Submitted:
26 September 2024
Posted:
26 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.3. Hydrogen Peroxide (H2O2) Production
2.4. LPS Assay
2.5. Zonulin Assay
2.6. Plasma D-Dimer Assay
2.7. Albumin Assay
2.8. Assessment of Intrahospital Ischemic and Embolic Events
2.9. In Vitro Study
2.10. Statistical Analysis
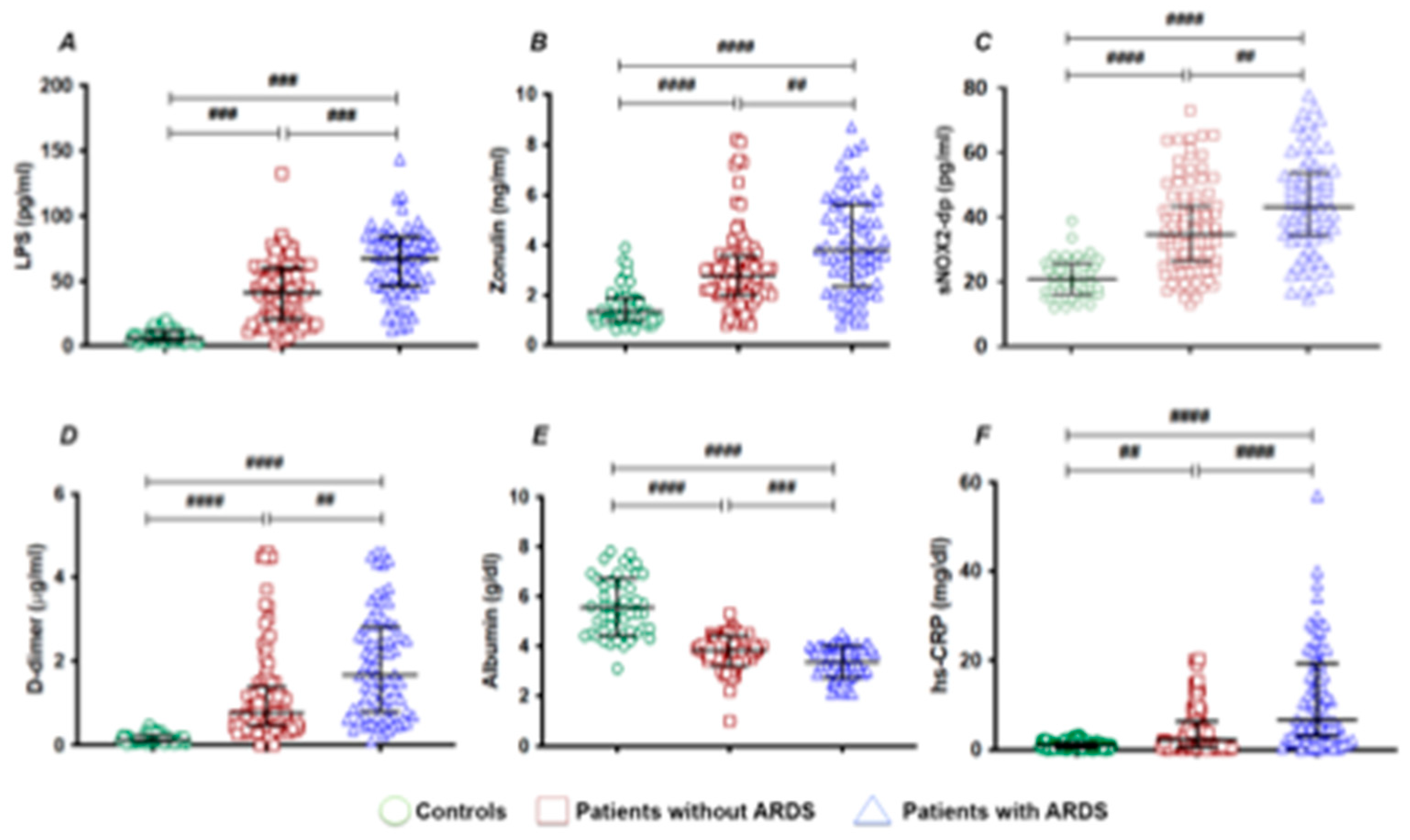
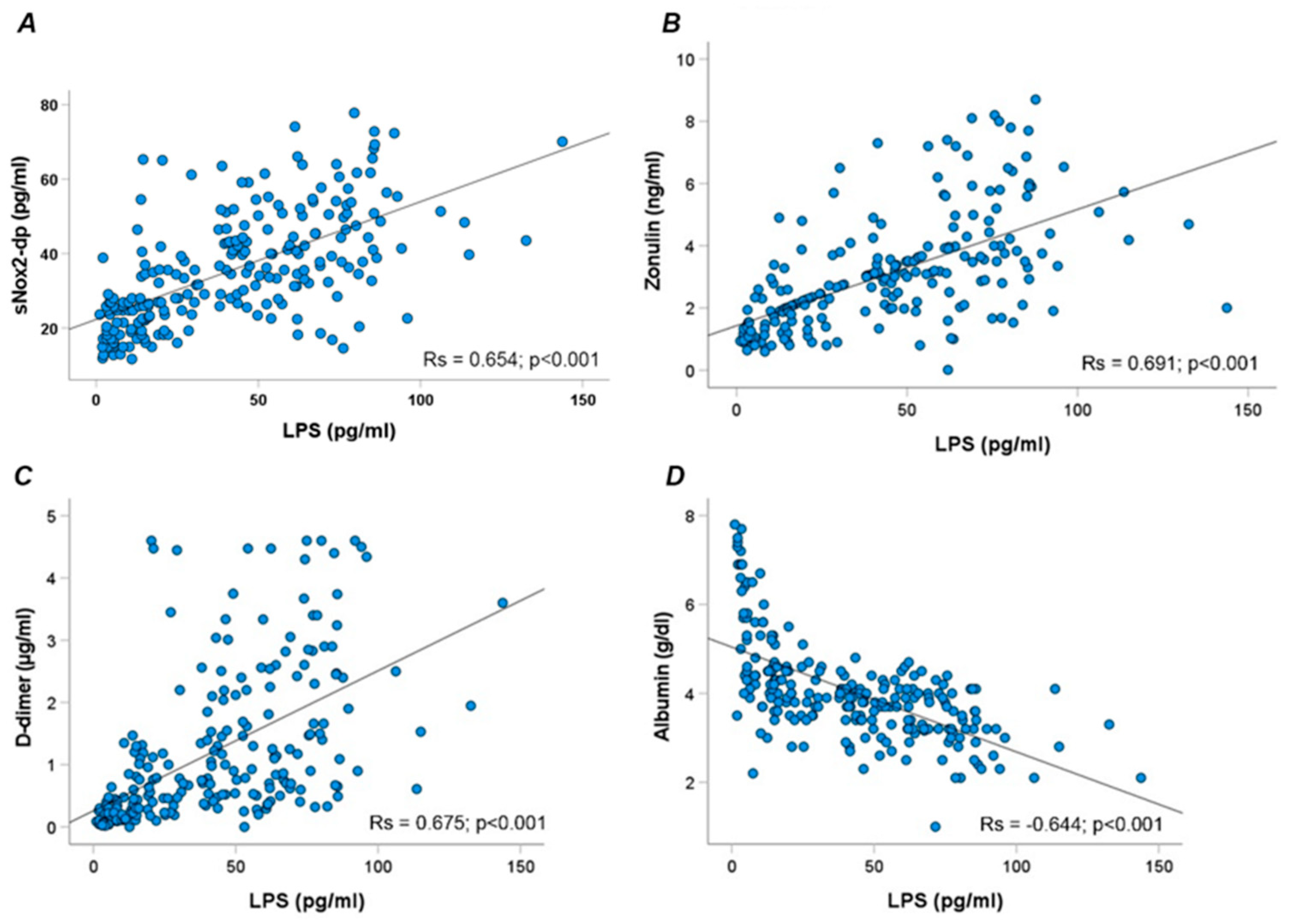
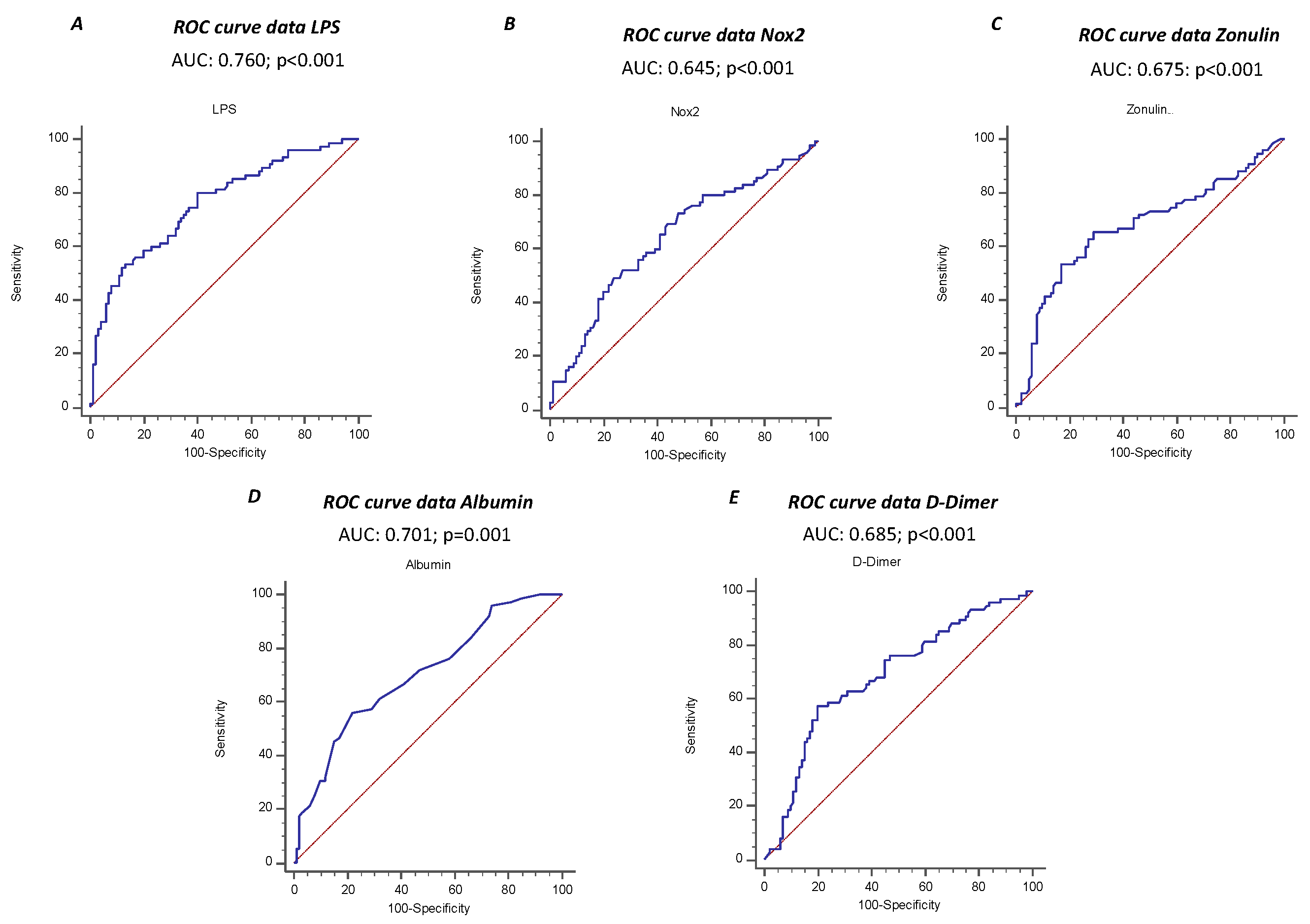
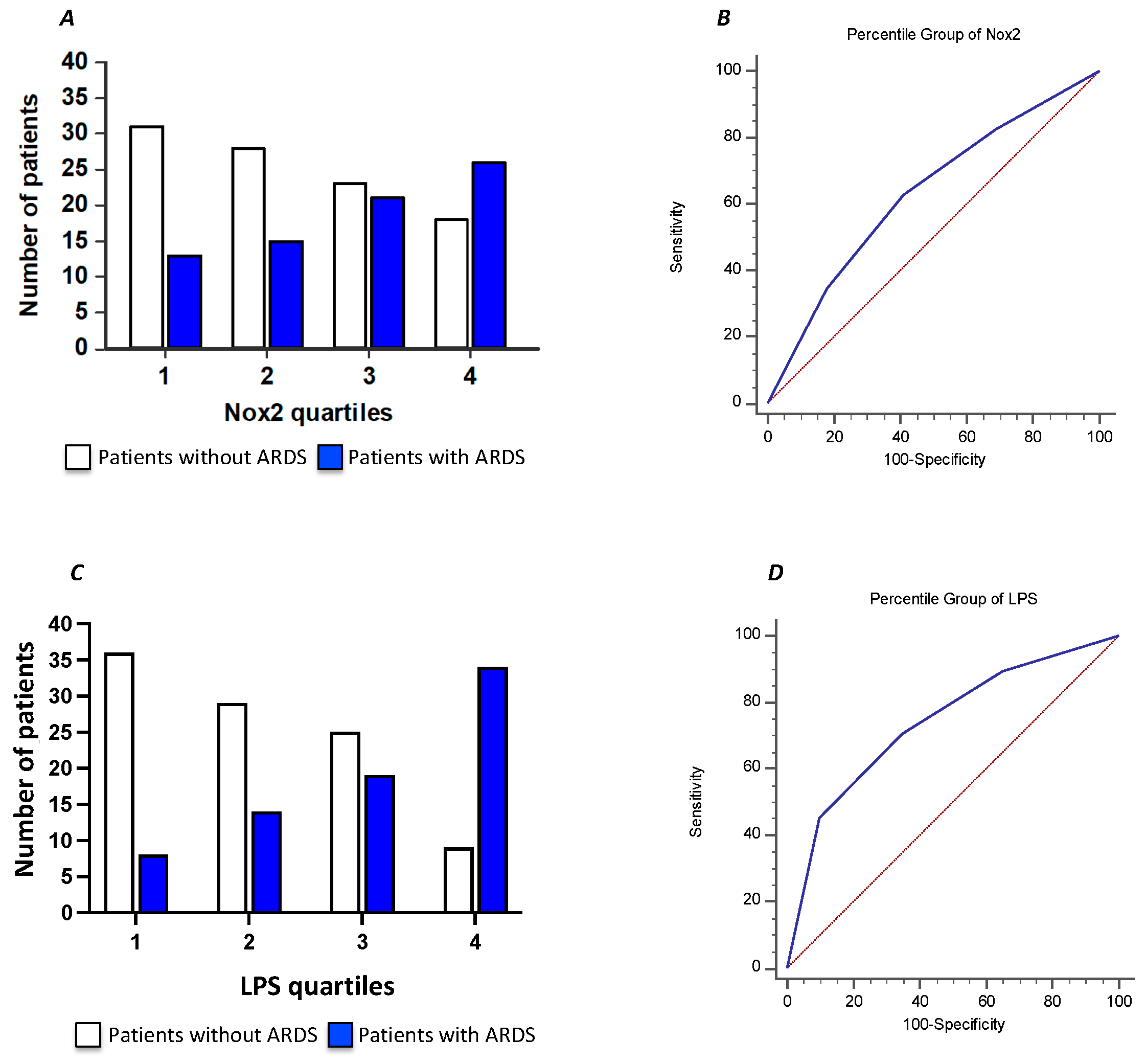
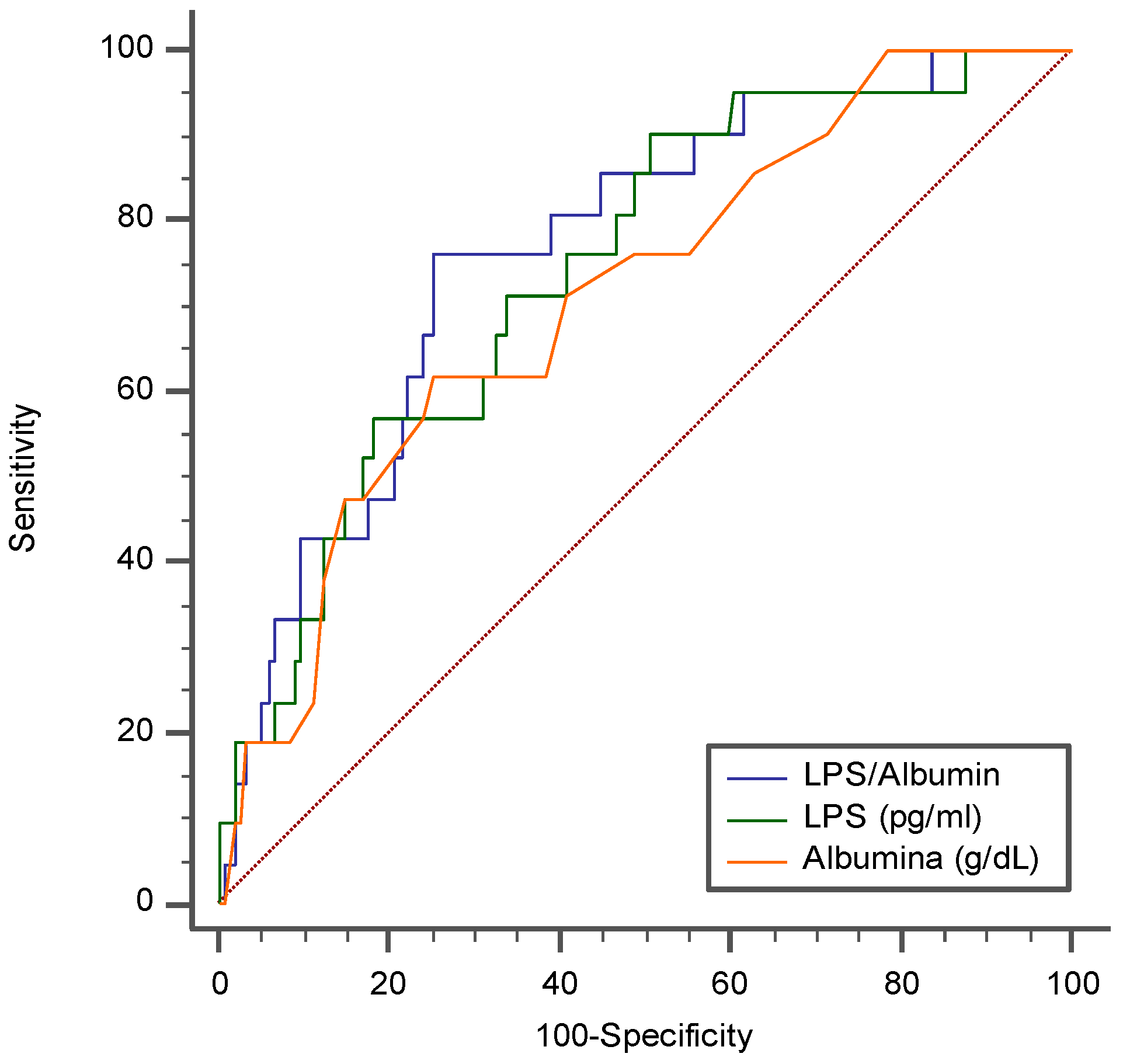
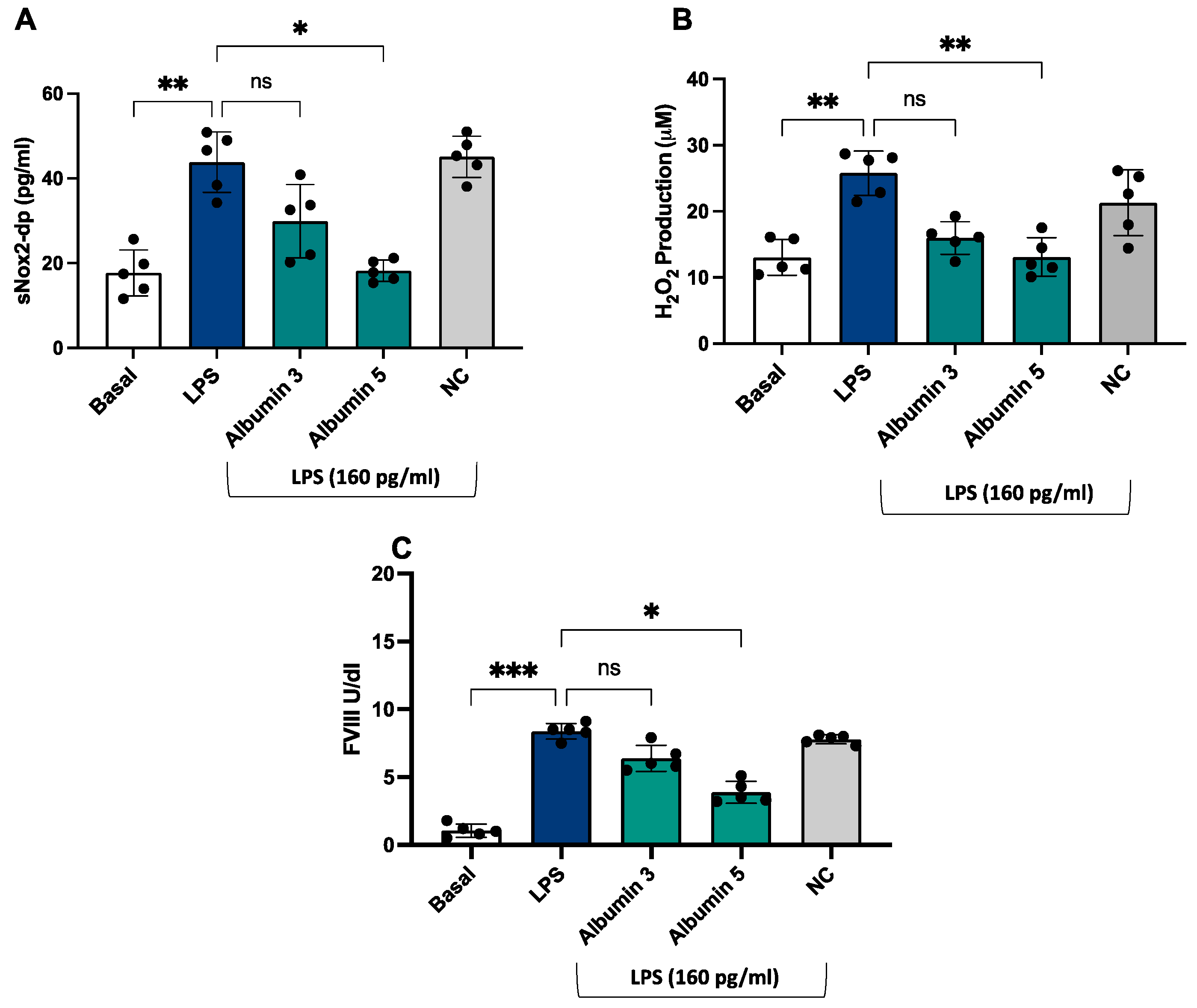
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Violi, F.; Pignatelli, P.; Cammisotto, V.; Carnevale, R.; Nocella, C. COVID-19 and thrombosis: Clinical features, mechanism of disease, and therapeutic implications. Kardiol Pol 2021, 79, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.D.; Schell, J.C.; Rodgers, G.M. The D-dimer assay. Am J Hematol 2019, 94, 833–839. [Google Scholar] [CrossRef]
- Berger, J.S.; Kunichoff, D.; Adhikari, S.; Ahuja, T.; Amoroso, N.; Aphinyanaphongs, Y.; Cao, M.; Goldenberg, R.; Hindenburg, A.; Horowitz, J.; et al. Prevalence and Outcomes of D-Dimer Elevation in Hospitalized Patients With COVID-19. Arterioscler Thromb Vasc Biol 2020, 40, 2539–2547. [Google Scholar] [CrossRef]
- Zhang, L.; Yan, X.; Fan, Q.; Liu, H.; Liu, X.; Liu, Z.; Zhang, Z. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J Thromb Haemost 2020, 18, 1324–1329. [Google Scholar] [CrossRef]
