Submitted:
04 October 2024
Posted:
07 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
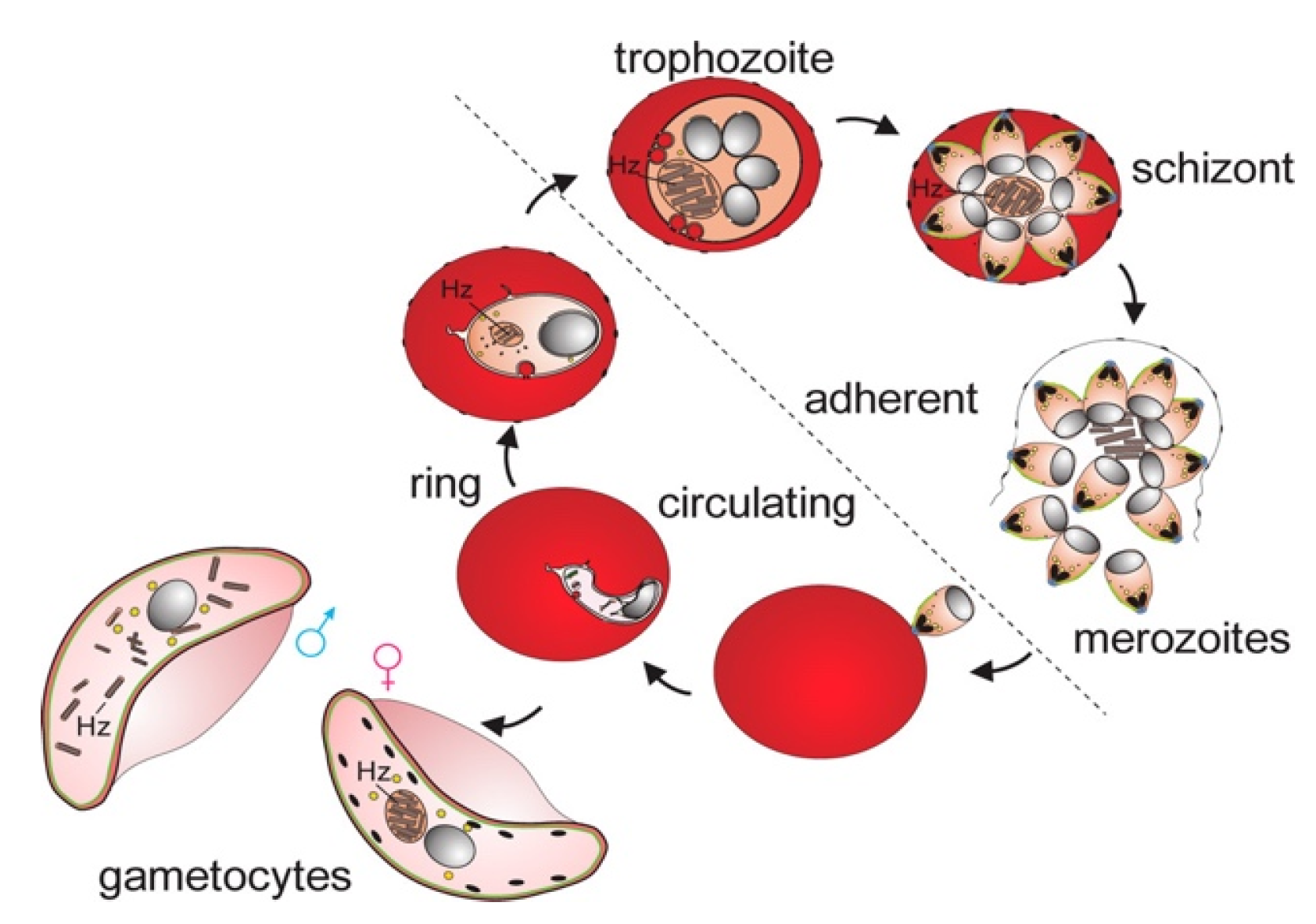
2. Life cycle of the parasite
3. Spectral biomarkers for malaria
3.1. Hemozoin
3.1.1. Discovery of hemozoin and malaria infection
3.1.1. Organisms producing hemozoin
3.1.2. Hemozoin location in humans
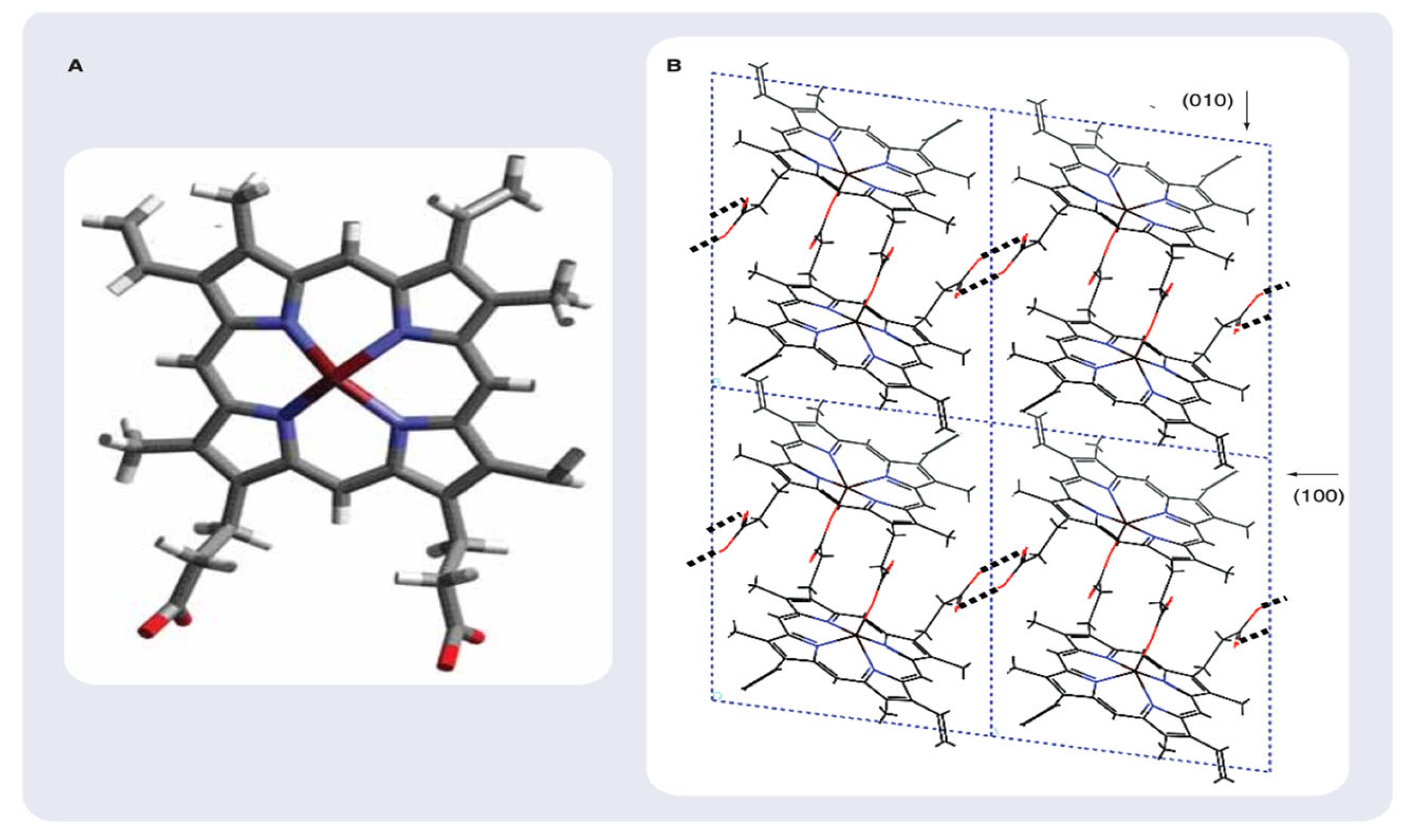
3.1.3. Crystal structure
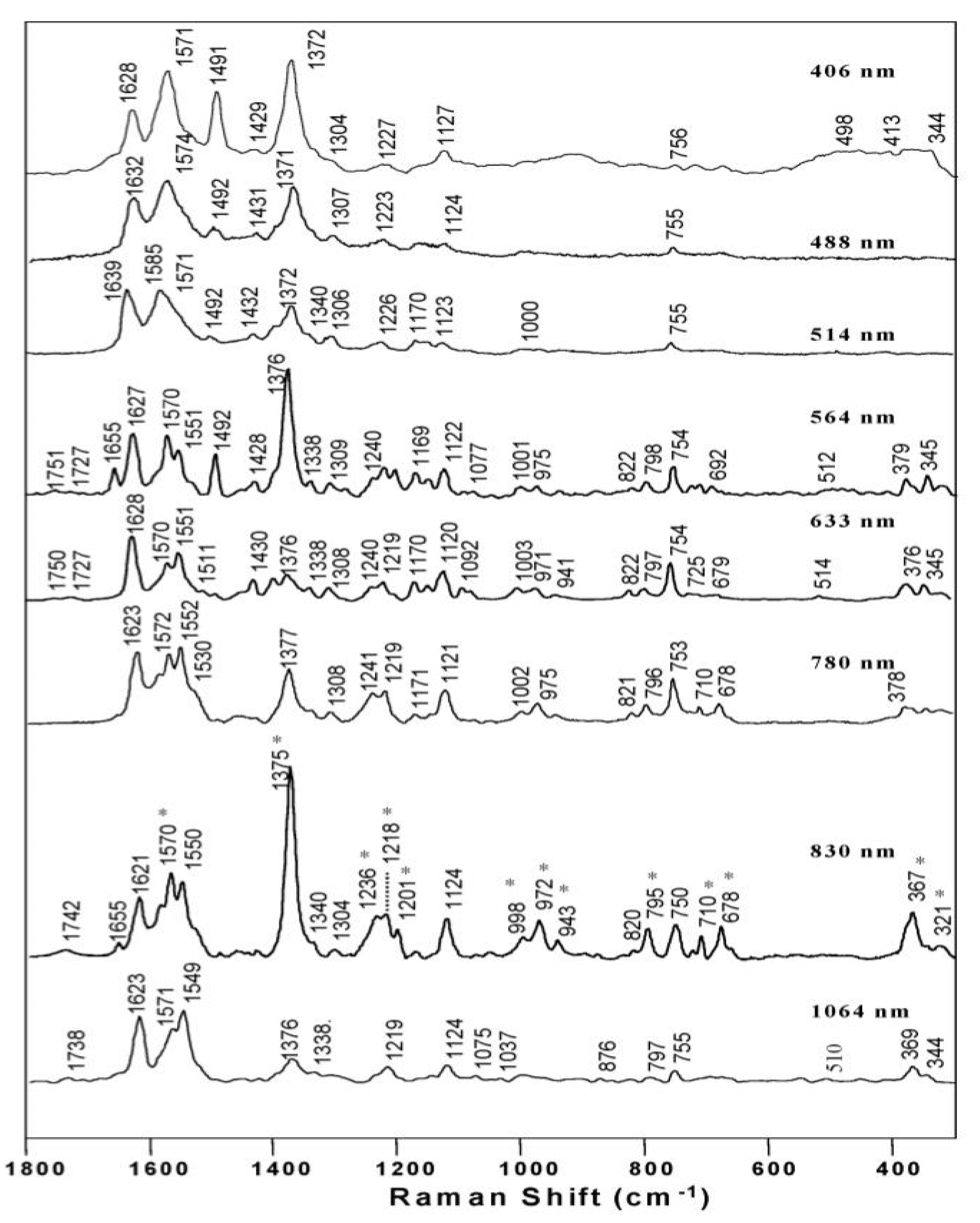
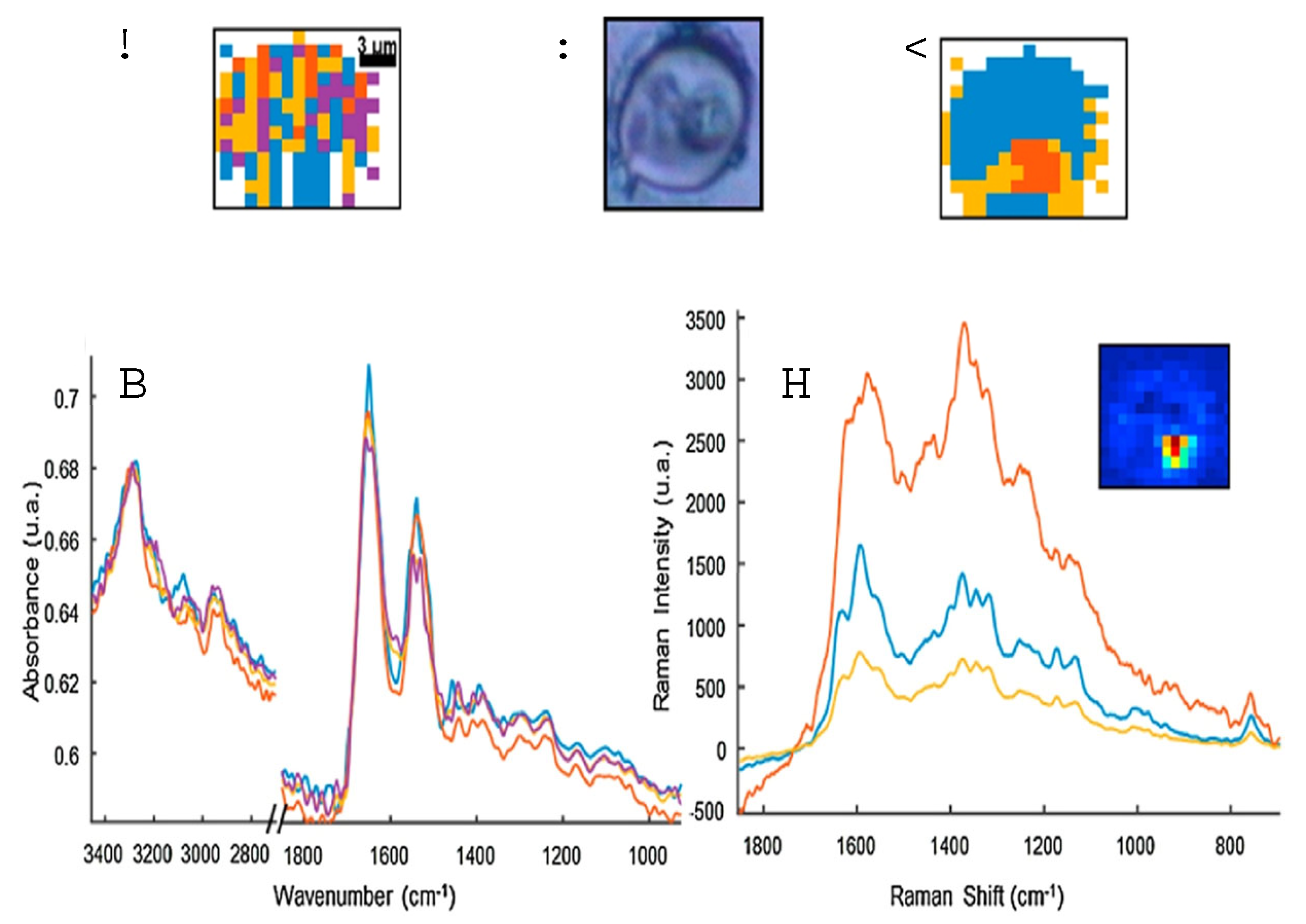
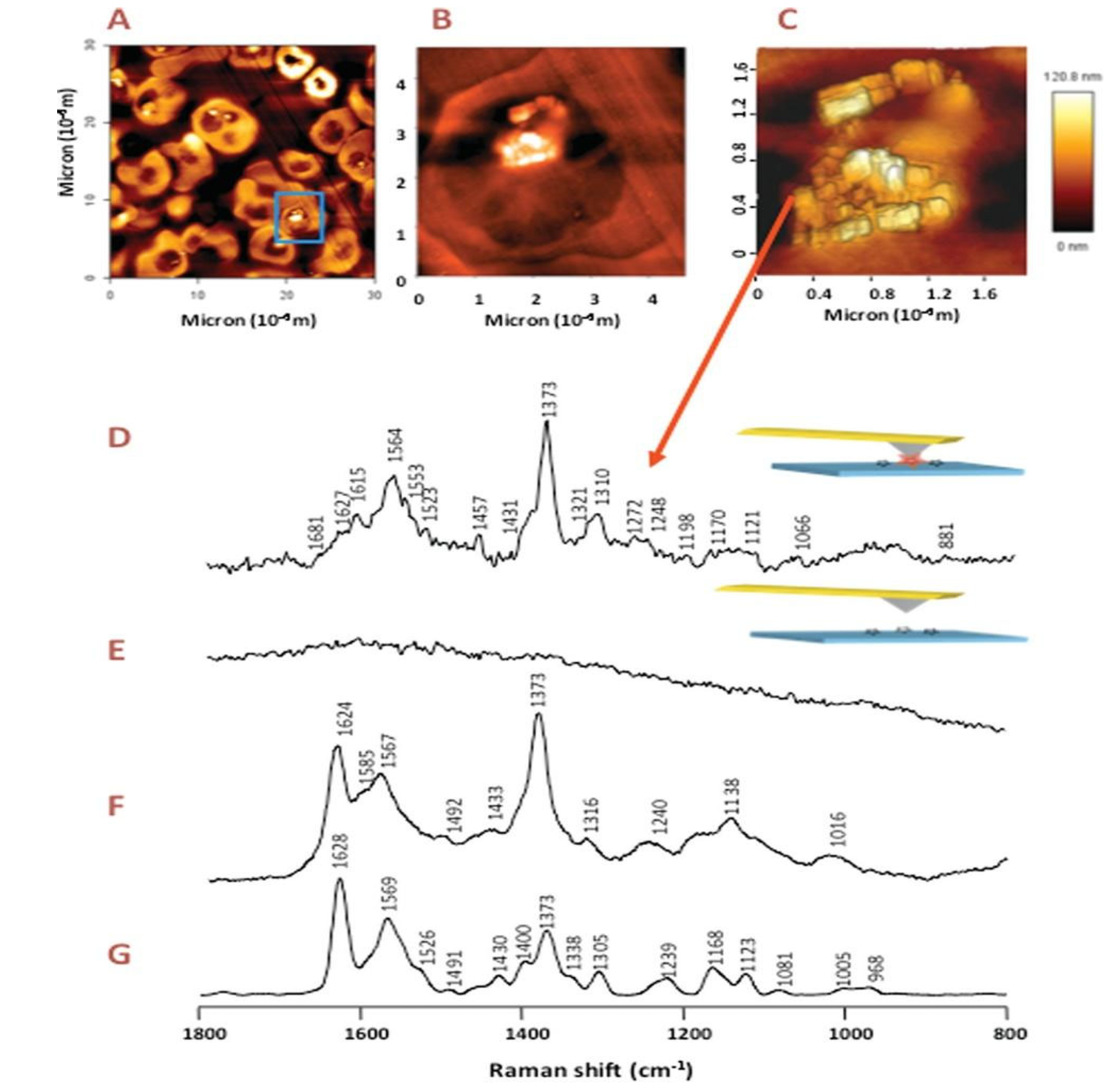
3.1.4. Raman spectroscopy of hemozoin
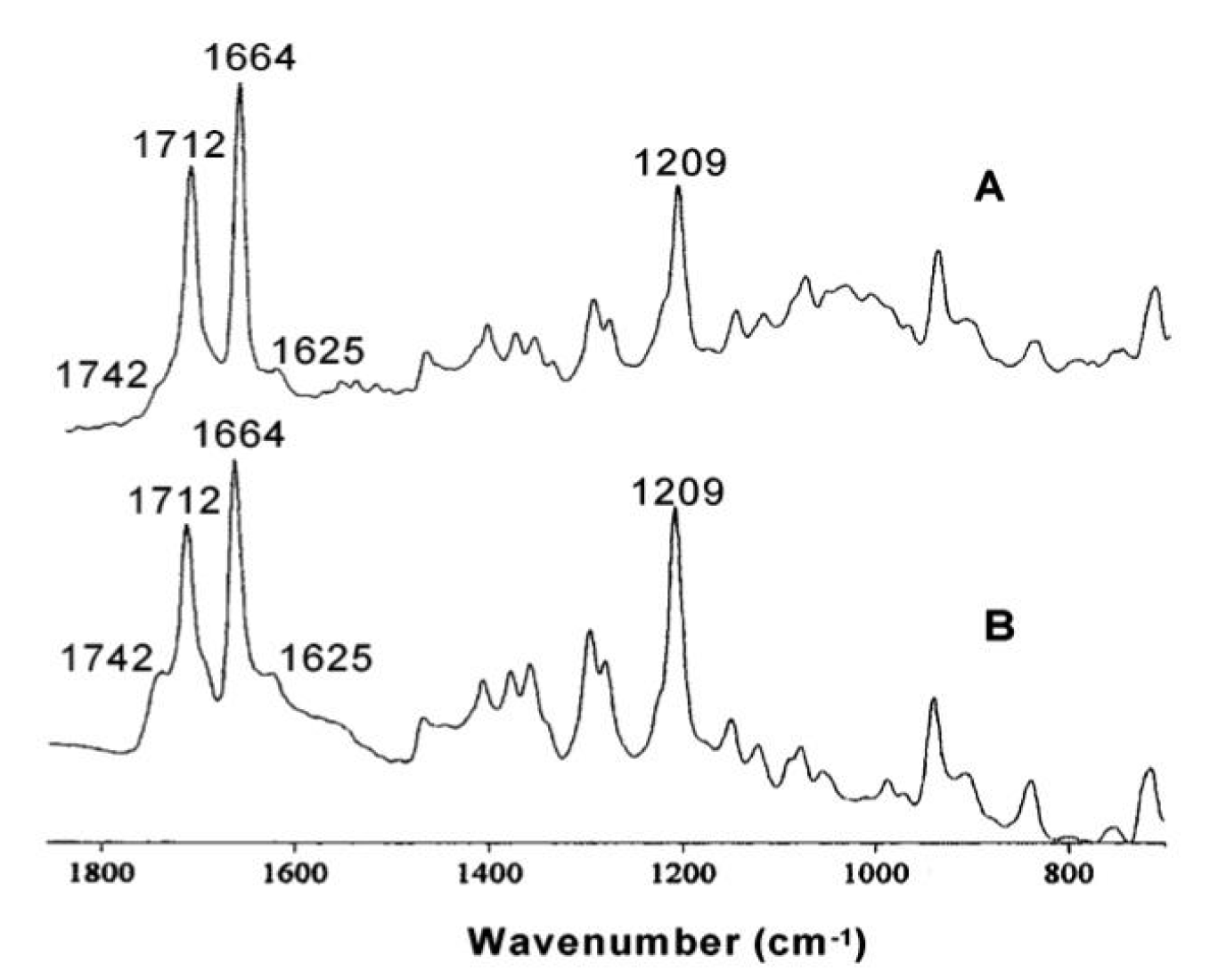
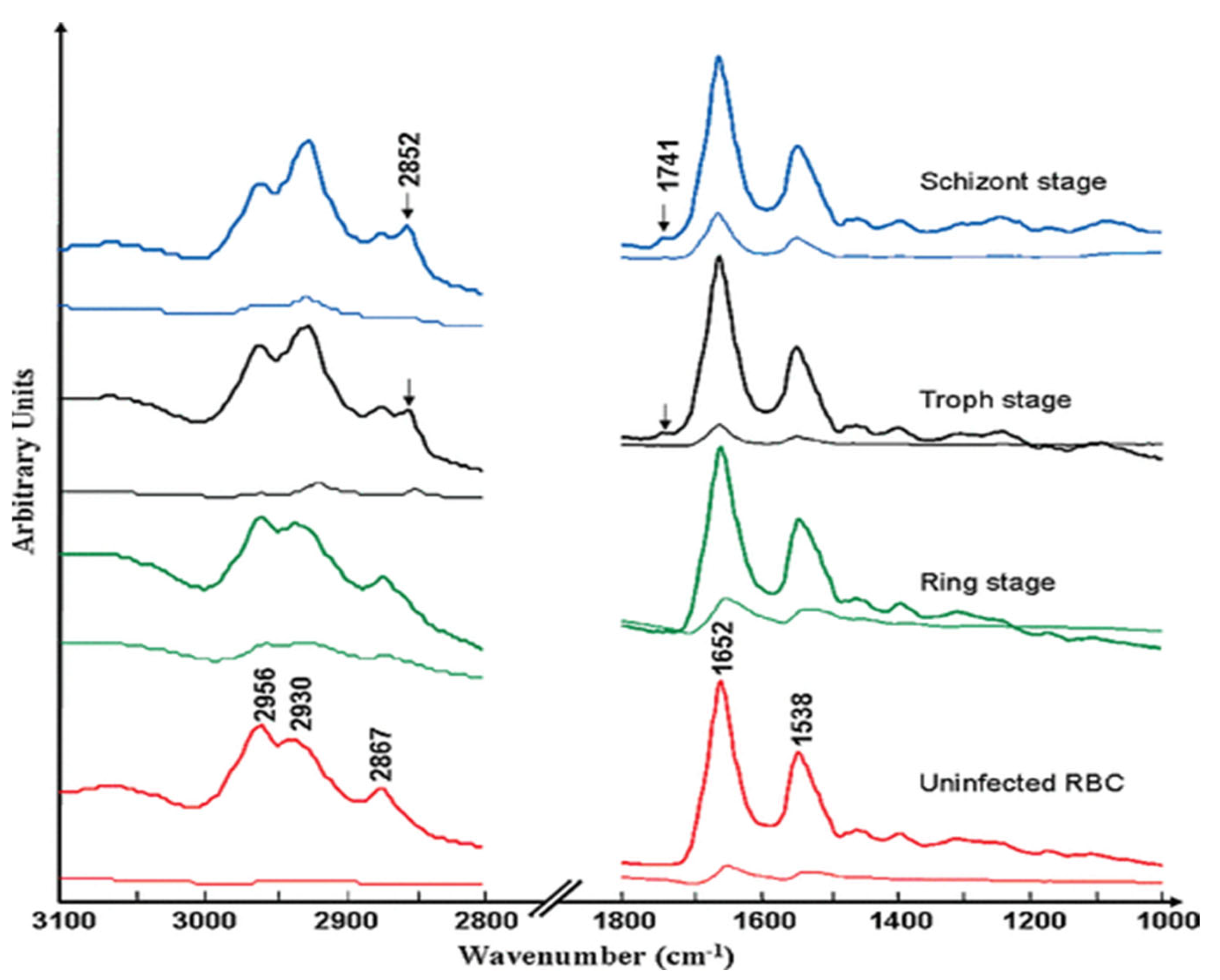
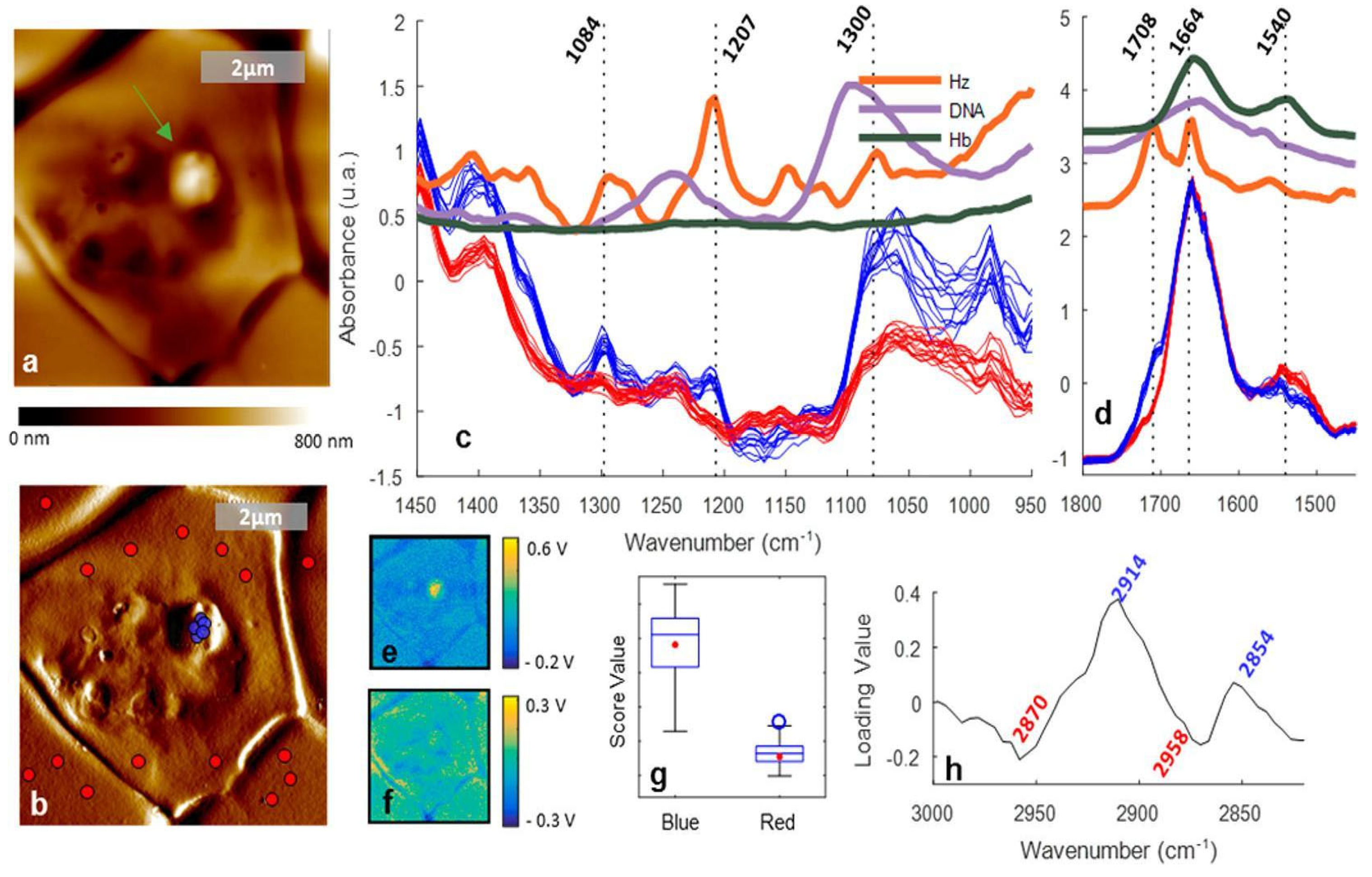
3.1.5. FTIR spectroscopy of hemozoin
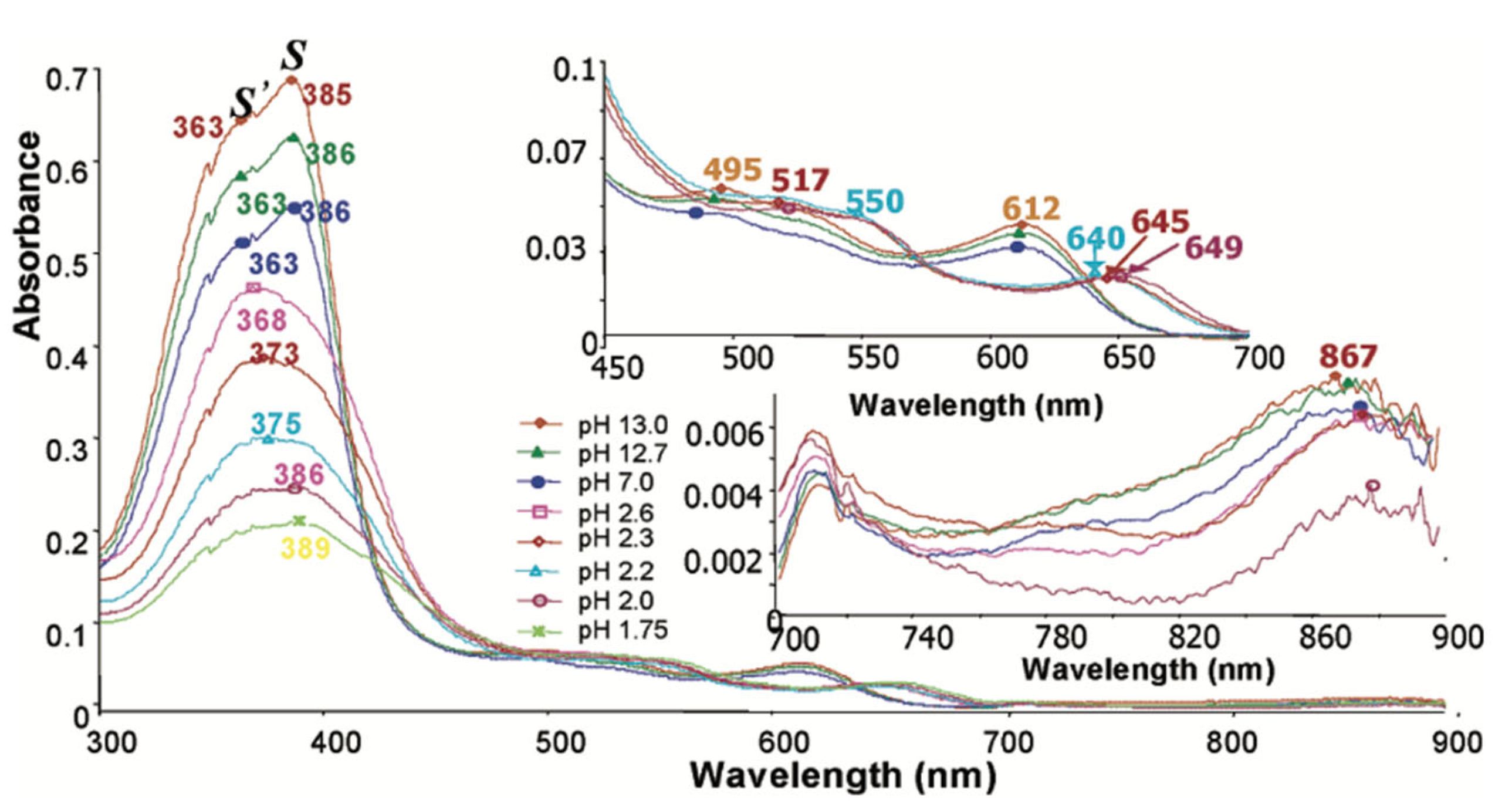
3.1.6. UV/Visible spectroscopy of hemozoin
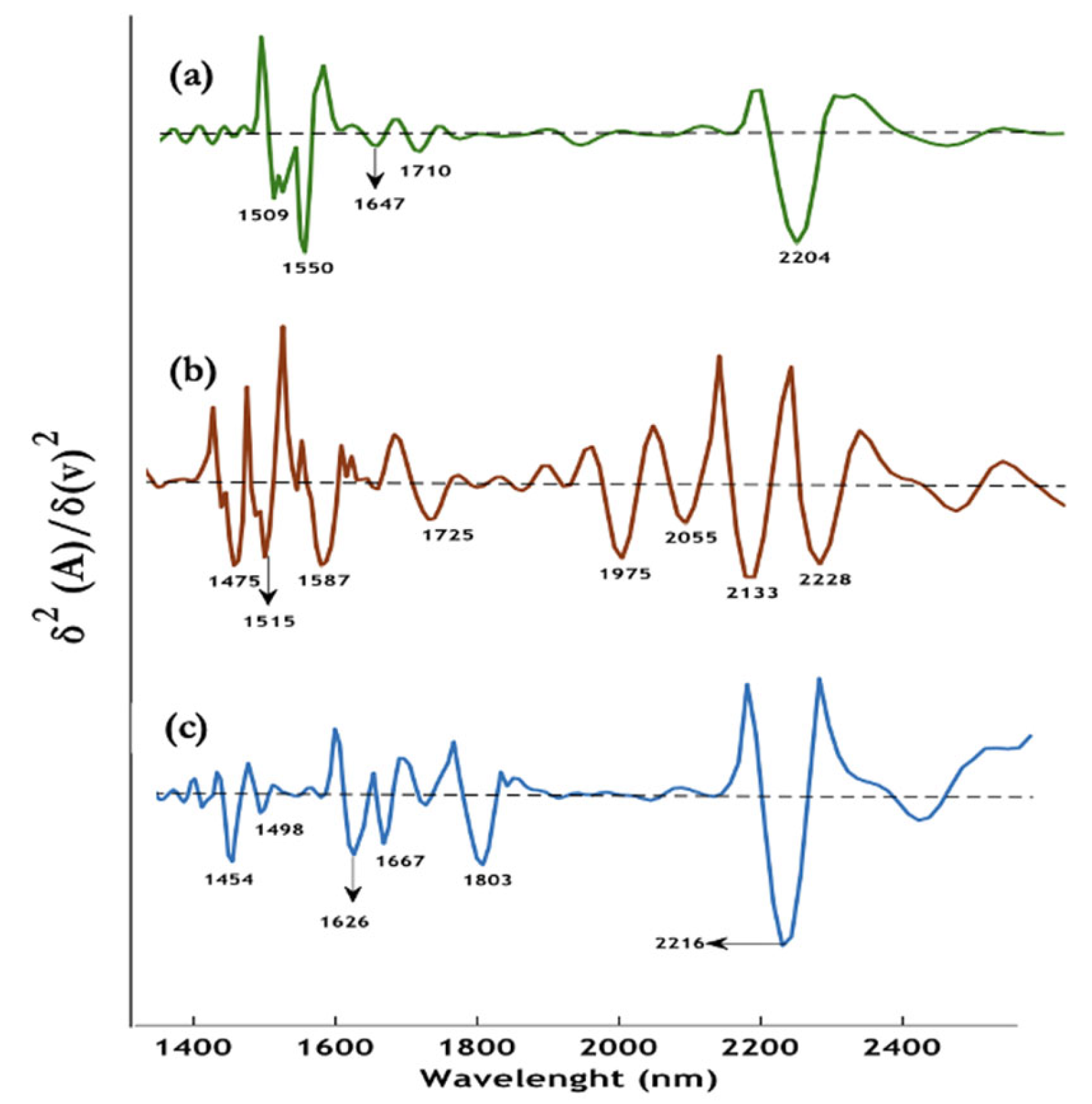
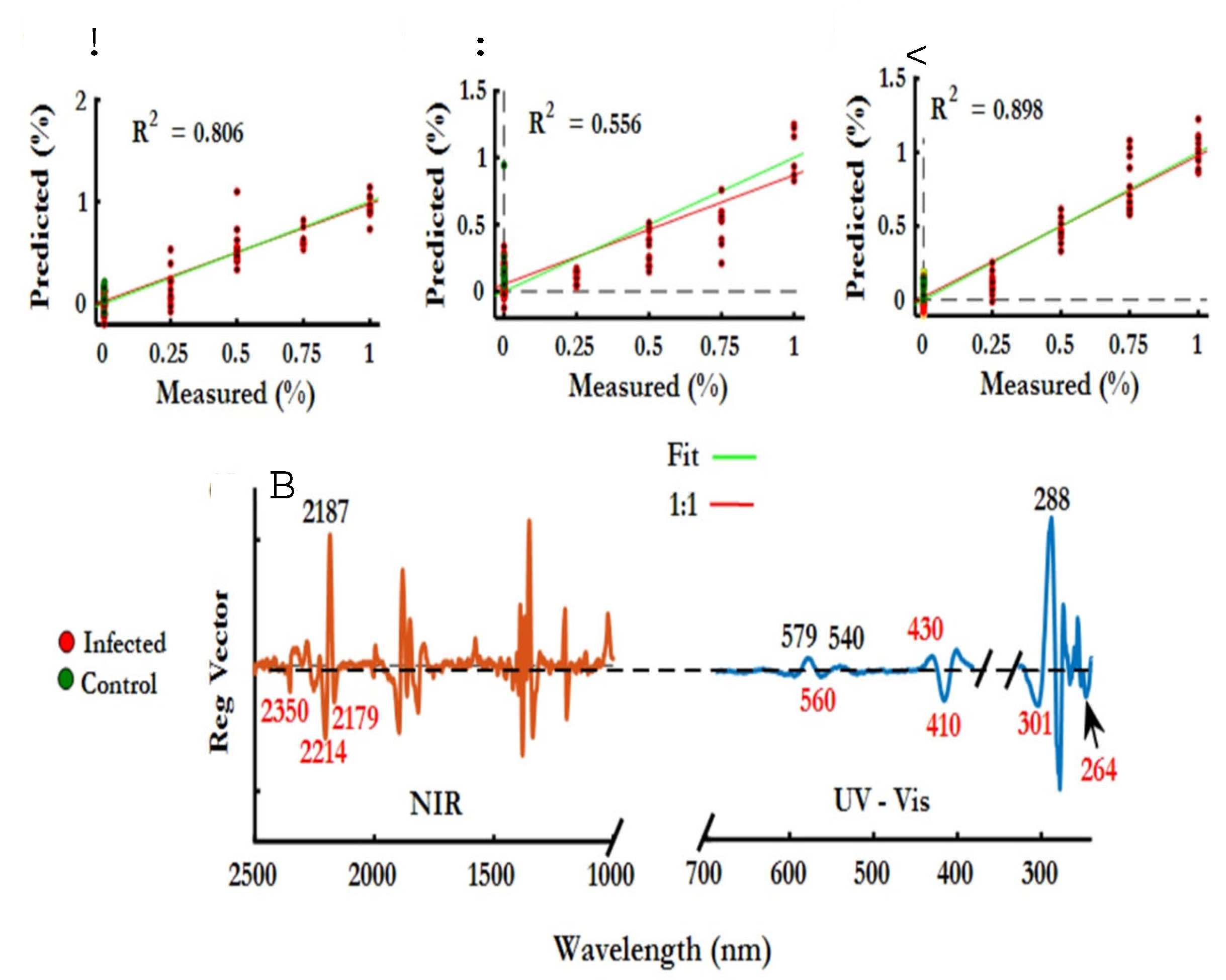
3.1.8. NIR spectroscopy of hemozoin
3.2. Lipids
3.3. Nucleic acids
3.4. Proteins
3.4.1. FTIR spectroscopy of proteins
3.4.2. Raman spectroscopy of proteins
3.4.3. UV/Visible spectroscopy of proteins
3.4.4. Near infrared spectroscopy of proteins
5. Clinical field trials of ATR-FTIR spectroscopy
