Submitted:
01 October 2024
Posted:
04 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
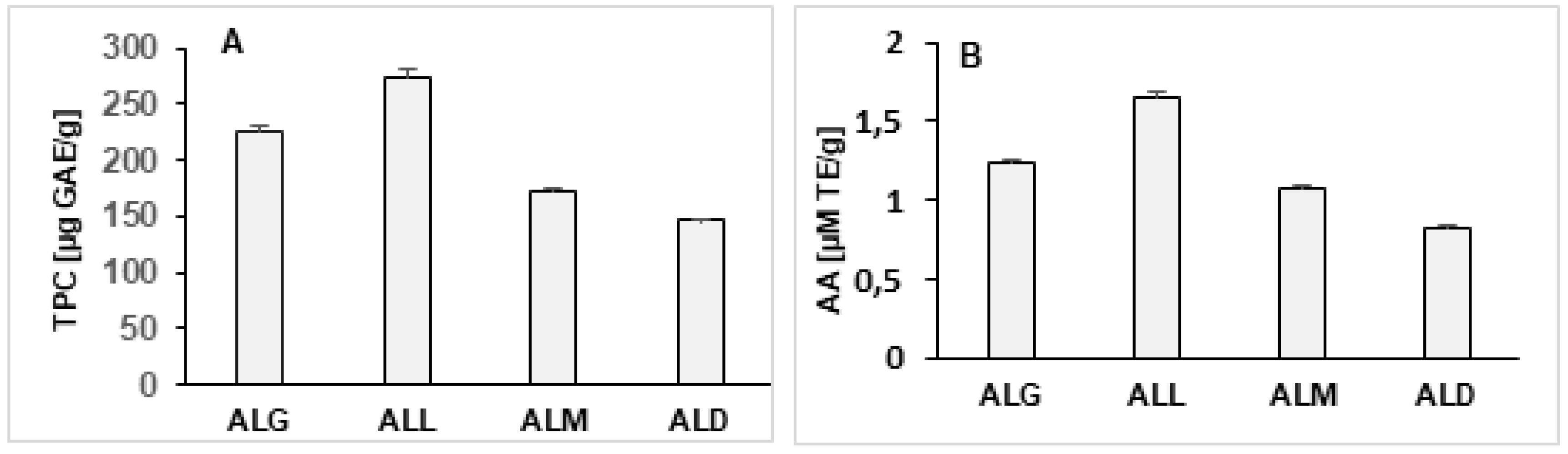
2.1. Total Phenolic Content (TPC) of the Coffee Extracts
2.2. Antioxidant Activity of the Coffee Extract
2.3. Chlorogenic Acids (CGAs) Content in the Coffee Extracts
2.4. Free phenolic Acids (FPAs) Content in the Coffee Extracts Determined after an Alkaline Hydrolysis
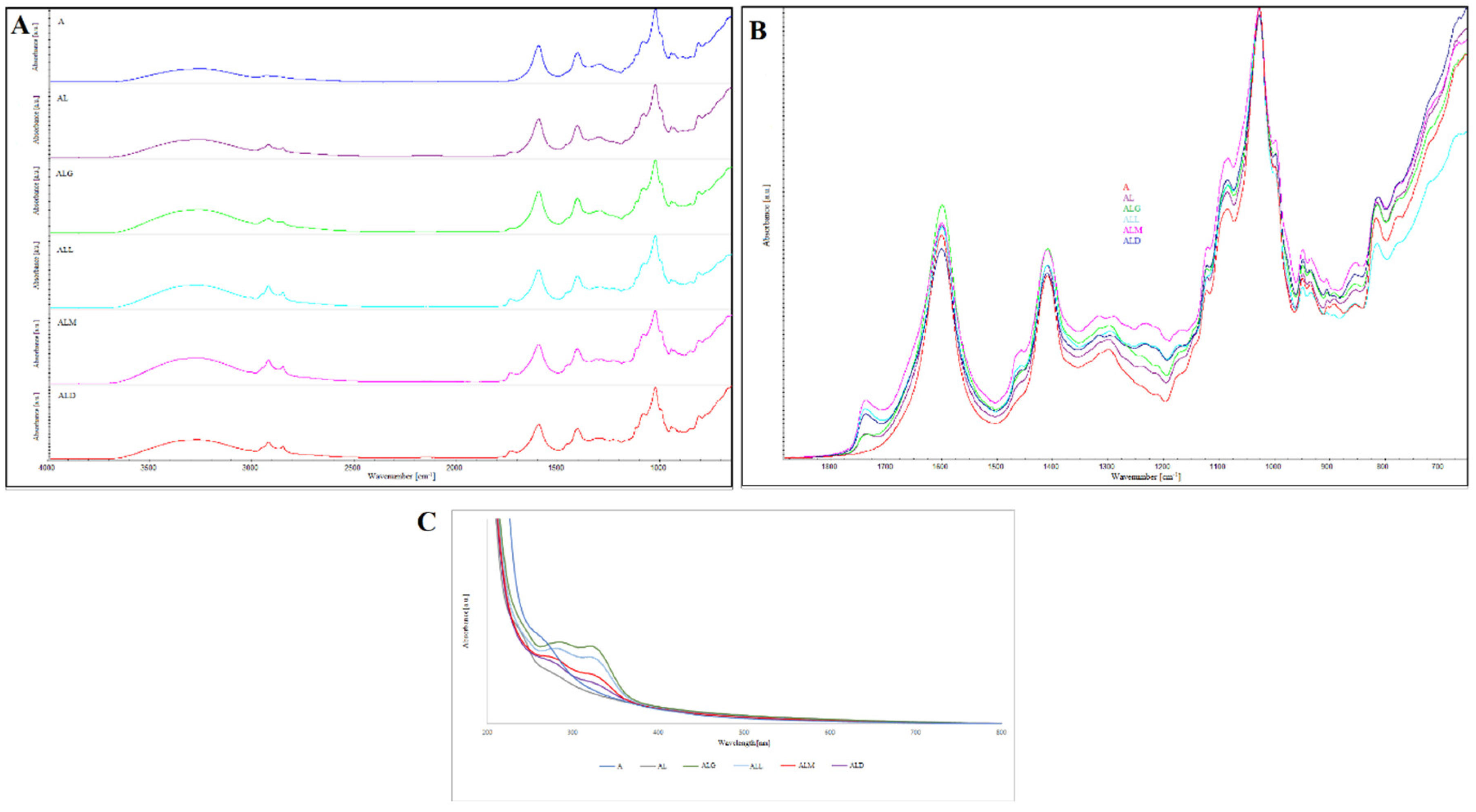
2.5. ATR-FTIR Spectrophotometry
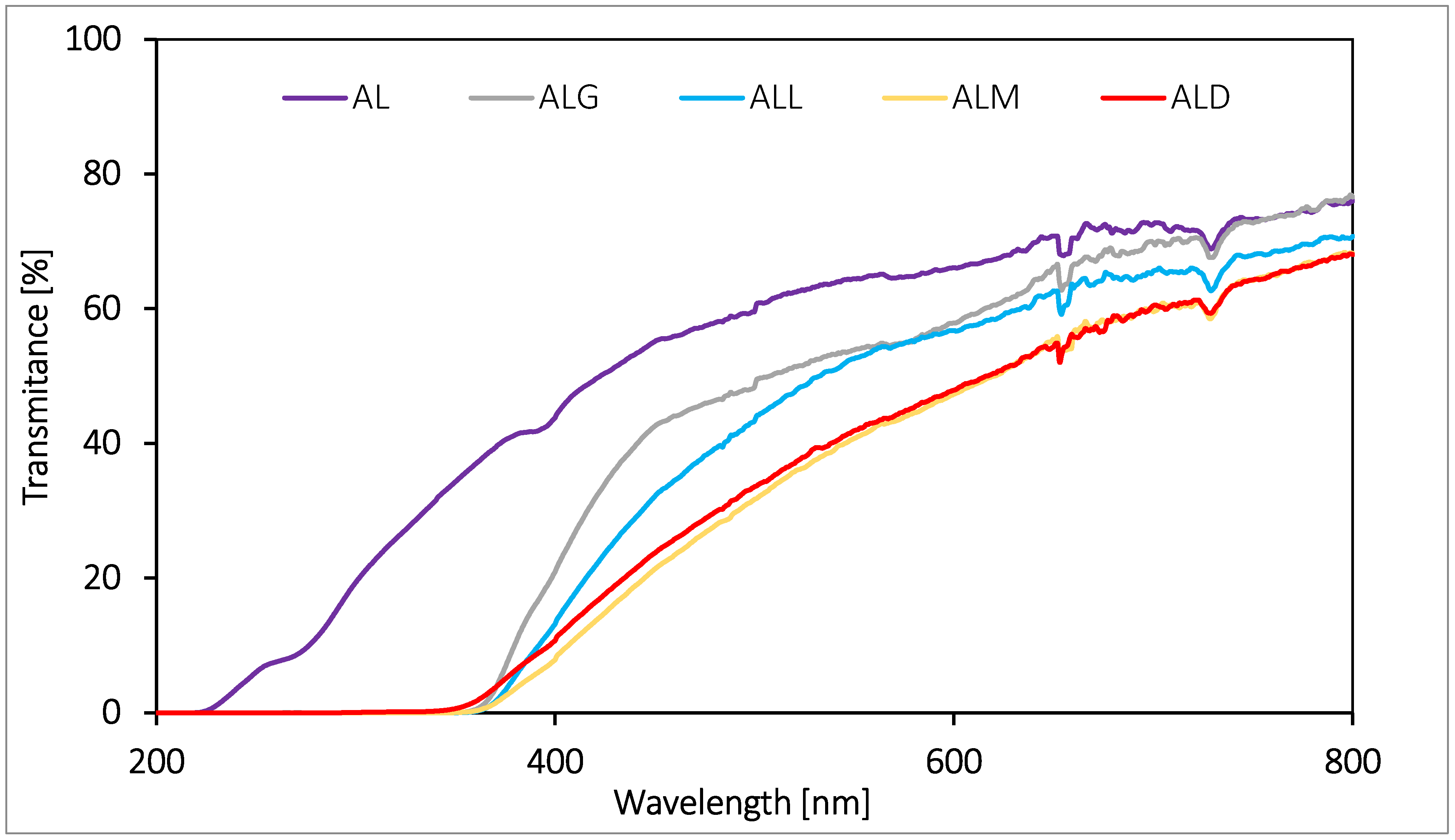
2.6. UV–Vis Spectrophotometry
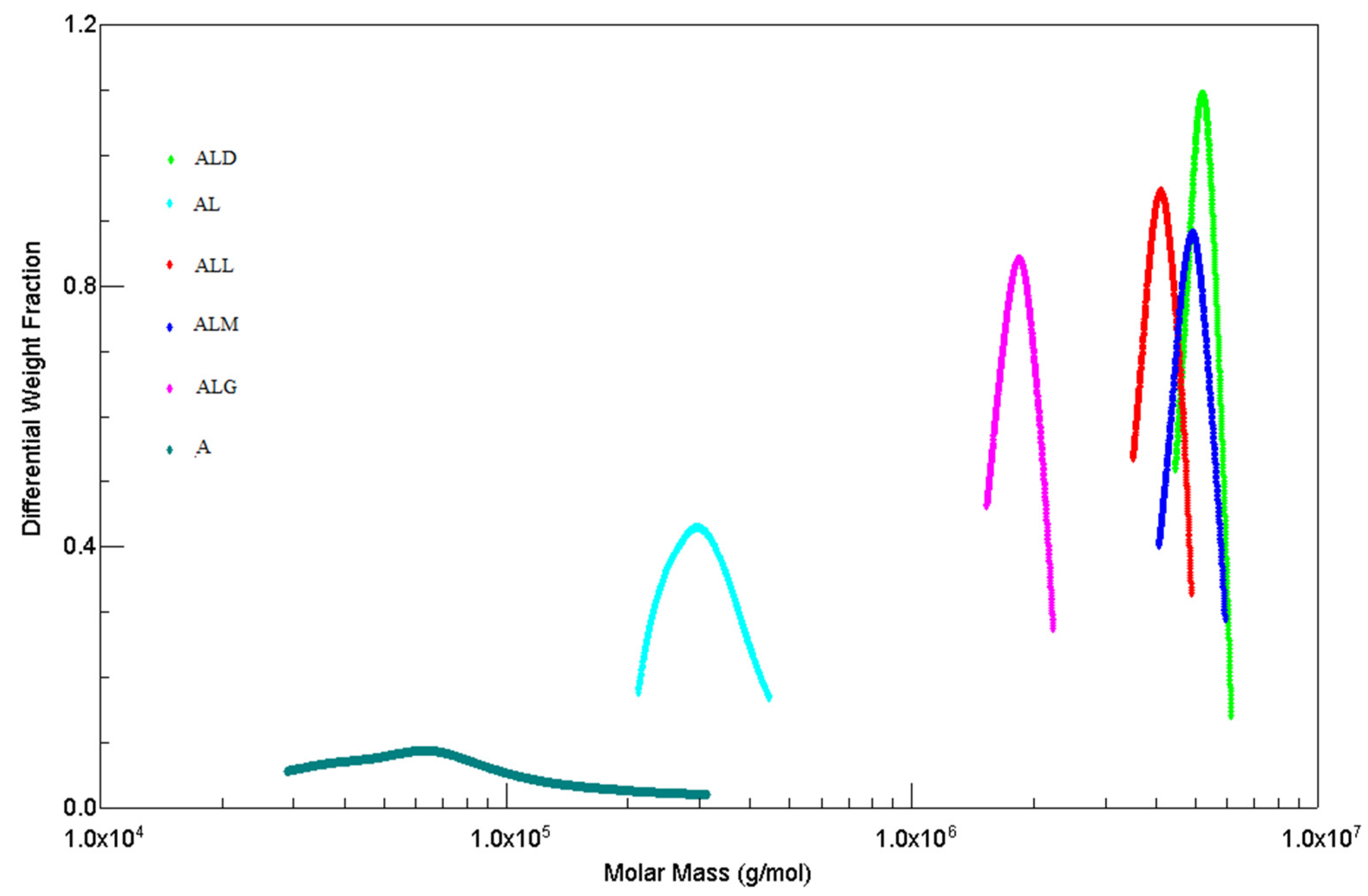
2.7. High-Performance Size Exclusion Chromatography (HPSEC–MALLS–RI)
2.8. Thermal (DSC) Analysis of the Alginate Films
2.9. Mechanical Properties of the Films
2.10. Water Absorption and Solubility
2.11. Water Contact Angle Determination
2.12. Foil Barrier Properties Estimated against UV-Vis Light
2.12. Color Parameters and Opacity of the Films
2.13. Determination of TPC and AA of the Coffee Extracts and Films
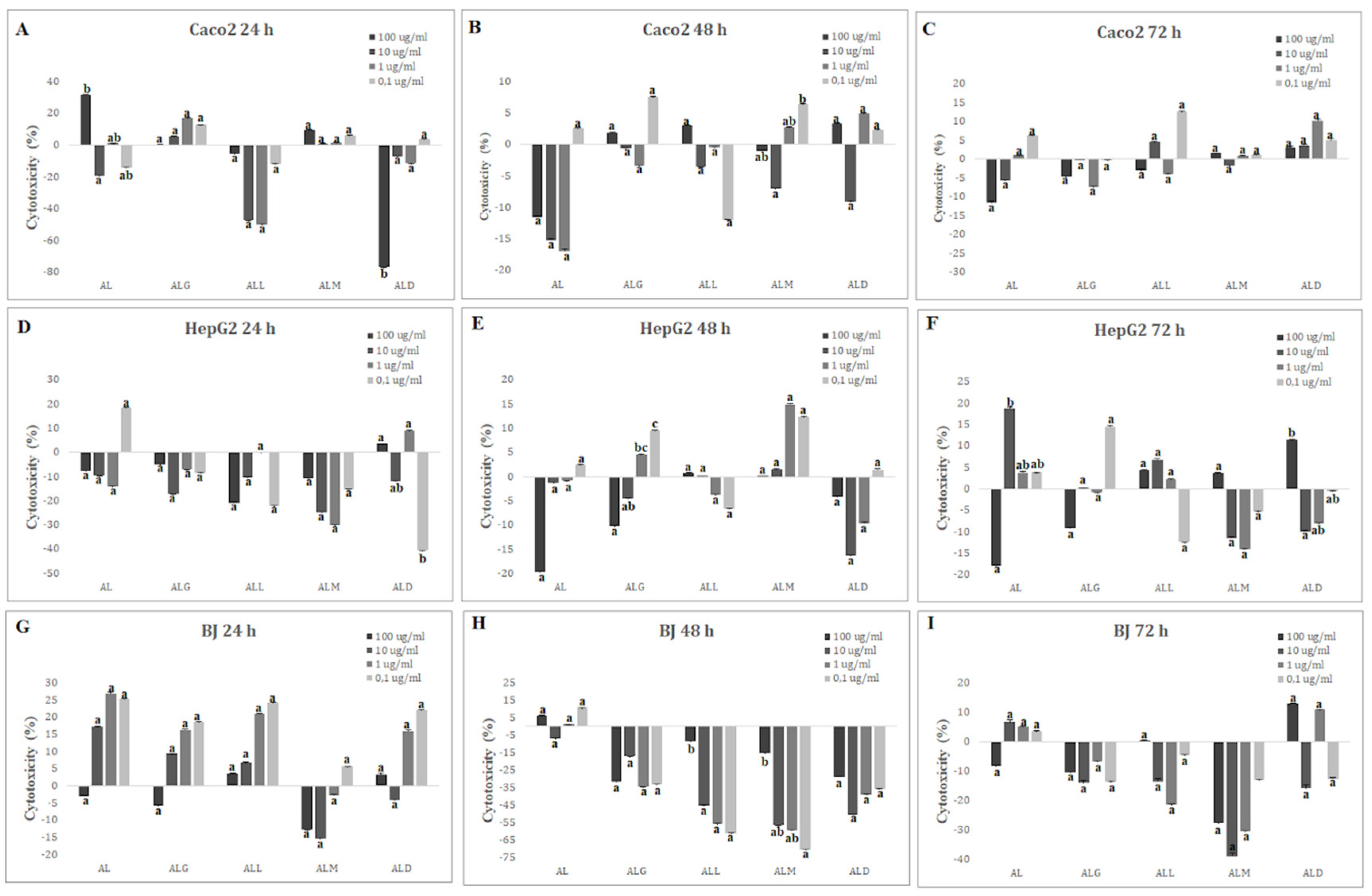
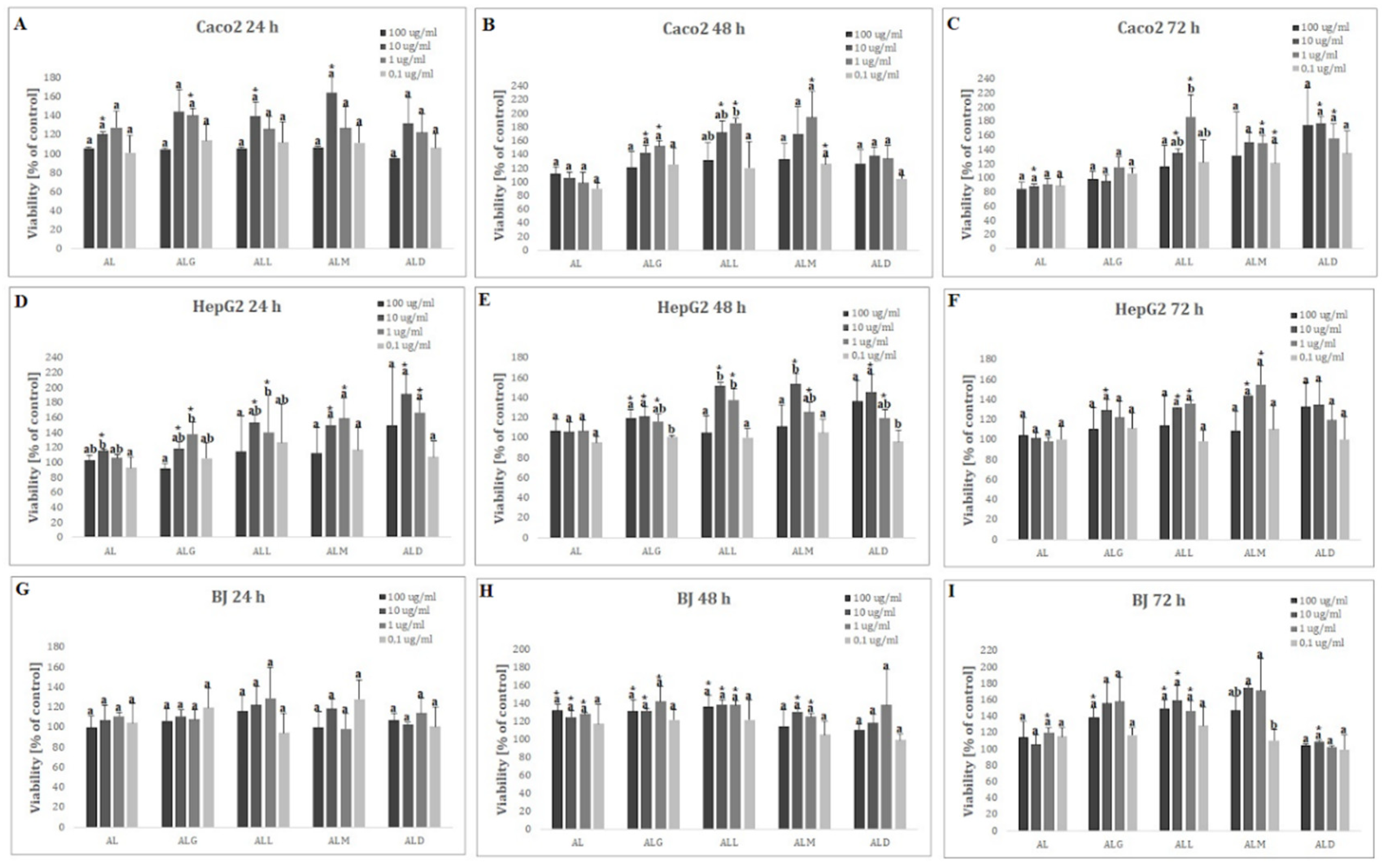
2.14. Cytotoxicity and Viability Analysis
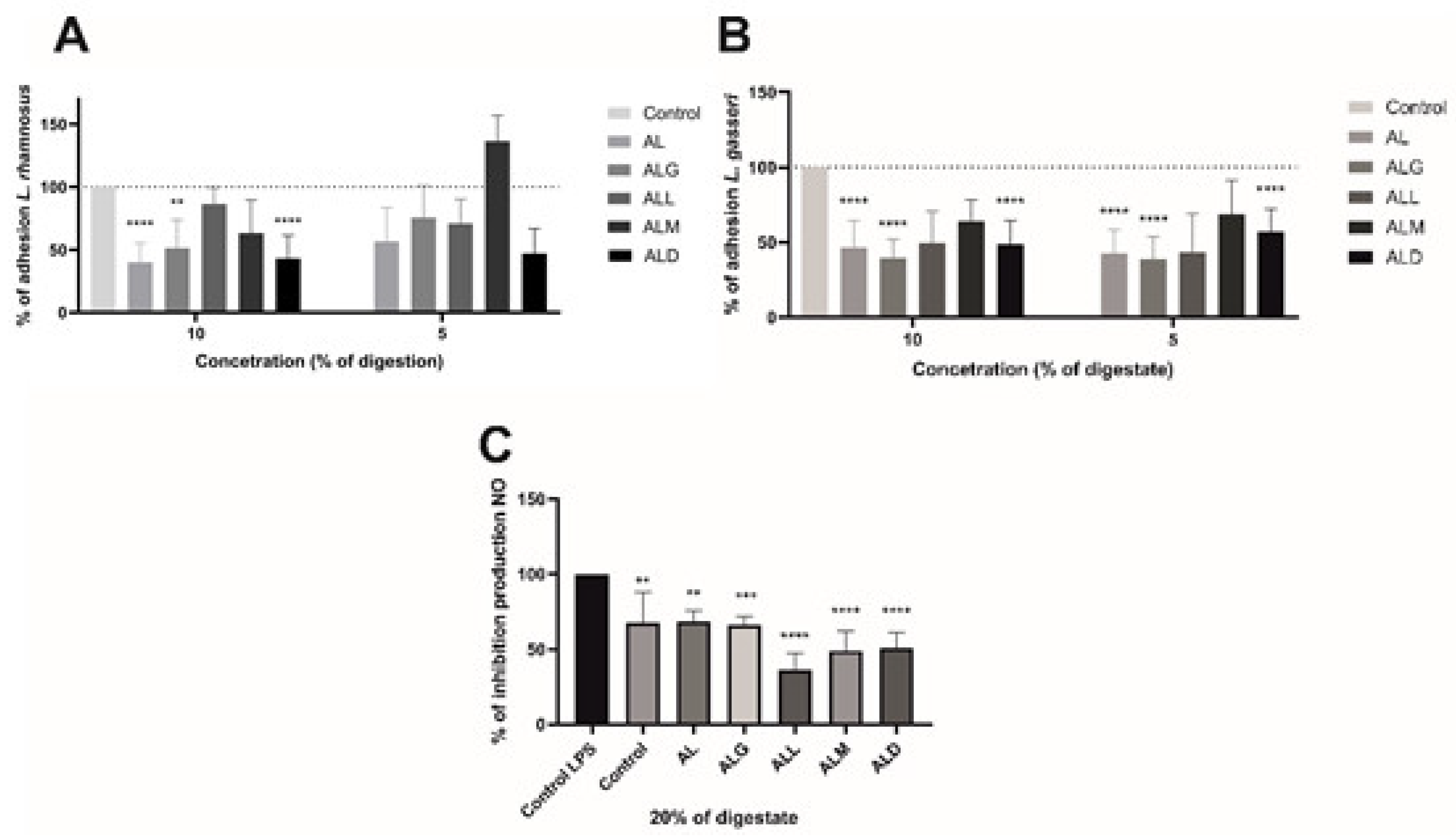
2.15. Influence of Digestion of Film to Adhesion of Lactic Acid Bacteria and Anti-Inflammatory Response in Murine Macrophages on Their Produce Nitric Oxide
2.16. Microbiological Tests
3. Materials and Methods
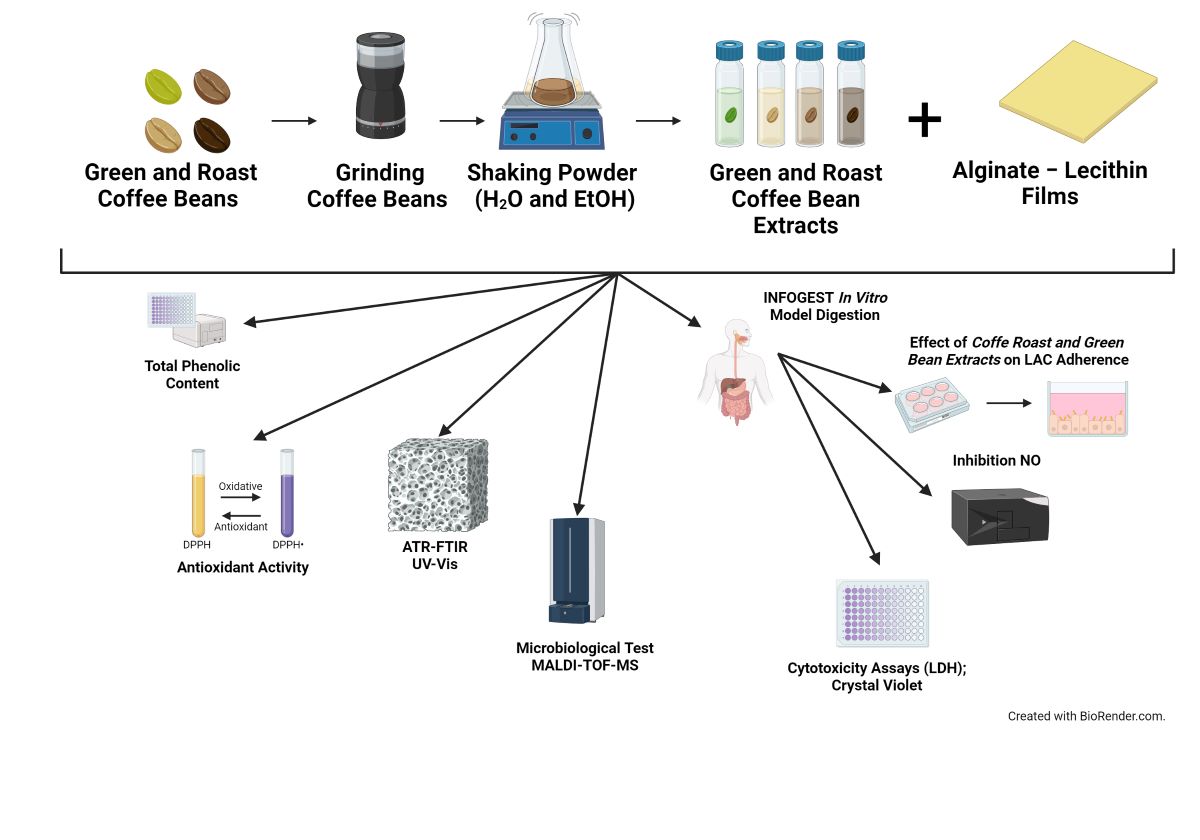
3.1. Coffee Beans
3.2. Preparation of Coffee Extract
3.3. Carbohydrate Polymer Characterization
3.4. Films Preparation
3.5. Determination of Total Phenolic Content (TPC) in the Coffee Extracts
3.6. Determination of Antioxidant Activity (AA) in the Coffee Extracts
3.7. Determination of chlorogenic Acids (CGAs) Content in the Studied Coffee Extracts
3.8. Determination of Free Phenolic Acids (FPA) in the Studied Coffee Extracts
3.9. Physicochemical Properties of the Films
3.9.1. ATR-FTIR Spectrophotometry
3.9.2. UV–Vis Spectrophotometry
3.9.3. High-Performance Size Exclusion Chromatography (HPSEC–MALLS–RI)
3.9.4. Differential Scanning Calorimetry (DSC) Analysis
3.10. Determination of Mechanical Properties of the Films
3.11. Water Content and Solubility of the Films
3.12. Water Contact Angle Determination
3.13. Determination of Foil Barrier Properties against UV-Vis Light
