Submitted:
14 September 2024
Posted:
16 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Tetrahymena Thermophila Culture and Treatments
2.2. RNA Extraction
2.3. cDNA Synthesis
2.4. Quantification of Relative mRNA Abundances of Mtt Genes
| NCBI ID | Sequence (5’-3’) | Oligo Type |
|---|---|---|
| AF537326-Mtt1 | FAM-TGC TGC ACA GAC CCT AAC AGC GGA-BHQ1 | Dual Labeled Probes |
| AY204351-Mtt2 | HEX-TGC TTG CAA TTG CAA ACC TTG CGA-BHQ1 | Dual Labeled Probes |
| AY884209-Mtt5 | RED-CTC ACT AGG GCA GCA GCA CCA CTT-BHQ2 | Dual Labeled Probes |
| M10932-17s | CY5-CCG CAG GTT CCA CTT CTG GTG TGC-BHQ2 | Dual Labeled Probes |
| AF537326-Mtt1F | TGC TGT GGT GTA AAT GCT AAG CC | Forward PCR Primer |
| AF537326-Mtt1R | TCC TGT ACC AGT GCA ACA TCC CT | Reverse PCR Primer |
| AY204351-Mtt2/4F | ACC TCT CTG CAA ATG TGG AAC TAC | Forward PCR Primer |
| AY204351-Mtt2/4R | CCA CAG CTT TCA GTA ACA CCA CAT | Reverse PCR Primer |
| AY884209-Mtt5F | TCT GGT GAA AGC ACT AAA ATT TGT | Forward PCR Primer |
| AY884209-Mtt5R | ATC AGA ATT GCA GCA ATT TTG GG | Reverse PCR Primer |
| M10932-17sF | CGA TCA GAT ACC GTC GTA GTC TTA | Forward PCR Primer |
| M10932-17sR | GAG TTT CCC CGT GTT GAG TCA | Reverse PCR Primer |
Statistics
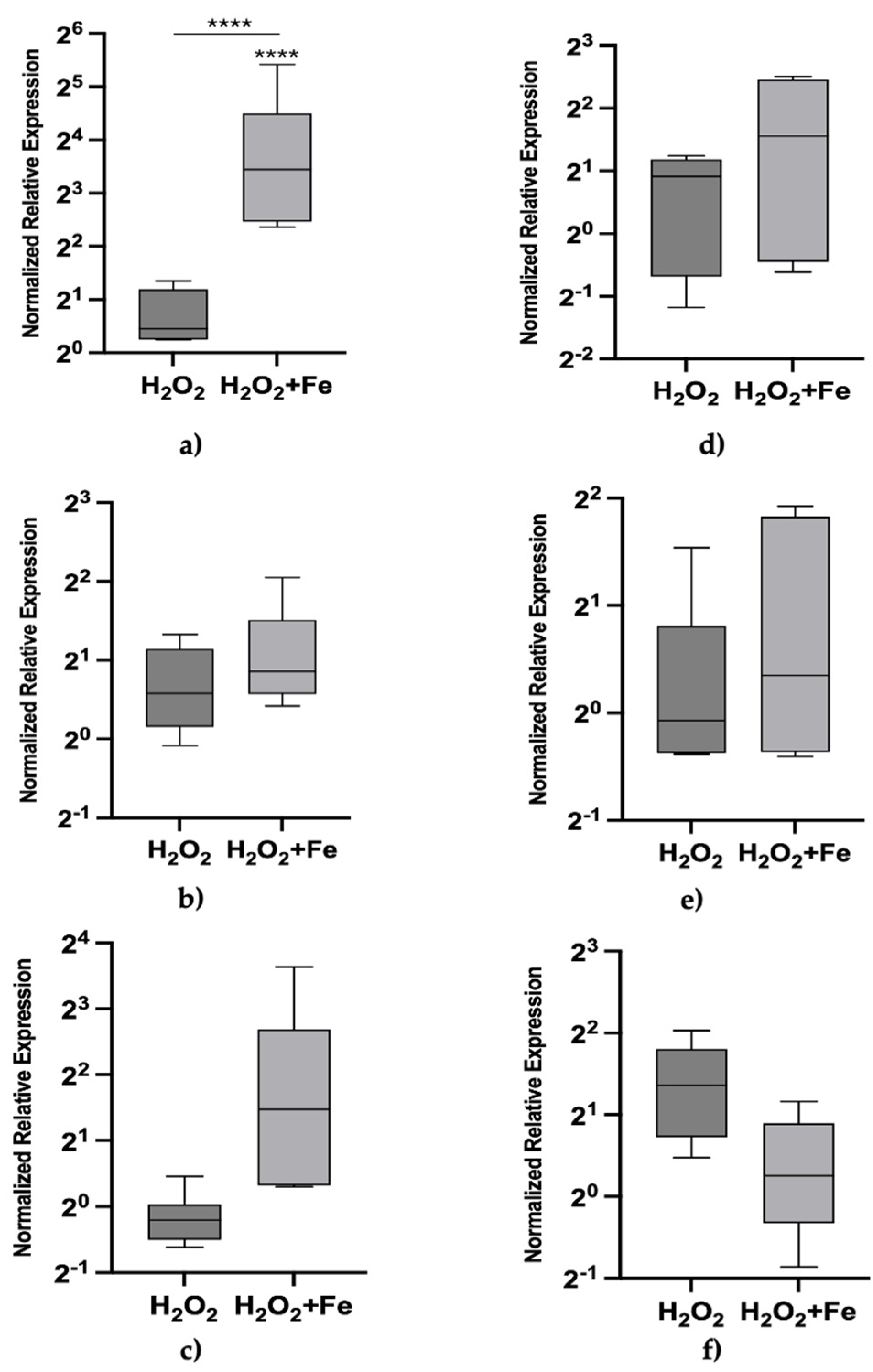
