Submitted:
28 September 2024
Posted:
29 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
- Screening of Phosphatidyl Conjugated Compounds in Different Cancer Cell Lines
- Metastatic Cell Sensitivity to diC6-THIO Correlates with Increased Telomerase Activity
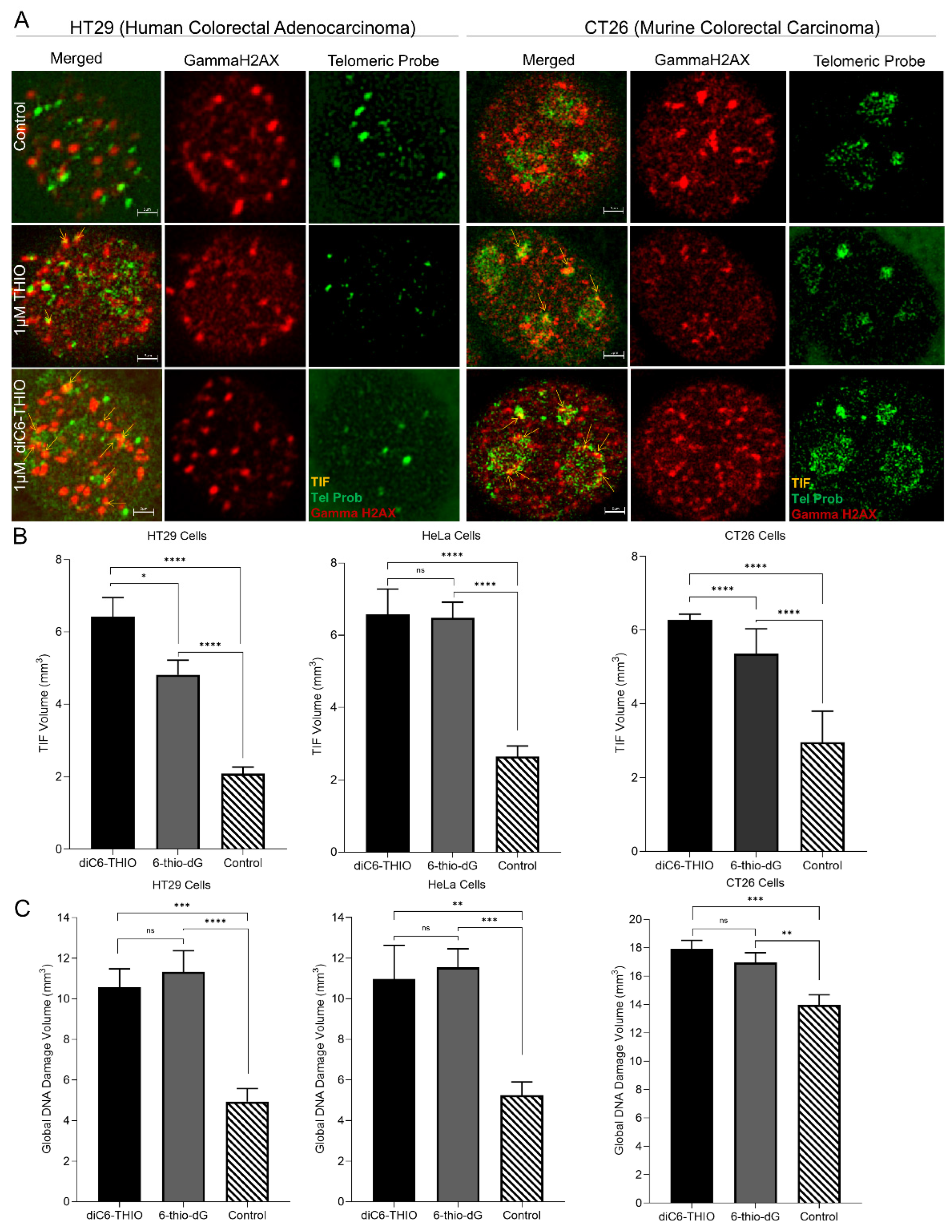
- diC6-THIO Treatment Induces Telomere Dysfunction Induced Foci (TIF) Formation
- diC6-THIO-Induced Dissociation of CT26 Spheroids
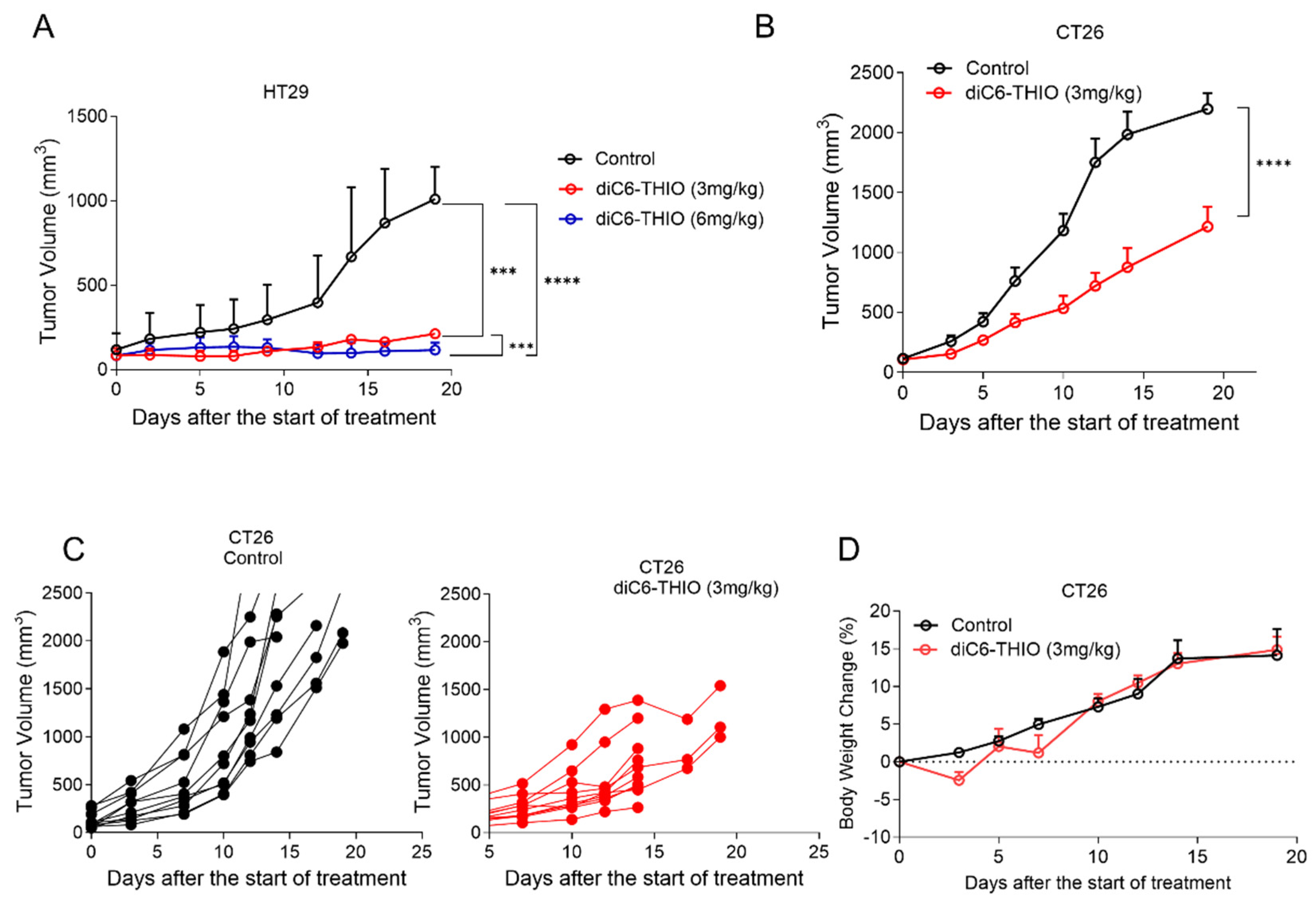
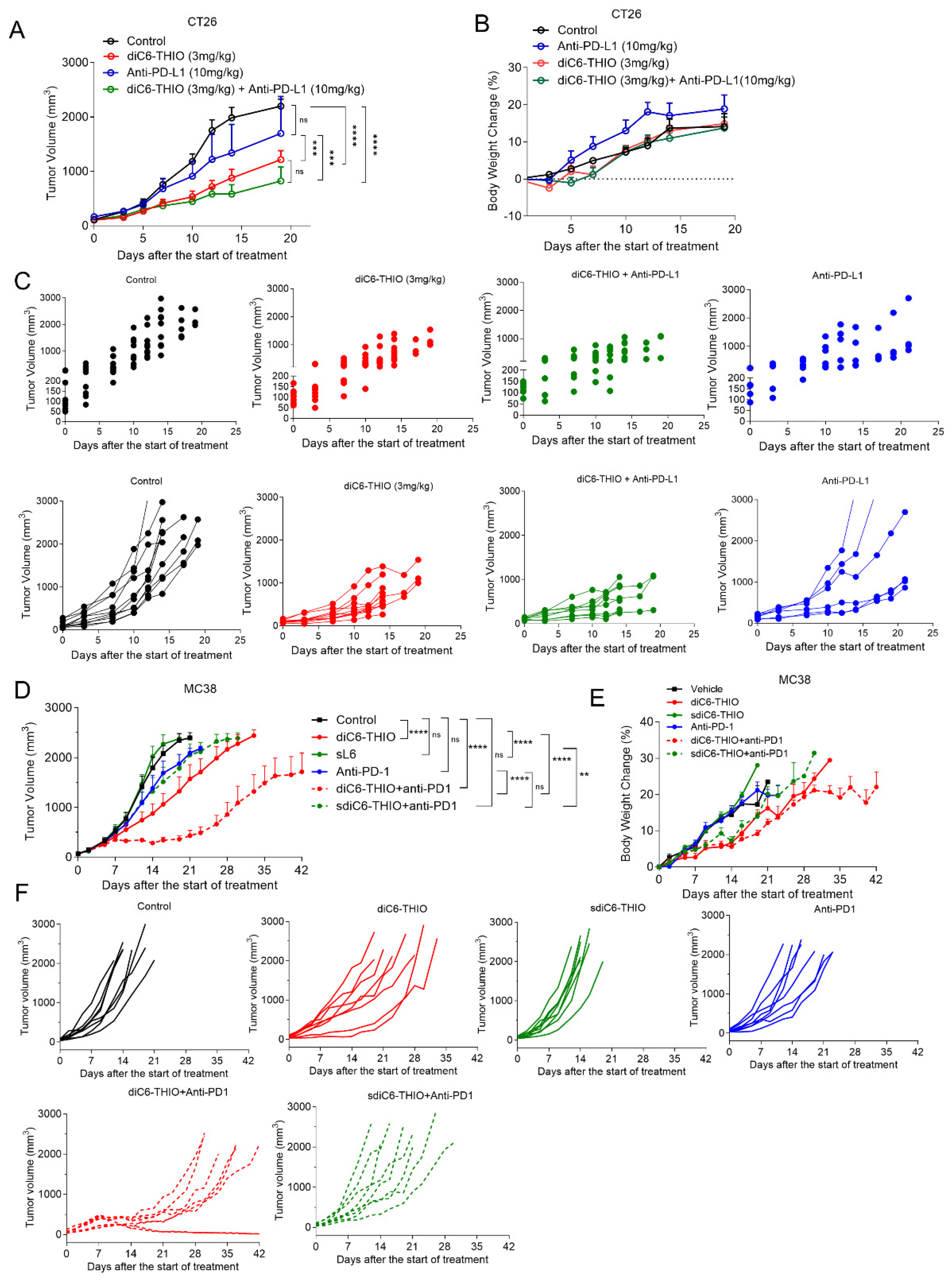