- Zingaropoli, M.A.; Nijhawan, P.; Carraro, A.; Pasculli, P.; Zuccala, P.; Perri, V.; Marocco, R.; Kertusha, B.; Siccardi, G.; Del Borgo, C.; et al. Increased sCD163 and sCD14 Plasmatic Levels and Depletion of Peripheral Blood Pro-Inflammatory Monocytes, Myeloid and Plasmacytoid Dendritic Cells in Patients With Severe COVID-19 Pneumonia. Front Immunol 2021, 12, 627548. [Google Scholar] [CrossRef] [PubMed]
- Cangemi, R.; Della Valle, P.; Calvieri, C.; Taliani, G.; Ferroni, P.; Falcone, M.; Carnevale, R.; Bartimoccia, S.; D’Angelo, A.; Violi, F.; et al. Low-grade endotoxemia and clotting activation in the early phase of pneumonia. Respirology 2016, 21, 1465–1471. [Google Scholar] [CrossRef]
- Cangemi, R.; Casciaro, M.; Rossi, E.; Calvieri, C.; Bucci, T.; Calabrese, C.M.; Taliani, G.; Falcone, M.; Palange, P.; Bertazzoni, G.; et al. Platelet activation is associated with myocardial infarction in patients with pneumonia. J Am Coll Cardiol 2014, 64, 1917–1925. [Google Scholar] [CrossRef] [PubMed]
- Cangemi, R.; Calvieri, C.; Bucci, T.; Carnevale, R.; Casciaro, M.; Rossi, E.; Calabrese, C.M.; Taliani, G.; Grieco, S.; Falcone, M.; et al. Is NOX2 upregulation implicated in myocardial injury in patients with pneumonia? Antioxid Redox Signal 2014, 20, 2949–2954. [Google Scholar] [CrossRef]
- Oliva, A.; Cammisotto, V.; Cangemi, R.; Ferro, D.; Miele, M.C.; De Angelis, M.; Cancelli, F.; Pignatelli, P.; Venditti, M.; Pugliese, F.; et al. Low-Grade Endotoxemia and Thrombosis in COVID-19. Clin Transl Gastroenterol 2021, 12, e00348. [Google Scholar] [CrossRef]
