3. Discussion
Among the phenolic compounds, 8 flavonoids were identified: rutin, quercetin, quercetin-3-glucoside, luteolin, apigenin, neohesperidin, naringin, and naringenin. Additionally, 4 tannin metabolites were identified: pyrocatechin, epicatechin, epicatechin gallate, and gallocatechin. 5 hydroxycinnamic acids were also identified: caffeic,
p-coumaric, sinapic, trans-cinnamic, and trans-ferulic acids. 3 phenol carboxylic acids were found: gallic, benzoic, and syringic acids. Quinic acid was also identified in the extracts. The predominant hydroxycinnamic acids in
A. reptans herb extracts were
p-coumaric, caffeic, and sinapic acids; among the flavonoids, rutin, quercetin, and neohesperidin were predominant; and among the tannin metabolites, epicatechin gallate and gallocatechin were the most significant. It was reported that in
A. reptans, raw materials from Turkey, caffeic and chlorogenic acids predominated among hydroxycinnamic acids and luteolin derivative - among flavonoids [
34]. In the other research isoquercitrin and ferulic acids were dominant phenolic compounds [
7]. Thus, Ukrainian raw materials have a similar qualitative composition but differ in dominant substances.
In the analysis of steroids, a total of 54 substances were detected, of which 9 steroids were identified: stigmasterol, pregna-5,17-dien-3-ol, ergosterol, 3β-steariloxy-ers-12-ene, stigmaster-5,22-dien-3-ol acetate, β-sitosterol, olean-12-ene, 3β-methoxy-5-cholestene, and traces of cholesterol. Stigmasterol was found to be the most concentrated. The steroid composition is quite well studied for this genus, so the results correspond to the previous research [
5].
As shown in
Table 2, 10 compounds were identified and quantified in the
A. reptans herb extracts, including 1 ketone, 2 alcohols, and 1 terpenoid compound. The alcohols and their derivatives included 3-heptanol and a-terpineol; the ketone identified was 2-heptanone. Among sesquiterpenes and their derivatives were α-bergamotene and hexahydrofarnesyl acetone; among aliphatic sesquiterpenes was farnesene; and among terpenoids was a-linalool. The extracts also contained acyclic triterpenes such as squalene and esters like methyl linoleate and linoleic acid. Other research has shown that teupolioside, martinoside, verbascoside, and isoverbascoside are the major chemical constituents among phenylproponoids, and teupolioside is the chemical marker of the
A. reptans extract, and major monoterpenoids include three iridoid glycosides: harpagide, 8-O-acetylharpagide and reptoside [
35]. Thus, our results extend the data on the chemical composition of the volatile fraction.
The qualitative composition and quantitative content of amino acids in the extracts of
A. reptans herb were studied for the first time. Sixteen amino acids were identified, including 7 essential amino acids: threonine, valine, methionine, isoleucine, leucine, phenylalanine, and lysine, as well as 1 conditionally essential amino acid, histidine. The identified amino acids included 10 monoaminomonocarboxylic acids: alanine, valine, glycine, isoleucine, leucine, methionine, serine, threonine, phenylalanine, and cysteine; 2 monoaminodicarboxylic acids: aspartic and glutamic acids; 2 diamino monocarboxylic acids: arginine and lysine; and 2 heterocyclic amino acids: histidine and tryptophan. According to experimental research data (
Table 3), glutamic acid, aspartic acid, arginine, leucine, serine, valine, and glycine predominated in
A. reptans herb extracts.
The extracts of
A. reptans herb are classified as practically non-toxic (toxicity class V) when administered intragastrically (LD
50 > 5000 mg/kg) according to the classification by K.K. Sidorov [
32].
The simultaneous administration of the extracts and the hepatotoxic poison led to a reduction (
Table 4) in the content of TBA-reactive substances by 1.58, 1.56, and 1.55 times, respectively, and a decrease in ALT activity by 1.52, 1.32, and 1.25 times in the blood serum of the experimental animals and by 1.88, 2.11, and 1.87 times, respectively, in the liver homogenate compared to the control group of animals.