- diC6-THIO Reduces Tumor Growth in HT29-Derived Xenograft and CT26 Syngeneic Mouse Models
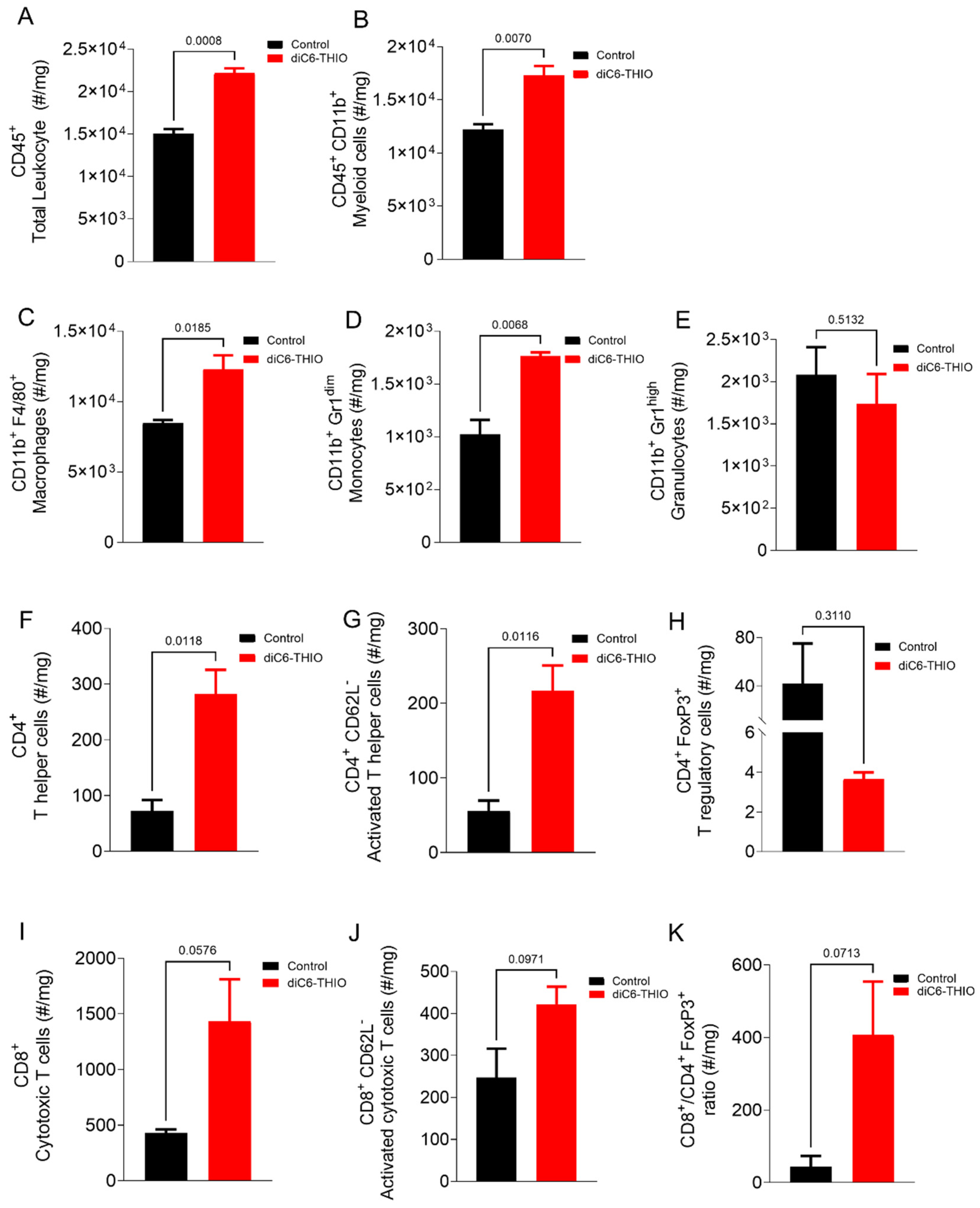
- diC6-THIO Treatment Enhances Activated CD4+ and CD8+ T Cells and Decreases T Regulatory Cells in the Tumor Microenvironment
- diC6-THIO Treatment Reduces Tumor Growth Compared to Control and Anti-PD-L1 Therapy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L., et al., Cancer statistics, 2023. CA Cancer J Clin, 2023. 73(1): p. 17-48.
- Kuipers, E.J., et al., Colorectal cancer. Nat Rev Dis Primers, 2015. 1: p. 15065.