3.14. Determination of the Color Parameters and Opacity
3.15. Determination of Total Phenolic Content
3.16. Determination of Antioxidant Activity of the Films
3.17. In Vitro Digestion of Films
3.18. Cell Culture
3.19. Caco-2 Cells Permeability Assay
3.20. In vitro Cytotoxicity Analysis
3.21. Effect of Digestate on the Adhesion of Lactic Acid Bacteria
3.22. Effect of Digested on Inhibition of NO
3.23. Microbiological Tests
3.24. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zactiti, E.M.; Kieckbusch, T.G. Potassium Sorbate Permeability in Biodegradable Alginate Films: Effect of the Antimicrobial Agent Concentration and Crosslinking Degree. J Food Eng 2006, 77, 462–467. [Google Scholar] [CrossRef]
- Senturk Parreidt, T.; Müller, K.; Schmid, M. Alginate-Based Edible Films and Coatings for Food Packaging Applications. Foods 2018, 7. [Google Scholar] [CrossRef]
- Senturk Parreidt, T.; Schott, M.; Schmid, M.; Müller, K. Effect of Presence and Concentration of Plasticizers, Vegetable Oils, and Surfactants on the Properties of Sodium-Alginate-Based Edible Coatings. Int J Mol Sci 2018, 19. [Google Scholar] [CrossRef]
- Leimann, F. V.; Gonçalves, O.H.; Sakanaka, L.S.; Azevedo, A.S.B.; Lima, M. V.; Barreiro, F.; Shirai, M.A. Active Food Packaging From Botanical, Animal, Bacterial, and Synthetic Sources. In Food Packaging and Preservation; Elsevier, 2018; pp. 87–135.
- Zhang, H.; Dudley, E.G.; Davidson, P.M.; Harte, F. Critical Concentration of Lecithin Enhances the Antimicrobial Activity of Eugenol against Escherichia Coli. Appl Environ Microbiol 2017, 83, e03467-16. [Google Scholar] [CrossRef]
- Perrone, D.; Farah, A.; Donangelo, C.; Paulis, T.; Martin, P. Comprehensive Analysis of Major and Minor Chlorogenic Acids and Lactones in Economically Relevant Brazilian Coffee Cultivars. Food Chem 2008, 106, 859–867. [Google Scholar] [CrossRef]
- van Zeeland, A.A.; de Groot, A.J.L.; Hall, J.; Donato, F. 8-Hydroxydeoxyguanosine in DNA from Leukocytes of Healthy Adults: Relationship with Cigarette Smoking, Environmental Tobacco Smoke, Alcohol and Coffee Consumption. Mutation Research/Genetic Toxicology and Environmental Mutagenesis 1999, 439, 249–257. [Google Scholar] [CrossRef]