- Carnevale, R.; Sciarretta, S.; Valenti, V.; di Nonno, F.; Calvieri, C.; Nocella, C.; Frati, G.; Forte, M.; d’Amati, G.; Pignataro, M.G.; et al. Low-grade endotoxaemia enhances artery thrombus growth via Toll-like receptor 4: implication for myocardial infarction. Eur Heart J 2020, 41, 3156–3165. [Google Scholar] [CrossRef]
- Giron, L.B.; Dweep, H.; Yin, X.; Wang, H.; Damra, M.; Goldman, A.R.; Gorman, N.; Palmer, C.S.; Tang, H.Y.; Shaikh, M.W.; et al. Plasma Markers of Disrupted Gut Permeability in Severe COVID-19 Patients. Front Immunol 2021, 12, 686240. [Google Scholar] [CrossRef] [PubMed]
- Yonker, L.M.; Gilboa, T.; Ogata, A.F.; Senussi, Y.; Lazarovits, R.; Boribong, B.P.; Bartsch, Y.C.; Loiselle, M.; Rivas, M.N.; Porritt, R.A.; et al. Multisystem inflammatory syndrome in children is driven by zonulin-dependent loss of gut mucosal barrier. J Clin Invest 2021, 131. [Google Scholar] [CrossRef] [PubMed]
- Violi, F.; Pignatelli, P.; Castellani, V.; Carnevale, R.; Cammisotto, V. Gut dysbiosis, endotoxemia and clotting activation: A dangerous trio for portal vein thrombosis in cirrhosis. Blood Rev 2023, 57, 100998. [Google Scholar] [CrossRef] [PubMed]
- Violi, F.; Ceccarelli, G.; Cangemi, R.; Alessandri, F.; D’Ettorre, G.; Oliva, A.; Pastori, D.; Loffredo, L.; Pignatelli, P.; Ruberto, F.; et al. Hypoalbuminemia, Coagulopathy, and Vascular Disease in COVID-19. Circ Res 2020, 127, 400–401. [Google Scholar] [CrossRef]
