Submitted:
08 October 2024
Posted:
08 October 2024
You are already at the latest version
Abstract
Formaldehyde is generated via the biochemical and chemical pathways, and it widely exists in foods. Its chronic exposure causes carcinogenic, neurotoxic, reproductive, allergic, immunological, genetic and respiratory toxicities. However, food-sourced formaldehyde is usually neglected due to its rapid metabolism after intake. Compared with its residual level in processed foods, a large proportion of formaldehyde disappears because of its high reactivity. Nevertheless, its elimination reaction mechanism and the potential consequences remain unclear. In this review, the disappearing pathway of formaldehyde from two aspects, the formation of formaldehyde-derived toxins and aromas, was reviewed. This review article may help food scientists take appropriate strategies to use the beneficial reactions of formaldehyde or divert its harmful effects to beneficial ones.
Keywords:
1. Introduction
2. Toxic Effects of Formaldehyde
2.1. Acute Toxication
2.2. Chronic Toxication
2.2.1. Carcinogenesis
2.2.2. Genotoxicity and Mutagenicity
2.2.3. Respiratory and Lung Toxicity
2.2.4. Neurotoxicity
2.2.5. Reproductive and Developmental Toxicity
3. Formaldehyde-Derived Harmful Compounds
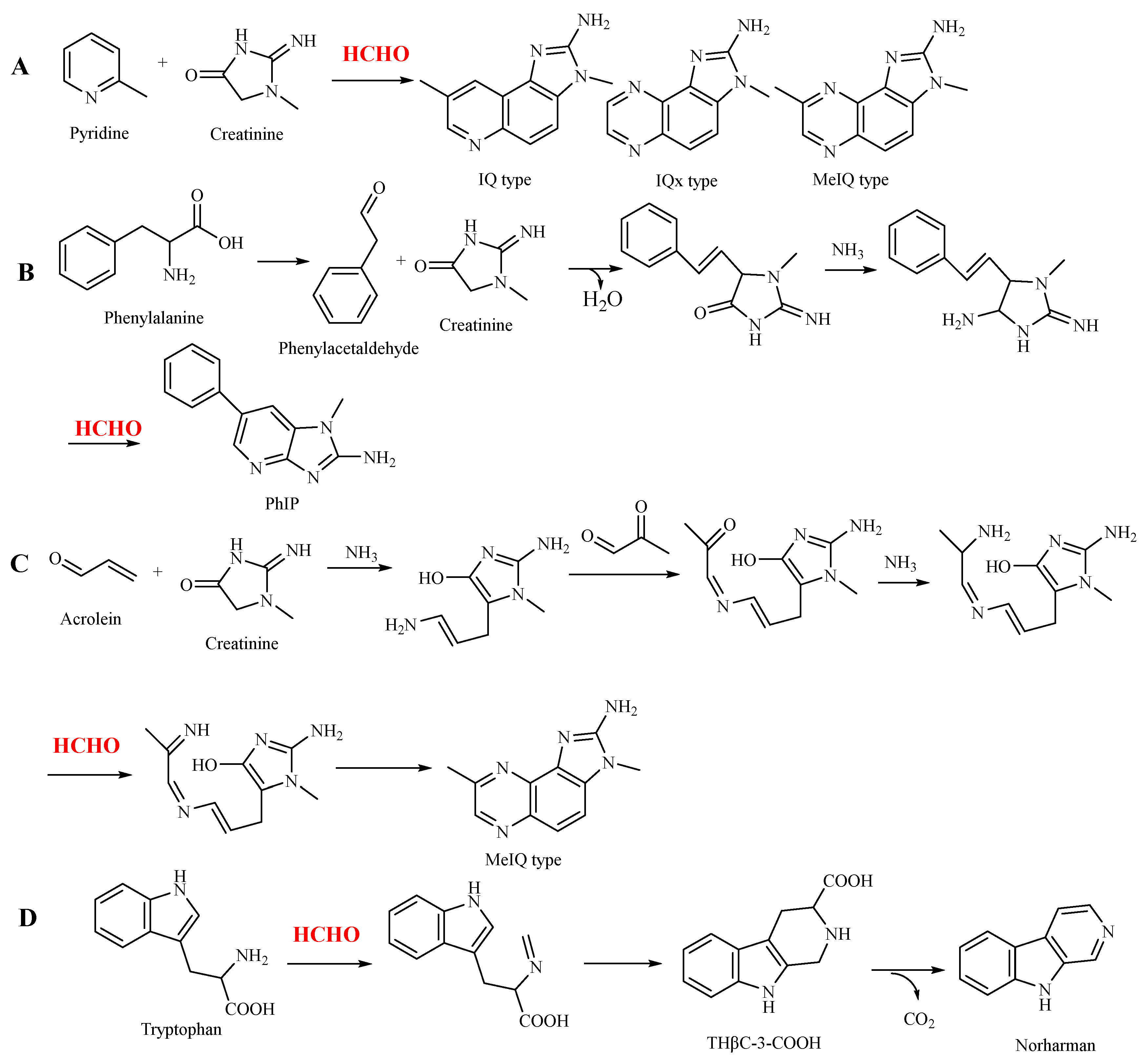
3.1. Participation of Formaldehyde in the Formation of Heterocyclic Aromatic Amines
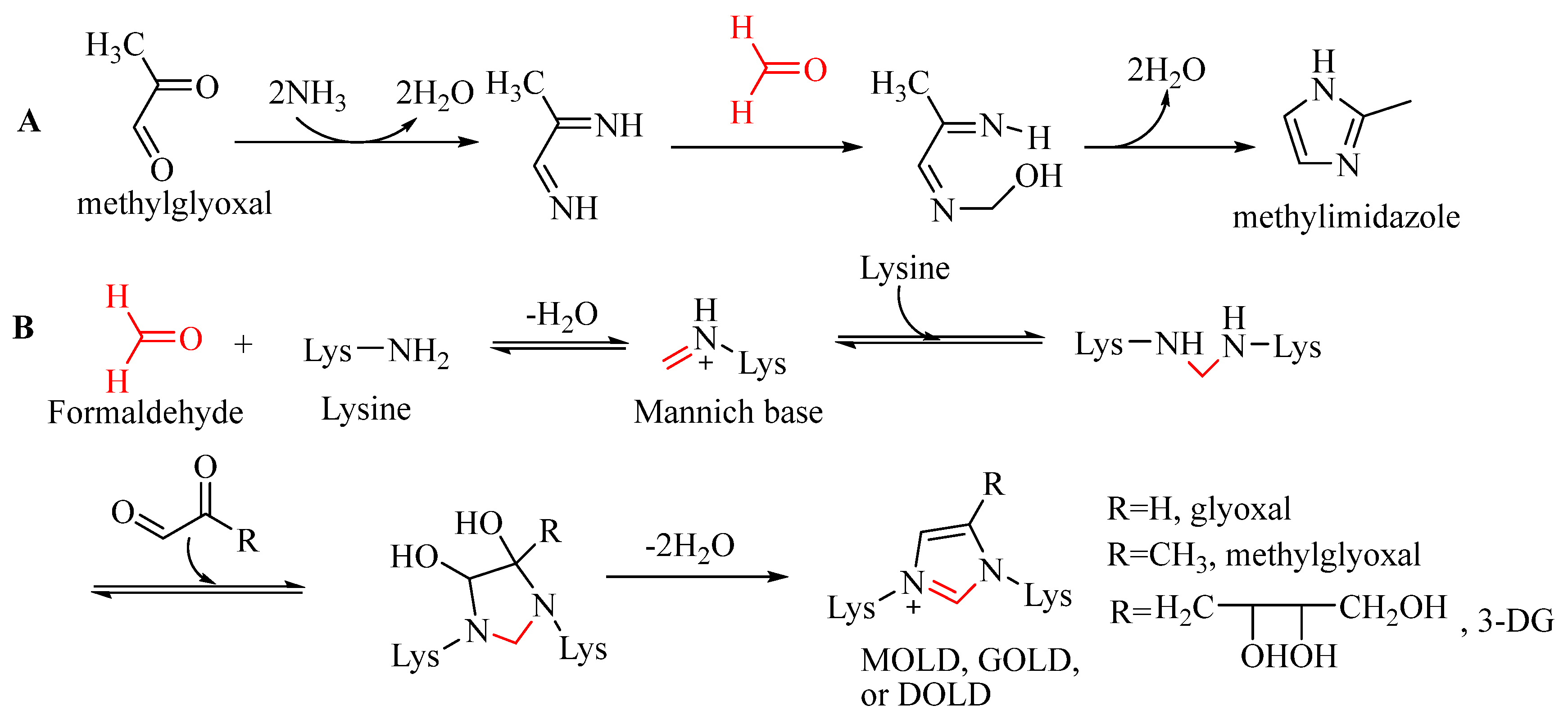
3.2. Participation of Formaldehyde in the Formation of Methylimidazole and Imidazole-Type Advanced Glycation End Products
3.3. N-Nitrosamines
4. Flavour Formation in Foods
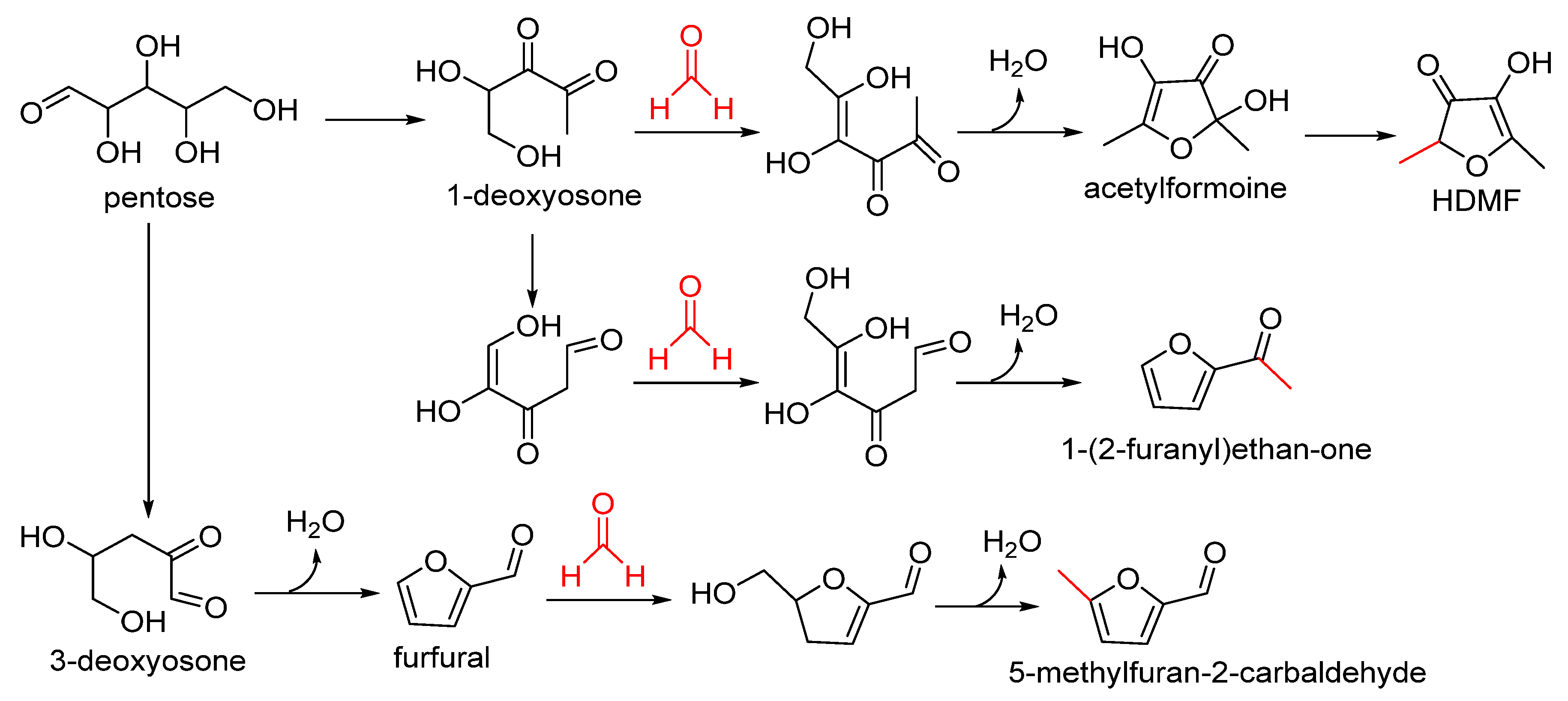
2.1. Participation of Furanone Formation
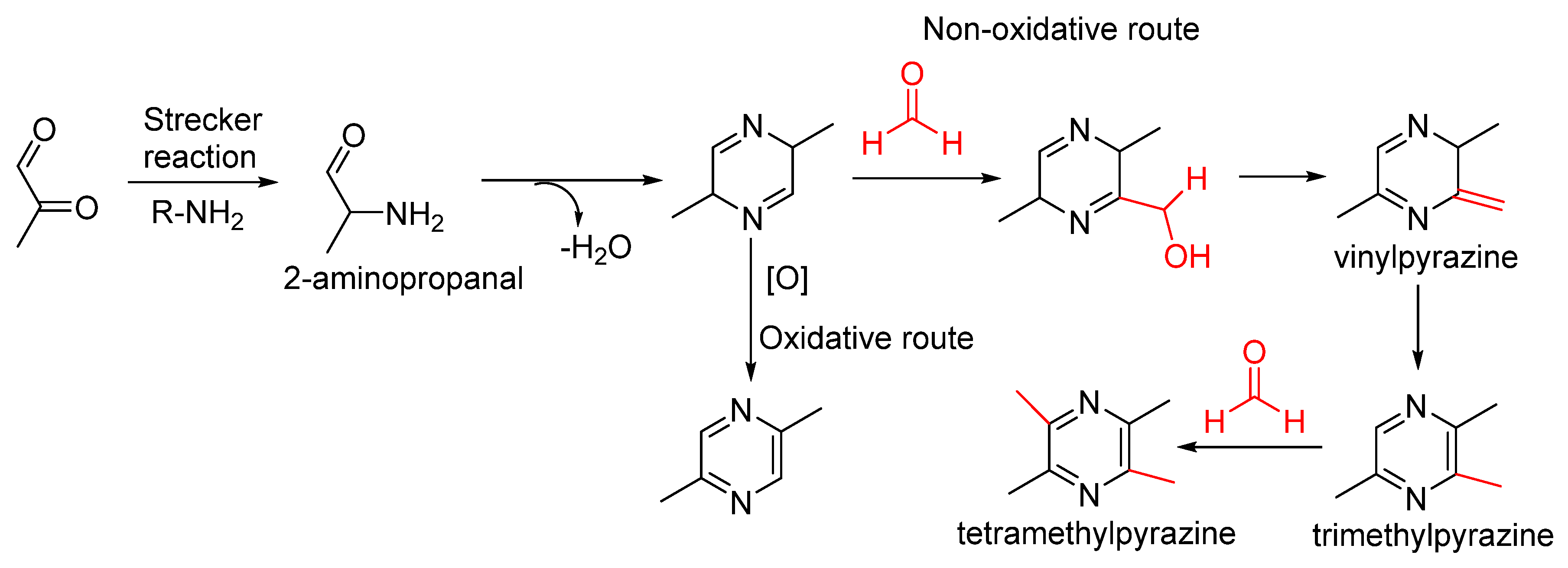
2.2. Formation of Pyrazines
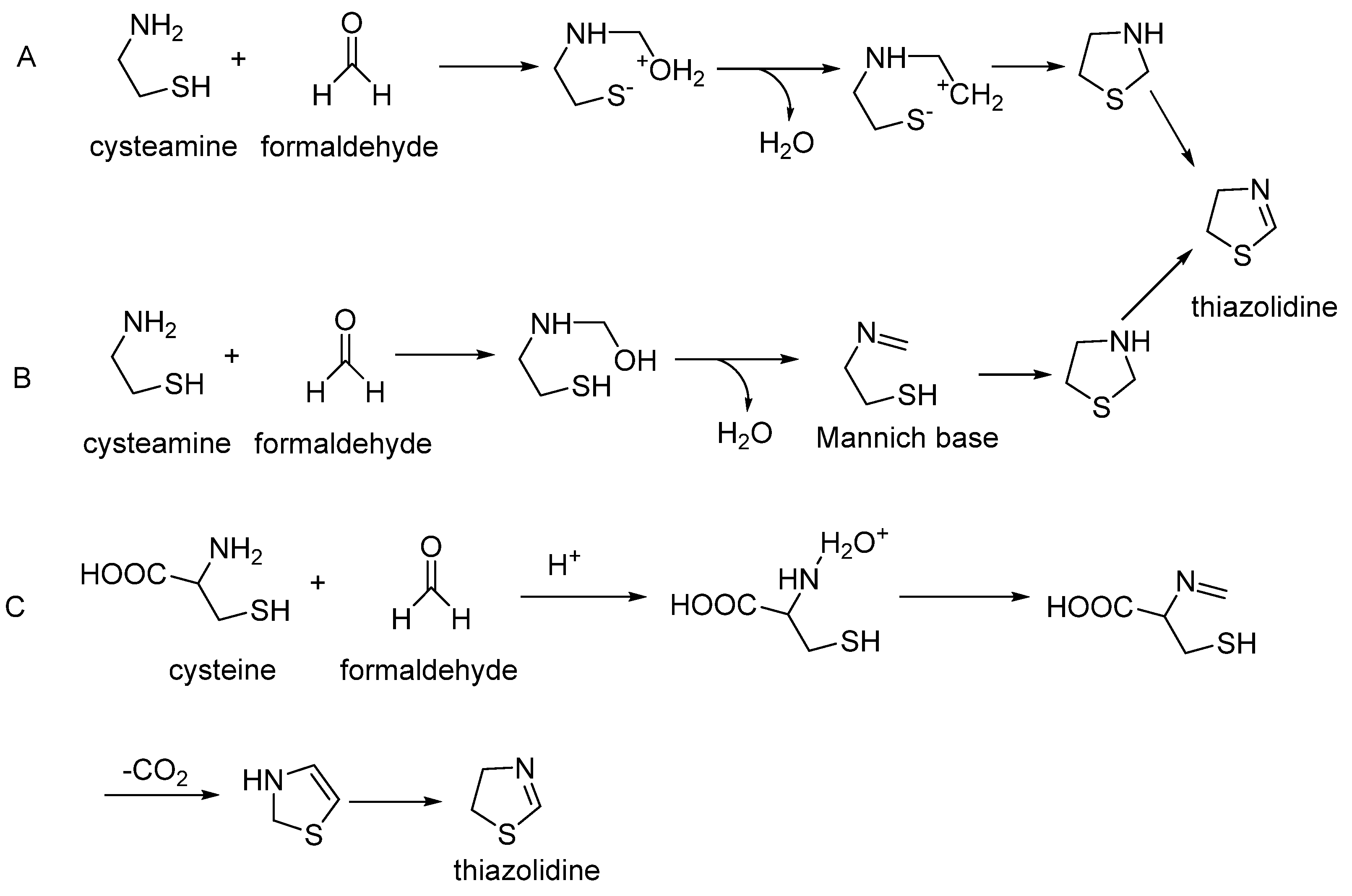
2.3. Thiazoles
5. Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Food Safety Authority. Endogenous Formaldehyde Turnover in Humans Compared with Exogenous Contribution from Food Sources. EFSA J. 2014, 12. [Google Scholar]
- Xiao, R.; He, R. Metabolism of Formaldehyde In Vivo. In Formaldehyde and Cognition; Springer Netherlands: Dordrecht, 2017; pp 21–46. [Google Scholar]