The destruction of cell membrane components (
Table 4) resulted in the development of a pronounced cytolytic syndrome, as indicated by a 5.7-fold increase in ALT activity in the blood serum. The development of acute toxic hepatitis was characterized by an intensification of peroxide catabolic transformations, as evidenced by a 1.7 and 2.3-fold increase in the content of TBA-reactive substances in the blood serum and liver homogenate of the control group animals, respectively, compared to the indicators of intact animals.
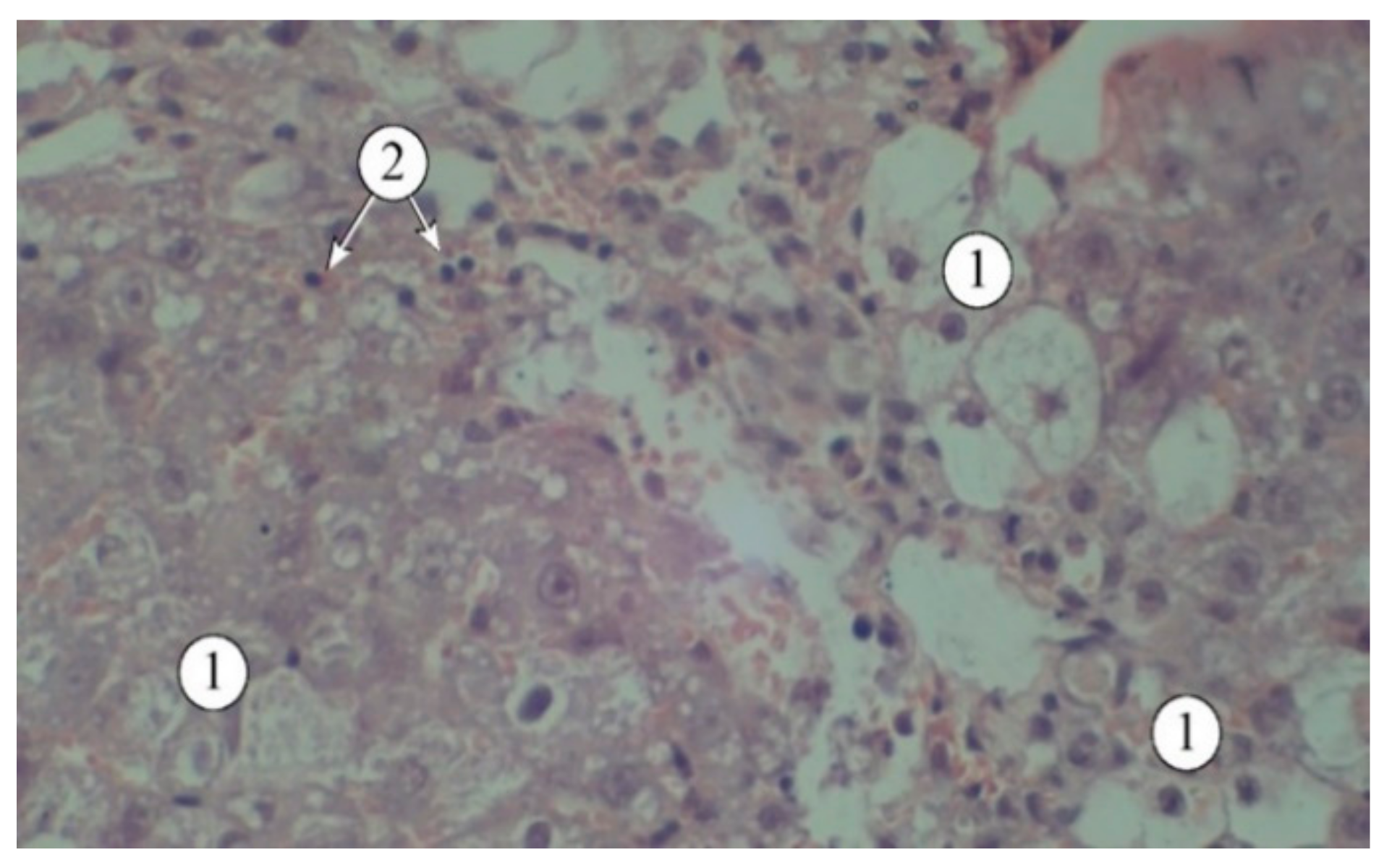
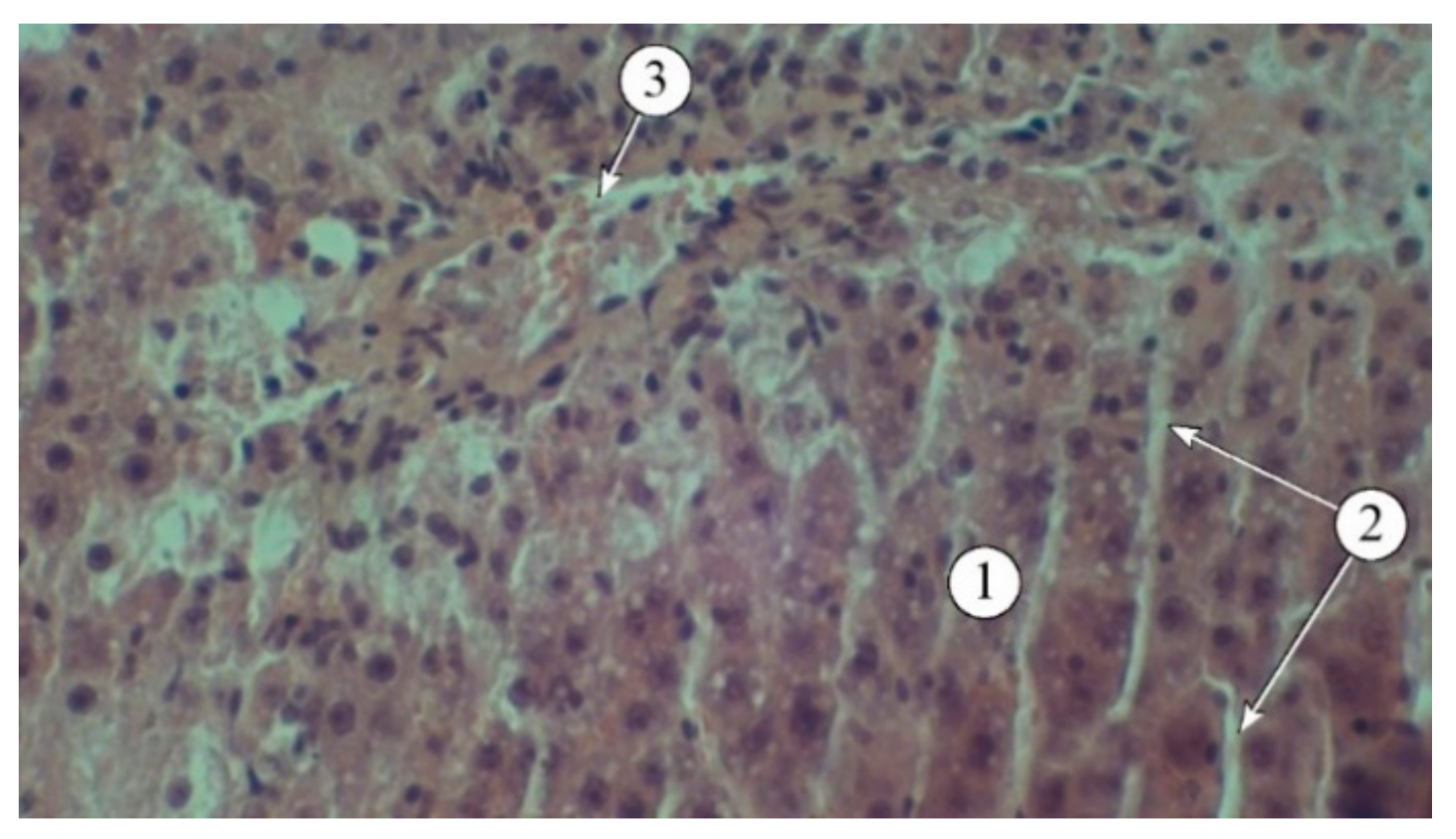
The results of the studies (
Figure 4) indicate that a single administration of carbon tetrachloride was accompanied by the development of acute toxic liver damage, with a loss of structural integrity, disorganized hepatic plates, and the portal triad not being visualized. The interlobular spaces were expanded and deformed, with pronounced lymphocytic infiltration. The borders of hepatocytes were blurred, and in many fields of view, the nuclei were not visible. There was a pronounced macro- and microvesicular component in the cytoplasm of hepatocytes in the peripheral zone.
