Submitted:
11 October 2024
Posted:
15 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
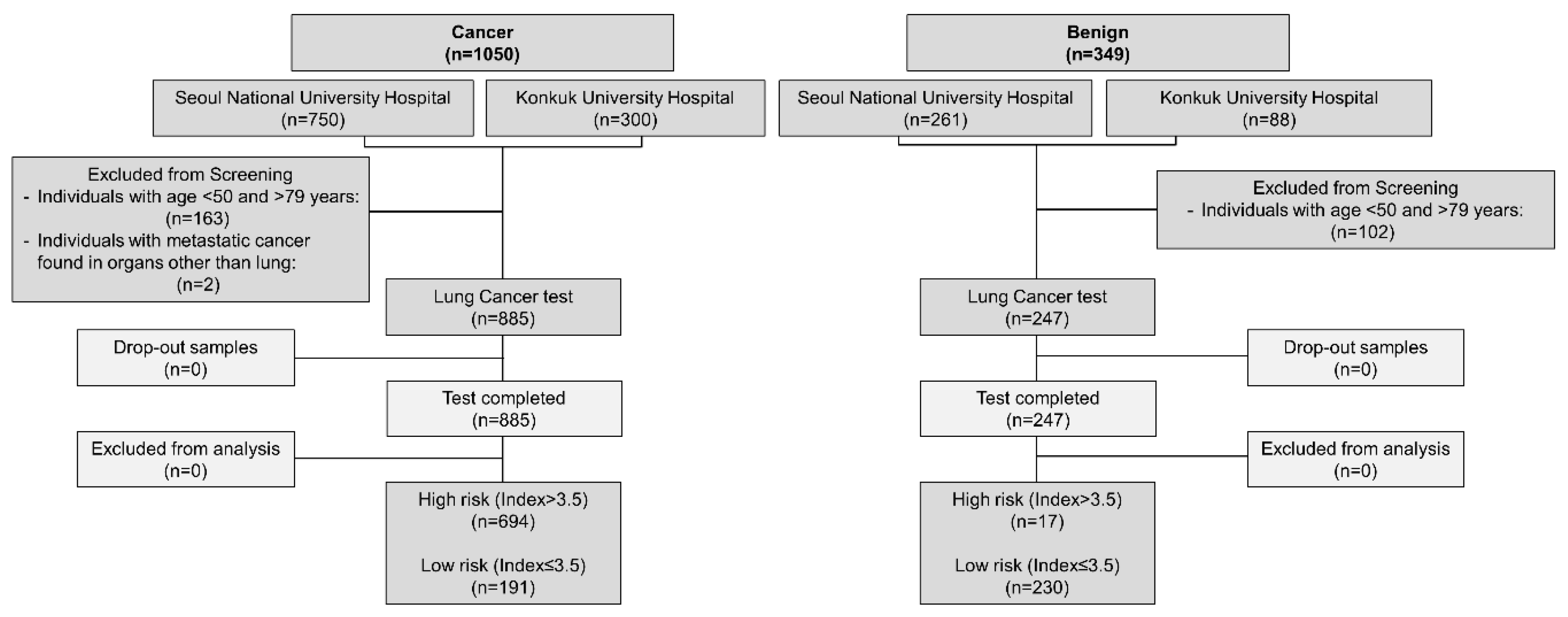
2.1. Study Design
2.2. Study Population
2.3. Laboratory Methods
2.3.1. Blood Sample Storage and Processing Method
2.3.2. Diagnostic Test
2.4. Statistical Analysis
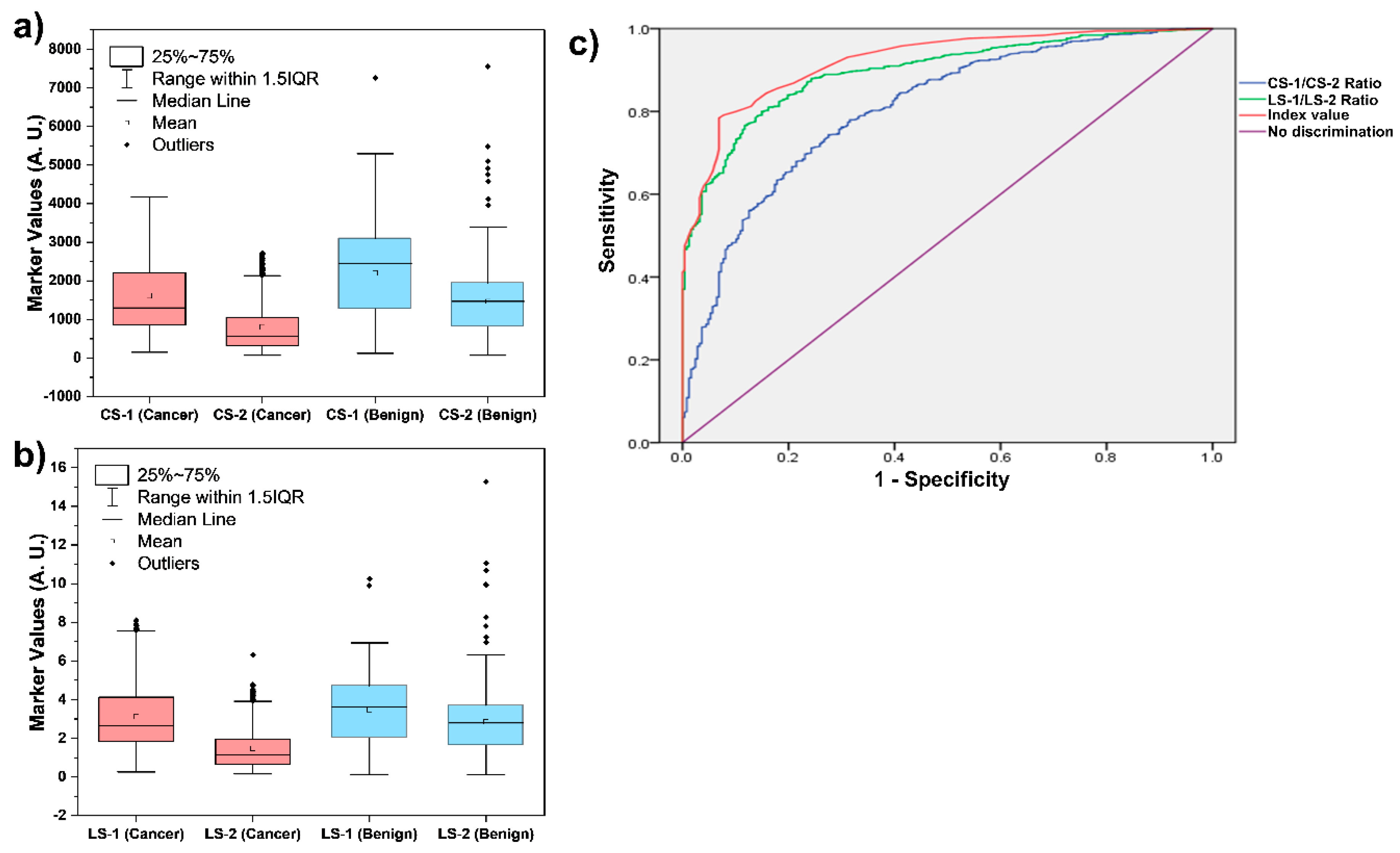
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA: A Cancer Journal for Clinicians 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA: A Cancer Journal for Clinicians 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA: A Cancer Journal for Clinicians 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Pastorino, U.; Silva, M.; Sestini, S.; Sabia, F.; Boeri, M.; Cantarutti, A.; Sverzellati, N.; Sozzi, G.; Corrao, G.; Marchianò, A. Prolonged lung cancer screening reduced 10-year mortality in the MILD trial: new confirmation of lung cancer screening efficacy. Annals of Oncology 2019, 30, 1162–1169. [Google Scholar] [CrossRef] [PubMed]
- Oudkerk, M.; Liu, S.; Heuvelmans, M.A.; Walter, J.E.; Field, J.K. Lung cancer LDCT screening and mortality reduction — evidence, pitfalls and future perspectives. Nature Reviews Clinical Oncology 2021, 18, 135–151. [Google Scholar] [CrossRef]
- Wang, X.; Liu, H.; Shen, Y.; Li, W.; Chen, Y.; Wang, H. Low-dose computed tomography (LDCT) versus other cancer screenings in early diagnosis of lung cancer: A meta-analysis. Medicine 2018, 97. [Google Scholar] [CrossRef]
- Siegel, R.L.; Giaquinto, A.N.; Jemal, A. Cancer statistics, 2024. CA: A Cancer Journal for Clinicians 2024, 74, 12–49. [Google Scholar] [CrossRef]
- de Koning, H.J.; van der Aalst, C.M.; de Jong, P.A.; Scholten, E.T.; Nackaerts, K.; Heuvelmans, M.A.; Lammers, J.J.; Weenink, C.; Yousaf-Khan, U.; Horeweg, N.; et al. Reduced Lung-Cancer Mortality with Volume CT Screening in a Randomized Trial. N Engl J Med 2020, 382, 503–513. [Google Scholar] [CrossRef]