- Toh, J.D.W.; Crossley, S.W.M.; Bruemmer, K.J.; Ge, E.J.; He, D.; Iovan, D.A.; Chang, C.J. Distinct RNA N-Demethylation Pathways Catalyzed by Nonheme Iron Alkbh5 and FTO Enzymes Enable Regulation of Formaldehyde Release Rates. Proc. Natl. Acad. Sci. 2020, 117, 25284–25292. [Google Scholar] [CrossRef] [PubMed]
- Dorokhov, Y.L.; Sheshukova, E.V.; Komarova, T.V. Methanol in Plant Life. Front. Plant Sci. 2018, 9, 1623. [Google Scholar] [CrossRef]
- Reingruber, H.; Pontel, L.B. Formaldehyde Metabolism and Its Impact on Human Health. Curr. Opin. Toxicol. 2018, 9, 28–34. [Google Scholar] [CrossRef]
- 6 Li, Y.; Ou, J.; Huang, C.; Liu, F.; Ou, S.; Zheng, J. Chemistry of Formation and Elimination of Formaldehyde in Foods. Trends Food Sci. Technol. 2023, 139, 104134. [Google Scholar] [CrossRef]
- Cheng, J.; Zhang, L.; Tang, Y.; & Li, Z. The Toxicity of Continuous Long-Term Low-Dose Formaldehyde Inhalation in Mice. Immunopharmacol Immunotoxicol. 2016, 38, 495–501. [Google Scholar] [CrossRef]
