Submitted:
11 October 2024
Posted:
11 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
Telomeres and Telomerase Targeting in Cancer
2. Materials and Methods
Cell Culture
Inhibitors
Cell Treatment
- TMPyP4 (5µM, 10µM or 20µM)
- BIBR 1532 (1µM, 5µM or 10µM)
- Imetelstat (1µM,5µM or 10µM)
- Mismatch negative control for Imetestat (1µM, 5µM or 10µM).
Data Assessment
3. Results
Effect of Telomerase Inhibitors on the Cell-to-Cell Interactions and Spheroid Formation Abilities
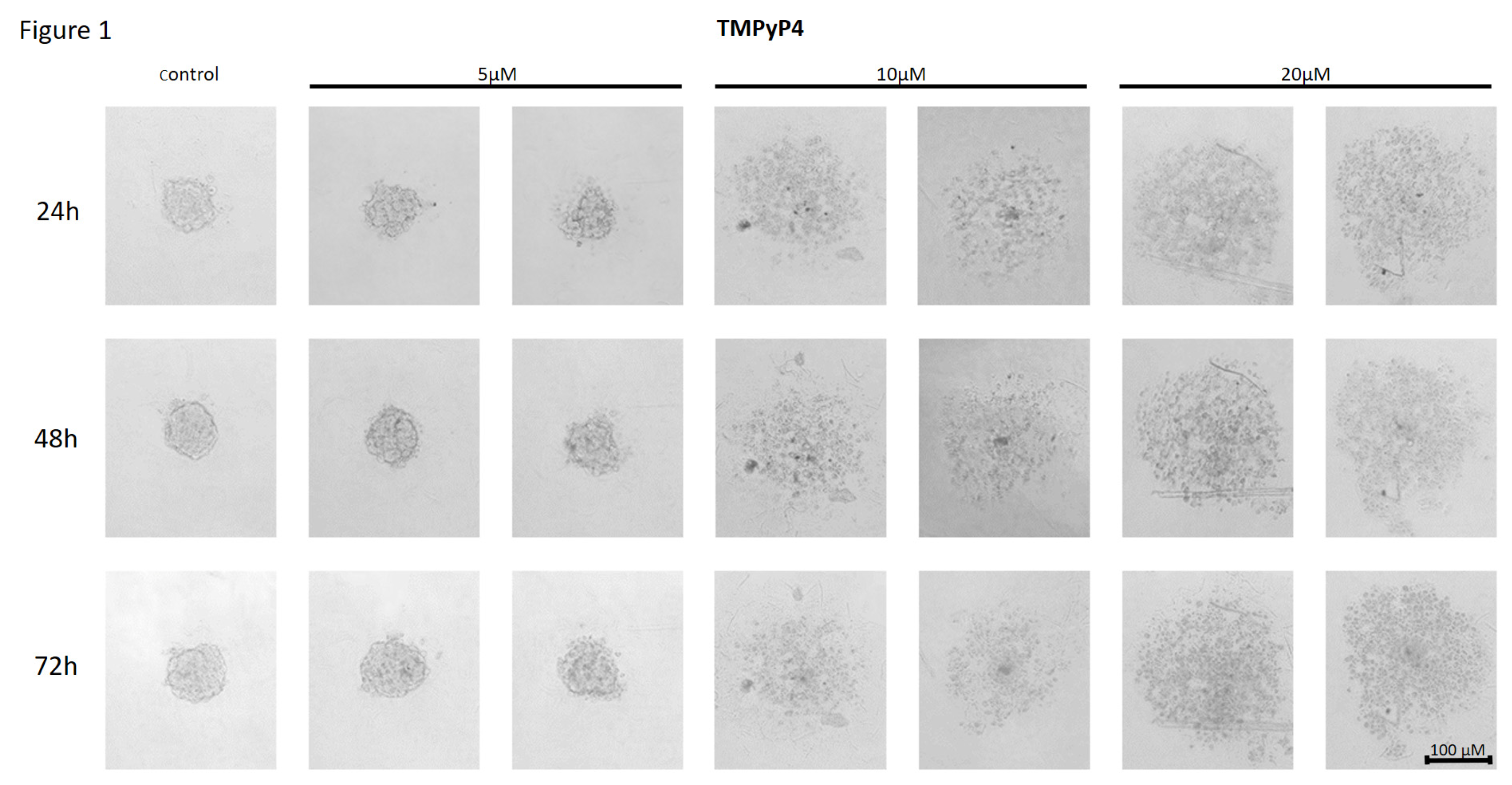
Effect of TMPyP4 on the Cell-to-Cell Interactions and Spheroid Formation in MCF7 Cells
Effect of TMPyP4 on the Cell-to-Cell Interactions and Spheroid Formation in MDA-MB-231 Cells
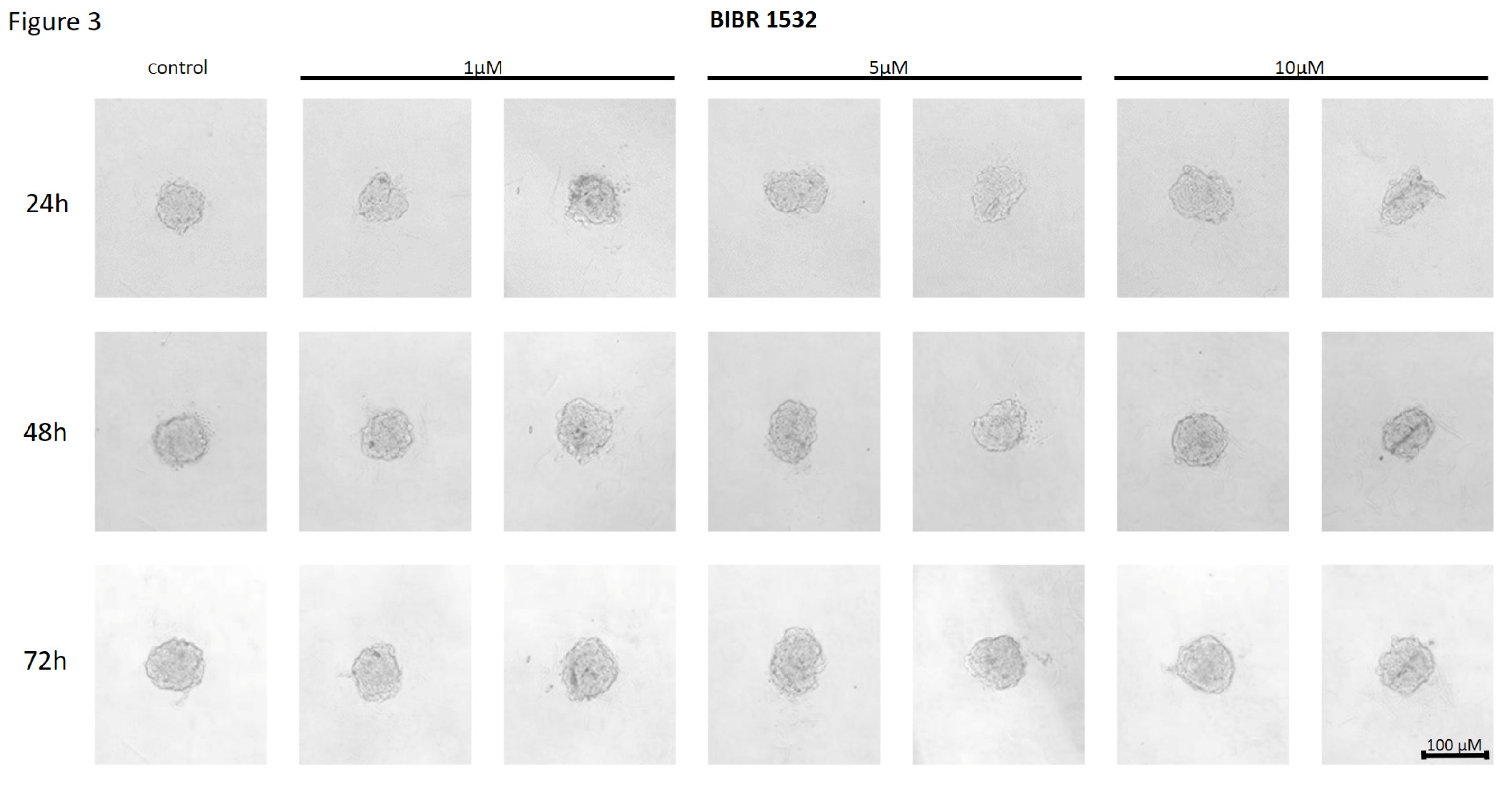
Effect of BIBR 1532 on the Cell-to-Cell Interactions and Spheroid Formation in MCF7 Cells
Effect of BIBR 1532 on the Cell-to-Cell Interactions and Spheroid Formation in MDA-MB-231 Cells
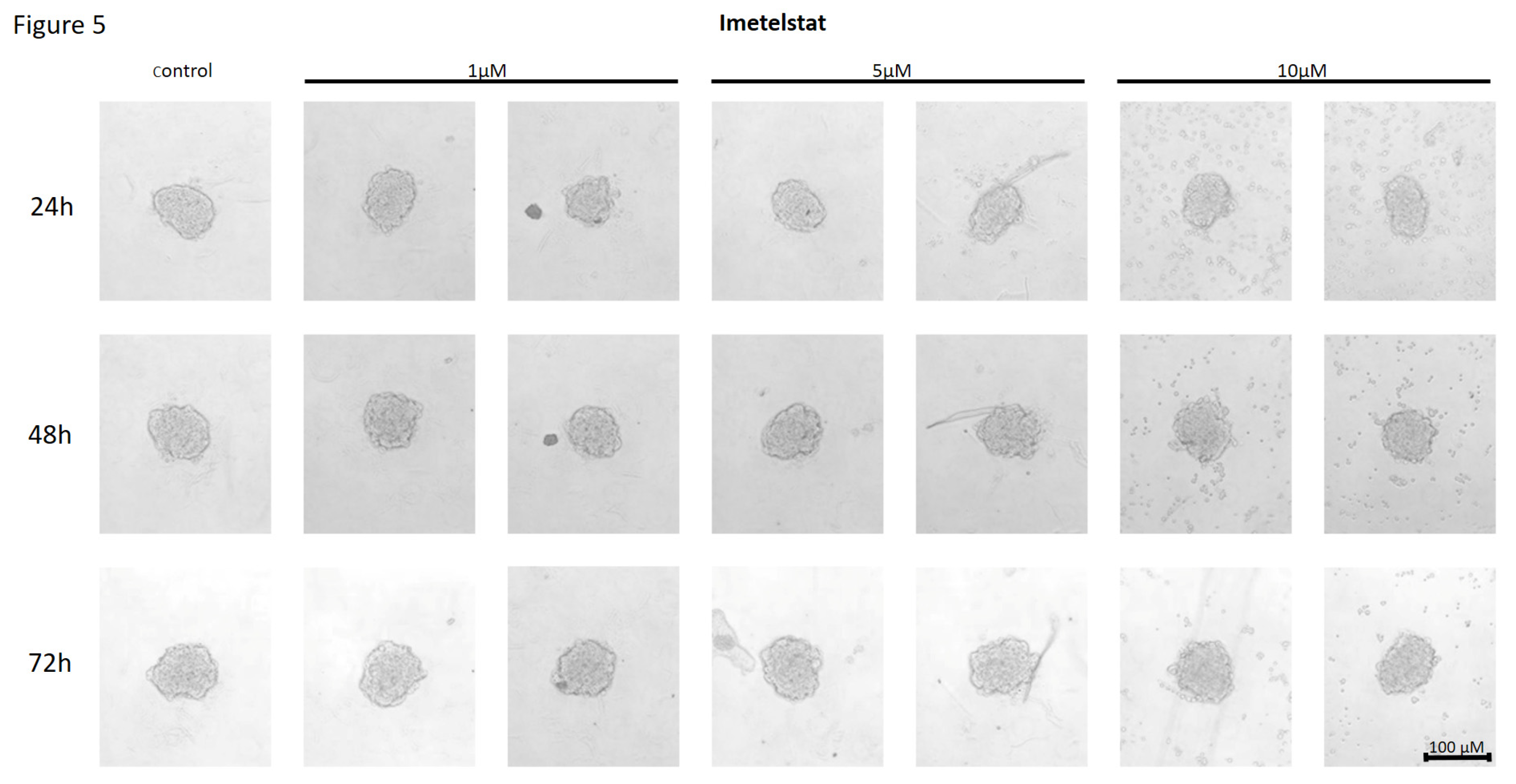
Effect of Imetelstat on the Cell-to-Cell Interactions and Spheroid Formation in MCF7 Cells
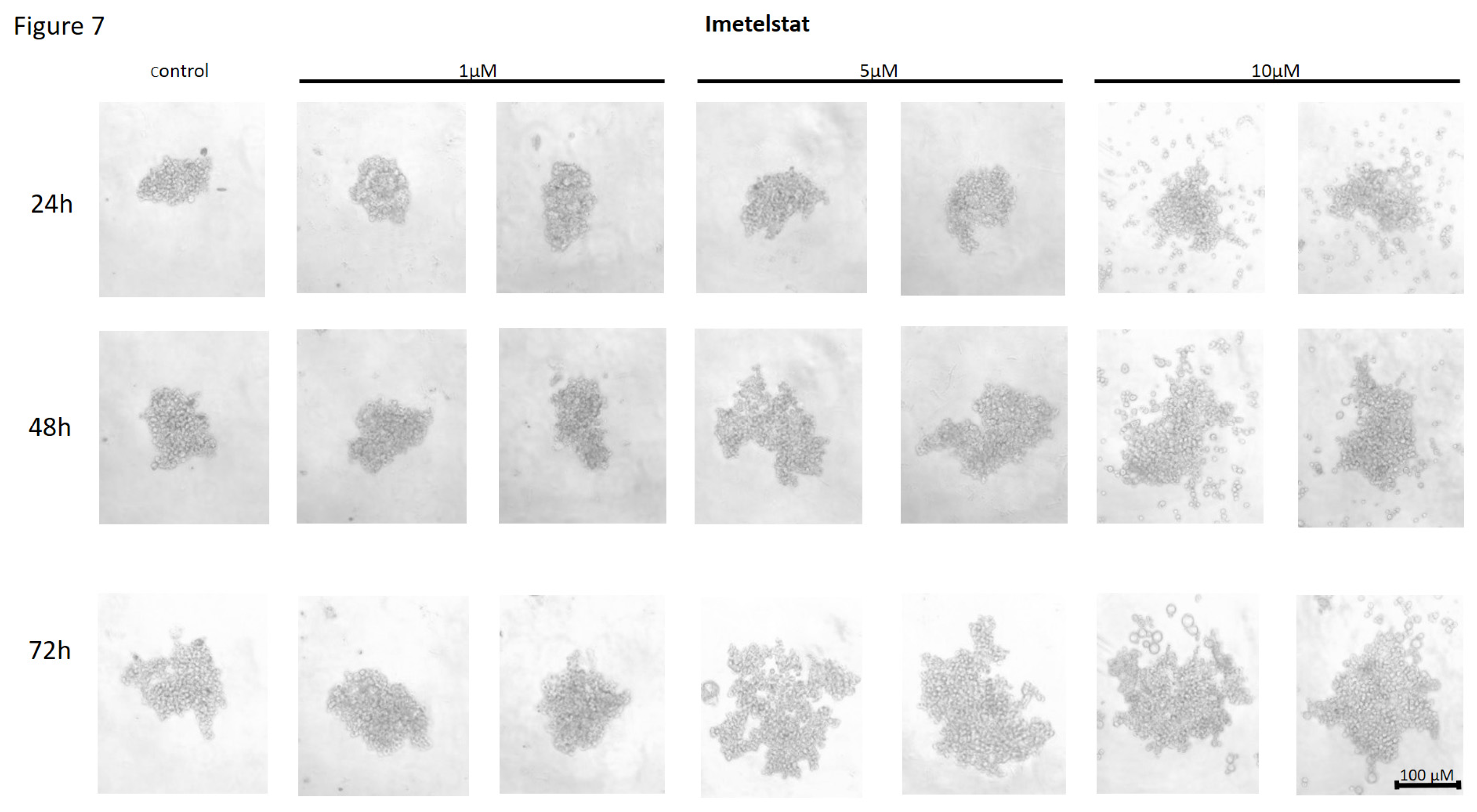
Effect of Imetelstat on the Cell-to-Cell Interactions and Spheroid Formation in MDA-MB-231 Cells
4. Discussion
