Submitted:
23 September 2024
Posted:
24 September 2024
You are already at the latest version
Abstract
Keywords:
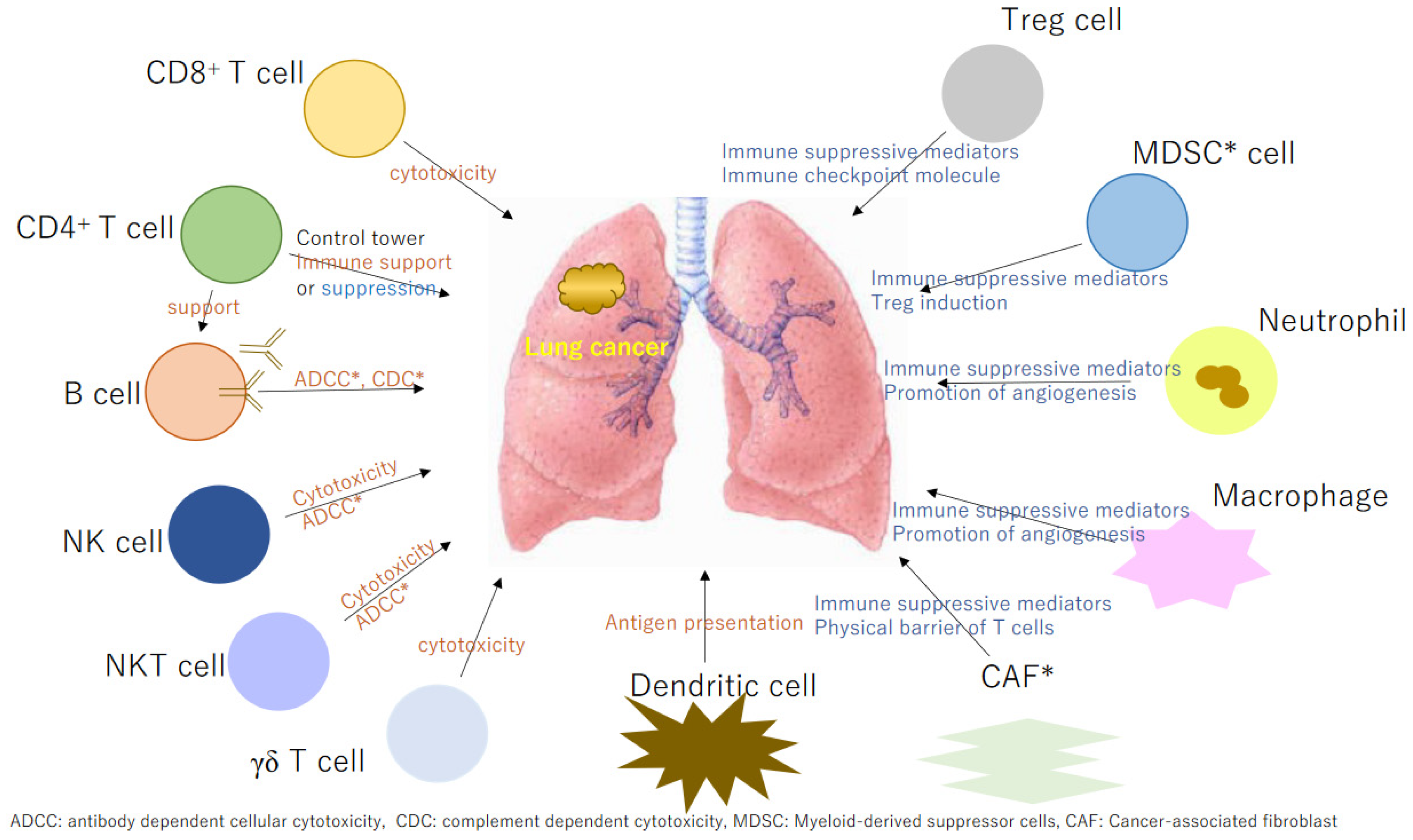
1. Introduction
2. Tertiary Lymphoid Structures
3. Tumor-Infiltrating Lymphocytes
4. Tumor Mutation Burden
4.1. CD8+ T Cells
4.2. CD4+ T Cells
4.3. B Cells
4.4. Regulatory T Cells
4.5. Natural Killer Cells
4.6. Natural Killer T Cells
4.7. γδ T Cells
5. Tumor-Associated Macrophages
6. Dendritic Cells
7. Myeloid-Derived Suppressor Cells
8. Neutrophils
9. Cancer-Associated Fibroblasts
10. Chimeric Antigen Receptor-T Cells
11. T Cell Receptor-Transduced Effector Cells
12. Conclusion
Author Contributions
Funding
Conflicts of Interest
Ethical Statement
Abreviations
| ICI | immune checkpoint inhibitor |
| TLS | Tertiary lymphoid structures |
| DC | dendritic cell |
| TIL | Tumor-infiltrating lymphocyte |
| MDSC | myeloid-derived suppressor cell |
| TAM | tumor-associated macrophage |
| CAF | cancer-associated fibroblast |
| MSC | mesenchymal stem cell |
| ECM | extracellular matrix |
| PD-1 | programmed cell death -1 |
| Treg | regulatory T cell |
| TMB | Tumor mutation burden |
| PD-L1 | programmed cell death - ligand 1 |
| MMR | mismatch repair |
| dMMR | MMR deficient |
| EPCAM | epithelial cell adhesion molecule |
| MHC | major histocompatibility complex |
| CTL | cytotoxic T lymphocyte |
| TCR | T cell receptor |
| PCR | polymerase chain reaction |
| SCID mouse | severe combined immunodeficiency mouse |
| IFN | interferon |
| TNF | tumor necrosis factor |
| IL | interleukin |
| LAG-3 | lymphocyte activation gene 3 protein |
| TIM-3 | T cell immunoglobulin domain and mucin domain protein 3 |
| BCR | B-cell receptor |
| TIB | tumor infiltrating B lymphocyte |
| IPEX | immunedysregulation polyendocrinopathy enteropathy X-linked |
| Foxp3 | forkhead box protein P3 |
| CTLA-4 | cytotoxic T-lymphocyte associated antigen-4 |
| NK cell | Natural killer cell |
| TRAIL | TNF-related apoptosis-inducing ligand |
| MICA/B | MHC class I chain-related gene A and B |
| NKG2D | natural killer group 2 member D |
| KIR | killer cell immunoglobulin-like receptors |
| NKT cell | Natural killer T cell |
| α-GalCer | α-galactosylceramide |
| TME | tumor microenvironment |
| RT-MPS | Microparticles released from irradiated tumor cells |
| DSB | DNA double-strand break |
| LTB | lymphotoxin beta |
| TAN | tumor-associated neutrophils |
| ROS | reactive oxygen species |
| NE | neutrophil elastase |
| MMP-9 | matrix metalloproteinase-9 |
| NSCLC | non-small cell lung cancer |
| NLR | neutrophil-to-lymphocyte ratio |
| PFS | progression-free survival |
| OS | overall survival |
| CAR-T cell | Chimeric antigen receptor- T cell |
| CXCR4 | CXC chemokine receptor 4 |
References
- Sautès-Fridman, C.; Petitprez, F.; Calderaro, J.; Fridman, W.H. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat. Rev. Cancer 2019, 19, 307–325. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Romero, K.; Rodríguez, R.M.; Amedei, A.; Barceló-Coblijn, G.; Lopez, D.H. Immune Landscape in Tumor Microenvironment: Implications for Biomarker Development and Immunotherapy. Int. J. Mol. Sci. 2020, 21, 5521. [Google Scholar] [CrossRef]
