Submitted:
17 October 2024
Posted:
18 October 2024
You are already at the latest version
Abstract
Keywords:
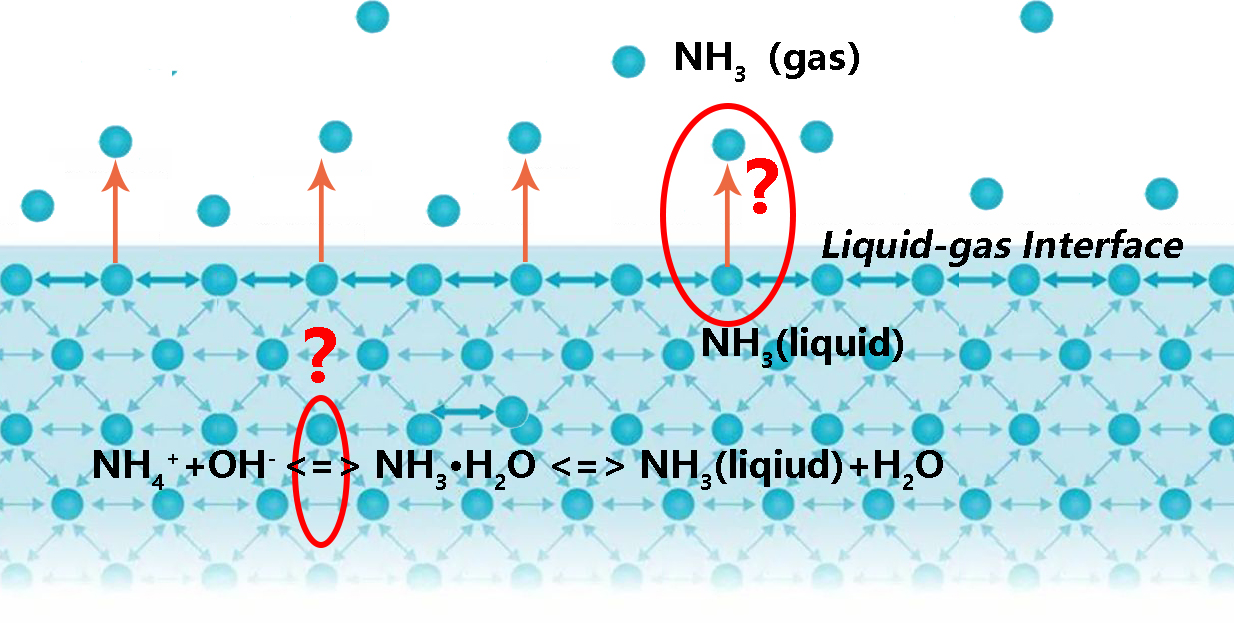
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
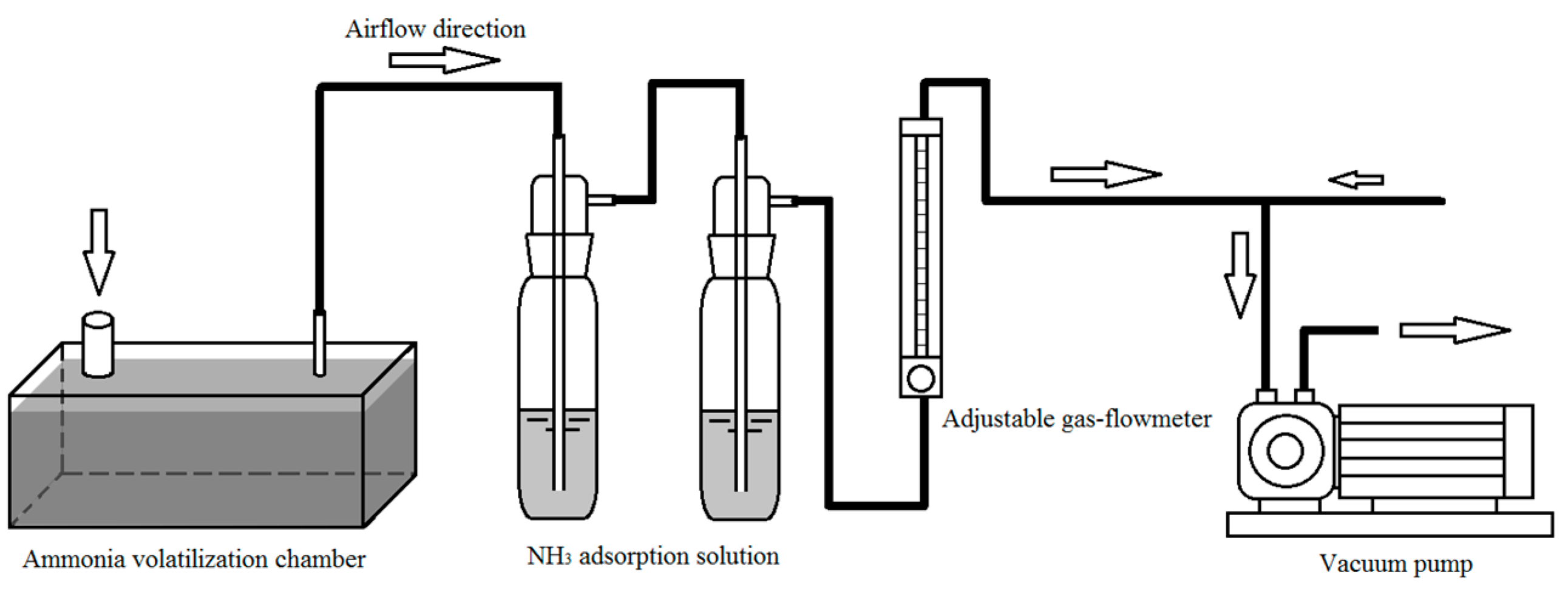
2.2. Experimental of Ammonia Volatilization
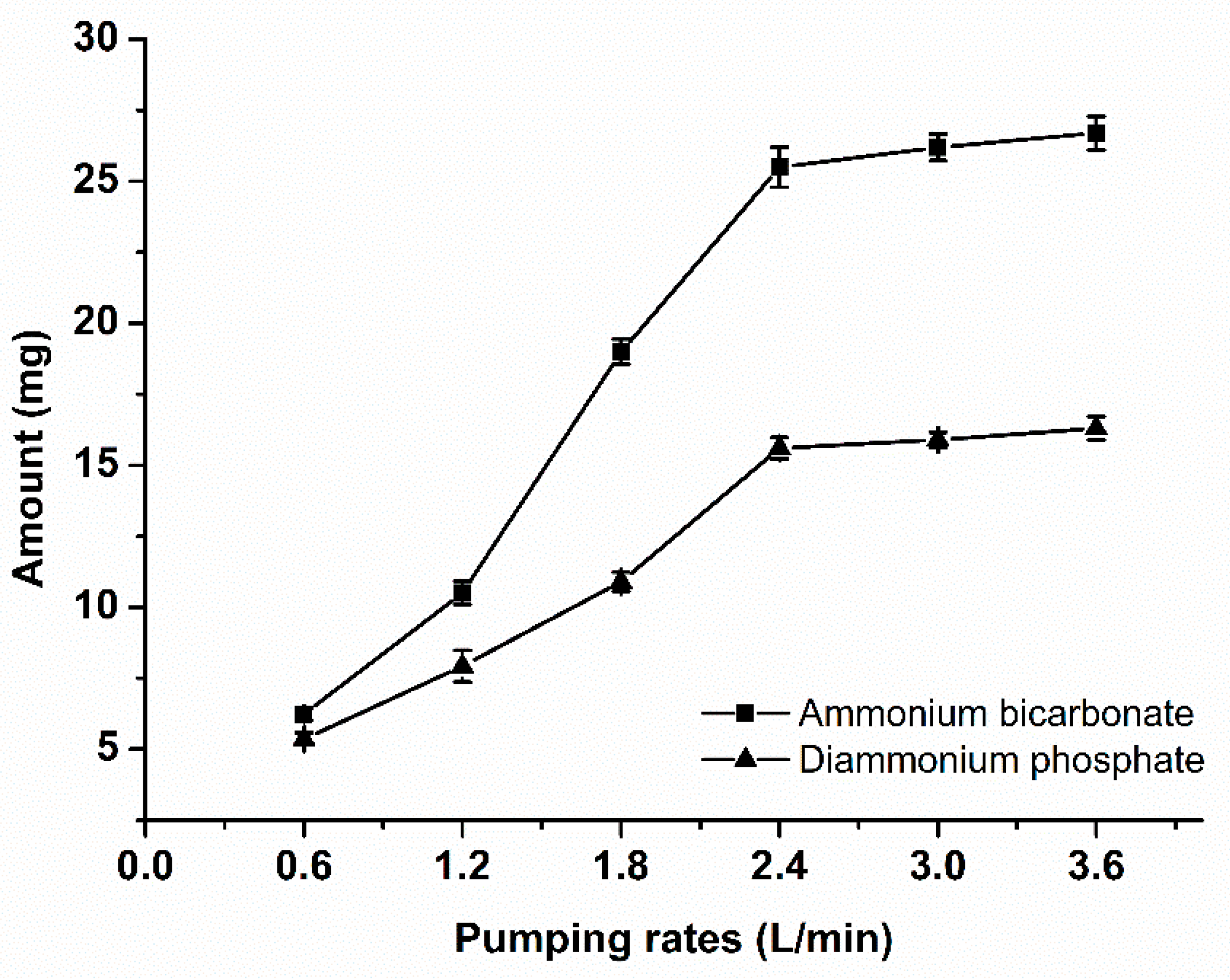
2.3. Experimental of Gas Flow Rate
2.5. Data Analysis and Processing
3. Results
3.1. Calculation of Gas Velocity in Gas-Liquid Interface
3.2. Kinetics Study on Ammonia Volatilization
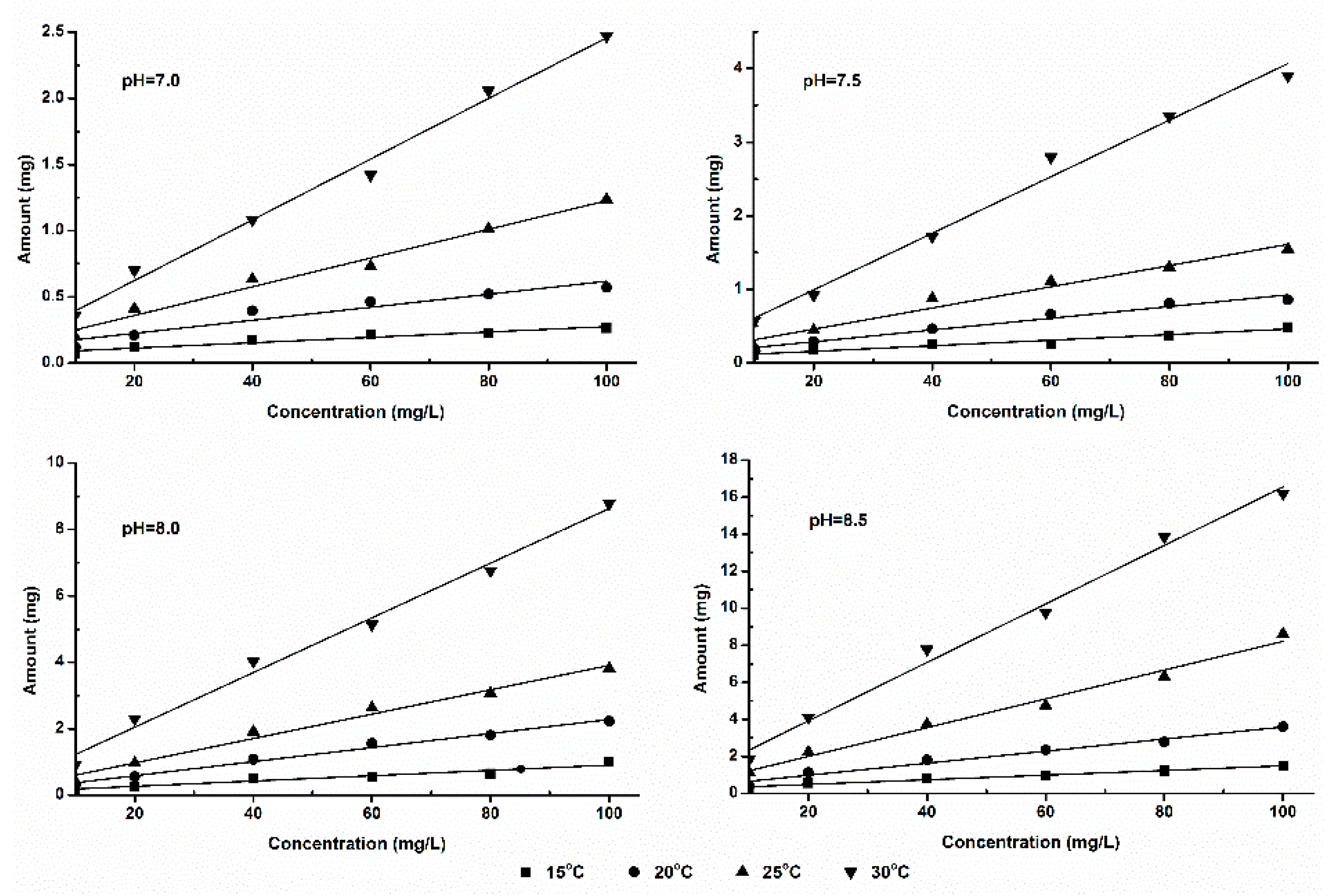
3.3. Equation Fitting of Ammonia Volatilization Fluxes
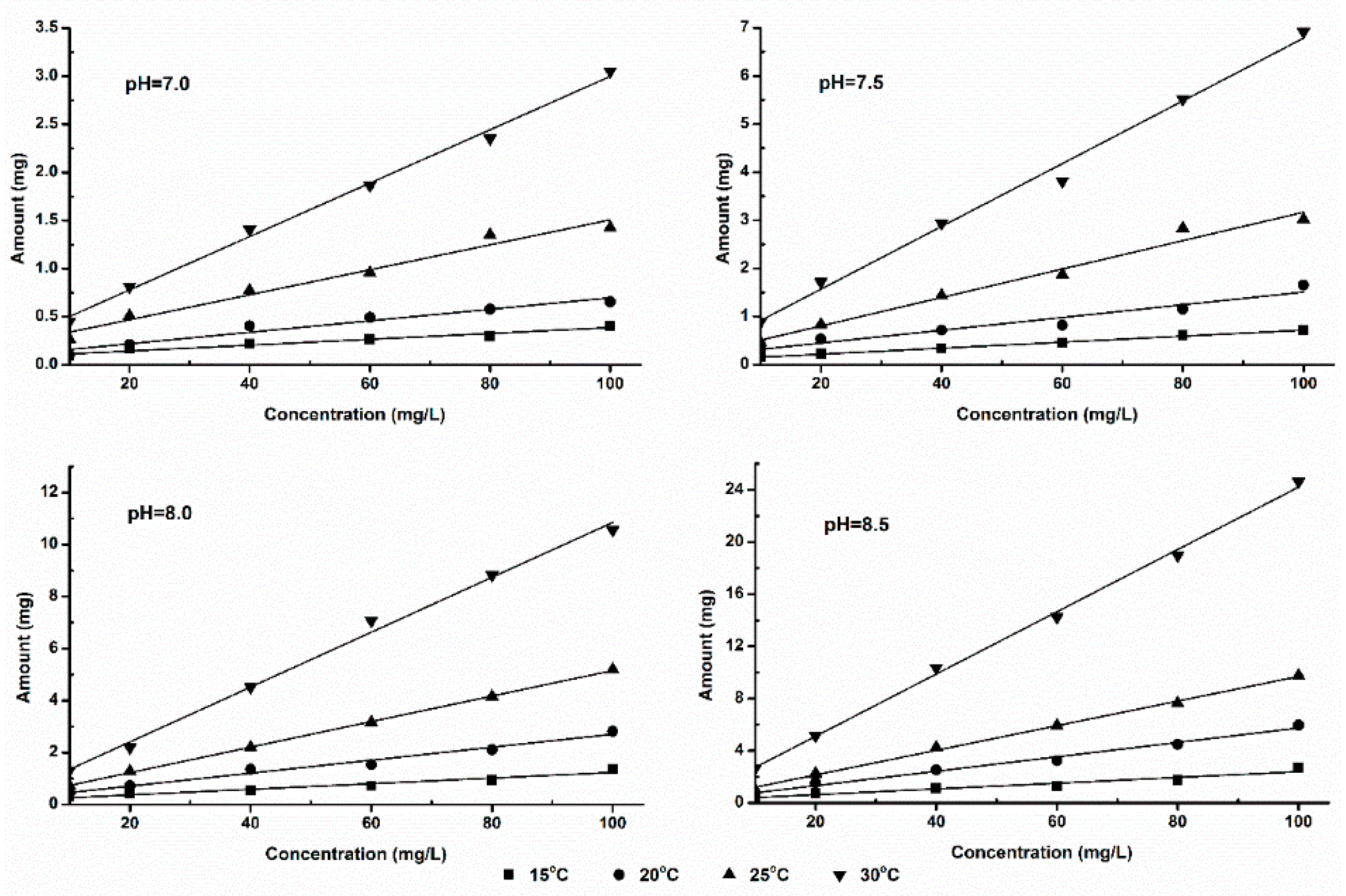
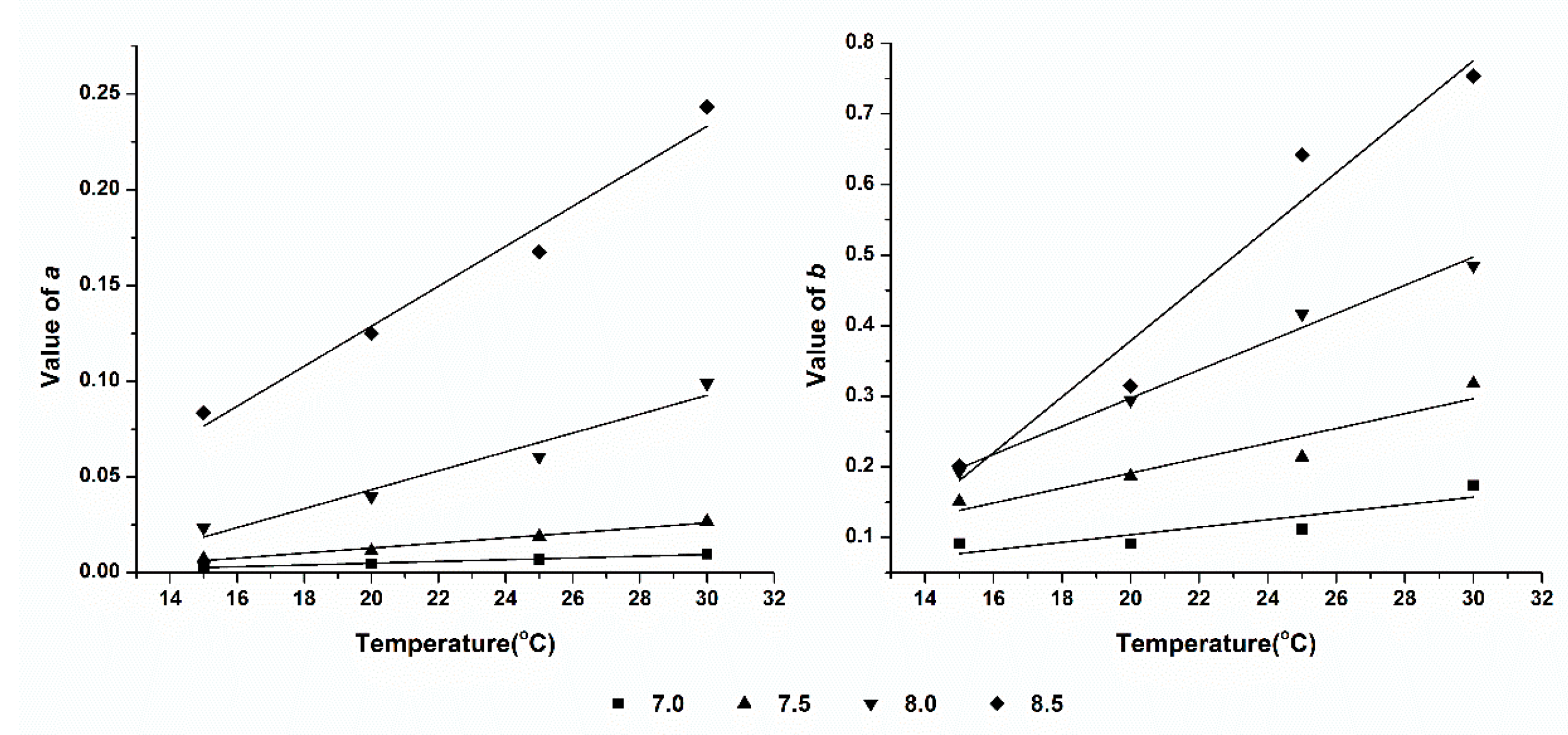
3.4. Effect of Temperature on Ammonia Volatilization
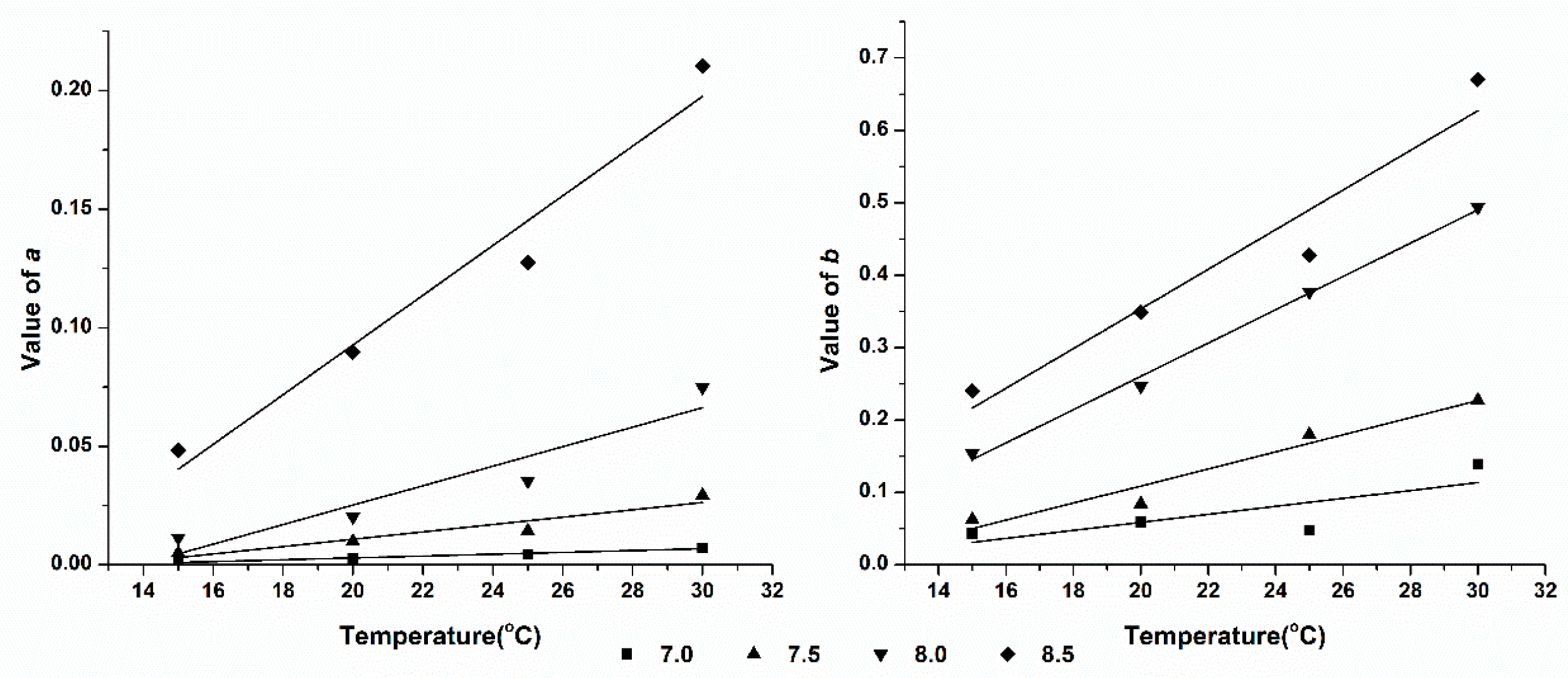
3.5. Effect of pH on Ammonia Volatilization
4. Discussion
4.1. Effect of Temperature and pH on Volatilization Process
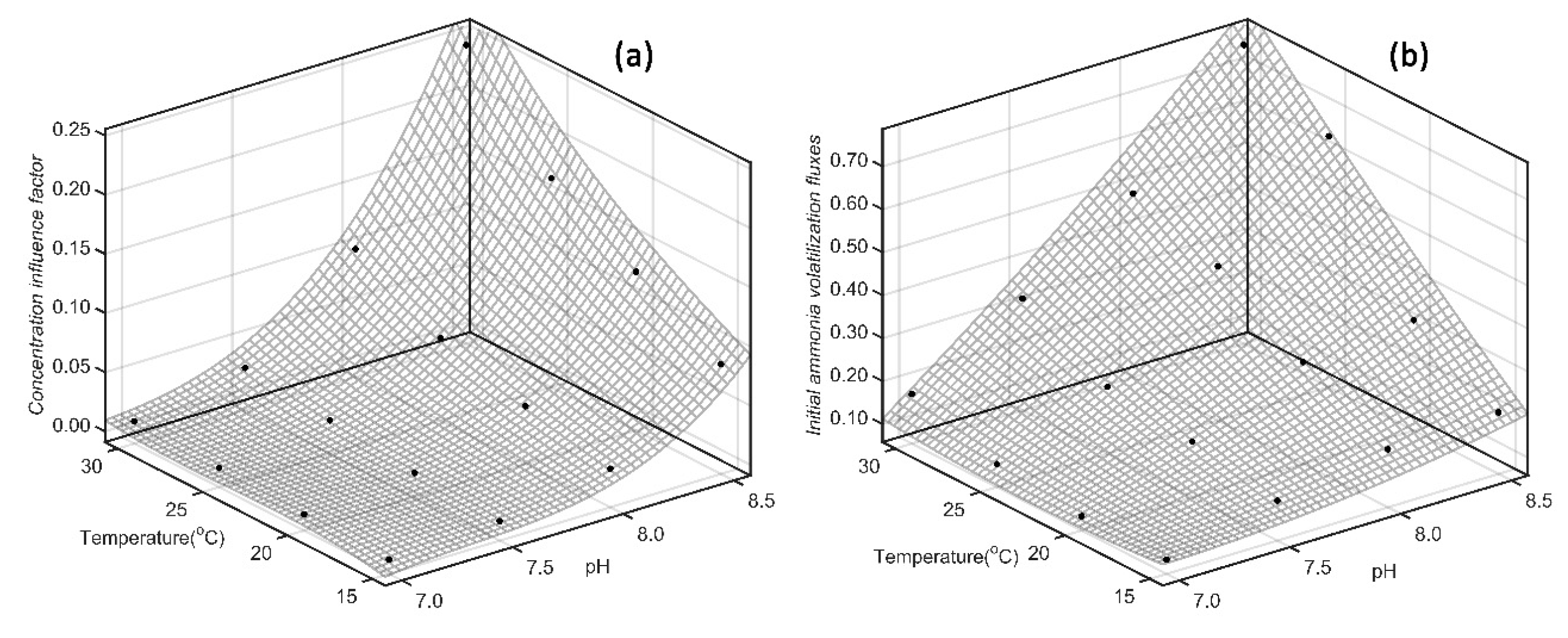
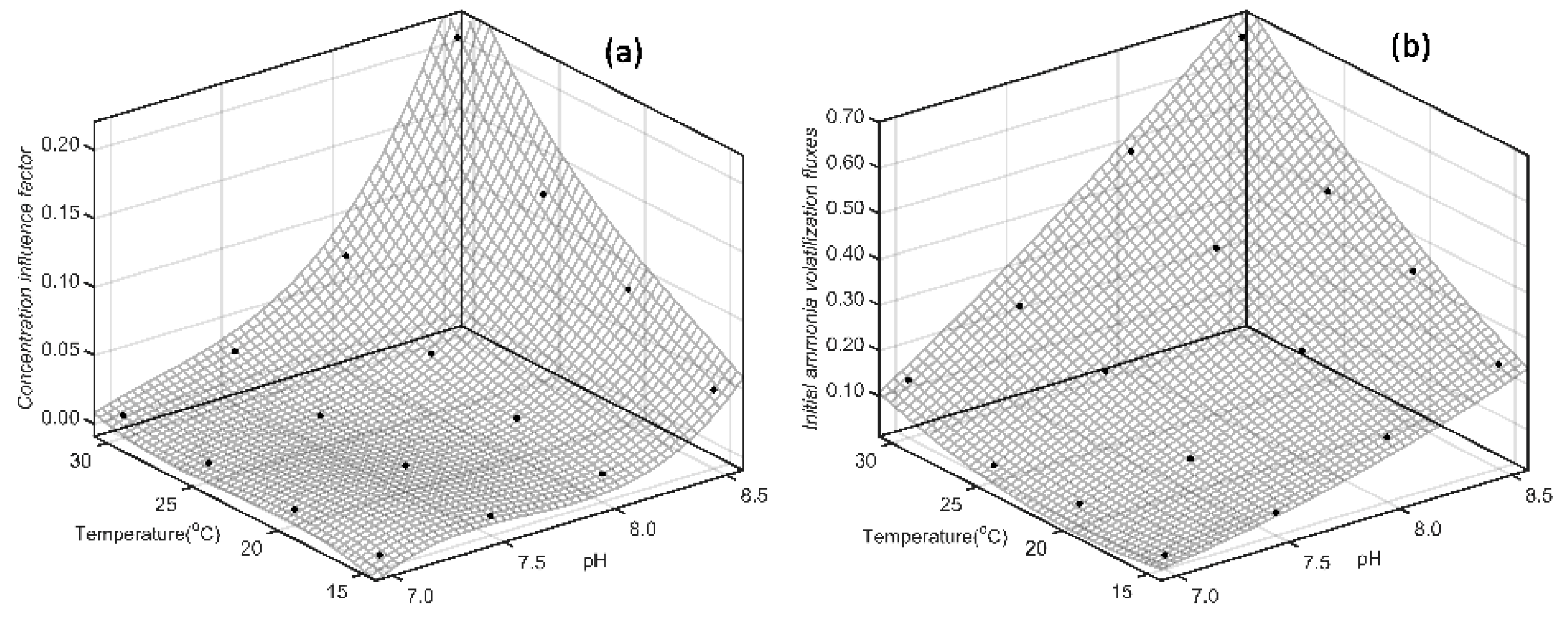
4.2. Synergistic Effect of Temperature and pH on Concentration Influence Factor and Initial Ammonia Volatilization Fluxes
4.3. Synergistic Effect of Ammonia Volatilization Rate Among Concentration, Temperature and pH
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhatnagar, A.; Hogland, W.; Marques, M.; Sillanpaa, M. An overview of the modification methods of activated carbon for its water treatment applications. Chem Eng J. 2013, 219, 499–511. [Google Scholar] [CrossRef]