- De Simone, G.; di Masi, A.; Ascenzi, P. Serum Albumin: A Multifaced Enzyme. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Chen, T.A.; Tsao, Y.C.; Chen, A.; Lo, G.H.; Lin, C.K.; Yu, H.C.; Cheng, L.C.; Hsu, P.I.; Tsai, W.L. Effect of intravenous albumin on endotoxin removal, cytokines, and nitric oxide production in patients with cirrhosis and spontaneous bacterial peritonitis. Scand J Gastroenterol 2009, 44, 619–625. [Google Scholar] [CrossRef]
- Violi, F.; Cangemi, R.; Romiti, G.F.; Ceccarelli, G.; Oliva, A.; Alessandri, F.; Pirro, M.; Pignatelli, P.; Lichtner, M.; Carraro, A.; et al. Is Albumin Predictor of Mortality in COVID-19? Antioxid Redox Signal 2021, 35, 139–142. [Google Scholar] [CrossRef]
- Bartimoccia, S.; Cammisotto, V.; Nocella, C.; Del Ben, M.; D’Amico, A.; Castellani, V.; Baratta, F.; Pignatelli, P.; Loffredo, L.; Violi, F.; et al. Extra Virgin Olive Oil Reduces Gut Permeability and Metabolic Endotoxemia in Diabetic Patients. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Matuschak, G.M.; Lechner, A.J. Acute lung injury and the acute respiratory distress syndrome: pathophysiology and treatment. Mo Med 2010, 107, 252–258. [Google Scholar]
- Konstantinides, S.V.; Meyer, G.; Becattini, C.; Bueno, H.; Geersing, G.J.; Harjola, V.P.; Huisman, M.V.; Humbert, M.; Jennings, C.S.; Jimenez, D.; et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). Eur Respir J 2019, 54. [Google Scholar] [CrossRef]
- Ibanez, B.; James, S.; Agewall, S.; Antunes, M.J.; Bucciarelli-Ducci, C.; Bueno, H.; Caforio, A.L.P.; Crea, F.; Goudevenos, J.A.; Halvorsen, S.; et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J 2018, 39, 119–177. [Google Scholar] [CrossRef] [PubMed]