- Johdi, N.A. and N.F. Sukor, Colorectal Cancer Immunotherapy: Options and Strategies. Front Immunol, 2020. 11: p. 1624. [CrossRef]
- Marcus, L., et al., FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors. Clin Cancer Res, 2019. 25(13): p. 3753-3758. [CrossRef]
- Jacome, A.A. and C. Eng, Role of immune checkpoint inhibitors in the treatment of colorectal cancer: focus on nivolumab. Expert Opin Biol Ther, 2019. 19(12): p. 1247-1263. [CrossRef]
- Li, J. and X. Xu, Immune Checkpoint Inhibitor-Based Combination Therapy for Colorectal Cancer: An Overview. Int J Gen Med, 2023. 16: p. 1527-1540. [CrossRef]
- Giang, I., E.L. Boland, and G.M. Poon, Prodrug applications for targeted cancer therapy. AAPS J, 2014. 16(5): p. 899-913. [CrossRef]
- Walther, R., J. Rautio, and A.N. Zelikin, Prodrugs in medicinal chemistry and enzyme prodrug therapies. Adv Drug Deliv Rev, 2017. 118: p. 65-77. [CrossRef]
- Fattahi, N., et al., Emerging insights on drug delivery by fatty acid mediated synthesis of lipophilic prodrugs as novel nanomedicines. J Control Release, 2020. 326: p. 556-598. [CrossRef]
- Dahan, A., et al., The prospects of lipidic prodrugs: an old approach with an emerging future. Future Med Chem, 2019. 11(19): p. 2563-2571. [CrossRef]
- Mura, S., et al., Lipid prodrug nanocarriers in cancer therapy. J Control Release, 2015. 208: p. 25-41. [CrossRef]
- Sreekanth, V. and A. Bajaj, Recent Advances in Engineering of Lipid Drug Conjugates for Cancer Therapy. ACS Biomater Sci Eng, 2019. 5(9): p. 4148-4166. [CrossRef]
- Irby, D., C. Du, and F. Li, Lipid-Drug Conjugate for Enhancing Drug Delivery. Mol Pharm, 2017. 14(5): p. 1325-1338. [CrossRef]
- Huang, L., et al., Engineering of small-molecule lipidic prodrugs as novel nanomedicines for enhanced drug delivery. J Nanobiotechnology, 2022. 20(1): p. 49. [CrossRef]
- Griffith, J.D., et al., Mammalian telomeres end in a large duplex loop. Cell, 1999. 97(4): p. 503-14. [CrossRef]
- Blackburn, E.H., Structure and function of telomeres. Nature, 1991. 350(6319): p. 569-73. [CrossRef]
- Shay, J.W. and W.E. Wright, Telomerase: a target for cancer therapeutics. Cancer Cell, 2002. 2(4): p. 257-65. [CrossRef]
- Shay, J.W. and S. Bacchetti, A survey of telomerase activity in human cancer. Eur J Cancer, 1997. 33(5): p. 787-91. [CrossRef]
- Mender, I., S. Gryaznov, and J.W. Shay, A novel telomerase substrate precursor rapidly induces telomere dysfunction in telomerase positive cancer cells but not telomerase silent normal cells. Oncoscience, 2015. 2(8): p. 693-5. [CrossRef]
- Mender, I., et al., Induction of telomere dysfunction mediated by the telomerase substrate precursor 6-thio-2'-deoxyguanosine. Cancer Discov, 2015. 5(1): p. 82-95. [CrossRef]
- Gilles, J.F., et al., DiAna, an ImageJ tool for object-based 3D co-localization and distance analysis. Methods, 2017. 115: p. 55-64. [CrossRef]
- Ludlow, A.T., et al., Quantitative telomerase enzyme activity determination using droplet digital PCR with single cell resolution. Nucleic Acids Research, 2014. 42(13): p. e104-e104. [CrossRef]
- Zhang, J., et al., Enrichment and characterization of cancer stem-like cells in ultra-low concentration of serum and non-adhesive culture system. Am J Transl Res, 2018. 10(5): p. 1552-1561.
