Submitted:
26 September 2024
Posted:
27 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Capsid Variant Development
2.2. Recombinant AAV Preparations
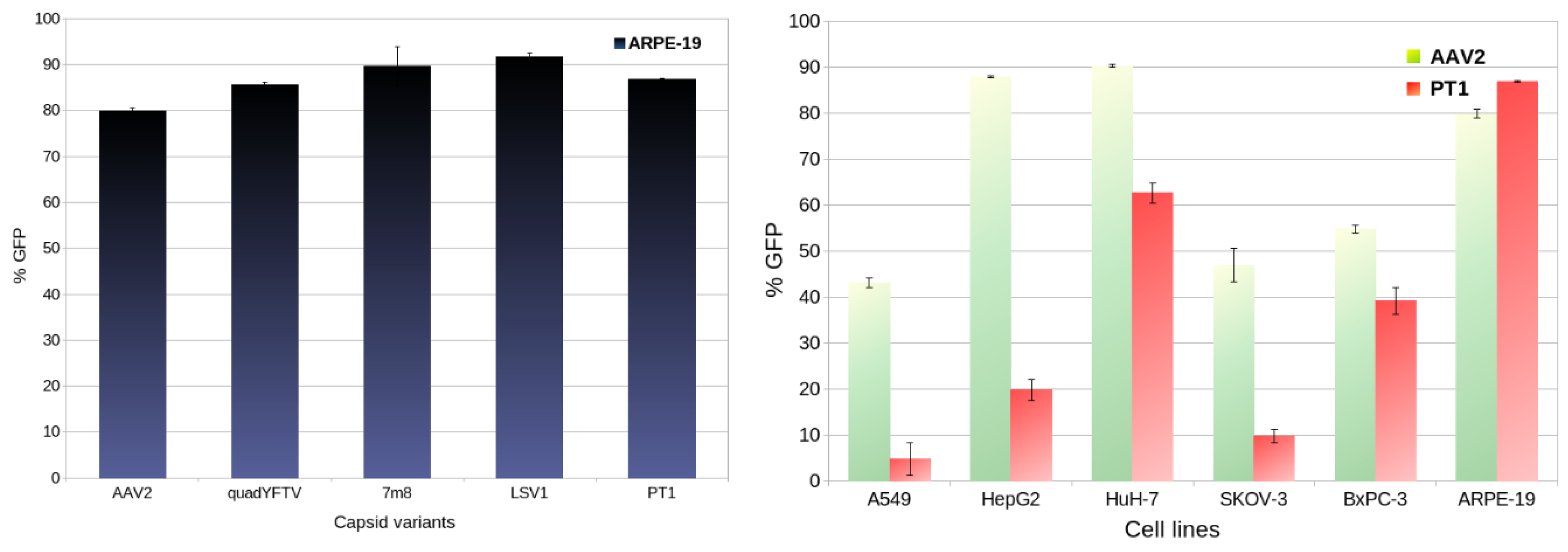
2.3. In Vitro Transduction
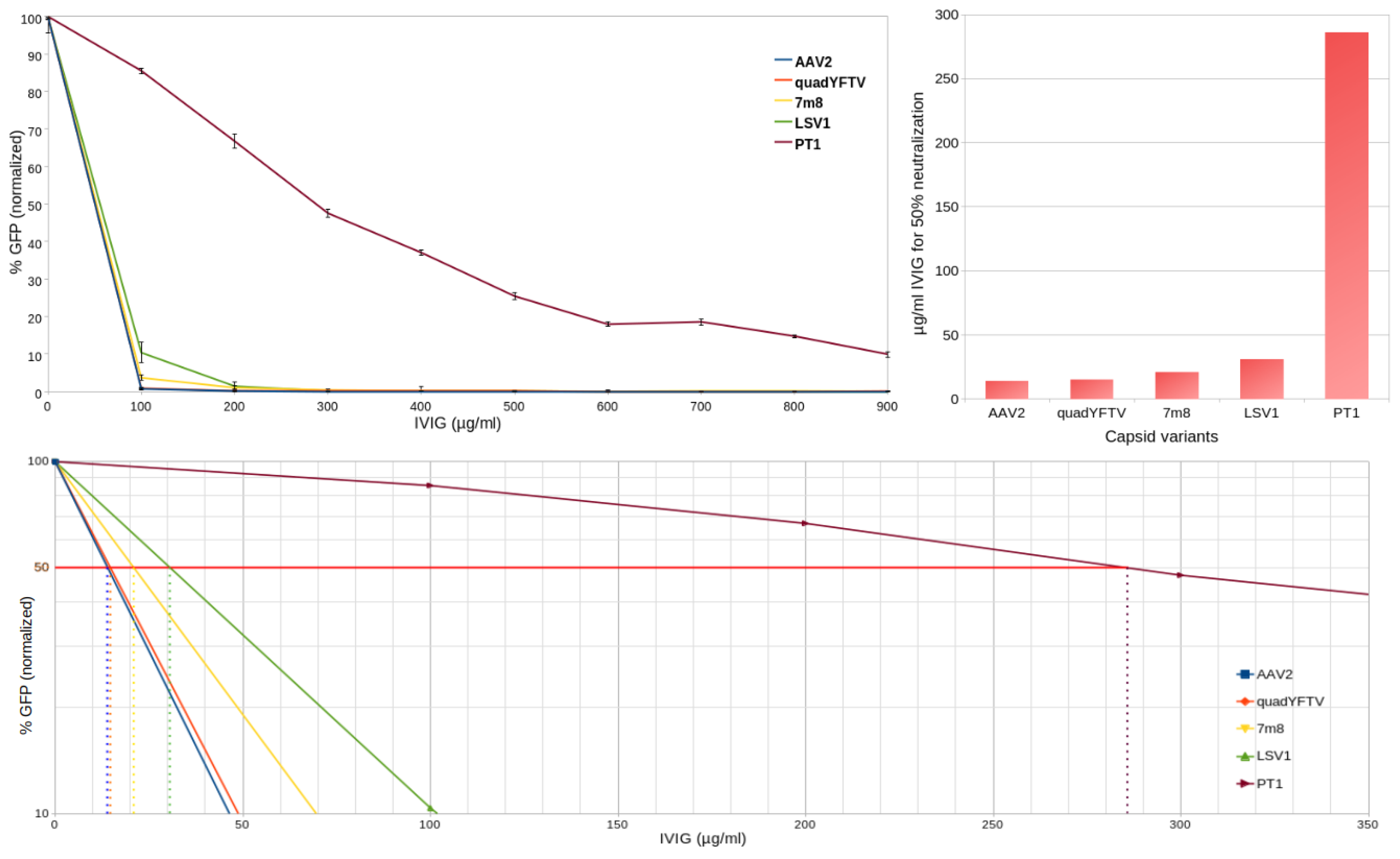
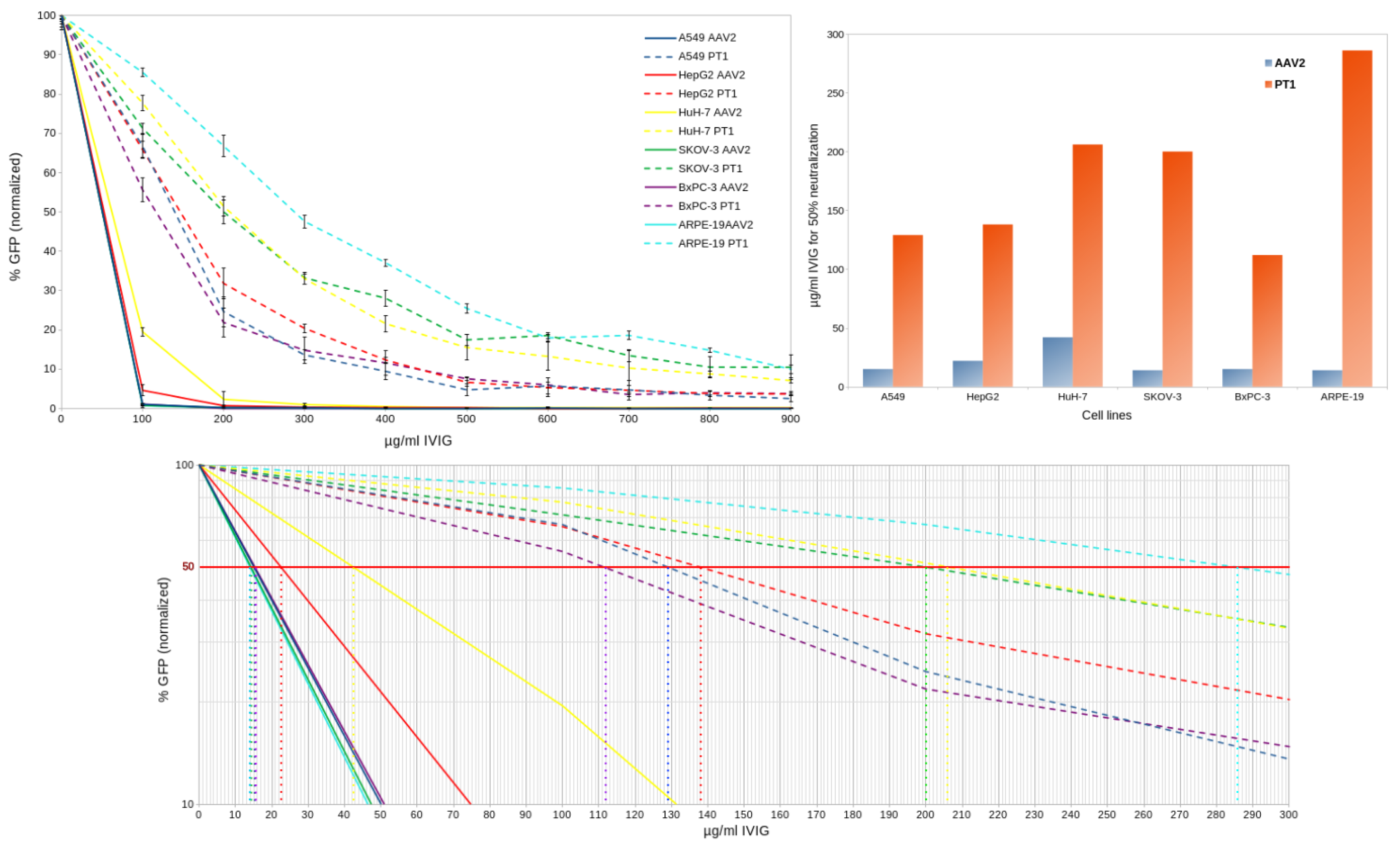
2.4. Immune Evasion
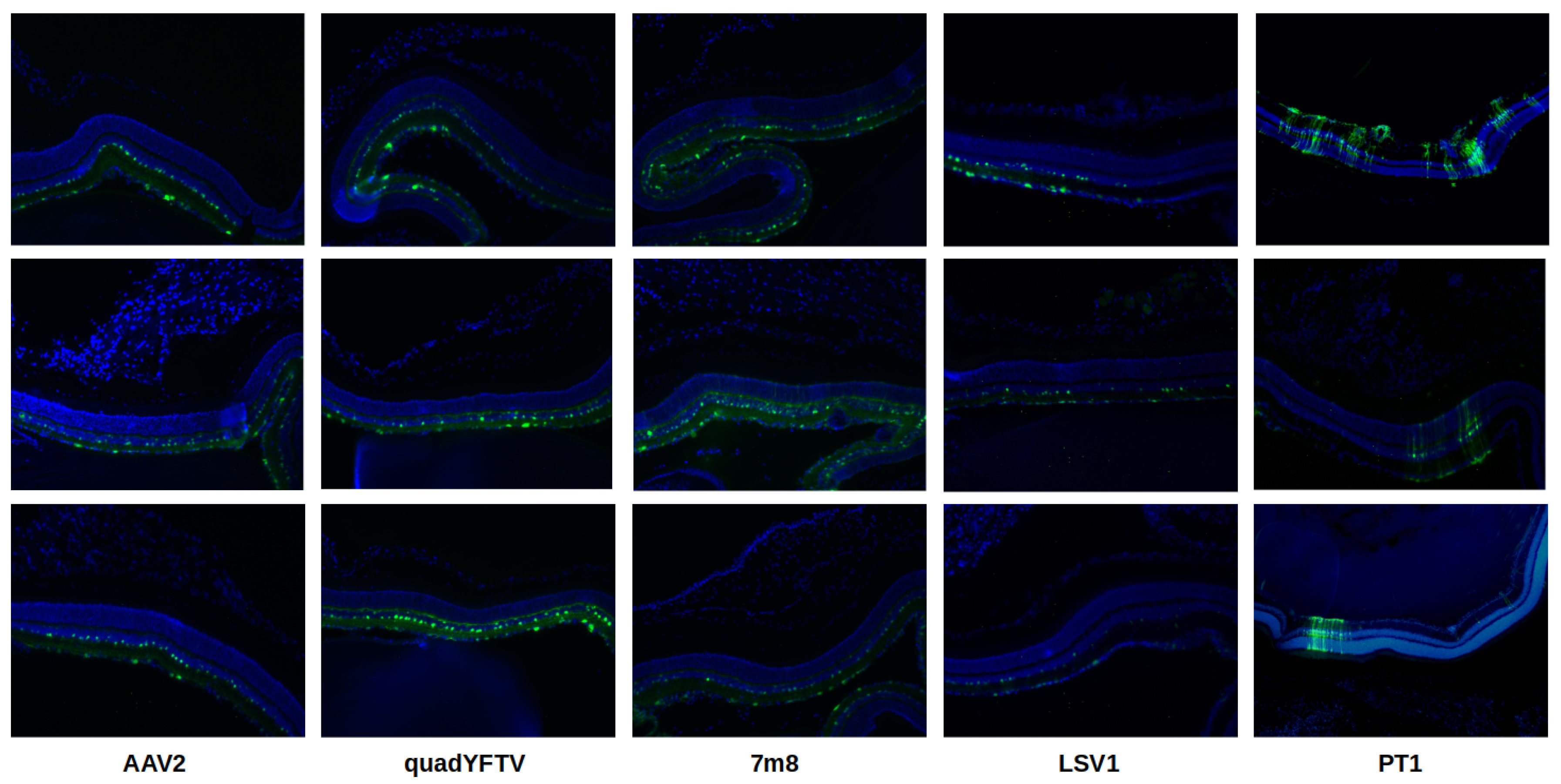
2.5. In Vivo Evaluation
3. Discussion
4. Materials and Methods
4.1. AAV Capsid Libraries
4.2. AAV Preparations
4.3. AAV Quantification
4.4. In Vitro Transduction and Neutralization Assay
4.5. Animals
4.6. Intravitreal rAAV Injections
4.7. Retinal Harvesting
4.8. Retinal Flat Mounts
4.9. Retinal Cryosections
4.10. Data Visualization
4.11. Sequence Deposition
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| VR | Variable region |
| GFP | Green fluorescent protein |
| vg | viral genome |
| rAAV | Recombinant AAV |
| NHP | Non-human primate |
| EDTA | Ethylenediaminetetraacetic acid |
| SDS | Sodium dodecyl sulfate |