- National Lung Screening Trial Research, T. Lung Cancer Incidence and Mortality with Extended Follow-up in the National Lung Screening Trial. J Thorac Oncol 2019, 14, 1732–1742. [Google Scholar] [CrossRef]
- National Lung Screening Trial Research, T.; Aberle, D.R.; Adams, A.M.; Berg, C.D.; Black, W.C.; Clapp, J.D.; Fagerstrom, R.M.; Gareen, I.F.; Gatsonis, C.; Marcus, P.M.; et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011, 365, 395–409. [Google Scholar] [CrossRef]
- Baptiste, J.V.; Jankowich, M.; Nici, L.L. Lung Cancer Screening with Low Dose CT: Two Year Experience at Providence Veteran Affairs Medical Center. In C30. LUNG CANCER SCREENING: WHO, WHY, WHERE, AND HOW MUCH; American Thoracic Society International Conference Abstracts; American Thoracic Society: 2017; pp. A5178–A5178.
- Hammer, M.M.; Byrne, S.C.; Kong, C.Y. Factors Influencing the False Positive Rate in CT Lung Cancer Screening. Acad Radiol 2022, 29 Suppl 2, S18–S22. [Google Scholar] [CrossRef]
- Hammer, M.M.; Byrne, S.C.; Kong, C.Y. Factors Influencing the False Positive Rate in CT Lung Cancer Screening. Academic Radiology 2022, 29, S18–S22. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Tan, F.; Xie, Y.; Tang, W.; Wang, F.; Xu, Y.; Cao, W.; Qin, C.; Dong, X.; Zheng, Y.; et al. A strategy to reduce the false-positive rate after low-dose computed tomography in lung cancer screening: A multicenter prospective cohort study. Cancer Med 2023, 12, 14781–14793. [Google Scholar] [CrossRef] [PubMed]
- Toumazis, I.; Erdogan, S.A.; Bastani, M.; Leung, A.; Plevritis, S.K. A Cost-Effectiveness Analysis of Lung Cancer Screening With Low-Dose Computed Tomography and a Diagnostic Biomarker. JNCI Cancer Spectr 2021, 5. [Google Scholar] [CrossRef] [PubMed]
- Boiselle, P.M. Computed tomography screening for lung cancer. JAMA 2013, 309, 1163–1170. [Google Scholar] [CrossRef]