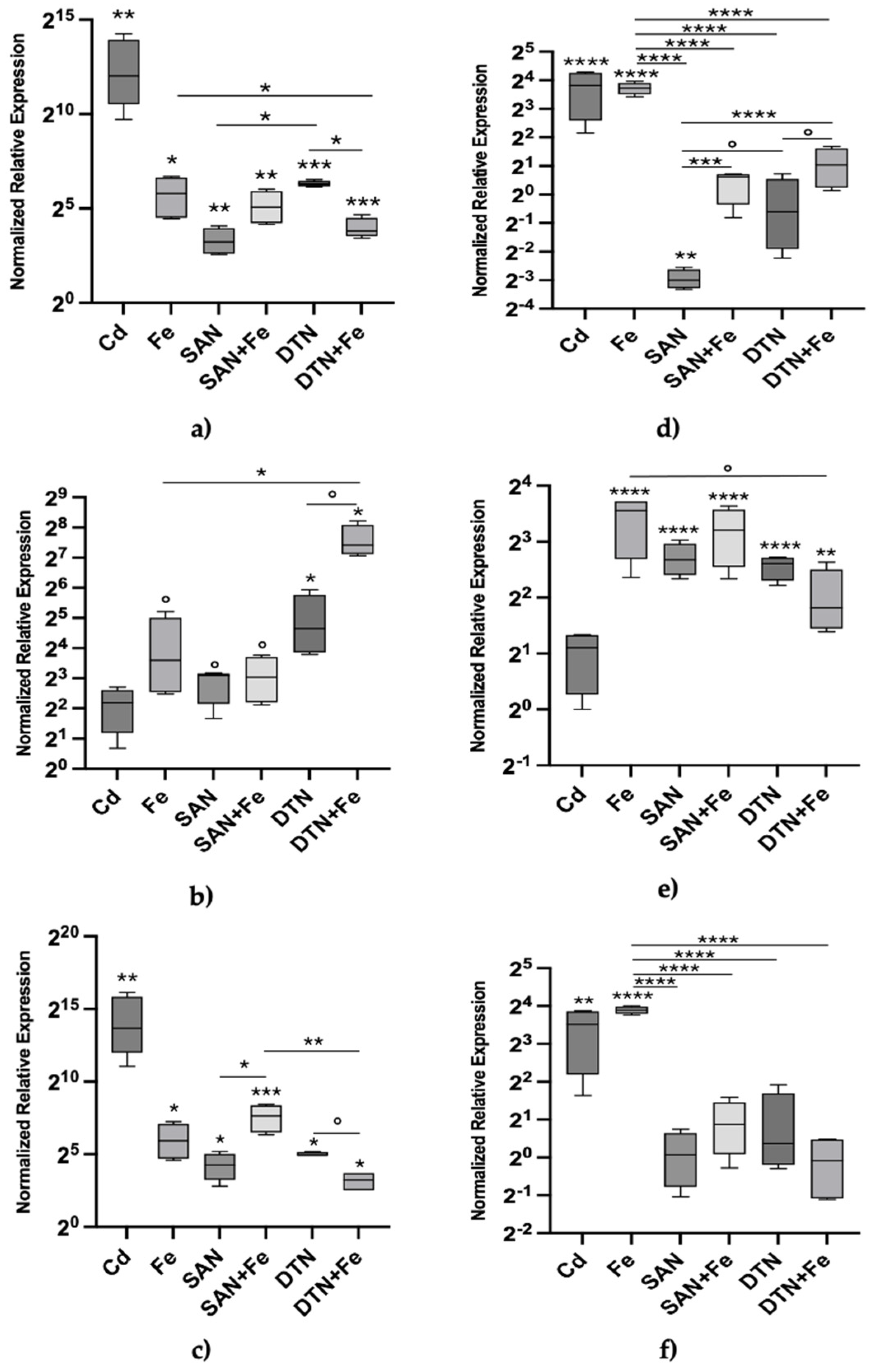
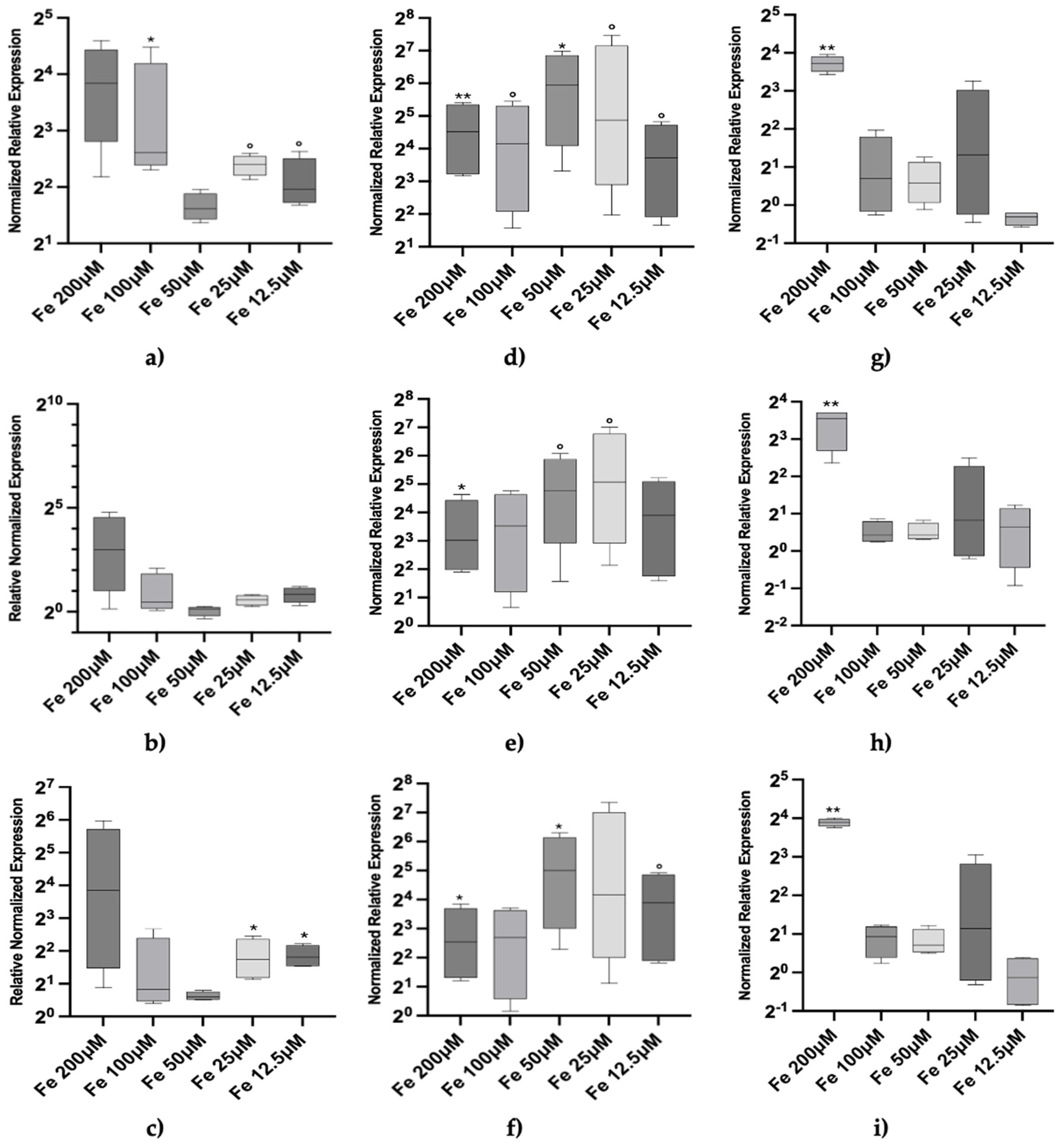
3. Results
| Treatment | NOEC |
|---|---|
| Sodium azide (SAN) | 200 μM |
| Sodium diethyldithiocarbamate (DTN) | 100 μM |
| Cadmium chloride (Cd) | 20 μM |
| Ferric chloride (Fe) | 200 μM |
| Hydrogen peroxide (H2O2) | 100 μM |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Capdevila, M., Atrian S. Metallothionein protein evolution: a miniassay. J Biol Inorg Chem. 2011 Oct;16(7):977-89. [CrossRef]
- Blindauer, C.A. Bacterial metallothioneins: past, present, and questions for the future. J Biol Inorg Chem. 2011 Oct;16(7):1011-24. [CrossRef]
- Drozd A., Wojewska D., Peris-Díaz M.D., Jakimowicz P., Krężel A., Crosstalk of the structural and zinc buffering properties of mammalian metallothionein-2, Metallomics, Volume 10, Issue 4, April 2018, Pages 595–613. [CrossRef]
- Vallee, B.L., Falchuk K.H., The biochemical basis of zinc physiology. Physiol Rev. 1993 Jan;73(1):79-118. [CrossRef]
- Kägi, J.H., Kojima Y. Chemistry and biochemistry of metallothionein. Experientia Suppl. 1987; 52:25-61. [CrossRef]
- Hunziker, P.E., and Kagi J.H.R., Metaltothionein. In: Metalloproteins: part 2. Metal Proteins with Non-redox roles. Harrison, P. (Edtor), pp. 149-181, Verlag Chemie, Basel, Switzerland, 1985.