| qPCR | Quantitative polymerase chain reaction |
| MOI | Multiplicity of infection |
| PBS | Phosphate-buffered saline |
| CCD | Charge-coupled device |
| PFA | Paraformaldehyde |
| DAPI | 4’,6-diamidino-2-phenylindole |
References
- Wang, J.H.; Gessler, D.J.; Zhan, W.; Gallagher, T.L.; Gao, G. Adeno-associated virus as a delivery vector for gene therapy of human diseases. Signal Transduction and Targeted Therapy 2024, 9, 78. [Google Scholar] [CrossRef]
- Liu, F.; Li, R.; Zhu, Z.; Yang, Y.; Lu, F. Current developments of gene therapy in human diseases. MedComm 2024, 5, e645. [Google Scholar] [CrossRef]
- Gao, J.; Hussain, R.M.; Weng, C.Y. Voretigene Neparvovec in Retinal Diseases: A Review of the Current Clinical Evidence. Clinical Ophthalmology 2020, Volume 14, 3855–3869. [Google Scholar] [CrossRef]
- Kotterman, M.A.; Yin, L.; Strazzeri, J.M.; Flannery, J.G.; Merigan, W.H.; Schaffer, D.V. Antibody neutralization poses a barrier to intravitreal adeno-associated viral vector gene delivery to non-human primates. Gene Therapy 2015, 22, 116–126. [Google Scholar] [CrossRef]