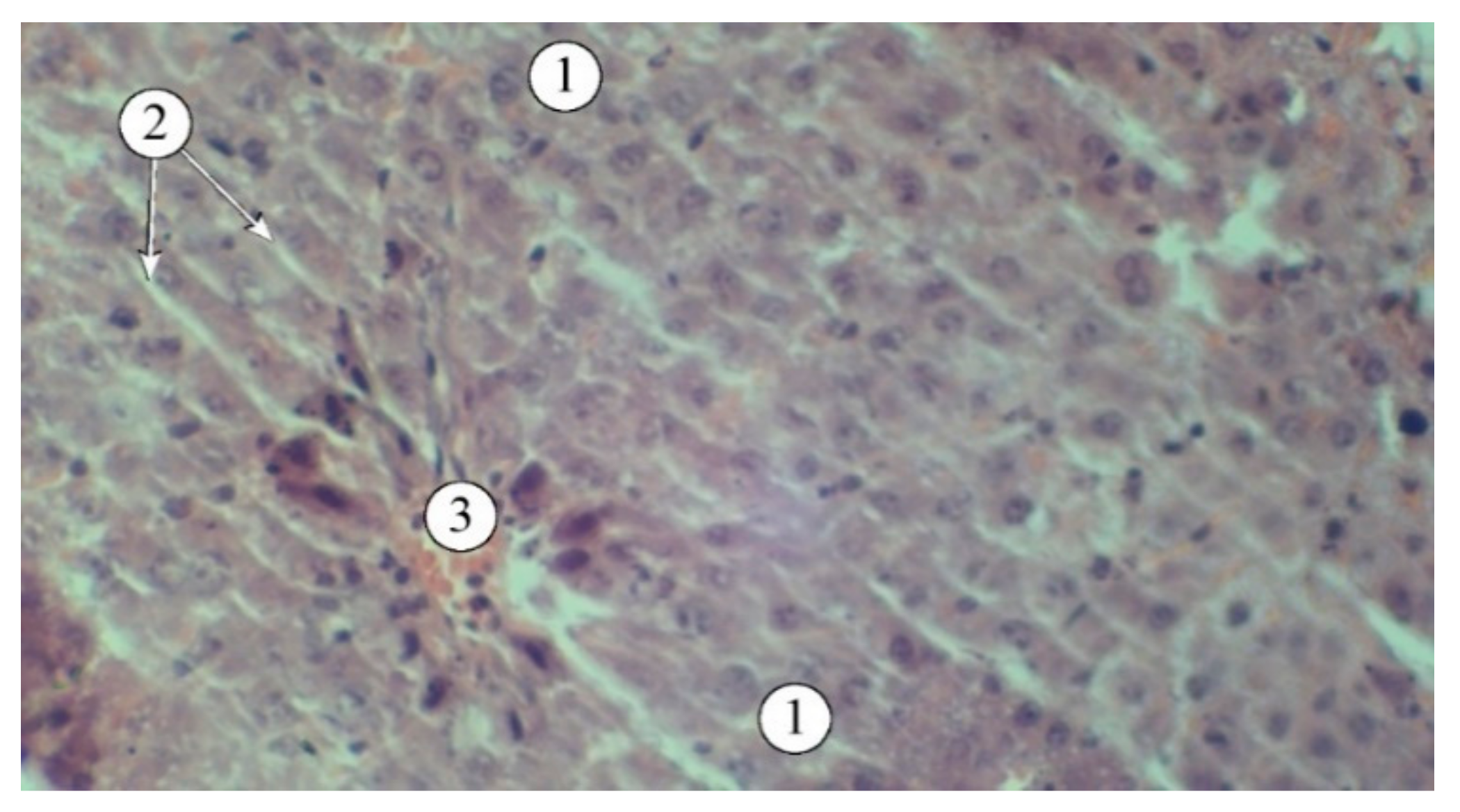
The use of extracts AR1, AR2, AR3, and the comparison drug "Silibor" in the context of experimental hepatitis was associated with a noticeable reduction in pathological manifestations (
Figure 5). The best results were observed in animals that received AR2; the histostructure of the liver was characterized by the organization of hepatic plates and visualization of the components of the portal triad. Hepatocytes in the peripheral zone had a polygonal shape, with their cytoplasm filled with fine-dispersed content and uniformly filled with small vesicles. The intermediate and central zones of the liver lobule demonstrated structured hepatocytes and sinusoids.
In the liver of animals that received the "Silibor" preparation, light-optical examination revealed hepatic plates that were radially oriented towards the central vein and composed of two rows of hepatocytes. The sinusoids appeared as optically translucent slits, without infiltration. The cytoplasm of the hepatocytes was finely granular, with rounded nuclei and visible nucleoli. Individual dystrophically altered hepatocytes were observed in certain fields of view, displaying moderately hypochromatic cytoplasm and nuclei (
Figure 6).
The results obtained from the conducted studies indicate that A. reptans herb extracts exhibit a pronounced hepatoprotective activity in cases of acute toxic liver damage by suppressing peroxide destructive processes and reducing the development of cytolysis syndrome, and they are practically not inferior to the hepatoprotective action of the comparison drug "Silibor." The hepatoprotective activity of A. reptans herb extracts has been studied for the first time.
The study results (