- Cai, Z.; Wang, B.; Xu, M.; Zhang, H.; He, X.; Zhang, L.; Gao, S. Intensified soil acidification from chemical N fertilization and prevention by manure in an 18-year field experiment in the red soil of southern China. J. Soils Sediments. 2015, 15, 260–270. [Google Scholar] [CrossRef]
- Egyir, M.; Luyima, D.; Park, S. J.; Lee, K. S.; Oh, T. K. Volatilisations of ammonia from the soils amended with modified and nitrogen-enriched biochars. Sci. Total Environ. 2022, 835, 155453. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.; He, Y.; Chen, J.; Huang, Q.; Wang, H. Ammonia volatilization from a Chinese cabbage field under different nitrogen treatments in the Taihu Lake Basin, China. J Environ Sci China. 2015, 38, 14–23. [Google Scholar] [CrossRef]
- Tsai, C. C.; Chang, Y. F. Nitrogen availability in biochar-amended soils with excessive compost application. Agronomy. 2020, 10, 444. [Google Scholar] [CrossRef]
- Pan, B.; Lam, SK.; Mosier, A.; Luo, Y.; Chen, D. Ammonia volatilization from synthetic fertilizers and its mitigation strategies: A global synthesis. Agriculture, Ecosystems & Environment. 2016, 232, 283–289. [Google Scholar]
- Panday, D.; Mikha, M.M.; Collins, H.P.; Jin, V.L.; Kaiser, M.; Cooper, J.; Malakar, A.; Maharjan, B. Optimum rates of surface-applied coal char decreased soil ammonia volatilization loss. J. Environ. Qual. 2020, 49, 256–267. [Google Scholar] [CrossRef]