- Palmiter, R.D. The elusive function of metallothioneins. Proc Natl Acad Sci U S A. 1998 Jul 21;95(15):8428-30. [CrossRef]
- Egli, D., Domènech J., Selvaraj A., Balamurugan K., Hua H., Capdevila M., Georgiev O., Schaffner W., Atrian S. The four members of the Drosophila metallothionein family exhibit distinct yet overlapping roles in heavy metal homeostasis and detoxification. Genes Cells. 2006 Jun;11(6):647-58. [CrossRef]
- Coyle, P., Philcox J.C., Carey L.C., Rofe A.M. Metallothionein: the multipurpose protein. Cell Mol Life Sci. 2002 Apr;59(4):627-47. [CrossRef]
- De, S.K., McMaster M.T., Andrews G.K. Endotoxin induction of murine metallothionein gene expression. J Biol Chem. 1990 Sep 5;265(25):15267-74. [PubMed]
- Thornalley, P.J., Vasák M. Possible role for metallothionein in protection against radiation-induced oxidative stress. Kinetics and mechanism of its reaction with superoxide and hydroxyl radicals. Biochim Biophys Acta. 1985 Jan 21;827(1):36-44. [CrossRef]
- Hanada, K., Baba T., Hashimoto I., Fukui R., Watanabe S. Possible role of cutaneous metallothionein in protection against photo-oxidative stress--epidermal localization and scavenging activity for superoxide and hydroxyl radicals. Photodermatol Photoimmunol Photomed. 1992-1993 Oct;9(5):209-13. [PubMed]
- Bauman, J.W., Madhu C., McKim J.M. Jr, Liu Y., Klaassen C.D. Induction of hepatic metallothionein by paraquat. Toxicol Appl Pharmacol. 1992 Dec;117(2):233-41. [CrossRef]
- Palmiter, R.D. Molecular biology of metallothionein gene expression. Experientia Suppl. 1987;52:63-80. [CrossRef]
- Díaz, S., Amaro F., Rico D., Campos V., Benítez L., Martín-González A., Hamilton E.P., Orias E., Gutiérrez J.C. Tetrahymena metallothioneins fall into two discrete subfamilies. PLoS One. 2007 Mar 14;2(3):e291. [CrossRef] [PubMed]
- Espart, A., Marín M., Gil-Moreno S., Palacios Ò., Amaro F., Martín-González A., Gutiérrez J.C., Capdevila M., Atrian S. Hints for metal-preference protein sequence determinants: different metal binding features of the five tetrahymena thermophila metallothioneins. Int J Biol Sci. 2015 Mar 18;11(4):456-71. [CrossRef] [PubMed]
- de Francisco P, Martín-González A, Turkewitz AP, Gutiérrez JC. Extreme metal adapted, knockout and knockdown strains reveal a coordinated gene expression among different Tetrahymena thermophila metallothionein isoforms. PLoS One. 2017 Dec 5;12(12):e0189076. [CrossRef]
- Shang, Y., Song X., Bowen J., Corstanje R., Gao Y., Gaertig J., Gorovsky M.A. A robust inducible-repressible promoter greatly facilitates gene knockouts, conditional expression, and overexpression of homologous and heterologous genes in Tetrahymena thermophila. Proc Natl Acad Sci U S A. 2002 Mar 19;99(6):3734-9. [CrossRef]
- Boldrin, F., Santovito G., Gaertig J., Wloga D., Cassidy-Hanley D., Clark T.G., Piccinni E. Metallothionein gene from Tetrahymena thermophila with a copper-inducible-repressible promoter. Eukaryot Cell. 2006 Feb;5(2):422-5. [CrossRef]
- Formigari, A., Boldrin F., Santovito G., Cassidy-Hanley D., Clark T.G., Piccinni E. Functional characterization of the 5’-upstream region of MTT5 metallothionein gene from Tetrahymena thermophila. Protist. 2010 Jan;161(1):71-7. [CrossRef]
