Submitted:
09 October 2024
Posted:
10 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Equipment
2.1. Materials
2.2. Equipment
3. Methods and Experimental Part
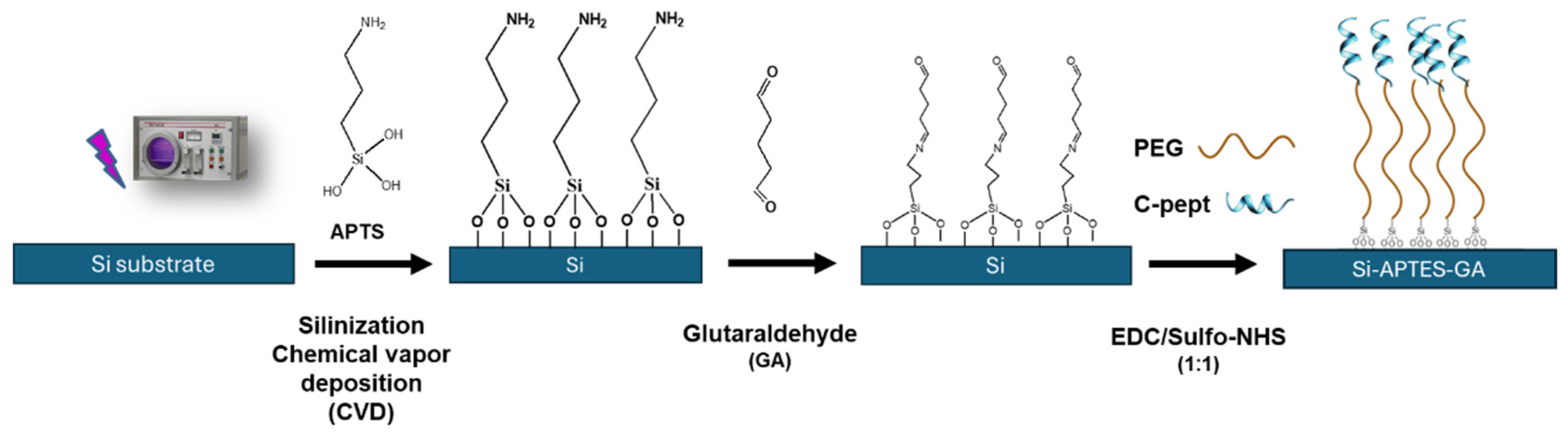
3.1. Silica Surface Functionalization with Glutaraldehyde Chemistry for Peptide Immobilization
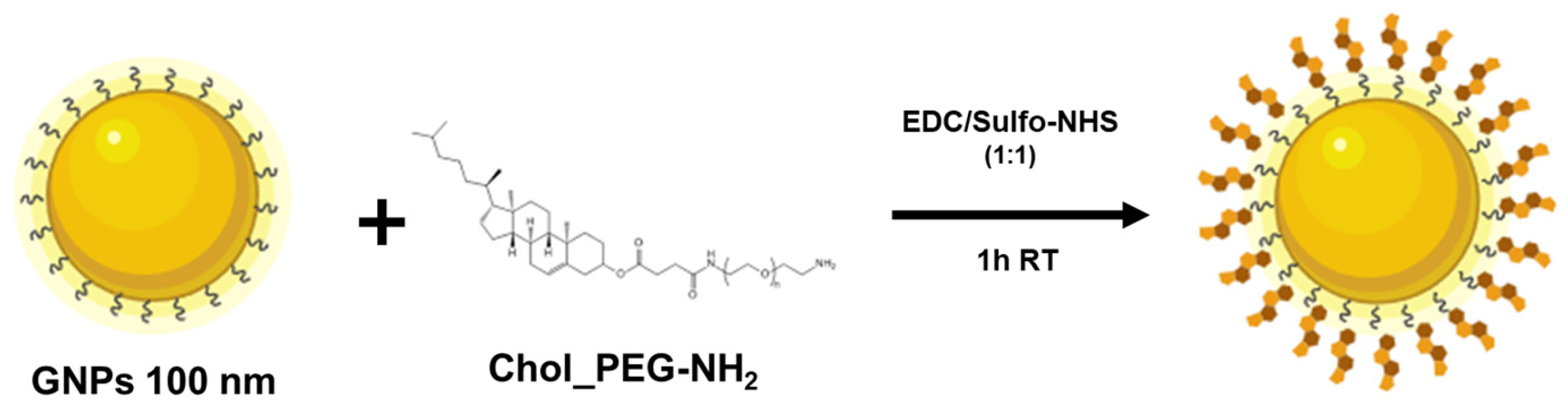
3.2. Functionalization of Gold Nanoparticles (GNPs)
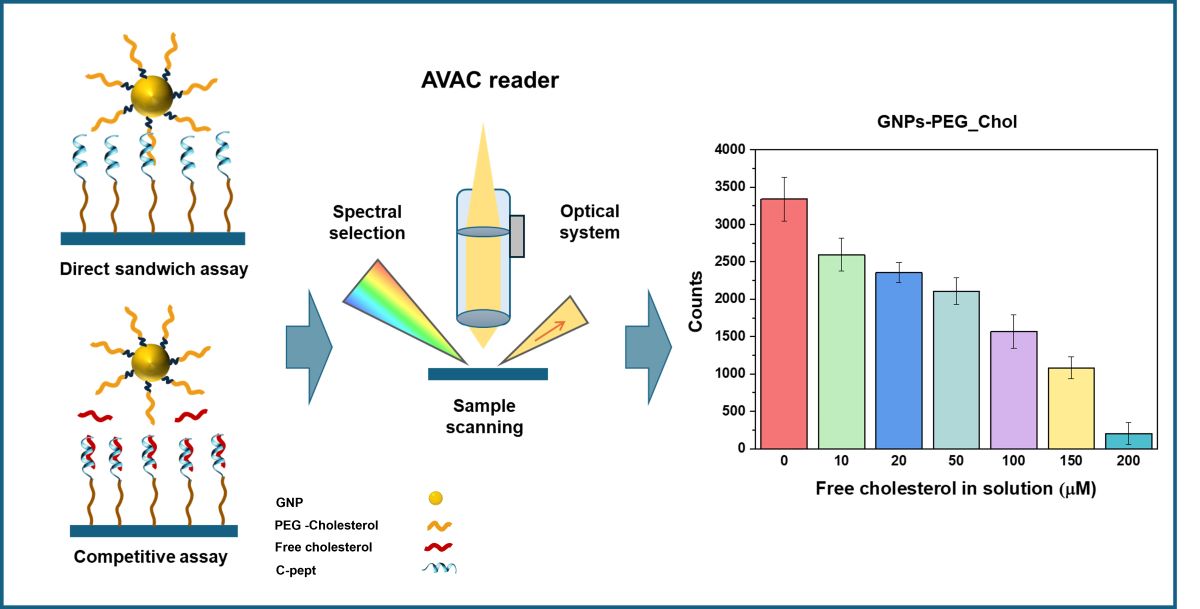
3.3. Competitive Plasmonic Bioassay for Cholesterol Determination
3.4. AVAC Analyzer
3.5. Surface Characterization
4. Results and Discussion
4.1. Functionalization of Silicon Surface with C-pept as Recognition Element
4.2. GNPs Functionalization with Cholesterol
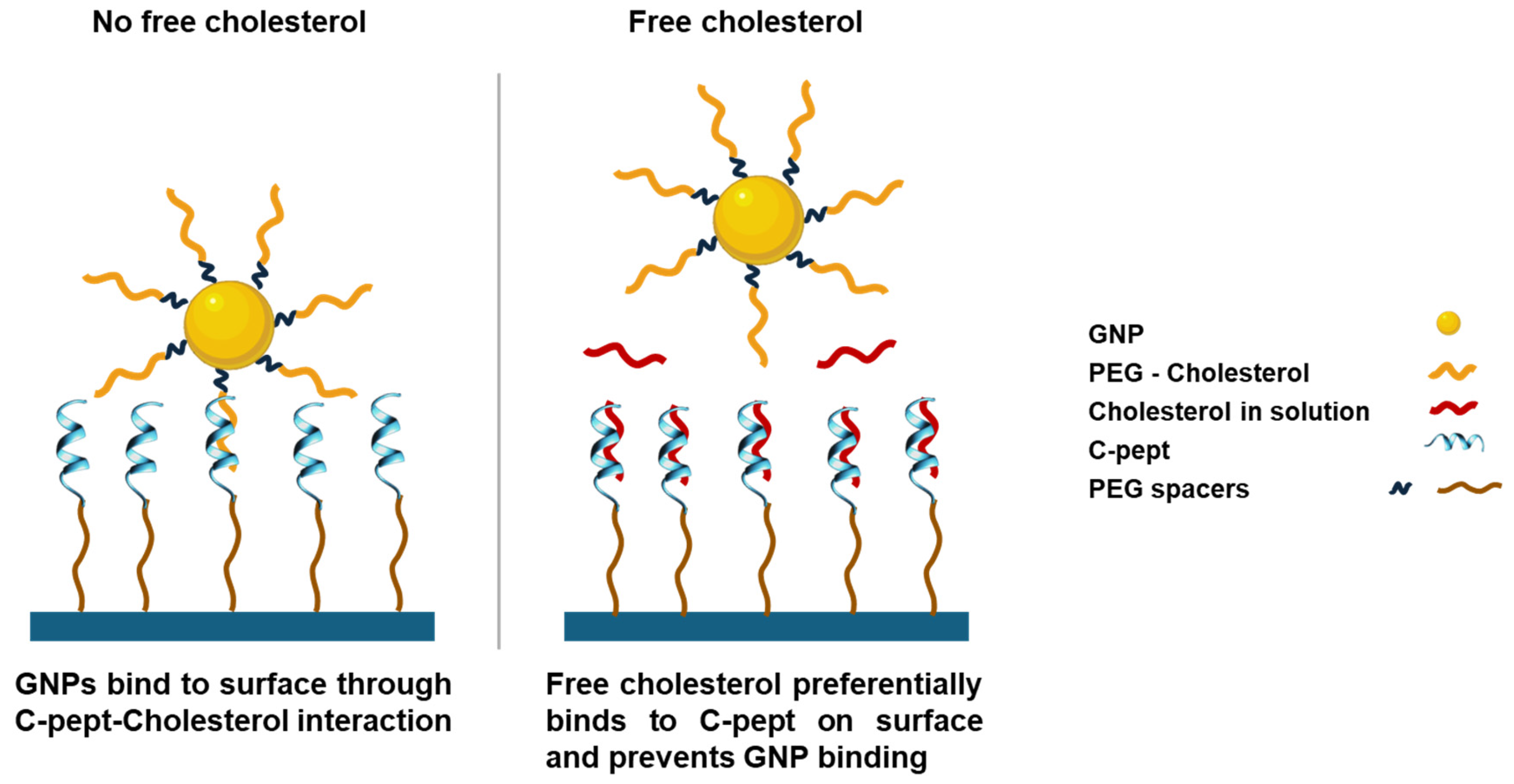
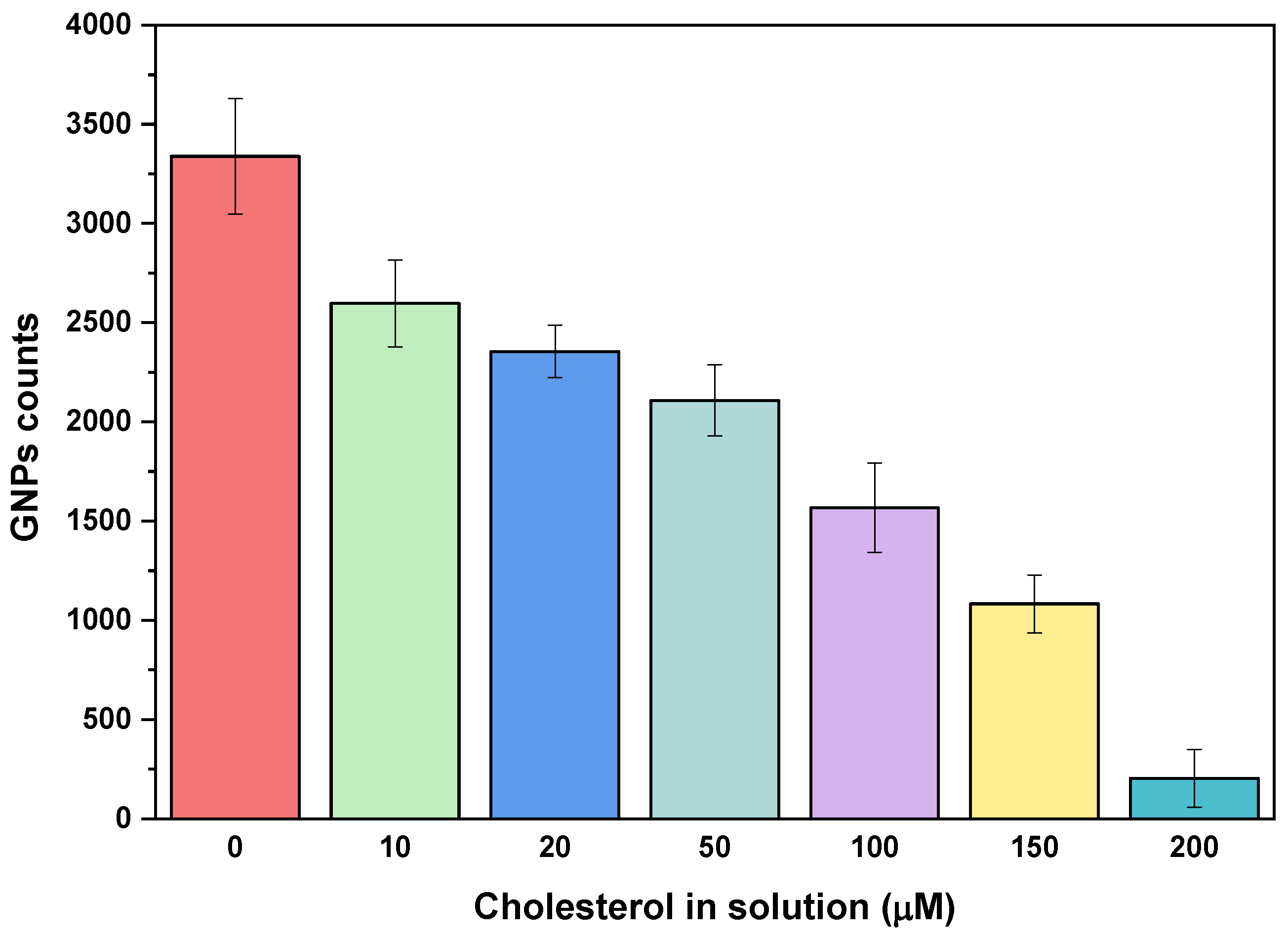
4.3. Peptide-Based Plasmonic Competitive Assay for Cholesterol Quantification
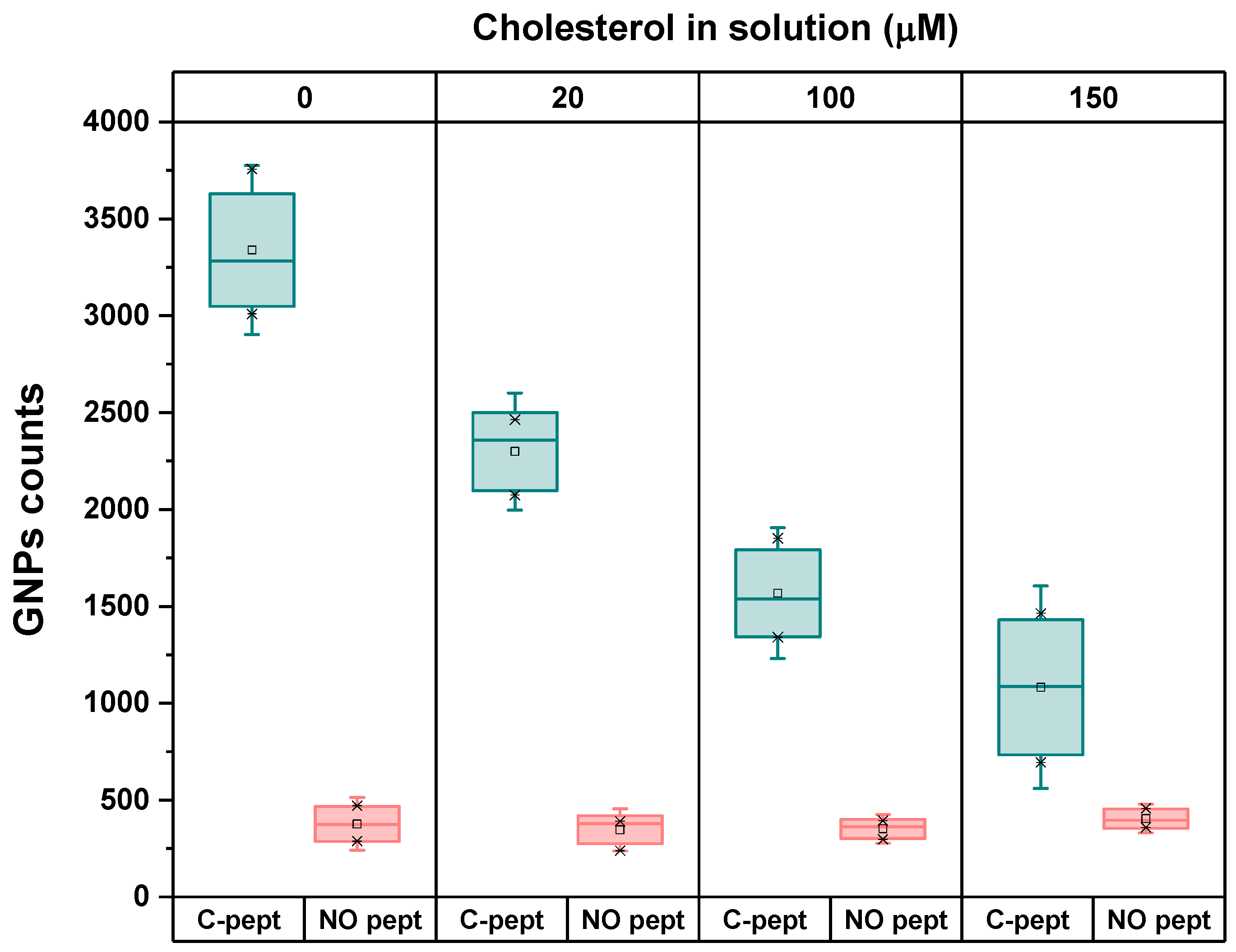
4.4. Selection of the System Configuration
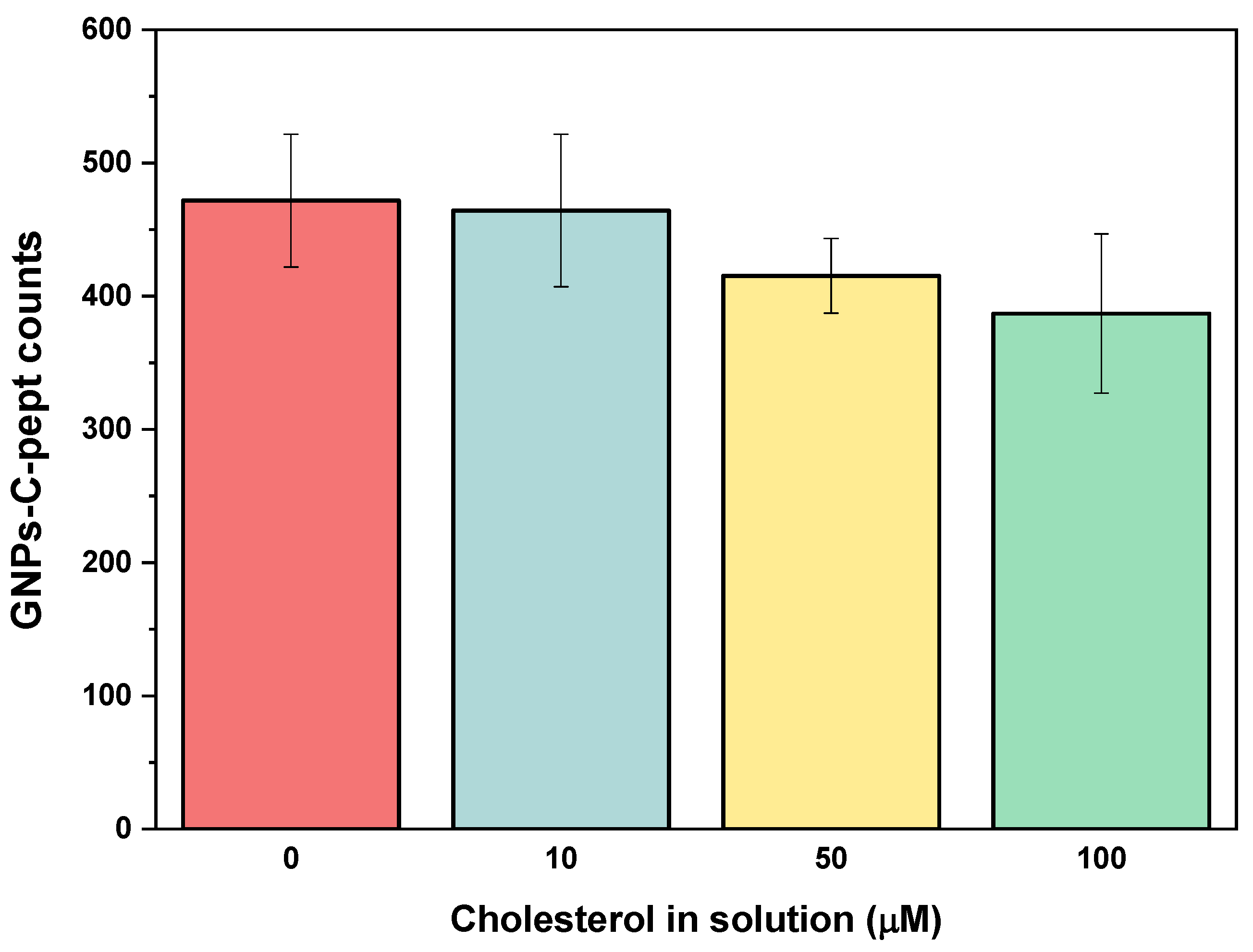
4.5. Specificity of the Biorecognition Peptide Element
5. Conclusion
Authors Contribution
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oddi, S.; Dainese, E.; Fezza, F.; Lanuti, M.; Barcaroli, D.; De Laurenzi, V.; Centonze, D.; MacCarrone, M. Functional Characterization of Putative Cholesterol Binding Sequence (CRAC) in Human Type-1 Cannabinoid Receptor. J Neurochem 2011, 116, 858–865. [CrossRef]
- Morales-Lázaro, S.L.; Rosenbaum, T. Cholesterol as a Key Molecule That Regulates TRPV1 Channel Function. Adv Exp Med Biol 2019, 1135, 105–117. [CrossRef]
- Dainese, E.; De Fabritiis, G.; Sabatucci, A.; Oddi, S.; Angelucci, C.B.; Di Pancrazio, C.; Giorgino, T.; Stanley, N.; Del Carlo, M.; Cravatt, B.F.; et al. Membrane Lipids Are Key Modulators of the Endocannabinoid-Hydrolase FAAH. Biochemical Journal 2014, 457, 463–472. [CrossRef]