- Elias, E.; Okoth, P. F.; Stoorvogel, J. J.; Berecha, G.; Mellisse, B.T.; Mekuriaw, A.; Gebresamuel, G.; Selassie, Y.G.; Biratu, G.K.; Smaling, E.M.A. Cereal yields in Ethiopia relate to soil properties and N and P fertilizers. Nutr Cycl Agroecosystems. 2023, 126, 279–292. [Google Scholar] [CrossRef]
- Sha, Z.; Li, Q.; Lv, T.; Misselbrook, T.; Liu, X. Response of ammonia volatilization to biochar addition: a meta-analysis. Sci. Total Environ. 2019, 655, 1387–1396. [Google Scholar] [CrossRef]
- Tsai, C. C.; Chang, Y. F. Nitrogen availability in biochar-amended soils with excessive compost application. Agronomy. 2020, 10, 444. [Google Scholar] [CrossRef]
- Qi, S.; Ding, J.; Yang, S.; Jiang, Z.; Xu, Y. Impact of biochar application on ammonia volatilization from paddy fields under controlled irrigation. Sustainability. 2022, 14, 1337. [Google Scholar] [CrossRef]
- Zhang, Y.; Luan, S.; Chen, L.; Shao, M. Estimating the volatilization of ammonia from synthetic nitrogenous fertilizers used in China. Journal of Environmental Management. 2011, 92, 480–493. [Google Scholar] [CrossRef]
- Mandal, S.; Thangarajan, R.; Bolan, N.S.; Sarkar, B.; Khan, N.; Ok, Y.S.; Naidu, R. Biochar-induced concomitant decrease in ammonia volatilization and increase in nitrogen use efficiency by wheat. Chemosphere. 2016, 142, 120–127. [Google Scholar] [CrossRef]
- Ni, K.; Pacholski, A.; Kage, H. Ammonia volatilization after application of urea to winter wheat over 3 years affected by novel urease and nitrification inhibitors. Agriculture, Ecosystems & Environment 2014, 197, 184–194. [Google Scholar]