- de Francisco, P., Amaro F., Martín-González A., Gutiérrez J.C. AP-1 (bZIP) Transcription Factors as Potential Regulators of Metallothionein Gene Expression in Tetrahymena thermophila. Front Genet. 2018 Oct 23;9:459. [CrossRef]
- Kling, P., Modig C., Mujahed H., Khalaf H., von Hofsten J., Olsson P.E. Differential regulation of the rainbow trout (Oncorhynchus mykiss) MT-A gene by nuclear factor interleukin-6 and activator protein-1. BMC Mol Biol. 2013 Dec 17;14:28. [CrossRef]
- Dalton, T., Palmiter R.D., Andrews G.K. Transcriptional induction of the mouse metallothionein-I gene in hydrogen peroxide-treated Hepa cells involves a composite major late transcription factor/antioxidant response element and metal response promoter elements. Nucleic Acids Res. 1994 Nov 25;22(23):5016-23. [CrossRef]
- Dalton, T.P., Li Q., Bittel D., Liang L., Andrews G.K. Oxidative stress activates metal-responsive transcription factor-1 binding activity. Occupancy in vivo of metal response elements in the metallothionein-I gene promoter. J Biol Chem. 1996 Oct 18;271(42):26233-41. [CrossRef]
- Fenton, H.J.H. Oxidation of tartaric acid in presence of iron. J Chem Soc, 1894 65, 899-910. [CrossRef]
- Haber F., andWeiss J. The catalytic decomposition of hydrogen peroxide by iron salts Proc. R. Soc. Lond.1934 A147332–35. [CrossRef]
- Kienzle, N., Young D., Zehntner S., Bushell G., Sculley T.B. DNaseI treatment is a prerequisite for the amplification of cDNA from episomal-based genes. Biotechniques. 1996 Apr;20(4):612-6. [CrossRef]
- Shi, X., Liu X., Wang Q., Das A., Ma G., Xu L., Sun Q., Peddireddi L., Jia W., Liu Y., Anderson G., Bai J., Shi J. A multiplex real-time PCR panel assay for simultaneous detection and differentiation of 12 common swine viruses. J Virol Methods. 2016 Oct;236:258-265. [CrossRef]
- Bustin, S.A., Benes V., Garson J.A., Hellemans J., Huggett J., Kubista M., Mueller R., Nolan T., Pfaffl M.W., Shipley G.L., Vandesompele J., Wittwer C.T. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem. 2009 Apr;55(4):611-22. [CrossRef]
- Sanchis-Segura, C., Miquel M., Correa M., Aragon C.M. The catalase inhibitor sodium azide reduces ethanol-induced locomotor activity. Alcohol. 1999 Aug;19(1):37-42. [CrossRef]
- Heikkila R.E., Cabbat F.S., Cohen G. In vivo inhibition of superoxide dismutase in mice by diethyldithiocarbamate. J Biol Chem. 1976 doi:10;251(7):2182-5.
- Zuo, Y., Hu J., Xu X., Gao X., Wang Y., Zhu S. Sodium azide induces mitochondria-mediated apoptosis in PC12 cells through Pgc-1α-associated signaling pathway. Mol Med Rep. 2019 Mar;19(3):2211-2219. [CrossRef]
- de Freitas Oliveira J.W., Torres T.M., Moreno C.J.G., Amorim-Carmo B., Damasceno I.Z., Soares A.K.M.C., da Silva Barbosa J., Rocha H.A.O., Silva M.S. Insights of antiparasitic activity of sodium diethyldithiocarbamate against different strains of Trypanosoma cruzi. Sci Rep. 2021 May 27;11(1):11200. [CrossRef]
- Gebicka, L., Krych-Madej J. The role of catalases in the prevention/promotion of oxidative stress. J Inorg Biochem. 2019 Aug;197:110699. [CrossRef]
- Wang, Y., Branicky R., Noë A., Hekimi S. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol. 2018 Jun 4;217(6):1915-1928. [CrossRef]