- Almeida, A.A.P.; Farah, A.; Silva, D.A.M.; Nunan, E.A.; Glória, M.B.A. Antibacterial Activity of Coffee Extracts and Selected Coffee Chemical Compounds against Enterobacteria. J Agric Food Chem 2006, 54, 8738–8743. [Google Scholar] [CrossRef]
- da Silva, C.Q.; Fernandes, A. da S. ; Teixeira, G.F.; França, R.J.; Marques, M.R. da C.; Felzenszwalb, I.; Falcão, D.Q.; Ferraz, E.R.A. Risk Assessment of Coffees of Different Qualities and Degrees of Roasting. Food Research International 2021, 141, 110089. [Google Scholar] [CrossRef]
- Amigo-Benavent, M.; Wang, S.; Mateos, R.; Sarriá, B.; Bravo, L. Antiproliferative and Cytotoxic Effects of Green Coffee and Yerba Mate Extracts, Their Main Hydroxycinnamic Acids, Methylxanthine and Metabolites in Different Human Cell Lines. Food and Chemical Toxicology 2017, 106, 125–138. [Google Scholar] [CrossRef]
- Nunes, F.M.; Coimbra, M.A. Role of Hydroxycinnamates in Coffee Melanoidin Formation. Phytochemistry Reviews 2010, 9, 171–185. [Google Scholar] [CrossRef]
- Fujioka, K.; Shibamoto, T. Chlorogenic Acid and Caffeine Contents in Various Commercial Brewed Coffees. Food Chem 2008, 106, 217–221. [Google Scholar] [CrossRef]
- Andrade, P.B.; Leitão, R.; Seabra, R.M.; Oliveira, M.B.; Ferreira, M.A. 3,4-Dimethoxycinnamic Acid Levels as a Tool for Differentiation of Coffea Canephora Var. Robusta and Coffea Arabica. Food Chem 1998, 61, 511–514. [Google Scholar] [CrossRef]
- Abdel Aziz, M.S.; Salama, H.E. Developing Multifunctional Edible Coatings Based on Alginate for Active Food Packaging. Int J Biol Macromol 2021, 190, 837–844. [Google Scholar] [CrossRef]
- Badita, C.R.; Aranghel, D.; Burducea, C.; Mereuta, P. Characterization of Sodium Alginate Based Films. Rom. J. Phys 2020, 65, 1–8. [Google Scholar]
- Othman, F.; Idris, S.N.; Nasir, N.A.H.A.; Nawawi, M.A. Preparation and Characterization of Sodium Alginate-Based Edible Film with Antibacterial Additive Using Lemongrass Oil. Sains Malays 2022. [CrossRef]
- Kuligowski, J.; Quintás, G.; Esteve-Turrillas, F.A.; Garrigues, S.; De la Guardia, M. On-Line Gel Permeation Chromatography–Attenuated Total Reflectance–Fourier Transform Infrared Determination of Lecithin and Soybean Oil in Dietary Supplements. J Chromatogr A 2008, 1185, 71–77. [Google Scholar] [CrossRef]