- Li, Q.; Yang, A.; Wang, Z.; Roelcke, M.; Chen, X.; Zhang, F.; Pasda, G.; Zerulla, W.; Wissemeier, A.H.; Liu, X. Effect of a new urease inhibitor on ammonia volatilization and nitrogen utilization in wheat in north and northwest China. Field Crops Research. 2015, 175, 96–105. [Google Scholar] [CrossRef]
- Sun, X.; Zhong, T.; Zhang, L.; Zhang, K.; Wu, W. Reducing ammonia volatilization from paddy field with rice straw derived biochar. Sci. Total Environ. 2019, 660, 512–518. [Google Scholar] [CrossRef]
- Liu, T. Q.; Fan, D. J.; Zhang, X. X.; Chen, J.; Li, C. F.; Cao, C. G. Deep placement of nitrogen fertilizers reduces ammonia volatilization and increases nitrogen utilization efficiency in no-tillage paddy fields in central China. Field Crops Research. 2015, 184, 80–90. [Google Scholar] [CrossRef]
- Xu, J.; Peng, S.; Yang, S.; Wang, W. Ammonia volatilization losses from a rice paddy with different irrigation and nitrogen managements. Agricultural Water Management. 2012, 104, 184–192. [Google Scholar] [CrossRef]
- Amaral, V.; Ortega, T.; Romera, C. C.; Forja, J. Linkages between greenhouse gases (CO2, CH4, and N2O) and dissolved organic matter composition in a shallow estuary. Sci. Total Environ. 2021, 788, 147863. [Google Scholar] [CrossRef]
