Submitted:
03 October 2024
Posted:
04 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
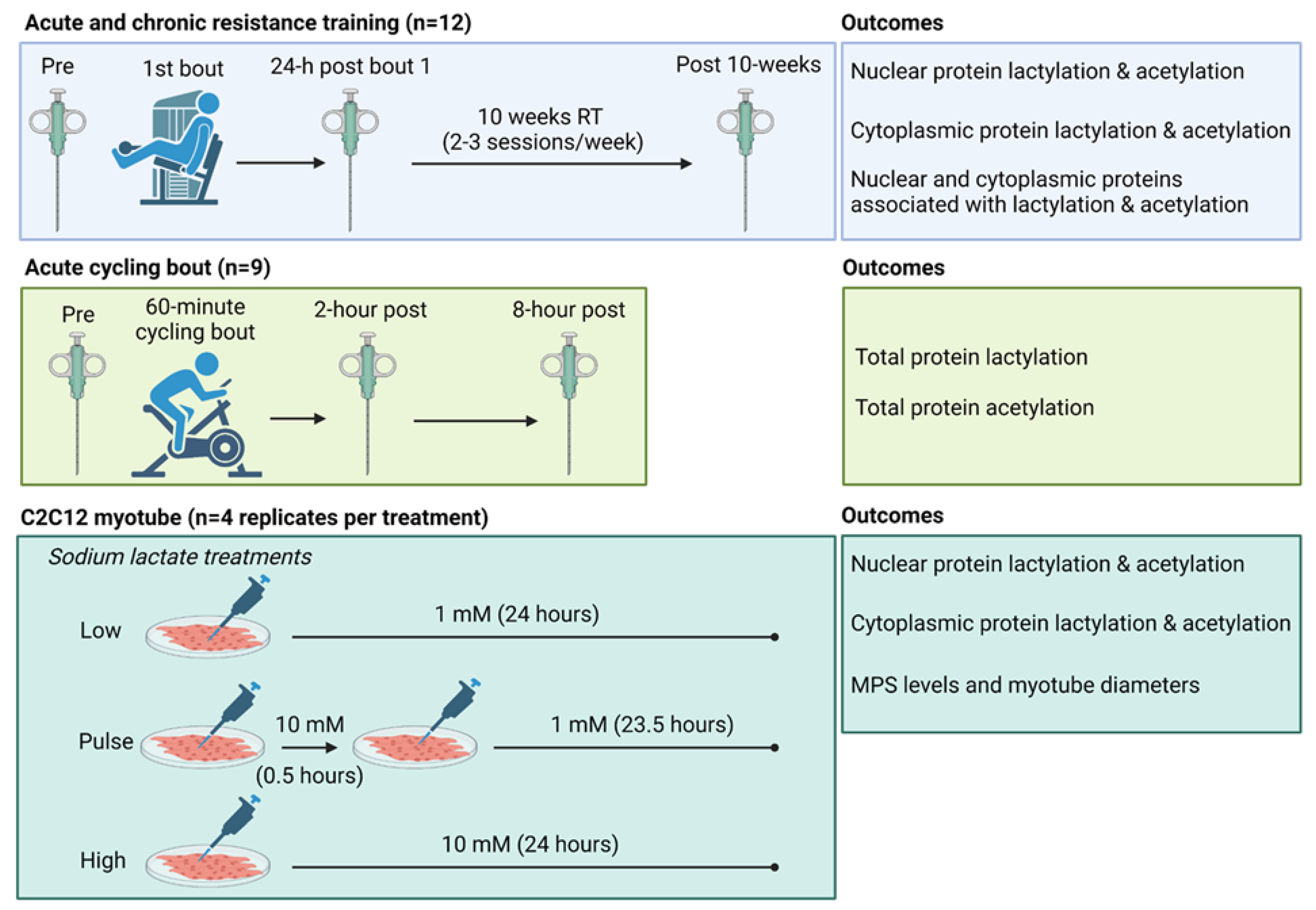
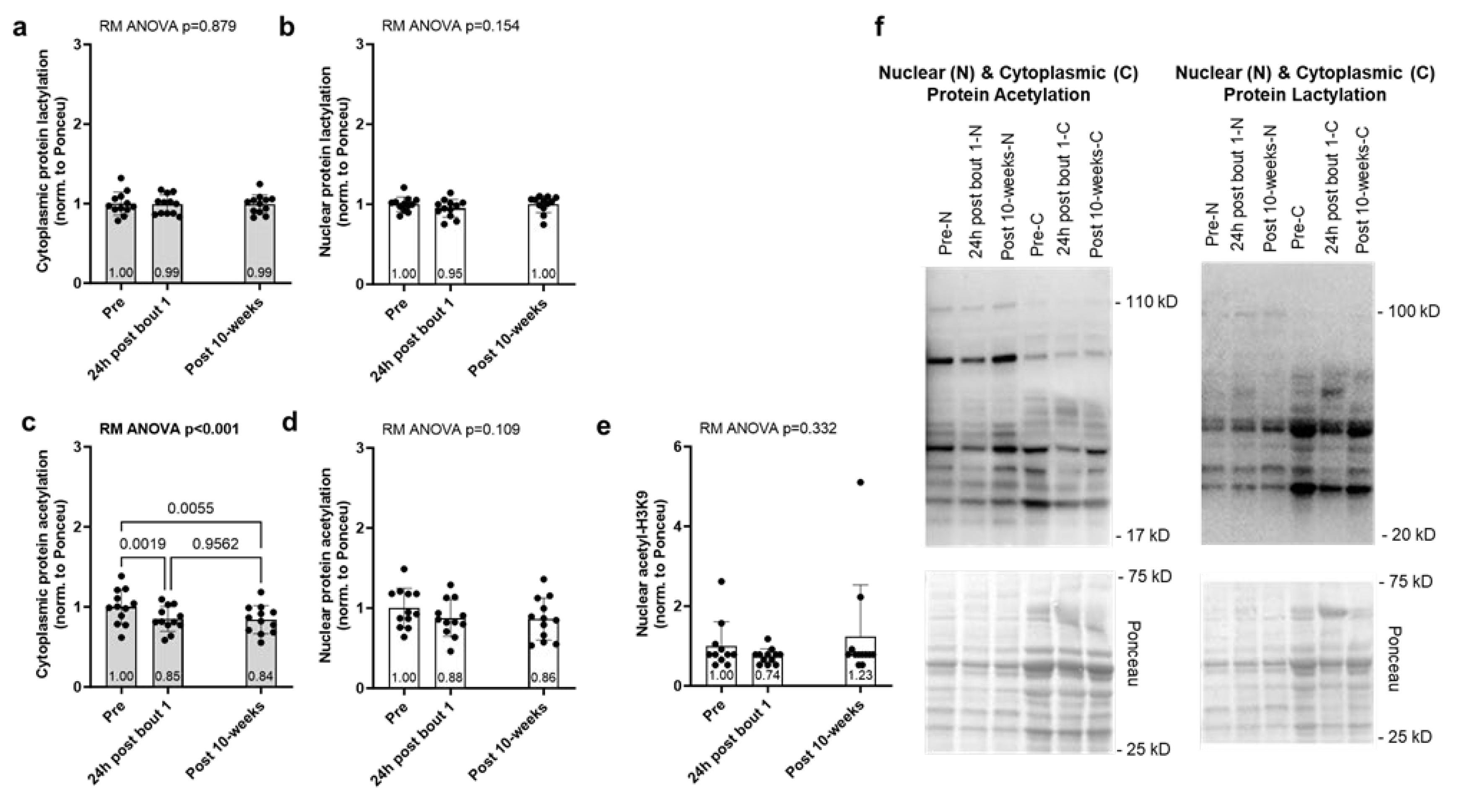
2.1. One Bout of Resistance Exercise, But Not Chronic Training, Significantly Decreases Select Enzymes Involved in Protein Acetylation and Lactylation
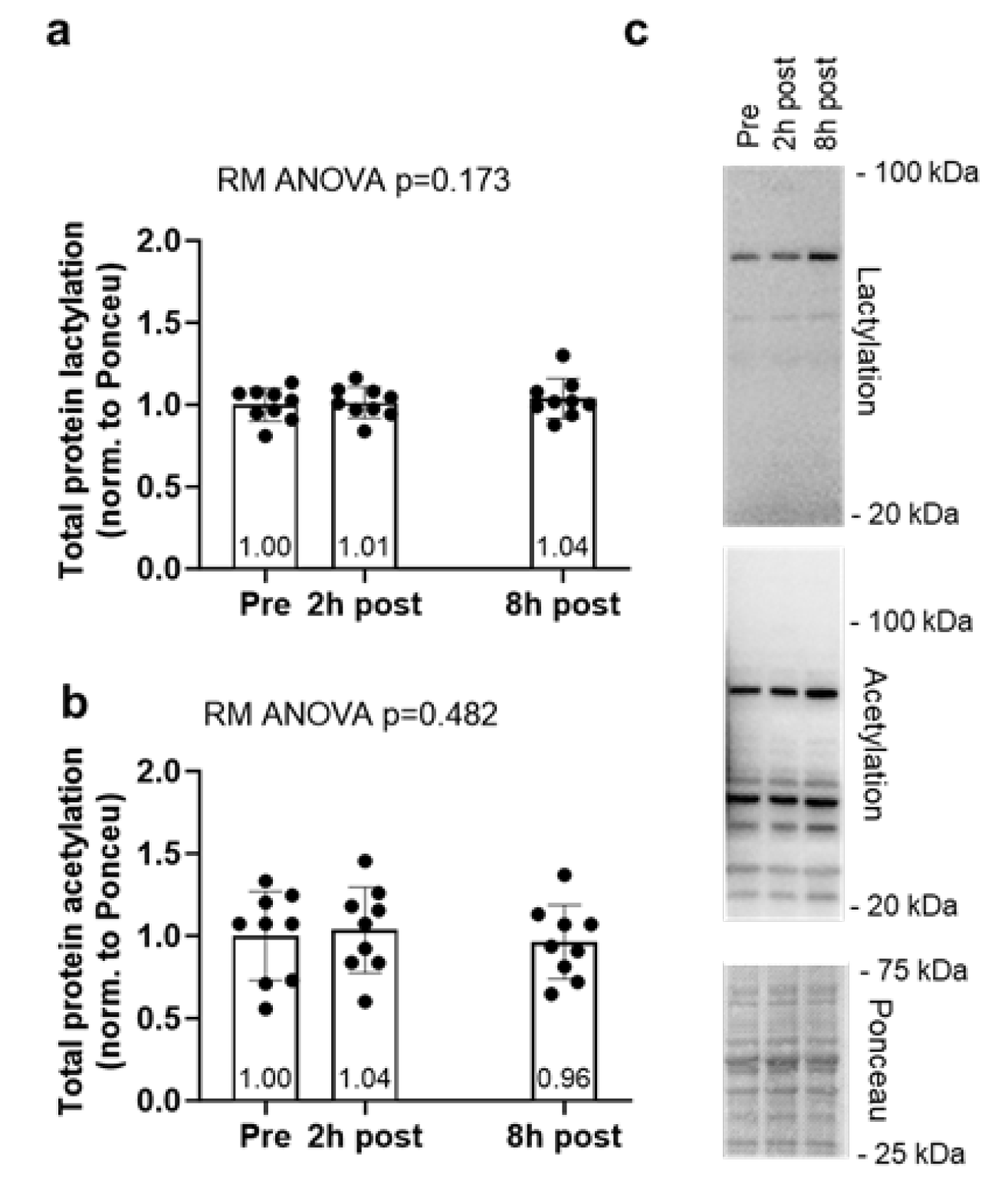
2.2. Global Protein Lactylation and Acetylation Markers Remain Unaltered Following 60 Minutes of Cycling
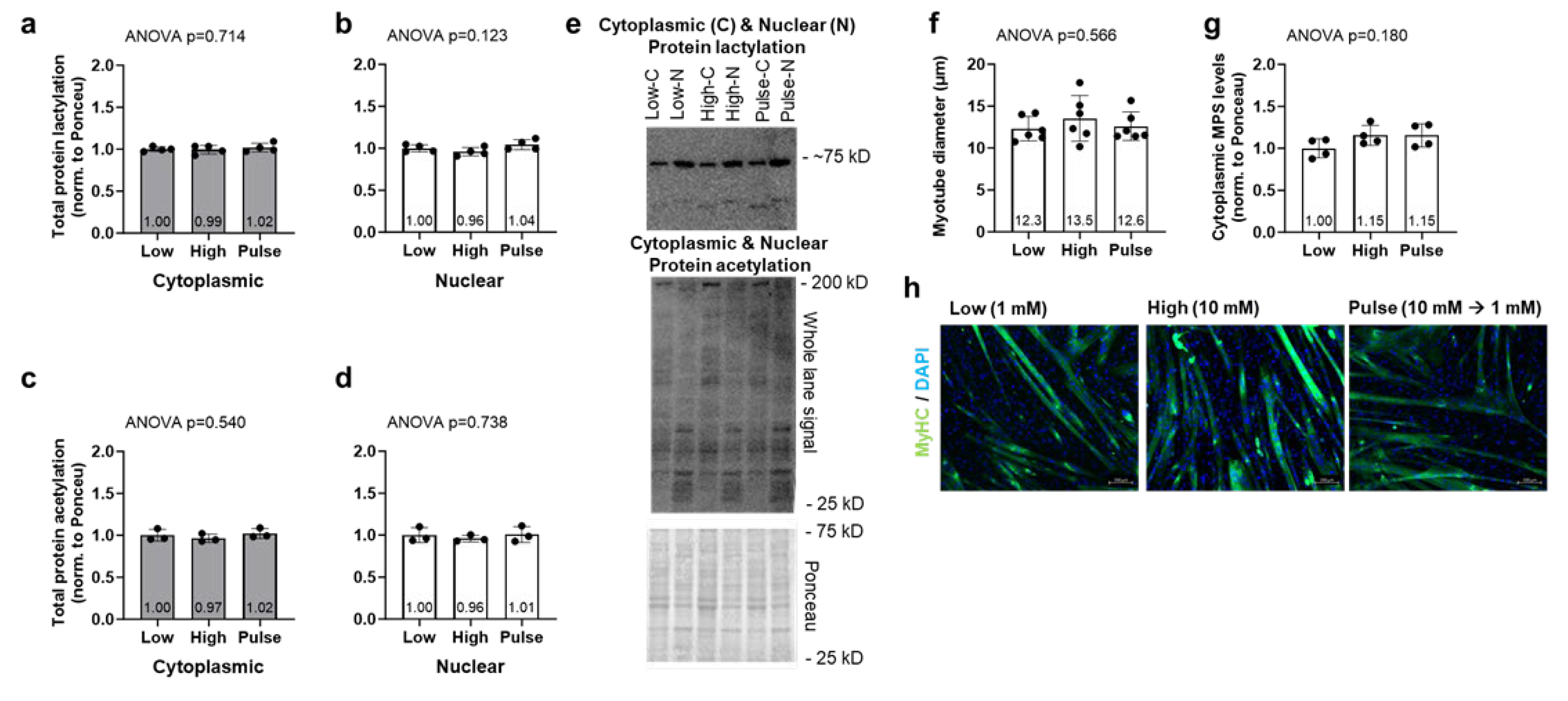
2.3. Lactate Administration Does Not Alter Myotube Diameter, Nuclear, or Cytoplasmic Puromycin-Labeled Proteins in C2C12 Cell Line
3. Discussion
3.1. Experimental Considerations
3.2. Conclusions
4. Materials and Methods
4.1. Ethical Approval for Human Work
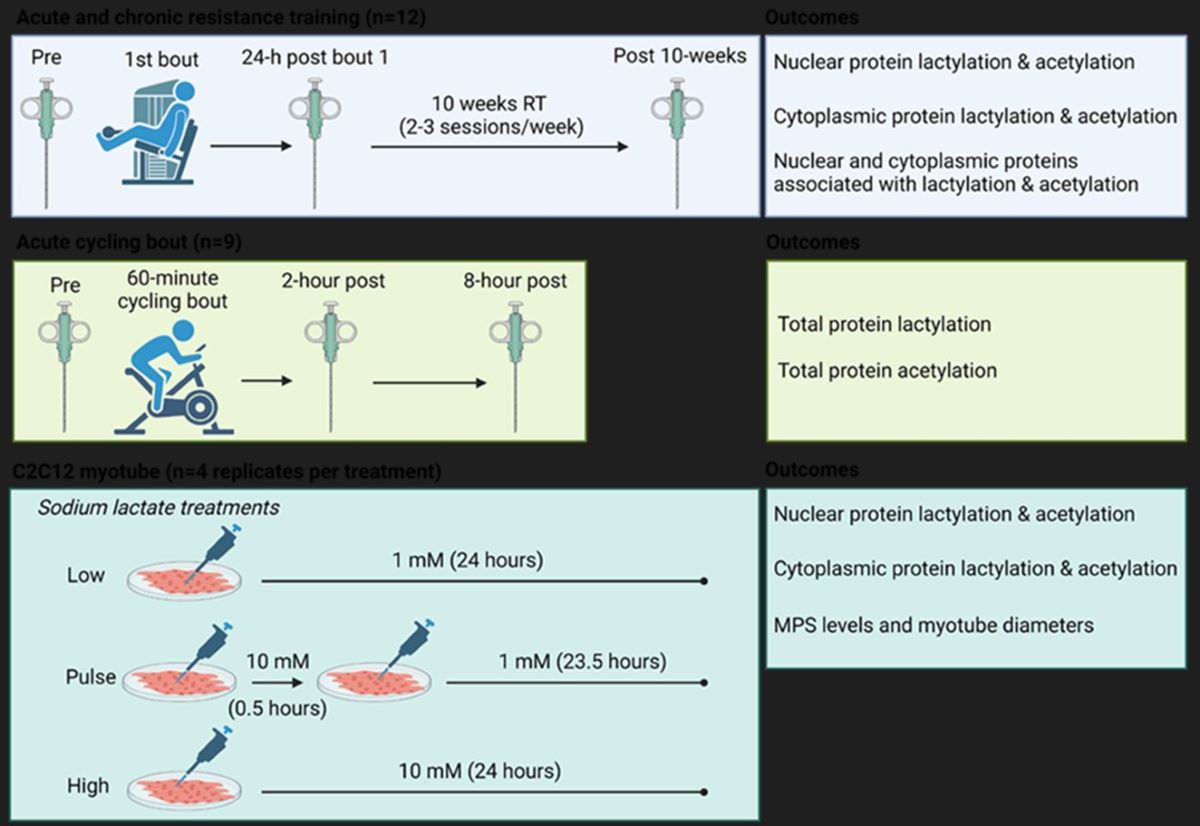
4.2. Study Designs
4.2.1. Acute and Chronic Resistance Training Study
4.2.2. Acute Endurance Training Study
4.2.3. Cell Culture Experiments
4.3. Western Blotting
4.4. Statistics
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Roberts, M.D.; McCarthy, J.J.; Hornberger, T.A.; Phillips, S.M.; Mackey, A.L.; Nader, G.A.; Boppart, M.D.; Kavazis, A.N.; Reidy, P.T.; Ogasawara, R.; et al. Mechanisms of mechanical overload-induced skeletal muscle hypertrophy: current understanding and future directions. Physiol Rev 2023, 103, 2679–2757. [Google Scholar] [CrossRef]
