Submitted:
03 October 2024
Posted:
04 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Optical Properties of Polymer Solutions
2.3. Rheological Properties
2.4. Electrospinning Technique
2.5. Morphology and Diameters of Nanofibers
2.6. Fourier–Transform Infrared (FTIR) Spectroscopy
2.7. X–ray Diffraction (XRD) Analysis
2.8. Differential Scanning Calorimetry (DSC) Analysis
2.9. Thermogravimetric Analysis (TGA)
2.10. Tensile Property
2.11. Statistical Analysis
3. Results and Discussion
3.1. Effect of Ethanol– Acetic Acid Ratio on Optical Properties of PVA–CS Solution
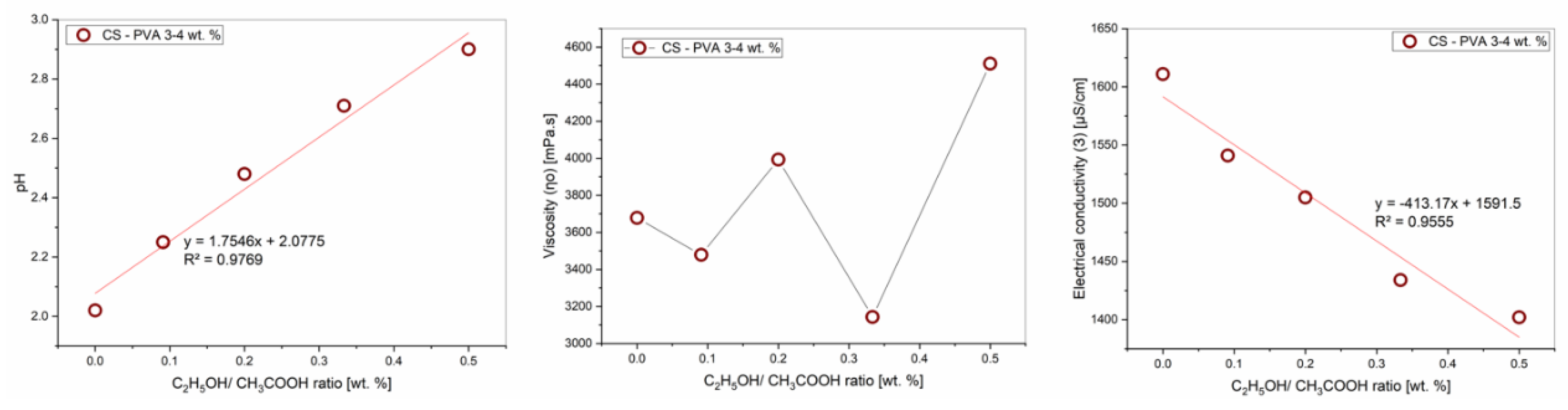
3.2. Effect of Ethanol– Acetic Acid Ratio on Rheological Properties of PVA–CS Solution
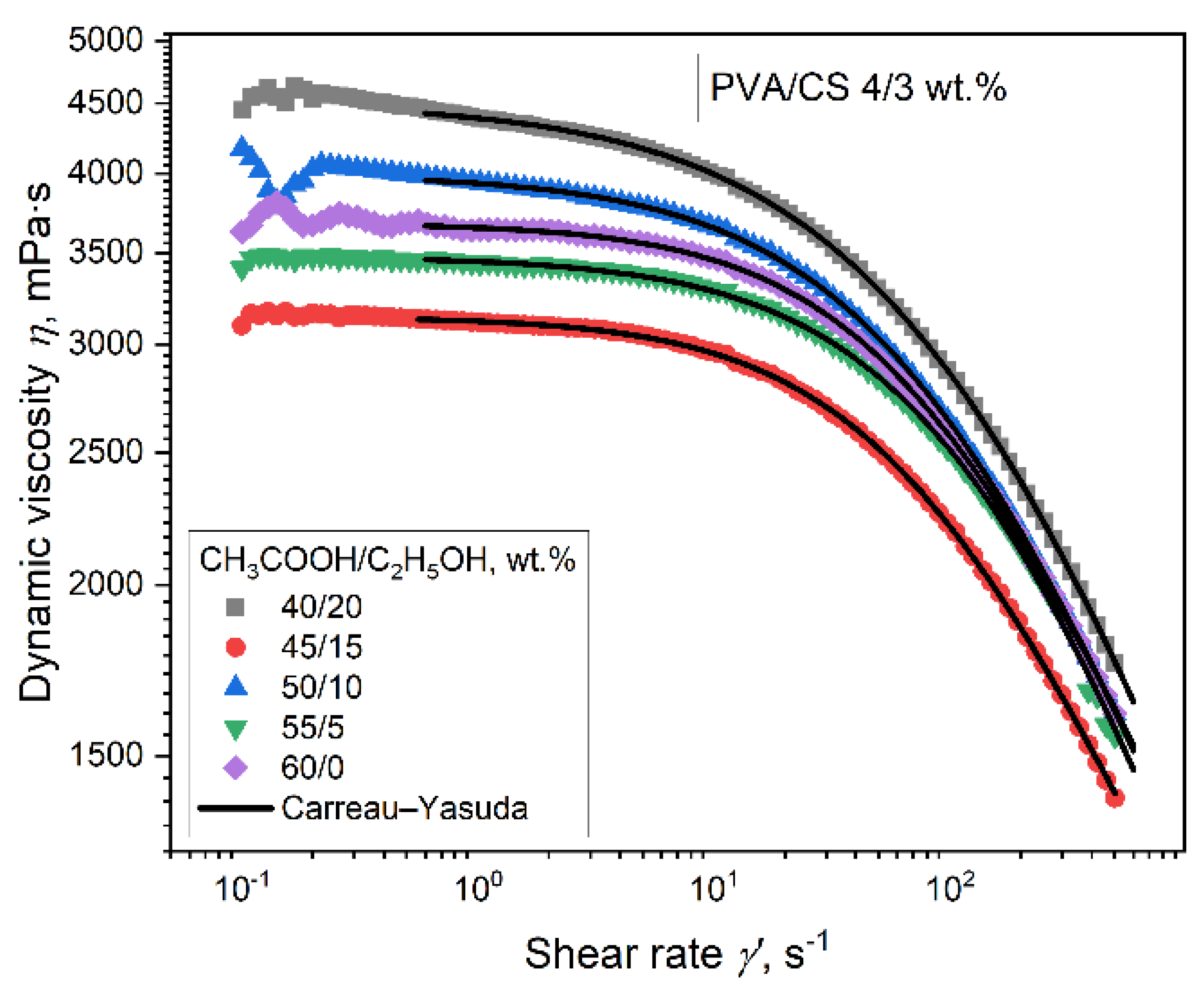
| CH3COOH/C2H5OH, (%) | η0, (mPa·s) | λ · 103 (s) | a | m | R2 |
|---|---|---|---|---|---|
| 60/0 | 3678 | 13,86 | 0,93 | 0,611 | 0,9999 |
| 55/5 | 3479 | 7,11 | 0,84 | 0,507 | 0,9995 |
| 50/10 | 3993 | 11,54 | 0,80 | 0,553 | 0,9999 |
| 45/15 | 3143 | 13,70 | 0,92 | 0,617 | 0,9999 |
| 40/20 | 4511 | 7,56 | 0,68 | 0,485 | 0,9999 |
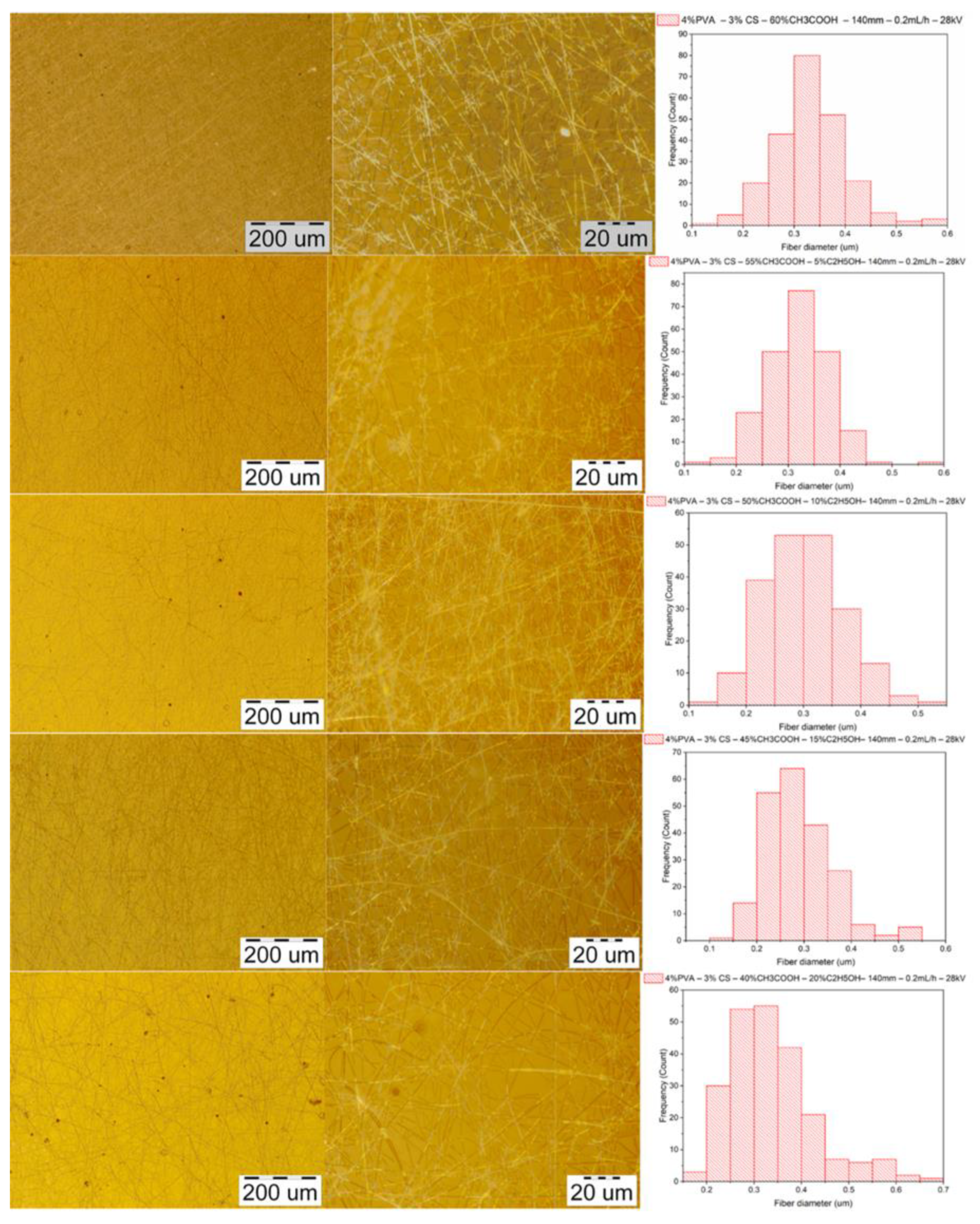
3.3. Effect of Ethanol– Acetic Acid Ratio on Fabrication of PVA–CS Nanofibers
3.3.1. The Ability to Fabricate PVA–CS Nanofibers