TMPyP4 Potential to Alter Cancer Cells Adhesion Properties
BIBR 1532 Potential to Alter Cancer Cells Adhesion Properties
Imetelstat Potential to Alter Cancer Cells Adhesion Properties
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vasan, N.; Baselga, J.; Hyman, D.M. A view on drug resistance in cancer. Nature 2019, 575, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Lipinska, N.; Romaniuk, A.; Paszel-Jaworska, A.; Toton, E.; Kopczynski, P.; Rubis, B. Telomerase and drug resistance in cancer. Cell. Mol. Life Sci. 2017, 74, 4121–4132. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liu, Q.; Ge, Y.; Zhao, Q.; Zheng, X.; Zhao, Y. hTERT promotes cell adhesion and migration independent of telomerase activity. Sci. Rep. 2016, 6, 22886. [Google Scholar] [CrossRef]
- Janiszewska, M.; Primi, M.C.; Izard, T. Cell adhesion in cancer: Beyond the migration of single cells. J. Biol. Chem. 2020, 295, 2495–2505. [Google Scholar] [CrossRef]
- Sajjad, H.; Imtiaz, S.; Noor, T.; Siddiqui, Y.H.; Sajjad, A.; Zia, M. Cancer models in preclinical research: A chronicle review of advancement in effective cancer research. Anim. Model. Exp. Med. 2021, 4, 87–103. [Google Scholar] [CrossRef]