6. Pathway and obstacles to translation
7. Application of spectroscopy to monitor drug interactions
8. Conclusions
Funding
Conflicts of Interest
References
- Venkatesan, P. The 2023 WHO World malaria report. Lancet Microbe 2024, 5, e214. [Google Scholar] [CrossRef] [PubMed]
- Abebaw, A.; Aschale, Y.; Kebede, T.; Hailu, A. The prevalence of symptomatic and asymptomatic malaria and its associated factors in Debre Elias district communities, Northwest Ethiopia. Malar J 2022, 21, 167. [Google Scholar] [CrossRef] [PubMed]
- Danwang, C.; Kirakoya-Samadoulougou, F.; Samadoulougou, S. Assessing field performance of ultrasensitive rapid diagnostic tests for malaria: a systematic review and meta-analysis. Malar J 2021, 20, 245. [Google Scholar] [CrossRef]
- Mantsch, H.; Jackson, M. Molecular spectroscopy in biodiagnostics (from Hippocrates to Herschel and beyond). Journal of Molecular Structure 1995, 347, 187–206. [Google Scholar] [CrossRef]
- Adegoke, J.A.; Kochan, K.; Heraud, P.; Wood, B.R. A Near-Infrared "Matchbox Size" Spectrometer to Detect and Quantify Malaria Parasitemia. Anal Chem 2021, 93, 5451–5458. [Google Scholar] [CrossRef]