- Dainese, E.; Oddi, S.; Bari, M.; Maccarrone, M. Modulation of the Endocannabinoid System by Lipid Rafts. Curr Med Chem 2007, 14, 2702–2715. [CrossRef]
- Maccarrone, M.; Bernardi, G.; Agrò, A.F.; Centonze, D. Cannabinoid Receptor Signalling in Neurodegenerative Diseases: A Potential Role for Membrane Fluidity Disturbance. Br J Pharmacol 2011, 163, 1379–1390. [CrossRef]
- Oddi, S.; Caporali, P.; Dragotto, J.; Totaro, A.; Maiolati, M.; Scipioni, L.; Angelucci, C.B.; Orsini, C.; Canterini, S.; Rapino, C.; et al. The Endocannabinoid System Is Affected by Cholesterol Dyshomeostasis: Insights from a Murine Model of Niemann Pick Type C Disease. Neurobiol Dis 2019, 130. [CrossRef]
- Sniderman, A.; McQueen, M.; Contois, J.; Williams, K.; Furberg, C.D. Why Is Non−high-Density Lipoprotein Cholesterol a Better Marker of the Risk of Vascular Disease than Low-Density Lipoprotein Cholesterol? J Clin Lipidol 2010, 4, 152–155. [CrossRef]
- Golier, J.A.; Marzuk, P.M.; Leon, A.C.; Weiner, C.; Tardiff, K. Low Serum Cholesterol Level and Attempted Suicide. 2006, 152, 419–423. [CrossRef]
- Rong, S.; Li, B.; Chen, L.; Sun, Y.; Du, Y.; Liu, B.; Robinson, J.G.; Bao, W. Association of Low-Density Lipoprotein Cholesterol Levels with More than 20-Year Risk of Cardiovascular and All-Cause Mortality in the General Population. J Am Heart Assoc 2022, 11, 23690. [CrossRef]
- Wang, X.; Cohen, L.; Wang, J.; Walt, D.R. Competitive Immunoassays for the Detection of Small Molecules Using Single Molecule Arrays. J Am Chem Soc 2018, 140, 18132–18139. [CrossRef]