- Egan, B.; Sharples, A.P. Molecular responses to acute exercise and their relevance for adaptations in skeletal muscle to exercise training. Physiol Rev 2023, 103, 2057–2170. [Google Scholar] [CrossRef]
- Ohsawa, I.; Kawano, F. Chronic exercise training activates histone turnover in mouse skeletal muscle fibers. FASEB J 2021, 35, e21453. [Google Scholar] [CrossRef]
- McGee, S.L.; Hargreaves, M. Histone modifications and exercise adaptations. J Appl Physiol (1985) 2011, 110, 258–263. [Google Scholar] [CrossRef]
- McGee, S.L.; Fairlie, E.; Garnham, A.P.; Hargreaves, M. Exercise-induced histone modifications in human skeletal muscle. J Physiol 2009, 587, 5951–5958. [Google Scholar] [CrossRef]
- Zhang, D.; Tang, Z.; Huang, H.; Zhou, G.; Cui, C.; Weng, Y.; Liu, W.; Kim, S.; Lee, S.; Perez-Neut, M.; et al. Metabolic regulation of gene expression by histone lactylation. Nature 2019, 574, 575–580. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Z.; Wang, Q.; Li, X.; Guo, Y. Ubiquitous protein lactylation in health and diseases. Cell Mol Biol Lett 2024, 29, 23. [Google Scholar] [CrossRef]
- Ghosh-Choudhary, S.; Finkel, T. Lactylation regulates cardiac function. Cell Res 2023, 33, 653–654. [Google Scholar] [CrossRef]