- Adegoke, J.A.; Raper, H.; Gassner, C.; Heraud, P.; Wood, B.R. Visible microspectrophotometry coupled with machine learning to discriminate the erythrocytic life cycle stages of P. falciparum malaria parasites in functional single cells. Analyst 2022, 147, 2662–2670. [Google Scholar] [CrossRef]
- Khoshmanesh, A.; Dixon, M.W.; Kenny, S.; Tilley, L.; McNaughton, D.; Wood, B.R. Detection and quantification of early-stage malaria parasites in laboratory infected erythrocytes by attenuated total reflectance infrared spectroscopy and multivariate analysis. Anal Chem 2014, 86, 4379–4386. [Google Scholar] [CrossRef]
- Rathi, A.; Chowdhry, Z.; Patel, A.; Zuo, S.; Veettil, T.C.P.; Adegoke, J.A.; Heidari, H.; Wood, B.R.; Bhallamudi, V.P.; Peng, W.K. Hemozoin in malaria eradication—from material science, technology to field test. NPG Asia Materials 2023, 15, 70. [Google Scholar] [CrossRef]
- Drouin, E.; Hautecoeur, P.; Markus, M. Who was the first to visualize the malaria parasite? Parasit Vectors 2024, 17, 184. [Google Scholar] [CrossRef]
- Klencke, P.F.H. Neue physiologische Abhandlungen auf selbständige Beobachtungen gegründet für Aerzte und Naturforscher; Leipzig, 1843; Volume VIII.