- Fappiano, L.; Carriera, F.; Iannone, A.; Notardonato, I.; & Avino, P. A Review on Recent Sensing Methods for Determining Formaldehyde in Agri-Food Chain: A Comparison with the Conventional Analytical Approaches. Foods, 2022, 11, 1351. [Google Scholar] [CrossRef]
- Dhareshwar, S.S.; Stella, V.J. Your Prodrug Releases Formaldehyde: Should You Be Concerned? No! J. Pharm. Sci. 2008, 97, 4184–4193. [Google Scholar] [CrossRef] [PubMed]
- Hagos, S.; Gebeya, D.; & Teklay, A. Effects of Formalin Toxicity among Preclinical I Medical Students, Adigrat University, Ethiopia. Cough, 2018, 42, 43–8. [Google Scholar] [CrossRef]
- Augenreich, M.; Stickford, J.; Stute, N.; Koontz, L.; Cope, J.; Bennett, C.; & Ratchford, S.M. Vascular Dysfunction and Oxidative Stress Caused by Acute Formaldehyde Exposure in Female Adults. Am J Physiol Heart Circ Physiol. 2020, 319, H1369–H1379. [Google Scholar] [CrossRef]
- Arici, S.; Karaman, S.; Dogru, S.; Cayli, S.; Arici, A.; Suren, M.; Karaman, T.; & Kaya, Z. Central Nervous System Toxicity after Acute Oral Formaldehyde Exposure in Rabbits: An experimental study. Hum Exp Toxicol 2014, 33(11),1141-1149.