- Cao, Y.; Tian, Y.; Yin, B.; Zhu, Z. Assessment of ammonia volatilization from paddy fields under crop management practices aimed to increase grain yield and N efficiency. Field Crops Research. 2013, 147, 23–31. [Google Scholar] [CrossRef]
- Moriyama, Y.; Sato, S. Effect of biochar application on suppression of ammonia volatilization from anaerobic digestion effluent mixed with soil as a nitrogen source. Wood Carbonization Res Soc. 2020, 17, 8–16. [Google Scholar]
- Zhang, Z.; Xu, Z.; Song, X.; Zhang, B.; Li, G.; Hu, N.; Luo, W. Membrane processes for resource recovery from anaerobically digested livestock manure effluent: opportunities and challenges. Curr Pollut Rep. 2020, 6, 123–136. [Google Scholar] [CrossRef]
- Bornø, M.L.; Müller-Stover, D.S.; Liu, F. Biochar modifies the content of primary metabolites in the rhizosphere of well-watered and drought-stressed Zea mays L. (maize). Biol. Fertil. Soils. 2022, 58, 633–647. [Google Scholar] [CrossRef]
- Ding, Z.; Zhou, Z.; Lin, X.; Zhao, F.; Wang, B.; Lin, F.; Ge, Y.; Eissa, M.A. Biochar impacts on NH3-volatilization kinetics and growth of sweet basil (Ocimum basilicum L.) under saline conditions. Ind. Crop. Prod 2020, 157, 112903. [Google Scholar] [CrossRef]
- Liu, K.; Ran, Q.; Li, F.; Shaheen, S.M.; Wang, H.; Rinklebe, J.; Liu, C.; Fang, L. Carbon-based strategy enables sustainable remediation of paddy soils in harmony with carbon neutrality. Carbon Res. 2022, 1, 12. [Google Scholar] [CrossRef]
