Submitted:
12 October 2024
Posted:
15 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Dung Beetle Colony
2.2. Field Cage
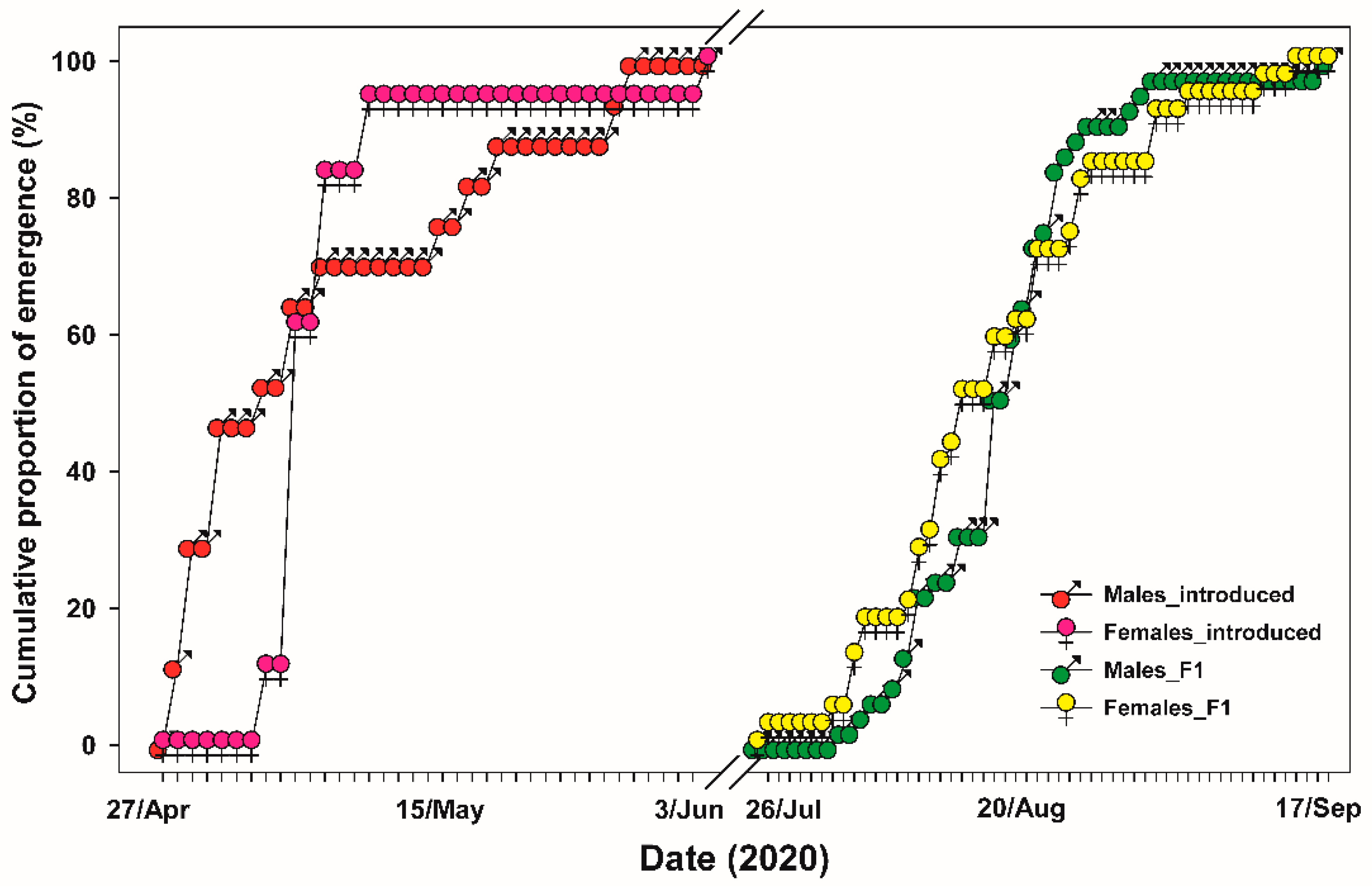
2.3. Overwintering and Spring Emergence
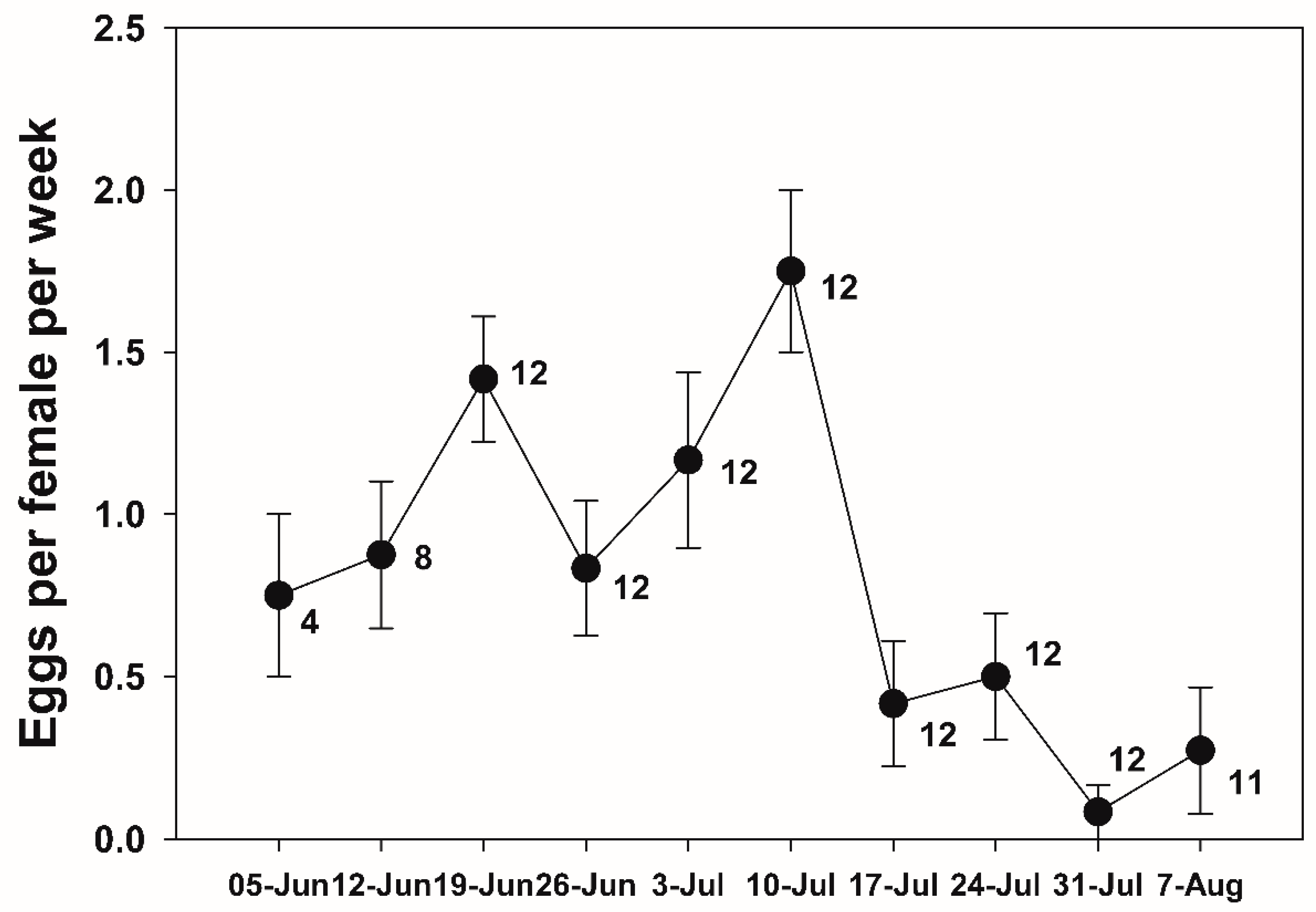
2.4. Survival and Reproduction Monitoring