- Oudkerk, M.; Devaraj, A.; Vliegenthart, R.; Henzler, T.; Prosch, H.; Heussel, C.P.; Bastarrika, G.; Sverzellati, N.; Mascalchi, M.; Delorme, S.; et al. European position statement on lung cancer screening. Lancet Oncol 2017, 18, e754–e766. [Google Scholar] [CrossRef]
- Song, K.-S.; Balasaheb Nimse, S.; Kim, J.; Kim, J.; Nguyen, V.-T.; Ta, V.-T.; Kim, T. 9G DNAChip: microarray based on the multiple interactions of 9 consecutive guanines. Chemical Communications 2011, 47, 7101–7103. [Google Scholar] [CrossRef]
- Choe, W.; Chae, J.D.; Lee, B.-H.; Kim, S.-H.; Park, S.Y.; Nimse, S.B.; Kim, J.; Warkad, S.D.; Song, K.-S.; Oh, A.-C.; et al. 9G TestTM Cancer/Lung: A Desirable Companion to LDCT for Lung Cancer Screening. Cancers 2020, 12. [Google Scholar] [CrossRef]
- Song, K.-S.; Nimse, S.B.; Warkad, S.D.; Oh, A.-C.; Kim, T.; Hong, Y.J. Quantification of CYFRA 21-1 and a CYFRA 21-1–anti-CYFRA 21-1 autoantibody immune complex for detection of early stage lung cancer. Chemical Communications 2019, 55, 10060–10063. [Google Scholar] [CrossRef]
- Tan, E.M.; Zhang, J. Autoantibodies to tumor-associated antigens: reporters from the immune system. Immunol Rev 2008, 222, 328–340. [Google Scholar] [CrossRef]
- Agarwal, S.; Saini, S.; Parashar, D.; Verma, A.; Sinha, A.; Jagadish, N.; Batra, A.; Suri, S.; Gupta, A.; Ansari, A.S.; et al. The novel cancer-testis antigen A-kinase anchor protein 4 (AKAP4) is a potential target for immunotherapy of ovarian serous carcinoma. Oncoimmunology 2013, 2, e24270. [Google Scholar] [CrossRef] [PubMed]
- Zaenker, P.; Gray, E.S.; Ziman, M.R. Autoantibody Production in Cancer--The Humoral Immune Response toward Autologous Antigens in Cancer Patients. Autoimmun Rev 2016, 15, 477–483. [Google Scholar] [CrossRef]