- Lipińska, N.; Rubiś, B. Telomerase as a target in diagnosis and treatment of cancer in postmenopausal women. Menopausal Rev. 2013, 6, 478–483. [Google Scholar] [CrossRef]
- Konieczna, N.; Romaniuk-Drapała, A.; Lisiak, N.; Totoń, E.; Paszel-Jaworska, A.; Kaczmarek, M.; Rubiś, B. Telomerase Inhibitor TMPyP4 Alters Adhesion and Migration of Breast-Cancer Cells MCF7 and MDA-MB-231. Int. J. Mol. Sci. 2019, 20, 2670. [Google Scholar] [CrossRef]
- Yin, L.; Duan, J.-J.; Bian, X.-W.; Yu, S.-C. Triple-negative breast cancer molecular subtyping and treatment progress. Breast Cancer Res. 2020, 22, 1–13. [Google Scholar] [CrossRef]
- Herbert, B.-S.; E Wright, W.; Shay, J.W. Telomerase and breast cancer. Breast Cancer Res. 2001, 3, 1–4. [Google Scholar] [CrossRef]
- Shay, J.W.; Wright, W.E. Hayflick, his limit, and cellular ageing. Nat. Rev. Mol. Cell Biol. 2000, 1, 72–76. [Google Scholar] [CrossRef]
- Palm, W.; de Lange, T. How Shelterin Protects Mammalian Telomeres. Annu. Rev. Genet. 2008, 42, 301–334. [Google Scholar] [CrossRef] [PubMed]
- Clark, G.M.; Osborne, C.K.; Levitt, D.; Wu, F.; Kim, N.W. Telomerase Activity and Survival of Patients With Node-Positive Breast Cancer. JNCI J. Natl. Cancer Inst. 1997, 89, 1874–1881. [Google Scholar] [CrossRef]