- Zhang, L.; Li, Y.; Wang, L.; Zhang, S.; Zhang, G.; Zuo, M. . & Cong, B. A Fatal Case of Accidental Oral Formaldehyde Poisoning and Its Pathomorphological Characteristics. Int J Legal Med. 2022, 136, 1303–1307. [Google Scholar]
- Metz, B.; Kersten, G.F.; Hoogerhout, P.; Brugghe, H.F.; Timmermans, H.A.; De Jong, A.D. . & Jiskoot, W. Identification of Formaldehyde-Induced Modifications in Proteins: Reactions With Model Peptides. J Biol Chem. 2004, 279, 6235–6243. [Google Scholar] [PubMed]
- Tayri-Wilk, T.; Slavin, M.; Zamel, J.; Blass, A.; Cohen, S.; Motzik, A. . & Kalisman, N. Mass Spectrometry Reveals the Chemistry of Formaldehyde Cross-Linking in Structured Proteins. Nat Commun. 2020, 11, 3128. [Google Scholar]
- Bernardini, L.; Barbosa, E.; Charao, M.F.; & Brucker, N. Formaldehyde Toxicity Reports from In Vitro and In Vivo Studies: A Review and Updated Data. Drug Chem Toxicol. 2022, 45, 972–984. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Jahan, S.A.; & Lee, J.T. T. (2011). Exposure to Formaldehyde and Its Potential Human Health Hazards. J Environ Sci Health. C. 2011, 29, 277–299. [Google Scholar] [CrossRef]
- Protano, C.; Buomprisco, G.; Cammalleri, V.; Pocino, R.N.; Marotta, D.; Simonazzi, S. . & Vitali, M. The Carcinogenic Effects of Formaldehyde Occupational Exposure: A Systematic Review. Cancers, 2021, 14, 165. [Google Scholar]
- Kang, D.S.; Kim, H.S.; Jung, J.H.; Lee, C.M.; Ahn, Y.S.; & Seo, Y.R. R. Formaldehyde Exposure and Leukemia Risk: A Comprehensive Review and Network-Based Toxicogenomic Approach. Genes and Environ. 2021, 43, 1–10. [Google Scholar] [CrossRef]
- Umansky, C.; Morellato, A.E.; Rieckher, M.; Scheidegger, M.A.; Martinefski, M.R.; Fernández, G.A. . & Pontel, L.B. Endogenous Formaldehyde Scavenges Cellular Glutathione Resulting in Redox Disruption and Cytotoxicity. Nat commun. 2022, 13, 745. [Google Scholar] [PubMed]
- Ghelli, F.; Cocchi, E.; Bellisario, V.; Buglisi, M.; Squillacioti, G.; Santovito, A. Bono. R. The Formation of SCEs as an Effect of Occupational Exposure to Formaldehyde. Arch Toxikol. 2022, 96, 1101–1108. [Google Scholar] [CrossRef]
- Leng, J.; Liu, C.W.; Hartwell, H.J.; Yu, R.; Lai, Y.; Bodnar, W.M. . & Swenberg, J.A. Evaluation of Inhaled Low-Dose Formaldehyde-Induced DNA Adducts and DNA–Protein Cross-Links by Liquid Chromatography–Tandem Mass Spectrometry. Arch Toxikol. 2019, 93, 763–773. [Google Scholar]
- Chen, P.W.; Kuo, T.C.; Liu, Z.S.; & Lu, H.F. F. Assessment of the Mutagenicity of Two Common Indoor Air Pollutants, Formaldehyde and Toluene. Indoor Air. 2021, 31, 1353–1363. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.A.; Afzal, M.; Goyal, A.; Gupta, G.; Thapa, R.; Kazmi, I. ;... & Dua, K. The Impact of Formaldehyde Exposure on Lung Inflammatory Disorders: Insights Into Asthma, Bronchitis, and Pulmonary Fibrosis. Chem Biol Interact, 2024; 111002. [Google Scholar]
- Squillacioti, G.; Bellisario, V.; Ghelli, F.; & Bono, R. Occupational Exposure to Formaldehyde and Oxidative Stress in Italian Workers. Eur J Public Health. 2021, 31, 584. [Google Scholar] [CrossRef]
- [26] Huang, J.W.; Lu, Y.; Zhang, B.; Yang, S.P.; Zhang, Q.; Cui, H.Y.; Lu, X.X.; Zhao, Y.; Yang, X.; & Li, R. Antagonistic Effffect of Epigallocatechin-3-Gallate on Neurotoxicity Induced by Formaldehyde. Toxicol. 2019, 412, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Tulpule, K.; & Dringen, R. Formaldehyde in Brain: an Overlooked Player in Neurodegeneration? J Neurochem. 2013, 127, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Li, B.; Ismail, N.; Smith, K.; Li, T.; Dai, R.; & Deng, Y. Neurotoxicity and Underlying Mechanisms of Endogenous Neurotoxins. Int J Mol Sci. 2021, 22, 12805. [Google Scholar] [CrossRef]
- Rana, I.; Rieswijk, L.; Steinmaus, C.; & Zhang, L.P. P. Formaldehyde and Brain Disorders: A Meta Analysis and Bioinformatics Approach. Neurotox Res. 2021, 39, 924–948. [Google Scholar] [CrossRef]
- Kou, Y.; Zhao, H.; Cui, D. , Han, H.; & Tong, Z. Formaldehyde Toxicity in Age-Related Neurological Dementia. Ageing Res Rev. 2022, 73, 101512. [Google Scholar]
- Aydin, D.D.; Taşkınalp, O.; Uluçam, E.; Karadağ, H.; Özfidan, G.K.; Topuz, R.; & Ersoy, O. The Effect of Acute and Chronic Formaldehyde Exposure on Learning and Memory in Male and Female Rats. Int J Neurosci. 2023, 1–9. [Google Scholar] [CrossRef]
- Duong, A.; Steinmaus, C.; McHale, C.M.; Vaughan, C.P.; & Zhang, L. Reproductive and Developmental Toxicity of Formaldehyde: A Systematic Review. Mutat Res Rev Mutat Res. 2011, 728, 118–138. [Google Scholar] [CrossRef]
- Treesh, S.A.; Aburawi, S.M.; Elghedamsi, M.T.; Jaafari, H.A.E.; Alzowam, R.; Shibani, N.; & Khair, N.S. Protective Role of Vitamin C on Histopathological Effect of Formaldehyde on Reproductive System in Female Albino Mice (Histological Study). Int J Adv Res. 2019, 7, 529–540. [Google Scholar] [CrossRef]
- Peng, G.; Zhong, C.; Zhang, Q.; Xie, Y.; & Gong, F. Effect of Formaldehyde on E xpressions of Fas Apoptosis Pathway-Related Genes of Ovary Tissues in Female Rats. Journal of Central South University Medical Sciences, 2010, 35, 341–345. [Google Scholar] [PubMed]
- Xu, W.; Zhang, W.; Zhang, X.; Dong, T.; Zeng, H.; & Fan, Q. Association between Formaldehyde Exposure and Miscarriage in Chinese Women. Medicine, 96, e7146.