- Kernan, W.N.; Ovbiagele, B.; Black, H.R.; Bravata, D.M.; Chimowitz, M.I.; Ezekowitz, M.D.; Fang, M.C.; Fisher, M.; Furie, K.L.; Heck, D.V.; et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2014, 45, 2160–2236. [Google Scholar] [CrossRef] [PubMed]
- Bonaca, M.P.; Gutierrez, J.A.; Creager, M.A.; Scirica, B.M.; Olin, J.; Murphy, S.A.; Braunwald, E.; Morrow, D.A. Acute Limb Ischemia and Outcomes With Vorapaxar in Patients With Peripheral Artery Disease: Results From the Trial to Assess the Effects of Vorapaxar in Preventing Heart Attack and Stroke in Patients With Atherosclerosis-Thrombolysis in Myocardial Infarction 50 (TRA2 degrees P-TIMI 50). Circulation 2016, 133, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- De Falco, E.; Carnevale, R.; Pagano, F.; Chimenti, I.; Fianchini, L.; Bordin, A.; Siciliano, C.; Monticolo, R.; Equitani, F.; Carrizzo, A.; et al. Role of NOX2 in mediating doxorubicin-induced senescence in human endothelial progenitor cells. Mech Ageing Dev 2016, 159, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Loffredo, L.; Perri, L.; Catasca, E.; Pignatelli, P.; Brancorsini, M.; Nocella, C.; De Falco, E.; Bartimoccia, S.; Frati, G.; Carnevale, R.; et al. Dark chocolate acutely improves walking autonomy in patients with peripheral artery disease. J Am Heart Assoc 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Menna, C.; De Falco, E.; Pacini, L.; Scafetta, G.; Ruggieri, P.; Puca, R.; Petrozza, V.; Ciccone, A.M.; Rendina, E.A.; Calogero, A.; et al. Axitinib affects cell viability and migration of a primary foetal lung adenocarcinoma