- Dieu-Nosjean, M.-C.; Goc, J.; Giraldo, N.A.; Sautès-Fridman, C.; Fridman, W.H. Tertiary lymphoid structures in cancer and beyond. Trends Immunol. 2014, 35, 571–580. [Google Scholar] [CrossRef]
- Petitprez, F.; de Reyniès, A.; Keung, E.Z.; Chen, T.W.-W.; Sun, C.-M.; Calderaro, J.; Jeng, Y.-M.; Hsiao, L.-P.; Lacroix, L.; Bougoüin, A.; et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature 2020, 577, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Helmink, B.A.; Reddy, S.M.; Gao, J.; Zhang, S.; Basar, R.; Thakur, R.; Yizhak, K.; Sade-Feldman, M.; Blando, J.; Han, G.; et al. B cells and tertial lymphoid structures promote immunotherapy response. Nature. 2020, 577, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Cabrita, R.; Lauss, M.; Sanna, A.; Donia, M.; Larsen, M.S.; Mitra, S.; Johansson, I.; Phung, B.; Harbst, K.; Vallon-Christersson, J.; et al. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature 2020, 577, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Mellman, I.; Chen, D.S.; Powles, T.; Turley, S.J. The cancer-immunity cycle: Indication, genotype, and immunotype. Immunity 2023, 56, 2188–2205. [Google Scholar] [CrossRef]
- Kumagai, S.; Togashi, Y.; Kamada, T.; Sugiyama, E.; Nishinakamura, H.; Takeuchi, Y.; Vitaly, K.; Itahashi, K.; Maeda, Y.; Matsui, S.; et al. The PD-1 expression balance between effector and regulatory T cells predicts the clinical efficacy of PD-1 blockade therapies. Nat. Immunol. 2020, 21, 1346–1358. [Google Scholar] [CrossRef]
- McGivern, A.; Wynter, C.; Whitehall, V.; Kambara, T.; Spring, K.; Walsh, M.D.; Barker, M.; Arnold, S.; Simms, L.; Leggett, B.; et al. Promoter Hypermethylation Frequency and BRAF Mutations Distinguish Hereditary Non-Polyposis Colon Cancer from Sporadic MSI-H Colon Cancer. Fam. Cancer 2004, 3, 101–107. [Google Scholar] [CrossRef]
- Kuiper, R.P.; Vissers, L.E.; Venkatachalam, R.; Bodmer, D.; Hoenselaar, E.; Goossens, M.; Haufe, A.; Kamping, E.; Niessen, R.C.; Hogervorst, F.B.; et al. Recurrence and variability of germline EPCAM deletions in Lynch syndrome. Hum. Mutat. 2011, 32, 407–414. [Google Scholar] [CrossRef]
- Niessen, R.C.; Hofstra, R.M.W.; Westers, H.; Lightenberg, M.J.L; Kooi, K.; Jager, P.O.J; Groote, M.L. Dijikhuizen, T. ; Olderode-Berends, M.J.W.; Hollema, H.; et al. Germline hypermethylation of MLH1 and EPCAM deletions are a frequent cause of Lynch syndrome. Genes Chromosomes Cancer. 2009, 48, 737–744. [Google Scholar]
- Goel, A.; Nguyen, T.P; Leung, H.C.E.; Nagasaka, T.; Rhees, J.; Hotchkiss, E.; Arnold, M.; Banerji, P.; Koi, M.; Kwok, C.T; et al. De novo constitutional MLH1 epimutations confer early-onset colorectal cancer in two new sporadic Lynch syndrome cases, with derivation of the epimutation on the paternal allele in one. Int J Cancer. 2011, 128, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, R.; Chitre, A.S.; Scherl, A.; Wu, T.D.; Patil, N.S.; de Almeida, P.; Kadel, I.E.E.; Madireddi, S.; Au-Yeung, A.; Takahashi, C.; et al. Intratumoral CD103+ CD8+ T cells predict response to PD-L1 blockade. J. Immunother. Cancer 2021, 9, e002231. [Google Scholar] [CrossRef] [PubMed]
- Duhen, T.; Duhen, R.; Montler, R.; Moses, J.; Moudgil, T.; de Miranda, N.F.; Goodall, C.P.; Blair, T.C.; Fox, B.A.; McDermott, J.E.; et al. Co-expression of CD39 and CD103 identifies tumor-reactive CD8 T cells in human solid tumors. Nat. Commun. 2018, 9, 2724. [Google Scholar] [CrossRef]
