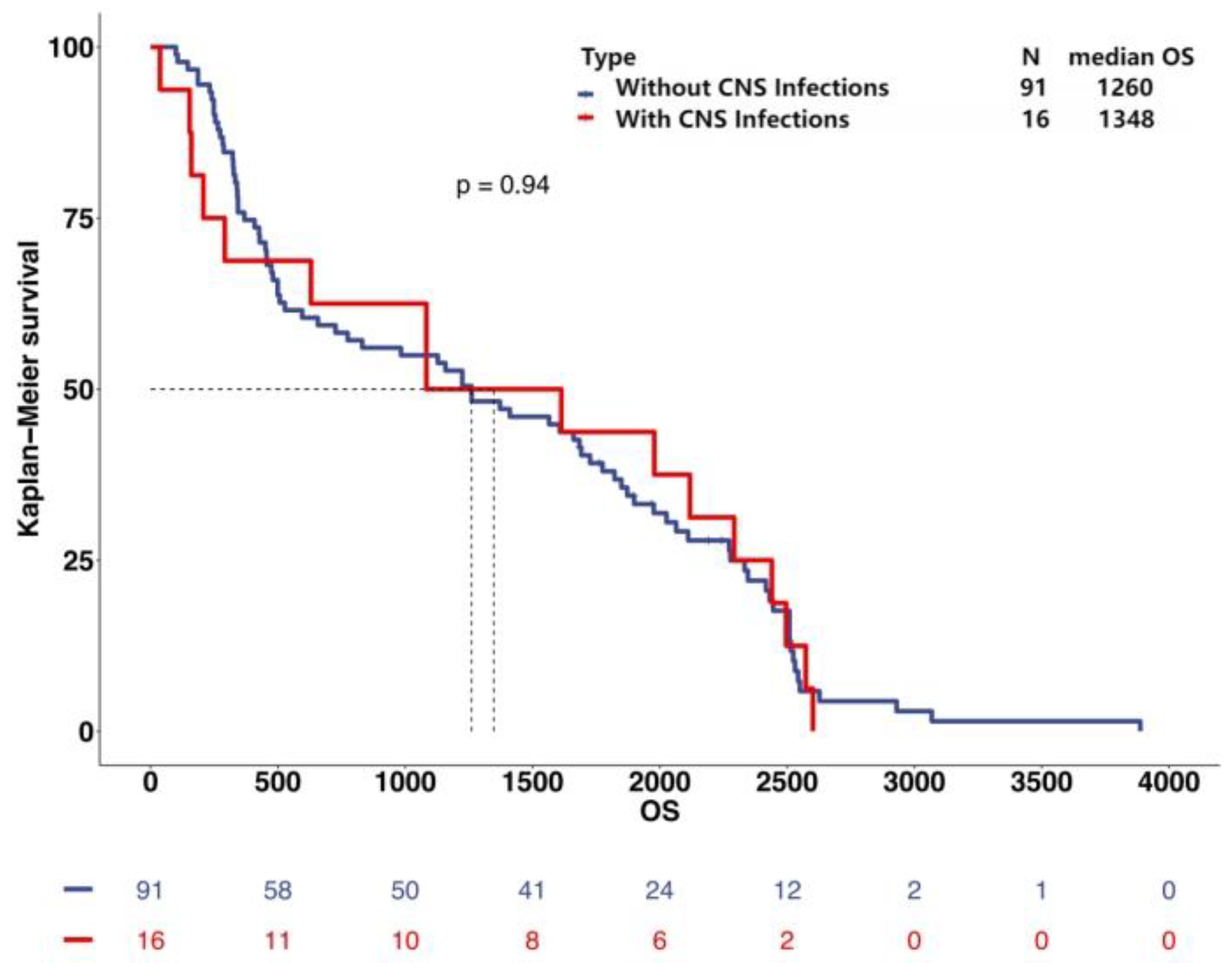
Objective: Glioma surgery, involving manipulation of brain parenchyma, can lead to elevated levels of inflammatory molecules in the central nervous system and alterations in cerebrospinal fluid circulation, potentially increasing the risk of central nervous system infection (CNSI). However, research on risk factors, clinical characteristics, and predictive models for CNSI following glioma resection remains scarce. Methods: We studied 435 patients undergoing glioma resection at Peking Union Medical College Hospital (2015-2023), collecting demographic, clinical, surgical, laboratory, and survival data. We analyzed risk factors for postoperative CNSI, developed a predictive model, and summarized cerebrospinal fluid (CSF) characteristics and antibiotic use in CNSI patients. The impact of CNSI on long-term prognosis was also assessed. Results: Of 435 patients, 65 (14.9%) developed CNSI. After excluding 98 patients with missing data, 61 of 337 analyzed patients had postoperative CNSI. Independent risk factors included ventricular opening, postoperative systemic infection, maximum cavity diameter, and preoperative peripheral blood mononuclear cell percentage. A nomogram based on these factors showed good predictive performance (C statistic = 0.797) and was validated internally and externally (AUC = 0.731). Decision curve analysis indicated positive net benefits within a 0-30% risk threshold. CSF in CNSI patients showed elevated protein (median 1.73 g/L), decreased glucose (median 2.7 mmol/L), and increased white blood cells (median 1478 × 10^6 /L) with a high polymorphonuclear cell percentage (median 84.1%). Meropenem and vancomycin were the most used antibiotics, with average durations of 7.79 and 8.41 days, respectively. Survival analysis showed no impact of CNSI on long-term prognosis. Conclusions: This study identifies risk factors for CNSI following glioma resection and develops a predictive model, aiding clinical decision-making. Summarized CSF characteristics and antibiotic use provide diagnostic and treatment references for CNSI post-glioma surgery.
