Submitted:
13 September 2024
Posted:
14 September 2024
You are already at the latest version
Abstract
Keywords:
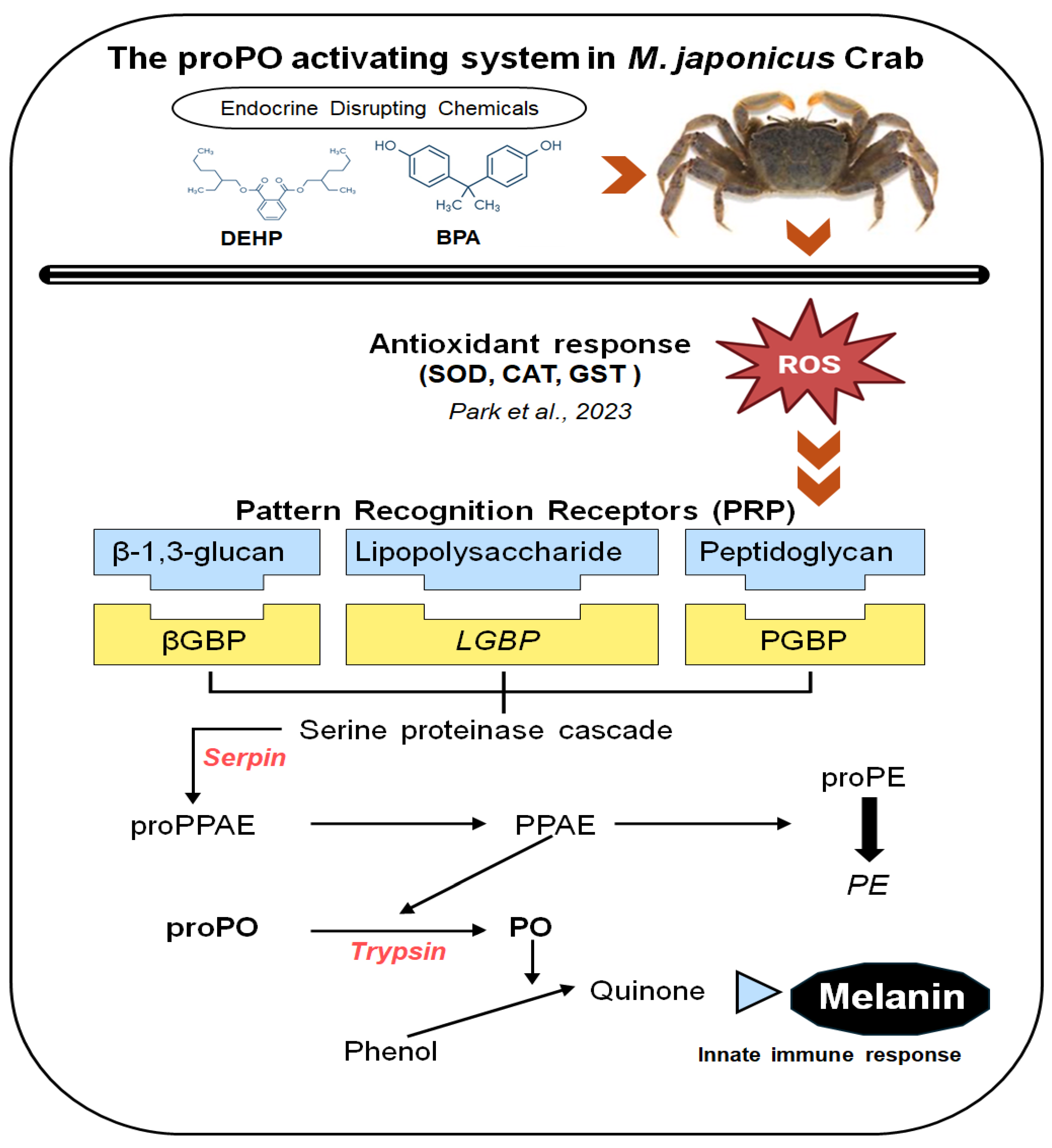
1. Introduction
2. Materials and Methods
2.1. Test Organism
2.2. Chemicals, Reagents, and Exposure Experiment Design
2.3. Total RNA Extraction and cDNA Synthesis
2.4. Gene Expression Quantification in M. japonicus
2.5. Data Analysis
3. Results
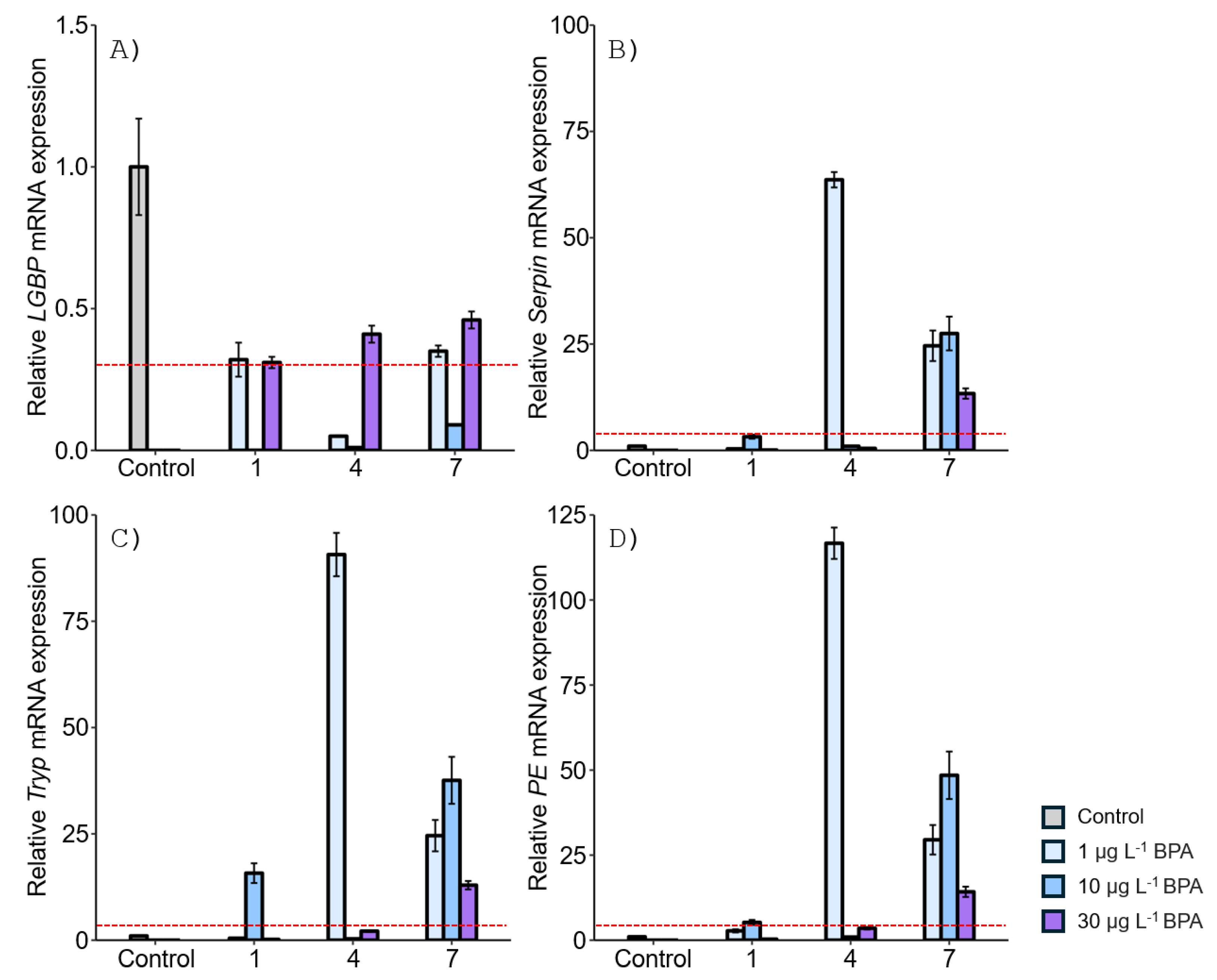
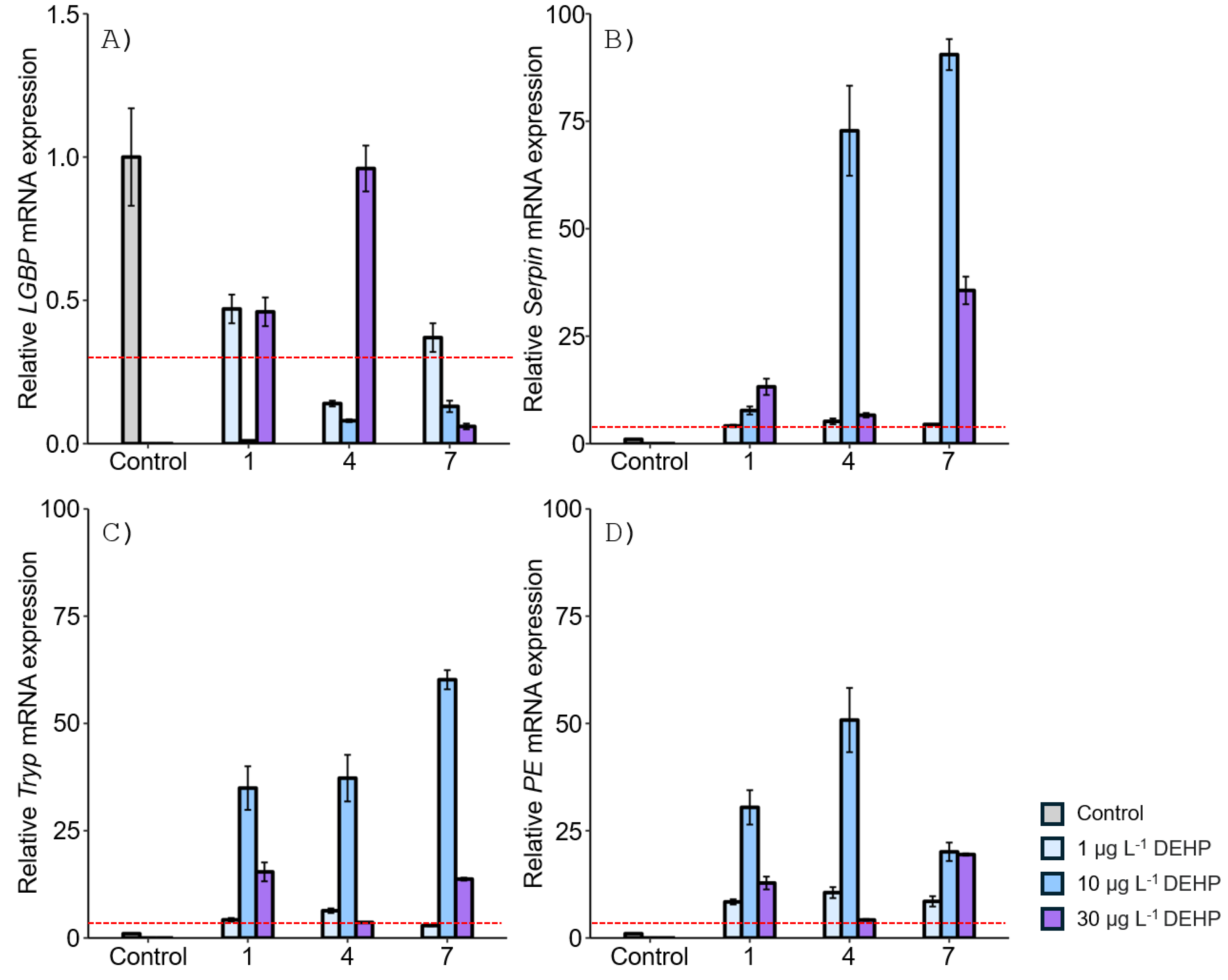
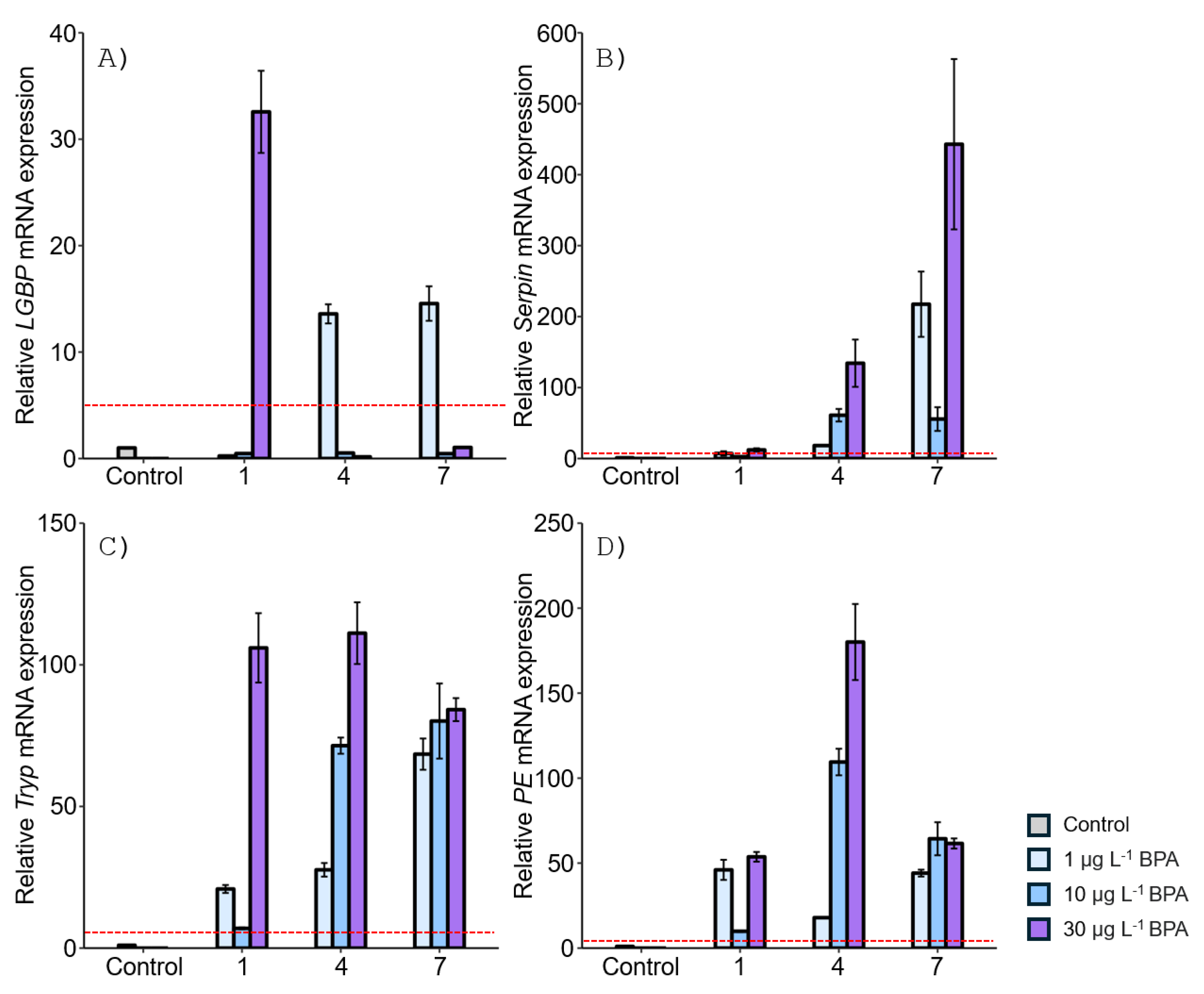
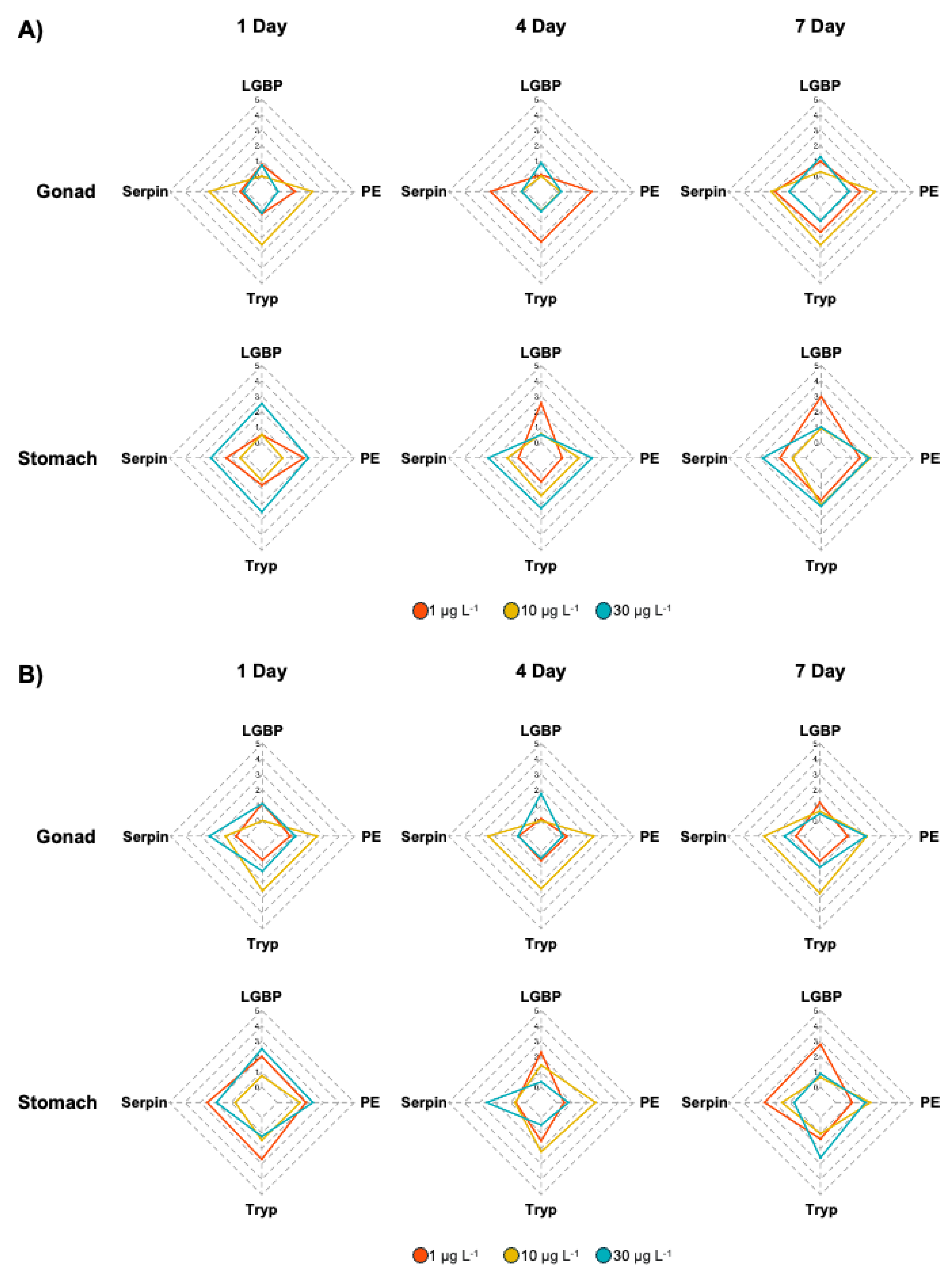
3.1. Expression of proPO System-Related Genes in Gonads after Exposure to EDCs
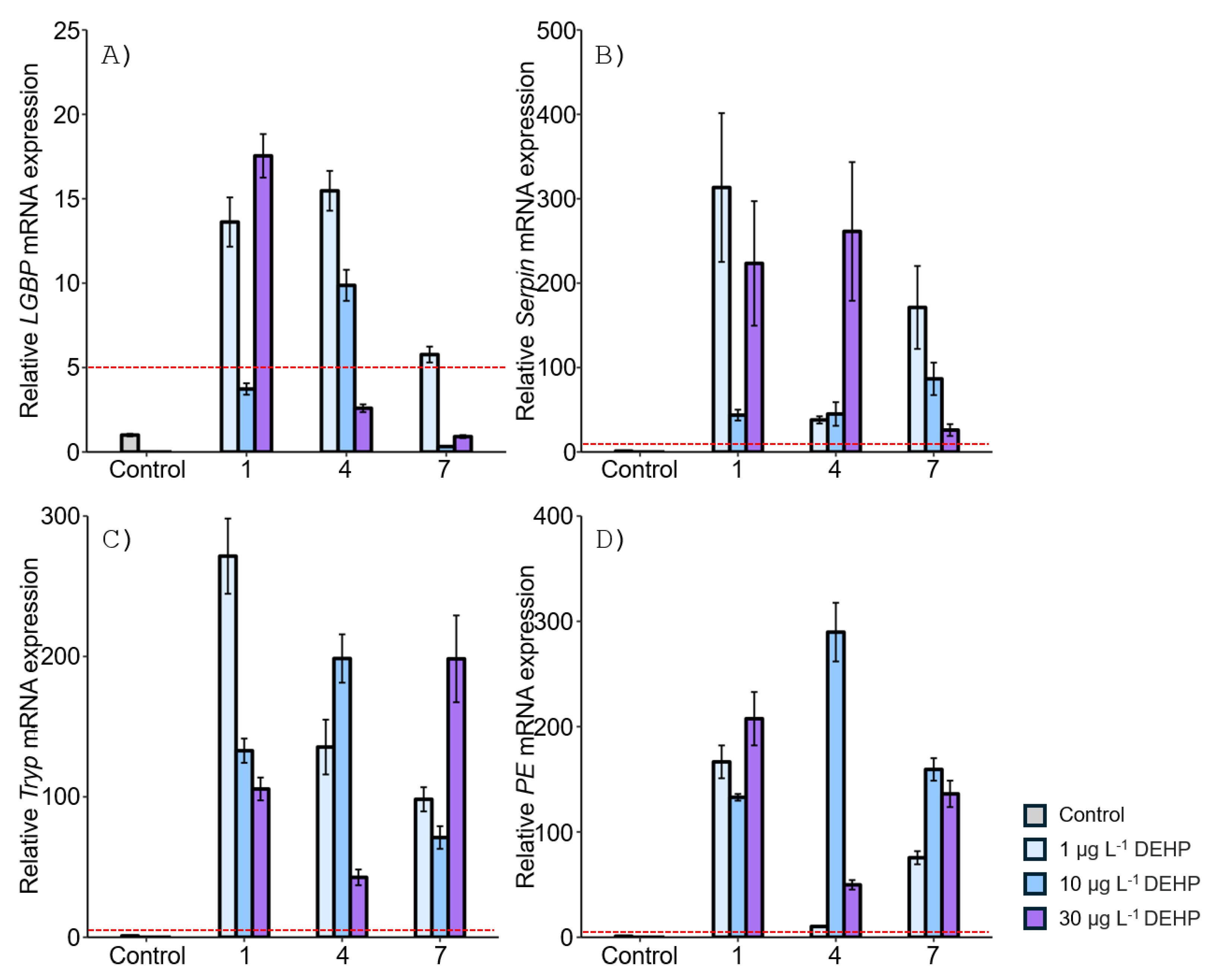
3.2. Expression of proPO System-Related Genes in the Stomach after EDC Exposure
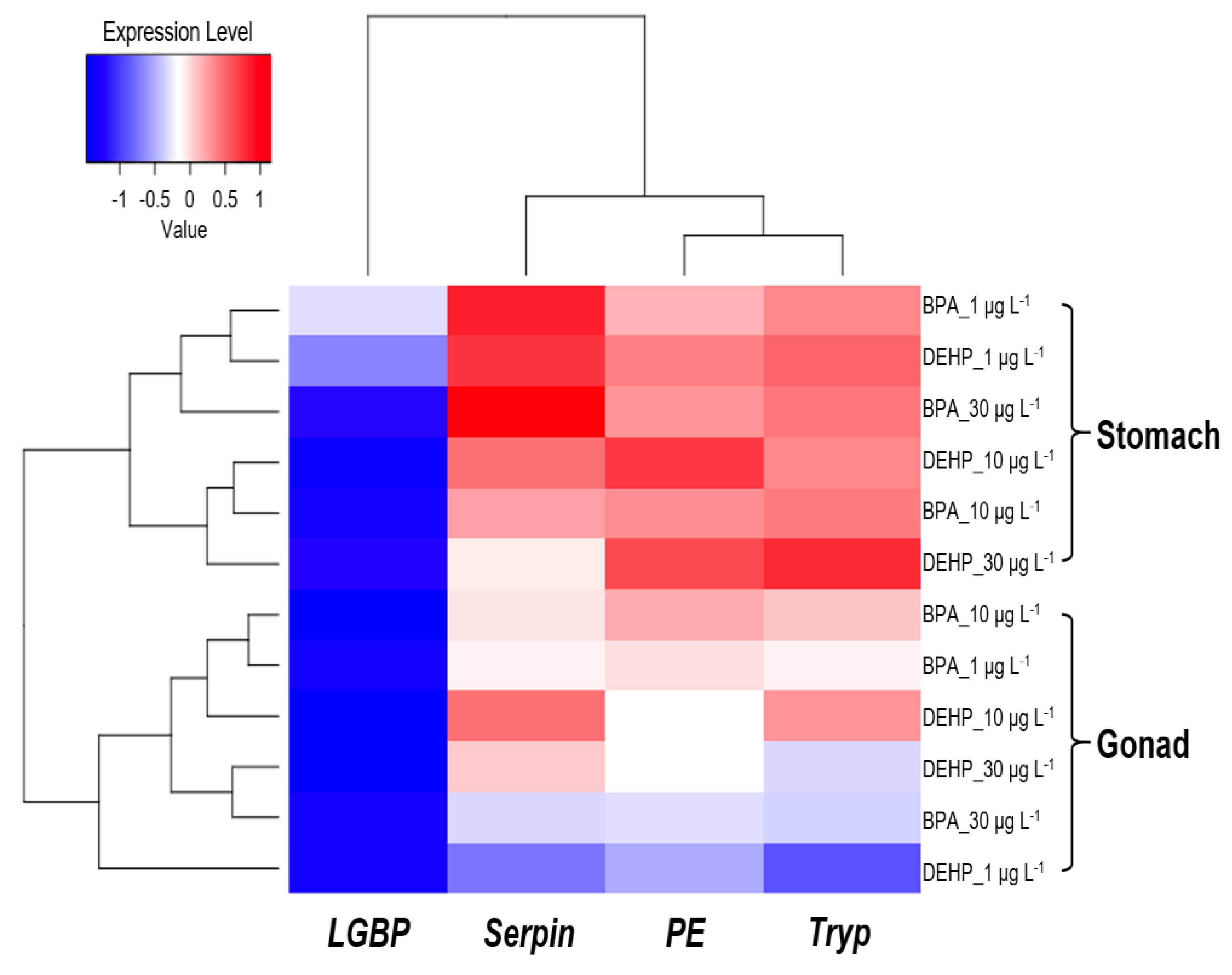
3.3. Integrated Biomarker Response (IBR) Index and Heatmap Analysis on M. japonicus Exposed to BPA and DEHP
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alrumman, S.; Keshk, S.; El Kott, A. Water pollution: source & treatment. Am. J. Environ. Enginner. 2016, 88–98. [Google Scholar]
- Morin-Crini, N.; Lichtfouse, E.; Liu, G.; Balaram, V.; Ribeiro, A.R.L.; Lu, Z.; Stock, F.; Carmona, E.; Teixeira, M.R.; Picos-Corrales, L.A.; Moreno-Piraján, J.C.; Giraldo, L.; Li, C.; Pandey, A.; Hocquet, D.; Torri, G.; Crini, G. Worldwide cases of water pollution by emerging contaminants: a review. Environ. Chem. Lett. 2022, 20, 2311–2338. [Google Scholar] [CrossRef]