- Setiadi, S.; Hidayah, N. The Effect of Papain Enzyme Dosage on the Modification of Egg-Yolk Lecithin Emulsifier Product through Enzymatic Hydrolysis Reaction. International Journal of Technology 2018, 9, 380–389. [Google Scholar] [CrossRef]
- Shah, P.R.; Gaitonde, U.N.; Ganesh, A. Influence of Soy-Lecithin as Bio-Additive with Straight Vegetable Oil on CI Engine Characteristics. Renew Energy 2018, 115, 685–696. [Google Scholar] [CrossRef]
- Obeidat, S.M.; Hammoudeh, A.Y.; Alomary, A.A. Application of FTIR Spectroscopy for Assessment of Green Coffee Beans According to Their Origin. J Appl Spectrosc 2018, 84, 1051–1055. [Google Scholar] [CrossRef]
- Sahachairungrueng, W.; Meechan, C.; Veerachat, N.; Thompson, A.K.; Teerachaichayut, S. Assessing the Levels of Robusta and Arabica in Roasted Ground Coffee Using NIR Hyperspectral Imaging and FTIR Spectroscopy. Foods 2022, 11, 3122. [Google Scholar] [CrossRef]
- Lyman, D.J.; Benck, R.; Dell, S.; Merle, S.; Murray-Wijelath, J. FTIR-ATR Analysis of Brewed Coffee: Effect of Roasting Conditions. J Agric Food Chem 2003, 51, 3268–3272. [Google Scholar] [CrossRef]
- Ribeiro, J.S.; Salva, T.J.; Ferreira, M.M.C. Chemometric Studies for Quality Control of Processed Brazilian Coffees Using Drifts. J Food Qual 2010, 33, 212–227. [Google Scholar] [CrossRef]
- Craig, A.P.; Franca, A.S.; Oliveira, L.S.; Irudayaraj, J.; Ileleji, K. Application of Elastic Net and Infrared Spectroscopy in the Discrimination between Defective and Non-Defective Roasted Coffees. Talanta 2014, 128, 393–400. [Google Scholar] [CrossRef]
- Zhang, Q.; Lian, H.-Z.; Wang, W.; Chen, H.-Y. Separation of Caffeine and Theophylline in Poly(Dimethylsiloxane) Microchannel Electrophoresis with Electrochemical Detection. J Chromatogr A 2006, 1098, 172–176. [Google Scholar] [CrossRef]
- Belay, A.; Ture, K.; Redi, M.; Asfaw, A. Measurement of Caffeine in Coffee Beans with UV/Vis Spectrometer. Food Chem 2008, 108, 310–315. [Google Scholar] [CrossRef]
- Gemta, A.; Gholap, A. Characterization and Determination of Chlorogenic Acid (CGA) in Coffee Beans by UV-Vis Spectroscopy. African Journal of Pure and Applied Chemistry 2009, 3, 234–240. [Google Scholar] [CrossRef]