- Nian, F.; Qian, Y.; Xu, F.; Yang, M.; Wang, H.; Zhang, Z. LDHA promotes osteoblast differentiation through histone lactylation. Biochem Biophys Res Commun 2022, 615, 31–35. [Google Scholar] [CrossRef]
- Yang, K.; Fan, M.; Wang, X.; Xu, J.; Wang, Y.; Tu, F.; Gill, P.S.; Ha, T.; Liu, L.; Williams, D.L.; et al. Lactate promotes macrophage HMGB1 lactylation, acetylation, and exosomal release in polymicrobial sepsis. Cell Death Differ 2022, 29, 133–146. [Google Scholar] [CrossRef]
- Dai, W.; Wu, G.; Liu, K.; Chen, Q.; Tao, J.; Liu, H.; Shen, M. Lactate promotes myogenesis via activating H3K9 lactylation-dependent up-regulation of Neu2 expression. J Cachexia Sarcopenia Muscle 2023, 14, 2851–2865. [Google Scholar] [CrossRef]
- Lim, C.; Shimizu, J.; Kawano, F.; Kim, H.J.; Kim, C.K. Adaptive responses of histone modifications to resistance exercise in human skeletal muscle. PLoS One 2020, 15, e0231321. [Google Scholar] [CrossRef]
- de Meireles, L.C.; Bertoldi, K.; Cechinel, L.R.; Schallenberger, B.L.; da Silva, V.K.; Schroder, N.; Siqueira, I.R. Treadmill exercise induces selective changes in hippocampal histone acetylation during the aging process in rats. Neurosci Lett 2016, 634, 19–24. [Google Scholar] [CrossRef]