- Lavéran, A. Note sur un nouveau parasite trouvé dans le sang de plusieurs malades atteints de fièvre palustre. Bull Acad Méd. 1880, 1880, 1235–1236. [Google Scholar]
- Egan, T.J. Hemozoin Biophysics and Drug Target. Encylopedia of Malaria 2013, 1–11. [Google Scholar] [CrossRef]
- Xiao, S.H.; Sun, J. Schistosoma hemozoin and its possible roles. Int J Parasitol 2017, 47, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.A.; Udeinya, I.J.; Leech, J.H.; Hay, R.J.; Aikawa, M.; Barnwell, J.; Green, I.; Miller, L.H. Plasmodium falciparum malaria. An amelanotic melanoma cell line bears receptors for the knob ligand on infected erythrocytes. J Clin Invest 1982, 70, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Qadir, H.; Baig, M.M.; Ojla, M.D.; Aisha, M.; Adil, A. Malarial Pigment Hemozoin in Neutrophils: A Manifestation of Severe Malaria. Turk J Haematol 2023, 40, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Dixon, M.W.; Thompson, J.; Gardiner, D.L.; Trenholme, K.R. Sex in Plasmodium: a sign of commitment. Trends Parasitol 2008, 24, 168–175. [Google Scholar] [CrossRef]
- Birhanu, M.; Asres, Y.; Adissu, W.; Yemane, T.; Zemene, E.; Gedefaw, L. Hematological Parameters and Hemozoin-Containing Leukocytes and Their Association with Disease Severity among Malaria Infected Children: A Cross-Sectional Study at Pawe General Hospital, Northwest Ethiopia. Interdiscip Perspect Infect Dis 2017, 2017, 8965729. [Google Scholar] [CrossRef]
- Pagola, S.; Stephens, P.W.; Bohle, D.S.; Kosar, A.D.; Madsen, S.K. The structure of malaria pigment beta-haematin. Nature 2000, 404, 307–310. [Google Scholar] [CrossRef]
- Solomonov, I.; Osipova, M.; Feldman, Y.; Baehtz, C.; Kjaer, K.; Robinson, I.K.; Webster, G.T.; McNaughton, D.; Wood, B.R.; Weissbuch, I.; et al. Crystal nucleation, growth, and morphology of the synthetic malaria pigment beta-hematin and the effect thereon by quinoline additives: the malaria pigment as a target of various antimalarial drugs. J Am Chem Soc 2007, 129, 2615–2627. [Google Scholar] [CrossRef]
- Wood, B.R.; Langford, S.J.; Cooke, B.M.; Lim, J.; Glenister, F.K.; Duriska, M.; Unthank, J.K.; McNaughton, D. Resonance Raman spectroscopy reveals new insight into the electronic structure of beta-hematin and malaria pigment. J Am Chem Soc 2004, 126, 9233–9239. [Google Scholar] [CrossRef]
- Marzec, K.M.; Perez-Guaita, D.; de Veij, M.; McNaughton, D.; Baranska, M.; Dixon, M.W.A.; Tilley, L.; Wood, B.R. Red Blood Cells Polarize Green Laser Light Revealing Hemoglobin′s Enhanced Non-Fundamental Raman Modes. ChemPhysChem 2014, 15, 3963–3968. [Google Scholar] [CrossRef]
- Slater, A.F. Malaria pigment. Exp Parasitol 1992, 74, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Slater, A.F.; Swiggard, W.J.; Orton, B.R.; Flitter, W.D.; Goldberg, D.E.; Cerami, A.; Henderson, G.B. An iron-carboxylate bond links the heme units of malaria pigment. Proc Natl Acad Sci U S A 1991, 88, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Bohle, D.S.; Conklin, B.J.; Cox, D.; Madsen, S.K.; Paulson, S.; Stephens, P.W.; Yee, G.T. Structural and Spectroscopic Studies of beta-Hematin (the Heme Coordination Polymer in Malaria Pigment). In Inorganic and Organometallic Polymers II: AdVanced Materials and Intermediates; ACS Symposium Series, Wisian-Neilson, P., Allcock, H.R., Wynne, K.J., Eds.; American Chemical Society: Washington, DC, 1994; Volume 572, pp. 497–515. [Google Scholar]
- Morselt, A.F.; Glastra, A.; James, J. Microspectrophotometric analysis of malarial pigment. Exp Parasitol 1973, 33, 17–22. [Google Scholar] [CrossRef]
- Lukianova-Hleb, E.Y.; Campbell, K.M.; Constantinou, P.E.; Braam, J.; Olson, J.S.; Ware, R.E.; Sullivan, D.J., Jr.; Lapotko, D.O. Hemozoin-generated vapor nanobubbles for transdermal reagent- and needle-free detection of malaria. Proc Natl Acad Sci U S A 2014, 111, 900–905. [Google Scholar] [CrossRef]
- Bendrat, K.; Berger, B.J.; Cerami, A. Haem polymerization in malaria. Nature 1995, 378, 138–139. [Google Scholar] [CrossRef]
- Ancelin, M.L.; Vial, H.J. Saturable and non-saturable components of choline transport in Plasmodium-infected mammalian erythrocytes: possible role of experimental conditions. Biochem J 1992, 283 Pt 2, 619–621. [Google Scholar] [CrossRef]
- Holz, G.G. Lipids and the malarial parasite. Bull World Health Organ 1977, 55, 237–248. [Google Scholar]
- Vielemeyer, O.; McIntosh, M.T.; Joiner, K.A.; Coppens, I. Neutral lipid synthesis and storage in the intraerythrocytic stages of Plasmodium falciparum. Mol Biochem Parasitol 2004, 135, 197–209. [Google Scholar] [CrossRef]
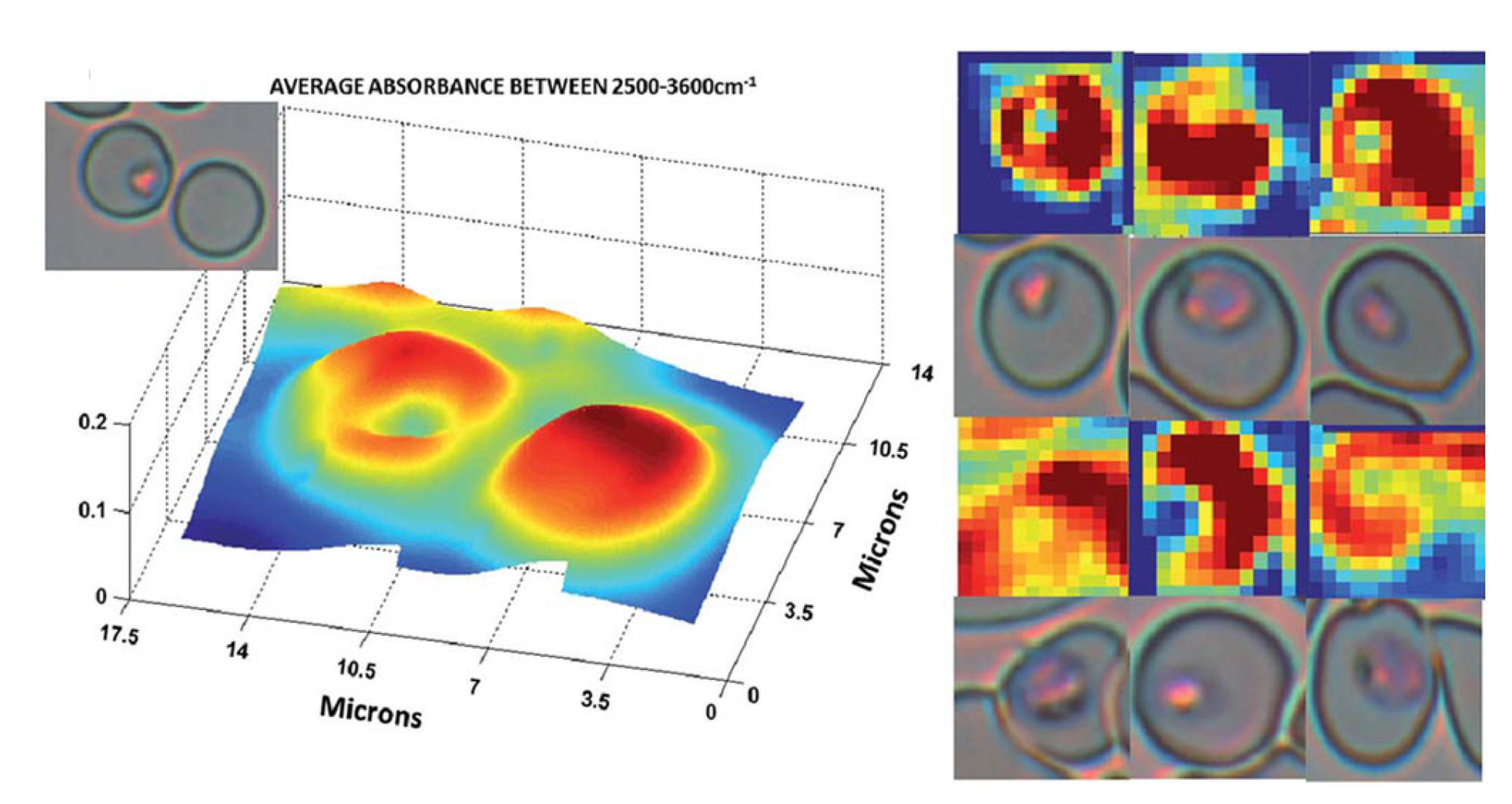
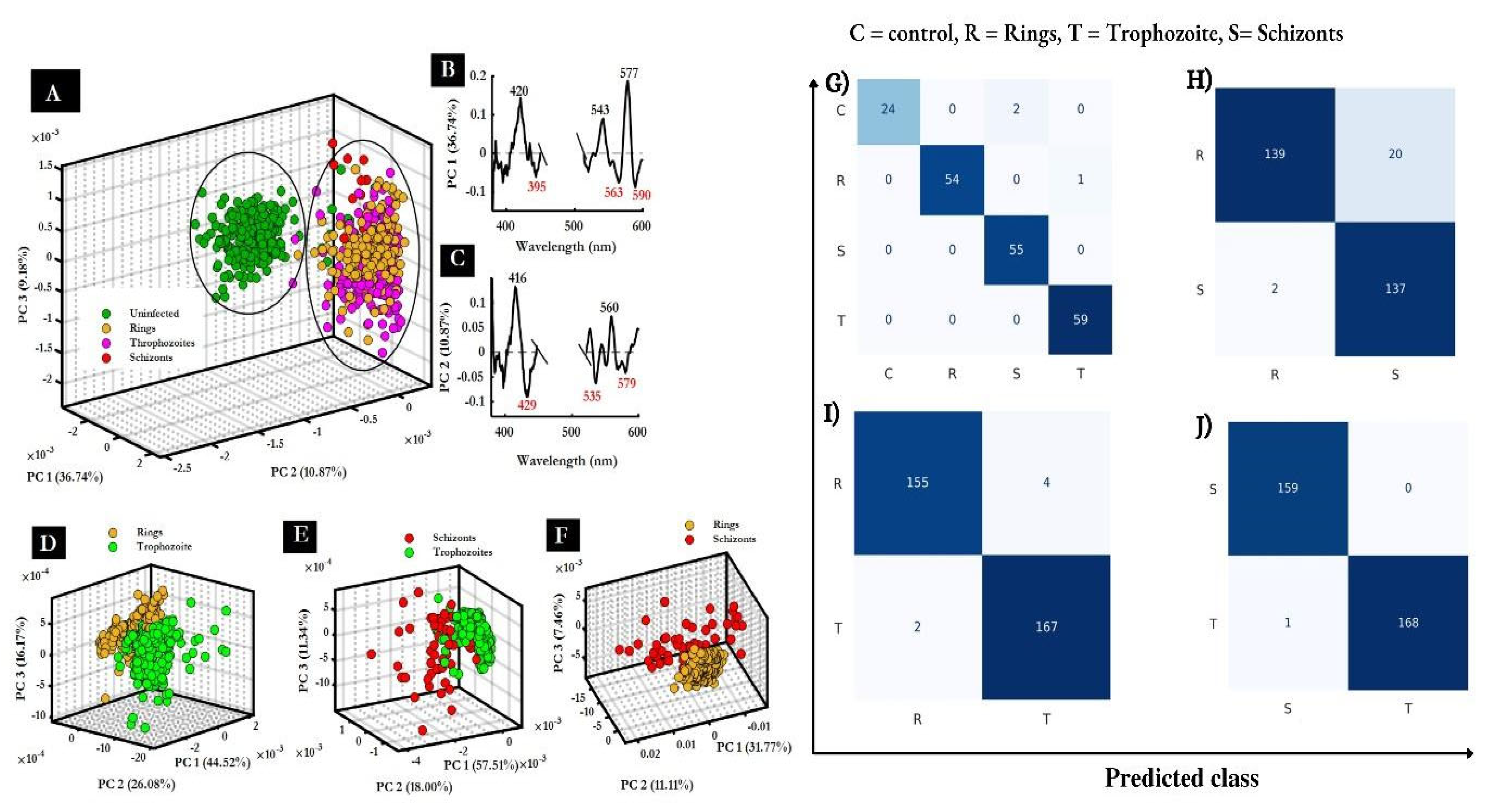
- Webster, G.T.; de Villiers, K.A.; Egan, T.J.; Deed, S.; Tilley, L.; Tobin, M.J.; Bambery, K.R.; McNaughton, D.; Wood, B.R. Discriminating the intraerythrocytic lifecycle stages of the malaria parasite using synchrotron FT-IR microspectroscopy and an artificial neural network. Anal Chem 2009, 81, 2516–2524. [Google Scholar] [CrossRef]
- Pisciotta, J.M.; Coppens, I.; Tripathi, A.K.; Scholl, P.F.; Shuman, J.; Bajad, S.; Shulaev, V.; Sullivan, D.J., Jr. The role of neutral lipid nanospheres in Plasmodium falciparum haem crystallization. Biochem J 2007, 402, 197–204. [Google Scholar] [CrossRef]
- Palacpac, N.M.; Hiramine, Y.; Seto, S.; Hiramatsu, R.; Horii, T.; Mitamura, T. Evidence that Plasmodium falciparum diacylglycerol acyltransferase is essential for intraerythrocytic proliferation. Biochem Biophys Res Commun 2004, 321, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Jackson, K.E.; Klonis, N.; Ferguson, D.J.; Adisa, A.; Dogovski, C.; Tilley, L. Food vacuole-associated lipid bodies and heterogeneous lipid environments in the malaria parasite, Plasmodium falciparum. Mol Microbiol 2004, 54, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Nawabi, P.; Lykidis, A.; Ji, D.; Haldar, K. Neutral-lipid analysis reveals elevation of acylglycerols and lack of cholesterol esters in Plasmodium falciparum-infected erythrocytes. Eukaryot Cell 2003, 2, 1128–1131. [Google Scholar] [CrossRef] [PubMed]
- Egan, T.J.; Chen, J.Y.; de Villiers, K.A.; Mabotha, T.E.; Naidoo, K.J.; Ncokazi, K.K.; Langford, S.J.; McNaughton, D.; Pandiancherri, S.; Wood, B.R. Haemozoin (beta-haematin) biomineralization occurs by self-assembly near the lipid/water interface. FEBS Lett 2006, 580, 5105–5110. [Google Scholar] [CrossRef]
- Birczyńska-Zych, M.; Czepiel, J.; Łabanowska, M.; Kraińska, M.; Biesiada, G.; Moskal, P.; Kozicki, M.; Garlicki, A.; Wesełucha-Birczyńska, A. Could Raman spectroscopy distinguish between P. falciparum and P. vivax Infection? Clinical Spectroscopy 2021, 3, 100015. [Google Scholar] [CrossRef]
- Gardner, M.J.; Hall, N.; Fung, E.; White, O.; Berriman, M.; Hyman, R.W.; Carlton, J.M.; Pain, A.; Nelson, K.E.; Bowman, S.; et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 2002, 419, 498–511. [Google Scholar] [CrossRef]
- Ganter, M.; Goldberg, J.M.; Dvorin, J.D.; Paulo, J.A.; King, J.G.; Tripathi, A.K.; Paul, A.S.; Yang, J.; Coppens, I.; Jiang, R.H.; et al. Plasmodium falciparum CRK4 directs continuous rounds of DNA replication during schizogony. Nat Microbiol 2017, 2, 17017. [Google Scholar] [CrossRef]
- Machado, M.; Steinke, S.; Ganter, M. Plasmodium Reproduction, Cell Size, and Transcription: How to Cope With Increasing DNA Content? Front Cell Infect Microbiol 2021, 11, 660679. [Google Scholar] [CrossRef]
- Sambe, B.S.; Diagne, A.; Diatta, H.A.M.; Gaba, F.M.; Sarr, I.; Diatta, A.S.; Diaw, S.O.M.; Sané, R.; Diouf, B.; Vigan-Womas, I.; et al. Molecular detection and quantification of Plasmodium vivax DNA in blood pellet and plasma samples from patients in Senegal. Frontiers in Parasitology 2023, 2. [Google Scholar] [CrossRef]
- Hobro, A.J.; Konishi, A.; Coban, C.; Smith, N.I. Raman spectroscopic analysis of malaria disease progression via blood and plasma samples. Analyst 2013, 138, 3927–3933. [Google Scholar] [CrossRef]
- Wood, B.R. The importance of hydration and DNA conformation in interpreting infrared spectra of cells and tissues. Chem Soc Rev 2016, 45, 1980–1998. [Google Scholar] [CrossRef] [PubMed]
- Reuterswärd, P.; Bergström, S.; Orikiiriza, J.; Lindquist, E.; Bergström, S.; Andersson Svahn, H.; Ayoglu, B.; Uhlén, M.; Wahlgren, M.; Normark, J.; et al. Levels of human proteins in plasma associated with acute paediatric malaria. Malaria Journal 2018, 17, 426. [Google Scholar] [CrossRef] [PubMed]
- Ikegbunam, M.; Maurer, M.; Abone, H.; Ezeagwuna, D.; Sandri, T.L.; Esimone, C.; Ojurongbe, O.; Woldearegai, T.G.; Kreidenweiss, A.; Held, J.; et al. Evaluating Malaria Rapid Diagnostic Tests and Microscopy for Detecting Plasmodium Infection and Status of Plasmodium falciparum Histidine-Rich Protein 2/3 Gene Deletions in Southeastern Nigeria. Am J Trop Med Hyg 2024, 110, 902–909. [Google Scholar] [CrossRef]
- Kojom Foko, L.P.; Eboumbou Moukoko, C.E.; Jakhan, J.; Narang, G.; Hawadak, J.; Kouemo Motse, F.D.; Pande, V.; Singh, V. Deletions of Histidine-Rich Protein 2/3 Genes in Natural Plasmodium falciparum Populations from Cameroon and India: Role of Asymptomatic and Submicroscopic Infections. Am J Trop Med Hyg 2024, 110, 1100–1109. [Google Scholar] [CrossRef]
- Oyegoke, O.O.; Akoniyon, O.P.; Maharaj, L.; Adewumi, T.S.; Malgwi, S.A.; Aderoju, S.A.; Fatoba, A.J.; Adeleke, M.A.; Maharaj, R.; Okpeku, M. Molecular detection of sub-microscopic infections and Plasmodium falciparum histidine-rich protein-2 and 3 gene deletions in pre-elimination settings of South Africa. Sci Rep 2024, 14, 16024. [Google Scholar] [CrossRef]
- Ranjan, P.; Ghoshal, U.; Prakash, S.; Pandey, A.; Shukla, R. Genetic variability of histidine-rich protein 2 repeat sequences: Misleading factor in true determination of Plasmodium falciparum in different population. Indian J Med Microbiol 2024, 49, 100616. [Google Scholar] [CrossRef]
- Schreidah, C.; Giesbrecht, D.; Gashema, P.; Young, N.W.; Munyaneza, T.; Muvunyi, C.M.; Thwai, K.; Mazarati, J.B.; Bailey, J.A.; Juliano, J.J.; et al. Expansion of artemisinin partial resistance mutations and lack of histidine rich protein-2 and -3 deletions in Plasmodium falciparum infections from Rukara, Rwanda. Malar J 2024, 23, 150. [Google Scholar] [CrossRef]
- Xu, S.; Tang, J. Biological threats to global malaria elimination II Deletion in the malaria rapid diagnostic test target Plasmodium falciparum histidine-rich protein 2/3 genes. Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi 2024, 36, 239–242. [Google Scholar] [CrossRef]
- Bachman, C.M.; Cate, D.M.; Grant, B.; Burkot, S.; Mulondo, J.; Hsieh, H.V.; Chamai, M.; Odongo, B.; Olwoch, P.; Nalubega, M.; et al. A Novel Malaria Lateral Flow Assay for Detecting Plasmodium falciparum Lactate Dehydrogenase in Busia, Uganda. Am J Trop Med Hyg 2022, 106, 850–852. [Google Scholar] [CrossRef]
- Feleke, S.M.; Gidey, B.; Mohammed, H.; Nega, D.; Dillu, D.; Haile, M.; Solomon, H.; Parr, J.B.; Tollera, G.; Tasew, G.; et al. Field performance of Plasmodium falciparum lactate dehydrogenase rapid diagnostic tests during a large histidine-rich protein 2 deletion survey in Ethiopia. Malar J 2022, 21, 236. [Google Scholar] [CrossRef]
- Tan, A.F.; Sakam, S.S.B.; Rajahram, G.S.; William, T.; Abd Rachman Isnadi, M.F.; Daim, S.; Barber, B.E.; Kho, S.; Sutherland, C.J.; Anstey, N.M.; et al. Diagnostic accuracy and limit of detection of ten malaria parasite lactate dehydrogenase-based rapid tests for Plasmodium knowlesi and P. falciparum. Front Cell Infect Microbiol 2022, 12, 1023219. [Google Scholar] [CrossRef] [PubMed]
- Saxena, N.; Pandey, V.C.; Dutta, G.P.; Ghatak, S. Levels of FDP-aldolase enzyme activity in Plasmodium knowlesi parasitized monkey erythrocytes. Indian J Exp Biol 1986, 24, 502–504. [Google Scholar]
- Francis, S.E.; Sullivan, D.J., Jr.; Goldberg, D.E. Hemoglobin metabolism in the malaria parasite Plasmodium falciparum. Annu Rev Microbiol 1997, 51, 97–123. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Boysen, R.I.; Wood, B.R.; Kansiz, M.; McNaughton, D.; Hearn, M.T. Determination of the secondary structure of proteins in different environments by FTIR-ATR spectroscopy and PLS regression. Biopolymers 2008, 89, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Heraud, P.; Chatchawal, P.; Wongwattanakul, M.; Tippayawat, P.; Doerig, C.; Jearanaikoon, P.; Perez-Guaita, D.; Wood, B.R. Infrared spectroscopy coupled to cloud-based data management as a tool to diagnose malaria: a pilot study in a malaria-endemic country. Malar J 2019, 18, 348. [Google Scholar] [CrossRef]
- Elliott, A.; Ambrose, E.J. Evidence of chain folding in polypeptides and proteins. Discussions of the Faraday Society 1950, 9, 246–251. [Google Scholar] [CrossRef]
- Salzer, R. Practical Guide to Interpretive Near-Infrared Spectroscopy. By Jerry Workman, Jr. and Lois Weyer. Angewandte Chemie International Edition 2008, 47, 4628–4629. [Google Scholar] [CrossRef]
- Miyazawa, M.; Sonoyama, M. Second Derivative near Infrared Studies on the Structural Characterisation of Proteins. Journal of Near Infrared Spectroscopy 1998, 6, A253–A257. [Google Scholar] [CrossRef]
- Beć, K.B.; Huck, C.W. Breakthrough Potential in Near-Infrared Spectroscopy: Spectra Simulation. A Review of Recent Developments. Front Chem 2019, 7, 48. [Google Scholar] [CrossRef]
- Beć, K.B.; Grabska, J.; Huck, C.W. Principles and Applications of Miniaturized Near-Infrared (NIR) Spectrometers. Chemistry – A European Journal 2021, 27, 1514–1532. [Google Scholar] [CrossRef]
- Antila, J.; Tuohiniemi, M.; Rissanen, A.; Kantojärvi, U.; Lahti, M.; Viherkanto, K.; Kaarre, M.; Malinen, J. MEMS- and MOEMS-Based Near-Infrared Spectrometers. In Encyclopedia of Analytical Chemistry; 2014; pp. 1-36.