- Søgaard, R.; Poulsen, P.B.; Gelardi, R.M.; Geschke, S.; Schwensen, J.F.B.; & Johansen, J.D. Hidden Formaldehyde in Cosmetic Products. Contact Dermatitis. 2024. [Google Scholar] [CrossRef] [PubMed]
- Goossens, A.; & Aerts, O. Contact Allergy to and Allergic Contact Dermatitis from Formaldehyde and Formaldehyde Releasers: A Clinical Review and Update. Contact Dermatitis, 2022, 87, 20–27. [Google Scholar] [CrossRef]
- Zhu, R.; Zhang, G.; Jing, M.; Han, Y.; Li, J.; Zhao, J. . & Chen, P.R. Genetically Encoded Formaldehyde Sensors Inspired by A Protein Intra-Helical Crosslinking Reaction. Nat Commun. 2021, 12, 581. [Google Scholar]
- Nadeem, H.R.; Akhtar, S.; Ismail, T.; Sestili, P.; Lorenzo, J.M.; Ranjha, M.M.A.N. . & Aadil, R.M. Heterocyclic Aromatic Amines in Meat: Formation, Isolation, Risk Assessment, and Inhibitory Effect of Plant Extracts. Foods, 2021, 10, 1466. [Google Scholar]
- Iammarino, M.; Marino, R.; Nardelli, V.; Ingegno, M.; & Albenzio, M. Red Meat Heating Processes, Toxic Compounds Production and Nutritional Parameters Changes: What about Risk–Benefit? Foods, 2024, 13, 445. [Google Scholar] [CrossRef]
- Zamora, R.; Hidalgo, F.J. Formation of Heterocyclic Aromatic Amines with the Structure of Aminoimidazoazarenes in Food Products. Food Chem. 2020, 313, 126128. [Google Scholar] [CrossRef]
- Hidalgo, F.J.; Zamora, R. Carbonyl Chemistry and the Formation of Heterocyclic Aromatic Amines with the Structure of Aminoimidazoazaarene. J. Agric. Food Chem. 2022, 70, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Herraiz, T.; Peña, A.; Salgado, A. Identification, Formation, and Occurrence of Perlolyrine: A β-Carboline Alkaloid with a Furan Moiety in Foods. J. Agric. Food Chem. 2023, 71, 13451–13461. [Google Scholar] [CrossRef]
- Chen, X.; Jia, W.; Zhu, L.; Mao, L.; Zhang, Y. Recent Advances in Heterocyclic Aromatic Amines: An Update on Food Safety and Hazardous Control from Food Processing to Dietary Intake. Compr. Rev. Food Sci. Food Saf. 2020, 19, 124–148. [Google Scholar] [CrossRef]
- Hengel, M.; Shibamoto, T. Carcinogenic 4-Methylimidazole Found in Beverages, Sauces, and Caramel Colors: Chemical Properties, Analysis, and Biological Activities. J. Agric. Food Chem. 2013, 61, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Raters, M.; Elsinghorst, P.W.; Goetze, S.; Dingel, A.; Matissek, R. Determination of 2-Methylimidazole, 4-Methylimidazole, and 2-Acetyl-4-(1,2,3,4-Tetrahydroxybutyl)Imidazole in Licorice Using High-Performance Liquid Chromatography–Tandem Mass Spectrometry Stable-Isotope Dilution Analysis. J. Agric. Food Chem. 2015, 63, 5930–5934. [Google Scholar] [CrossRef]
- Akbari, N.; Shafaroodi, H.; Jahanbakhsh, M.; Sabah, S.; Molaee- Aghaee, E.; Sadighara, P. 4-Methylimidazole, a Carcinogenic Component in Food, Amount, Methods Used for Measurement; a Systematic Review. Food Chem. X 2023, 18, 100739. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Ma, W.; Tang, H.; Wu, X.; Yang, X.; Sun, F. 4-Methylimidazole Exposure Impairs Sperm Mobility by Reducing the Expression of Blood-Testis Barrier Junction Protein in Mouse Testes. Reprod. Biol. 2024, 24, 100928. [Google Scholar] [CrossRef]
- Moon, J.-K.; Shibamoto, T. Formation of Carcinogenic 4-Methylimidazole in Maillard Reaction Systems. J. Agric. Food Chem. 2011, 59, 615–618. [Google Scholar] [CrossRef]
- Jang, H.W.; Jiang, Y.; Hengel, M.; Shibamoto, T. Formation of 4-Methylimidazole and Its Precursors, α-Dicarbonyl Compounds, in Maillard Model Systems. J. Agric. Food Chem. 2013, 61, 6865–6872. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Y.; Fu, L. Dietary Advanced Glycation End-products: Perspectives Linking Food Processing with Health Implications. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2559–2587. [Google Scholar] [CrossRef] [PubMed]
- Eggen, M.D.; Glomb, M.A. Analysis of Glyoxal- and Methylglyoxal-Derived Advanced Glycation End Products during Grilling of Porcine Meat. J. Agric. Food Chem. 2021, 69, 15374–15383. [Google Scholar] [CrossRef]
- Brinkmann, E.; Wells-Knecht, K.J.; Thorpe, S.R.; Baynes, J.W. ChemInform Abstract: Characterization of an Imidazolium Compound Formed by Reaction of Methylglyoxal and Nα-Hippuryllysine. ChemInform 1996, 27, chin–199610265. [Google Scholar] [CrossRef]
- Hu, J.; Jiang, K.; Huang, C.; Zheng, J.; Zhou, H.; Ou, J.; Ou, S. Glycine and Serine Markedly Eliminate Methylglyoxal in the Presence of Formaldehyde via the Formation of Imidazole Salts. Food Chem. 2022, 369, 130952. [Google Scholar] [CrossRef]
- Li, H.; Li, Q.; Wang, Q.; Chen, J.; Xia, W.; & Liao, E. Effects of Inoculating Autochthonous Starter Cultures on Changes of N-Nitrosamines and Their Precursors in Chinese Traditional Fermented Fish during In Vitro Human Digestion. Foods, 2024, 13, 2021. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, I.W.; Dirat, O.; Teasdale, A.; Whiting, M. Potential for the Formation of N -Nitrosamines during the Manufacture of Active Pharmaceutical Ingredients: An Assessment of the Risk Posed by Trace Nitrite in Water. Org. Process Res. Dev. 2020, 24, 1629–1646. [Google Scholar] [CrossRef]