3.3.2. Intermolecular interactions for the PVA–CS nanofibers fabrication
| CH3COOH wt.% | C2H5OH wt.% | H2O wt.% |
100fd | 100fp | 100fh | fh-fp | fh-fd | fp-fd |
|---|---|---|---|---|---|---|---|---|
| 60 | 0 | 33 | 32,19 | 24,13 | 43,68 | 19,55 | 11,48 | -8,06 |
| 55 | 5 | 33 | 31,98 | 23,91 | 44,11 | 20,19 | 12,13 | -8,06 |
| 50 | 10 | 33 | 31,76 | 23,70 | 44,54 | 20,84 | 12,77 | -8,06 |
| 45 | 15 | 33 | 31,55 | 23,48 | 44,97 | 21,48 | 13,42 | -8,06 |
| 40 | 20 | 33 | 31,33 | 23,27 | 45,40 | 22,13 | 14,06 | -8,06 |
| 35 | 25 | 33 | 31,12 | 23,05 | 45,83 | 22,77 | 14,71 | -8,06 |
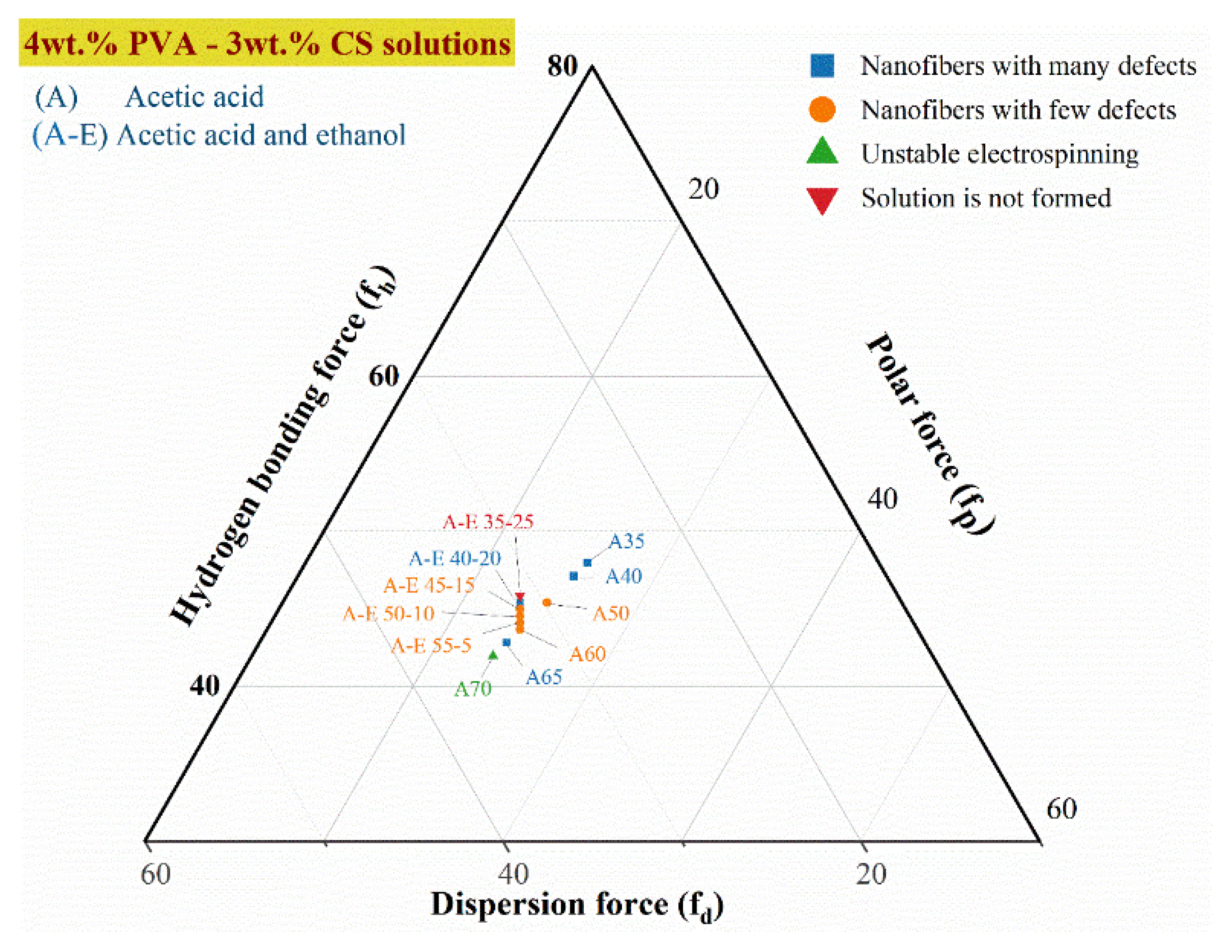
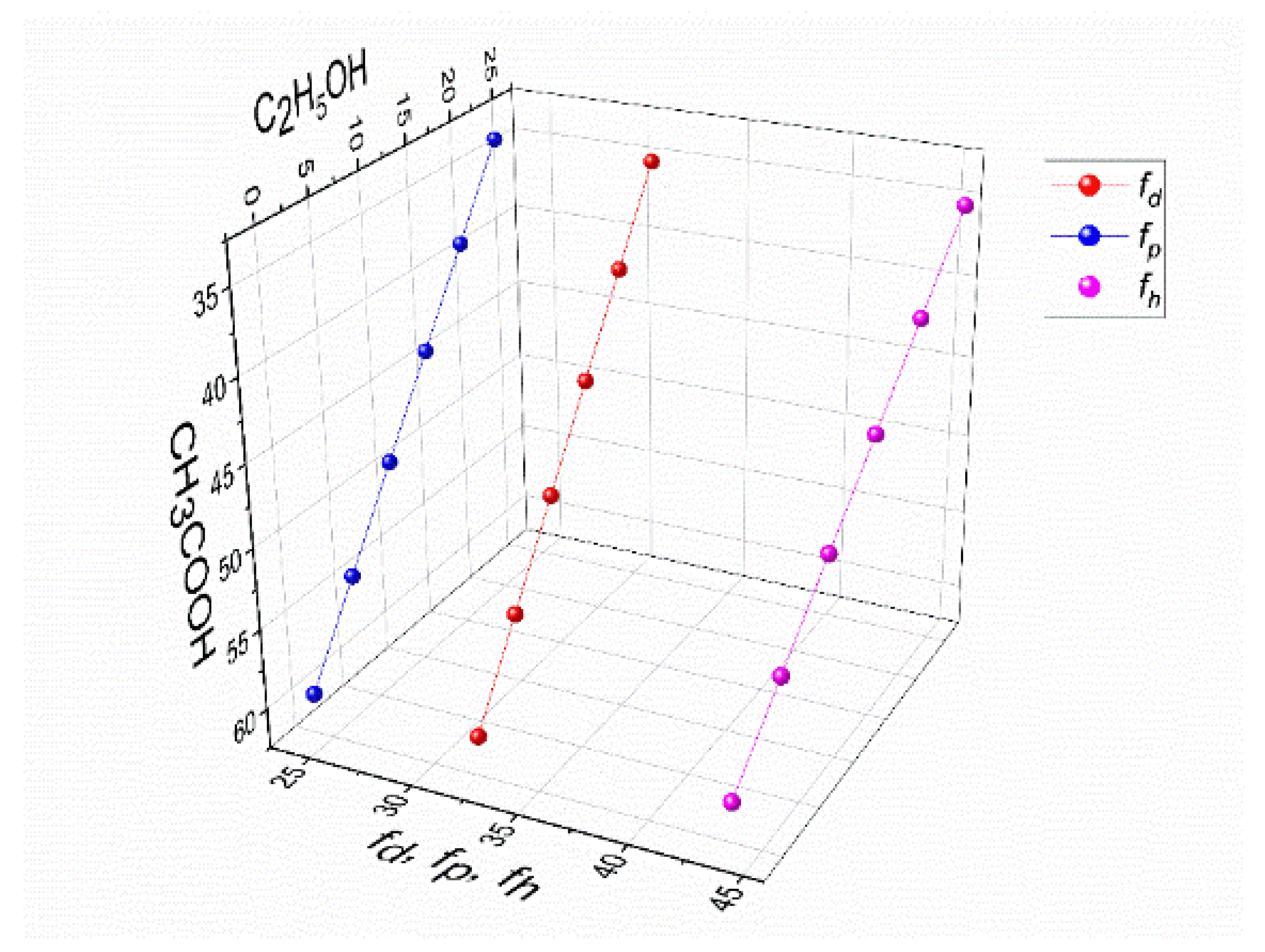
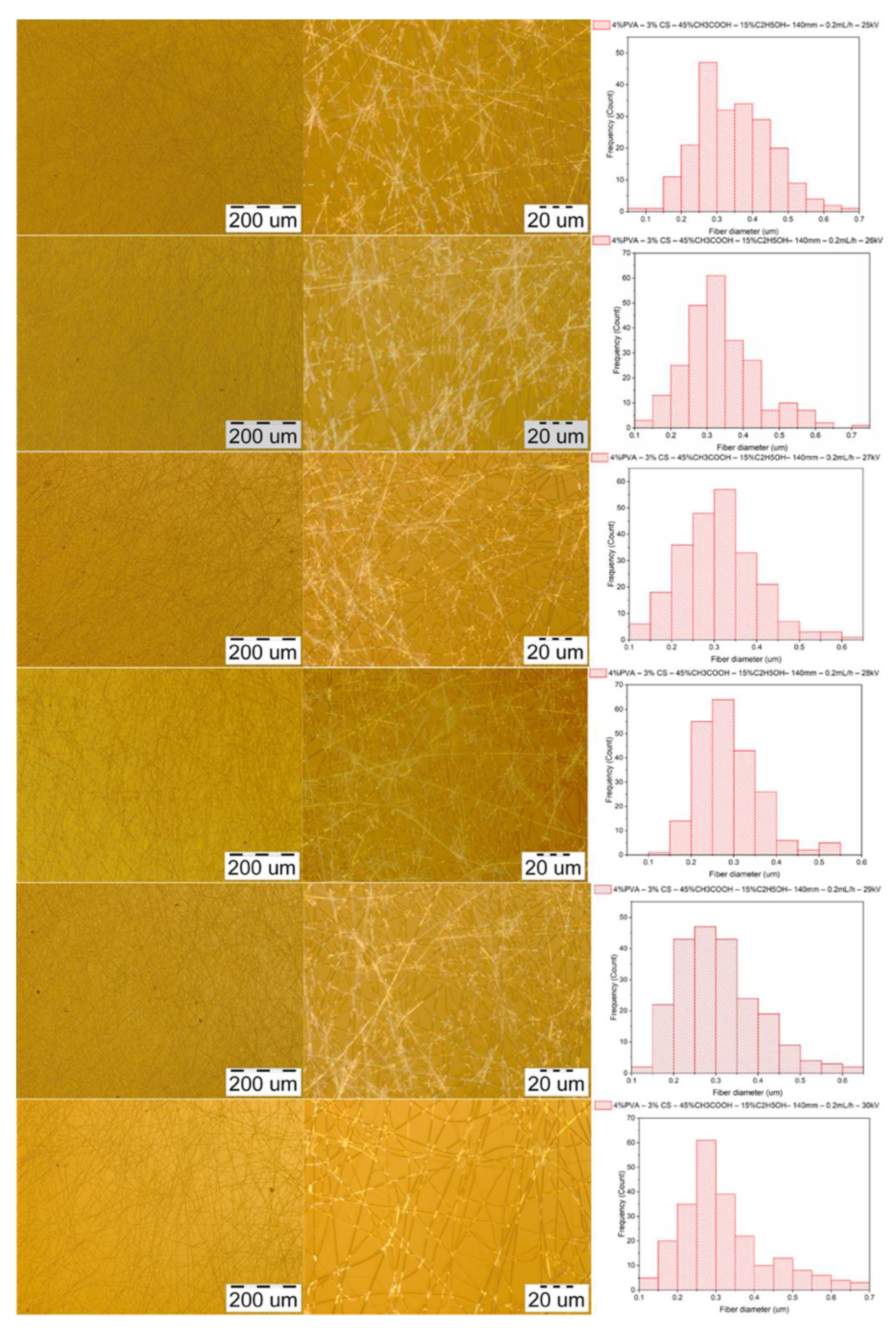
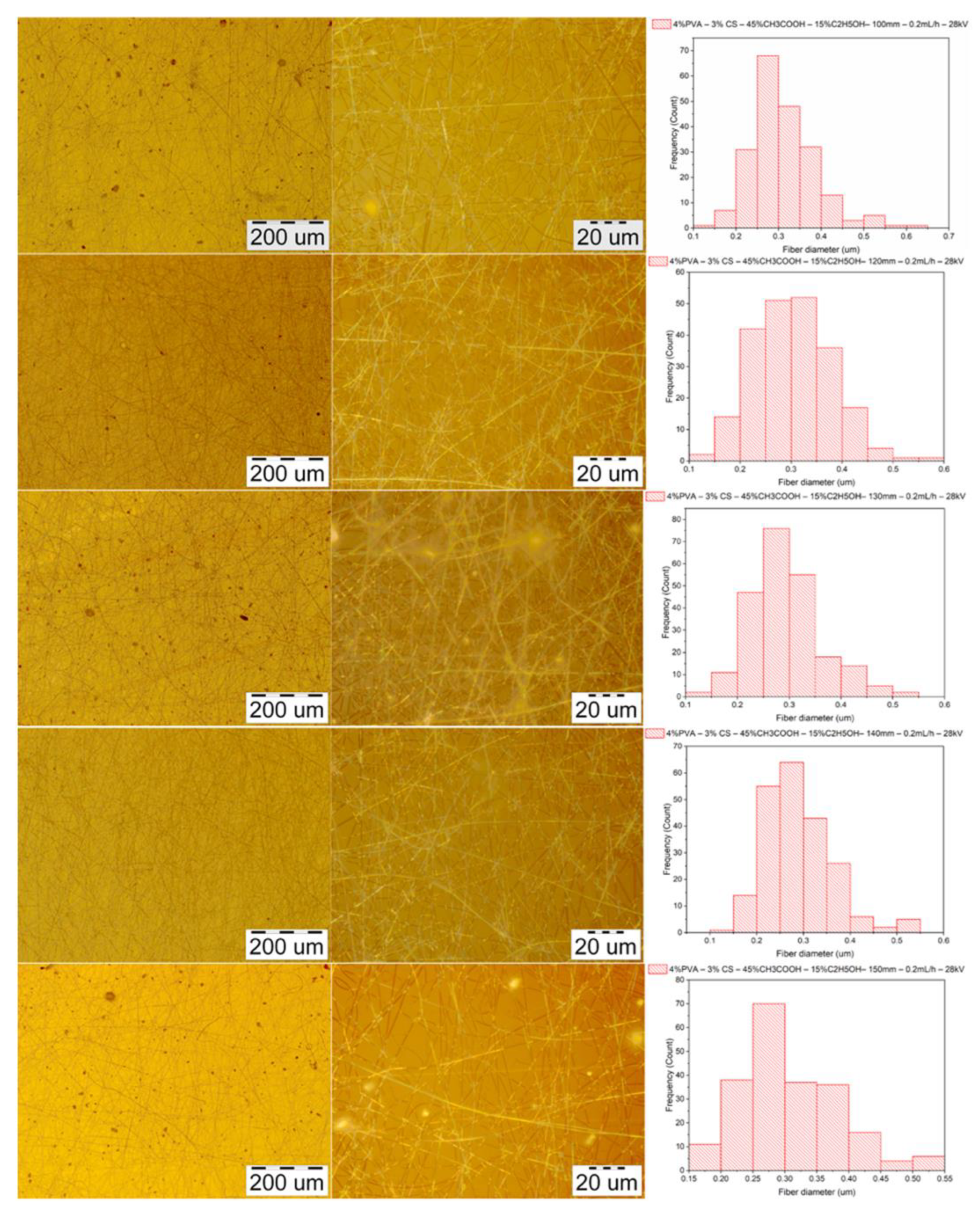
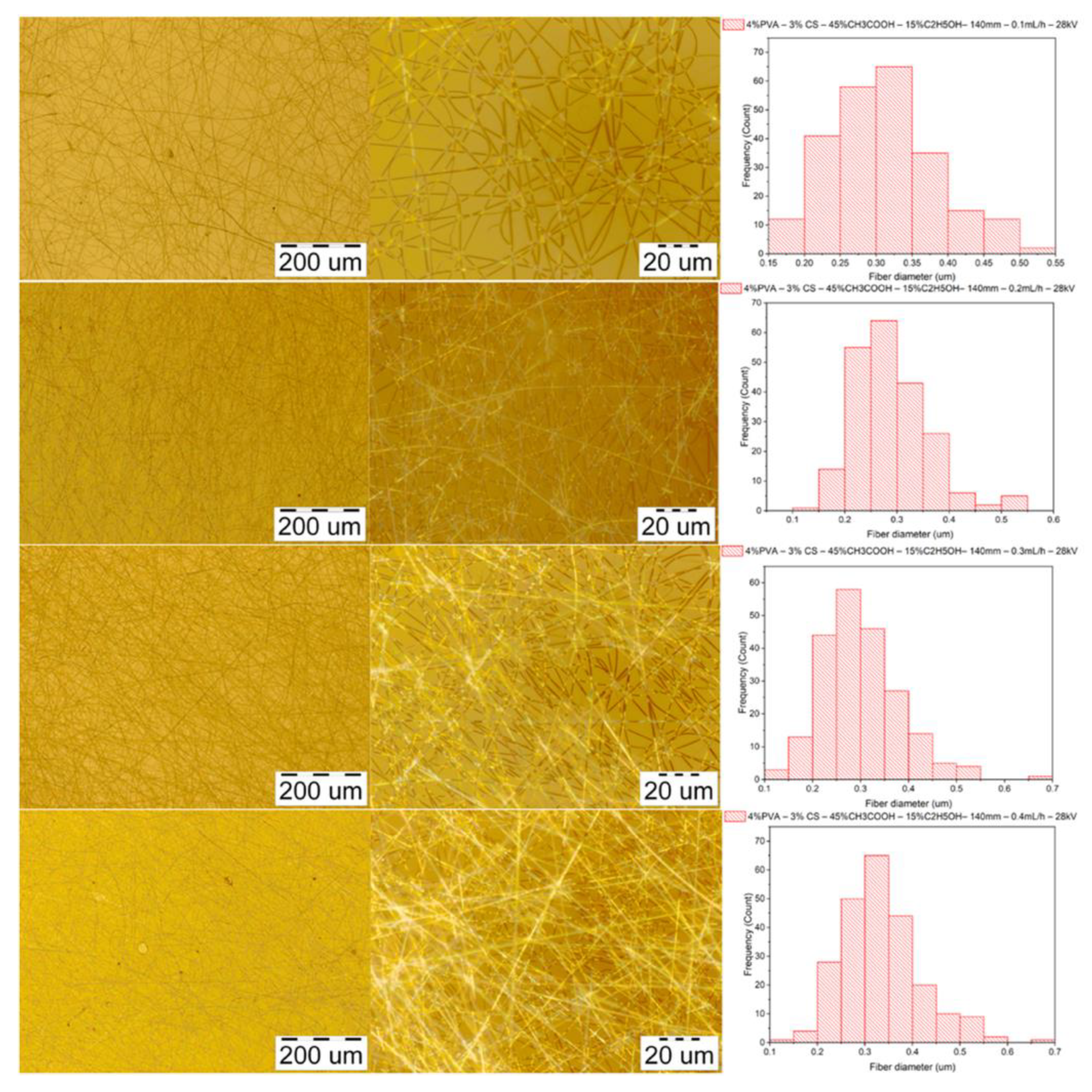
3.3.3. Morphology of PVA–CS Nanofibers
3.4. Effect of Ethanol– Acetic Acid Ratio on Optimal Electrospinning Parameters
3.4.1. Parameter of the Distance between the Needle Tip and the Collector
3.4.2. Parameters of Feed Rate
3.4.3. Parameters of the Voltage
| Diameter (nm) | Voltage (kV) | |||||
|---|---|---|---|---|---|---|
| 25 | 26 | 27 | 28 | 29 | 30 | |
| Mean | 348 | 334 | 309 | 285 | 307 | 318 |