- Tsuda, M.; Fukushima, A.; Matsumoto, J.; Takada, S.; Kakutani, N.; Nambu, H.; Yamanashi, K.; Furihata, T.; Yokota, T.; Okita, K.; et al. Protein acetylation in skeletal muscle mitochondria is involved in impaired fatty acid oxidation and exercise intolerance in heart failure. J Cachexia Sarcopenia Muscle 2018, 9, 844–859. [Google Scholar] [CrossRef]
- Huang, W.; Su, J.; Chen, X.; Li, Y.; Xing, Z.; Guo, L.; Li, S.; Zhang, J. High-Intensity Interval Training Induces Protein Lactylation in Different Tissues of Mice with Specificity and Time Dependence. Metabolites 2023, 13. [Google Scholar] [CrossRef]
- Mattingly, M.L.; Ruple, B.A.; Sexton, C.L.; Godwin, J.S.; McIntosh, M.C.; Smith, M.A.; Plotkin, D.L.; Michel, J.M.; Anglin, D.A.; Kontos, N.J.; et al. Resistance training in humans and mechanical overload in rodents do not elevate muscle protein lactylation. Front Physiol 2023, 14, 1281702. [Google Scholar] [CrossRef]
- Maschari, D.; Saxena, G.; Law, T.D.; Walsh, E.; Campbell, M.C.; Consitt, L.A. Lactate-induced lactylation in skeletal muscle is associated with insulin resistance in humans. Front Physiol 2022, 13, 951390. [Google Scholar] [CrossRef]
- Moreno-Yruela, C.; Zhang, D.; Wei, W.; Baek, M.; Liu, W.; Gao, J.; Dankova, D.; Nielsen, A.L.; Bolding, J.E.; Yang, L.; et al. Class I histone deacetylases (HDAC1-3) are histone lysine delactylases. Sci Adv 2022, 8, eabi6696. [Google Scholar] [CrossRef]