- Vos, J.G.; Dybing, E.; Greim, H.A.; Ladefoged, O.; Lambré, C.; Tarazona, J.V.; Brandt, I.; Vethaal, A.D. Health effects of endocrine-disrupting chemicals on wildlife, with special reference to the European situation. Crit. Rev. Toxicol. 2000, 30, 71–133. [Google Scholar] [CrossRef]
- Darbre, P.D. The history of endocrine-disrupting chemicals. Curr. Opin. Endocr. Metab. Res. 2019, 7, 26–33. [Google Scholar] [CrossRef]
- Chen, D.; Kannan, K.; Tan, H.; Zheng, Z.; Feng, Y.L.; Wu, Y.; Widelka, M. Bisphenol analogues other than BPA: Environmental occurrence, human exposure, and toxicity – a review. Environ. Sci. Technol. 2016, 50, 5438–5453. [Google Scholar] [CrossRef]
- Lin, C.; Lee, C.J.; Mao, W.M.; Nadim, F. Identifying the potential sources of di-(2-ethylhexyl) phthalate contamination in the sediment of the Houjing River in southern Taiwan. J. Hazard. Mater. 2009, 161, 270–275. [Google Scholar] [CrossRef]
- Rowdhwal, S.S.S.; Chen, J. Toxic Effects of Di-2-ethylhexyl Phthalate: An Overview. Biomed. Res. Int. 2018, 2018, 1750368. [Google Scholar] [CrossRef]
- Park, K.; Kim, W.S.; Kwak, I.S. Endocrine-disrupting chemicals impair the innate immune T prophenoloxidase system in the intertidal mud crab, Macrophthalmus japonicus. Fish. Shellfish. Immunol. 2019, 87, 322–332. [Google Scholar] [CrossRef]
- Wetherill, Y.B.; Akingbemi, B.T.; Kanno, J.; McLachlan, J.A.; Nadal, A.; Sonnenschein, C.; Watson, C.S.; Zoeller, R.T.; Belcher, S.M. In vitro molecular mechanisms of bisphenol A action. Reprod. Toxicol. 2007, 24, 178–198. [Google Scholar] [CrossRef]