- Ichiki, Y.; Ueno, M.; Yanagi, S.; Kanasaki, Y.; Goto, H.; Fukuyama, T.; Mikami, S.; Nakanishi, K.; Ishida, T. An analysis of the immunological tumor microenvironment of primary tumors and regional lymph nodes in squamous cell lung cancer. Transl. Lung Cancer Res. 2021, 10, 3520–3537. [Google Scholar] [CrossRef]
- Ichiki, Y.; Fukuyama, T.; Ueno, M.; Kanasaki, Y.; Goto, H.; Takahashi, M.; Mikami, S.; Kobayashi, N.; Nakanishi, K.; Hayashi, S.; et al. Immune profile analysis of peripheral blood and tumors of lung cancer patients treated with immune checkpoint inhibitors. Transl. Lung Cancer Res. 2022, 11, 2192–2207. [Google Scholar] [CrossRef]
- Ichiki, Y.; Takenoyama, M.; Mizukami, M.; So, T.; Sugaya, M.; Yasuda, M.; So, T.; Hanagiri, T.; Sugio, K.; Yasumoto, K. Simultaneous Cellular and Humoral Immune Response against Mutated p53 in a Patient with Lung Cancer. J. Immunol. 2004, 172, 4844–4850. [Google Scholar] [CrossRef]
- Ichiki, Y; Shigematsu, Y.; Baba, T.; Shiota, H.; Fukuyama, T.; Nagata, Y.; So, T.; Yasuda, M.; Takenoyama, M.; Yasumoto, K. Development of adoptive immunotherapy with KK-LC-1 specific TCR transduced γδT cells against lung cancer cells. Cancer Sci. 2020, 111, 4021–4030. [Google Scholar] [CrossRef]
- Kagamu, H.; Kitano, S.; Yamaguchi, O.; Yoshimura, K.; Horimoto, K.; Kitazawa, M.; Fukui, K.; Shiono, A.; Mouri, A.; Nishihara, F.; et al. Cancer Immunol Res 2020, 8, 334–344. [CrossRef]
- Coussens, L.M.; Zitvogel, L.; Palucka, A.K. Neutralizing tumor-promoting chronic inflammation: a magic bullet? Science. 2013, 339, 286–291. [Google Scholar] [CrossRef]
- Speiser, D.E.; Utzschneider, D.T.; Oberle, S.G.; Munz, C.; Romero, P.; Zehn, D. T cell differentiation in chronicinfection and cancer: functional adaptation or exhaustion? Nat Rev Immunol. 2014, 14, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Fridman, WH.; Pages, F.; Sautes-Fridman, C.; Galon, J. The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer. 2012, 12, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Wouters, M.C.A.; Nelson, B.H. Prognostic Significance of Tumor-Infiltrating B Cells and Plasma Cells in Human Cancer. Clin. Cancer Res. 2018, 24, 6125–6135. [Google Scholar] [CrossRef] [PubMed]
- Cipponi, A.; Mercier, M.; Seremet, T.; Baurain, J.-F.; Théate, I.; Oord, J.v.D.; Stas, M.; Boon, T.; Coulie, P.G.; van Baren, N. Neogenesis of Lymphoid Structures and Antibody Responses Occur in Human Melanoma Metastases. Cancer Res. 2012, 72, 3997–4007. [Google Scholar] [CrossRef] [PubMed]
- Germain, C.; Gnjatic, S.; Dieu-Nosjean, M.-C. Tertiary Lymphoid Structure-Associated B Cells are Key Players in Anti-Tumor Immunity. Front. Immunol. 2015, 6, 67–67. [Google Scholar] [CrossRef]
- Sautès-Fridman, C.; Petitprez, F.; Calderaro, J.; Fridman, W.H. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat. Rev. Cancer 2019, 19, 307–325. [Google Scholar] [CrossRef]
- Petitprez, F.; de Reyniès, A.; Keung, E.Z.; Chen, T.W.-W.; Sun, C.-M.; Calderaro, J.; Jeng, Y.-M.; Hsiao, L.-P.; Lacroix, L.; Bougoüin, A.; et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature 2020, 577, 556–560. [Google Scholar] [CrossRef]