- Wang, S.; Chen, S.; Shang, K.; Gao, X.; Wang, X. Sensitive Electrochemical Detection of Cholesterol Using a Portable Paper Sensor Based on the Synergistic Effect of Cholesterol Oxidase and Nanoporous Gold. Int J Biol Macromol 2021, 189, 356–362. [CrossRef]
- Salazar, P.; Martín, M.; González-Mora, J.L. In Situ Electrodeposition of Cholesterol Oxidase-Modified Polydopamine Thin Film on Nanostructured Screen Printed Electrodes for Free Cholesterol Determination. Journal of Electroanalytical Chemistry 2019, 837, 191–199. [CrossRef]
- Singh, S.; Solanki, P.R.; Pandey, M.K.; Malhotra, B.D. Cholesterol Biosensor Based on Cholesterol Esterase, Cholesterol Oxidase and Peroxidase Immobilized onto Conducting Polyaniline Films. Sensors and Actuators B 2006, 115, 534–541. [CrossRef]
- Mukai, M.; Krause, M.R.; Regen, S.L. Peptide Recognition of Cholesterol in Fluid Phospholipid Bilayers. J Am Chem Soc 2015, 137, 12518–12520. [CrossRef]
- Sinha, A.; Basiruddin, S.K.; Chakraborty, A.; Jana, N.R. β-Cyclodextrin Functionalized Magnetic Mesoporous Silica Colloid for Cholesterol Separation. ACS Appl Mater Interfaces 2015, 7, 1340–1347. [CrossRef]
- Ballesta-Claver, J.; Salinas Velázquez, P.; Valencia-Mirón, M.C.; Capitán-Vallvey, L.F. SPE Biosensor for Cholesterol in Serum Samples Based on Electrochemiluminescent Luminol Copolymer. Talanta 2011, 86, 178–185. [CrossRef]
- Hu, S.R.; Yang, C.R.; Huang, Y.F.; Huang, C.C.; Chen, Y.L.; Chang, H.T. Ratiometric Fluorescence Probe of Vesicle-like Carbon Dots and Gold Clusters for Quantitation of Cholesterol. Chemosensors 2022, 10, 160. [CrossRef]