Table 5) show that the anti-exudative activity of AR1, AR3 extracts, and quercetin gradually manifested and reached its maximum within 4 hours from the start of the experiment, indicating a moderate effect of these substances on the suppression of all inflammation mediators, especially prostaglandins. The anti-inflammatory effect is minor and has no pharmacological significance, as a level of pharmacological activity of at least 20% is considered significant for the experimental study of anti-inflammatory agents [
36]. The activity of the AR2 extract increased within 2 hours during the release period of early inflammation mediators (kinin and histamine), confirming the presence of identified polyphenolic compounds with antihistaminic and polyoxygenase activity. The results indicate that, after 2 hours, the AR2 extract exceeded the anti-exudative effect of the reference drug quercetin by more than 1.2 times and maintained this effect for 2 hours. The effect of diclofenac sodium manifested immediately, increased significantly within 2 hours, and reached its maximum 4 hours after the start of the experiment, indicating significant inhibition of all groups of inflammation mediators. In the control group of untreated animals, oedema increased within 4 hours. The study of the anti-inflammatory activity of the tested extracts established that the AR2 extract at a dose of 50 mg/kg exceeded the anti-exudative effect of the reference drug quercetin (anti-exudative effect – 20.52%) after 2 hours. Previously in vivo and in vitro studies have shown that the representatives of
Ajuga L. genus inhibit inflammatory responses by suppressing inflammatory factors (cyclooxygenase-1 (COX-1), cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS), nitric oxide (NO), interleukin 8 (IL-8), interleukin 6 (IL-6), and tumour necrosis factor-α (TNF-α) [
5,
37]. Such data is available for some
Ajuga species but lacks them for
A. reptans. The best anti-oxidative and anti-inflammatory activity was observed for
the A. reptans 100 mg dw/mL extract when compared with diclofenac [
7], but in our research the effective therapeutic dose was 50 mg/kg.