| Standard deviation | 102 | 99 | 91 | 65 | 96 | 114 |
| Min | 97 | 139 | 110 | 121 | 131 | 120 |
| Max | 652 | 734 | 600 | 592 | 644 | 689 |
3.5. Fourier–Transform Infrared (FTIR) Spectroscopy
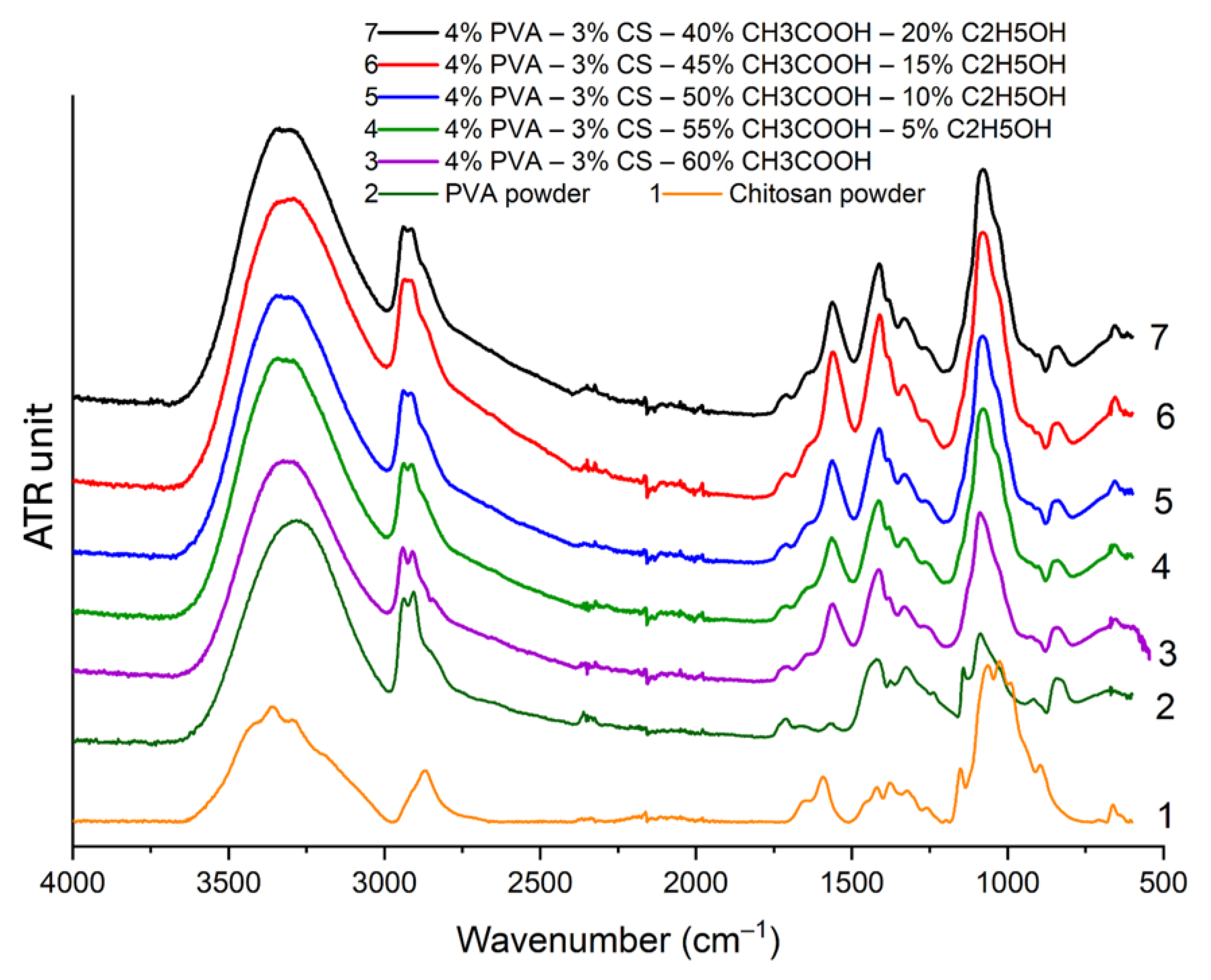
3.6. X–ray diffraction (XRD) Analysis
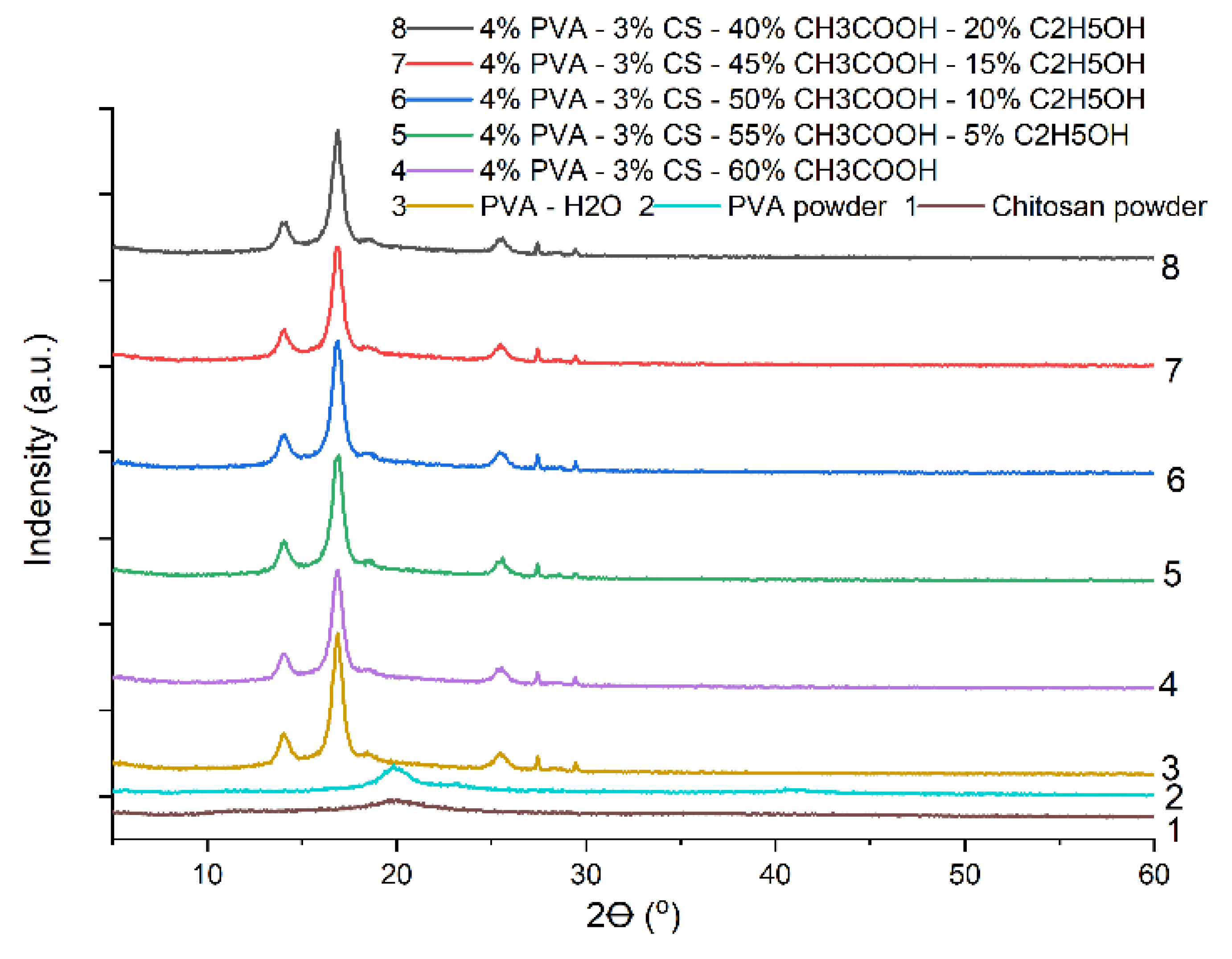
| Lattice parameters | Axial Lengths [Å] | Angles [°] | Cell volume [Å3] | Crystal-linity (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | α | β | γ | |||||
| Powder | CS | 15.7371 | 8.3352 | 3.0609 | 90 | 90 | 90 | 401.5017 | 48.29 | |
| PVA | 15.2596 | 5.2416 | 9.7092 | 90 | 97.188 | 90 | 770.4844 | 57.69 | ||
| Ethanol– acetic acid | 0–60 | 12.6928 | 3.5873 | 10.8013 | 90 | 95.237 | 90 | 489.7613 | 60.64 | |
| 5–55 | 7.6994 | 16.7927 | 6.8328 | 90 | 96.878 | 90 | 877.0803 | 54.64 | ||
| 10–50 | 15.9762 | 5.5865 | 9.3954 | 90 | 93.823 | 90 | 836.6833 | 62.36 | ||
| 15–45 | 12.7499 | 7.2561 | 8.637 | 90 | 92.457 | 90 | 798.3136 | 57.45 | ||
| 20–40 | 16.0564 | 4.1401 | 11.3214 | 90 | 93.814 | 90 | 750.9244 | 52.22 | ||
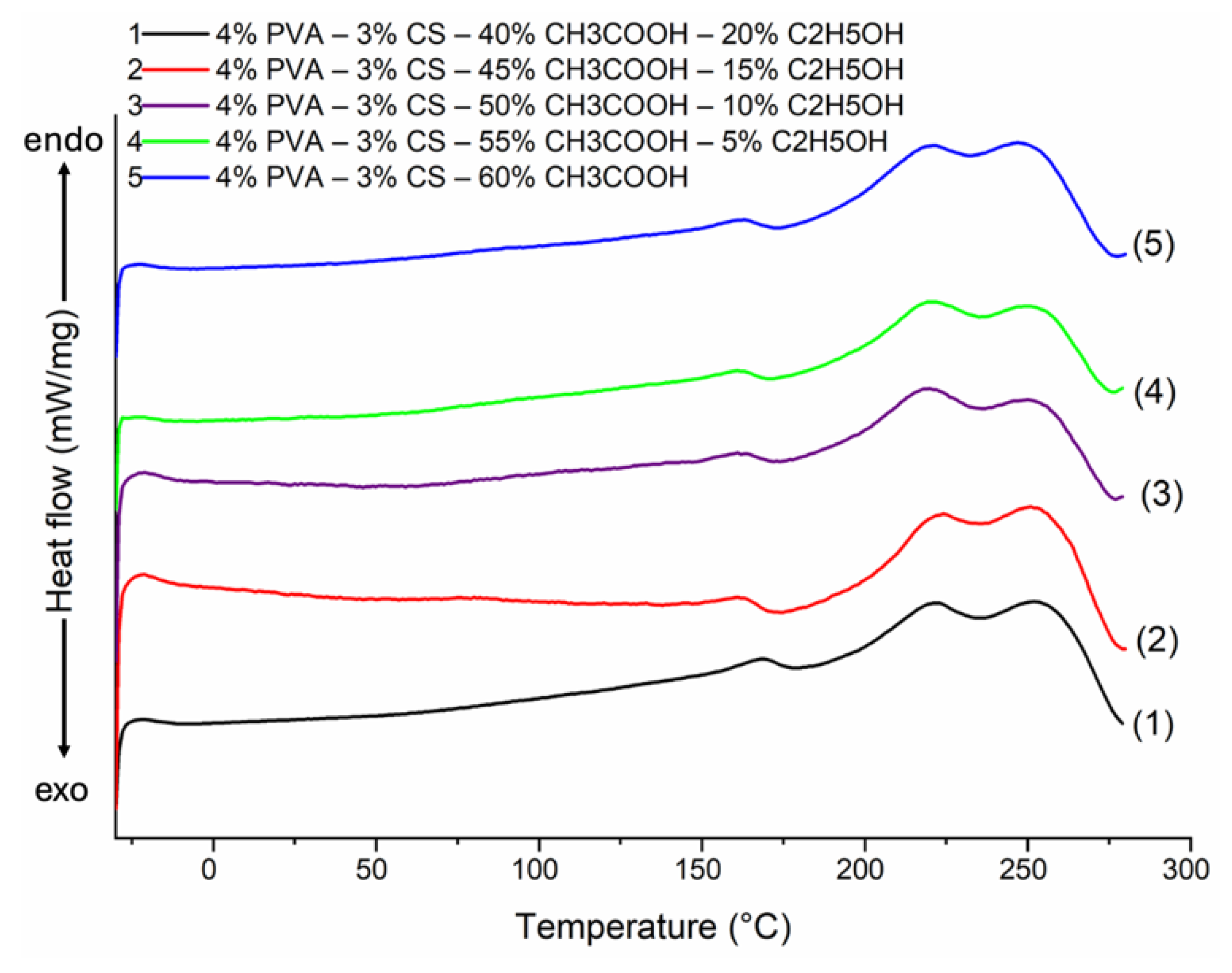
3.7. Differential Scanning Calorimetry (DSC) Analysis
| Ethanol– acetic acid ratio | ΔHPVA (J/g) | χPVA (%) | ΔHCS (J/g) | Tg (°C) | Tm(°C) |