culture. Cancer Invest 2014, 32, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Osband, A.J.; Deitch, E.A.; Hauser, C.J.; Lu, Q.; Zaets, S.; Berezina, T.; Machiedo, G.W.; Rajwani, K.K.; Xu, D.Z. Albumin protects against gut-induced lung injury in vitro and in vivo. Ann Surg 2004, 240, 331–339. [Google Scholar] [CrossRef]
- Violi, F.; Cammisotto, V.; Bartimoccia, S.; Pignatelli, P.; Carnevale, R.; Nocella, C. Gut-derived low-grade endotoxaemia, atherothrombosis and cardiovascular disease. Nat Rev Cardiol 2023, 20, 24–37. [Google Scholar] [CrossRef]
- Carnevale, R.; Pastori, D.; Nocella, C.; Cammisotto, V.; Bartimoccia, S.; Novo, M.; Del Ben, M.; Farcomeni, A.; Angelico, F.; Violi, F. Gut-derived lipopolysaccharides increase post-prandial oxidative stress via Nox2 activation in patients with impaired fasting glucose tolerance: effect of extra-virgin olive oil. Eur J Nutr 2019, 58, 843–851. [Google Scholar] [CrossRef]
- Pignatelli, P.; Farcomeni, A.; Menichelli, D.; Pastori, D.; Violi, F. Serum albumin and risk of cardiovascular events in primary and secondary prevention: a systematic review of observational studies and Bayesian meta-regression analysis. Intern Emerg Med 2020, 15, 135–143. [Google Scholar] [CrossRef]
- Meng, F.; Lowell, C.A. Lipopolysaccharide (LPS)-induced macrophage activation and signal transduction in the absence of Src-family kinases Hck, Fgr, and Lyn. J Exp Med 1997, 185, 1661–1670. [Google Scholar] [CrossRef] [PubMed]