- Eisenbrand, G.; Baum, M.; Cartus, A.T.; Diel, P.; Engel, K.-H.; Engeli, B. .. & Steinberg, P. Salivary Nitrate/Nitrite and Acetaldehyde in Humans: Potential Combination Effects in the Upper Gastrointestinal Tract and Possible Consequences for the in Vivo Formation of N-Nitroso Compounds—a Hypothesis. Arch. Toxicol. 2022, 96, 1905–1914. [Google Scholar]
- Kurechi, T.; Kikugawa, K.; Ozawa, M. Effect of Malondialdehyde on Nitrosamine Formation. Food Cosmet. Toxicol. 1980, 18, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Breider, F.; Barrios, B.; Minakata, D.; Deng, H.; Von Gunten, U. Role of Carbonyl Compounds for N -Nitrosamine Formation during Nitrosation: Kinetics and Mechanisms. Environ. Sci. Technol. 2024, 58, 4792–4801. [Google Scholar] [CrossRef] [PubMed]
- Kamps, J.J.A.G.; Hopkinson, R.J.; Schofield, C.J.; Claridge, T.D.W. How Formaldehyde Reacts with Amino Acids. Commun. Chem. 2019, 2, 126. [Google Scholar] [CrossRef]
- Tricker, A.R.; Kubacki, S.J. Review of the Occurrence and Formation of Non-volatile N -nitroso Compounds in Foods†. Food Addit. Contam. 1992, 9, 39–69. [Google Scholar] [CrossRef]
- Jiang, K.; Huang, C.; Liu, F.; Zheng, J.; Ou, J.; Zhao, D.; Ou, S. Origin and Fate of Acrolein in Foods. Foods 2022, 11, 1976. [Google Scholar] [CrossRef]
- Hong, J.; Song, Y.; Xie, J.; Xie, J.; Chen, Y.; Li, P. . & Yu, Q. Acrolein Promotes Aging and Oxidative Stress via the Stress Response Factor DAF-16/FOXO in Caenorhabditis Elegans. Foods, 2022, 11, 1590. [Google Scholar]
- Folliard, V.; Postole, G.; Devaux, J.F.; Dubois, J.L.; Marra, L.; & Auroux, A. Oxidative Coupling of A Mixture of Bio-Alcohols to Produce A More Sustainable Acrolein: an in Depth Look in the Mechanism Implying Aldehydes Co-Adsorption and Acid/Base Sites. Appl Catal B. 2020, 268, 118421.
- Govindaraju, I.; Sana, M.; Chakraborty, I.; Rahman, M.H.; Biswas, R.; & Mazumder, N. Dietary acrylamide: A Detailed Review on Formation, Detection, Mitigation, and Its Health Impacts. Foods.
- Pérez-Lucas, G.; Navarro, G.; & Navarro, S. Understanding How Chemical Pollutants Arise and Evolve in the Brewing Supply Chain: A Scoping Review. Foods, 2024, 13, 1709. [Google Scholar] [CrossRef] [PubMed]
- Chemistry and Technology of Flavors and Fragrances, 1st ed.; Rowe, D.J., Ed.; Wiley, 2004.
- Xiao, Q.; Huang, Q.; Ho, C.-T. Occurrence, Formation, Stability, and Interaction of 4-Hydroxy-2,5-Dimethyl-3(2H)-Furanone. ACS Food Sci. Technol. 2021, 1, 292–303. [Google Scholar] [CrossRef]
- Blank, I.; Fay, L.B. Formation of 4-Hydroxy-2,5-Dimethyl-3(2 H )-Furanone and 4-Hydroxy-2(or 5)-Ethyl-5(or 2)-Methyl-3(2 H )-Furanone through Maillard Reaction Based on Pentose Sugars. J. Agric. Food Chem. 1996, 44, 531–536. [Google Scholar] [CrossRef]
- Wang, Y.; Rodolfo Juliani, H.; Simon, J.E.; Ho, C.-T. Amino Acid-Dependent Formation Pathways of 2-Acetylfuran and 2,5-Dimethyl-4-Hydroxy-3[2H]-Furanone in the Maillard Reaction. Food Chem. 2009, 115, 233–237. [Google Scholar] [CrossRef]
- Hwang, Young-Sook; Kim, Sang-Hyun; Sin, Tae-Seon; Cho, Jun-Hyun; Lee, In-Seok; Oh, Kwang-Soo. Volatile Flavor Constituents of Cooked Oyster Sauce Prepared from Individually Quick-Frozen Oyster Crassostrea Gigas Extract. Korean J. Fish. Aquat. Sci. 2015, 48, 668–673. [Google Scholar]
- Lu, J.-W.; Lin, C.-Y.; Fang, M. Roasted Fish Reaction Flavor by Plant-Based Ingredients. Food Chem. 2024, 460, 140492. [Google Scholar] [CrossRef]
- Peralta, R.R.; Shimoda, M.; Osajima, Y. Further Identification of Volatile Compounds in Fish Sauce. J. Agric. Food Chem. 1996, 44, 3606–3610. [Google Scholar] [CrossRef]
- Sha, K.; Zhang, Z.-J.; Sun, B.-Z.; Li, H.-P.; Song, H.-L.; Lang, Y.-M.; Lei, Y.-H.; Li, H.-D.; Zhang, Y. Investigation of Physicochemical and Textural Characteristics and Volatile Compounds of Kazakh Dry-Cured Beef. Food Sci. Technol. Res. 2017, 23, 375–383. [Google Scholar] [CrossRef]
- Zhan, H.; Cui, H.; Yu, J.; Hayat, K.; Wu, X.; Zhang, X.; Ho, C.-T. Characteristic Flavor Formation of Thermally Processed N-(1-Deoxy-α-d-Ribulos-1-Yl)-Glycine: Decisive Role of Additional Amino Acids and Promotional Effect of Glyoxal. Food Chem. 2022, 371, 131137. [Google Scholar] [CrossRef]
- Cai, W.; Feng, T.; Yao, L.; Sun, M.; Song, S.; Wang, H.; Yu, C.; Liu, Q. Characterisation of Differential Aroma Markers in Roasted Coffee Powder Samples by GC×GC- TOF- MS and Multivariate Statistical Analysis. Food Biosci. 2024, 59, 104207. [Google Scholar] [CrossRef]
- Fayek, N.M.; Xiao, J.; Farag, M.A. A Multifunctional Study of Naturally Occurring Pyrazines in Biological Systems; Formation Mechanisms, Metabolism, Food Applications and Functional Properties. Crit. Rev. Food Sci. Nutr. 2023, 63, 5322–5338. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Cao, H.; & Xiao, J. Pyrazines in Food. Handbook of Dietary Phytochemicals, 2020, 1-25.