- Liu, H.; Liu, Q.; Ge, Y.; Zhao, Q.; Zheng, X.; Zhao, Y. hTERT promotes cell adhesion and migration independent of telomerase activity. Sci. Rep. 2016, 6, 22886. [Google Scholar] [CrossRef]
- Altamura, G.; Martano, M.; Licenziato, L.; Maiolino, P.; Borzacchiello, G. Telomerase Reverse Transcriptase (TERT) Expression, Telomerase Activity, and Expression of Matrix Metalloproteinases (MMP)-1/-2/-9 in Feline Oral Squamous Cell Carcinoma Cell Lines Associated With Felis catus Papillomavirus Type-2 Infection. Front. Veter- Sci. 2020, 7, 148. [Google Scholar] [CrossRef]
- Yamashita, T.; Uno, T.; Ishikawa, Y. Stabilization of guanine quadruplex DNA by the binding of porphyrins with cationic side arms. Bioorganic Med. Chem. 2005, 13, 2423–2430. [Google Scholar] [CrossRef]
- Izbicka, E.; Wheelhouse, R.T.; Raymond, E.; Davidson, K.K.; A Lawrence, R.; Sun, D.; E Windle, B.; Hurley, L.H.; Von Hoff, D.D. Effects of cationic porphyrins as G-quadruplex interactive agents in human tumor cells. Cancer Res. 1999, 59, 639–44. [Google Scholar]
- Anantha, N.V.; Azam, M.; Sheardy, R.D. Porphyrin Binding to Quadruplexed T4G4. Biochemistry 1998, 37, 2709–2714. [Google Scholar] [CrossRef]