Keywords:
1. Introduction
2. Methods
2.1. Patient Identification
2.2. Data Extraction
2.3. Statistical Analysis
3. Results
3.1. Study Population and Surgical Characteristics
3.2. Risk Factors for Postoperative CNSI
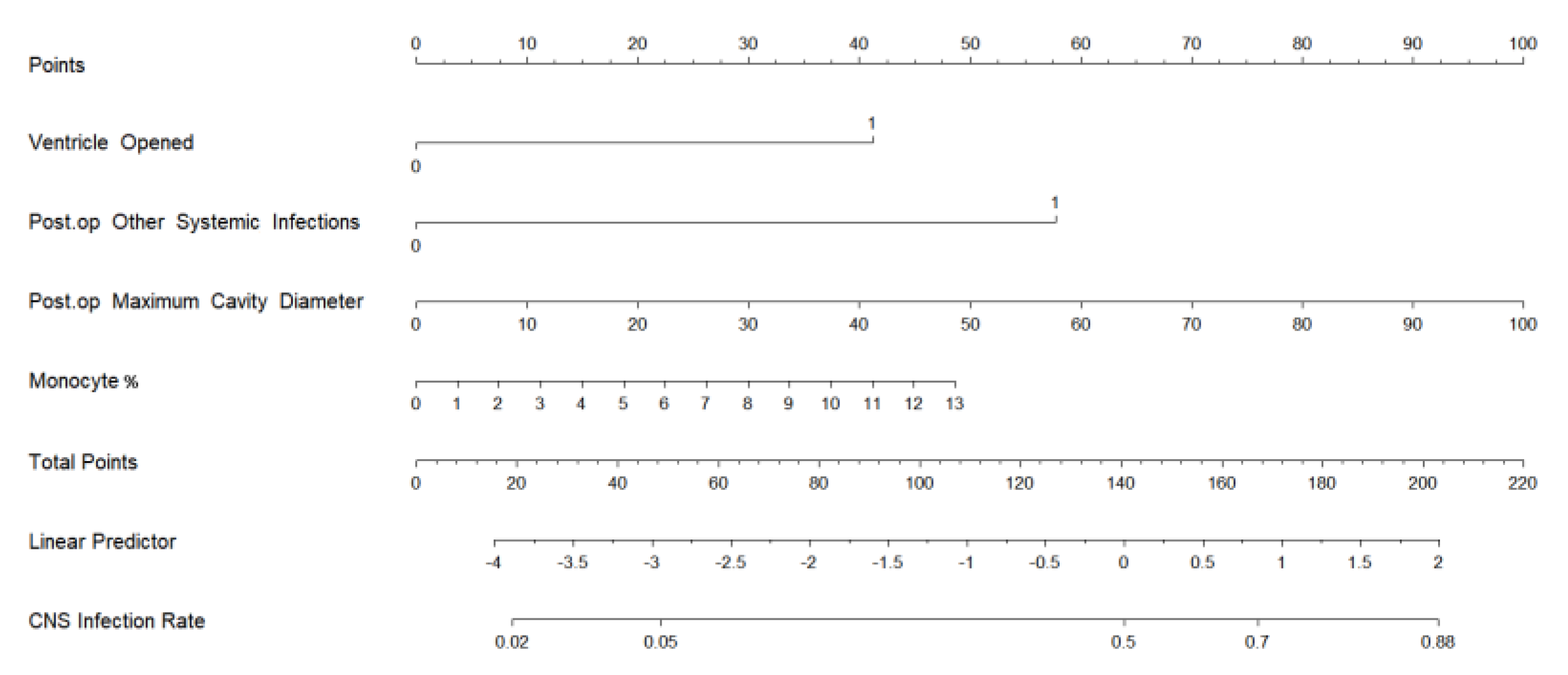
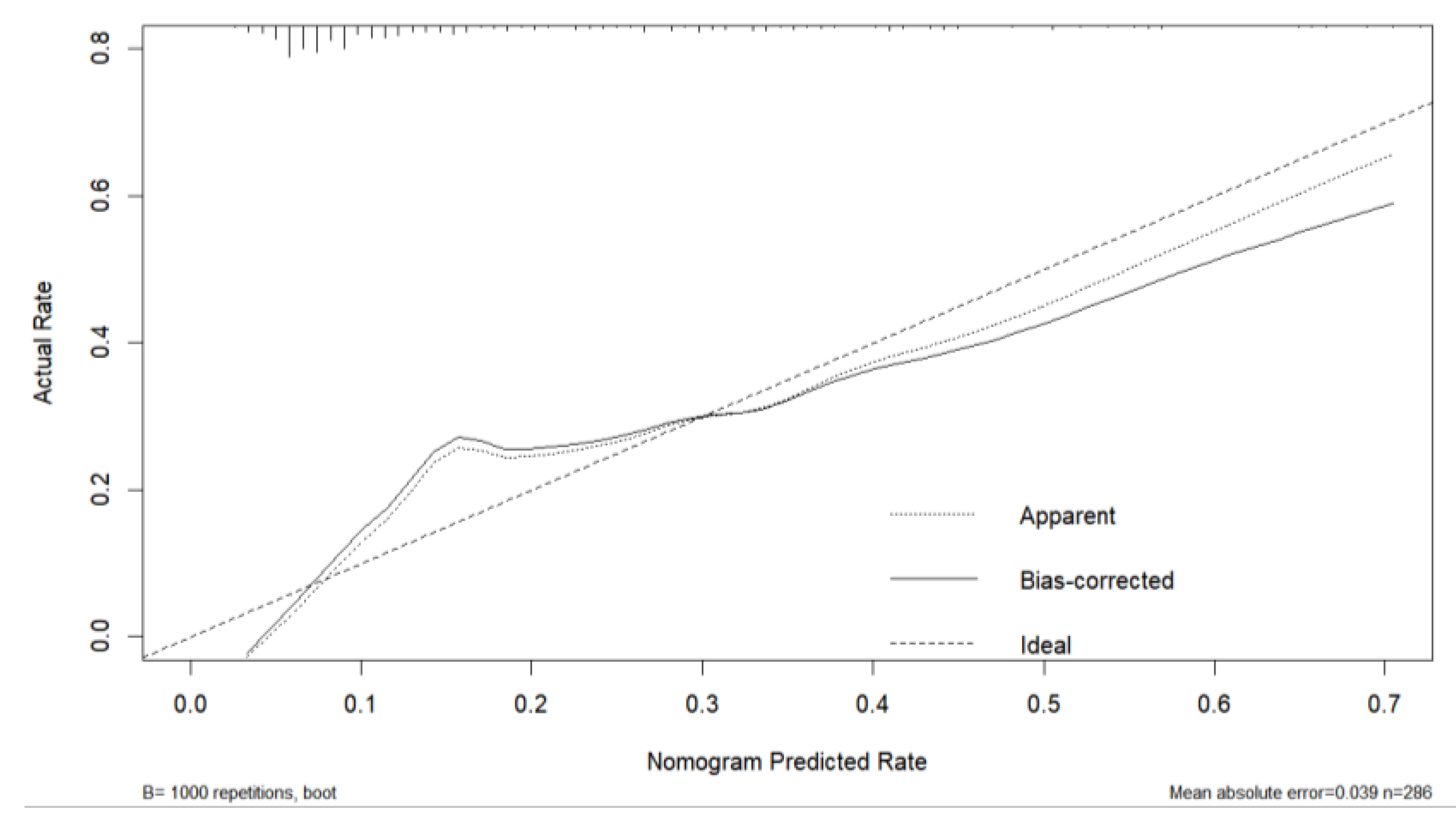
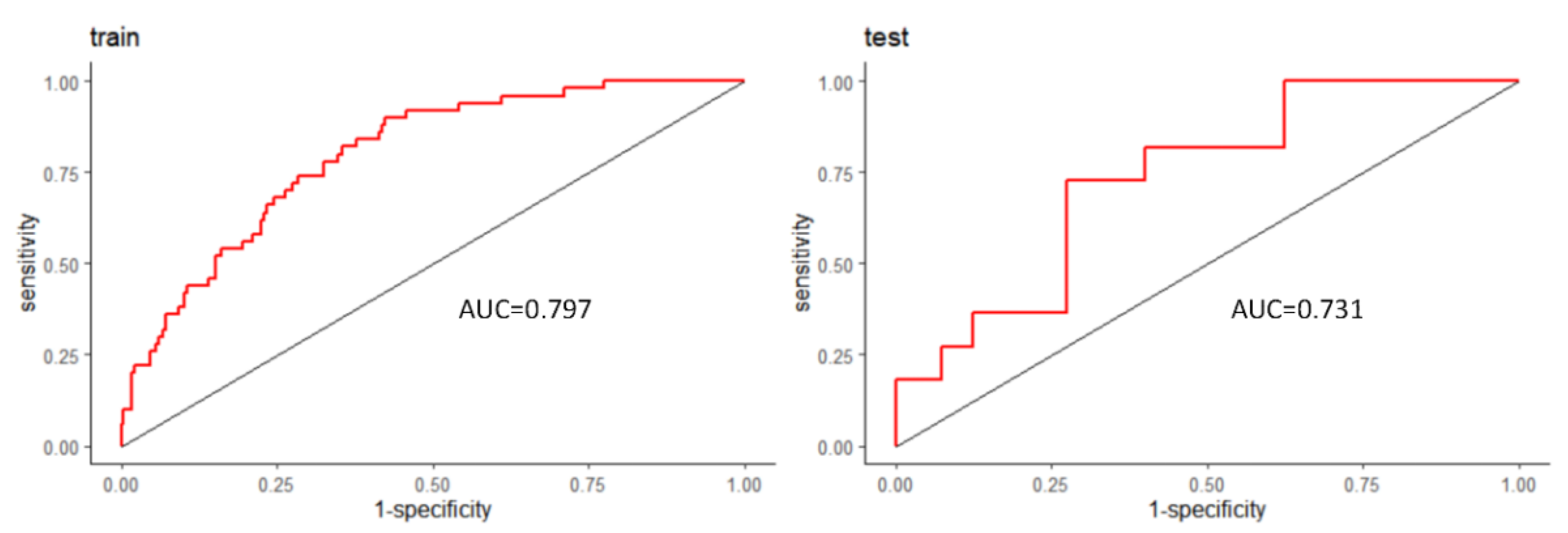
3.3. Model Development and Performance Validation
3.4. CSF Characteristics of Patients with CNSI
3.5. Microbiological Characteristics of Central Nervous System Infection and Antibiotic Use
3.6. Long-Term Follow-Up and Survival Analysis
4. Discussion
4.1. Risk Factors and Predictive Model
4.2. CSF Characteristics and Antibiotic Use
4.3. Survival Analysis
5. Limitation
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Reichert, M.C.; Medeiros, E.A.; Ferraz, F.A. Hospital-acquired meningitis in patients undergoing craniotomy: incidence, evolution, and risk factors. Am J Infect Control 2002, 30, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Korinek, A.M. , et al., Risk factors for adult nosocomial meningitis after craniotomy: role of antibiotic prophylaxis. Neurosurgery, 2008. 62 Suppl 2: p. 532-9.
- McClelland, S. , 3rd and W.A. Hall, Postoperative central nervous system infection: incidence and associated factors in 2111 neurosurgical procedures. Clin Infect Dis, 2007. 45(1): p. 55-9.
- Tang, K. and J.A. Zhou, Risk factors analysis of postoperative intracranial infection in patients with recurrent glioma. Chin Med J (Engl), 2013. 126(9): p. 1796.