- Khachatryan, G.; Krzeminska-Fiedorowicz, L.; Nowak, E.; Fiedorowicz, M. Molecular Structure and Physicochemical Properties of Hylon V and Hylon VII Starches Illuminated with Linearly Polarised Visible Light. LWT-Food Science and Technology 2014, 58, 256–262. [Google Scholar] [CrossRef]
- Kadzińska, J.; Bryś, J.; Ostrowska-Ligȩza, E.; Estéve, M.; Janowicz, M. Influence of Vegetable Oils Addition on the Selected Physical Properties of Apple–Sodium Alginate Edible Films. Polymer Bulletin 2019, 77, 883–900. [Google Scholar] [CrossRef]
- Mimmo, T.; Marzadori, C.; Montecchio, D.; Gessa, C. Characterisation of Ca- and Al-Pectate Gels by Thermal Analysis and FT-IR Spectroscopy. Carbohydr Res 2005, 340, 2510—2519. [Google Scholar] [CrossRef]
- Pereira, R.; Carvalho, A.; Vaz, D.C.; Gil, M.H.; Mendes, A.; Bártolo, P. Development of Novel Alginate Based Hydrogel Films for Wound Healing Applications. Int J Biol Macromol 2013, 52, 221–230. [Google Scholar] [CrossRef]
- Tongdeesoontorn, W.; Mauer, L.J.; Wongruong, S.; Sriburi, P.; Reungsang, A.; Rachtanapun, P. Antioxidant Films from Cassava Starch/Gelatin Biocomposite Fortified with Quercetin and TBHQ and Their Applications in Food Models. Polymers (Basel) 2021, 13. [Google Scholar] [CrossRef]
- Dou, L.; Li, B.; Zhang, K.; Chu, X.; Hou, H. Physical Properties and Antioxidant Activity of Gelatin-Sodium Alginate Edible Films with Tea Polyphenols. Int J Biol Macromol 2018, 118, 1377–1383. [Google Scholar] [CrossRef]
- Bisht, A.; Alam, M.; Bhatia, S.; Gupta, S. Studies on Development and Evaluation of Glycerol Incorporated Cellulose and Alginate Based Edible Films. Indian Journal of Agricultural Biochemistry 2017, 30, 67. [Google Scholar] [CrossRef]
- Such, A.; Wisła-Świder, A.; Węsierska, E.; Nowak, E.; Szatkowski, P.; Kopcińska, J.; Koronowicz, A. Edible Chitosan-Alginate Based Coatings Enriched with Turmeric and Oregano Additives: Formulation, Antimicrobial and Non-Cytotoxic Properties. Food Chem 2023, 426, 136662. [Google Scholar] [CrossRef]
- Hutachok, N.; Koonyosying, P.; Pankasemsuk, T.; Angkasith, P.; Chumpun, C.; Fucharoen, S.; Srichairatanakool, S. Chemical Analysis, Toxicity Study, and Free-Radical Scavenging and Iron-Binding Assays Involving Coffee (Coffea Arabica) Extracts. Molecules 2021, 26. [Google Scholar] [CrossRef]