- Valentino, R.; D’Esposito, V.; Ariemma, F.; Cimmino, I.; Beguinot, F.; Formisano, P. Bisphenol A environmental exposure and the detrimental effects on human metabolic health: is it necessary to revise the risk assessment in vulnerable population? J. Endocrinol. Invest. 2016, 39, 259–263. [Google Scholar] [CrossRef]
- Seoane, M.; Cid, Á.; Esperanza, M. Toxicity of bisphenol A on marine microalgae: Single- and multispecies bioassays based on equivalent initial cell biovolume. Sci. Total. Environ. 2021, 767, 144363. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Li, X.; Li, H.; Yuan, S.; Li, J.; Liu, C. Greater toxic potency of bisphenol AF than bisphenol A in growth, reproduction, and transcription of genes in Daphnia magna. Environ. Sci. Pollut. Res. Int. 2021, 28, 25218–25227. [Google Scholar] [CrossRef]
- Yin, X.; Zeb, R.; Wei, H.; Cai, L. Acute exposure of di(2-ethylhexyl) phthalate (DEHP) induces immune signal regulation and ferroptosis in oryzias melastigma. Chemosphere. 2021, 265, 129053. [Google Scholar] [CrossRef]
- Rogers, J.A.; Metz, L.; Yong, V.W. Review: Endocrine disrupting chemicals and immune responses: A focus on bisphenol-A and its potential mechanisms. Mol. Immunol. 2013, 53, 421–430. [Google Scholar] [CrossRef]
- Huang, R.G.; Li, X.B.; Wang, Y.Y.; Wu, H.; Li, K.D.; Jin, X.; Du, Y.J.; Wang, H.; Qian, F.Y.; Li, B.Z. Endocrine-disrupting chemicals and autoimmune diseases. Environ. Res. 2023, 231, 116222. [Google Scholar] [CrossRef]
- Vivier, E.; Malissen, B. Innate and adaptive immunity: specificities and signaling hierarchies revisited. Nat. Immunol. 2005, 6, 17–21. [Google Scholar] [CrossRef]