2.5. Second Generation Emergence
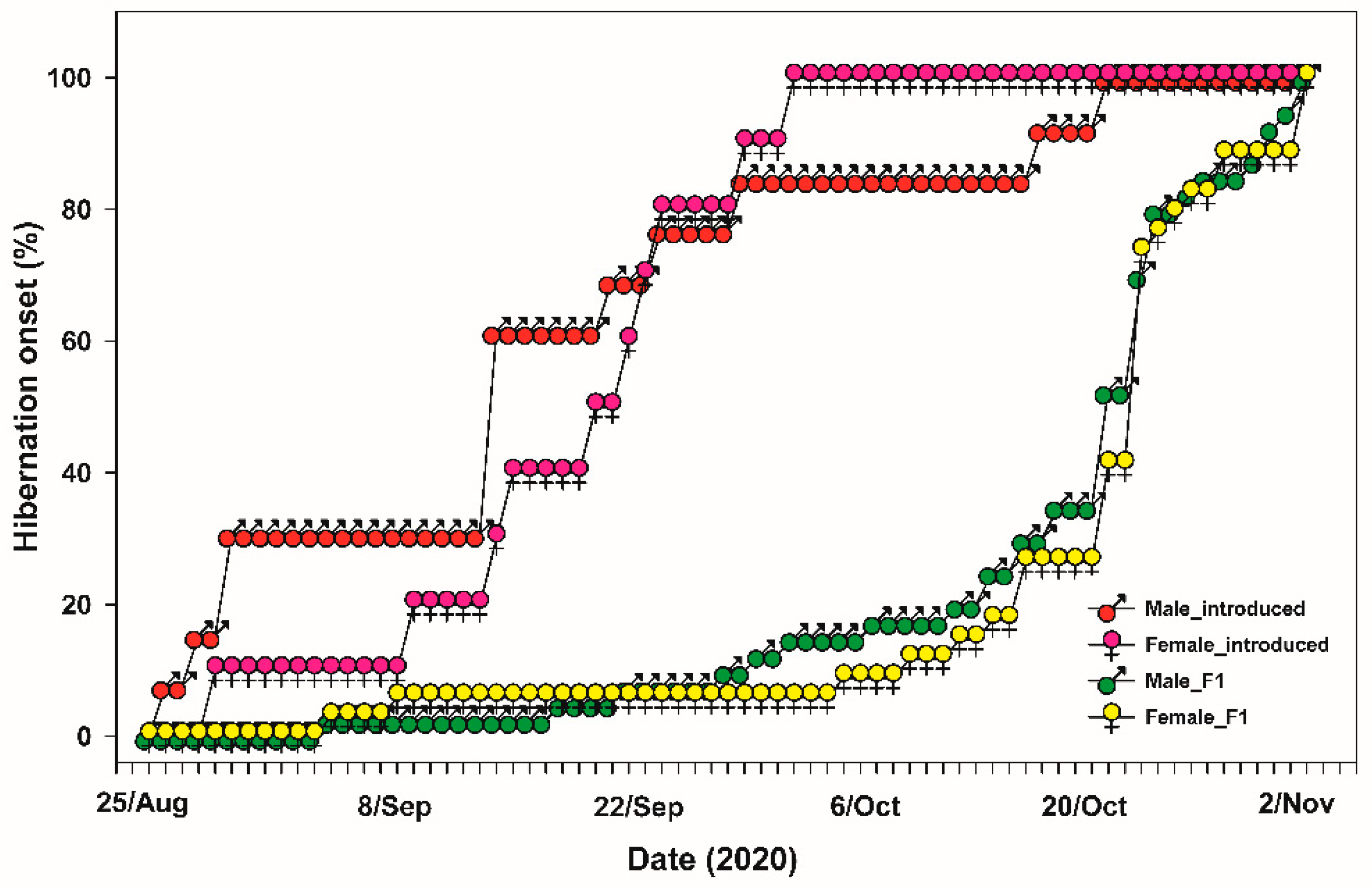
2.6. Hibernation Onset
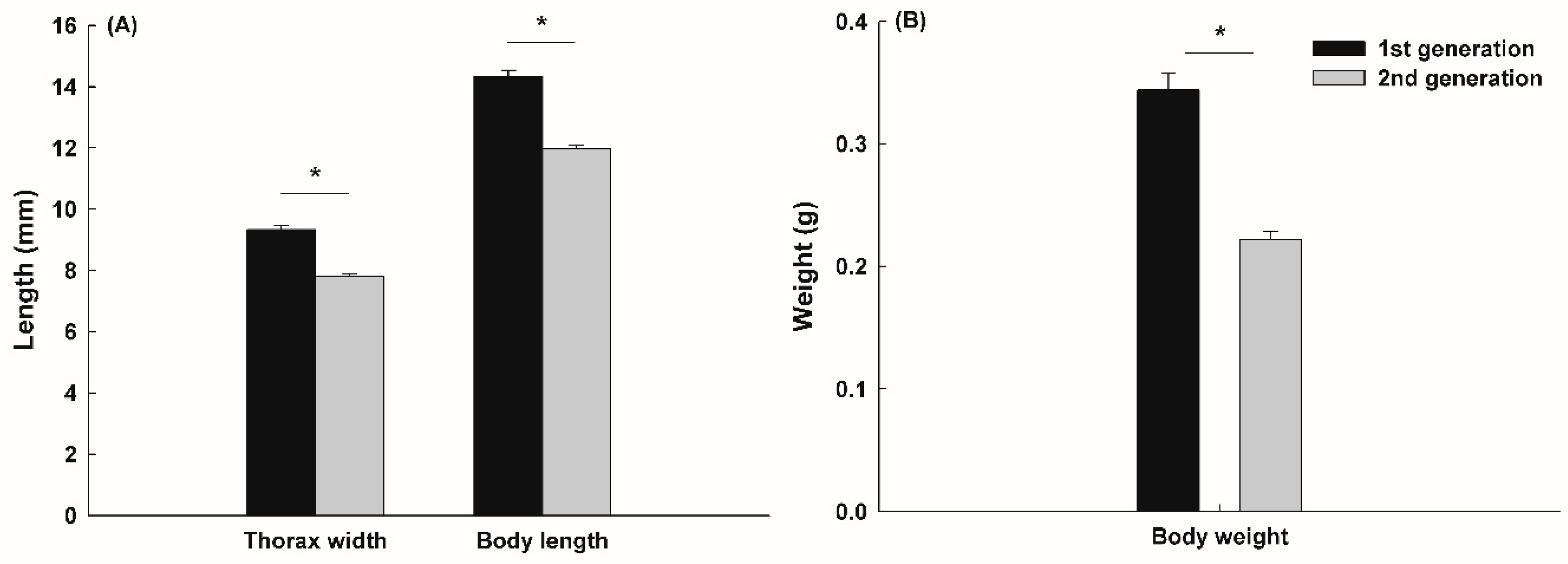
2.7. Measurement of Adult Beetle and Brood Dung Ball
2.8. Data Analysis
3. Results
3.1. Emergence and Hibernation Onset under Field Cage Condition
