Submitted:
21 September 2024
Posted:
24 September 2024
You are already at the latest version
Abstract
Keywords:
1. Background
2. Methods
2.1. Genome Sequencing and Promoter Analysis of Lacticaseibacillus paracasei BCRC-16100
2.2. Production of NIMO-CH
2.3. Collection, Animal Care, and Acclimatization of Experimental Animals
2.4. Excision of Wounds in ICR Mice and Application of Treatments
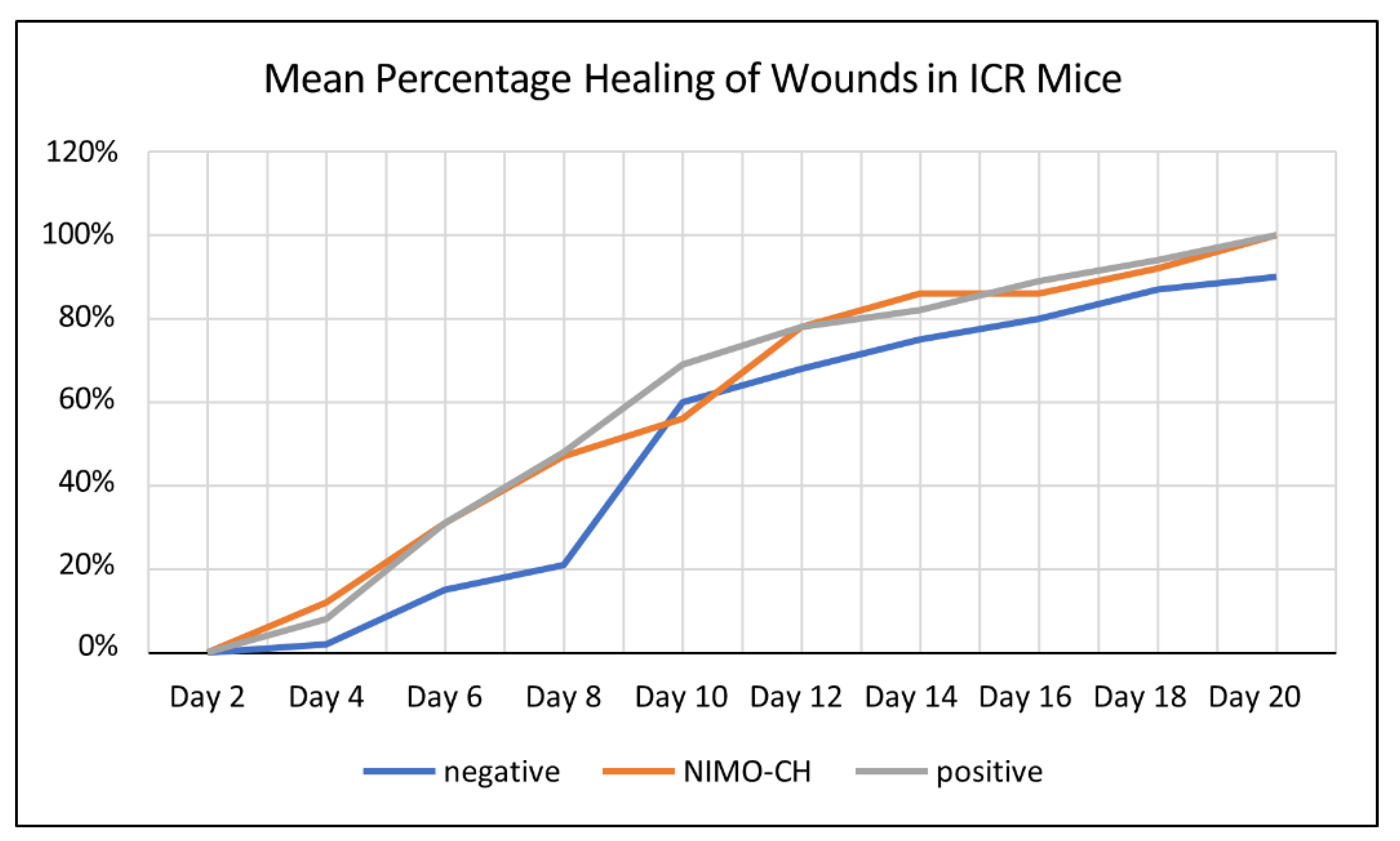
2.5. Assessment of the Wound Healing Activity
2.6. Morphoanatomical Assessment of Wounded ICR Mice
2.7. Histological Assessment of Wounded ICR Mice
3. Results
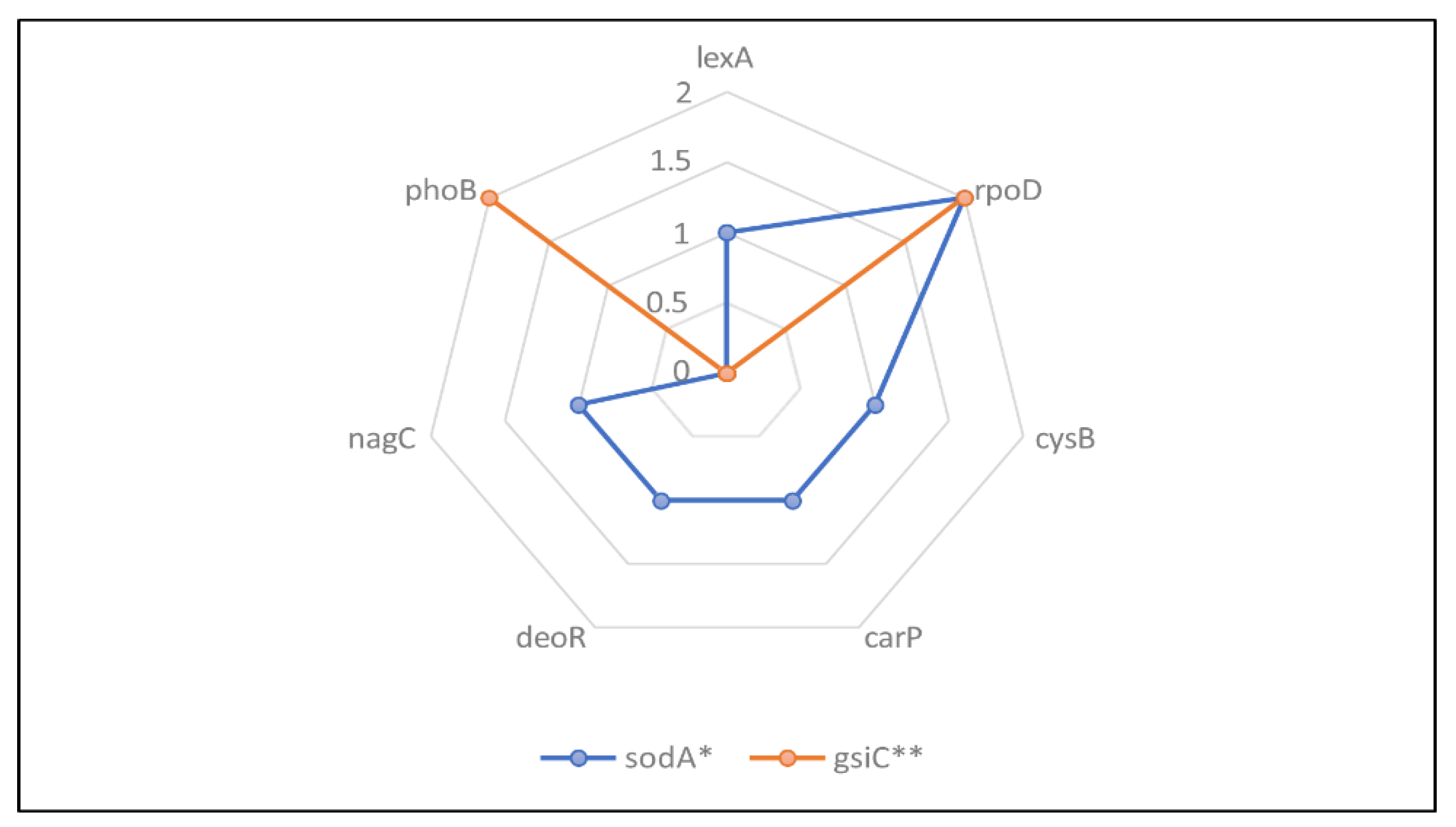
3.1. Genomic Analysis
3.2. Morphoanatomical Assessment
| Treatment Groups | Observation Period Day | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2 | 4 | 6 | 8 | 10 | 12 | 14 | 16 | 18 | 20 | |
| Untreated (-) Control | A | A | B | B | C | C | D | E | E | F |
| NIMO-CH | A | B | C | C | C | D | E | E | F | F |