- Guo, X.; Shi, Y.; Liu, D.; Li, Y.; Chen, W.; Wang, Y.; Wang, Y.; Xing, H.; Xia, Y.; Li, J.; et al. Clinical updates on gliomas and implications of the 5th edition of the WHO classification of central nervous system tumors. Front Oncol 2023, 13, 1131642. [Google Scholar] [CrossRef] [PubMed]
- Horbinski, C.; Nabors, L.B.; Portnow, J.; Baehring, J.; Bhatia, A.; Bloch, O.; Brem, S.; Butowski, N.; Cannon, D.M.; Chao, S.; et al. NCCN Guidelines® Insights: Central Nervous System Cancers, Version 2.2022. J Natl Compr Canc Netw 2023, 21, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Kourbeti, I.S.; Vakis, A.F.; Ziakas, P.; Karabetsos, D.; Potolidis, E.; Christou, S.; Samonis, G. Infections in patients undergoing craniotomy: risk factors associated with post-craniotomy meningitis. J Neurosurg 2015, 122, 1113–1119. [Google Scholar] [CrossRef]
- Gundamraj, S.; Hasbun, R. The Use of Adjunctive Steroids in Central Nervous Infections. Front Cell Infect Microbiol 2020, 10, 592017. [Google Scholar] [CrossRef]
- Durand, M.L.; Calderwood, S.B.; Weber, D.J.; Miller, S.I.; Southwick, F.S.; Caviness, V.S., Jr.; Swartz, M.N. Acute bacterial meningitis in adults. A review of 493 episodes. N Engl J Med 1993, 328, 21–28. [Google Scholar] [CrossRef]
- Horan, T.C.; Andrus, M.; Dudeck, M.A. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 2008, 36, 309–332. [Google Scholar] [CrossRef]
- Yau, B.; Hunt, N.H.; Mitchell, A.J.; Too, L.K. Blood‒Brain Barrier Pathology and CNS Outcomes in Streptococcus pneumoniae Meningitis. Int J Mol Sci 2018, 19. [Google Scholar] [CrossRef]
- Hirst, R.A.; Kadioglu, A.; O'Callaghan, C.; Andrew, P.W. The role of pneumolysin in pneumococcal pneumonia and meningitis. Clin Exp Immunol 2004, 138, 195–201. [Google Scholar] [CrossRef]
- Fitzgerald, D.; Waterer, G.W. Invasive Pneumococcal and Meningococcal Disease. Infect Dis Clin North Am 2019, 33, 1125–1141. [Google Scholar] [CrossRef]
- Green, E.W.; Ndiaye, M.; Hossain, I.M.; Olatunji, Y.A.; Sahito, S.M.; Salaudeen, R.; Badji, H.; Manjang, A.; Ceesay, L.; Hill, P.C.; et al. Pneumonia, Meningitis, and Septicemia in Adults and Older Children in Rural Gambia: 8 Years of Population-Based Surveillance. Clin Infect Dis 2023, 76, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xiu, L.; Xiao, Y.; Xie, Z.; Ren, L.; Peng, J. Simultaneous Detection of Key Bacterial Pathogens Related to Pneumonia and Meningitis Using Multiplex PCR Coupled With Mass Spectrometry. Front Cell Infect Microbiol 2018, 8, 107. [Google Scholar] [CrossRef] [PubMed]
- Rafferty, A.; Drew, R.J.; Cunney, R.; Bennett, D.; Marriott, J.F. Infant Escherichia coli urinary tract infection: is it associated with meningitis? Arch Dis Child 2022, 107, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Tebruegge, M.; Pantazidou, A.; Curtis, N. Question 1. How common is co-existing meningitis in infants with urinary tract infection? Arch Dis Child 2011, 96, 602–606. [Google Scholar] [CrossRef]
- Wallace, S.S.; Brown, D.N.; Cruz, A.T. Prevalence of Concomitant Acute Bacterial Meningitis in Neonates with Febrile Urinary Tract Infection: A Retrospective Cross-Sectional Study. J Pediatr 2017, 184, 199–203. [Google Scholar] [CrossRef]
- Nugent, J.; Childers, M.; Singh-Miller, N.; Howard, R.; Allard, R.; Eberly, M. Risk of Meningitis in Infants Aged 29 to 90 Days with Urinary Tract Infection: A Systematic Review and Meta-Analysis. J Pediatr 2019, 212, 102–110.e105. [Google Scholar] [CrossRef]