3.2. Growth and Morphological Traits on Field Cage
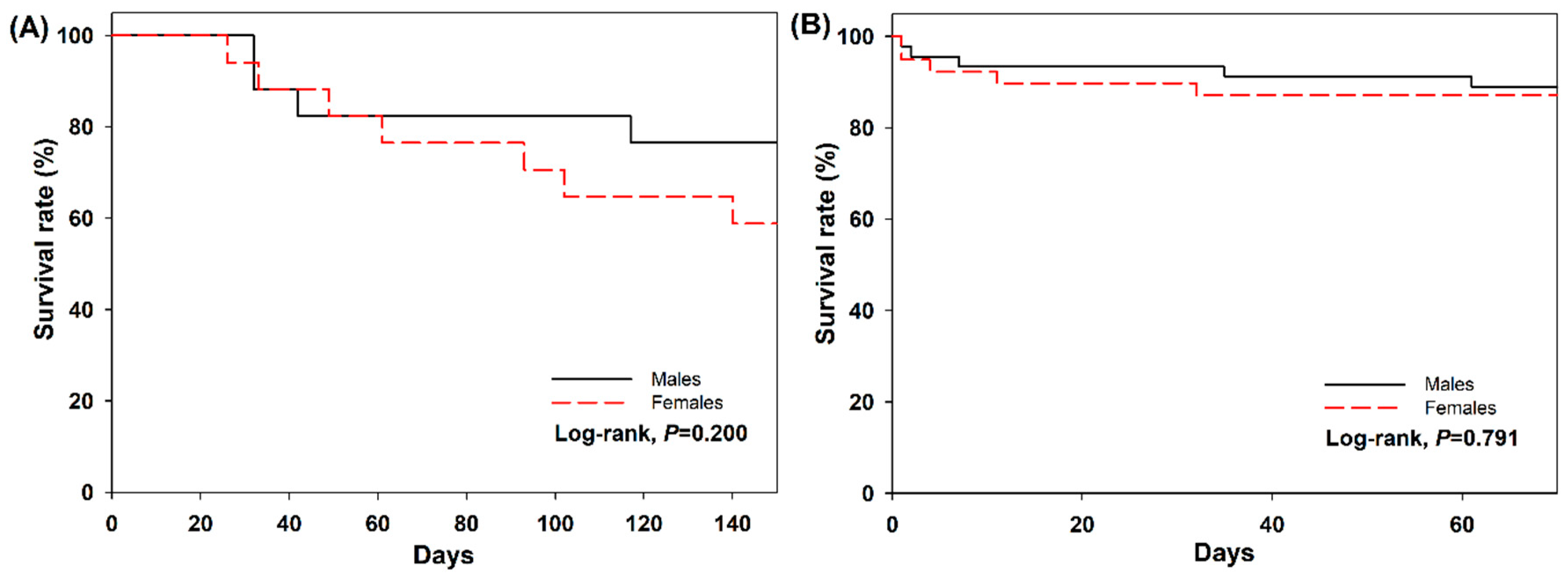
3.3. Fecundity and Survival on Field Cage
4. Discussion
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nichols, E.; Larsen, T.; Spector, S.; Davis, A.L.; Escobar, F.; Favila, M.; Vulinec, K. Global dung beetle response to tropical forest modification and fragmentation: A quantitative literature review and meta-analysis. Biological Conservation 2007, 137 (1),1-19.
- Anderson, D.E.; Berson, J. D.; Didham, R.K.; Simmons, L. W.; Evans, T.A. Dung beetles increase plant growth: a meta-analysis. Proceedings B 2024, 291, 20232885. [CrossRef]
- Andresen, E. Dung beetles in a Central Amazonian rainforest and their ecologicalrole as secondary seed dispersers. Ecological Entomology 2002, 27, 257–270.
- Bornemissza, G.F. The Australian dung beetle research unit in Pretoria. South Agrican Journal of Science 1979, 75 (6), 257-260.