- Xiao, W.; Yang, Z.; Liu, J.; Chen, Z.C.; Li, H. Sensitive Cholesterol Determination by β-Cyclodextrin Recognition Based on Fluorescence Enhancement of Gold Nanoclusters. Microchemical Journal 2022, 175, 107125. [CrossRef]
- Calvo, R.; Rodriguez Mariblanca, I.; Pini, V.; Dias, M.; Cebrian, V.; Thon, A.; Saad, A.; Salvador-Matar, A.; Ahumada, Ó.; Manso Silván, M.; et al. Novel Characterization Techniques for Multifunctional Plasmonic–Magnetic Nanoparticles in Biomedical Applications. Nanomaterials 2023, 13, 2929. [CrossRef]
- Li, Z.; Leustean, L.; Inci, F.; Zheng, M.; Demirci, U.; Wang, S. Plasmonic-Based Platforms for Diagnosis of Infectious Diseases at the Point-of-Care. Biotechnol Adv 2019, 37, 107440. [CrossRef]
- Mauriz, E.; Lechuga, L.M. Plasmonic Biosensors for Single-Molecule Biomedical Analysis. Biosensors (Basel) 2021, 11, 123. [CrossRef]
- Nyembe, S.; Mkhohlakali, A.; May, B.; Mhlanga, N.; Nyembe, S.; Mkhohlakali, A.; May, B.; Mhlanga, N. Application of Plasmonic Nanostructures in Molecular Diagnostics and Biosensor Technology: Challenges and Current Developments. Plasmonic Nanostructures—Basic Concepts, Optimization and Applications 2022. [CrossRef]
- Rosman, C.; Prasad, J.; Neiser, A.; Henkel, A.; Edgar, J.; Sönnichsen, C. Multiplexed Plasmon Sensor for Rapid Label-Free Analyte Detection. Nano Lett 2013, 13, 3243–3247. [CrossRef]
- Kumalasari, M.R.; Alfanaar, R.; Andreani, A.S. Gold Nanoparticles (AuNPs): A Versatile Material for Biosensor Application. Talanta Open 2024, 9, 100327. [CrossRef]
- Pellas, V.; Hu, D.; Mazouzi, Y.; Mimoun, Y.; Blanchard, J.; Guibert, C.; Salmain, M.; Boujday, S. Gold Nanorods for LSPR Biosensing: Synthesis, Coating by Silica, and Bioanalytical Applications. Biosensors (Basel) 2020, 10, 146. [CrossRef]