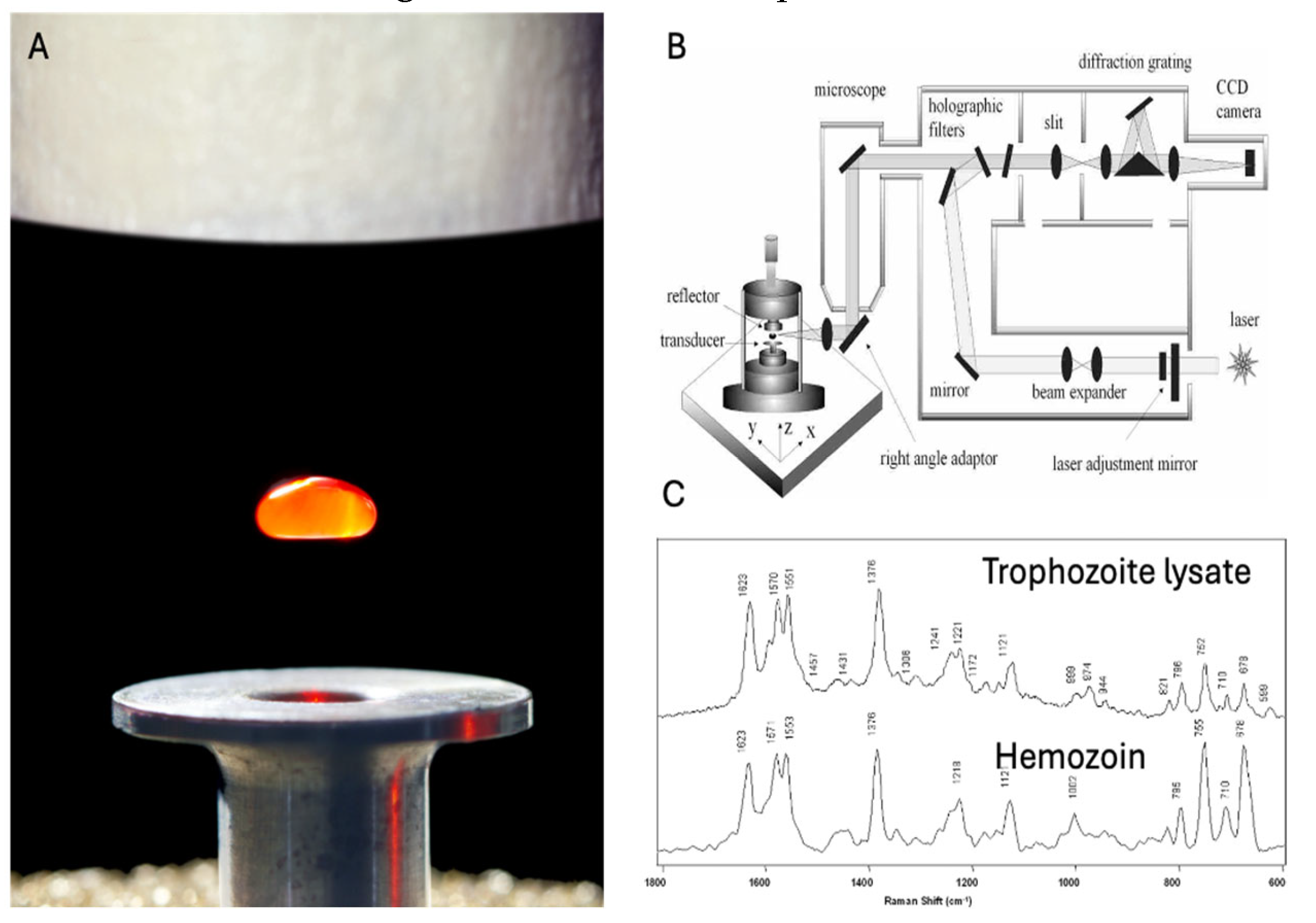
- Wood, B.R.; McNaughton, D. Resonance Raman spectroscopy in malaria research. Expert Review of Proteomics 2006, 3, 525–544. [Google Scholar] [CrossRef] [PubMed]
- Luthra, A.; Denisov, I.G.; Sligar, S.G. Spectroscopic features of cytochrome P450 reaction intermediates. Archives of Biochemistry and Biophysics 2011, 507, 26–35. [Google Scholar] [CrossRef]
- Ong, C.W.; Shen, Z.X.; Ang, K.K.H.; Kara, U.A.K.; Tang, S.H. Ong, C.W.; Shen, Z.X.; Ang, K.K.H.; Kara, U.A.K.; Tang, S.H. Resonance Raman Microspectroscopy of Normal Erythrocytes and Plasmodium Berghei-Infected Erythrocytes. http://dx.doi.org/10.1366/0003702991947874 1999, 53, 1097-1101. [CrossRef]
- Wood, B.R.; Langford, S.J.; Cooke, B.M.; Glenister, F.K.; Lim, J.; McNaughton, D. Raman imaging of hemozoin within the food vacuole of Plasmodium falciparum trophozoites. FEBS Letters 2003, 554, 247–252. [Google Scholar] [CrossRef]
- Wood, B.R.; Hermelink, A.; Lasch, P.; Bambery, K.R.; Webster, G.T.; Khiavi, M.A.; Cooke, B.M.; Deed, S.; Naumann, D.; McNaughton, D. Resonance Raman microscopy in combination with partial dark-field microscopy lights up a new path in malaria diagnostics. Analyst 2009, 134, 1119–1125. [Google Scholar] [CrossRef]
- Puskar, L.; Tuckermann, R.; Frosch, T.; Popp, J.; Ly, V.; McNaughton, D.; Wood, B.R. Raman acoustic levitation spectroscopy of red blood cells and Plasmodium falciparum trophozoites. Lab on a Chip 2007, 7, 1125–1131. [Google Scholar] [CrossRef]
- Kang, J.W.; Lue, N.; Kong, C.-R.; Barman, I.; Dingari, N.C.; Goldfless, S.J.; Niles, J.C.; Dasari, R.R.; Feld, M.S.; Choi, W.; et al. Combined confocal Raman and quantitative phase microscopy system for biomedical diagnosis. Biomedical Optics Express, Vol. 2, Issue 9, pp. 2484-2492 2011, 2, 2484–2492. [Google Scholar] [CrossRef]
- Brückner, M.; Becker, K.; Popp, J.; Frosch, T. Fiber array based hyperspectral Raman imaging for chemical selective analysis of malaria-infected red blood cells. Analytica Chimica Acta 2015, 894, 76–84. [Google Scholar] [CrossRef]
- Kozicki, M.; Czepiel, J.; Biesiada, G.; Nowak, P.; Garlicki, A.; Wesełucha-Birczyńska, A. The ring-stage of Plasmodium falciparum observed in RBCs of hospitalized malaria patients. Analyst 2015, 140, 8007–8016. [Google Scholar] [CrossRef]
- Patel, S.K.; Rajora, N.; Kumar, S.; Sahu, A.; Kochar, S.K.; Krishna, C.M.; Srivastava, S. Rapid Discrimination of Malaria- and Dengue-Infected Patients Sera Using Raman Spectroscopy. Analytical Chemistry 2019, 91, 7054–7062. [Google Scholar] [CrossRef]
- Hackett, M.J.; Aitken, J.B.; El-Assaad, F.; McQuillan, J.A.; Carter, E.A.; Ball, H.J.; Tobin, M.J.; Paterson, D.; De Jonge, M.D.; Siegele, R.; et al. Neuroscience: Mechanisms of murine cerebral malaria: Multimodal imaging of altered cerebral metabolism and protein oxidation at hemorrhage sites. Science Advances 2015, 1. [Google Scholar] [CrossRef]
- Frame, L.; Brewer, J.; Lee, R.; Faulds, K.; Graham, D. Development of a label-free Raman imaging technique for differentiation of malaria parasite infected from non-infected tissue. Analyst 2017, 143, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Hobro, A.J.; Pavillon, N.; Fujita, K.; Ozkan, M.; Coban, C.; Smith, N.I. Label-free Raman imaging of the macrophage response to the malaria pigment hemozoin. Analyst 2015, 140, 2350–2359. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, D.J. Introduction to Raman Scattering. In Practical Raman Spectroscopy; Gardiner, D.J., Graves, P.R., Eds.; Springer Berlin Heidelberg: Berlin, Heidelberg, 1989; pp. 1–12. [Google Scholar]
- Bell, S.E.J.; Charron, G.; Cortés, E.; Kneipp, J.; de la Chapelle, M.L.; Langer, J.; Procházka, M.; Tran, V.; Schlücker, S. Towards Reliable and Quantitative Surface-Enhanced Raman Scattering (SERS): From Key Parameters to Good Analytical Practice. Angewandte Chemie International Edition 2020, 59, 5454–5462. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Flaherty, B.R.; Cohen, C.E.; Peterson, D.S.; Zhao, Y. Direct detection of malaria infected red blood cells by surface enhanced Raman spectroscopy. Nanomedicine 2016, 12, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Perlaki, C.; Xiong, A.; Preiser, P.; Liu, Q. Review of Surface Enhanced Raman Spectroscopy for Malaria Diagnosis and a New Approach for the Detection of Single Parasites in the Ring Stage. IEEE Journal of Selected Topics in Quantum Electronics 2016, 22, 179–187. [Google Scholar] [CrossRef]
- Garrett, N.L.; Sekine, R.; Dixon, M.W.; Tilley, L.; Bambery, K.R.; Wood, B.R. Bio-sensing with butterfly wings: naturally occurring nano-structures for SERS-based malaria parasite detection. Phys Chem Chem Phys 2015, 17, 21164–21168. [Google Scholar] [CrossRef]
- Radziuk, D.; Moehwald, H. Highly effective hot spots for SERS signatures of live fibroblasts. Nanoscale 2014, 6, 6115–6126. [Google Scholar] [CrossRef]
- Heidelberger, M.; Prout, C.; Hindle, J.A.; Rose, A.S. Studies in Human Malaria1: III. An Attempt at Vaccination of Paretics Against Blood-Borne Infection with Pl. Vivax. The Journal of Immunology 1946, 53, 109–112. [Google Scholar] [CrossRef]
- Liu, Q.; Yuen, C.; Chen, K.; Ju, J.; Xiong, A.; Preiser, P. Surface enhanced Raman spectroscopy for malaria diagnosis and intradermal measurements; SPIE: 2018; Volume 10509.