- Tao, W.; Ukwuani, A. T. Coupling thermal stripping and acid absorption for ammonia recovery from dairy manure: Ammonia volatilization kinetics and effects of temperature, pH and dissolved solids content. Chemical Engineering Journal. 2015, 280, 188–196. [Google Scholar] [CrossRef]
- Maqbool, T.; Ly, Q.V.; Asif, M.B.; Ng, H.Y.; Zhang, Z. Fate and role of fluorescence moieties in extracellular polymeric substances during biological wastewater treatment: A review. Sci. Total Environ. 2020, 718, 137291. [Google Scholar] [CrossRef]
| pH |
Temperature (oC) |
SAB | pH |
Temperature (oC) |
SDP | ||||
| a | b | R2 | a | b | R2 | ||||
| 7.0 | 15 | 0.0030 | 0.0915 | 0.9214 | 7.0 | 15 | 0.0012 | 0.0433 | 0.8932 |
| 20 | 0.0046 | 0.0913 | 0.9664 | 20 | 0.0026 | 0.0591 | 0.9314 | ||
| 25 | 0.0070 | 0.1118 | 0.9727 | 25 | 0.0044 | 0.0477 | 0.9877 | ||
| 30 | 0.0098 | 0.1735 | 0.9513 | 30 | 0.0071 | 0.1393 | 0.9549 | ||
| 7.5 | 15 | 0.0074 | 0.1510 | 0.9651 | 7.5 | 15 | 0.0050 | 0.0623 | 0.9363 |
| 20 | 0.0114 | 0.1870 | 0.9718 | 20 | 0.0099 | 0.0843 | 0.9759 | ||
| 25 | 0.0190 | 0.2138 | 0.9779 | 25 | 0.0143 | 0.1801 | 0.9668 | ||
| 30 | 0.0268 | 0.3183 | 0.9839 | 30 | 0.0294 | 0.2273 | 0.9659 | ||
| 8.0 | 15 | 0.0235 | 0.1931 | 0.9456 | 8.0 | 15 | 0.0114 | 0.1539 | 0.9527 |
| 20 | 0.0397 | 0.2949 | 0.9650 | 20 | 0.0202 | 0.2471 | 0.9650 | ||
| 25 | 0.0606 | 0.4168 | 0.9789 | 25 | 0.0354 | 0.3770 | 0.9712 | ||
| 30 | 0.0990 | 0.4854 | 0.9869 | 30 | 0.0749 | 0.4938 | 0.9861 | ||