- Arthanari, H.; Basu, S.; Kawano, T.L.; Bolton, P.H. Fluorescent dyes specific for quadruplex DNA. Nucleic Acids Res. 1998, 26, 3724–3728. [Google Scholar] [CrossRef]
- Zhuang, X.-Y.; Yao, Y.-G. Mitochondrial dysfunction and nuclear-mitochondrial shuttling of TERT are involved in cell proliferation arrest induced by G-quadruplex ligands. FEBS Lett. 2013, 587, 1656–1662. [Google Scholar] [CrossRef]
- Grand, C.L.; Han, H.; Muñoz, R.M.; Weitman, S.; Von Hoff, D.D.; Hurley, L.H.; Bearss, D.J. The cationic porphyrin TMPyP4 down-regulates c-MYC and human telomerase reverse transcriptase expression and inhibits tumor growth in vivo. Mol Cancer Ther. 2002, 1, 565–73. [Google Scholar]
- Cheng, M.-J.; Cao, Y.-G. TMPYP4 exerted antitumor effects in human cervical cancer cells through activation of p38 mitogen-activated protein kinase. Biol. Res. 2017, 50, 24–24. [Google Scholar] [CrossRef] [PubMed]
- Pascolo, E.; Wenz, C.; Lingner, J.; Hauel, N.; Priepke, H.; Kauffmann, I.; Garin-Chesa, P.; Rettig, W.J.; Damm, K.; Schnapp, A. Mechanism of Human Telomerase Inhibition by BIBR1532, a Synthetic, Non-nucleosidic Drug Candidate. J. Biol. Chem. 2002, 277, 15566–15572. [Google Scholar] [CrossRef] [PubMed]