- Chen, K.; Yuen, C.; Aniweh, Y.; Preiser, P.; Liu, Q. Towards ultrasensitive malaria diagnosis using surface enhanced Raman spectroscopy. Sci Rep 2016, 6, 20177. [Google Scholar] [CrossRef]
- Wang, W.; Dong, R.-l.; Gu, D.; He, J.-a.; Yi, P.; Kong, S.-K.; Ho, H.-P.; Loo, J.; Wang, W.; Wang, Q. Antibody-free rapid diagnosis of malaria in whole blood with surface-enhanced Raman Spectroscopy using Nanostructured Gold Substrate. Advances in Medical Sciences 2020, 65, 86–92. [Google Scholar] [CrossRef]
- Yuen, C.; Gao, X.; Yong, J.J.M.; Prakash, P.; Shobana, C.R.; Kaushalya, P.A.T.; Luo, Y.; Bai, Y.; Yang, C.; Preiser, P.R.; et al. Towards malaria field diagnosis based on surface-enhanced Raman scattering with on-chip sample preparation and near-analyte nanoparticle synthesis. Sensors and Actuators B: Chemical 2021, 343, 130162. [Google Scholar] [CrossRef]
- Yuen, C.; Liu, Q. Optimization of Fe3O4@Ag nanoshells in magnetic field-enriched surface-enhanced resonance Raman scattering for malaria diagnosis. Analyst 2013, 138, 6494–6500. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Perlaki, C.; Xiong, A.; Preiser, P.; Liu, Q. Review of Surface Enhanced Raman Spectroscopy for Malaria Diagnosis and a New Approach for the Detection of Single Parasites in the Ring Stage. Ieee Journal of Selected Topics in Quantum Electronics 2016, 22. [Google Scholar] [CrossRef]
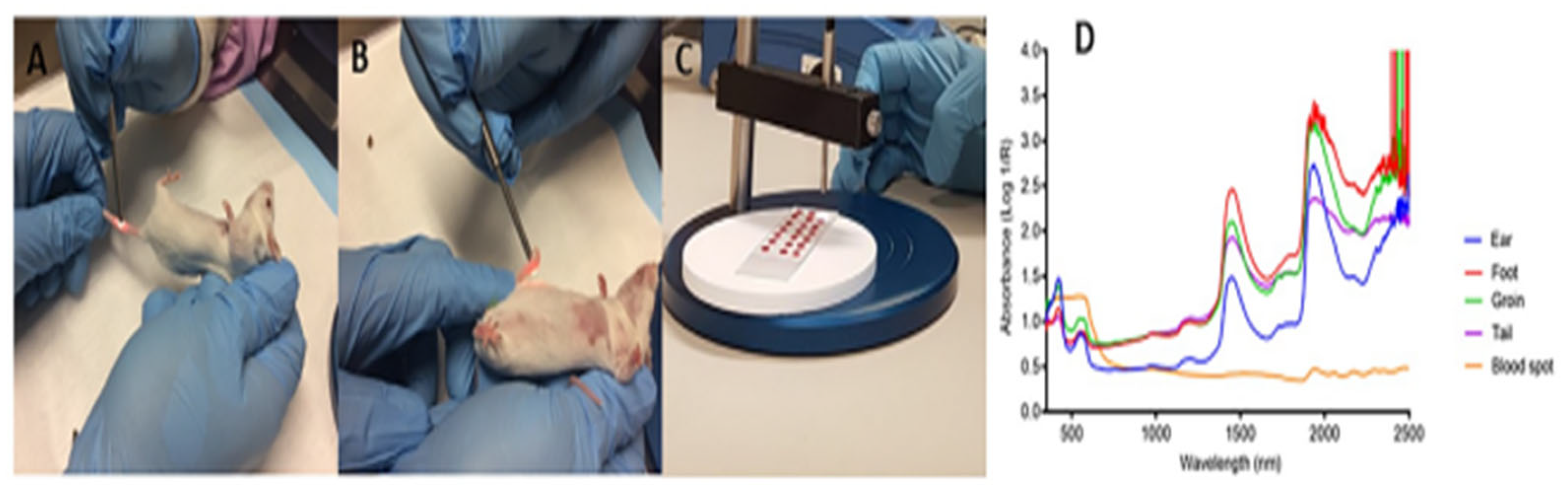
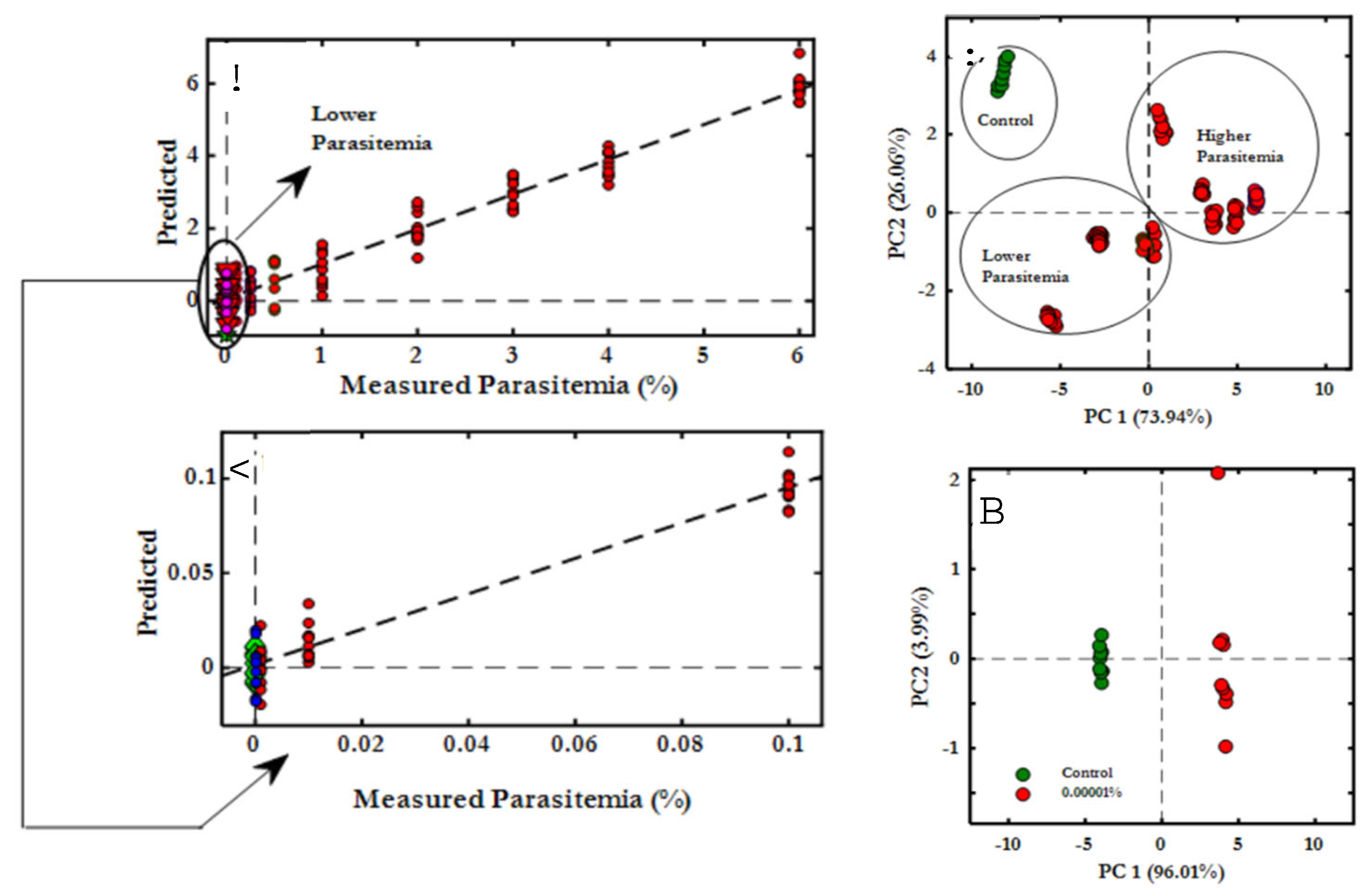
- Martin, M.; Perez-Guaita, D.; Andrew, D.W.; Richards, J.S.; Wood, B.R.; Heraud, P. The effect of common anticoagulants in detection and quantification of malaria parasitemia in human red blood cells by ATR-FTIR spectroscopy. Analyst 2017, 142, 1192–1199. [Google Scholar] [CrossRef]
- Roy, S.; Perez-Guaita, D.; Andrew, D.W.; Richards, J.S.; McNaughton, D.; Heraud, P.; Wood, B.R. Simultaneous ATR-FTIR Based Determination of Malaria Parasitemia, Glucose and Urea in Whole Blood Dried onto a Glass Slide. Anal Chem 2017, 89, 5238–5245. [Google Scholar] [CrossRef]
- Mwanga, E.P.; Minja, E.G.; Mrimi, E.; Jimenez, M.G.; Swai, J.K.; Abbasi, S.; Ngowo, H.S.; Siria, D.J.; Mapua, S.; Stica, C.; et al. Detection of malaria parasites in dried human blood spots using mid-infrared spectroscopy and logistic regression analysis. Malar J 2019, 18, 341. [Google Scholar] [CrossRef]
- Perez-Guaita, D.; Perez-Guaita, D.; Andrew, D.; Heraud, P.; Beeson, J.; Anderson, D.; Richards, J.; Wood, B.R. High resolution FTIR imaging provides automated discrimination and detection of single malaria parasite infected erythrocytes on glass. Faraday discussions. 2016, 187, 341–352. [Google Scholar] [CrossRef]
- Perez-Guaita, D.; Kochan, K.; Martin, M.; Andrew, D.W.; Heraud, P.; Richards, J.S.; Wood, B.R. Multimodal vibrational imaging of cells. Vibrational Spectroscopy 2017, 91, 46–58. [Google Scholar] [CrossRef]
- Banas, A.M.; Banas, K.; Chu, T.T.T.; Naidu, R.; Hutchinson, P.E.; Agrawal, R.; Lo, M.K.F.; Kansiz, M.; Roy, A.; Chandramohanadas, R.; et al. Comparing infrared spectroscopic methods for the characterization of Plasmodium falciparum-infected human erythrocytes. Commun Chem 2021, 4, 129. [Google Scholar] [CrossRef]
- Idro, R.; Jenkins, N.E.; Newton, C.R.J.C. Pathogenesis, clinical features, and neurological outcome of cerebral malaria. The Lancet Neurology 2005, 4, 827–840. [Google Scholar] [CrossRef]
- Dvorin, J.D. Getting Your Head around Cerebral Malaria. Cell Host & Microbe 2017, 22, 586–588. [Google Scholar] [CrossRef] [PubMed]
- Hackett, M.J.; Aitken, J.B.; El-Assaad, F.; McQuillan, J.A.; Carter, E.A.; Ball, H.J.; Tobin, M.J.; Paterson, D.; De Jonge, M.D.; Siegele, R.; et al. Mechanisms of murine cerebral malaria: Multimodal imaging of altered cerebral metabolism and protein oxidation at hemorrhage sites. Science Advances 2015, 1, e1500911. [Google Scholar] [CrossRef] [PubMed]
- Hackett, M.J.; Lee, J.; El-Assaad, F.; McQuillan, J.A.; Carter, E.A.; Grau, G.E.; Hunt, N.H.; Lay, P.A. FTIR Imaging of Brain Tissue Reveals Crystalline Creatine Deposits Are an ex Vivo Marker of Localized Ischemia during Murine Cerebral Malaria: General Implications for Disease Neurochemistry. ACS Chemical Neuroscience 2012, 3, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Wood, B.R.; Bambery, K.R.; Dixon, M.W.A.; Tilley, L.; Nasse, M.J.; Mattson, E.; Hirschmugl, C.J. Diagnosing malaria infected cells at the single cell level using focal plane array Fourier transform infrared imaging spectroscopy. The Analyst 2014, 139, 4769–4774. [Google Scholar] [CrossRef]
- Lasch, P.; Naumann, D. Spatial resolution in infrared microspectroscopic imaging of tissues. Biochimica et Biophysica Acta (BBA) - Biomembranes 2006, 1758, 814–829. [Google Scholar] [CrossRef]
- Edgar, R.C.S.; Counihan, N.A.; McGowan, S.; de Koning-Ward, T.F. Methods Used to Investigate the Plasmodium falciparum Digestive Vacuole. Front Cell Infect Microbiol 2021, 11, 829823. [Google Scholar] [CrossRef]
- Shibeshi, M.A.; Kifle, Z.D.; Atnafie, S.A. Antimalarial Drug Resistance and Novel Targets for Antimalarial Drug Discovery. Infect Drug Resist 2020, 13, 4047–4060. [Google Scholar] [CrossRef]
- Matz, J.M. Plasmodium’s bottomless pit: properties and functions of the malaria parasite's digestive vacuole. Trends in Parasitology 2022, 38, 525–543. [Google Scholar] [CrossRef]
- Noland, G.S.; Briones, N.; Sullivan, D.J. The shape and size of hemozoin crystals distinguishes diverse Plasmodium species. Molecular and Biochemical Parasitology 2003, 130, 91–99. [Google Scholar] [CrossRef]
- Bannister, L.H.; Hopkins, J.M.; Margos, G.; Dluzewski, A.R.; Mitchell, G.H. Three-Dimensional Ultrastructure of the Ring Stage ofPlasmodium falciparum: Evidence for Export Pathways. Microscopy and Microanalysis 2004, 10, 551–562. [Google Scholar] [CrossRef]
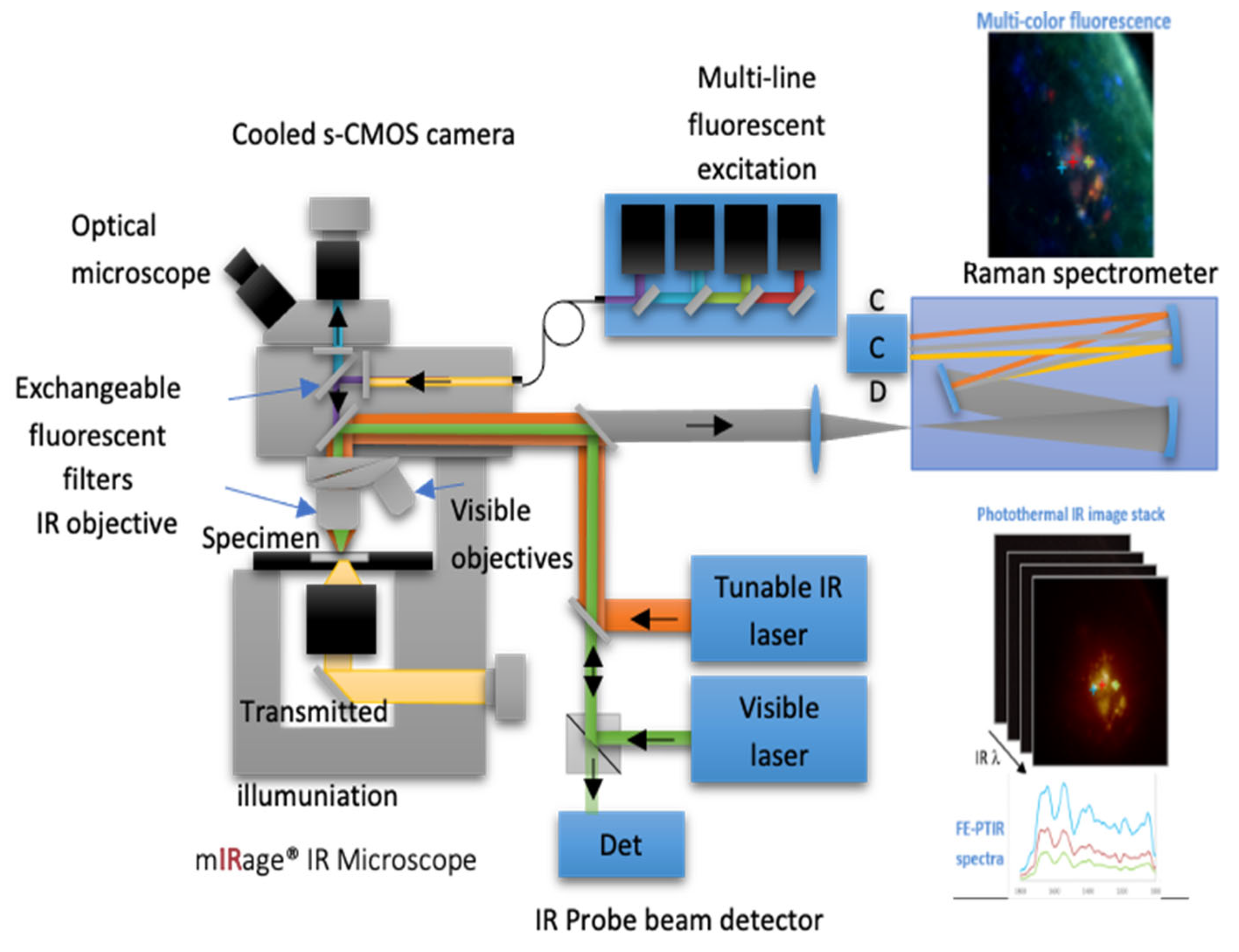
- Kansiz, M.; Prater, C.; Dillon, E.; Lo, M.; Anderson, J.; Marcott, C.; Demissie, A.; Chen, Y.; Kunkel, G. Optical Photothermal Infrared Microspectroscopy with Simultaneous Raman – A New Non-Contact Failure Analysis Technique for Identification of <10 μm Organic Contamination in the Hard Drive and other Electronics Industries. Microscopy Today 2020, 28, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Reffner, J. Advances in infrared microspectroscopy and mapping molecular chemical composition at submicrometer spatial resolution. Spectroscopy (Santa Monica) 2018, 33, 12–17. [Google Scholar]
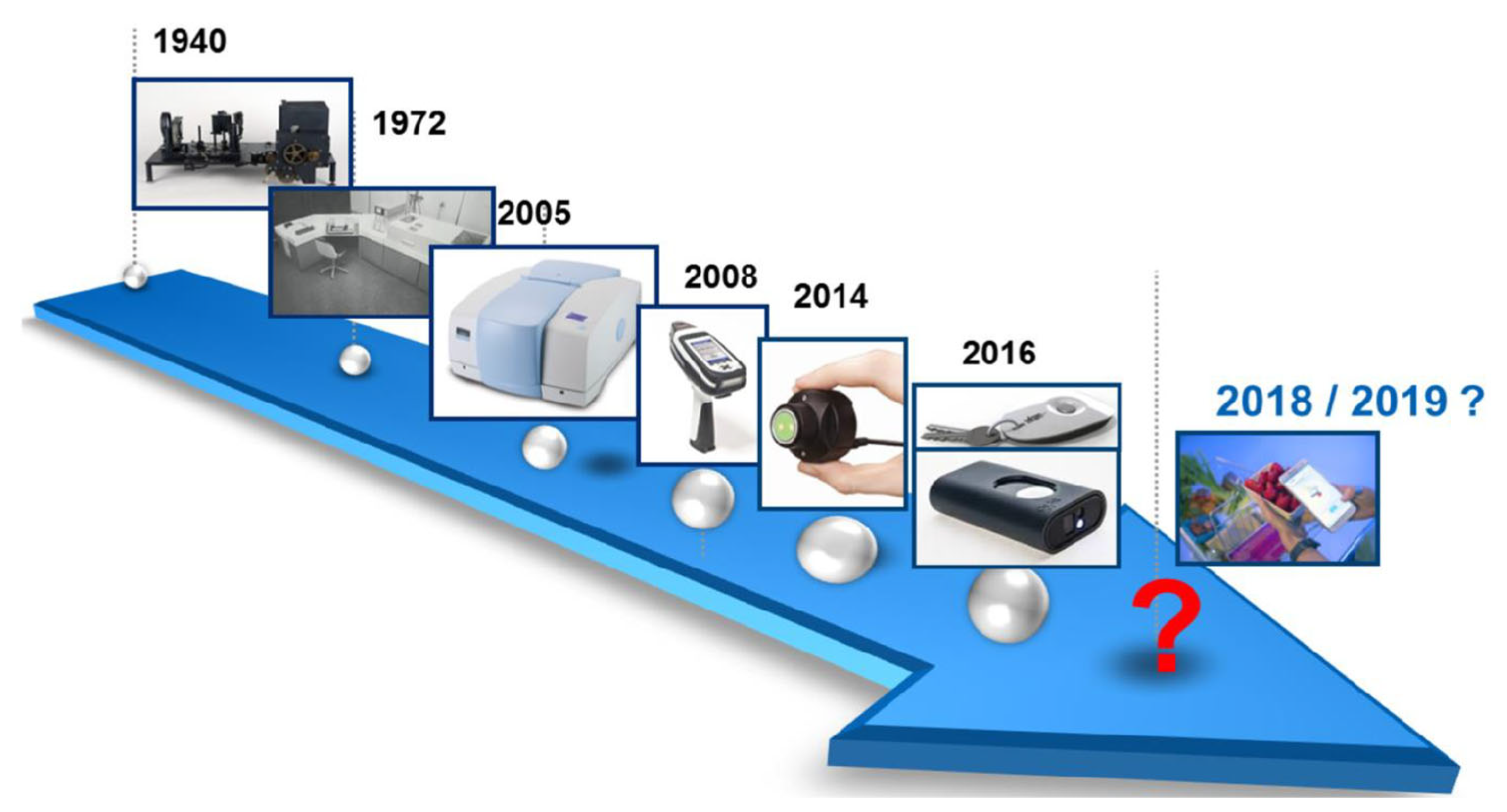
- Mclean, A.; Veettil, T.C.P.; Giergiel, M.; Wood, B.R. Evolution of Vibrational Biospectroscopy: Multimodal techniques and Miniaturisation supported by Machine Learning. Vibrational Spectroscopy 2024, 103708. [Google Scholar] [CrossRef]
- Manley, M. Near-infrared spectroscopy and hyperspectral imaging: non-destructive analysis of biological materials. Chem Soc Rev 2014, 43, 8200–8214. [Google Scholar] [CrossRef]
- Blanco, M.; Villarroya, I. NIR spectroscopy: a rapid-response analytical tool. TrAC Trends in Analytical Chemistry 2002, 21, 240–250. [Google Scholar] [CrossRef]
- Reich, G. Near-infrared spectroscopy and imaging: basic principles and pharmaceutical applications. Adv Drug Deliv Rev 2005, 57, 1109–1143. [Google Scholar] [CrossRef]
- Schwanninger, M.; Rodrigues, J.C.; Fackler, K. A Review of Band Assignments in near Infrared Spectra of Wood and Wood Components. Journal of Near Infrared Spectroscopy 2011, 19, 287–308. [Google Scholar] [CrossRef]
- Huck, C.W.; Ozaki, Y.; Huck-Pezzei, V.A. Critical Review Upon the Role and Potential of Fluorescence and Near-Infrared Imaging and Absorption Spectroscopy in Cancer Related Cells, Serum, Saliva, Urine and Tissue Analysis. Current medicinal chemistry 2016, 23, 3052–3077. [Google Scholar] [CrossRef]
- Manley, M. Near-infrared spectroscopy and hyperspectral imaging: non-destructive analysis of biological materials. Chemical Society Reviews 2014, 43, 8200–8214. [Google Scholar] [CrossRef]
- Reich, G. Near-infrared spectroscopy and imaging: Basic principles and pharmaceutical applications. Advanced Drug Delivery Reviews 2005, 57, 1109–1143. [Google Scholar] [CrossRef]
- Ozaki, Y. Near-infrared spectroscopy--its versatility in analytical chemistry. Anal Sci 2012, 28, 545–563. [Google Scholar] [CrossRef] [PubMed]
- Beć, K.B.; Huck, C.W. Breakthrough Potential in Near-Infrared Spectroscopy: Spectra Simulation. A Review of Recent Developments.
