Submitted:
12 September 2024
Posted:
12 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
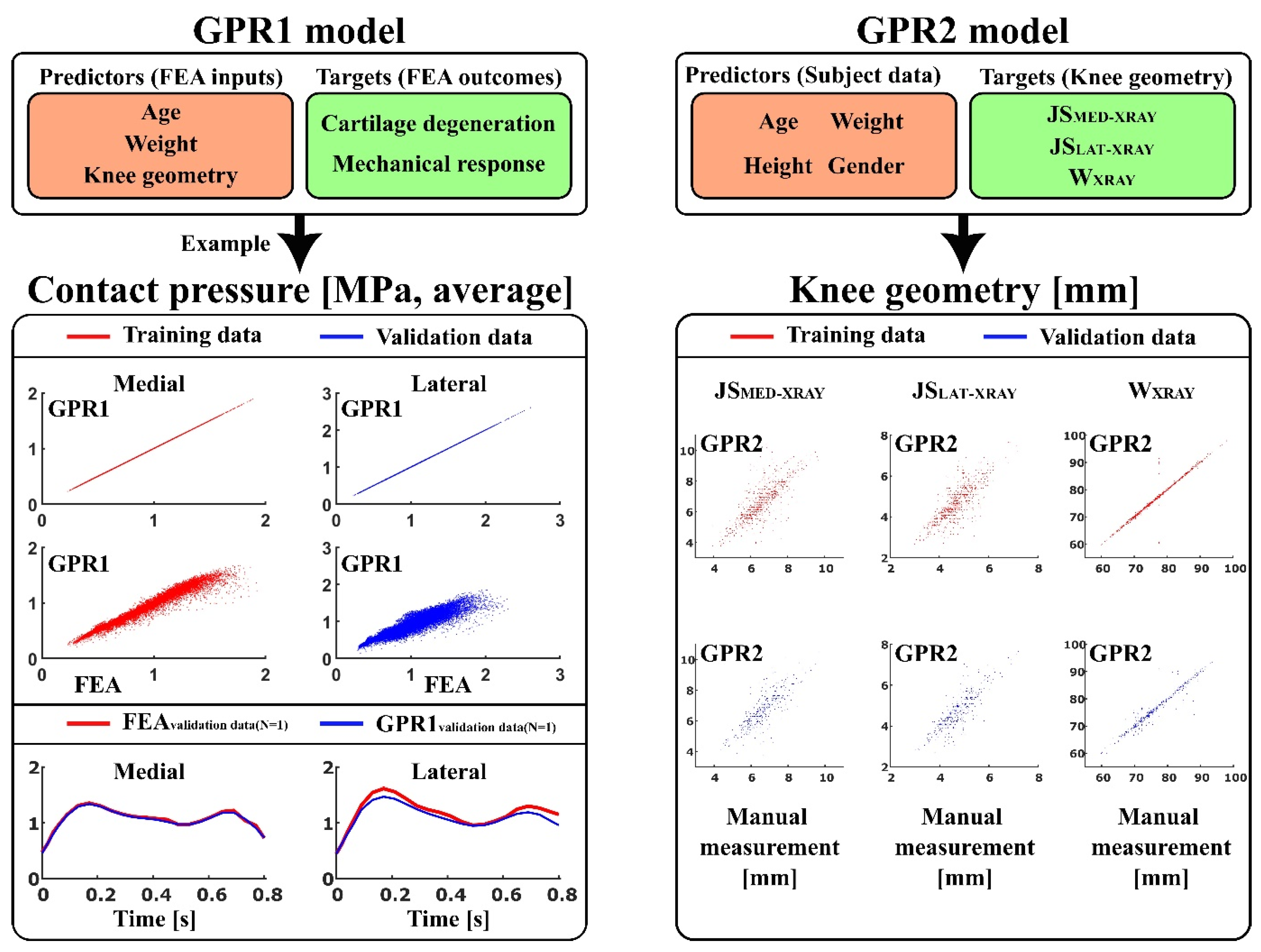
2.1. Workflow
- GPR1 - to predict atlas-based FEA method [17] outcomes: contact area, contact pressure, tensile stress, tensile strain, shear stress, shear strain, and pore pressure (peaks and averages over the tibiofemoral contact area), as well as simulated cartilage degeneration using age, weight, and anatomical knee joint dimensions: lateral joint space (JSLAT-MRI), medial joint space (JSMED-MRI), maximum lateral anterior-posterior dimension (APLAT-MRI), maximum medial anterior-posterior dimensions (APMED-MRI), and condyle distance (CDMRI), as a set of predictor variables.