- Amparyup, P.; Charoensapsri, W.; Tassanakajon, A. Prophenoloxidase system and its role in shrimp immune responses against major pathogens. Fish. Shellfish. Immunol. 2013, 34, 990–1001. [Google Scholar] [CrossRef] [PubMed]
- Binggeli, O.; Neyen, C.; Poidevin, M.; Lemaitre, B. Prophenoloxidase Activation Is Required for Survival to Microbial Infections in Drosophila. PLoS. Pathog. 2014, 10, e1004067. [Google Scholar] [CrossRef] [PubMed]
- Cerenius, L.; Söderhäll, K. ; The prophenoloxidase-activating system in invertebrates. Immunol. Rev. 2004, 198, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Falfushynska, H.; Delwig, O.; Piontkivska, H.; Sokolova, I.M. Interactive effects of salinity variation and exposure to ZnO nanoparticles on the innate immune system of a sentinel marine bivalve, Mytilus edulis. Sci. Total. Environ. 2020, 712, 136473. [Google Scholar] [CrossRef]
- Wang, Q.; Zhou, X.; Jin, Q.; Zhu, F. Effects of the aquatic pollutant sulfamethoxazole on the innate immunity and antioxidant capacity of the mud crab Scylla paramamosain. Chemosphere. 2024, 349, 140775. [Google Scholar] [CrossRef] [PubMed]
- Kitaura, J.; Nishida, M.; Wada, K. Genetic and behavioral diversity in the Macrophthalmus japonicus species complex (Crustacea: Brachyura: Ocypodidae). Mar. Biol. 2002, 140, 1–8. [Google Scholar]
- Henmi, Y. Life-history patterns in two forms of Macrophthalmus japonicus (Crustacea: Brachyura). Mar. Biol. 1989, 101, 53–60. [Google Scholar] [CrossRef]
- Davis, J.A.; Wille, M.; Hecht, T.; Sorgeloos, P. Optimal first feed organism for South African mud crab Scylla serrata (Forskål) larvae. Aquac. Int. 2005, 13, 187–201. [Google Scholar] [CrossRef]