The obtained results (
Table 6) indicate that the local application of
A. reptans extracts (using a gauze pad soaked in the tested extracts directly on the wound surface immediately after the wound was made), significantly decreased bleeding time compared to the control group of animals. The most significant reduction in bleeding was caused by applying the AR1 extract, reducing bleeding time by 40.59%, while the hemostatic activity of the "Rotokan" preparation reduced it by 52.61% compared to the control group. Our conducted studies indicate that
A. reptans extracts have a local hemostatic effect. The study's results on the dynamics of the wound healing process with the application of
A. reptans extracts (
Table 7) show that from the 9th day of the local application of the AR2 extract and the comparison drug "Rotokan," complete wound healing occurred. There was no scientific conformation of the hemostatic and wound healing activity of
A. reptans extracts, although, in folk medicine, it is commonly recommended in these cases [
5,
14,
38].
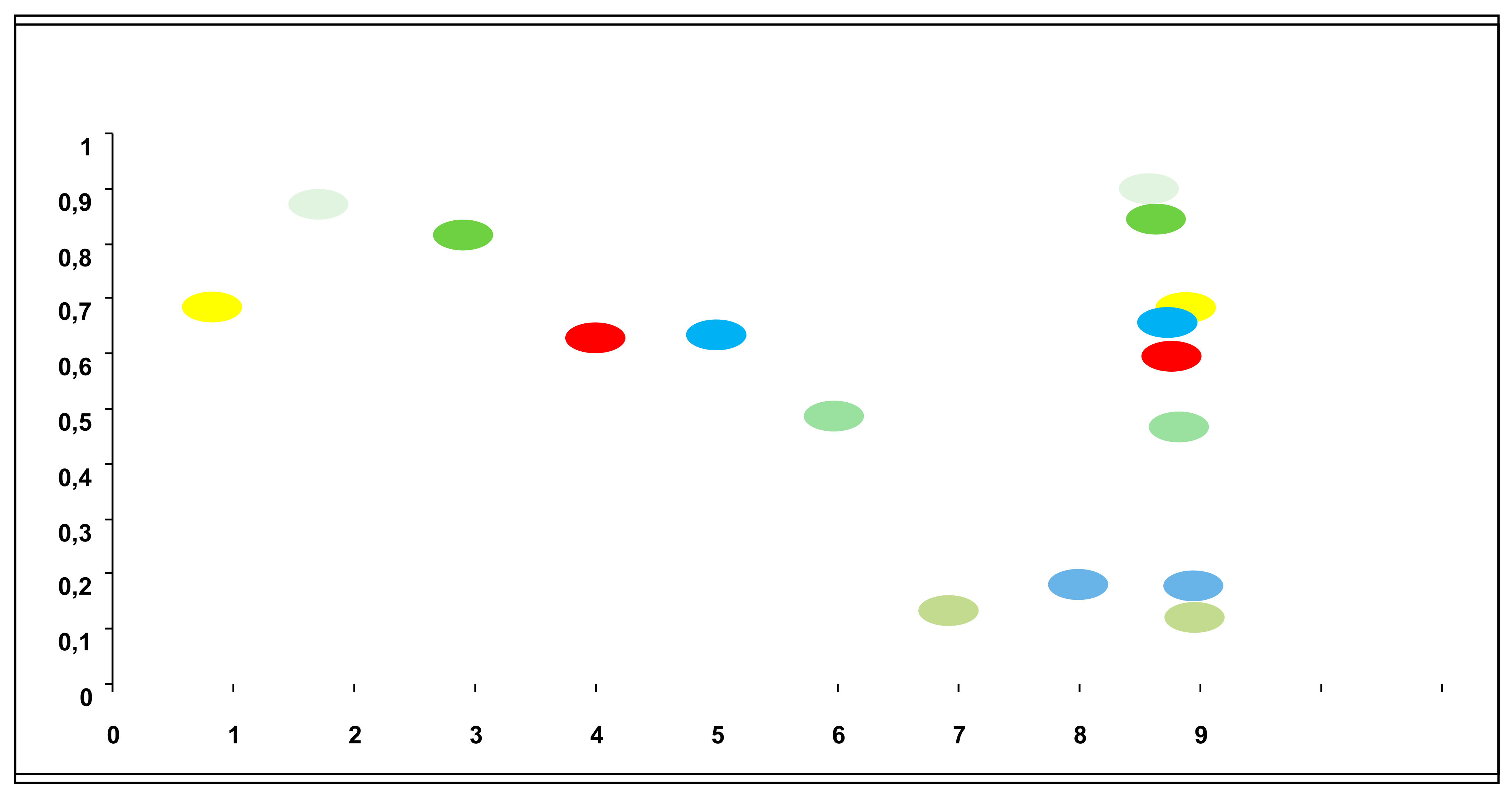
The conducted studies established that A. reptans herb extracts can inhibit the growth of microorganisms to varying degrees, depending on the ethanol concentration used as an extraction agent.
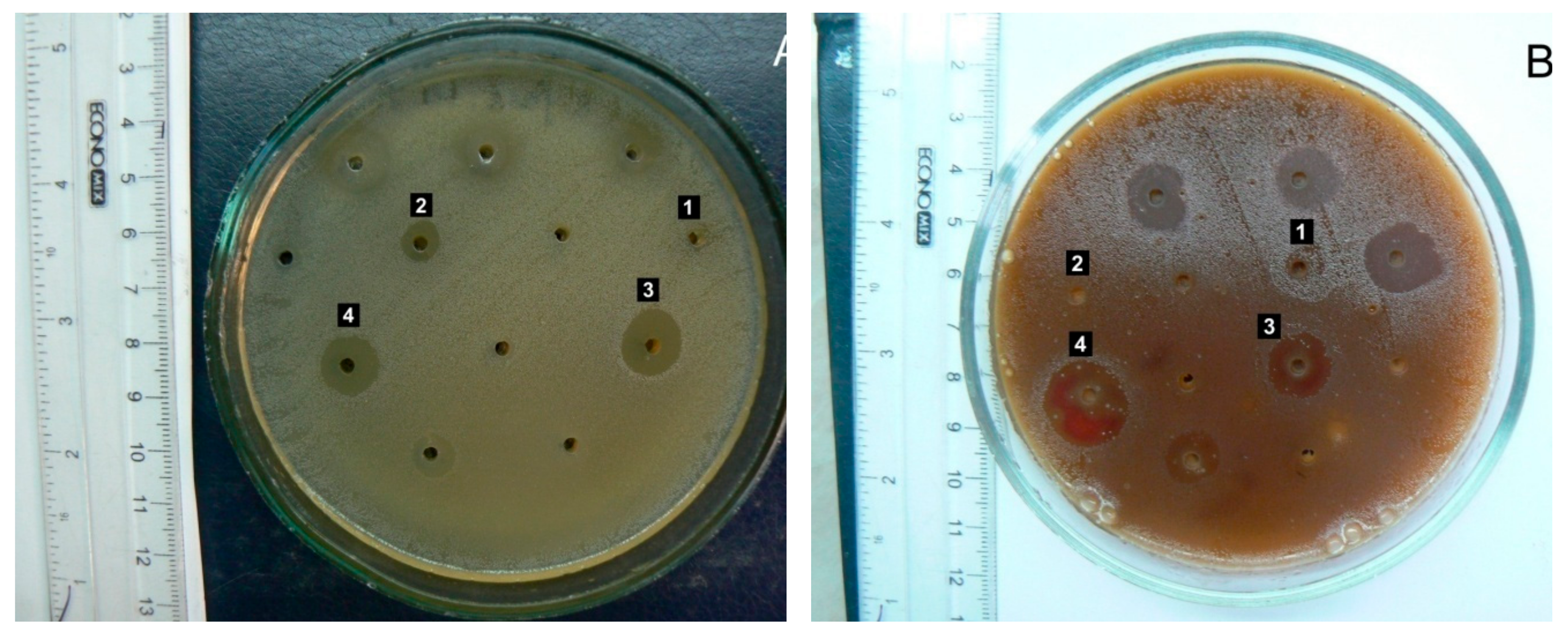
A. reptans extracts demonstrated higher activity against gram-positive microorganisms, with streptococci being particularly sensitive. The AR3 herb extract (extraction agent: 70% ethanol) caused significant inhibition of the growth of pathogenic β-hemolytic streptococci of groups A and G – causative agents of tonsillitis and angina (