- Ferrari, E. Gold Nanoparticle-Based Plasmonic Biosensors. Biosensors (Basel) 2023, 13, 411. [CrossRef]
- Saha, K.; Agasti, S.S.; Kim, C.; Li, X.; Rotello, V.M. Gold Nanoparticles in Chemical and Biological Sensing. Chem Rev 2012, 112, 2739–2779. [CrossRef]
- Yeh, Y.C.; Creran, B.; Rotello, V.M. Gold Nanoparticles: Preparation, Properties, and Applications in Bionanotechnology. Nanoscale 2012, 4, 1871–1880. [CrossRef]
- Zeng, S.; Yong, K.T.; Roy, I.; Dinh, X.Q.; Yu, X.; Luan, F. A Review on Functionalized Gold Nanoparticles for Biosensing Applications. Plasmonics 2011 6:3 2011, 6, 491–506. [CrossRef]
- Jazayeri, M.H.; Amani, H.; Pourfatollah, A.A.; Pazoki-Toroudi, H.; Sedighimoghaddam, B. Various Methods of Gold Nanoparticles (GNPs) Conjugation to Antibodies. Sens Biosensing Res 2016, 9, 17–22. [CrossRef]
- Chen, Y.; Xianyu, Y.; Jiang, X. Surface Modification of Gold Nanoparticles with Small Molecules for Biochemical Analysis. Acc Chem Res 2017, 50, 310–319. [CrossRef]
- Pellas, V.; Sallem, F.; Blanchard, J.; Miche, A.; Concheso, S.M.; Méthivier, C.; Salmain, M.; Boujday, S. Silica-Coated Gold Nanorods Biofunctionalization for Localized Surface Plasmon Resonance (LSPR) Biosensing. Talanta 2023, 255, 124245. [CrossRef]
- Ghosh, S.K.; Pal, T. Interparticle Coupling Effect on the Surface Plasmon Resonance of Gold Nanoparticles: From Theory to Applications. Chem Rev 2007, 107, 4797–4862. [CrossRef]
- Taylor, A.B.; Zijlstra, P. Single-Molecule Plasmon Sensing: Current Status and Future Prospects. ACS Sens 2017, 2, 1103–1122. [CrossRef]
- Yu, T.; Wei, Q. Plasmonic Molecular Assays: Recent Advances and Applications for Mobile Health. Nano Res 2018, 11, 5439. [CrossRef]