- Mohapatra, A.; Mohanty, R.K.; Mohanty, S.K.; Bhatta, K.S.; Das, N.R. Fisheries enhancement and biodiversity assessment of fish, prawn and mud crab in Chilika lagoon through hydrological intervention. Wetl. Ecol. Manag. 2007, 15, 229–251. [Google Scholar] [CrossRef]
- Leoville, A.; Lagarde, R.; Grondin, H.; Faivre, L.; Rasoanirina, E.; Teichert, N. Influence of environmental conditions on the distribution of burrows of the mud crab, Scylla serrata, in a fringing mangrove ecosystem. Reg. Stud. Mar. Sci. 2021, 43, 101684. [Google Scholar] [CrossRef]
- Hsieh, S.L.; Hsieh, S.; Xu, R.Q.; Chen, Y.T.; Chen, C.W.; Singhania, R.R.; Chen, Y.C.; Tsai, T.H.; Dong, C.D. Toxicological effects of polystyrene nanoplastics on marine organisms on marine organisms. Environ. Technol. Innov. 2023, 30, 103073. [Google Scholar] [CrossRef]
- Yifei, Y.; Zhixiong, Z.; Luna, C.; Qihui, C.; Zuoyuan, W.; Xinqi, L.; Zhexiang, L.; Fei, Z.; Xiujuan, Z. Marine pollutant Phenanthrene (PHE) exposure causes immunosuppression of hemocytes in crustacean species, Scylla paramamosain. Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 2024, 275, 109761. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔct method. Methods. 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Avellan, A.; Duarte, A.; Rocha-Santos, T. Organic contaminants in marine sediments and seawater: A review for drawing environmental diagnostics and searching for informative predictors. Sci. Total. Environ. 2022, 808, 152012. [Google Scholar] [CrossRef]