- Varner, E.L.; Trefely, S.; Bartee, D.; von Krusenstiern, E.; Izzo, L.; Bekeova, C.; O’Connor, R.S.; Seifert, E.L.; Wellen, K.E.; Meier, J.L.; et al. Quantification of lactoyl-CoA (lactyl-CoA) by liquid chromatography mass spectrometry in mammalian cells and tissues. Open Biol 2020, 10, 200187. [Google Scholar] [CrossRef]
- Xu, J.; Li, C.; Kang, X. The epigenetic regulatory effect of histone acetylation and deacetylation on skeletal muscle metabolism-a review. Front Physiol 2023, 14, 1267456. [Google Scholar] [CrossRef]
- Hostrup, M.; Lemminger, A.K.; Stocks, B.; Gonzalez-Franquesa, A.; Larsen, J.K.; Quesada, J.P.; Thomassen, M.; Weinert, B.T.; Bangsbo, J.; Deshmukh, A.S. High-intensity interval training remodels the proteome and acetylome of human skeletal muscle. Elife 2022, 11. [Google Scholar] [CrossRef]
- Hain, B.A.; Kimball, S.R.; Waning, D.L. Preventing loss of sirt1 lowers mitochondrial oxidative stress and preserves C2C12 myotube diameter in an in vitro model of cancer cachexia. Physiol Rep 2024, 12, e16103. [Google Scholar] [CrossRef]