| DuoDERM (+) control | A | B | C | C | C | D | D | E | E | F |
| Treatment Groups | Observation Period Day |
|---|---|
| 2 4 6 8 10 12 14 16 18 20 | |
| Untreated (-) Control NIMO-CH DuoDERM (+) Control |
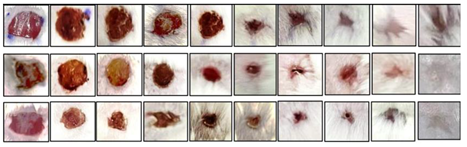 |
| Treatment Groups | Observation Period Day | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2 | 4 | 6 | 8 | 10 | 12 | 14 | 16 | 18 | 20 | |
| Untreated (-) Control | 0 | 2 | 15 | 21 | 60 | 68 | 75 | 80 | 87 | 90b |
| NIMO-CH | 0 | 12 | 31 | 47 | 56 | 78 | 86 | 86 | 92 | 100a |
| DuoDERM (+) control | 0 | 8 | 31 | 48 | 69 | 78 | 82 | 89 | 94 | 100a |
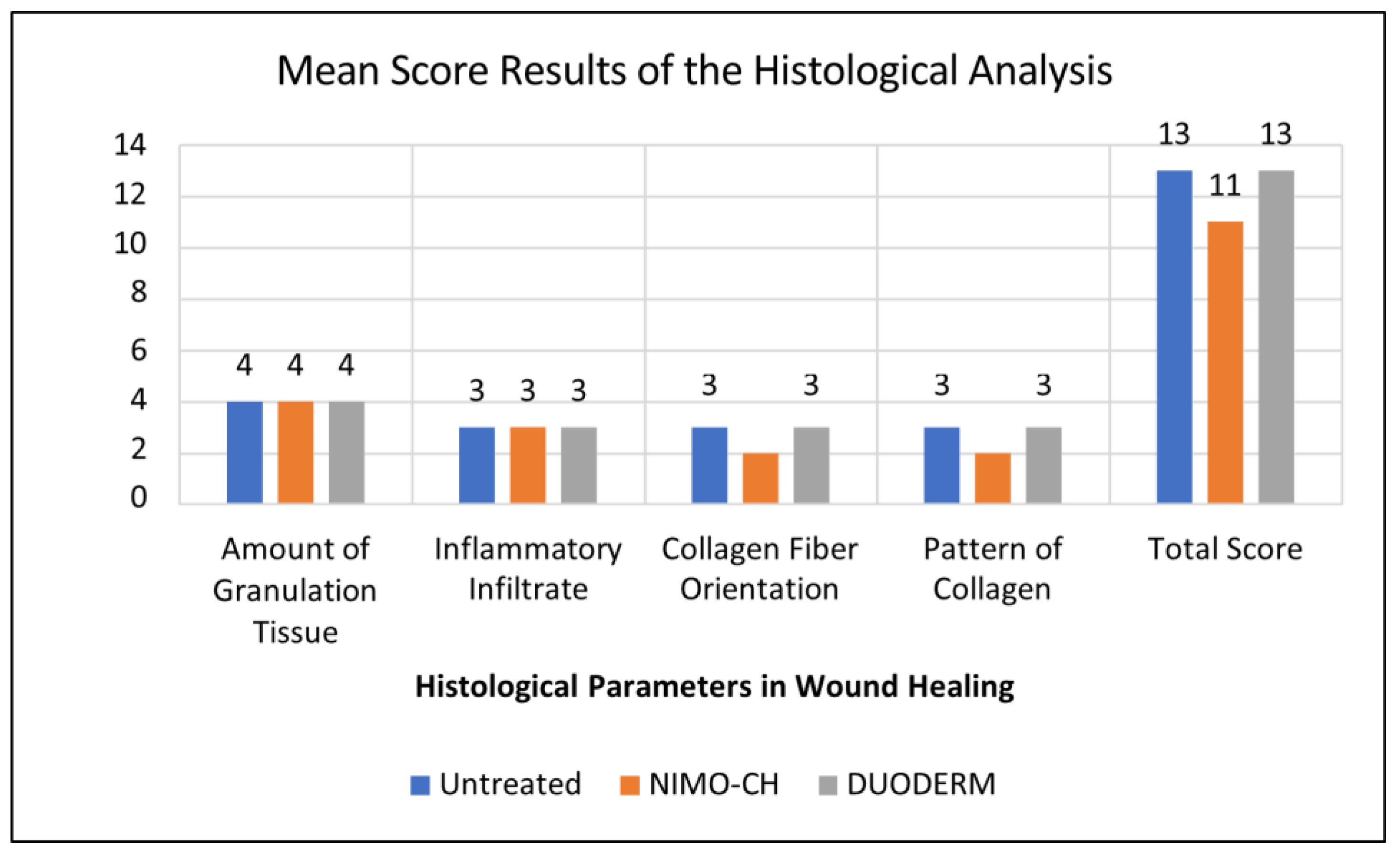
3.3. Histological Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Asl, S., Mousavi, M., & Labbafi, M. (2017). Synthesis and Characterization of Carboxymethyl Cellulose from Sugarcane Bagasse. Journal of Food Processing & Technology, 08(08). [CrossRef]