Figure 3), as well as β-hemolytic streptococcus group B – a causative agent of inflammatory processes in the female external genital organs. Conditionally pathogenic α-hemolytic streptococci of the oral microflora,
St. oralis,
St. sanguinis, and
St. gordonii (which can cause purulent-inflammatory processes of the oral mucosa and periodontal tissues in dental practice), also showed high sensitivity to the AR3 extract. The growth of
S. sanguinis and
St. oralis was also notably inhibited by the AR2 extract. Regarding the primary causative agent of bacterial respiratory infections (bronchitis, otitis, sinusitis), pneumococcus
St. pneumonia, the AR2 and AR3 extracts exhibited a bacteriostatic effect. Therefore, it can be suggested that
A. reptans herb extracts have potential use in treating streptococcal infections in dental, pediatric, and ENT practices.
Enterococcus faecalis, a common causative agent of urological and wound infections, demonstrated quite high sensitivity to AR3. This result deserves special attention due to enterococcus's high natural resistance to most antibiotics groups.
The most common causative agents of purulent-inflammatory processes, staphylococci, were significantly less sensitive to A. reptans herb extracts than streptococci. S. aureus and S. haemolyticus exhibited weak sensitivity to all tested preparations.
Gram-negative bacteria generally showed significantly lower sensitivity to the biologically active compounds of A. reptans extracts. Normal E. coli was found to be completely insensitive to them. However, there was a tendency for AR3 to inhibit the growth of E. fergusonii, which has reduced enzymatic activity, as well as Citrobacter and representatives of the putrefactive intestinal microflora Providencia and Morganella. Therefore, the inclusion of A. reptans herb as an auxiliary component can be considered when developing herbal mixtures and complex herbal remedies for treating mild forms of intestinal dysbiosis.
The antibacterial effects of the essential oil isolated from the aerial part of
A. pseudoiva have been tested before against Gram-positive and Gram-negative bacteria in vitro, including
B. subtilis,
B. cereus,
S. aureus,
E. coli,
E. faecalis,
K. pneumonia,
P. aeruginosa,
S. typhimurium,
L. monocytogenes, and
E. faecium [
5,
39]. The ethanol and methanol extracts from the aerial parts of
A. laxmannii have been shown to exhibit antibacterial activity against
P. aeruginosa,
L. monocytogenes,
E. coli, and
S. typhimurium [
7]. Thus, the results of the antibacterial effects of
A. reptans herb extracts provide new knowledges.
The
A. reptans herb extracts showed weak fungistatic activity against most types of
Candida yeast-like fungi. Previously, it was also shown that
A. reptans extracts are active against Canada spp. and
E. coli [
34,
35].
During the conducted studies, an important pattern was established: the antimicrobial activity of A. reptans herb extracts increases proportionally with the increase in ethanol concentration in the extracting solution. This may indicate the predominantly hydrophobic properties of biologically active compounds that provide the antimicrobial activity of the extracts.