- Zhong, Y., et al., Spheres derived from the human SK-RC-42 renal cell carcinoma cell line are enriched in cancer stem cells. Cancer Lett, 2010. 299(2): p. 150-60. [CrossRef]
- Godoy, P., et al., Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells and their use in investigating mechanisms of hepatotoxicity, cell signaling and ADME. Archives of Toxicology, 2013. 87(8): p. 1315-1530. [CrossRef]
- Boudreau, N., Z. Werb, and M.J. Bissell, Suppression of apoptosis by basement membrane requires three-dimensional tissue organization and withdrawal from the cell cycle. Proc Natl Acad Sci U S A, 1996. 93(8): p. 3509-13. [CrossRef]
- Mender, I., et al., Activating an Adaptive Immune Response with a Telomerase-Mediated Telomere Targeting Therapeutic in Hepatocellular Carcinoma. Mol Cancer Ther, 2023. 22(6): p. 737-750. [CrossRef]
- Delahousse, J., C. Skarbek, and A. Paci, Prodrugs as drug delivery system in oncology. Cancer Chemother Pharmacol, 2019. 84(5): p. 937-958. [CrossRef]
- Wang, H., et al., Doxorubicin conjugated phospholipid prodrugs as smart nanomedicine platforms for cancer therapy. J Mater Chem B, 2015. 3(16): p. 3297-3305. [CrossRef]
- Bui, D.T., et al., Multifunctional squalene-based prodrug nanoparticles for targeted cancer therapy. Chem Commun (Camb), 2014. 50(40): p. 5336-8. [CrossRef]
- Emamzadeh, M., et al., Dual controlled delivery of squalenoyl-gemcitabine and paclitaxel using thermo-responsive polymeric micelles for pancreatic cancer. J Mater Chem B, 2018. 6(15): p. 2230-2239. [CrossRef]
- Mougin, J., et al., Stacking as a Key Property for Creating Nanoparticles with Tunable Shape: The Case of Squalenoyl-Doxorubicin. ACS Nano, 2019. 13(11): p. 12870-12879. [CrossRef]
- Gobeaux, F., et al., Albumin-driven disassembly of lipidic nanoparticles: the specific case of the squalene-adenosine nanodrug. Nanoscale, 2020. 12(4): p. 2793-2809. [CrossRef]
- Sauraj, et al., Lipophilic 5-fluorouracil prodrug encapsulated xylan-stearic acid conjugates nanoparticles for colon cancer therapy. Int J Biol Macromol, 2019. 128: p. 204-213.
- Wu, L., et al., Self-Assembled Gemcitabine Prodrug Nanoparticles Show Enhanced Efficacy against Patient-Derived Pancreatic Ductal Adenocarcinoma. ACS Appl Mater Interfaces, 2020. 12(3): p. 3327-3340. [CrossRef]