| 8.5 | 15 | 0.0836 | 0.2014 | 0.9537 | 8.5 | 15 | 0.0483 | 0.2405 | 0.9495 |
| 20 | 0.1251 | 0.3148 | 0.9602 | 20 | 0.0897 | 0.3491 | 0.9799 | ||
| 25 | 0.1675 | 0.6422 | 0.9863 | 25 | 0.1274 | 0.4277 | 0.9904 | ||
| 30 | 0.2434 | 0.7535 | 0.9897 | 30 | 0.2103 | 0.6705 | 0.9756 | ||
| Fertilizer | pH | Regression Equation | |||
| Value of a | R2 | Value of b | R2 | ||
| SAB | 7.0 | =0.0005T-0.0042 | 0.9792 | =0.0053T-0.0029 | 0.6763 |
| 7.5 | =0.0013T-0.0134 | 0.9716 | =0.0106T-0.0204 | 0.8502 | |
| 8.0 | =0.0049T-0.0555 | 0.9381 | =0.0200T-0.1020 | 0.9837 | |
| 8.5 | =0.0104T-0.0799 | 0.9625 | =0.0397T-0.4147 | 0.9326 | |
| SDP | 7.0 | =0.0004T-0.00492 | 0.9625 | =0.0055T-0.0521 | 0.4384 |
| 7.5 | =0.0016T-0.02025 | 0.8546 | =0.0118T-0.1273 | 0.9258 | |
| 8.0 | =0.0041T-0.05713 | 0.8409 | =0.0230T-0.1994 | 0.9940 | |
| 8.5 | =0.0105T-0.11673 | 0.9423 | =0.0274T-0.1940 | 0.9042 | |
| Fertilizer | Temperature(oC) | Regression Equation | |||
| Value of a | R2 | Value of b | R2 | ||
| SAB | 15 | =0.0557pH2-0.8114pH+2.9564 | 0.9624 | =0.0744pH-0.4170 | 0.8679 |
| 20 | =0.0785 pH2-1.1385pH+4.1306 | 0.9794 | =0.1557pH-0.9846 | 0.9099 | |
| 25 | =0.0950 pH2-1.3679pH+4.9289 | 0.9880 | =0.3589pH-2.4349 | 0.9626 | |
| 30 | =0.1274 pH2-1.8207pH+6.5112 | 0.9987 | =0.3814pH-2.5234 | 0.9668 | |
| SDP | 15 | =0.0332 pH2-0.4846pH+1.7695 | 0.9166 | =0.1366pH-0.9338 | 0.9126 |
| 20 | =0.0623 pH2-0.9111pH+3.331 | 0.9019 | =0.2066pH-1.4163 | 0.9091 | |