- Helmink, B.A.; Reddy, S.M.; Gao, J.; Zhang, S.; Basar, R.; Thakur, R.; Yizhak, K.; Sade-Feldman, M.; Blando, J.; Han, G.; et al. B cells and tertiary lymphoid structures promote immunotherapy response. Nature 2020, 577, 549–555. [Google Scholar] [CrossRef]
- Cabrita, R.; Lauss, M.; Sanna, A.; Donia, M.; Larsen, M.S.; Mitra, S.; Johansson, I.; Phung, B.; Harbst, K.; Vallon-Christersson. J. Nature. 2020, 577, 561–565. [Google Scholar]
- Hori, S.; Nomura, T.; Sakaguchi, S. Control of Regulatory T Cell Development by the Transcription Factor Foxp3. Science 2003, 299, 1057–1061. [Google Scholar] [CrossRef]
- Turner, J.A.; Stephen-Victor, E.; Wang, S.; Rivas, M.N.; Abdel-Gadir, A.; Harb, H.; Cui, Y.; Fanny, M. ; Charbonnier, L-M. ; Fong, J.J.H.; et al. Regulatory T cell-derived TGF-β1 controls multiple checkpoints governing allergy and autoimmunity. Immunity. 2020, 53, 1202–1214e6. [Google Scholar] [CrossRef]
- Miyara, M.; Sakaguchi, S. Natural regulatory T cells: mechanisms of suppression. Trends Mol. Med. 2007, 13, 108–116. [Google Scholar] [CrossRef]
- Nagai, H.; Horikawa, T.; Hara, I.; Fukunaga, A.; Oniki, S.; Oka, M.; Nishigori, C.; Ichihashi, M. In vivo elimination of CD25+ regulatory T cells leads to tumor rejection of B16F10 melanoma, when combined with interleukin-12 gene transfer. Exp. Dermatol. 2004, 13, 613–620. [Google Scholar] [CrossRef]
- Dannull, J.; Su, Z.; Rizzieri, D.; Yang, B.K.; Coleman, D.; Yancey, D.; Zhang, A.; Dahm, P.; Chao, N.; Gilboa, E.; et al. Enhancement of vaccine-mediated antitumor immunity in cancer patients after depletion of regulatory T cells. J. Clin. Investig. 2005, 115, 3623–3633. [Google Scholar] [CrossRef]
- Wing, K.; Onishi, Y.; Martin, P.P.; Tamaguchi, T.; Miyara, M.; Fehervari, Z.; Nomura, T.; Sakaguchi, S. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008, 322, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Smyth, M.J.; Hayakawa, Y.; Takeda, K.; Yagita, H. New aspects of natural-killer-cell surveillance and therapy of cancer. Nat. Rev. Cancer 2002, 211, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Campos-Martín, Y.; del Moral, M.G.; Gozalbo-López, B.; Suela, J.; Martínez-Naves, E. Expression of Human CD1d Molecules Protects Target Cells from NK Cell-Mediated Cytolysis. J. Immunol. 2004, 172, 7297–7305. [Google Scholar] [CrossRef]
- Bauer, S.; Groh, V.; Wu, J.; Steinle, A.; Phillips, J.H.; Lanier, L.L.; Spies, T. Activation of NK Cells and T Cells by NKG2D, a Receptor for Stress-Inducible MICA. Science 1999, 285, 727–729. [Google Scholar] [CrossRef]
- Armeanu, S.; Bitzer, M.; Lauer, U.M.; Venturelli, S.; Pathil, A.; Krusch, M.; Kaiser, S.; Jobst, J.; Smirnow, I.; Wagner, A.; et al. Natural Killer Cell–Mediated Lysis of Hepatoma Cells via Specific Induction of NKG2D Ligands by the Histone Deacetylase Inhibitor Sodium Valproate. Cancer Res. 2005, 65, 6321–6329. [Google Scholar] [CrossRef]
- Skov, S.; Pedersen, M.T.; Andresen, L.; Straten, P.T.; Woetmann, A.; Ødum, N. Cancer Cells Become Susceptible to Natural Killer Cell Killing after Exposure to Histone Deacetylase Inhibitors Due to Glycogen Synthase Kinase-3–Dependent Expression of MHC Class I–Related Chain A and B. Cancer Res. 2005, 65, 11136–11145. [Google Scholar] [CrossRef]