- Li, Q., et al., A syntaxin 1, Galpha(o), and N-type calcium channel complex at a presynaptic nerve terminal: analysis by quantitative immunocolocalization. J Neurosci, 2004. 24(16): p. 4070-81. [CrossRef]
- Manders, E.M., et al., Dynamics of three-dimensional replication patterns during the S-phase, analysed by double labelling of DNA and confocal microscopy. J Cell Sci, 1992. 103 ( Pt 3): p. 857-62. [CrossRef]
- Lachmanovich, E., et al., Co-localization analysis of complex formation among membrane proteins by computerized fluorescence microscopy: application to immunofluorescence co-patching studies. J Microsc, 2003. 212(Pt 2): p. 122-31. [CrossRef]
- Obara, B., et al., A novel method for quantified, superresolved, three-dimensional colocalisation of isotropic, fluorescent particles. Histochem Cell Biol, 2013. 139(3): p. 391-402. [CrossRef]
- Bolte, S. and F.P. Cordelieres, A guided tour into subcellular colocalization analysis in light microscopy. J Microsc, 2006. 224(Pt 3): p. 213-32. [CrossRef]
- Jaskolski, F., C. Mulle, and O.J. Manzoni, An automated method to quantify and visualize colocalized fluorescent signals. J Neurosci Methods, 2005. 146(1): p. 42-9. [CrossRef]
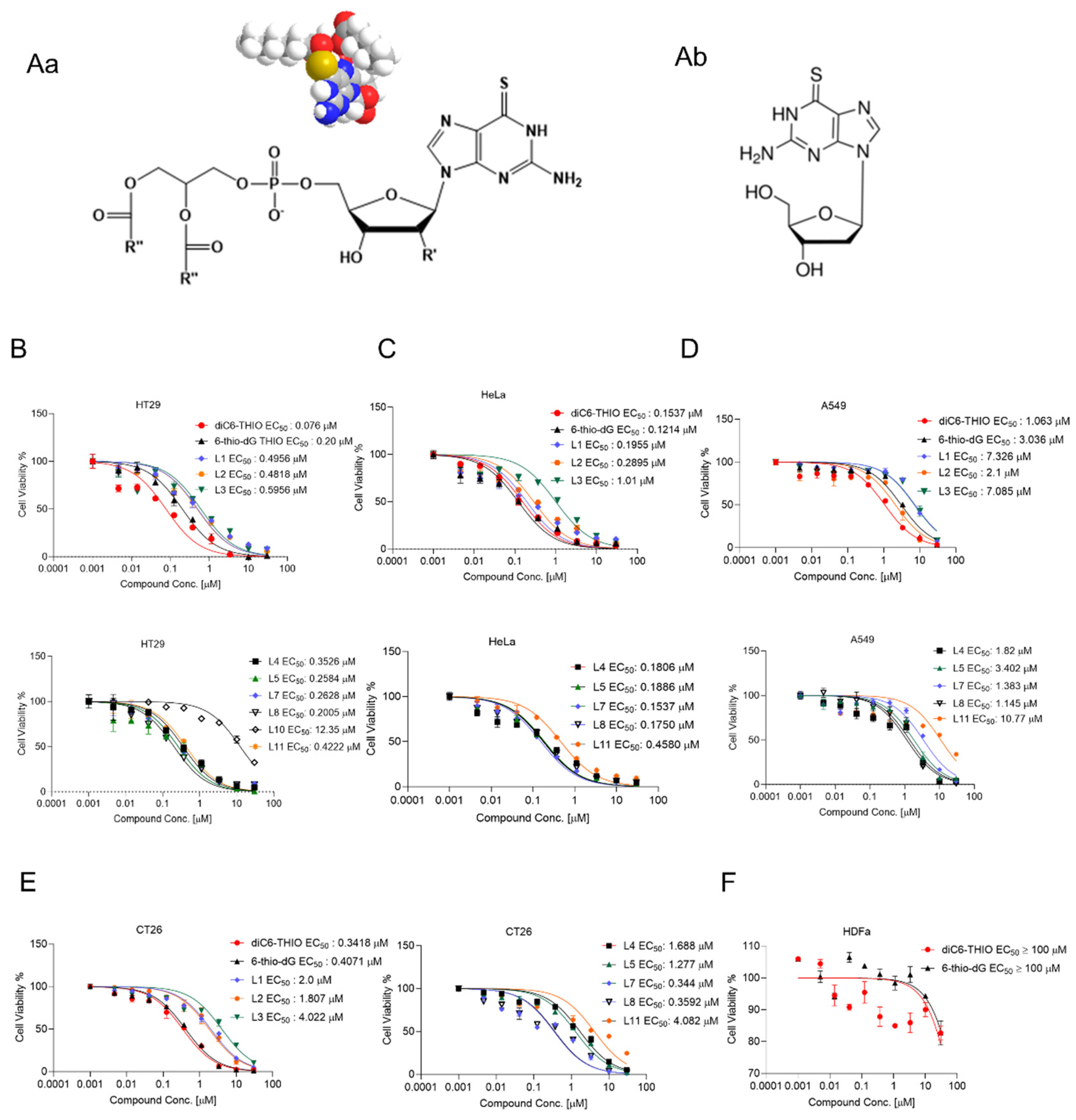
| EC50 (µM) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Compounds | HT29 | HeLa | A549 | CT26 | MC38 | LLC | HDFa | U87 |
| 6-thio-dG | 0.2 | 0.1214 | 3.036 | 0.4071 | 1.507 | 0.172 | >100 | 0.8985 |
| L1 | 0.4956 | 0.1955 | 7.326 | 2 | - | - | - | - |
| L2 | 0.4818 | 0.2895 | 2.1 | 1.807 | - | - | - | - |
| L3 | 0.5956 | 1.01 | 7.085 | 4.022 | - | - | - | - |
| L4 | 0.3526 | 0.186 | 1.82 | 1.688 | - | - | - | - |
| L5 | 0.2584 | 0.1886 | 3.402 | 1.277 | - | - | - | - |
| L6 (diC6-THIO) | 0.076 | 0.1537 | 1.063 | 0.3418 | 3.527 | 0.3418 | >100 | 0.7878 |
| sdiC6-THIO | - | - | - | - | > 50 | 23.94 | - | - |
| L7 | 0.2628 | 0.1537 | 1.383 | 0.344 | - | - | - | - |
| L8 | 0.2 | 0.175 | 1.145 | 0.3592 | - | - | - | - |
| L10 | 12.35 | - | - | 43.07 | - | - | - | - |
| L11 | 0.4222 | 0.458 | 10.77 | 4.02 | - | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