- Zolotukhin, S.; Vandenberghe, L. AAV capsid design: A Goldilocks challenge. Trends in Molecular Medicine 2022, 28, 183–193. [Google Scholar] [CrossRef]
- Srivastava, A. Rationale and strategies for the development of safe and effective optimized AAV vectors for human gene therapy. Molecular Therapy - Nucleic Acids 2023, 32, 949–959. [Google Scholar] [CrossRef]
- Dalkara, D.; Byrne, L.C.; Klimczak, R.R.; Visel, M.; Yin, L.; Merigan, W.H.; Flannery, J.G.; Schaffer, D.V. In Vivo–Directed Evolution of a New Adeno-Associated Virus for Therapeutic Outer Retinal Gene Delivery from the Vitreous. Science Translational Medicine 2013, 5. [Google Scholar] [CrossRef]
- Pavlou, M.; Schön, C.; Occelli, L.M.; Rossi, A.; Meumann, N.; Boyd, R.F.; Bartoe, J.T.; Siedlecki, J.; Gerhardt, M.J.; Babutzka, S.; Bogedein, J.; Wagner, J.E.; Priglinger, S.G.; Biel, M.; Petersen-Jones, S.M.; Büning, H.; Michalakis, S. Novel AAV capsids for intravitreal gene therapy of photoreceptor disorders. EMBO Molecular Medicine 2021, 13, e13392. [Google Scholar] [CrossRef]