|---|---|---|---|---|---|
| 0–60 | 57.22 | 38.15 | 5.86 | 75 | 222, 247 |
| 5–55 | 42.23 | 28.15 | 11.19 | 75 | 220, 249 |
| 10–50 | 44.09 | 29.39 | 9.79 | 84 | 220, 250 |
| 15–45 | 38.72 | 25.81 | 13.82 | 75 | 222, 251 |
| 20–40 | 53.73 | 35.82 | 2.56 | 87 | 223, 252 |
3.8. Thermogravimetric Analysis (TGA)
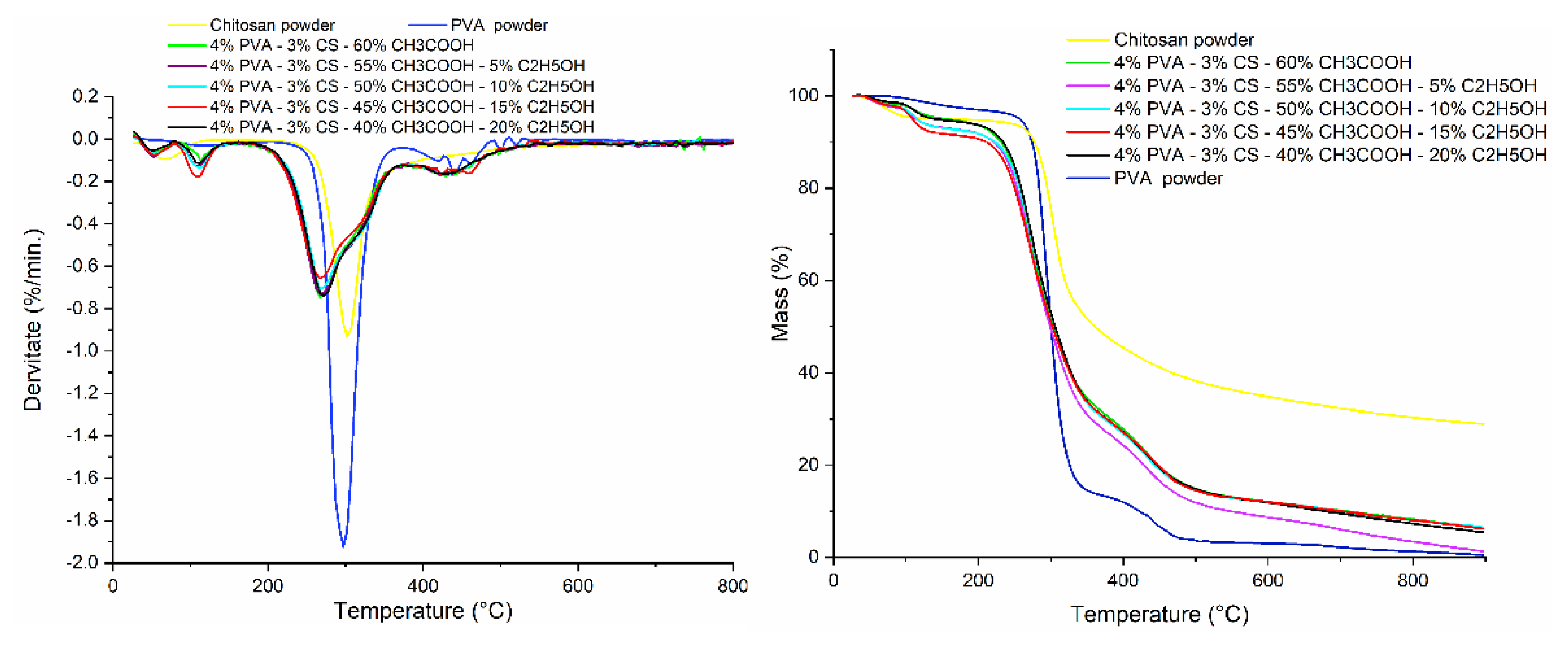
| Degradation stages | Powder | PVA–CS nanofibers with different CH3COOH/C2H5OH ratios | ||||||
|---|---|---|---|---|---|---|---|---|
| PVA | CS | 60 – 0 | 55 – 5 | 50 – 10 | 45 – 15 | 40 – 20 | ||
| First stage | Range (oС) | 25–202 | 25–177 | 25–172 | 25–172 | 25–172 | 25–172 | 25–172 |
| Peaks (oC) | 112 | 67 | 52; 114 | 52; 112 | 52; 112 | 52; 107 | 52; 112 | |
| Weight loss (%) | 3.04 | 5.21 | 5.42 | 7.34 | 5.63 | 8.58 | 5.63 | |
| Second stage | Range (oС) | 202–367 | 177–462 | 172–377 | 172–377 | 172–377 | 172–377 | 172–377 |
| Peaks (oC) | 297 | 302 | 267 | 272 | 272 | 267 | 272 | |
| Weight loss (%) | 83.37 | 77.04 | 63.79 | 64.19 | 62.94 | 61.40 | 64.31 | |
| Third stage | Range (oС) | 367–527 | 377–547 | 377–547 | 377–547 | 377–547 | 377–547 | |
| Peaks (oC) | 422; 442; 462 | 429; 459 | 422; 437 | 427 | 422; 437; 457 | 427 | ||