- Carnevale, R.; Raparelli, V.; Nocella, C.; Bartimoccia, S.; Novo, M.; Severino, A.; De Falco, E.; Cammisotto, V.; Pasquale, C.; Crescioli, C.; et al. Gut-derived endotoxin stimulates factor VIII secretion from endothelial cells. Implications for hypercoagulability in cirrhosis. J Hepatol 2017, 67, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Ma, X.; Huang, G. Understanding thrombosis: the critical role of oxidative stress. Hematology 2024, 29, 2301633. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, K.A.; Stoffersen, E. On the inhibitory effect of albumin on platelet aggregation. Thromb Res 1980, 17, 13–18. [Google Scholar] [CrossRef]
- Maclouf, J.; Kindahl, H.; Granstrom, E.; Samuelsson, B. Interactions of prostaglandin H2 and thromboxane A2 with human serum albumin. Eur J Biochem 1980, 109, 561–566. [Google Scholar] [CrossRef]
- Basili, S.; Carnevale, R.; Nocella, C.; Bartimoccia, S.; Raparelli, V.; Talerico, G.; Stefanini, L.; Romiti, G.F.; Perticone, F.; Corazza, G.R.; et al. Serum Albumin Is Inversely Associated With Portal Vein Thrombosis in Cirrhosis. Hepatol Commun 2019, 3, 504–512. [Google Scholar] [CrossRef]
- Violi, F.; Ceccarelli, G.; Loffredo, L.; Alessandri, F.; Cipollone, F.; D’Ardes, D.; D’Ettorre, G.; Pignatelli, P.; Venditti, M.; Mastroianni, C.M.; et al. Albumin Supplementation Dampens Hypercoagulability in COVID-19: A Preliminary Report. Thromb Haemost 2021, 121, 102–105. [Google Scholar] [CrossRef]
| Variable | Controls | COVID-19 patients |
p | Patients without ARDS | Patients with ARDS | p |
|---|---|---|---|---|---|---|
| N Age (years)a Female sex (%) |
50 | 175 | 100 | 75 | ||