- Kellish, P.C.; Marsic, D.; Crosson, S.M.; Choudhury, S.; Scalabrino, M.L.; Strang, C.E.; Hill, J.; McCullough, K.T.; Peterson, J.J.; Fajardo, D.; Gupte, S.; Makal, V.; Kondratov, O.; Kondratova, L.; Iyer, S.; Witherspoon, C.D.; Gamlin, P.D.; Zolotukhin, S.; Boye, S.L.; Boye, S.E. Intravitreal injection of a rationally designed AAV capsid library in non-human primate identifies variants with enhanced retinal transduction and neutralizing antibody evasion. Molecular Therapy 2023, 31, 3441–3456. [Google Scholar] [CrossRef]
- Kay, C.N.; Ryals, R.C.; Aslanidi, G.V.; Min, S.H.; Ruan, Q.; Sun, J.; Dyka, F.M.; Kasuga, D.; Ayala, A.E.; Van Vliet, K.; Agbandje-McKenna, M.; Hauswirth, W.W.; Boye, S.L.; Boye, S.E. Targeting Photoreceptors via Intravitreal Delivery Using Novel, Capsid-Mutated AAV Vectors. PLoS ONE 2013, 8, e62097. [Google Scholar] [CrossRef]
- Marsic, D.; Govindasamy, L.; Currlin, S.; Markusic, D.M.; Tseng, Y.S.; Herzog, R.W.; Agbandje-McKenna, M.; Zolotukhin, S. Vector Design Tour de Force: Integrating Combinatorial and Rational Approaches to Derive Novel Adeno-associated Virus Variants. Molecular Therapy 2014, 22, 1900–1909. [Google Scholar] [CrossRef]
- Biswas, M.; Marsic, D.; Li, N.; Zou, C.; Gonzalez-Aseguinolaza, G.; Zolotukhin, I.; Kumar, S.R.; Rana, J.; Butterfield, J.S.; Kondratov, O.; De Jong, Y.P.; Herzog, R.W.; Zolotukhin, S. Engineering and In Vitro Selection of a Novel AAV3B Variant with High Hepatocyte Tropism and Reduced Seroreactivity. Molecular Therapy - Methods & Clinical Development 2020, 19, 347–361. [Google Scholar] [CrossRef]
- Baker, C.K.; Bennett, A.; Belbellaa, B.; Nieves, J.; Tai, A.; NcKenna, R.; Cepeda, D.; Agbandje-McKenna, M.; Riley, B.E. Novel Capsid LSV1 Has a Unique 3D Structure at the Loop Substitution Area - Confers Superior Retinal Transduction from Intravitreal Injection. 2022 ASGCT Annual Meeting Abstracts. Molecular Therapy, 2022, Vol. 30, p. 575.