- Hill, R.T. Plasmonic Biosensors. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2015, 7, 152. [CrossRef]
- D’Agata, R.; Bellassai, N.; Spoto, G. Exploiting the Design of Surface Plasmon Resonance Interfaces for Better Diagnostics: A Perspective Review. Talanta 2024, 266, 125033. [CrossRef]
- Karimzadeh, A.; Hasanzadeh, M.; Shadjou, N.; Guardia, M. de la Peptide Based Biosensors. TrAC Trends in Analytical Chemistry 2018, 107, 1–20. [CrossRef]
- Liu, Q.; Wang, J.; Boyd, B.J. Peptide-Based Biosensors. Talanta 2015, 136, 114–127. [CrossRef]
- Saadati, A.; Hassanpour, S.; Guardia, M. de la; Mosafer, J.; Hashemzaei, M.; Mokhtarzadeh, A.; Baradaran, B. Recent Advances on Application of Peptide Nucleic Acids as a Bioreceptor in Biosensors Development. TrAC Trends in Analytical Chemistry 2019, 114, 56–68. [CrossRef]
- Koyiloth, M.; Gummadi, S.N. Interaction of Human Phospholipid Scramblase 1 with Cholesterol via CRAC Motif Is Essential for Functional Regulation and Subcellular Localization. Int J Biol Macromol 2022, 209, 850–857. [CrossRef]
- Volynsky, P.E.; Galimzyanov, T.R.; Akimov, S.A. Interaction of Peptides Containing CRAC Motifs with Lipids in Membranes of Various Composition. Biochem. Moscow Suppl. Ser. A 2021, 15, 98–108. [CrossRef]
- Li, H.; Yao, Z.X.; Degenhardt, B.; Teper, G.; Papadopoulos, V. Cholesterol Binding at the Cholesterol Recognition/Interaction Amino Acid Consensus (CRAC) of the Peripheral-Type Benzodiazepine Receptor and Inhibition of Steroidogenesis by an HIV TAT-CRAC Peptide. Proc Natl Acad Sci U S A 2001, 98, 1267–1272. [CrossRef]