- Beć, K.B.; Grabska, J.; Huck, C.W. The essential role of omni-capable research laboratories in advancing analytical spectroscopy. NIR news 2019, 30, 30–34. [Google Scholar] [CrossRef]
- Sakudo, A. Near-infrared spectroscopy for medical applications: Current status and future perspectives. Clin Chim Acta 2016, 455, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Domján, G.; Kaffka, K.J.; Jákó, J.M.; Vályi-Nagy, I.T. Rapid Analysis of Whole Blood and Blood Serum Using near Infrared Spectroscopy. Journal of Near Infrared Spectroscopy 1994, 2, 67–78. [Google Scholar] [CrossRef]
- Lukianova-Hleb, E.Y.; Lapotko, D.O. Malaria theranostics using hemozoin-generated vapor nanobubbles. Theranostics 2014, 4, 761–769. [Google Scholar] [CrossRef]
- Maia, M.F.; Kapulu, M.; Muthui, M.; Wagah, M.G.; Ferguson, H.M.; Dowell, F.E.; Baldini, F.; Ranford-Cartwright, L. Detection of Plasmodium falciparum infected Anopheles gambiae using near-infrared spectroscopy. Malar J 2019, 18, 85. [Google Scholar] [CrossRef]
- Da, D.F.; McCabe, R.; Some, B.M.; Esperanca, P.M.; Sala, K.A.; Blight, J.; Blagborough, A.M.; Dowell, F.; Yerbanga, S.R.; Lefevre, T.; et al. Detection of Plasmodium falciparum in laboratory-reared and naturally infected wild mosquitoes using near-infrared spectroscopy. Sci Rep 2021, 11, 10289. [Google Scholar] [CrossRef]
- Ong, O.T.W.; Kho, E.A.; Esperança, P.M.; Freebairn, C.; Dowell, F.E.; Devine, G.J.; Churcher, T.S. Ability of near-infrared spectroscopy and chemometrics to predict the age of mosquitoes reared under different conditions. Parasites & Vectors 2020, 13, 160. [Google Scholar] [CrossRef]
- Fernandes, J.N.; Dos Santos, L.M.B.; Chouin-Carneiro, T.; Pavan, M.G.; Garcia, G.A.; David, M.R.; Beier, J.C.; Dowell, F.E.; Maciel-de-Freitas, R.; Sikulu-Lord, M.T. Rapid, noninvasive detection of Zika virus in Aedes aegypti mosquitoes by near-infrared spectroscopy. Sci Adv 2018, 4, eaat0496. [Google Scholar] [CrossRef]
- Garcia, G.A.; Lord, A.R.; Santos, L.M.B.; Kariyawasam, T.N.; David, M.R.; Couto-Lima, D.; Tátila-Ferreira, A.; Pavan, M.G.; Sikulu-Lord, M.T.; Maciel-de-Freitas, R. Rapid and Non-Invasive Detection of Aedes aegypti Co-Infected with Zika and Dengue Viruses Using Near Infrared Spectroscopy. Viruses 2022, 15. [Google Scholar] [CrossRef]
- Some, B.M.; Da, D.F.; McCabe, R.; Djegbe, N.D.C.; Pare, L.I.G.; Werme, K.; Mouline, K.; Lefevre, T.; Ouedraogo, A.G.; Churcher, T.S.; et al. Adapting field-mosquito collection techniques in a perspective of near-infrared spectroscopy implementation. Parasit Vectors 2022, 15, 338. [Google Scholar] [CrossRef] [PubMed]
- Lambert, B.; Sikulu-Lord, M.T.; Mayagaya, V.S.; Devine, G.; Dowell, F.; Churcher, T.S. Monitoring the Age of Mosquito Populations Using Near-Infrared Spectroscopy. Sci Rep 2018, 8, 5274. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.C.D.; Viana, J.L.S.; Monteiro, J.D.; Freire, R.C.M.; Freitas, D.L.D.; Câmara, I.M.; da Silva, G.J.S.; Gama, R.A.; Araújo, J.M.G.; Lima, K.M.G. Infrared spectroscopy (NIRS and ATR-FTIR) together with multivariate classification for non-destructive differentiation between female mosquitoes of Aedes aegypti recently infected with dengue vs. uninfected females. Acta Tropica 2022, 235, 106633. [Google Scholar] [CrossRef] [PubMed]
- Sikulu-Lord, M.T.; Edstein, M.D.; Goh, B.; Lord, A.R.; Travis, J.A.; Dowell, F.E.; Birrell, G.W.; Chavchich, M. Rapid and non-invasive detection of malaria parasites using near-infrared spectroscopy and machine learning. PLoS One 2024, 19, e0289232. [Google Scholar] [CrossRef]
- Adegoke, J.A.; De Paoli, A.; Afara, I.O.; Kochan, K.; Creek, D.J.; Heraud, P.; Wood, B.R. Ultraviolet/Visible and Near-Infrared Dual Spectroscopic Method for Detection and Quantification of Low-Level Malaria Parasitemia in Whole Blood. Anal Chem 2021, 93, 13302–13310. [Google Scholar] [CrossRef]
- Briehl, R.W.; Hobbs, J.F. Ultraviolet Difference Spectra in Human Hemoglobin: I. DIFFERENCE SPECTRA IN HEMOGLOBIN A AND THEIR RELATION TO THE FUNCTION OF HEMOGLOBIN. Journal of Biological Chemistry 1970, 245, 544–554. [Google Scholar] [CrossRef]
- Briehl, R.W.; Ranney, H.M. Ultraviolet Difference Spectra in Human Hemoglobin: II. DIFFERENCE SPECTRA IN ISOLATED SUBUNITS OF HEMOGLOBIN. Journal of Biological Chemistry 1970, 245, 555–558. [Google Scholar] [CrossRef]
- Serebrennikova, Y.M.; Patel, J.; Garcia-Rubio, L.H. Interpretation of the ultraviolet-visible spectra of malaria parasite Plasmodium falciparum. Appl. Opt. 2010, 49, 180–188. [Google Scholar] [CrossRef]
- Frosch, T.; Koncarevic, S.; Becker, K.; Popp, J. Morphology-sensitive Raman modes of the malaria pigment hemozoin. Analyst 2009, 134, 1126–1132. [Google Scholar] [CrossRef]
- Queral-Beltran, A.; Marín-García, M.; Lacorte, S.; Tauler, R. Multivariate curve resolution of incomplete and partly trilinear multiblock datasets. Chemometrics and Intelligent Laboratory Systems 2024, 247, 105081. [Google Scholar] [CrossRef]
- Scepanovic, O.R.; Volynskaya, Z.; Kong, C.R.; Galindo, L.H.; Dasari, R.R.; Feld, M.S. A multimodal spectroscopy system for real-time disease diagnosis. Rev Sci Instrum 2009, 80, 043103. [Google Scholar] [CrossRef] [PubMed]
- Szymoński, K.; Chmura, Ł.; Lipiec, E.; Adamek, D. Vibrational spectroscopy - are we close to finding a solution for early pancreatic cancer diagnosis? World J Gastroenterol 2023, 29, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, D.J., Graves, P.R., Eds.Sensitivity of photoacoustic microscopy. [CrossRef]
- Memeu, D.M.; Sallorey, A.M.; Maina, C.; Kinyua, D.M. Review of Photoacoustic Malaria Diagnostic Techniques. Open Journal of Clinical Diagnostics 2021, 11, 59–75. [Google Scholar] [CrossRef]
- Samson, E.B.; Goldschmidt, B.S.; Whiteside, P.J.; Sudduth, A.S.; Custer, J.R.; Beerntsen, B.; Viator, J.A. Photoacoustic spectroscopy of β-hematin. J Opt 2012, 14. [Google Scholar] [CrossRef]
- Menyaev, Y.A.; Carey, K.A.; Nedosekin, D.A.; Sarimollaoglu, M.; Galanzha, E.I.; Stumhofer, J.S.; Zharov, V.P. Preclinical photoacoustic models: application for ultrasensitive single cell malaria diagnosis in large vein and artery. Biomed Opt Express 2016, 7, 3643–3658. [Google Scholar] [CrossRef]
- Cai, C.; Carey, K.A.; Nedosekin, D.A.; Menyaev, Y.A.; Sarimollaoglu, M.; Galanzha, E.I.; Stumhofer, J.S.; Zharov, V.P. In vivo photoacoustic flow cytometry for early malaria diagnosis. Cytometry A 2016, 89, 531–542. [Google Scholar] [CrossRef]
- Lukianova-Hleb, E.; Bezek, S.; Szigeti, R.; Khodarev, A.; Kelley, T.; Hurrell, A.; Berba, M.; Kumar, N.; D'Alessandro, U.; Lapotko, D. Transdermal Diagnosis of Malaria Using Vapor Nanobubbles. Emerg Infect Dis 2015, 21, 1122–1127. [Google Scholar] [CrossRef]
- Dean-Ben, X.L.; Gottschalk, S.; Mc Larney, B.; Shoham, S.; Razansky, D. Advanced optoacoustic methods for multiscale imaging of in vivo dynamics. Chem Soc Rev 2017, 46, 2158–2198. [Google Scholar] [CrossRef]
- Veverka, M.; Menozzi, L.; Yao, J. The sound of blood: photoacoustic imaging in blood analysis. Med Nov Technol Devices 2023, 18. [Google Scholar] [CrossRef]
- Hu, S.; Wang, V. , Lihong. Optical-Resolution Photoacoustic Microscopy: Auscultation of Biological Systems at the Cellular Level. Biophysical Journal 2013, 105, 841–847. [Google Scholar] [CrossRef]
- Park, S.; Lee, C.; Kim, J.; Kim, C. Acoustic resolution photoacoustic microscopy. Biomedical Engineering Letters 2014, 4, 213–222. [Google Scholar] [CrossRef]