- Aronson, P.L.; Louie, J.P.; Kerns, E.; Jennings, B.; Magee, S.; Wang, M.E.; Gupta, N.; Kovaleski, C.; McDaniel, L.M.; McDaniel, C.E. Prevalence of Urinary Tract Infection, Bacteremia, and Meningitis Among Febrile Infants Aged 8 to 60 Days With SARS-CoV-2. JAMA Netw Open 2023, 6, e2313354. [Google Scholar] [CrossRef]
- McLellan, L.K.; Hunstad, D.A. Urinary Tract Infection: Pathogenesis and Outlook. Trends Mol Med 2016, 22, 946–957. [Google Scholar] [CrossRef]
- Bowyer, G.S.; Loudon, K.W.; Suchanek, O.; Clatworthy, M.R. Tissue Immunity in the Bladder. Annu Rev Immunol 2022, 40, 499–523. [Google Scholar] [CrossRef]
- Phuong, P.C.; Hung, N.Q.; Ngoc, T.B.; Rades, D.; Khoa, M.T. Rotating Gamma System Irradiation: A Promising Treatment for Low-grade Brainstem Gliomas. In Vivo 2017, 31, 957–960. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Y.; Zhang, X.S.; Shen, B.Z. Expression of VEGF and MMP-9 and MRI imaging changes in cerebral glioma. Oncol Lett 2011, 2, 1171–1175. [Google Scholar] [CrossRef] [PubMed]
- Krivosheya, D.; Prabhu, S.S.; Weinberg, J.S.; Sawaya, R. Technical principles in glioma surgery and preoperative considerations. J Neurooncol 2016, 130, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Ille, S.; Schwendner, M.; Zhang, W.; Schroeder, A.; Meyer, B.; Krieg, S.M. Tractography for Subcortical Resection of Gliomas Is Highly Accurate for Motor and Language Function: ioMRI-Based Elastic Fusion Disproves the Severity of Brain Shift. Cancers (Basel) 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Auger, J.P.; Rivest, S.; Benoit-Biancamano, M.O.; Segura, M.; Gottschalk, M. Inflammatory Monocytes and Neutrophils Regulate Streptococcus suis-Induced Systemic Inflammation and Disease but Are Not Critical for the Development of Central Nervous System Disease in a Mouse Model of Infection. Infect Immun 2020, 88. [Google Scholar] [CrossRef]
- Lepennetier, G.; Hracsko, Z.; Unger, M.; Van Griensven, M.; Grummel, V.; Krumbholz, M.; Berthele, A.; Hemmer, B.; Kowarik, M.C. Cytokine and immune cell profiling in the cerebrospinal fluid of patients with neuro-inflammatory diseases. J Neuroinflammation 2019, 16, 219. [Google Scholar] [CrossRef]
- Djukic, M.; Mildner, A.; Schmidt, H.; Czesnik, D.; Brück, W.; Priller, J.; Nau, R.; Prinz, M. Circulating monocytes engraft in the brain, differentiate into microglia and contribute to the pathology following meningitis in mice. Brain 2006, 129, 2394–2403. [Google Scholar] [CrossRef]
- Montemurro, N.; Pahwa, B.; Tayal, A.; Shukla, A.; De Jesus Encarnacion, M.; Ramirez, I.; Nurmukhametov, R.; Chavda, V.; De Carlo, A. Macrophages in Recurrent Glioblastoma as a Prognostic Factor in the Synergistic System of the Tumor Microenvironment. Neurol Int 2023, 15, 595–608. [Google Scholar] [CrossRef]
- Bauler, T.J.; Starr, T.; Nagy, T.A.; Sridhar, S.; Scott, D.; Winkler, C.W.; Steele-Mortimer, O.; Detweiler, C.S.; Peterson, K.E. Salmonella Meningitis Associated with Monocyte Infiltration in Mice. Am J Pathol 2017, 187, 187–199. [Google Scholar] [CrossRef]
- Seidelman, J.L.; Mantyh, C.R.; Anderson, D.J. Surgical Site Infection Prevention: A Review. Jama 2023, 329, 244–252. [Google Scholar] [CrossRef]
- Medikonda, R.; Patel, K.; Jackson, C.; Saleh, L.; Srivastava, S.; Feghali, J.; Mohan, A.; Pant, A.; Jackson, C.M.; Weingart, J.; et al. The safety and efficacy of dexamethasone in the perioperative management of glioma patients. J Neurosurg 2022, 136, 1062–1069. [Google Scholar] [CrossRef]