- Li, H.; Papadopoulos, V. Peripheral-Type Benzodiazepine Receptor Function in Cholesterol Transport. Identification of a Putative Cholesterol Recognition/Interaction Amino Acid Sequence and Consensus Pattern. Endocrinology 1998, 139, 4991–4997. [CrossRef]
- Giarola, J.F.; Santos, J.; Estevez, M.C.; Ventura, S.; Pallarès, I.; Lechuga, L.M. An α-Helical Peptide-Based Plasmonic Biosensor for Highly Specific Detection of α-Synuclein Toxic Oligomers. Anal Chim Acta 2024, 1304, 342559. [CrossRef]
- Heo, N.S.; Oh, Y.; Ryu, M.Y.; Hoon Baek, S.; Park, J.; Choi, C.; Huh, Y.S.; Park, J.P. Affinity Peptide-Guided Plasmonic Biosensor for Detection of Noroviral Protein and Human Norovirus. Biotechnology and Bioprocess Engineering 2019, 24, 318–325. [CrossRef]
- Dey, S.; Dolci, M.; Zijlstra, P. Single-Molecule Optical Biosensing: Recent Advances and Future Challenges. ACS Physical Chemistry Au 2023, 3, 143–156. [CrossRef]
- Calvo, R.; Thon, A.; Saad, A.; Salvador-Matar, A.; Manso-Silván, M.; Ahumada, Ó.; Pini, V. Size Characterization of Plasmonic Nanoparticles with Dark-Field Single Particle Spectrophotometry. Scientific Reports | 123AD, 12, 17231. [CrossRef]
- Sriram, M.; Markhali, B.P.; Nicovich, P.R.; Bennett, D.T.; Reece, P.J.; Brynn Hibbert, D.; Tilley, R.D.; Gaus, K.; Vivekchand, S.R.C.; Gooding, J.J. A Rapid Readout for Many Single Plasmonic Nanoparticles Using Dark-Field Microscopy and Digital Color Analysis. Biosens Bioelectron 2018, 117, 530–536. [CrossRef]
- Pini, V.; Thon, A.; Salvador -Matar Renteria, A.; Cebrián Hernando, V.; García Aguado, C.; Ahumada Heredero, J.Ó. Biosensor Platform and Method for the Simultaneous, Multiplexed, Ultra-Sensitive and High Throughput Optical Detection of Biomarkers. U.S. Patent and Trademark Office 2020, US11519843B2.