- Slade, E.M.; Mann, D.J.; Villanueva, J.F.; Lewis, O.T. Experimental evidence for the effects of dung beetle functional group richness and composition on ecosystem function in a tropical forest. Journal of Animal Ecology 2007, 76 (6), 1094-1104. [CrossRef]
- Barragán, F; Moreno, C.E.; Escobar, F.; Halffter, G.; Navarrete, D. Negative impacts of human land use on dung beetle functional diversity. PLoS One 2011, 6, e17976. [CrossRef]
- Braga, R.F.; Korasaki, V.; Andresen, E.; Louzada, J. Dung beetle community and functions along a habitat-disturbance gradient in the Amazon: a rapid assessment of ecological functions associated to biodiversity. PLoS One 2013, 8 (2), e57786. [CrossRef]
- Salomao, R.P.; Alvarado, F.; Baena-Diaz, F.; Favila, M.E.; Iannuzzi, L.; Liberal, C.N., Santos, B.A.; Vaz-de-Mello, F.Z.; Gonzalez-Tokman, D. Urbanization effects on dung beetle assemblages in a tropical city. Ecological Indicators 2019, 103, 665-675. [CrossRef]
- Noriega, J.A.; March-Salas, M.; Castillo, S.; García-Q, H.; Hortal, J.; Santos, A.M. Human perturbations reduce dung beetle diversity and dung removal ecosystem function. Biotropica 2021, 53 (3), 753-766. [CrossRef]
- López-Bedoya, P.A.; Bohada-Murillo, M.; Ángel-Vallejo, M.C.; Audino, L.D.; Davis, A.L.; Gurr, G.; Noriega, J. A. Primary forest loss and degradation reduces biodiversity and ecosystem functioning: a global meta-analysis using dung beetles as an indicator taxon. Journal of Applied Ecology 2022, 59 (6), 1572-1585. [CrossRef]
- Wall, R.; Strong, L. Environmental consequences of treating cattle with the antiparasitic drug invermectin. Nature 1987, 327, 418-421.
- Lumaret, J.P.; Galante, E.; Lumbreras, C.; Mena, J.; Bertrand, M.; Bernal, J.L.; Cooper, J.F.; Kadiri, N.; Crowe, D. Field effects of invermectin residues on dung beetles. Journal of Applied Ecology 1993, 30, 428-436.
- Verdú, J.R.; Lobo, J.M.; Sánchez-Piñero, F.; Gallego, B.; Numa, C.; Lumaret, J.; Cortez, V.; Ortiz, A.J.; Tonelli, M.; Garcia-Teba, J.P.; Rey, A.; Rodriguez, A.; Duran, J. Ivermectin residues disrupt dung beetle diversity, soil properties and ecosystem functioning: An interdisciplinary field study. Science of the Total Environment 2018, 618, 219-228. [CrossRef]
- Davis, A.L.V.; Scholtz, C.H.; Dooley, P.W.; Bham, N.; Kryger, U. Scarabaine dung beetles as indicators of biodiversity, habitat transformation and pest control chemicals in agro-ecosystems. South Africal Journal of Science 2004, 100 (9), 415-424.
- Bang, H.S.; Lee, J.H.; Na, Y E.; Wall, R. Reproduction of the dung beetle (Copris tripartitus) in the dung of cattle treated with cis-cypermethrin and chlorpyrifos. Applied Soil Ecology 2007, 35(3), 546-552. [CrossRef]
- Sands, B., Wall, R. Sustained parasiticide use in cattle farming affects dung beetle functional assemblages. Agriculture, Ecosystems & Environment 2018, 265, 226-235. [CrossRef]
- Dortel, E.; Thuiller, W.; Lobo, J.M.; Bohbot, H.; Lumaret, J.P.; Jay-Robert, P. Potential effects of climate change on the distribution of Scarabaeidae dung beetles in Western Europe. Journal of Insect Conservation 2013, 17, 1059-1070. [CrossRef]
- Audino, L.D; Louzada, J.; Comita, L. Dung beetles as indicators of tropical forest restoration success: Is it possible to recover species and functional diversity? Biological conservation 2014, 169, 248-257.