- Neves, R.A.; Miralha, A.; Guimarães, T.B.; Sorrentino, R.; Calderari, M.R.; Santos, L.N. Phthalates contamination in the coastal and marine sediments of Rio de Janeiro, Brazil. Mar. Pollut. Bull. 2023, 190, 114819. [Google Scholar] [CrossRef] [PubMed]
- Rolfo, A.; Nuzzo, A.M.; De Amicis, R.; Moretti, L.; Bertoli, S.; Leone, A. Fetal-Maternal Exposure to Endocrine Disruptors: Correlation with Diet Intake and Pregnancy Outcomes. Nutrients. 2020, 12, 1744. [Google Scholar] [CrossRef] [PubMed]
- Hamid, N.; Junaid, M.; Pei, D.S. Combined toxicity of endocrine-disrupting chemicals: A review. Ecotoxicol. Environ. Saf. 2021, 215, 112136. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Flaws, J.A.; Spinella, M.J.; Irudayaraj, J. The relationship between typical environmental endocrine disruptors and kidney disease. Toxics. 2022, 11, 32. [Google Scholar] [CrossRef]
- Pu, C.; Liu, Y.; Ma, J.; Li, J.; Sun, R.; Zhou, Y.; Zhang, C. The effects of bisphenol S exposure on the growth, physiological and biochemical indices, and ecdysteroid receptor gene expression in red swamp crayfish, Procambarus clarkii. Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 2024, 276, 109811. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.S.; Park, K.; Kim, J.H.; Kwak, I.S. Effect of endocrine-disrupting chemicals on the expression of a calcium ion channel receptor (ryanodine receptor) in the mud crab (Macrophthalmus japonicus). Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 2024, 283, 109972. [Google Scholar] [CrossRef]