- El-Daly, H.; Kull, M.; Zimmermann, S.; Pantic, M.; Waller, C.F.; Martens, U.M. Selective cytotoxicity and telomere damage in leukemia cells using the telomerase inhibitor BIBR1532. Blood 2005, 105, 1742–1749. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.J.; Autexier, C. Pharmacological Telomerase Inhibition Can Sensitize Drug-Resistant and Drug-Sensitive Cells to Chemotherapeutic Treatment. Mol Pharmacol. 2005, 68, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Röth, A.; Dürig, J.; Himmelreich, H.; Bug, S.; Siebert, R.; Dührsen, U.; Lansdorp, P.M.; Baerlocher, G.M. Short telomeres and high telomerase activity in T-cell prolymphocytic leukemia. Leukemia 2007, 21, 2456–2462. [Google Scholar] [CrossRef]
- Meng, E.; Taylor, B.; Ray, A.; Shevde, L.A.; Rocconi, R.P. Targeted inhibition of telomerase activity combined with chemotherapy demonstrates synergy in eliminating ovarian cancer spheroid-forming cells. Gynecol. Oncol. 2012, 124, 598–605. [Google Scholar] [CrossRef]
- FDA.gov. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-imetelstat-low-intermediate-1-risk-myelodysplastic-syndromes-transfusion-dependent. (accessed 25 September 2024).