- Slade, E.M.; Roslin, T. Dung beetle species interactions and multifunctionality are affected by an experimentally warmed climate. Oikos 2016, 125 (11), 1607-1616. [CrossRef]
- Holley, J.M.; Andrew, N.R. Warming effects on dung beetle ecosystem services: brood production and dung burial by a tunnelling dung beetle, Onthophagus taurus (Coleoptera: Scarabaeidae), is reduced by experimental warming. Austral Entomology 2020, 59 (2), 353-367. [CrossRef]
- Heddle, T.C.; Nash, M.; Henry, K. Indigenous and introduced dung beetles (Coleoptera: scarabaeidae) of temperate Australia: A review of biology, importance and effect of climate change on population distributions. General and Applied Entomology: The Journal of the Entomological Society of New South Wales 2021, 49, 1-11.
- Maldaner, M.E.; Sobral-Souza, T.; Prasniewski, V.M.; Vaz-de-Mello, F.Z. Effects of climate change on the distribution of key native dung beetles in South American grasslands. Agronomy 2021, 11(10), 2033. [CrossRef]
- Fatah, A.M.A.; Zalat, S.; Elbanna, S.M.; Al-Khalaf, A.A.; Nasser, M. Climate Change and the Dung Beetle: Evaluation of Global Warming Impact on the Distribution of Phyllognathus excavatus (Forster, 1771) through the Mediterranean Region. Applied Sciences 2023, 13 (22), 12107.
- Barbero, E.; Palestrini, C.; Rolando, A. Dung beetle conservation: effects of habitat and resource selection (Coleoptera: Scarabaeoidea). Journal of Insect Conservation 1999, 3, 75-84. [CrossRef]
- Koch, S.O.; Chown, S.L.; Davis, A.L.V.; Endrody-Younga, S.; Jaarsveld, A.S. Conservation strategies for poorly surveyed taxa: a dung beetle (Coleoptera, Scarabaeidae) case study from southern Africa. Journal of Insect Conservation 2000, 4, 45-56. [CrossRef]
- Bayartogtokh, B.; Kim, J.I.; Bae, Y.J. Lamellicorn beetles (Coleoptera: Scarabaeoidea) in Korea and Mongolia. Entomological Research 2012, 42, 211-218. [CrossRef]
- Kang, J.H.; Lim, C.S.; Park, S.H.; Seok, S.W.; Yoon, T.J.; Bayartogtokh, B.; Bae, Y.J. Historical domestication-driven population expansion of the dung beetle Gymnopleurus mopsus (Coleoptera: Scarabaeidae) from its last refuge in Mongolia. Scientific Reports 2018, 8, 3963. [CrossRef]
- Kim, M.; Choi, Y.J.; Koh, M.H.; Jang, K.H.; Kim, Y.J. Hibernation Durations Affect Life-history Traits of Gymnopleurus mopsus (Coleoptera: Scarabaeidae), an Endangered Dung Beetle. Proceedings of the national institute of ecology of the Republic of Korea. Proceedings of the National Institute of Ecology 2021, 2 (4), 279-284.
- Ministry of Environment. Red Data Book of Republic of Korea. National Institute of Biological Resources: Incheon, S. Korea, 2023, 8: Insects Ⅱ.
- Yoon, T.J.; Park, H.C.; Kang, J.H.; Bayartogtokh, B.; Bae, Y.J. Genetic divergence between the South Korean and Mongolian populations of the dung beetle, Gymnopleurus mopsus (Coleoptera: Scarabaeidae) based on mitochondrial cytochrome c oxidase subunit I (COI) gene sequences. Entomological Research 2017, 47(6), 366-372.
- Lim, C.S.; Baek, H.M.; Park, S.H.; Purevdorj, J.; Hong, M.H.; Han, K.D.; Bae, Y.J. Morphological characteristics of Gymnopleurus mopsus (Coleoptera: Scarabaeidae) with a new sex discrimination method. Entomological Research Bulletin 2015, 31 (2), 126-129.