- Ren, A.; Zhang, Y.; Bian, Y.; Liu, Y.; Zhang, Y.; Ren, C.; Zhou, Y.; Zhang, T.; Feng, X. Pyrazines in Food Samples: Recent Update on Occurrence, Formation, Sampling, Pretreatment and Analysis Methods. Food Chem. 2024, 430, 137086. [Google Scholar] [CrossRef] [PubMed]
- Poisson, L.; Schmalzried, F.; Davidek, T.; Blank, I.; Kerler, J. Study on the Role of Precursors in Coffee Flavor Formation Using In-Bean Experiments. J. Agric. Food Chem. 2009, 57, 9923–9931. [Google Scholar] [CrossRef]
- Xing, H.; Yaylayan, V. Mechanochemistry of Strecker Degradation: Interaction of Glyoxal with Amino Acids. Food Chem. 2024, 439, 138071. [Google Scholar] [CrossRef]
- Shu, C.-K.; Lawrence, B.M. Formation of 2-(1-Hydroxyalkyl)-3-Oxazolines from the Reaction of Acyloins and Ammonia Precursors under Mild Conditions. J. Agric. Food Chem. 1995, 43, 2922–2924. [Google Scholar] [CrossRef]
- Rizzacasa, M.; Ricca, M. Chemistry and Biology of Acyloin Natural Products. Synthesis 2023, 55, 2273–2284. [Google Scholar] [CrossRef]
- Ma, Y.-J.; Wang, X.-Y.; Zhu, B.-W.; Du, M.; Dong, L.; Dong, X.-P.; Xu, X.-B. Model Studies on the Formation of 2-Vinylpyrazine and 2-Vinyl-6-Methylpyrazine in Maillard-Type Reactions. Food Chem. 2022, 374, 131652. [Google Scholar] [CrossRef]
- Low, M.Y.; Parker, J.K.; Mottram, D.S. Mechanisms of Alkylpyrazine Formation in a Potato Model System Containing Added Glycine. J. Agric. Food Chem. 2007, 55, 4087–4094. [Google Scholar] [CrossRef]
- Parker, J.K.; Balagiannis, D.P.; Desforges, N.; Mottram, D.S. Flavor Development in a Meat-Based Petfood Containing Added Glucose and Glycine. In ACS Symposium Series; Mottram, D.S., Taylor, A.J., Eds.; American Chemical Society: Washington, DC, 2010; Vol. 1042, pp 85–93. [Google Scholar]
- Jiang, W.; Wang, X.; Ma, Y.; Du, M.; Wu, C.; Xu, X. Mechanism of Carbon Skeleton Formation of 2,3,5-Trimethylpyrazine via a Conversion Reaction between Methylglyoxal and Glyoxal. J. Agric. Food Chem. 2023, 71, 5337–5344. [Google Scholar] [CrossRef]
- Sakaguchi, M.; Shibamoto, T. Formation of Heterocyclic Compounds from the Reaction of Cysteamine and D-Glucose, Acetaldehyde, or Glyoxal. J. Agric. Food Chem. 1978, 26, 1179–1183. [Google Scholar] [CrossRef]
- Liu, M.; Yu, J.; Zhou, T.; Xu, H.; Hayat, K.; Zhang, X.; Ho, C.-T. Formation Priority of Pyrazines and 2-Acetylthiazole Dependent on the Added Cysteine and Fragments of Deoxyosones during the Thermal Process of the Glycine–Ribose Amadori Compound. J. Agric. Food Chem. 2022, 70, 11643–11651. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Wang, Y.; Ma, Q.; Li, Y.; Wang, B.; Bai, S.; Fan, B.; Wang, F. The Number and Position of Unsaturated Bonds in Aliphatic Aldehydes Affect Meat Flavorings System: Insights on Initial Maillard Reaction Stage and Meat Flavor Formation from Thiazolidine Derivatives. Curr. Res. Food Sci. 2024, 8, 100719. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Rakotondrabe, T.F.; Chen, G.; & Guo, M. Advances in Microbial Analysis: Based on Volatile Organic Compounds of Microorganisms in Food. Food Chem. 2023, 418, 135950. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Xia, X.; Deng, S.; Cui, H.; Hayat, K.; Zhang, X.; & Ho, C.T. Reduced Asynchronism Between Regenerative Cysteine and Fragments of Deoxyosones Promoting Formation of Sulfur-Containing Compounds Through Extra-Added Xylose and Elevated Temperature During Thermal Processing of 2 Threityl-Thiazolidine-4-Carboxylic Acid. Food Chem. 2023, 404, 134420. [Google Scholar] [CrossRef]
- Feng, L.; Yang, Y.; Xie, Y.T.; Yan, W.Y.; Ma, Y.K.; Hu, S.; & Yu, A.N. The Volatile Organic Compounds Generated from the Maillard Reaction Between L-Ascorbic Acid And L-Cysteine in Hot Compressed Water. J Sci Food Agric. 2024, 104, 5764–5775. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