- Wang, S.; Cao, Y.; Wang, S.; Cai, J.; Zhang, Z. DEHP induces immunosuppression through disturbing inflammatory factors T and CYPs system homeostasis in common carp neutrophils. Fish. Shellfish. Immunol. 2020, 96, 26–31. [Google Scholar] [CrossRef]
- Han, Y.; Shi, W.; Tang, Y.; Zhou, W.; Sun, H.; Zhang, J.; Yan, M.; Hu, L.; Liu, G. Microplastics and bisphenol A hamper gonadal development of whiteleg shrimp (Litopenaeus vannamei) by interfering with metabolism and disrupting hormone regulation. Sci. Total. Environ. 2022, 810, 152354. [Google Scholar] [CrossRef]
- Montero, V.; Chinchilla, Y.; Gómez, L.; Flores, A.; Medaglia, A.; Guillén, R.; Montero, E. Human health risk assessment for consumption of microplastics and plasticizing substances through marine species. Environ. Res. 2023, 237, 116843. [Google Scholar] [CrossRef]
- Park, K.; Kim, W.S.; Choi, B.; Kwak, I.S. Expression levels of the immune-related p38 mitogen-activated protein kinase transcript in response to environmental pollutants on Macrophthalmus japonicus crab. Genes. 2020, 11, 958. [Google Scholar] [CrossRef]
- Kim, W.S.; Kwak, I.S. EDCs trigger immune-neurotransmitter related gene expression, and cause histological damage in sensitive mud crab Macrophthalmus japonicus gills and hepatopancreas. Fish. Shellfish. Immunol. 2022, 122, 484–494. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, T.; Mukai, K. Innate immunity signalling and membrane trafficking. Curr. Opin. Cell. Biol. 2019, 59, 1–7. [Google Scholar] [CrossRef] [PubMed]
- De Winther, M.P.; Palaga, T. Epigenetic Regulation of Innate Immunity. Front. Immunol. 2021, 12, 713758. [Google Scholar] [CrossRef] [PubMed]