- Halffter, G.; Halffter, V.; Favila, M.E. Food relocation and nesting behavior In Scarabaeus and Kheper (Coleoptera: Scarabaeinae). Acta Zoologica Mexicana, 2011, 27 (2), 305-324. [CrossRef]
- Pokhrel, M.R.; Cairns, S.C.; Hemmings, Z.; Floate, K.D.; Andrew, N.R.A. review of dung beetle introductions in the antipodes and north America: Status, opportunities, and challenges. Environmental Entomology 2021, 50(4), 762-780. [CrossRef]
- Edwards, P. Introduced dung beetles in Australia 1967-2003: current status and future directions. Dung beetle for Landcare Farming Committee: Brisbane, Australia, 2007, pp. 55.
- CSIRO (Commonwealth Scientific and Industrial Research Organisation). Information supporting the application to amend the list of specimens suitable for live import to include three new species of dung beetles (Coleoptera: Scarabaeidae) as suitable for import and for release Terms of Reference Biocoltrol: Euonthophagus crocatus, Onthophagus andalusicus and Gymnopleurus sturmi. CSIRO Health and Biosecurity: Canberra, Austalia, 2018, pp. 57.
- Bezanson, G.A.; Floate, K.D. An updated checklist of the Coleoptera associated with livestock dung on pastures in America North of Mexico. The Coleopterists Bulletin 2019, 73, 655–683. [CrossRef]
- Knapp, M.; Nedved, O. Gender and timing during ontogeny Matter: Effects of a temporary high temperature on survival, body size and colouration in Harmonia axyridis. Plos One 2013, e74948. [CrossRef]
- Carter, A.W.; Shedon, K.S. Life stages differ in plasticity to temperature fluctuations and uniquely contribute to adult phenotype in Onthophagus taurus dung beetles. Journal of Experimental Biology 2020, 223 (20), jeb227884.
- Callier, V.; Nijhout, H.F. Control of body size by oxygen supply reveals size-dependant and size-independent mechanisms of molting and metamorphosis. Proceedings of the National Academy of Sciences 2011, 108, 35. [CrossRef]
- Matalin, A.V, Evolution of biennial life cycles in ground beetles (Coleoptera, Carabidae) of the western palaearctic. In: Back to the roots and back to the future. Proceedings of XIII European Carabidologist meeting, Blagoevgrad 2008, 259-284.
- Osberg, D.C.; Doube, B.M.; Hanrahan, S.A. Habitat specificity in African dung beetles: the effect of soil type on dung burial by two species of ball-rolling dung beetles (Coleoptera Scarabaeidae). Tropical Zoology 1993, 6, 243-251. [CrossRef]
| Females | Males | t-value | Degrees of freedom | P-value* | |
| Thorax width (introduced, mm) | 9.21 ± 0.17 (17) | 9.43 ± 0.22 (17) | 0.79 | 32 | 0.438 |
| Body length (introduced, mm) | 14.26 ± 0.23 (17) | 14.41 ± 0.33 (17) | 0.38 | 32 | 0.706 |
| Body weight (introduced, g) | 0.33 ± 0.02 (17) | 0.35 ± 0.02 (17) | 0.54 | 32 | 0.592 |
| Thorax width (F1, mm) | 7.86 ± 0.12 (36) | 7.77 ± 0.11 (44) | -0.57 | 78 | 0.569 |
| Body length (F1, mm) | 12.15 ± 0.19 (36) | 11.84 ± 0.15 (44) | -1.31 | 78 | 0.195 |
| Body weight (F1, mm) | 0.22 ± 0.01 (36) | 0.22 ± 0.01 (44) | -0.30 | 78 | 0.767 |
| Brood ball length (mm) | 19.43 ± 0.31 (36) | 20.00 ± 0.29 (44) | 1.33 | 78 | 0.187 |
| Brood ball width (mm) | 17.15 ± 0.24 (36) | 17.30 ± 0.23 (44) | 0.45 | 78 | 0.656 |
| Brood ball weight (g) | 3.25 ± 0.12 (36) | 3.49 ± 0.12 (44) | 1.37 | 78 | 0.176 |
| Larval dev. time (days) | 45.67 ± 1.06 (36) | 45.25 ± 0.75 (44) | -0.33 | 78 | 0.743 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