- Comino-Sanz, I. M., López-Franco, M. D., Castro, B., & Pancorbo-Hidalgo, P. L. (2021). The Role of Antioxidants on Wound Healing: A Review of the Current Evidence. Journal of Clinical Medicine, 10(16). [CrossRef]
- Cariaga, J. F., Domingo, A. G., Santos, B. S., & Agrupis, S. C. (2023). Isolation of a - Cellulose from Nipa (Nypa fruticans Wurmb) Frond using Physico-Chemical Treatment. 16(23), 1754–1759. [CrossRef]
- Dai, T., Kharkwal, G. B., Tanaka, M., Huang, Y.-Y., Bil, V. J., & Hamblin, M. R. (2011). Animal models of external traumatic wound infections. 2(4), 296–315. [CrossRef]
- Domingo AG, Cariaga JF, Santos BS, Agrupis SC (2023) Production of Cellulose Hydrogel from Nipa (Nypa fruticans Wurmb) Frond. Indian Journal of Science and Technology 16(21): 1-8.
- Iuchi, Y., Roy, D., Okada, F., Kibe, N., Tsunoda, S., Suzuki, S., … Fujii, J. (2010). Spontaneous skin damage and delayed wound healing in SOD1-deficient mice. Molecular and Cellular Biochemistry, 341(1-2), 181–194. [CrossRef]
- Jair, W., Lu, F., Huang, W., Pan, Y., Lin, L., Huang, H., & Yang, C. (2019). Roles of the Two-MnSOD System of Stenotrophomonas maltophilia in the Alleviation of Superoxide Stress. International Journal of Molecular Sciences, 20(7). [CrossRef]
- Järbrink, K., Ni, G., Sönnergren, H., Schmidtchen, A., Pang, C., Bajpai, R., & Car, J. (2016). Prevalence and incidence of chronic wounds and related complications: a protocol for a systematic review. Systematic Reviews, 5(1). [CrossRef]
- Jose, G., Shalumon, K. T., Chen, J.-P., & Yu, C. (2019). Natural Polymers Based Hydrogels for Cell Culture Applications. Current Medicinal Chemistry, 26. [CrossRef]
- Martinengo L, Olsson M, Bajpai R, et al. (2019) Prevalence of chronic wounds in the general population: systematic review and meta-analysis of observational studies. Ann Epidemiol. 2019;29:8-15.