- Thon, A.; Pini, V.; Salvador-Matar Renteria, A.; Cebrián Hernardo, V.; García Aguado, C.; Ahumada Heredero, J.Ó. Method for Optically Detecting Biomarkers. U.S. Patent and Trademark Office 2022, US11519856B2.
- Bernardo, A.L.; Mohammed-Sadhakathullah, A.H.M.; Angelucci, C.B.; Estrany, F.; Berghella, A.; Torras, J.; Armelin, E.; Oddi, S.; Dainese, E. Non-Enzymatic Cholesterol Biosensor: Electrochemical Sensing Based on Peptide-PLA Thin Film. Int J Biol Macromol 2024. [CrossRef]
- Bañuls, M.J.; Puchades, R.; Maquieira, Á. Chemical Surface Modifications for the Development of Silicon-Based Label-Free Integrated Optical (IO) Biosensors: A Review. Anal Chim Acta 2013, 777, 1–16. [CrossRef]
- Antoniou, M.; Tsounidi, D.; Petrou, P.S.; Beltsios, K.G.; Kakabakos, S.E. Functionalization of Silicon Dioxide and Silicon Nitride Surfaces with Aminosilanes for Optical Biosensing Applications. Med Devices Sens 2020, 3. [CrossRef]
- Soler, M.; Lechuga, L.M. Biochemistry Strategies for Label-Free Optical Sensor Biofunctionalization: Advances towards Real Applicability. Anal Bioanal Chem 2022, 414, 5071. [CrossRef]
- Oliveira, J.P.; Prado, A.R.; Keijok, W.J.; Antunes, P.W.P.; Yapuchura, E.R.; Guimarães, M.C.C. Impact of Conjugation Strategies for Targeting of Antibodies in Gold Nanoparticles for Ultrasensitive Detection of 17β-Estradiol. Sci Rep 2019, 9. [CrossRef]
- Gopalan, A.I.; Lee, K.-P.; Ragupathy, D. Development of a Stable Cholesterol Biosensor Based on Multi-Walled Carbon Nanotubes-Gold Nanoparticles Composite Covered with a Layer of Chitosan-Room-Temperature Ionic Liquid Network. Biosens Bioelectron 2009, 24, 2211–2217. [CrossRef]
- Carvalho-De-Souza, J.L.; Nag, O.K.; Oh, E.; Huston, A.L.; Vurgaftman, I.; Pepperberg, D.R.; Bezanilla, F.; Delehanty, J.B. Cholesterol Functionalization of Gold Nanoparticles Enhances Photoactivation of Neural Activity. ACS Chem Neurosci 2019, 10, 1478–1487. [CrossRef]
- Gautam, S.; Loh, K.C. Immobilization of Hydrophobic Peptidic Ligands to Hydrophilic Chromatographic Matrix: A Preconcentration Approach. Anal Biochem 2012, 423, 202–209. [CrossRef]

| Diameter (nm) | 100 |
| Peak SPR Wavelength (nm) | 569 |
| NPS/ml | 2.48 × 10 11 |
| Molarity (pM) | 4.10 × 10 2 |
| Moles | 4.14 × 10 -13 |
| Molar Ext. (M-1cm-1) | 1.21 × 10 11 |
| Absorption Molar Ext. (M-1cm-1) | 5.82 × 10 10 |
| Scattering Molar Ext. (M-1cm-1) | 6.27 × 10 10 |
| Size Dispersity %PDI | < 4% |
| Size Accuracy (+/- nm) | 2 |
| Surface modification | WCA (º) |
| Si | 39 ± 2 |
| Si-APTES | 59 ± 1 |
| PEG (linker) | 67 ± 3 |
| PEG-C-pept | 78 ± 3 |
| PEG- C-pept /GNPs-Chol | 83 ± 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