| Weight loss (%) | 10.32 | 17.64 | 18.43 | 16.72 | 16.97 | 16.97 | ||
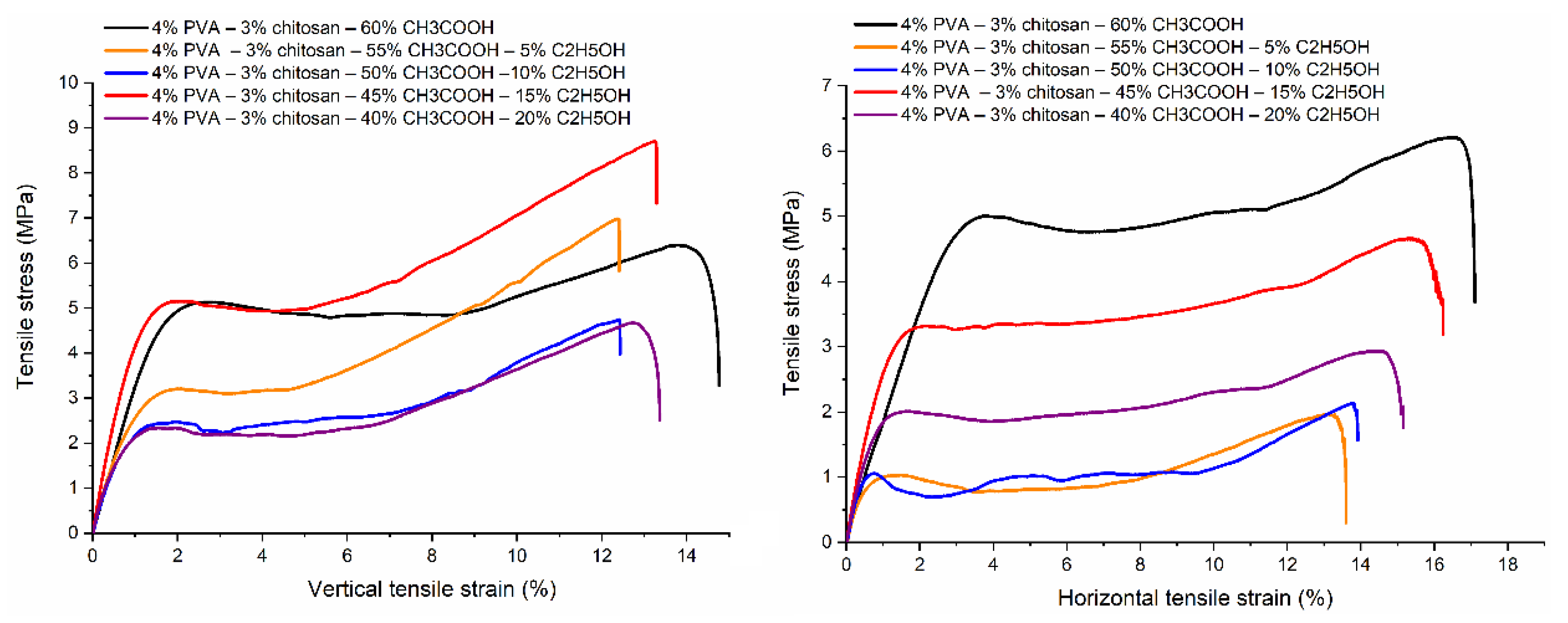
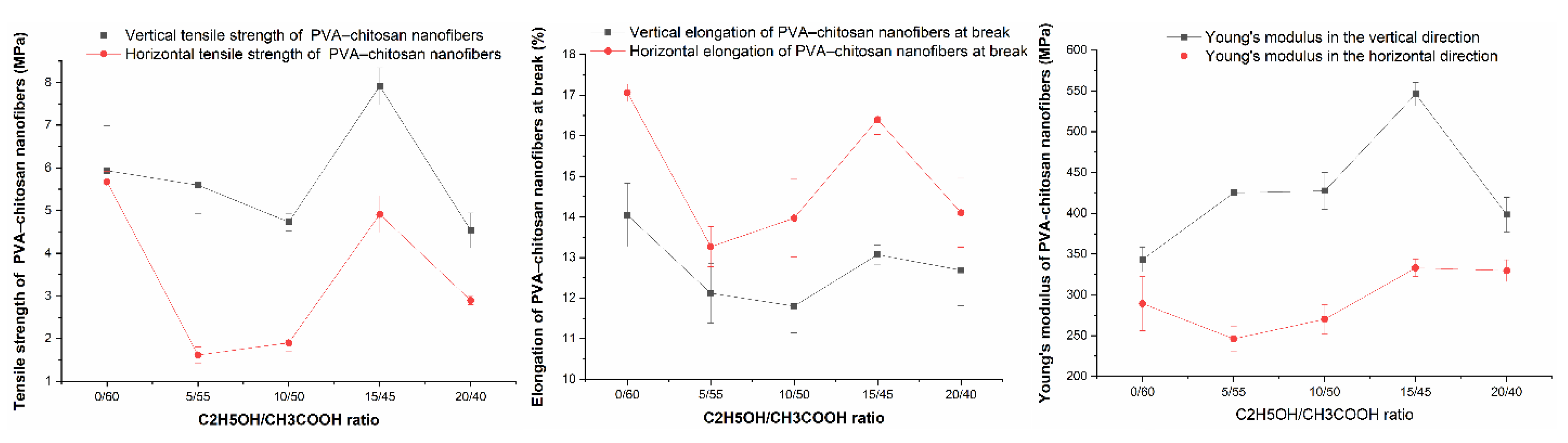
3.9. Tensile Property
4. Conclusions
- improved the morphology, diameter, and manufacturing productivity of PVA–CS nanofibers by 2–3 times due to the reduction of defects, reducing the diameter of nanofibers from 326 ± 62 nm (0.1 mL/h without ethanol) to 285 ± 65 nm (0.2 mL/h) or 300 ± 79 nm (0.3 mL/h);
- did not change the chemical nature of the resulting system of nanofibers, thereby maintaining the thermal properties of the PVA–CS nanofiber system;
- changed the structure of the crystal lattice of the nanofibers, which leads to a modification in the mechanical properties of the nanofibers. The PVA–CS nanofibers appear to become stronger by the increasing of the vertical tensile strength by 33%, the increasing of the vertical and horizontal Young's moduli by 59% and 15%, and the reducing of the elongation at break by 1%.
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Teodorescu, M.; Bercea, M.; Morariu, S. Biomaterials of PVA and PVP in Medical and Pharmaceutical Applications: Perspectives and Challenges. Biotechnol Adv 2019, 37, 109–131. [CrossRef]