- Lundby, A.; Lage, K.; Weinert, B.T.; Bekker-Jensen, D.B.; Secher, A.; Skovgaard, T.; Kelstrup, C.D.; Dmytriyev, A.; Choudhary, C.; Lundby, C.; et al. Proteomic analysis of lysine acetylation sites in rat tissues reveals organ specificity and subcellular patterns. Cell Rep 2012, 2, 419–431. [Google Scholar] [CrossRef]
- Baeza, J.; Smallegan, M.J.; Denu, J.M. Mechanisms and Dynamics of Protein Acetylation in Mitochondria. Trends Biochem Sci 2016, 41, 231–244. [Google Scholar] [CrossRef]
- Lawson, D.; Vann, C.; Schoenfeld, B.J.; Haun, C. Beyond Mechanical Tension: A Review of Resistance Exercise-Induced Lactate Responses & Muscle Hypertrophy. J Funct Morphol Kinesiol 2022, 7. [Google Scholar] [CrossRef]
- Shirai, T.; Kitaoka, Y.; Uemichi, K.; Tokinoya, K.; Takeda, K.; Takemasa, T. Effects of lactate administration on hypertrophy and mTOR signaling activation in mouse skeletal muscle. Physiol Rep 2022, 10, e15436. [Google Scholar] [CrossRef]
- Liegnell, R.; Apro, W.; Danielsson, S.; Ekblom, B.; van Hall, G.; Holmberg, H.C.; Moberg, M. Elevated plasma lactate levels via exogenous lactate infusion do not alter resistance exercise-induced signaling or protein synthesis in human skeletal muscle. Am J Physiol Endocrinol Metab 2020, 319, E792–E804. [Google Scholar] [CrossRef]