- Plikus, M. V., Guerrero-Juarez, C. F., Ito, M., Li, Y. R., Dedhia, P. H., Zheng, Y., Shao, M., Gay, D. L., Ramos, R., Hsi, T.-C., Oh, J. W., Wang, X., Ramirez, A., Konopelski, S. E., Elzein, A., Wang, A., Supapannachart, R. J., Lee, H.-L., Lim, C. H., & Nace, A. (2017). Regeneration of fat cells from myofibroblasts during wound healing. Science, 355(6326), 748–752. [CrossRef]
- Rhea, L., & Dunnwald, M. (2020). Murine Excisional Wound Healing Model and Histological Morphometric Wound Analysis. (162). [CrossRef]
- Schultz, G. S., Chin, G. A., Moldawer, L., & Diegelmann, R. F. (2011). Principles of Wound Healing (R. Fitridge & M. Thompson, Eds.). PubMed; University of Adelaide Press. https://www.ncbi.nlm.nih.gov/books/NBK534261/.
- Su, C., Liu, T., Wang, H., & Yang, W. (2023). Histopathological Study on Collagen in Full-Thickness Wound Healing in Fraser’s Dolphins (Lagenodelphis hosei). Animals, 13(10), 1681–1681. [CrossRef]
- Sultana, J., Molla, M. R., Kamal, M., Shahidullah, M., Begum, F., & Bashar, M. A. (2009). Histological differences in wound healing in Maxillofacial region in patients with or without risk factors. Bangladesh Journal of Pathology, 24(1), 3–8. [CrossRef]
- Wallace, H., Basehore, B., & Zito, P. (2023). Wound Healing Phases. Statpearls Publishing. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK470443/.
- Wang, C., Wang, M., Xu, T., Zhang, X., Lin, C., Gao, W., Xu, H., Lei, B., & Mao, C. (2019). Engineering Bioactive Self-Healing Antibacterial Exosomes Hydrogel for Promoting Chronic Diabetic Wound Healing and Complete Skin Regeneration. Theranostics, 9(1), 65–76. [CrossRef]
- Wang, Z., Qi, F., Luo, H., Xu, G., & Wang, D. (2022). Inflammatory Microenvironment of Skin Wounds. Frontiers in Immunology, 13. [CrossRef]
- Yaseen, H. S., Asif, M., Saadullah, M., Mahrukh, Asghar, S., Shams, M. U., Bazmi, R. R., Saleem, M., Yousaf, H. M., & Yaseen, M. (2020). Methanolic extract of Ephedra ciliata promotes wound healing and arrests inflammatory cascade in vivo through downregulation of TNF-α. Inflammopharmacology, 28(6), 1691– 1704. [CrossRef]
| Number | Histological Parameter |
|---|---|
| 1 | Amount of granulation tissue (profound-1, moderate-2, scanty-3, absent-4) |
| 2 | Inflammatory infiltrate (plenty-1, moderate-2, a few-3) |
| 3 | Collagen fiber orientation (vertical-1, mixed-2, horizontal-3) |
| 4 | Pattern of collagen (reticular-1, mixed-2, fascicle-3) |
| 5 | Amount of early collagen (profound-1, moderate-2, minimal-3) |
| 6 | Amount of mature collagen (profound-1, moderate-2, minimal-3) |
| Treatment Groups | Initial Weight (g) | Final Weight (g) |
| Untreated (-) | 32.30g | 34.72 |
| NIMO-CH | 25.80g | 28.52 |
| DuoDERM (+) | 29.02g | 29.75 |
| Significance | ns | ns |
| CV% | 13.02 | 14.24 |
| Treatment Groups | Mean Score Histological Parameter | ||||
|---|---|---|---|---|---|
| Amount of Granulation Tissue | Inflammatory Infiltrate | Collagen Fiber Orientation | Pattern of Collagen | Total Mean Score | |
| Untreated (-) Control | 4 Absent |
3 Few |
3 Horizontal |
3 Fascicle |
13 |
| NIMO-CH | 4 Absent |
3 Few |
2 Mixed |
2 Mixed |
11 |
| DuoDERM (+) Control | 4 Absent |
3 Few |
3 Mixed |
3 Mixed |
13 |
| Significance | ns | ns | ns | ns | ns |
| CV % | 10.65% | 11.76% | 22.59% | 22.59% | 11.58% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