- DeMerlis, C.C.; Schoneker, D.R. Review of the Oral Toxicity of Polyvinyl Alcohol (PVA). Food Chem Toxicol 2003, 41, 319–326. [CrossRef]
- Iqbal, D.N.; Tariq, M.; Khan, S.M.; Gull, N.; Sagar Iqbal, S.; Aziz, A.; Nazir, A.; Iqbal, M. Synthesis and Characterization of Chitosan and Guar Gum Based Ternary Blends with Polyvinyl Alcohol. Int J Biol Macromol 2020, 143, 546–554. [CrossRef]
- Nataraj, D.; Reddy, R.; Reddy, N. Crosslinking Electrospun Poly (Vinyl) Alcohol Fibers with Citric Acid to Impart Aqueous Stability for Medical Applications. Eur Polym J 2020, 124, 109484. [CrossRef]
- Pervez, M.N.; Stylios, G.K.; Liang, Y.; Ouyang, F.; Cai, Y. Low-Temperature Synthesis of Novel Polyvinylalcohol (PVA) Nanofibrous Membranes for Catalytic Dye Degradation. J Clean Prod 2020, 262, 121301. [CrossRef]
- Muppalaneni, S.; Omidian, H. Polyvinyl Alcohol in Medicine and Pharmacy: A Perspective. J Dev Drugs 2013, 02, 1–5. [CrossRef]
- Lizardi-Mendoza, J.; Argüelles Monal, W.M.; Goycoolea Valencia, F.M. Chapter 1 - Chemical Characteristics and Functional Properties of Chitosan. In; Bautista-Baños, S., Romanazzi, G., Jiménez-Aparicio, A.B.T.-C. in the P. of A.C., Eds.; Academic Press: San Diego, 2016; pp. 3–31 ISBN 978-0-12-802735-6.