- Patel, A.J.; Tan, T.-M.; Richter, A.G.; Naidu, B.; Blackburn, J.M.; Middleton, G.W. A highly predictive autoantibody-based biomarker panel for prognosis in early-stage NSCLC with potential therapeutic implications. British Journal of Cancer 2022, 126, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Karjalainen, A.; Koskinen, H.; Hemminki, K.; Vainio, H.; Shnaidman, M.; Ying, Z.; Pukkala, E.; Brandt-Rauf, P.W. p53 autoantibodies predict subsequent development of cancer. Int J Cancer 2005, 114, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.J.; Oh, I.-J.; Kim, Y.; Jung, J.H.; Seok, M.; Lee, W.; Park, C.K.; Lim, J.-H.; Kim, Y.-C.; Kim, W.-S.; et al. Clinical Validation of a Protein Biomarker Panel for Non-Small Cell Lung Cancer. J Korean Med Sci 2018, 33. [Google Scholar] [CrossRef]
- Baldwin, D.R.; Callister, M.E.; Crosbie, P.A.; Dowd, E.L.; Rintoul, R.C.; Robbins, H.A.; Steele, R.J.C. Biomarkers in lung cancer screening: the importance of study design. European Respiratory Journal 2021, 57, 2004367. [Google Scholar] [CrossRef]
- Doseeva, V.; Colpitts, T.; Gao, G.; Woodcock, J.; Knezevic, V. Performance of a multiplexed dual analyte immunoassay for the early detection of non-small cell lung cancer. Journal of Translational Medicine 2015, 13, 55. [Google Scholar] [CrossRef] [PubMed]
- Hajian-Tilaki, K. Sample size estimation in diagnostic test studies of biomedical informatics. Journal of Biomedical Informatics 2014, 48, 193–204. [Google Scholar] [CrossRef]
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more "personalized" approach to cancer staging. CA Cancer J Clin 2017, 67, 93–99. [Google Scholar] [CrossRef]
- Korea Institute of Radiological and Medical Sciences, K.C.C.H. Assesment of the diagnostic accuracy of 9G testTM Cancer/Lung using reciever operating curves based on CLSI EP24-A2 guideline. Index values ≤3.5 and >3.5 of 9G testTM Cancer/Lung test allows to discriminate the patients between high-risk and low-risk of lung cancer, respectively. 2020.