| 65.7±12.7 | 64.9±16.0 | 0.720 | 64.6±15.9 | 65.2±16.3 | 0.804 | |
| 34 | 34 | 0.970 | 40 | 27 | 0.066 | |
| BMI a Smokers (%) |
26.6±3.7 | 26.9±3.6 | 0.701 | 26.0±3.5 | 27.8±3.5 | 0.087 |
| 4 | 5 | 0.647 | 9 | 5 | 0.360 | |
| Hypertension (%) COPD (%) Atrial fibrillation (%) Diabetes (%) |
0 | 27 | <0.001 | 24 | 32 | 0.240 |
| 0 | 10 | 0.022 | 15 | 4 | 0.006 | |
| 0 | 5 | 0.124 | 7 | 0.75 | 0.076 | |
| 0 | 18 | 0.001 | 19 | 16 | 0.607 | |
| ACE-inhibitors (%) Thrombotic events |
0 | 25 | <0.001 | 20 | 31 | 0.105 |
| 0 | 21 | <0.001 | 6 | 15 | 0.004 | |
| HS-CRP (mg/dL) b | 1 [0.3-1.5] | 3.8 [1.1-10.8] | <0.001 | 2.1 [0.5-6.2] | 6.7 [3.0-19.2] | <0.001 |
| D-dimer (µg/mL) b | 0.12 [0.07-0.23] | 1.09 [0.54-2.25] | <0.001 | 0.77 [0.46-1.38] | 1.66 [0.79-2.81] | <0.001 |
| Albumin (g/dL) a | 5.56±1.14 | 3.62±0.66 | <0.001 | 3.82±0.61 | 3.36±0.63 | <0.001 |
| Zonulin (ng/mL) b | 1.33 [0.98-1.88] | 3.10 [2.10-4.22] | <0.001 | 2.79 [2.0-3.54] | 3.8 [2.37-5.60] | <0.001 |
| LPS (pg/mL) b | 6.0 [3.7-11.7] | 50.3 [29.4-71.7] | <0.001 | 41.5 [20.0-60.5] | 67.7 [46.3-83.9] | <0.001 |
| sNOX2-dp (pg/mL) | 21.9 [16.0-26.1] | 38.9 [28.0-50.2] | <0.001 | 34.8 [26.5-43.5] | 43.2 [34.5-53.8] | 0.001 |
| Variables | OR | 95% CI | p | |
| II quartile LPS vs I | 1.852 | 0.660 | 5.202 | 0.242 |
| III quartile LPS vs I | 2.158 | 0.772 | 6.035 | 0.143 |
| VI quartile LPS vs I | 6.819 | 2.231 | 20.843 | 0.001 |
| Albumin | 0.520 | 0.280 | 0.966 | 0.039 |
| hs-CRP | 1.079 | 1.023 | 1.138 | 0.005 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