- Karin, M., Liu Zg, Zandi E. AP-1 function and regulation. Curr Opin Cell Biol. 1997 Apr;9(2):240-6. [CrossRef]
- Valko, M., Morris H., Cronin M.T. Metals, toxicity and oxidative stress. Curr Med Chem. 2005;12(10):1161-208. [CrossRef]
- Chanarat, S., Svasti J. Stress-induced upregulation of the ubiquitin-relative Hub1 modulates pre-mRNA splicing and facilitates cadmium tolerance in Saccharomyces cerevisiae. Biochim Biophys Acta Mol Cell Res. 2020 Feb;1867(2):118565. [CrossRef]
- Sornaraj, P., Luang S., Lopato S., Hrmova M. Basic leucine zipper (bZIP) transcription factors involved in abiotic stresses: A molecular model of a wheat bZIP factor and implications of its structure in function. Biochim Biophys Acta. 2016;1860(1PtA):46-56. [CrossRef]
- Amaro, F., González D., Gutiérrez J.C. MicroRNAs in Tetrahymena thermophila: An epigenetic regulatory mechanism in the response to cadmium stress. Microbiol Res. 2024 Mar;280:127565. [CrossRef]
- Dondero, F., Cavaletto M., Ghezzi A.R., La Terza A., Banni M., Viarengo A. Biochemical characterization and quantitative gene expression analysis of the multi-stress inducible metallothionein from Tetrahymena thermophila. Protist. 2004 Jun;155(2):157-68. [CrossRef]
- Guo, R., Ma H., Gao F., Zhong L., Ren J. Metallothionein alleviates oxidative stress-induced endoplasmic reticulum stress and myocardial dysfunction. J Mol Cell Cardiol. 2009 Aug;47(2):228-37. [CrossRef]
- Martínez-Pastor, M.T., Perea-García A., Puig S. Mechanisms of iron sensing and regulation in the yeast Saccharomyces cerevisiae. World J Microbiol Biotechnol. 2017 Apr;33(4):75. [CrossRef]
- Askwith, C., Eide D., Van Ho A., Bernard P.S., Li L., Davis-Kaplan S., Sipe D.M., Kaplan J. The FET3 gene of S. cerevisiae encodes a multicopper oxidase required for ferrous iron uptake. Cell. 1994 Jan 28;76(2):403-10. [CrossRef]
- Mach, J., Sutak R. Iron in parasitic protists - from uptake to storage and where we can interfere. Metallomics. 2020 Sep 23;12(9):1335-1347. [CrossRef]
- Farnaud, S., Rapisarda C., Bui T., Drake A., Cammack R., Evans R.W. Identification of an iron-hepcidin complex. Biochem J. 2008 Aug 1;413(3):553-7. [CrossRef]
- Nemeth, E., Tuttle M.S., Powelson J., Vaughn M.B., Donovan A., Ward D.M., Ganz T., Kaplan J. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science. 2004 Dec 17;306(5704):2090-3. [CrossRef]
- Laity, J.H., Andrews G.K. Understanding the mechanisms of zinc-sensing by metal-response element binding transcription factor-1 (MTF-1). Arch Biochem Biophys. 2007 Jul 15;463(2):201-10. [CrossRef]
- Greenberg, J.T., Demple B. A global response induced in Escherichia coli by redox-cycling agents overlaps with that induced by peroxide stress. J Bacteriol. 1989 Jul;171(7):3933-9. [CrossRef]
- Li, H., Mapolelo D.T., Dingra N.N., Naik S.G., Lees N.S., Hoffman B.M., Riggs-Gelasco P.J., Huynh B.H., Johnson M.K., Outten C.E. The yeast iron regulatory proteins Grx3/4 and Fra2 form heterodimeric complexes containing a [2Fe-2S] cluster with cysteinyl and histidyl ligation. Biochemistry. 2009 Oct 13;48(40):9569-81. [CrossRef]
- Bell, S.G., Vallee B.L. The metallothionein/thionein system: an oxidoreductive metabolic zinc link. Chembiochem. 2009 Jan 5;10(1):55-62. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