- Khong, H.T.; Restifo, N.P. Natural selection of tumor variants in the generation of ‘tumor escape’ phenotypes. Nat Immunol. 2002, 3, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Moretta, L.; Bottino, C.; Pende, D.; Vitale, M.; Mingari, M.; Moretta, A. Different checkpoints in human NK activation. Trends Immunol. 2004, 25, 670–676. [Google Scholar] [CrossRef]
- Baba, T.; Hanagiri, T.; Ichiki, Y.; Kuroda, K.; Shigematsu, Y.; Mizukami, M.; Sugaya, M.; Takenoyama, M.; Sugio, K.; Yasumoto, K. Lack and restoration of sensitivity of lung cancer cells to cellular attack with special reference to expression of human leukocyte antigen class I and/or major histocompatibility complex class I chain related molecules A/B. Cancer Sci. 2007, 98, 1795–1802. [Google Scholar] [CrossRef] [PubMed]
- Shimasaki, N.; Jain, A.; Campana, D. NK cells for cancer immunotherapy. Nat. Rev. Drug Discov. 2020, 19, 200–218. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, D.I.; Le Nours, J.; Andrews, D.M.; Uldrich, A.P.; Rossjohn, J. Unconventional T Cell Targets for Cancer Immunotherapy. Immunity 2018, 48, 453–473. [Google Scholar] [CrossRef]
- Fujii, S.; Shimizu, K.; Kronenberg, M.; Steinman, R.M. Prolonged IFN-gamma-producing NKT response induced with alpha-galactosylceramide-loaded DCs. Nat Immunol. 2002, 3, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Fujii, S.-I.; Shimizu, K.; Smith, C.; Bonifaz, L.; Steinman, R.M. Activation of Natural Killer T Cells by α-Galactosylceramide Rapidly Induces the Full Maturation of Dendritic Cells In Vivo and Thereby Acts as an Adjuvant for Combined CD4 and CD8 T Cell Immunity to a Coadministered Protein. J. Exp. Med. 2003, 198, 267–279. [Google Scholar] [CrossRef]
- Shimizu, K.; Kurosawa, Y.; Taniguchi, M.; Steinman, R.M.; Fujii, S.-i. Cross-presentation of glycolipid from tumor cells loaded with α-galactosylceramide leads to potent and long-lived T cell–mediated immunity via dendritic cells. J. Exp. Med. 2007, 204, 2641–2653. [Google Scholar] [CrossRef]
- Hayday, A. Gammadelta T cells and the lymphoid stress-surveillance response. Immunity. 2009, 31, 184–196. [Google Scholar] [CrossRef]
- Nakajima, J.; Murakawa, T.; Fukami, T.; Goto, S.; Kaneko, T.; Yoshida, Y.; Takamoto, S.; Kakimi, K. A phase I study of adoptive immunotherapy for recurrent non-small-cell cancer patients with autologous gammadelta T cells. Eur J Cardiothorac Surg. 2010, 37, 1191–1197. [Google Scholar] [CrossRef]
- Sakamoto, M. ; J. ; Nakajima, J.; Murakawa, T.; Fukami, M.; Yoshida, Y.; Murayama, T.; Takamoto, S.; Matsushita, H.; Kakimi, K. Adoptive immunotherapy for advanced non-small cell lung cancer using zoledronate-expanded γδT cells: aphase I clinical study. Immunother. 2011, 34, 202–211. [Google Scholar] [CrossRef]
- Jin, C.; Lagoudas, G.K.; Zhao, C.; Bullman, S.; Bhutkar, A.; Hu, B.; Ameh, S.; Sandel, D.; Liang, X.S.; Mazzilli, S.; Whary, M.T.; et al. Commensal microbiota promote lung cancer development via γδ T cell. Cell. 2019, 176, 998–1013. [Google Scholar] [CrossRef] [PubMed]