- Kondratova, L.; Kondratov, O.; Ragheb, R.; Zolotukhin, S. Removal of Endotoxin from rAAV Samples Using a Simple Detergent-Based Protocol. Molecular Therapy - Methods & Clinical Development 2019, 15, 112–119. [Google Scholar] [CrossRef]
- Dunn, K.; Aotaki-Keen, A.; Putkey, F.; Hjelmeland, L. ARPE-19, A Human Retinal Pigment Epithelial Cell Line with Differentiated Properties. Experimental Eye Research 1996, 62, 155–170. [Google Scholar] [CrossRef]
- Chiorini, J.A.; Kim, F.; Yang, L.; Kotin, R.M. Cloning and Characterization of Adeno-Associated Virus Type 5. Journal of Virology 1999, 73, 1309–1319. [Google Scholar] [CrossRef]
- Mietzsch, M.; Jose, A.; Chipman, P.; Bhattacharya, N.; Daneshparvar, N.; McKenna, R.; Agbandje-McKenna, M. Completion of the AAV Structural Atlas: Serotype Capsid Structures Reveals Clade-Specific Features. Viruses 2021, 13, 101. [Google Scholar] [CrossRef]
- Bennett, A.; Patel, S.; Mietzsch, M.; Jose, A.; Lins-Austin, B.; Yu, J.C.; Bothner, B.; McKenna, R.; Agbandje-McKenna, M. Thermal Stability as a Determinant of AAV Serotype Identity. Molecular Therapy - Methods & Clinical Development 2017, 6, 171–182. [Google Scholar] [CrossRef]
- Chhabra, A.; Bashirians, G.; Petropoulos, C.J.; Wrin, T.; Paliwal, Y.; Henstock, P.V.; Somanathan, S.; Da Fonseca Pereira, C.; Winburn, I.; Rasko, J.E. Global seroprevalence of neutralizing antibodies against adeno-associated virus serotypes used for human gene therapies. Molecular Therapy - Methods & Clinical Development 2024, 32, 101273. [Google Scholar] [CrossRef]
- Gurda, B.L.; DiMattia, M.A.; Miller, E.B.; Bennett, A.; McKenna, R.; Weichert, W.S.; Nelson, C.D.; Chen, W.j.; Muzyczka, N.; Olson, N.H.; Sinkovits, R.S.; Chiorini, J.A.; Zolotutkhin, S.; Kozyreva, O.G.; Samulski, R.J.; Baker, T.S.; Parrish, C.R.; Agbandje-McKenna, M. Capsid Antibodies to Different Adeno-Associated Virus Serotypes Bind Common Regions. Journal of Virology 2013, 87, 9111–9124. [Google Scholar] [CrossRef]
- Jose, A.; Mietzsch, M.; Smith, J.K.; Kurian, J.; Chipman, P.; McKenna, R.; Chiorini, J.; Agbandje-McKenna, M. High-Resolution Structural Characterization of a New Adeno-associated Virus Serotype 5 Antibody Epitope toward Engineering Antibody-Resistant Recombinant Gene Delivery Vectors. Journal of Virology 2019, 93, e01394–18. [Google Scholar] [CrossRef]
- Tseng, Y.S.; Gurda, B.L.; Chipman, P.; McKenna, R.; Afione, S.; Chiorini, J.A.; Muzyczka, N.; Olson, N.H.; Baker, T.S.; Kleinschmidt, J.; Agbandje-McKenna, M. Adeno-Associated Virus Serotype 1 (AAV1)- and AAV5-Antibody Complex Structures Reveal Evolutionary Commonalities in Parvovirus Antigenic Reactivity. Journal of Virology 2015, 89, 1794–1808. [Google Scholar] [CrossRef]
- Crosson, S.M.; Dib, P.; Smith, J.K.; Zolotukhin, S. Helper-free Production of Laboratory Grade AAV and Purification by Iodixanol Density Gradient Centrifugation. Molecular Therapy - Methods & Clinical Development 2018, 10, 1–7. [Google Scholar] [CrossRef]
- Gray, J.T.; Zolotukhin, S. Design and Construction of Functional AAV Vectors. In Adeno-Associated Virus; Snyder, R.O., Moullier, P., Eds.; Humana Press: Totowa, NJ, 2012; Volume 807, pp. 25–46. Series Title: Methods in Molecular Biology. [Google Scholar] [CrossRef]
- Marsic, D. Parent-map: analysis of parental contributions to evolved or engineered protein or DNA sequences. Journal of Open Source Software 2021, 6, 2864. [Google Scholar] [CrossRef]
- Schrödinger, LLC. The PyMOL Molecular Graphics System, Version 3.0.