- Grzelczyk, J.; Szwajgier, D.; Baranowska-Wójcik, E.; Budryn, G.; Zakłos-Szyda, M.; Sosnowska, B. Bioaccessibility of Coffee Bean Hydroxycinnamic Acids during in Vitro Digestion Influenced by the Degree of Roasting and Activity of Intestinal Probiotic Bacteria, and Their Activity in Caco-2 and HT29 Cells. Food Chem 2022, 392, 133328. [Google Scholar] [CrossRef]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of Polyphenols on Gut Microbiota and Implications in Human Health. J Nutr Biochem 2013, 24, 1415–1422. [Google Scholar] [CrossRef]
- Vamanu, E.; Gatea, F. Correlations between Microbiota Bioactivity and Bioavailability of Functional Compounds: A Mini-Review. Biomedicines 2020, 8. [Google Scholar] [CrossRef]
- Corrêa, T.A.F.; Rogero, M.M.; Hassimotto, N.M.A.; Lajolo, F.M. The Two-Way Polyphenols-Microbiota Interactions and Their Effects on Obesity and Related Metabolic Diseases. Front Nutr 2019, 6. [Google Scholar] [CrossRef]
- Kim, S.-H.; Park, S.-Y.; Park, Y.-L.; Myung, D.-S.; Rew, J.-S.; Joo, Y.-E. Chlorogenic Acid Suppresses Lipopolysaccharide-Induced Nitric Oxide and Interleukin-1β Expression by Inhibiting JAK2/STAT3 Activation in RAW264. 7 Cells. Mol Med Rep 2017, 16, 9224–9232. [Google Scholar] [CrossRef]
- Funakoshi-Tago, M.; Nonaka, Y.; Tago, K.; Takeda, M.; Ishihara, Y.; Sakai, A.; Matsutaka, M.; Kobata, K.; Tamura, H. Pyrocatechol, a Component of Coffee, Suppresses LPS-Induced Inflammatory Responses by Inhibiting NF-ΚB and Activating Nrf2. Sci Rep 2020, 10, 2584. [Google Scholar] [CrossRef]
- Rebollo-Hernanz, M.; Zhang, Q.; Aguilera, Y.; Martín-Cabrejas, M.A.; Gonzalez de Mejia, E. Relationship of the Phytochemicals from Coffee and Cocoa By-Products with Their Potential to Modulate Biomarkers of Metabolic Syndrome In Vitro. Antioxidants 2019, 8, 279. [Google Scholar] [CrossRef]
- Zhong, R.; Miao, L.; Zhang, H.; Tan, L.; Zhao, Y.; Tu, Y.; Angel Prieto, M.; Simal-Gandara, J.; Chen, L.; He, C.; et al. Anti-Inflammatory Activity of Flavonols via Inhibiting MAPK and NF-ΚB Signaling Pathways in RAW264. 7 Macrophages. Curr Res Food Sci 2022, 5, 1176–1184. [Google Scholar] [CrossRef]
- Rosyidi, D.; Rosyidi, D.; Radiati, L.E.; Amri, I.A.; Prasetyo, D.; Qosimah, D.; Murwani, S. Antibacterial Activity of Green Coffee Bean Extract against Staphylococcus Aureus and Salmonella Enteritidis. Biotika 2018, v. 20, 12-16–2018 v.20 no.1.