- Herbert, B.-S.; Gellert, G.C.; Hochreiter, A.; Pongracz, K.; E Wright, W.; Zielinska, D.; Chin, A.C.; Harley, C.B.; Shay, J.W.; Gryaznov, S.M. Lipid modification of GRN163, an N3′ → P5′ thio-phosphoramidate oligonucleotide, enhances the potency of telomerase inhibition. Oncogene 2005, 24, 5262–5268. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, J.; Yang, S.; Kuang, Z.; Tan, G.; Yang, G.; Wei, Q.; Guo, Z. Telomerase antagonist imetelstat increases radiation sensitivity in esophageal squamous cell carcinoma. Oncotarget 2017, 8, 13600–13610. [Google Scholar] [CrossRef]
- Goldblatt, E.M.; Gentry, E.R.; Fox, M.J.; Gryaznov, S.M.; Shen, C.; Herbert, B.-S. The telomerase template antagonist GRN163L alters MDA-MB-231 breast cancer cell morphology, inhibits growth, and augments the effects of paclitaxel. Mol. Cancer Ther. 2009, 8, 2027–2035. [Google Scholar] [CrossRef]
- Koziel, J.E.; Herbert, B.-S. The telomerase inhibitor imetelstat alone, and in combination with trastuzumab, decreases the cancer stem cell population and self-renewal of HER2+ breast cancer cells. Breast Cancer Res. Treat. 2015, 149, 607–618. [Google Scholar] [CrossRef]
- Garnique, A.; Rezende-Teixeira, P.; Machado-Santelli, G. Telomerase inhibitors TMPyP4 and thymoquinone decreased cell proliferation and induced cell death in the non-small cell lung cancer cell line LC-HK2, modifying the pattern of focal adhesion. Braz. J. Med Biol. Res. 2023, 56, e12897. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.-H.; Nie, X.; Liu, H.-Y.; Fang, Y.-M.; Zhao, Y.; Xia, L.-X. TMPyP4 promotes cancer cell migration at low doses, but induces cell death at high doses. Sci. Rep. 2016, 6, 26592. [Google Scholar] [CrossRef] [PubMed]
- Nasrollahzadeh, A.; Bashash, D.; Kabuli, M.; Zandi, Z.; Kashani, B.; Zaghal, A.; Mousavi, S.A.; Ghaffari, S.H. Arsenic trioxide and BIBR1532 synergistically inhibit breast cancer cell proliferation through attenuation of NF-κB signaling pathway. Life Sci. 2020, 257, 118060. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Sun, L.; Chen, G.; Zheng, D.; Li, L.; Wei, W. A combination of the telomerase inhibitor, BIBR1532, and paclitaxel synergistically inhibit cell proliferation in breast cancer cell lines. Target. Oncol. 2015, 10, 565–573. [Google Scholar] [CrossRef]
- Joseph, I.; Tressler, R.; Bassett, E.; Harley, C.; Buseman, C.M.; Pattamatta, P.; Wright, W.E.; Shay, J.W.; Go, N.F. The Telomerase Inhibitor Imetelstat Depletes Cancer Stem Cells in Breast and Pancreatic Cancer Cell Lines. Cancer Res. 2010, 70, 9494–9504. [Google Scholar] [CrossRef]
- Frink, R.E.; Peyton, M.; Schiller, J.H.; Gazdar, A.F.; Shay, J.W.; Minna, J.D. Telomerase inhibitor imetelstat has preclinical activity across the spectrum of non-small cell lung cancer oncogenotypes in a telomere length dependent manner. Oncotarget 2016, 7, 31639–31651. [Google Scholar] [CrossRef]
- Bashash, D.; Ghaffari, S.H.; Zaker, F.; Hezave, K.; Kazerani, M.; Ghavamzadeh, A.; Alimoghaddam, K.; Mosavi, S.A.; Gharehbaghian, A.; Vossough, P. Direct Short-Term Cytotoxic Effects of BIBR 1532 on Acute Promyelocytic Leukemia Cells Through Induction of p21 Coupled with Downregulation of c-Myc and hTERT Transcription. Cancer Investig. 2012, 30, 57–64. [Google Scholar] [CrossRef]
- Baerlocher, G.M.; Haubitz, M.; Braschler, T.R.; Brunold, C.; Burington, B.; Leibundgut, E.O.; Go, N. Imetelstat inhibits growth of megakaryocyte colony-forming units from patients with essential thrombocythemia. Blood Adv. 2019, 3, 3724–3728. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