- Aam, B.B.; Heggset, E.B.; Norberg, A.L.; Sørlie, M.; Vårum, K.M.; Eijsink, V.G.H. Production of Chitooligosaccharides and Their Potential Applications in Medicine. Mar Drugs 2010, 8, 1482–1517. [CrossRef]
- Gajra, B.; Pandya, S.S.; Vidyasagar, G.; Rabari, H.; Dedania, R.R.; Rao, S. Poly Vinyl Alcohol Hydrogel and Its Pharmaceutical and Biomedical Applications: A Review. International Journal of Pharmaceutical Research 2012, 4, 20–26.
- Kita, M.; Ogura, Y.; Honda, Y.; Hyon, S.-H.; Cha, W.-I.; Ikada, Y. Evaluation of Polyvinyl Alcohol Hydrogel as a Soft Contact Lens Material. Graefe’s Archive for Clinical and Experimental Ophthalmology 1990, 228, 533–537. [CrossRef]
- Jiang, Y.; Schädlich, A.; Amado, E.; Weis, C.; Odermatt, E.; Mäder, K.; Kressler, J. In-Vivo Studies on Intraperitoneally Administrated Poly(Vinyl Alcohol). J Biomed Mater Res B Appl Biomater 2010, 93B, 275–284. [CrossRef]
- Zairy, H. M. Ibrahim, E.M.R.E.-Z. Chitosan as a Biomaterial — Structure, Properties, and Electrospun Nanofibers, Concepts, Compounds and the Alternatives of Antibacterials. Varaprasad Bobbarala, IntechOpen 2015, 81–101. [CrossRef]
- Martău, G.A.; Mihai, M.; Vodnar, D.C. The Use of Chitosan, Alginate, and Pectin in the Biomedical and Food Sector-Biocompatibility, Bioadhesiveness, and Biodegradability. Polymers (Basel) 2019, 11, 1837. [CrossRef]
- Koltai, T. Cancer: Fundamentals behind PH Targeting and the Double-Edged Approach. Onco Targets Ther 2016, 9, 6343–6360. [CrossRef]
- You, J.S.; Jones, P.A. Cancer Genetics and Epigenetics: Two Sides of the Same Coin? Cancer Cell 2012, 22, 9–20. [CrossRef]
- Lu, Y.; Aimetti, A.A.; Langer, R.; Gu, Z. Bioresponsive Materials. Nat Rev Mater 2016, 2, 16075. [CrossRef]
- Le, W.; Chen, B.; Cui, Z.; Liu, Z.; Shi, D. Detection of Cancer Cells Based on Glycolytic-Regulated Surface Electrical Charges. Biophys Rep 2019, 5, 10–18.
- Cascone, M.G.; Maltinti, S.; Barbani, N.; Laus, M. Effect of Chitosan and Dextran on the Properties of Poly(Vinyl Alcohol) Hydrogels. J Mater Sci Mater Med 1999, 10, 431–435. [CrossRef]
- Li, L.; Hsieh, Y.-L. Chitosan Bicomponent Nanofibers and Nanoporous Fibers. Carbohydr Res 2006, 341, 374–381. [CrossRef]
- Sanchez-Alvarado, D.I.; Guzmán-Pantoja, J.; Páramo-García, U.; Maciel-Cerda, A.; Martínez-Orozco, R.D.; Vera-Graziano, R. Morphological Study of Chitosan/Poly (Vinyl Alcohol) Nanofibers Prepared by Electrospinning, Collected on Reticulated Vitreous Carbon. Int J Mol Sci 2018, 19, 1718. [CrossRef]
- Lu, S.; Tao, J.; Liu, X.; Wen, Z. Baicalin-Liposomes Loaded Polyvinyl Alcohol-Chitosan Electrospinning Nanofibrous Films: Characterization, Antibacterial Properties and Preservation Effects on Mushrooms. Food Chem 2022, 371, 131372. [CrossRef]
- Bazzi, M.; Shabani, I.; Mohandesi, J.A. Enhanced Mechanical Properties and Electrical Conductivity of Chitosan/Polyvinyl Alcohol Electrospun Nanofibers by Incorporation of Graphene Nanoplatelets. J Mech Behav Biomed Mater 2022, 125, 104975. [CrossRef]
- Gupta, S.; Pramanik, A.K.; Kailath, A.; Mishra, T.; Guha, A.; Nayar, S.; Sinha, A. Composition Dependent Structural Modulations in Transparent Poly(Vinyl Alcohol) Hydrogels. Colloids Surf B Biointerfaces 2009, 74, 186–190. [CrossRef]
- BUNN, C.W. Crystal Structure of Polyvinyl Alcohol. Nature 1948, 161, 929–930. [CrossRef]
- British Standards Institution. Plastics. Part 3. Test Conditions for Films and Sheets. : Determination of Tensile Properties.; BSI, 1996; ISBN 0580249824.
- Vu, T.H.N.; Morozkina, S.N.; Sitnikova, V.E.; Olekhnovich, R.O.; Podshivalov, A. V.; Uspenskaya, M. V. A Systematic Investigation of Solution and Technological Parameters for the Fabrication and Characterization of Poly (Vinyl Alcohol) –Chitosan Electrospun Nanofibers. Polym Adv Technol 2024, 35. [CrossRef]
- Bohren, C.F. and H.D.R. Absorption and Scattering of Light by Small Particles. John Wiley and Sons, Inc. 1998, 544.
- Haiss, W.; Thanh, N.T.K.; Aveyard, J.; Fernig, D.G. Determination of Size and Concentration of Gold Nanoparticles from UV−Vis Spectra. Anal Chem 2007, 79, 4215–4221. [CrossRef]
- Doak, J.; Gupta, R.K.; Manivannan, K.; Ghosh, K.; Kahol, P.K. Effect of Particle Size Distributions on Absorbance Spectra of Gold Nanoparticles. Physica E Low Dimens Syst Nanostruct 2010, 42, 1605–1609. [CrossRef]
- Byron Bird, R.; Carreau, P.J. A Nonlinear Viscoelastic Model for Polymer Solutions and Melts—I. Chem Eng Sci 1968, 23, 427–434. [CrossRef]
- Cross, M.M. Rheology of Non-Newtonian Fluids: A New Flow Equation for Pseudoplastic Systems. J Colloid Sci 1965, 20, 417–437. [CrossRef]
- Miya, M.; Iwamoto, R.; Mima, S. FT-IR Study of Intermolecular Interactions in Polymer Blends. Journal of Polymer Science: Polymer Physics Edition 1984, 22, 1149–1151. [CrossRef]
- Krimm, S.; Liang, C.Y.; Sutherland, G.B.B.M. Infrared Spectra of High Polymers. V. Polyvinyl Alcohol. Journal of Polymer Science 1956, 22, 227–247. [CrossRef]
- Koosha, M.; Mirzadeh, H. Electrospinning, Mechanical Properties, and Cell Behavior Study of Chitosan/PVA Nanofibers. J Biomed Mater Res A 2015, 103, 3081–3093. [CrossRef]
- Zheng, H.; Du, Y.; Yu, J.; Huang, R.; Zhang, L. Preparation and Characterization of Chitosan/Poly(Vinyl Alcohol) Blend Fibers. J Appl Polym Sci 2001, 80, 2558–2565. [CrossRef]