- GPR2 - to predict anatomical knee joint dimensions: lateral joint space (JSLAT-XRAY), medial joint space (JSMED-XRAY), and full medial-lateral width of the distal femur (WXRAY) using age, weight, height, and gender as a set of predictor variables.
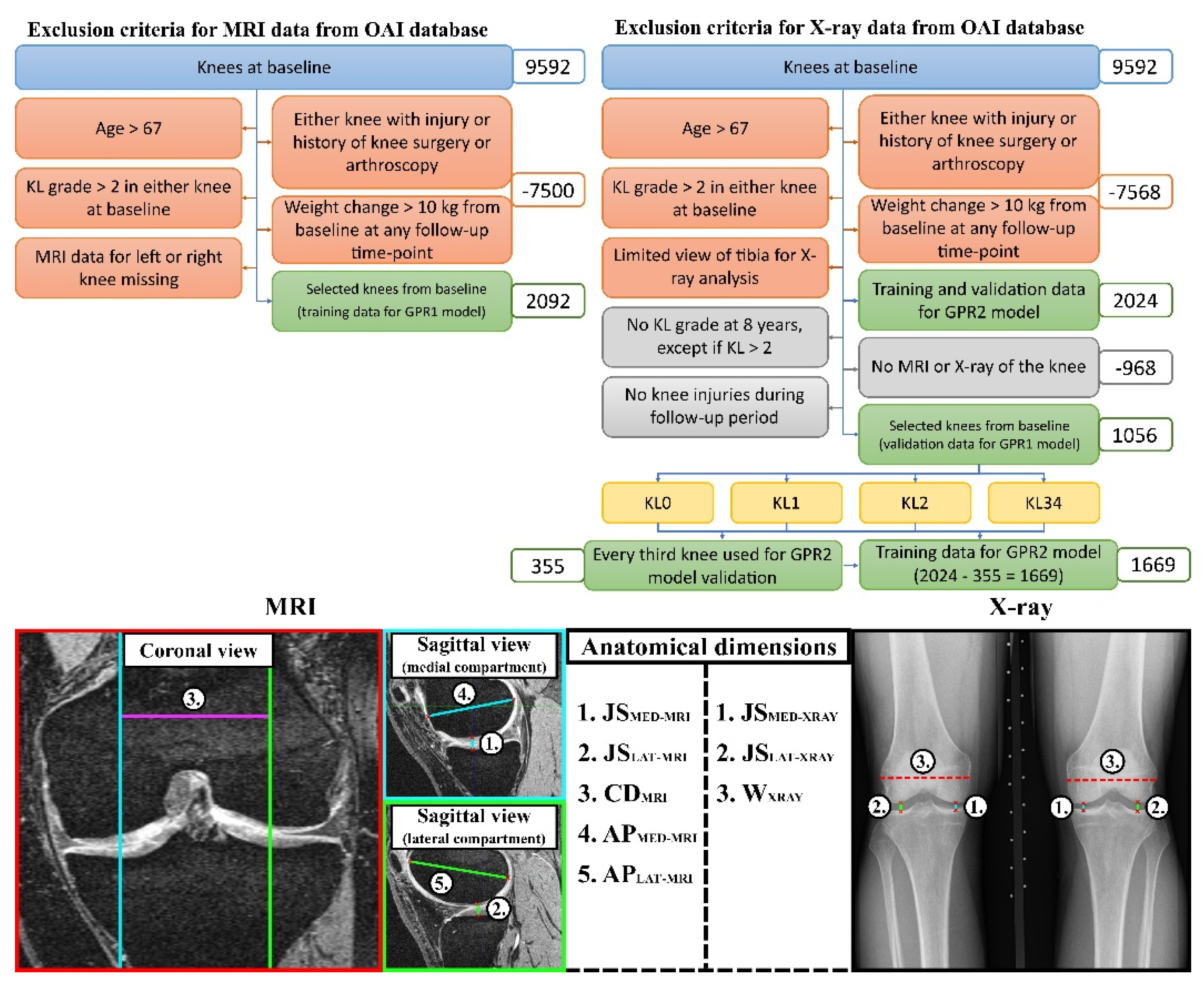
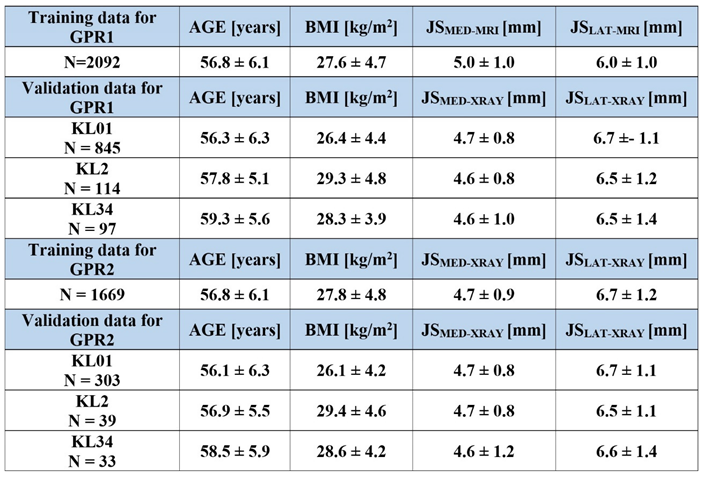
2.2. Data
2.3. Training Data in GPR1 and GPR2 Models
2.4. Atlas-Based FEA Method
- the tibiofemoral joint spaces (total cartilage thickness) at the medial compartment (JSMED-MRI)
- the tibiofemoral joint spaces (total cartilage thickness) at the lateral compartment (JSLAT-MRI)