| Gene | Primer Sequence (5’-3’) | Amplicon size (bp) | Efficiency (%) | Accession number |
| LGBP_F | AATGGCTTCTTCCCTGACGG | 131 | 100.0 | KJ653260 |
| LGBP_R | CTGATCTTGCCCTCACCCTG | |||
| Serpin_F | TTTGGAACGTGGGAGTATGC | 74 | 93.0 | MH41109 |
| Serpin_R | TGCACATTGGGAATCGCATG | |||
| Tryp_F | CCTAGAGGTCGGGGTCAAGA | 91 | 99.5 | KJ653261 |
| Tryp_R | CCTATCCAGCTCGAGCAGTG | |||
| PE_F | CTGACCACCATACACACGCT | 98 | 90.0 | KF804082 |
| PE_R | TGGAACACTTGCTCGTCCTG | |||
| GAPDH_F | TGCTGATGCACCCATGTTTG | 147 | 102.5 | KJ653265 |
| GAPDH_R | AGGCCCTGGACAATCTCAAAG |
| Chemical | Organ | Concentration (μg/L) | proPO system related gene | IBR value | ||||
| LGBP | Serpin | Tryp | PE | Mean | ||||
| BPA | Gonad | Control | 2.67 | 0.00 | 0.15 | 0.20 | 0.75 | 2.45 |
| 1 | 0.96 | 1.96 | 1.65 | 1.60 | 1.54 | 1.17 | ||
| 10 | 0.29 | 2.20 | 2.48 | 2.53 | 1.87 | 4.31 | ||
| 30 | 1.25 | 1.03 | 0.91 | 0.85 | 1.01 | 2.61 | ||
| Stomach | Control | 1.00 | 0.58 | 0 | 0.05 | 0.41 | 0.46 | |
| 1 | 2.98 | 1.67 | 1.74 | 1.52 | 1.98 | 1.38 | ||
| 10 | 0.92 | 0.85 | 2.04 | 2.22 | 1.51 | 3.90 | ||
| 30 | 1.01 | 2.81 | 2.14 | 2.12 | 2.02 | 4.24 | ||
| DEHP | Gonad | Control | 2.65 | 0.46 | 0.56 | 0 | 0.92 | 2.58 |
| 1 | 1.17 | 0.54 | 0.63 | 0.82 | 0.79 | 1.35 | ||
| 10 | 0.63 | 2.62 | 2.69 | 2.08 | 2.00 | 1.93 | ||
| 30 | 0.45 | 1.29 | 1.02 | 2.01 | 1.19 | 3.78 | ||
| Stomach | Control | 0.90 | 0.37 | 0.18 | 0 | 0.37 | 0.34 | |
| 1 | 2.79 | 2.62 | 1.37 | 1.05 | 1.96 | 1.31 | ||
| 10 | 0.64 | 1.50 | 1.04 | 2.24 | 1.35 | 3.40 | ||
| 30 | 0.87 | 0.70 | 2.60 | 1.91 | 1.52 | 2.97 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