- Montiel-Garcia, D.; Santoyo-Rivera, N.; Ho, P.; Carrillo-Tripp, M.; Iii, C.B.; Johnson, J.E.; Reddy, V.S. VIPERdb v3.0: a structure-based data analytics platform for viral capsids. Nucleic Acids Research 2021, 49, D809–D816. [Google Scholar] [CrossRef]
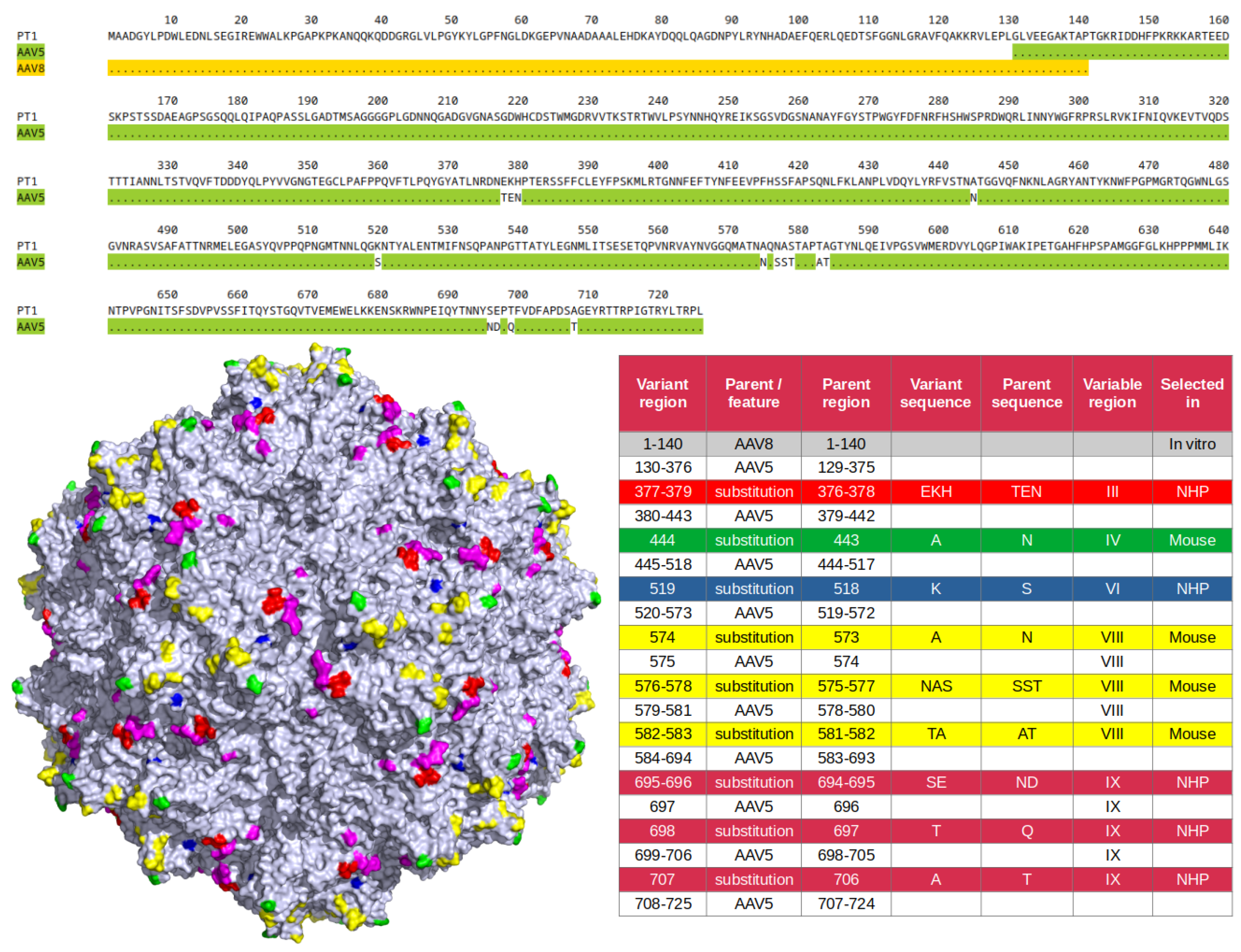

| Position | ADK5a [22] | ADK5b [22] | HL2476 [21] | 3C5 [20] | LSV1 | PT1 |
|---|---|---|---|---|---|---|
| 377 | + | E to K | ||||
| 378 | + | N to H | ||||
| 443 | + | + | N to A | |||
| 576 | + | S to A | ||||
| 577 | + | T to G | T to S | |||
| 578 | + | T to D | ||||
| 697 | + | + | Q to T | |||
| 706 | + | + | + | T to A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