- Dixit, K.S.; Kumthekar, P.U. Optimal Management of Corticosteroids in Patients with Intracranial Malignancies. Curr Treat Options Oncol 2020, 21, 77. [Google Scholar] [CrossRef] [PubMed]
- Mount, H.R.; Boyle, S.D. Aseptic and Bacterial Meningitis: Evaluation, Treatment, and Prevention. Am Fam Physician 2017, 96, 314–322. [Google Scholar] [PubMed]
- Eltwisy, H.O.; Twisy, H.O.; Hafez, M.H.; Sayed, I.M.; El-Mokhtar, M.A. Clinical Infections, Antibiotic Resistance, and Pathogenesis of Staphylococcus haemolyticus. Microorganisms 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.R.; Lu, C.H.; Wu, J.J.; Chang, H.W.; Chien, C.C.; Lei, C.B.; Chang, W.N. Coagulase-negative staphylococcal meningitis in adults: clinical characteristics and therapeutic outcomes. Infection 2005, 33, 56–60. [Google Scholar] [CrossRef]
- Sharma, R.; Goda, R.; Borkar, S.A.; Katiyar, V.; Agarwal, S.; Kumar, A.; Mohapatra, S.; Kapil, A.; Suri, A.; Kale, S.S. Outcome following postneurosurgical Acinetobacter meningitis: an institutional experience of 72 cases. Neurosurg Focus 2019, 47, E8. [Google Scholar] [CrossRef]
- Baldwin, C.M.; Lyseng-Williamson, K.A.; Keam, S.J. Meropenem: a review of its use in the treatment of serious bacterial infections. Drugs 2008, 68, 803–838. [Google Scholar] [CrossRef]
- Zhanel, G.G.; Wiebe, R.; Dilay, L.; Thomson, K.; Rubinstein, E.; Hoban, D.J.; Noreddin, A.M.; Karlowsky, J.A. Comparative review of the carbapenems. Drugs 2007, 67, 1027–1052. [Google Scholar] [CrossRef]
- Yew, K.S.; Cheng, E.M. Diagnosis of acute stroke. Am Fam Physician 2015, 91, 528–536. [Google Scholar]
- Shahan, B.; Choi, E.Y.; Nieves, G. Cerebrospinal Fluid Analysis. Am Fam Physician 2021, 103, 422–428. [Google Scholar]
- Bohman, L.E.; Gallardo, J.; Hankinson, T.C.; Waziri, A.E.; Mandigo, C.E.; McKhann, G.M., 2nd; Sisti, M.B.; Canoll, P.; Bruce, J.N. The survival impact of postoperative infection in patients with glioblastoma multiforme. Neurosurgery 2009, 64, 828–834. [Google Scholar] [CrossRef]
- De Bonis, P.; Albanese, A.; Lofrese, G.; de Waure, C.; Mangiola, A.; Pettorini, B.L.; Pompucci, A.; Balducci, M.; Fiorentino, A.; Lauriola, L.; et al. Postoperative infection may influence survival in patients with glioblastoma: simply a myth? Neurosurgery 2011, 69, 864–868. [Google Scholar] [CrossRef]
| Variable | No CNS Infections | CNS Infections | p Value |
|---|---|---|---|
| No. of Procedures | 276 | 61 | |
| Age(yrs) | 47.00 [33.75, 57.00] | 46.00 [34.00, 55.00] | 0.67 |
| Female Sex | 123 (44.57%) | 23 (37.70%) | 0.40 |
| BMI | 24.03 [20.29, 27.77] | 24.76 [21.03, 28.49] | 0.17 |
| Primary Glioma | 231 (83.70%) | 51 (83.61%) | 1.00 |
| Diabetes mellitus | 16 (5.80%) | 2 (3.28%) | 0.63 |
| Comorbidities in Other Systems | 113 (40.94%) | 27 (44.26%) | 0.74 |
| Pre-op Radiotherapy | 40 (14.49%) | 12 (19.67%) | 0.41 |
| Pre-op Chemotherapy | 36 (13.04%) | 12 (19.67%) | 0.26 |
| Pre-op Steroid Use | 76 (27.54%) | 16 (26.23%) | 0.96 |
| Pre-op Concomitant Organ Infections | 3 (1.09%) | 3 (4.92%) | 0.13 |
| Pre-op Tumor Necrosis | 173 (62.68%) | 41 (67.21%) | 0.60 |
| Maximum Tumor Diameter(mm) | 41.60 [32.15, 55.00] | 49.50 [40.00, 60.00] | <0.01* |
| Surgery Duration(hours) | 5.00 [4.00, 6.30] | 5.50 [4.70, 7.00] | 0.01* |