- Chaves, T.S.; Scarpelli, M.C.; Bergamasco, J.G.A.; Silva, D.G.D.; Medalha Junior, R.A.; Dias, N.F.; Bittencourt, D.; Carello Filho, P.C.; Angleri, V.; Nobrega, S.R.; et al. Effects of Resistance Training Overload Progression Protocols on Strength and Muscle Mass. Int J Sports Med 2024, 45, 504–510. [Google Scholar] [CrossRef]
- Roberson, P.A.; Romero, M.A.; Osburn, S.C.; Mumford, P.W.; Vann, C.G.; Fox, C.D.; McCullough, D.J.; Brown, M.D.; Roberts, M.D. Skeletal muscle LINE-1 ORF1 mRNA is higher in older humans but decreases with endurance exercise and is negatively associated with higher physical activity. J Appl Physiol (1985) 2019, 127, 895–904. [Google Scholar] [CrossRef]
- Mobley, C.B.; Fox, C.D.; Ferguson, B.S.; Amin, R.H.; Dalbo, V.J.; Baier, S.; Rathmacher, J.A.; Wilson, J.M.; Roberts, M.D. L-leucine, beta-hydroxy-beta-methylbutyric acid (HMB) and creatine monohydrate prevent myostatin-induced Akirin-1/Mighty mRNA down-regulation and myotube atrophy. Journal of the International Society of Sports Nutrition 2014, 11, 38. [Google Scholar] [CrossRef]
- Mobley, C.B.; Mumford, P.W.; McCarthy, J.J.; Miller, M.E.; Young, K.C.; Martin, J.S.; Beck, D.T.; Lockwood, C.M.; Roberts, M.D. Whey protein-derived exosomes increase protein synthesis and hypertrophy in C(2-)C(12) myotubes. J Dairy Sci 2017, 100, 48–64. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