- Kim, H.; Lee, J.K.; Kim, H.R.; Hong, Y.J. Enhanced Lung Cancer Detection Using a Combined Ratio of Antigen-Autoantibody Immune Complexes against CYFRA 21-1 and p53. Cancers (Basel) 2024, 16. [Google Scholar] [CrossRef]
- González Maldonado, S.; Motsch, E.; Trotter, A.; Kauczor, H.-U.; Heussel, C.-P.; Hermann, S.; Zeissig, S.R.; Delorme, S.; Kaaks, R. Overdiagnosis in lung cancer screening: Estimates from the German Lung Cancer Screening Intervention Trial. International Journal of Cancer 2021, 148, 1097–1105. [Google Scholar] [CrossRef]
- Patz, E.F., Jr.; Pinsky, P.; Gatsonis, C.; Sicks, J.D.; Kramer, B.S.; Tammemagi, M.C.; Chiles, C.; Black, W.C.; Aberle, D.R.; Team, N.O.M.W. Overdiagnosis in low-dose computed tomography screening for lung cancer. JAMA Intern Med 2014, 174, 269–274. [Google Scholar] [CrossRef]
- Oken, M.M.; Hocking, W.G.; Kvale, P.A.; Andriole, G.L.; Buys, S.S.; Church, T.R.; Crawford, E.D.; Fouad, M.N.; Isaacs, C.; Reding, D.J.; et al. Screening by chest radiograph and lung cancer mortality: the Prostate, Lung, Colorectal, and Ovarian (PLCO) randomized trial. JAMA 2011, 306, 1865–1873. [Google Scholar] [CrossRef] [PubMed]
- Marcus, P.M.; Bergstralh, E.J.; Zweig, M.H.; Harris, A.; Offord, K.P.; Fontana, R.S. Extended lung cancer incidence follow-up in the Mayo Lung Project and overdiagnosis. J Natl Cancer Inst 2006, 98, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Becker, N.; Motsch, E.; Trotter, A.; Heussel, C.P.; Dienemann, H.; Schnabel, P.A.; Kauczor, H.U.; Maldonado, S.G.; Miller, A.B.; Kaaks, R.; et al. Lung cancer mortality reduction by LDCT screening-Results from the randomized German LUSI trial. Int J Cancer 2020, 146, 1503–1513. [Google Scholar] [CrossRef] [PubMed]
- Henschke, C.I. Early lung cancer action project: overall design and findings from baseline screening. Cancer 2000, 89, 2474–2482. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.Z.; Kuo, P.L.; Huang, Y.L.; Tang, E.K.; Chen, C.S.; Wu, M.T.; Lin, Y.P. Differences in lung cancer characteristics and mortality rate between screened and non-screened cohorts. Sci Rep 2019, 9, 19386. [Google Scholar] [CrossRef]
- Henschke, C.I.; Yip, R.; Shaham, D.; Zulueta, J.J.; Aguayo, S.M.; Reeves, A.P.; Jirapatnakul, A.; Avila, R.; Moghanaki, D.; Yankelevitz, D.F.; et al. The Regimen of Computed Tomography Screening for Lung Cancer: Lessons Learned Over 25 Years From the International Early Lung Cancer Action Program. J Thorac Imaging 2021, 36, 6–23. [Google Scholar] [CrossRef]
- Sozzi, G.; Boeri, M. Potential biomarkers for lung cancer screening. Transl Lung Cancer Res 2014, 3, 139–148. [Google Scholar] [CrossRef]
- Seijo, L.M.; Peled, N.; Ajona, D.; Boeri, M.; Field, J.K.; Sozzi, G.; Pio, R.; Zulueta, J.J.; Spira, A.; Massion, P.P.; et al. Biomarkers in Lung Cancer Screening: Achievements, Promises, and Challenges. J Thorac Oncol 2019, 14, 343–357. [Google Scholar] [CrossRef]