- Vino, A.B.; Ramasamy, P.; Shanmugam, V.; Shanmugam, A. Extraction, Characterization and in Vitro Antioxidative Potential of Chitosan and Sulfated Chitosan from Cuttlebone of Sepia Aculeata Orbigny, 1848. Asian Pac J Trop Biomed 2012, 2, S334–S341. [CrossRef]
- Song, C.; Yu, H.; Zhang, M.; Yang, Y.; Zhang, G. Physicochemical Properties and Antioxidant Activity of Chitosan from the Blowfly Chrysomya Megacephala Larvae. Int J Biol Macromol 2013, 60, 347–354. [CrossRef]
- Fernandes Queiroz, M.; Melo, K.; Sabry, D.; Sassaki, G.; Rocha, H. Does the Use of Chitosan Contribute to Oxalate Kidney Stone Formation? Mar Drugs 2014, 13, 141–158. [CrossRef]
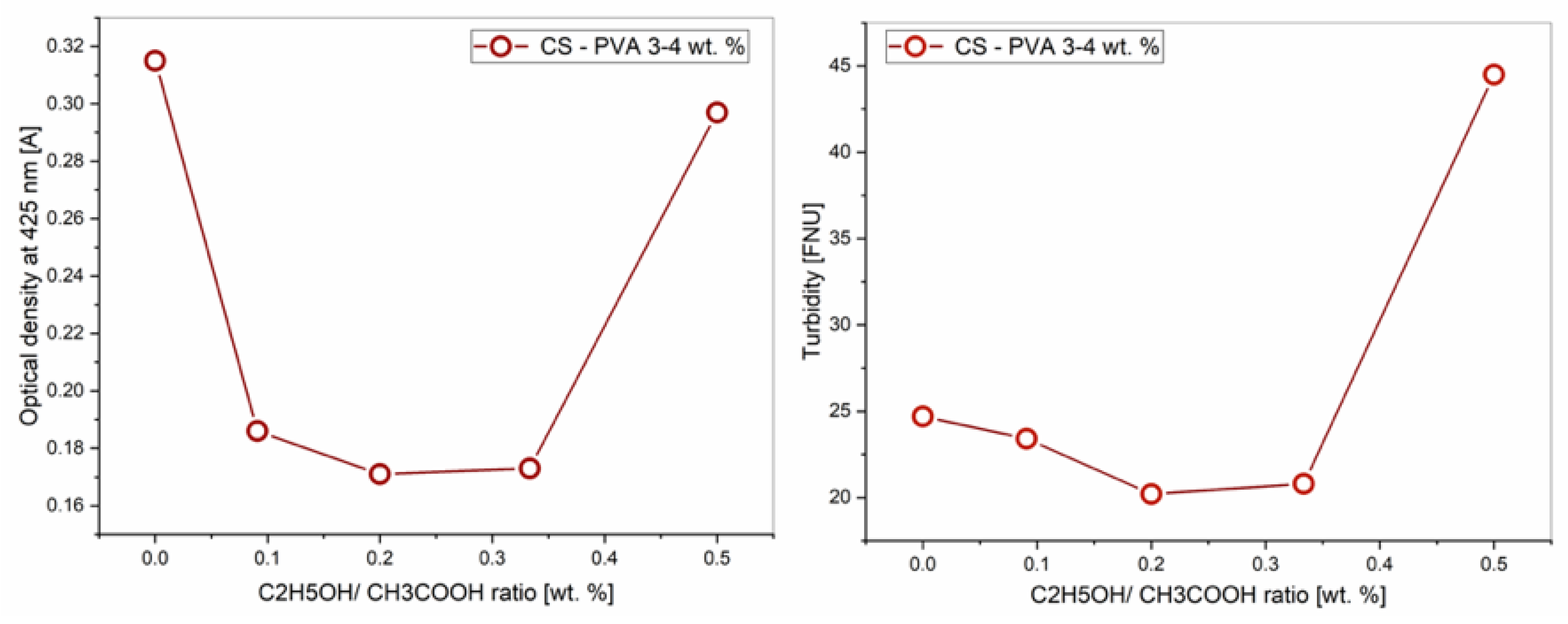
| CH3COOH/C2H5OH, % | Optical density | Turbidity, FNU | |
|---|---|---|---|
| Absorption (А) | Transmission (Т), % | ||
| 60/0 | 0,315 | 48,4 | 24,7 |
| 55/5 | 0,186 | 65,2 | 23,4 |
| 50/10 | 0,171 | 67,4 | 20,2 |
| 45/15 | 0,173 | 67,2 | 20,8 |
| 40/20 | 0,297 | 50,5 | 44,5 |
| C2H5OH/ CH3COOH, (%) | pH | Viscosity ⴄ, (mPa·s) | Electrical conductivity Ɜ, (μS / cm) |
|---|---|---|---|
| 0/60 | 2.02 | 2724.3 | 1611 |
| 5/55 | 2.25 | 2644.6 | 1541 |
| 10/50 | 2.48 | 2796.7 | 1505 |
| 15/45 | 2.71 | 2338.4 | 1434 |
| 20/40 | 2.90 | 3036.7 | 1402 |
| Distance (mm) | Feed rate (mL/h) | CCH3COOH (% w/w) |
CC2H5OH (% w/w) |
Voltage (kV) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 16 | 18 | 20 | 22 | 24 | 26 | 27 | 28 | 29 | 30 | ||||
| 150 | 0.1 | 60 | 0 | O | O | o | + | + | + | + | + | + | + |
| 55 | 5 | O | O | o | + | + | + | + | + | + | + | ||
| 50 | 10 | O | O | o | + | + | + | + | + | + | + | ||
| 45 | 15 | O | o | + | + | + | + | + | + | + | + | ||
| 40 | 20 | O | o | + | + | + | + | + | + | + | + | ||
| 0.2 | 60 | 0 | O | O | O | o | + | + | + | + | + | + | |
| 55 | 5 | O | O | o | o | + | + | + | + | + | + | ||
| 50 | 10 | O | O | o | o | + | + | + | + | + | + | ||
| 45 | 15 | O | O | o | + | + | + | + | + | + | + | ||
| 40 | 20 | O | O | o | + | + | + | + | + | + | + | ||
| 140 | 0.1 | 60 | 0 | O | o | + | + | + | + | + | + | + | + |
| 55 | 5 | O | O | o | + | + | + | + | + | + | + | ||
| 50 | 10 | O | O | o | + | + | + | + | + | + | + | ||
| 45 | 15 | O | o | + | + | + | + | + | + | + | + | ||
| 40 | 20 | O | o | + | + | + | + | + | + | + | + | ||
| 0.2 | 60 | 0 | O | O | O | o | + | + | + | + | + | + | |
| 55 | 5 | O | O | o | o | + | + | + | + | + | + | ||
| 50 | 10 | O | O | o | * | + | + | + | + | + | + | ||
| 45 | 15 | O | O | * | + | + | + | + | + | + | + | ||
| 40 | 20 | O | O | o | + | + | + | + | + | + | + | ||
| 120 | 0.1 | 60 | 0 | O | o | + | + | + | + | + | + | + | + |
| 55 | 5 | O | o | + | + | + | + | + | + | + | + | ||
| 50 | 10 | O | o | + | + | + | + | + | + | + | + | ||
| 45 | 15 | o | + | + | + | + | + | + | + | + | + | ||
| 40 | 20 | o | o | + | + | + | + | + | + | + | + | ||
| 0.2 | 60 | 0 | O | o | o | * | * | + | + | + | + | + | |
| 55 | 5 | O | O | o | + | + | + | + | + | + | + | ||
| 50 | 10 | O | O | o | + | + | + | + | + | + | + | ||
| 45 | 15 | O | o | + | + | + | + | + | + | + | + | ||
| 40 | 20 | O | o | o | + | + | + | + | + | + | + | ||
| 100 | 0.1 | 60 | 0 | O | o | + | + | + | + | + | + | + | + |
| 55 | 5 | O | o | + | + | + | + | + | + | + | + | ||
| 50 | 10 | O | o | + | + | + | + | + | + | + | + | ||
| 45 | 15 | o | + | + | + | + | + | + | + | + | + | ||
| 40 | 20 | o | + | + | + | + | + | + | + | + | + | ||
| 0.2 | 60 | 0 | O | o | + | + | + | + | + | + | + | + | |
| 55 | 5 | O | o | * | + | + | + | + | + | + | + | ||
| 50 | 10 | O | o | * | + | + | + | + | + | + | + | ||
| 45 | 15 | o | * | + | + | + | + | + | + | + | + | ||
| 40 | 20 | o | * | + | + | + | + | + | + | + | + | ||
|
(+) the formation of fibers; (O) the formation of droplets and a few fibers; (o) the formation of fibers and a few drops; (*) the formation of fibers, but the process is unstable | |||||||||||||
| Diameter (nm) | C2H5OH/CH3COOH ratio (% w/w) | ||||
|---|---|---|---|---|---|
| 0/60 | 5/55 | 10/50 | 15/45 | 20/40 | |
| Mean | 330 | 320 | 301 | 285 | 337 |
| Standard deviation | 68 | 59 | 68 | 65 | 91 |
| Min | 134 | 147 | 110 | 121 | 170 |
| Max | 587 | 573 | 533 | 592 | 654 |
| Diameter (nm) | Needle–collector distance (mm) | ||||
|---|---|---|---|---|---|
| 100 | 120 | 130 | 140 | 150 | |
| Mean | 313 | 302 | 295 | 285 | 308 |
| Standard deviation | 77 | 75 | 70 | 65 | 76 |
| Min | 139 | 124 | 124 | 121 | 155 |
| Max | 646 | 562 | 546 | 592 | 540 |
| Diameter (nm) | Feed rate (mL/h) | |||
|---|---|---|---|---|
| 0.1 | 0.2 | 0.3 | 0.4 | |
| Mean | 311 | 285 | 300 | 335 |
| Standard deviation | 75 | 65 | 79 | 83 |
| Min | 154 | 121 | 134 | 131 |
| Max | 513 | 592 | 670 | 650 |
| Ethanol– acetic acid ratio | Tensile strength [MPa] | Elongation at Break [%] | Young's modulus [MPa] | |||
|---|---|---|---|---|---|---|
| Vertical | Horizontal | Vertical | Horizontal | Vertical | Horizontal | |
| 0–60 | 5.937 ± 1.046 | 5.673 ± 0.560 | 14.05 ± 1.75 | 17.06 ± 0.47 | 343.532 ± 33.840 | 289.180 ± 74.027 |
| 5–55 | 5.594 ± 1.501 | 1.617 ± 0.416 | 12.12 ± 1.64 | 13.27 ± 1.10 | 424.921 ± 72.966 | 246.112 ± 34.894 |
| 10–50 | 4.732 ± 0.457 | 1.900 ± 0.450 | 11.80 ± 1.48 | 13.97 ± 2.14 | 427.543 ± 50.422 | 270.074 ± 40.440 |
| 15–45 | 7.916 ± 0.970 | 4.912 ± 0.965 | 13. 07 ± 0.53 | 16.40 ± 0.82 | 546.113 ± 32.150 | 333.086 ± 23.851 |
| 20–40 | 4.536 ± 0.918 | 2.893 ± 0.220 | 12.69 ± 1.96 | 14.10 ± 1.90 | 398.311 ± 47.879 | 329.703 ± 29.510 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