| 25 | =0.0822 pH2-1.195pH+4.3490 | 0.9437 | =0.2674pH-1.8144 | 0.9375 | |
| 30 | =0.1132 pH2-1.6235pH+5.8283 | 0.9733 | =0.3721pH-2.5007 | 0.9532 | |
| Fertilizers | Model types | Fitting equation | R2 |
| SAB | Binary | =-0.7915+ 0.09719pH + 0.004289T | 0.7755 |
| Binary quadratic | =5.779-1.436pH-0.05468T+0.08915pH2+0.006714pH·T+0.000154T2 | 0.9772 | |
| Binary cubic | =-20.01+7.513pH+0.3245T-0.9496pH2-0.07564pH·T-0.002611T2+0.0402pH3 +0.004628pH2·T + 0.000236pH·T2 +0.00001387T3 | 0.9980 | |
| SDP | Binary | =-0.6173+0.07323pH+0.004133T | 0.6681 |
| Binary quadratic | =4.966-1.2pH-0.05615T+0.07263pH2+0.006563pH·T+0.0002093T2 | 0.9447 | |
| Binary cubic | =-35.71+13.35pH+0.3913T-1.676pH2-0.08671pH·T-0.003744T2+ 0.07017pH3+0.005198pH2·T+0.0002822pH·T2+0.00002617T3 | 0.9946 |
| Fertilizers | Model types | Fitting equation | R2 |
| SAB | Binary | =-2.015+0.2426pH+0.01889T | 0.7999 |
| Binary quadratic | =3.80-0.7272pH-0.1661T+0.02993pH2+0.02249pH·T+0.0002378T2 | 0.9510 | |
| Binary cubic | =5.812-2.943pH+0.2989T+0.6076pH2-0.1751pH·T+0.0137T2-0.03883pH3+0.01445pH2·T-0.0005874pH·T2-0.000132T3 | 0.9608 | |
| SDP | Binary | =-2.047+0.2457pH+0.01693T | 0.8708 |
| Binary quadratic | =3.197-0.6862pH-0.1311T+0.03785pH2+0.01534pH·T+0.000647T2 | 0.9588 | |
| Binary cubic | =120.1-46.4pH-0.04929T+5.932pH2+0.02923pH·T-0.005468T2-0.2517pH3-0.001904pH2·T+0.0003472pH·T2 +0.00005073T3 | 0.9708 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