- Luo, C.; Walk, S.T.; Gordon, D.M.; Feldgarden, M.; Tiedje, J.M.; Konstantinidis, K.T. Genome Sequencing of Environmental Escherichia Coli Expands Understanding of the Ecology and Speciation of the Model Bacterial Species. Proceedings of the National Academy of Sciences 2011, 108, 7200–7205. [Google Scholar] [CrossRef]
- Mirzajani, F.; Ghassempour, A.; Aliahmadi, A.; Esmaeili, M.A. Antibacterial Effect of Silver Nanoparticles on Staphylococcus Aureus. Res Microbiol 2011, 162, 542–549. [Google Scholar] [CrossRef]
- Salgado-Pabón, W.; Schlievert, P.M. Models Matter: The Search for an Effective Staphylococcus Aureus Vaccine. Nat Rev Microbiol 2014, 12, 585–591. [Google Scholar] [CrossRef]
- Sondi, I.; Salopek-Sondi, B. Silver Nanoparticles as Antimicrobial Agent: A Case Study on E. Coli as a Model for Gram-Negative Bacteria. J Colloid Interface Sci 2004, 275, 177–182. [Google Scholar]
- Daglia, M.; Cuzzoni, M.T.; Dacarro, C. Antibacterial Activity of Coffee. J Agric Food Chem 1994, 42, 2270–2272. [Google Scholar] [CrossRef]
- Nonthakaew, A.; Matan, N.; Aewsiri, T.; Matan, N. Caffeine in Foods and Its Antimicrobial Activity. Int Food Res J 2015, 22. [Google Scholar]
- Dondapati, V.; Bandaru, S.; K, S.; Bandaru, N.; Perala, B.; Voleti, A.; B, V.; Devara, M. Antibacterial Activity of Coffee Extract against Common Human Bacterial Pathogens in a Teaching Hospital of Semi Urban Setup. Natl J Physiol Pharm Pharmacol 2023, 1. [Google Scholar] [CrossRef]
- Rante, H.; Wulandari, R.; Evary, Y.M. ANTIBACTERIAL ACTIVITY OF ROBUSTA COFFEE (Coffea Robusta L.) PEEL EXTRACT AGAINST HUMAN PATHOGENIC BACTERIA. Journal of Experimental Biology and Agricultural Sciences 2021.
- Martínez-Tomé, M.; Jiménez-Monreal, A.; García-Jiménez, L.; Almela, L.; Garcia-Diz, L.; Mariscal-Arcas, M.; Murcia, M. Assessment of Antimicrobial Activity of Coffee Brewed in Three Different Ways from Different Origins. European Food Research and Technology 2011, 233, 497–505. [Google Scholar] [CrossRef]
- de Farias, Y.B.; Coutinho, A.K.; Assis, R.Q.; Rios, A. de O. Biodegradable Sodium Alginate Films Incorporated with Norbixin Salts. J Food Process Eng 2020, 43. [Google Scholar] [CrossRef]
- Almeida, A.A.P.; Naghetini, C.C.; Santos, V.R.; Antonio, A.G.; Farah, A.; Glória, M.B.A. Influence of Natural Coffee Compounds, Coffee Extracts and Increased Levels of Caffeine on the Inhibition of Streptococcus Mutans. Food Research International 2012, 49, 459–461. [Google Scholar] [CrossRef]
- Monente, C.; Bravo, J.; Vitas, A.I.; Arbillaga, L.; De Peña, M.P.; Cid, C. Coffee and Spent Coffee Extracts Protect against Cell Mutagens and Inhibit Growth of Food-Borne Pathogen Microorganisms. J Funct Foods 2015, 12, 365–374. [Google Scholar] [CrossRef]
- Duangjai, A.; Suphrom, N.; Wungrath, J.; Ontawong, A.; Nuengchamnong, N.; Yosboonruang, A. Comparison of Antioxidant, Antimicrobial Activities and Chemical Profiles of Three Coffee (Coffea Arabica L. ) Pulp Aqueous Extracts. Integr Med Res 2016, 5, 324–331. [Google Scholar]
- Gómez-Ordóñez, E.; Rupérez, P. FTIR-ATR Spectroscopy as a Tool for Polysaccharide Identification in Edible Brown and Red Seaweeds. Food Hydrocoll 2011, 25, 1514–1520. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am J Enol Vitic 1965, 16, 144–158. [Google Scholar] [CrossRef]
- BLOIS, M.S. Antioxidant Determinations by the Use of a Stable Free Radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Nardini, M.; Ghiselli, A. Determination of Free and Bound Phenolic Acids in Beer. Food Chem 2004, 84, 137–143. [Google Scholar] [CrossRef]
- Nowak, E.; Khachatryan, G.; Wisła-Świder, A. Structural Changes of Different Starches Illuminated with Linearly Polarised Visible Light. Food Chem 2021, 344, 128693. [Google Scholar] [CrossRef]
- Bello-Perez, L.A.; Paredes-López, O.; Roger, P.; Colonna, P. Amylopectin—Properties and Fine Structure. Food Chem 1996, 56, 171–176. [Google Scholar] [CrossRef]
- Hanselmann, R.; Burchard, W.; Ehrat, M.; Widmer, H.M. Structural Properties of Fractionated Starch Polymers and Their Dependence on the Dissolution Process. Macromolecules 1996, 29, 3277–3282. [Google Scholar] [CrossRef]
- ASTM D882-18 Standard Test Method for Tensile Properties of Thin Plastic Sheeting 2018, 08.01. ASTM D882-18 Standard Test Method for Tensile Properties of Thin Plastic Sheeting 2018, 08.01.