- Noy, R.; Pollard, J.W. Tumor-associated macrophages: From mechanisms to therapy. Immunity 2014, 41, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Sica, A. Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr. Opin. Immunol. 2010, 22, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Wang, J.; Hu, Y.; Sun, Y.; Yang, X.; Zhang, B.; Deng, Y.; Wei, W.; Zhang, Z.; Wen, L.; et al. Irradiated tumour cell-derived microparticles upregulate MHC-I expression in cancer cells via DNA double-strand break repair pathway. Cancer Lett. 2024, 592, 216898. [Google Scholar] [CrossRef]
- Wan, C.; Sun, Y.; Tian, Y.; Lu, L.; Dai, X.; Meng, J.; Huang, J.; He, Q.; Wu, B.; Zhang, Z.; et al. Irradiated tumor cell–derived microparticles mediate tumor eradication via cell killing and immune reprogramming. Sci. Adv. 2020, 6, eaay9789. [Google Scholar] [CrossRef]
- Goc, J.; Germain, C.; Bourgais, T.K.D.; Lupo, A.; Klein, C.; Knockaert, S.; Chaisemartin, L.; Ouakrim, H.; Becht, E.; Alifano, M. Dendritic cells in tumor-associated tertial lymphoid structures signal a Th1 cytotoxic immune contexture and license the positive prognostic value of infiltrating CD8+ T cell. Cancer Res. 2014, 74, 705–715. [Google Scholar] [CrossRef]
- Dutertre, C.A.; Wang, L.F. ; Ginhoux. Aligning bona fide dendritic cell populationa cross species. Cell Immunol. 2014, 291, 3–10. [Google Scholar] [CrossRef]
- Haniffa, M.; Collin, M.; Ginhoux, F. Ontogeny and Functional Specialization of Dendritic Cells in Human and Mouse. Adv. Immunol. 2013, 120, 1–49. [Google Scholar] [CrossRef]
- Lavin, Y.; Kobayashi, S.; Leader, A.; Amir, E.D.; Elefant, N.; Bigenwald, C.; Remark, R.; Sweeney, R.; Becker, C.D.; Levine, J.H.; et al. Innate Immune Landscape in Early Lung Adenocarcinoma by Paired Single-Cell Analyses. Cell 2017, 169, 750–765. [Google Scholar] [CrossRef]
- Bossche, J.V.D.; De Laere, M.; Deschepper, K.; Germonpré, P.; Valcke, Y.; Lamont, J.; Stein, B.; Van Camp, K.; Germonpré, C.; Nijs, G.; et al. Integration of the PD-L1 inhibitor atezolizumab and WT1/DC vaccination into standard-of-care first-line treatment for patients with epithelioid malignant pleural mesothelioma—Protocol of the Immuno-MESODEC study. PLOS ONE 2024, 19, e0307204. [Google Scholar] [CrossRef]
- Land, F.R.v. .; Willemsen, M.; Bezemer, K.; van der Burg, S.H.; Bosch, T.P.v.D.; Doukas, M.; Fellah, A.; Kolijn, P.M.; Langerak, A.W.; Moskie, M.; et al. Dendritic Cell–Based Immunotherapy in Patients With Resected Pancreatic Cancer. J. Clin. Oncol. 2024, 42, 3083–3093. [Google Scholar] [CrossRef]
- Veglia, F.; Perego, M.; Gabrilovich, D. Myeloid-derived suppressor cells coming of age. Nat. Immunol. 2018, 19, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Hengesbach, L.M.; Hoag, K.A. Physiological Concentrations of Retinoic Acid Favor Myeloid Dendritic Cell Development over Granulocyte Development in Cultures of Bone Marrow Cells from Mice. J. Nutr. 2004, 134, 2653–2659. [Google Scholar] [CrossRef]
- Nefedova, Y.; Nagaraj, S.; Rosenbauer, A.; Muro-Cacho, C.; Sebti, S. M.; Gabrilovich, D. I. Regulation of dendritic cell differentiation and antitumor immune response in cancer by pharmacologic-selective inhibition of the Janus-activated kinase 2/signal transducers and activators of transcription 3 pathway. Cancer Research. 2005, 65, 9525–9535. [Google Scholar] [CrossRef]
- Kortylewski, M.; Kujawski, M.; Wang, T.; Wei, S.; Zhang, S.; Pilon-Thomas, S.; Niu, G.; Kay, H.; Mulé, J.; Kerr, W.G.; et al. Inhibiting Stat3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nat. Med. 2005, 11, 1314–1321. [Google Scholar] [CrossRef]