| Ventricle Opened | 48 (17.39%) | 29 (47.54%) | <0.01* |
| Frontal/Ethmoid Opened | 18 (6.52%) | 4 (6.56%) | 1.00 |
| Tumor Cavity Catheter Insertion | 95 (34.42%) | 37 (60.66%) | <0.01* |
| External Drain Duration(days) | 0.00 [0.00, 1.00] | 1.00 [0.00, 3.00] | <0.01* |
| Post-op Other Systemic Infections | 10 (3.62%) | 11 (18.03%) | <0.01* |
| Post-op Seizures | 16 (5.80%) | 3 (4.92%) | 1.00 |
| Post-op Steroid Use | 254 (92.03%) | 59 (96.72%) | 0.31 |
| Post-op Maximum Cavity Diameter(mm) | 43.15 [33.77, 54.82] | 53.90 [46.90, 63.70] | <0.01* |
| Multiple Hospital Surgeries | 1 (0.36%) | 4 (6.56%) | <0.01* |
| Pre-op Blood Cell Tests | |||
| Absolute WBC Count (×10^9/L) | 6.20 [5.19, 7.74] | 6.53 [4.94, 8.17] | 0.57 |
| Absolute Lymphocyte Count(×10^9/L) | 1.74 [1.34, 2.22] | 1.68 [1.32, 2.12] | 0.38 |
| Lymphocyte % | 28.79 [18.92,38.66] | 27.20 [18.43,35.97] | 0.25 |
| Absolute Monocyte Count(×10^9/L) | 0.35 [0.28, 0.43] | 0.37 [0.30, 0.47] | 0.14 |
| Monocyte % | 5.60 [4.68, 6.50] | 6.30 [4.90, 6.90] | 0.03* |
| Absolute Neutrophil Count(×10^9/L) | 3.75 [2.83, 4.87] | 3.70 [2.89, 5.42] | 0.48 |
| Neutrophil % | 60.80 [54.05, 68.82] | 62.60 [56.00, 69.60] | 0.24 |
| Absolute Eosinophil Count (×10^9/L) | 0.09 [0.05, 0.15] | 0.08 [0.05, 0.14] | 0.76 |
| Eosinophil % | 1.50 [0.80, 2.50] | 1.50 [0.80, 2.60] | 0.94 |
| Absolute Basophil Count (×10^9/L) | 0.03 [0.02, 0.03] | 0.02 [0.02, 0.04] | 0.97 |
| Basophil % | 0.40 [0.30, 0.60] | 0.40 [0.20, 0.60] | 0.83 |
| Variable | OR | CI | P |
|---|---|---|---|
| Ventricle Opened | 2.97 | 1.54-5.71 | <0.01* |
| Post-op Other Systemic Infections | 4.03 | 1.34-12.14 | 0.01* |
| Post-op Maximum Cavity Diameter | 1.03 | 1.01-1.06 | 0.02* |
| Pre-op Blood Monocyte % | 1.19 | 1.01-1.41 | 0.04* |
| Tumor Cavity Catheter Insertion | 1.63 | 0.66-4.02 | 0.29 |
| External Drain Duration | 1.07 | 0.84-1.38 | 0.57 |
| Maximum Tumor Diameter | 1.00 | 0.97-1.02 | 0.72 |
| Surgery Duration | 0.96 | 0.81-1.15 | 0.66 |
| Multiple Hospital Surgeries | 8.10 | 0.73-89.54 | 0.09 |
| Variable | Median | IQR | Reference |
|---|---|---|---|
| Protein(g/L) | 1.73 | [1.03, 3.06] | 0.15-0.45 |
| Glucose(mmol/L) | 2.7 | [1.90, 3.70] | 2.4-4.5 |
| Chloride(mmol/L) | 120 | [116.00, 122.00] | 120-132 |
| WBC(10^6/L) | 1478 | [467, 4204.25] | 0-8 |
| Multinucleated Cell % | 84.1 | [74.55, 90.83] | <70 |
| Bacteria | Sensitive Antibiotics | Resistant Antibiotics |
|---|---|---|
| Staphylococcus haemolyticus | Gentamicin,Linezolid, Selectrin,Teicoplanin, Vancomycin |
Ciprofloxacin,Oxacillin, Erythromycin,Penicillin G |
| Staphylococcus epidermidis | Gentamicin,Linezolid, Vancomycin,Rifampicin, Selectrin,Teicoplanin |
Oxacillin,Penicillin G |
| Acinetobacter baumanii |
Minocycline,Tigecycline | Amikacin,Ceftazidime, Ciprofloxacin,Levofloxacin,Cefperazone-Sulbactam,Meropenem,Selectrin, Sulbactam-Ampicillin, Doxycycline,Cefepime, Imipenem,Tobramycin, Piperacillin-Tazobactam |
| Antibiotic Varieties | Frequency of Use(n=50) | Average Time of Use(days) | |
|---|---|---|---|
| Gram-positive | Vancomycin | 43 | 7.79 |
| Linezolid | 4 | 7.75 | |
| Gram-negative | Meropenem | 27 | 8.41 |
| Cefperazone | 17 | 6.88 | |
| Ceftriaxone | 6 | 6.83 | |
| Ceftazidime | 7 | 8.43 | |
| Common Antibiotic Combinations | Meropenem+ Vancomycin |
23 | 8.13 |
| Cefperazone+ Vancomycin | 8 | 6.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