- Souza, V.; Fernando, A.; Pires, J.; Rodrigues, P.; Lopes, A.; Braz Fernandes, F. Physical Properties of Chitosan Films Incorporated with Natural Antioxidants. Ind Crops Prod 2017, 107. [Google Scholar] [CrossRef]
- Jamróz, E.; Janik, M.; Juszczak, L.; Kruk, T.; Kulawik, P.; Szuwarzyński, M.; Kawecka, A.; Khachatryan, K. Composite Biopolymer Films Based on a Polyelectrolyte Complex of Furcellaran and Chitosan. Carbohydr Polym 2021, 274, 118627. [Google Scholar] [CrossRef]
- Chavoshizadeh, S.; Pirsa, S.; Mohtarami, F. Conducting/Smart Color Film Based on Wheat Gluten/Chlorophyll/Polypyrrole Nanocomposite. Food Packag Shelf Life 2020, 24, 100501. [Google Scholar] [CrossRef]
- Sularz, O.; Koronowicz, A.; Boycott, C.; Smoleń, S.; Stefanska, B. Molecular Effects of Iodine-Biofortified Lettuce in Human Gastrointestinal Cancer Cells. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Krausova, G.; Hyrslova, I.; Hynstova, I. In Vitro Evaluation of Adhesion Capacity, Hydrophobicity, and Auto-Aggregation of Newly Isolated Potential Probiotic Strains. Fermentation 2019, 5. [Google Scholar] [CrossRef]
- Paudel, K.R.; Karki, R.; Kim, D.-W. Cepharanthine Inhibits in Vitro VSMC Proliferation and Migration and Vascular Inflammatory Responses Mediated by RAW264. 7. Toxicology in Vitro 2016, 34, 16–25. [Google Scholar] [CrossRef]
| Parameter | Green coffee | Light roasted | Medium roasted | Dark roasted |
| TPC1 | 113.36d ± 0.99 | 134.86c ± 2.03 | 125. 83b ± 1.65 | 102.89a ± 1.01 |
| AA2 | 0.73d ± 0.01 | 0.88c ± 0.03 | 0.82b ± 0.02 | 0.64a ± 0.01 |
| Chlorogenic acids | ||||
| 5-CQA3 | 2052.66d ± 45.73 | 1350.17c ± 21.92 | 1005.26b ± 5.38 | 232.04a ± 0.000 |
| 4-CQA4 | 340.42d ± 6.72 | 303.83c ± 10.92 | 231.16b ± 2.48 | 94.94a ± 1.44 |
| 3-CQA5 | 272.10d ± 6.38 | 263.29c ± 0.11 | 215.97b ± 2.54 | 61.67a ± 1.28 |
| 3,5-diCQA6 | 343.63d ± 4.55 | 69.93c ± 0.63 | 45.05b ± 1.62 | 6.45a ± 0.27 |
| 3,4-diCQA7 | 113.32d ± 0.39 | 60.29c ± 0.13 | 38.34b ± 0.21 | 3.72a ± 0.32 |
| 4.5-diCQA8 | 86.31d ± 1.70 | 53.46c ± 0.36 | 35.44b ± 0.22 | 6.24a ± 0.21 |
| Total CGAs | 3208.44 | 2100.97 | 1571.22 | 405.06 |
| FPAs3 | ||||
| Caffeic acid | 1594.74d ± 13.39 | 1251.51c ± 5.87 | 957.79b ± 18.37 | 294.20a ± 0.53 |
| p-Coumaric acid | 25.33c ± 0.85 | 17.63b ± 0.48 | 15.86b ± 0.42 | 9.19a ± 0.40 |
| Ferulic acid | 147.75d ± 1.69 | 120.00c ± 0.42 | 98.62b ± 0.33 | 45.97a ± 0.25 |
| Sample | Mw × 106 [g/mol] | Rg [nm] | ||
| Peak I | Peak II | Peak I | Peak II | |
| A | 0.112±0.15a | 0.077±0.15 | 95.3±1.4a | 59.8±1.1 |
| AL | 0.308±0.16b | n.m. | 46.9±1.4b | n.m. |
| ALD | 5.503±0.14c | n.m. | 37.3±1.3c | n.m. |
| ALM | 5.184±0.16d | n.m. | 35.5±1.4c,d | n.m. |
| ALL | 4.227±0.17e | n.m. | 33.2±1.4d | n.m. |
| ALG | 1.903±0.11f | n.m. | 34.9±1.4c,d | n.m. |
| Sample | A | AL | ALG | ALL | ALM | ALD |
| Endothermic fusion (melting point) | ||||||
| Tm (°C) | 186.6c ± 2.9 | 166.9a ± 2.1 | 176.7b ± 1.8 | 179.5b ± 3.2 | 183.1b,c ± 4.4 | 180.1b,c ± 6.0 |
| ΔHm (J g-1) | 76.96a ± 2.49 | 174.53c ± 4.77 | 89.86b ± 0.87 | 95.85b ± 2.01 | 93.43b ± 6.85 | 81.45a ± 6.02 |
| Exothermic fusion (decarboxylation) | ||||||
| TD (°C) | 232.4a ± 1.5 | 233.9a ± 0.8 | 233.3a ± 1.0 | 233.8a ± 1.7 | 232.5a ± 1.0 | 232.5a ± 0.8 |
| ΔHD (J g-1) | 170.6b ± 9.46 | 350.63d ± 6.31 | 235.53d ± 3.45 | 144.1a ± 19.87 | 224.10c ± 11.69 | 145.10a ± 11.48 |
| Mechanical properties | ||||||
| TS [MPa] | - | 168.9b ± 9.0 | 108.5a ± 5.9 | 112.9a ± 9.8 | 172.3b ± 13.6 | 122.9a ± 13.0 |
| MBL [N] | - | 19.08c ± 1.01 | 15.40b ± 0.84 | 13.78a ± 1.33 | 21.21d ± 0.73 | 16.47b ± 1.74 |
| ME [MPa] | - | 6709.9c ± 358.8 | 3693.2a ± 403.8 | 5357.7b± 190.4 | 5749.1b ± 210.3 | 5698.0b ± 477.7 |
| WCA [°] | - | 30.20a ± 2.62 | 32.46b ± 2.62 | 36.73c ± 1.17 | 38.78d ± 2.03 | 41.30e ± 1.84 |
| Water content [%] | - | 17.31c ±0,51 | 13.81b ±0.69 | 16.06c ± 0.56 | 11.29a ± 0.27 | 13.31a ± 0.68 |
| Sample | AL | ALG | ALL | ALM | ALD |
| L* | 92.69e ± 1.06 | 83.88d ± 0.73 | 77.12c ± 0.72 | 69.38b ± 0.89 | 64.21a ± 0.67 |
| a* | – 0.86a ± 0.03 | – 0.20b ± 0.07 | 3.39c ± 0.31 | 7.17d ± 0.48 | 8.74e ± 0.46 |
| b* | 10.68a ± 0.32 | 18.26b ± 0.52 | 28.56c ± 2.02 | 36.34d ± 0.78 | 36.26d ± 0.38 |
| WI | 87.03 | 75.63 | 63.24 | 51.95 | 48.31 |
| YI | 16.46 | 31.13 | 52.92 | 74.82 | 80.67 |
| ΔE | - | 11.66 | 24.09 | 35.58 | 28.11 |
| OP | 15.66a ± 0.04 | 17.47b ± 0.07 | 19.38c ± 0.05 | 24.9d ± 0.32 | 24.23d ± 0.04 |
| Microorganism | Zone of growth inhibition of tested microorganisms [mm] | ||||
| AL | ALG | ALL | ALM | ALD | |
| B. thuringiensis | 0a | 0a | 0a | 0a | 0a |
| B. cereus | 0a | 0a | 0a | 0a | 0a |
| B. megaterium | 0a | 0a | 7b | 7b | 7b |
| S. aureus | 0a | 22b | 0a | 0a | 0a |
| S. equorum | 0a | 16b | 14b | 13b | 14b |
| S. xylosus | 0a | 8b | 11b | 10b | 11b |
| M. luteus | 0a | 0a | 0a | 0a | 0a |
| E. faecalis | 0a | 8b | 8b | 8b | 10b |
| E. coli | 0a | 10b | 0a | 0a | 0a |
| S. typhimurium | 0a | 0a | 0a | 7b | 10b |
| S. enteritidis | 0a | 0a | 0a | 0a | 0a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