- Suzuki, E.; Kapoor, V.; Jassar, A.S.; Kaiser, L.R.; Albelda, S.M. ; Gemcitabine selectively eliminates splenic Gr-1+ / CD11b+ myeloid suppressor cells in tumor-bearing animals and enhances antitumor immune activity. Clin Cancer Res. 2005, 15, 6713–6721. [Google Scholar] [CrossRef]
- Pan, P.-Y.; Ma, G.; Weber, K.J.; Ozao-Choy, J.; Wang, G.; Yin, B.; Divino, C.M.; Chen, S.-H. Immune Stimulatory Receptor CD40 Is Required for T-Cell Suppression and T Regulatory Cell Activation Mediated by Myeloid-Derived Suppressor Cells in Cancer. Cancer Res. 2010, 70, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Aruga, A.; Aruga, E.; Cameron, M.J.; E Chang, A. Different cytokine profiles released by CD4+ and CD8+ tumor-draining lymph node cells involved in mediating tumor regression. J. Leukoc. Biol. 1997, 61, 507–516. [Google Scholar] [CrossRef]
- Uribe-Querol, E.; Rosales, C. Neutrophils in Cancer: Two Sides of the Same Coin. J. Immunol. Res. 2015, 2015, 1–21. [Google Scholar] [CrossRef]
- Canli. ; Nicolas, A.M.; Gupta, J.; Finkelmeier, F.; Goncharova, O.; Pesic, M.; Neumann, T.; Horst, D.; Löwer, M.; Sahin, U.; et al. Myeloid Cell-Derived Reactive Oxygen Species Induce Epithelial Mutagenesis. Cancer Cell 2017, 32, 869–883. [Google Scholar] [CrossRef] [PubMed]
- Houghton, A.M.; Rzymkiewicz, D.M.; Ji, H.; Gregory, A.D.; Egea, E.E.; Metz, H.E.; Stolz, D.B.; Land, S.R.; Marconcini, L.A.; Kliment, C.R.; et al. Neutrophil elastase-mediated degradation of IRS-1 accelerates lung tumor growth. Nat. Med. 2010, 16, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Nozawa, H.; Chiu, C.; Hanahan, D. Infiltrating neutrophils mediate the initial angiogenic switch in a mouse model of multistage carcinogenesis. Proc. Natl. Acad. Sci. 2006, 103, 12493–12498. [Google Scholar] [CrossRef]
- Kaltenmeier, C.; Simmons, R.L.; Tohme, S.; Yazdani, H.O. Neutrophil Extracellular Traps (NETs) in Cancer Metastasis. Cancers 2021, 13, 6131. [Google Scholar] [CrossRef]
- Diem, S.; Schmid, S.; Krapf, M.; Flatz, L.; Born, D.; Jochum, W.; Templeton, A.J.; Früh, M. Neutrophil-to-Lymphocyte ratio (NLR) and Platelet-to-Lymphocyte ratio (PLR) as prognostic markers in patients with non-small cell lung cancer (NSCLC) treated with nivolumab. Lung Cancer 2017, 111, 176–181. [Google Scholar] [CrossRef]
- Tanizaki, J.; Haratani, K.; Hayashi, H.; Chiba, Y.; Nakamura, Y.; Yonesaka, K.; Kudo, K.; Kaneda, H.; Hasegawa, Y.; Tanaka, K.; et al. Peripheral Blood Biomarkers Associated with Clinical Outcome in Non–Small Cell Lung Cancer Patients Treated with Nivolumab. J. Thorac. Oncol. 2017, 13, 97–105. [Google Scholar] [CrossRef]
- Ilie, M.; Hofman, V.; Ortholan, C.; Bonnetaud, C.; Coëlle, C.; Mouroux, J.; Hofman, P. Predictive clinical outcome of the intra-tumoral CD66b-positive neutrophil-to-CD8-positive T-cell ratio in patients with resectable nonsmall cell lung cancer. Cancer 2012, 118, 1726–1737. [Google Scholar] [CrossRef] [PubMed]
- Panciera, T.; Azzolin, L.; Cordenonsi, M.; Piccolo, S. Mechanobiology of YAP and TAZ in physiology and disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 758–770. [Google Scholar] [CrossRef]