- Zhou, Y.; Yao, J.; Wang, L.V. Tutorial on photoacoustic tomography. Journal of Biomedical Optics 2016, 21, 061007. [Google Scholar] [CrossRef] [PubMed]
- Periyasamy, V.; Das, N.; Sharma, A.; Pramanik, M. 1064 nm acoustic resolution photoacoustic microscopy. Journal of Biophotonics 2019, 12, e201800357. [Google Scholar] [CrossRef]
- Tian, C.; Zhang, C.; Zhang, H.; Xie, D.; Jin, Y. Spatial resolution in photoacoustic computed tomography. Reports on Progress in Physics 2021, 84, 036701. [Google Scholar] [CrossRef]
- Moothanchery, M.; Bi, R.; Kim, J.Y.; Jeon, S.; Kim, C.; Olivo, M. Optical resolution photoacoustic microscopy based on multimode fibers. Biomedical Optics Express 2018, 9, 1190. [Google Scholar] [CrossRef]
- Liu, W.; Yao, J. Photoacoustic microscopy: principles and biomedical applications. Biomedical Engineering Letters 2018, 8, 203–213. [Google Scholar] [CrossRef]
- Pleitez, M.A.; Khan, A.A.; Soldà, A.; Chmyrov, A.; Reber, J.; Gasparin, F.; Seeger, M.R.; Schätz, B.; Herzig, S.; Scheideler, M.; et al. Label-free metabolic imaging by mid-infrared optoacoustic microscopy in living cells. Nat Biotechnol 2020, 38, 293–296. [Google Scholar] [CrossRef]
- Perez-Guaita, D.; Kochan, K.; Batty, M.; Doerig, C.; Garcia-Bustos, J.; Espinoza, S.; McNaughton, D.; Heraud, P.; Wood, B.R. Multispectral Atomic Force Microscopy-Infrared Nano-Imaging of Malaria Infected Red Blood Cells. Analytical Chemistry 2018, 90, 3140–3148. [Google Scholar] [CrossRef]
- Wood, B.R.; Bailo, E.; Khiavi, M.A.; Tilley, L.; Deed, S.; Deckert-Gaudig, T.; McNaughton, D.; Deckert, V. Tip-Enhanced Raman Scattering (TERS) from Hemozoin Crystals within a Sectioned Erythrocyte. Nano Letters 2011, 11, 1868–1873. [Google Scholar] [CrossRef]
- Berna, A.Z.; Wang, X.R.; Bollinger, L.B.; Banda, J.; Mawindo, P.; Evanoff, T.; Culbertson, D.L.; Seydel, K.; John, A.R.O. Breath Biomarkers of Pediatric Malaria: Reproducibility and Response to Antimalarial Therapy. Journal of Infectious Diseases 2024. [Google Scholar] [CrossRef]
- Schaber, C.L.; Katta, N.; Bollinger, L.B.; Mwale, M.; Mlotha-Mitole, R.; Trehan, I.; Raman, B.; John, A.R.O. Breathprinting Reveals Malaria-Associated Biomarkers and Mosquito Attractants. Journal of Infectious Diseases 2018, 217, 1553–1560. [Google Scholar] [CrossRef] [PubMed]
- Chai, H.C.; Chua, K.H. Urine and Saliva: Relevant Specimens for Malaria Diagnosis? Diagnostics 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Wood, B.R.; Kochan, K.; Bedolla, D.E.; Salazar-Quiroz, N.; Grimley, S.L.; Perez-Guaita, D.; Baker, M.J.; Vongsvivut, J.; Tobin, M.J.; Bambery, K.R.; et al. Infrared Based Saliva Screening Test for COVID-19. Angewandte Chemie International Edition 2021, 60, 17102–17107. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Wongwattanakul, M.; Khemtonglang, N.; Kiatchoosakun, P.; Heraud, P.; Jearanaikoon, P.; Wood, B.R. Identification of Glucose-6 Phosphate Dehydrogenase Deficient Patients Using Attenuated Total Reflection Fourier Transform Infrared Spectroscopy Using Partial Least Squares Discriminant Analysis in Aqueous Blood Samples. Appl Spectrosc 2023, 77, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Perez-Guaita, D.; Marzec, K.M.; Hudson, A.; Evans, C.; Chernenko, T.; Matthäus, C.; Miljkovic, M.; Diem, M.; Heraud, P.; Richards, J.S.; et al. Parasites under the Spotlight: Applications of Vibrational Spectroscopy to Malaria Research. Chem Rev 2018, 118, 5330–5358. [Google Scholar] [CrossRef]
- Egan, T.J. Interactions of quinoline antimalarials with hematin in solution. J Inorg Biochem 2006, 100, 916–926. [Google Scholar] [CrossRef]
- Frosch, T.; Küstner, B.; Schlücker, S.; Szeghalmi, A.; Schmitt, M.; Kiefer, W.; Popp, J. In vitro polarization-resolved resonance Raman studies of the interaction of hematin with the antimalarial drug chloroquine. Journal of Raman Spectroscopy 2004, 35, 819–821. [Google Scholar] [CrossRef]
- Frosch, T.; Schmitt, M.; Bringmann, G.; Kiefer, W.; Popp, J. Structural Analysis of the Anti-Malaria Active Agent Chloroquine under Physiological Conditions. The Journal of Physical Chemistry B 2007, 111, 1815–1822. [Google Scholar] [CrossRef]
- Frosch, T.; Popp, J. Structural analysis of the antimalarial drug halofantrine by means of Raman spectroscopy and density functional theory calculations. J Biomed Opt 2010, 15, 041516. [Google Scholar] [CrossRef]
- Frosch, T.; Schmitt, M.; Schenzel, K.; Faber, J.H.; Bringmann, G.; Kiefer, W.; Popp, J. In vivo localization and identification of the antiplasmodial alkaloid dioncophylline A in the tropical liana Triphyophyllum peltatum by a combination of fluorescence, near infrared Fourier transform Raman microscopy, and density functional theory calculations. Biopolymers 2006, 82, 295–300. [Google Scholar] [CrossRef]
- Frosch, T.; Schmitt, M.; Noll, T.; Bringmann, G.; Schenzel, K.; Popp, J. Ultrasensitive in situ Tracing of the Alkaloid Dioncophylline A in the Tropical Liana Triphyophyllum peltatum by Applying Deep-UV Resonance Raman Microscopy. Analytical Chemistry 2007, 79, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Frosch, T.; Schmitt, M.; Popp, J. Raman spectroscopic investigation of the antimalarial agent mefloquine. Anal Bioanal Chem 2007, 387, 1749–1757. [Google Scholar] [CrossRef] [PubMed]
- Kozicki, M.; Creek, D.J.; Sexton, A.; Morahan, B.J.; Wesełucha-Birczyńska, A.; Wood, B.R. An attenuated total reflection (ATR) and Raman spectroscopic investigation into the effects of chloroquine on Plasmodium falciparum-infected red blood cells. Analyst 2015, 140, 2236–2246. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S.; Domes, R.; Domes, C.; Frosch, T. Spectrally Resolved and Highly Parallelized Raman Difference Spectroscopy for the Analysis of Drug–Target Interactions between the Antimalarial Drug Chloroquine and Hematin. Analytical Chemistry 2024, 96, 3345–3353. [Google Scholar] [CrossRef]
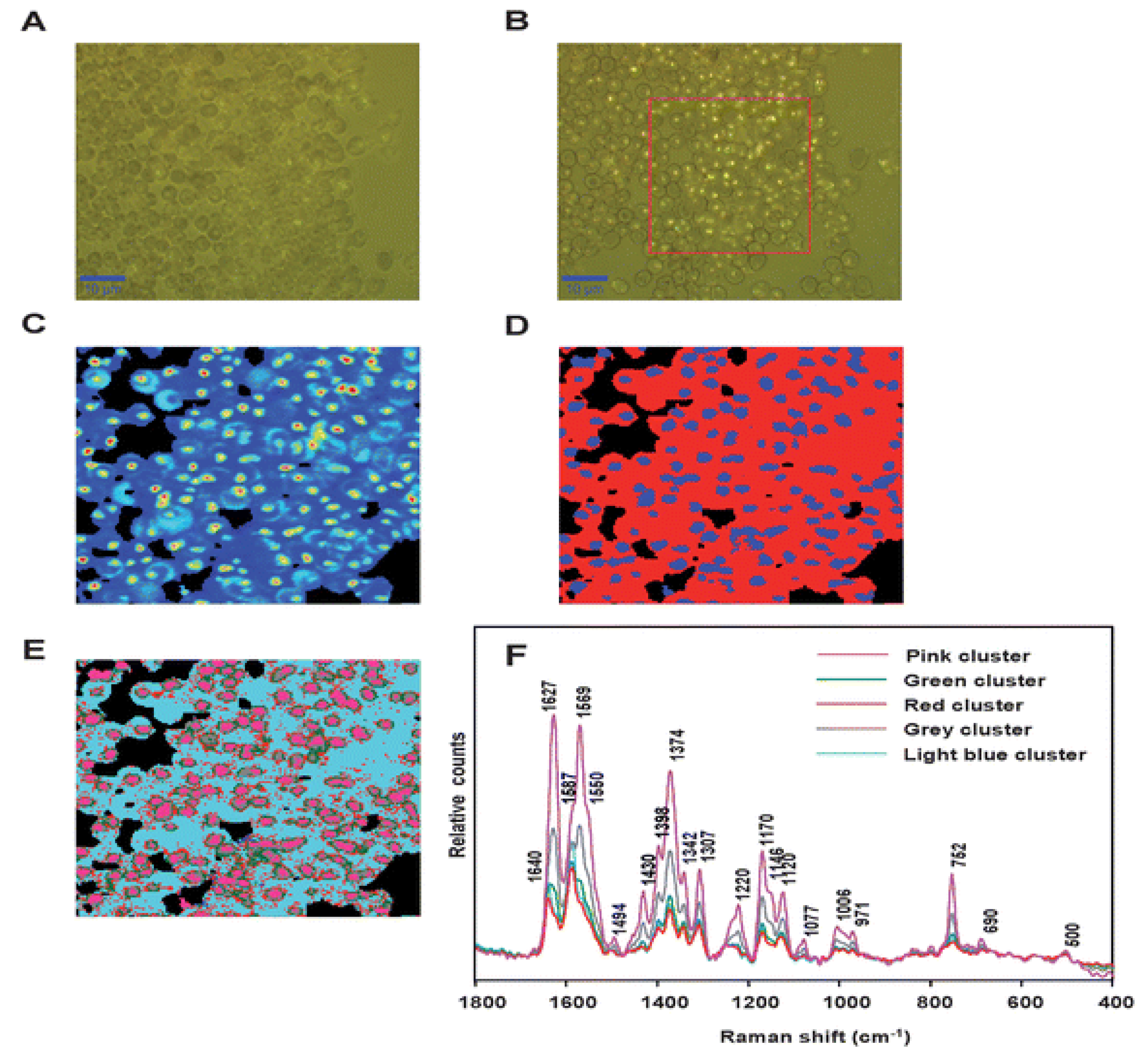
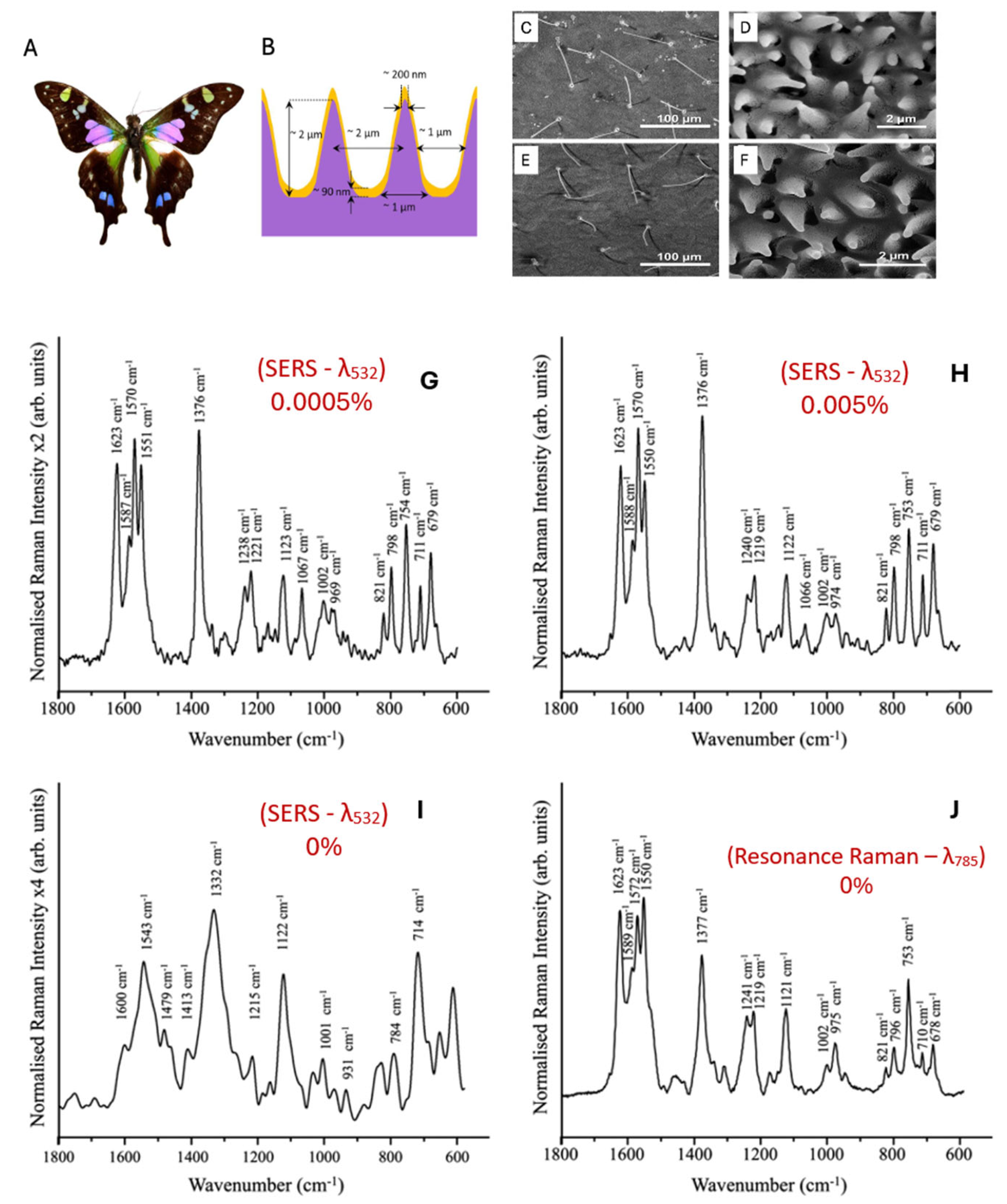
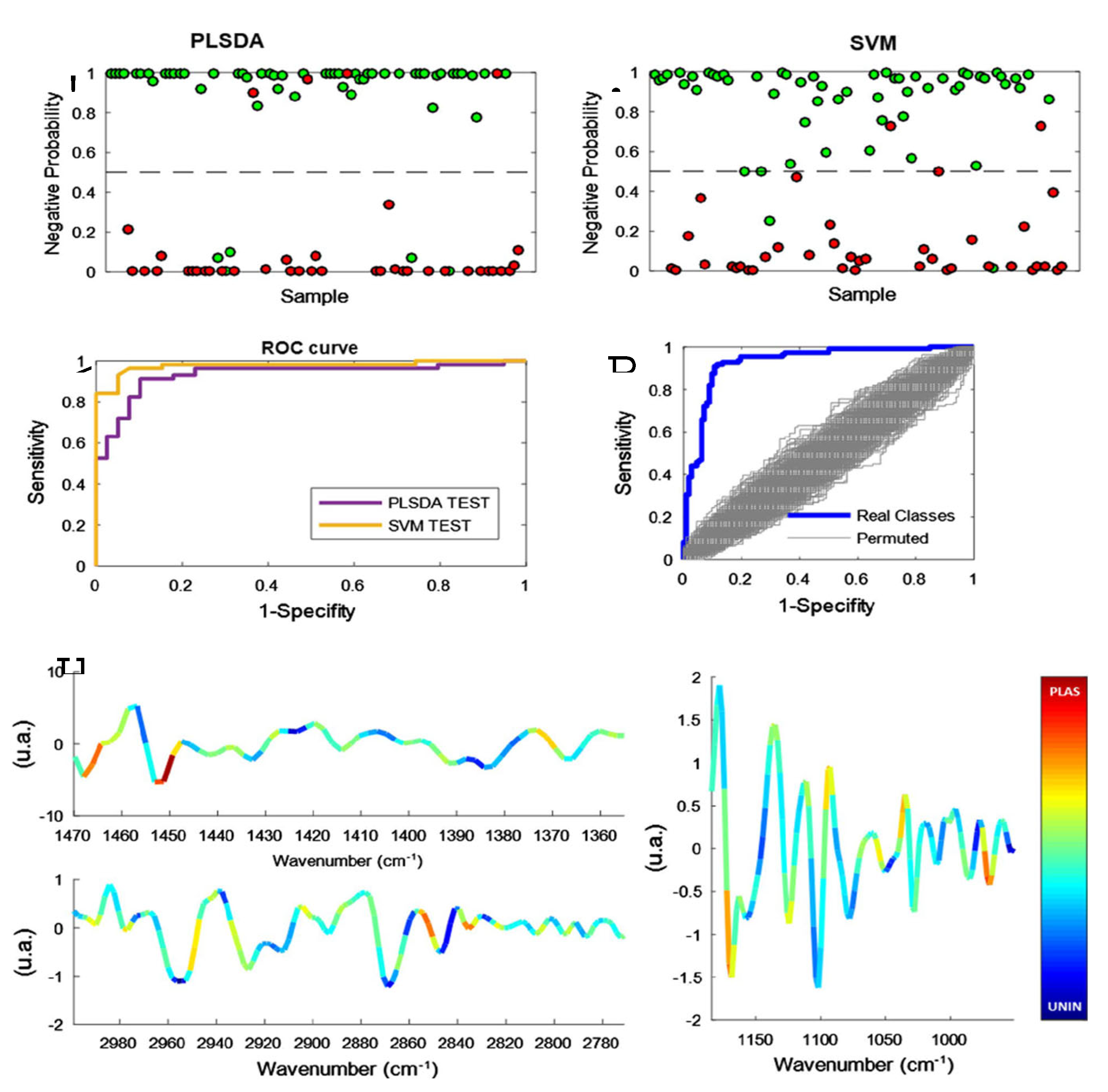
| Mode of detection | sensitivity | Methods | Citation |
| SERS – AgNP | 500 parasites/ µL (0.01% parasitemia level) |
silver nanoparticles were synthesized separately and then mixed with lysed blood | [85] |
| SERS – AgNP | 2.5 parasites/ µL (0.00005% parasitemia level) |
silver nanoparticles were synthesized directly inside the parasites of P. falciparum | [85] |
| SERS - AgNP | 1 parasite | AgNPs were synthesized inside P. falciparum. Giemsa stain was used to identify single parasite. | [89] |
| SERS -AgNP | 125 parasites/ µL (0.0025 % parasitemia) |
SERS-active silver nanoparticles are synthesized on site near hemozoin on a chip platform | [85] |
| Molecules | Absorption Bands (nm) |
| Protein Macromolecules (Amino acids) | |
| Tryptophan | 280 |
| Tyrosine | 275 |
| Phenylalanine | 258 |
| Haem group from RBCs | |
| Oxyhemoglobin | 416 |
| Oxyhemoglobin doublet | 540 |
| Oxyhemoglobin doublet | 575 |
| DNA nucleobases and chromophoric groups | |
| Guanine | 253 |
| Cytidine | 271 |
| Polymeric DNA and RNA | 260 |
| STUDY | SENSITIVITY | SPECIFICITY | CALIBRATION SET | VALIDATION SET |
| Heraud et al. , 2019 | 92 | 97 | Ntotal=232 Npositive=112 Nnegative=110 |
Ntotal=96 Npositive=39 Nnegatve=57 |
| Mwanga et al. , 2019 P. falciparum only |
92a | 93a | Ntotal=208 Npositive=86 Nnegative=122 |
Ntotal=52 Npositive=28 Nnegative=24 |
| Mwanga et al. , 2019 including P. falciparum and P. ovale mixed infection |
82b | 91b | Not Stated | Not stated |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).