- Bamji-Stocke, S.; van Berkel, V.; Miller, D.M.; Frieboes, H.B. A review of metabolism-associated biomarkers in lung cancer diagnosis and treatment. Metabolomics 2018, 14, 81. [Google Scholar] [CrossRef] [PubMed]
- Ostrin, E.J.; Bantis, L.E.; Wilson, D.O.; Patel, N.; Wang, R.; Kundnani, D.; Adams-Haduch, J.; Dennison, J.B.; Fahrmann, J.F.; Chiu, H.T.; et al. Contribution of a Blood-Based Protein Biomarker Panel to the Classification of Indeterminate Pulmonary Nodules. J Thorac Oncol 2021, 16, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, P.J.; Sears, C.R.; Arenberg, D.A.; Gaga, M.; Gould, M.K.; Massion, P.P.; Nair, V.S.; Powell, C.A.; Silvestri, G.A.; Vachani, A.; et al. Evaluating Molecular Biomarkers for the Early Detection of Lung Cancer: When Is a Biomarker Ready for Clinical Use? An Official American Thoracic Society Policy Statement. Am J Respir Crit Care Med 2017, 196, e15–e29. [Google Scholar] [CrossRef] [PubMed]
- Paez, R.; Kammer, M.N.; Tanner, N.T.; Shojaee, S.; Heideman, B.E.; Peikert, T.; Balbach, M.L.; Iams, W.T.; Ning, B.; Lenburg, M.E.; et al. Update on Biomarkers for the Stratification of Indeterminate Pulmonary Nodules. Chest 2023, 164, 1028–1041. [Google Scholar] [CrossRef]
| Parameter | Cancer (N = 885) |
Benign (N = 247) |
Overall (N = 1,132) |
|---|---|---|---|
| Gender, n (%) | |||
| a man | 566 (63.9) | 137 (55.5) | 703 (62.1) |
| a woman | 319 (36.1) | 110 (44.5) | 429 (37.9) |
| Age (years) | |||
| N | 885 | 247 | 1,132 |
| means | 67.0 ± 7.35 | 65.7 ± 8.31 | 66.7 ± 7.58 |
| median | 67.0 | 66.0 | 67.0 |
| min~max | 50.0 ~ 79.0 | 50.0 ~ 79.0 | 50.0 ~ 79.0 |
| Age category, n (%) | |||
| 50 ~ 59 | 153 (17.3) | 75 (30.4) | 228 (20.1) |
| 60 ~ 69 | 380 (42.9) | 72 (29.2) | 452 (39.9) |
| 70 ~ 79 | 352 (39.8) | 100 (40.5) | 452 (39.9) |
| Smoking history, n (%) | |||
| Smoker | 556 (62.8) | 125 (50.6) | 681 (60.2) |
| Never smoker | 329 (37.2) | 122 (49.4) | 451 (39.8) |
| Histological classification, n (%) | |||
| Non-Small Cell Lung Cancer (NSCLC) | 757 (85.5) | - | 757 (85.5) |
| Small Cell Lung Cancer (SCLC) | 128 (14.5) | - | 128 (14.5) |
| Stage of lung cancer - † for NSCLC, n (%) | |||
| Stage I | 408 (53.9) | - | 408 (53.9) |
| Stage II | 142 (18.8) | - | 142 (18.8) |
| Stage III | 146 (19.3) | - | 146 (19.3) |
| Stage IV | 61 (8.06) | - | 61 (8.06) |
| Stage of lung cancer - ‡ for SCLC, n (%) | |||
| Limited Disease (LD) | 57 (44.5) | - | 57 (44.5) |
| Extensive Disease (ED) | 71 (55.5) | - | 71 (55.5) |
| Parameter | High-risk of lung cancer (Index > 3.5) | Low-risk of lung cancer (Index ≤ 3.5) | Sensitivity (%) (95% CI) |
Specificity (%) (95% CI) |
||
|---|---|---|---|---|---|---|
|
Cancer (N = 885) |
694 (78.4%) | 191 (21.6%) | 78.4 (75.7, 81.1) |
93.1 (90.0, 96.3) |
||
|
Benign (N = 247) |
17 (6.98%) | 230 (93.1%) | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