- Plaks, V.; Kong, N.; Werb, Z. The Cancer Stem Cell Niche: How Essential Is the Niche in Regulating Stemness of Tumor Cells? Cell Stem Cell 2015, 16, 225–238. [Google Scholar] [CrossRef]
- Vermeulen, L.; Felipe De Sousa, E.M.; Van Der Heijden, M.; Cameron, K.; De Jong, J.H.; Borovski, T.; Tuynman, J.B.; Todaro, M.; Merz, C.; Rodermond, H.; et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat. Cell Biol. 2010, 12, 468–476. [Google Scholar] [CrossRef]
- Calvo, F.; Ege, N.; Grande-Garcia, A.; Hooper, S.; Jenkins, R.P.; Chaudhry, S.I.; Harrington, K.; Williamson, P.; Moeendarbary, E.; Charras, G.; et al. Mechanotransduction and YAP-dependent matrix remodelling is required for the generation and maintenance of cancer-associated fibroblasts. Nat. Cell Biol. 2013, 15, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Junttila, M.R.; de Sauvage, F.J. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature 2013, 501, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Gilkes, D.M.; Bajpai, S.; Chaturvedi, P.; Wirtz, D.; Semenza, G.L. Hypoxia-inducible factor (HIF-1) promotes extracellular matrix remodeling under hypoxic conditions by inducing P4HA1, P4HA2 and PLOD2 expression in fibroblasts. J Biol Chem, 2013, 288, 10819–10829. [Google Scholar] [CrossRef]
- Cords, L.; Engler, A.; Heberecker, M.; Ruschoff, J.H.; Moch, H.; Souza, N.; Bodenmiller, B. Cancer-associated fibroblast phenotypes are associated with patient outcome in non-small cell lung cancer. Cancer Cell. 2024, 42, 396–412. [Google Scholar] [CrossRef]
- Tan, E.; Gakhar, N.; Kirtane, K. TCR gene-engineered cell therapy for solid tumors. Best Pr. Res. Clin. Haematol. 2021, 34, 101285. [Google Scholar] [CrossRef] [PubMed]
- Sterner, R.C.; Sterner, R.M. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 2021, 11, 69. [Google Scholar] [CrossRef]
- Zhong, S.; Cui, Y.; Liu, Q.; Chen, S. CAR-T cell therapy for lung cancer: a promising but challenging future. J. Thorac. Dis. 2020, 12, 4516–4521. [Google Scholar] [CrossRef]
- Liang, T.; Wang, B.; Li, J.; Liu, Y. LINC00922 accelerates the proliferation, migration and invasion of lung cancer via the miRNA-204/CXCR4 Axis. Med Sci Monit. 2019, 25, 5075–5086. [Google Scholar] [CrossRef]
- Gangadhar, T.; Nandi, S.; Salgia, R. The role of chemokine receptor CXCR4 in lung cancer. Cancer Biol. Ther. 2010, 9, 409–416. [Google Scholar] [CrossRef]
- Mao, Y.; Fan, W.; Hu, H.; Zhang, L.; Michel, J.; Wu, Y.; Wang, J.; Jia, L.; Tang, X.; Chen, Y.; et al. MAGE-A1 in lung adenocarcinoma as a promising target of chimeric antigen receptor T cells. J. Hematol. Oncol. 2019, 12, 1–18. [Google Scholar] [CrossRef]
- Leidner, R.; Silva, N.S.; Huang, H.; Sprott, D.; Zheng, C.; Shih, Y.-P.; Leung, A.; Payne, R.; Sutcliffe, K.; Cramer, J.; et al. Neoantigen T-Cell Receptor Gene Therapy in Pancreatic Cancer. New Engl. J. Med. 2022, 386, 2112–2119. [Google Scholar] [CrossRef] [PubMed]
- Robbins, P.F.; Morgan, R.A.; Feldman, S.A.; Yang, J.C.; Sherry, R.M.; Dudley, M.E.; Wunderlich, J.R.; Nahvi, A.V.; Helman, L.J.; Mackall, C.L.; et al. Tumor Regression in Patients With Metastatic Synovial Cell Sarcoma and Melanoma Using Genetically Engineered Lymphocytes Reactive With NY-ESO-1. J. Clin. Oncol. 2011, 29, 917–924. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