- the maximum anterior-posterior dimensions at medial femoral condyles (APMED-MRI)
- the maximum anterior-posterior dimensions at lateral femoral condyles (APLAT-MRI)
- the medial-lateral condyle distance measured as a distance between the medial and lateral contact area (CDMRI)
- the tibiofemoral joint spaces at the medial compartment (JSMED-Xray)
- the tibiofemoral joint spaces at the lateral compartment (JSLAT-Xray)
- full medial-lateral width of the distal femur (WXray)
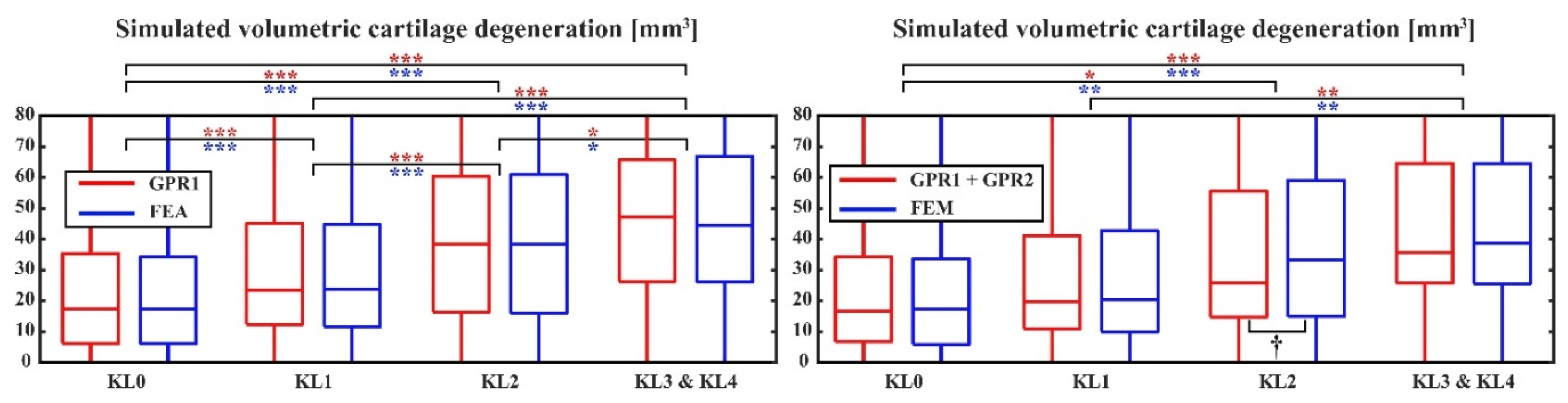
2.5. Simulation of Cartilage Degeneration
2.6. Statistical Analysis
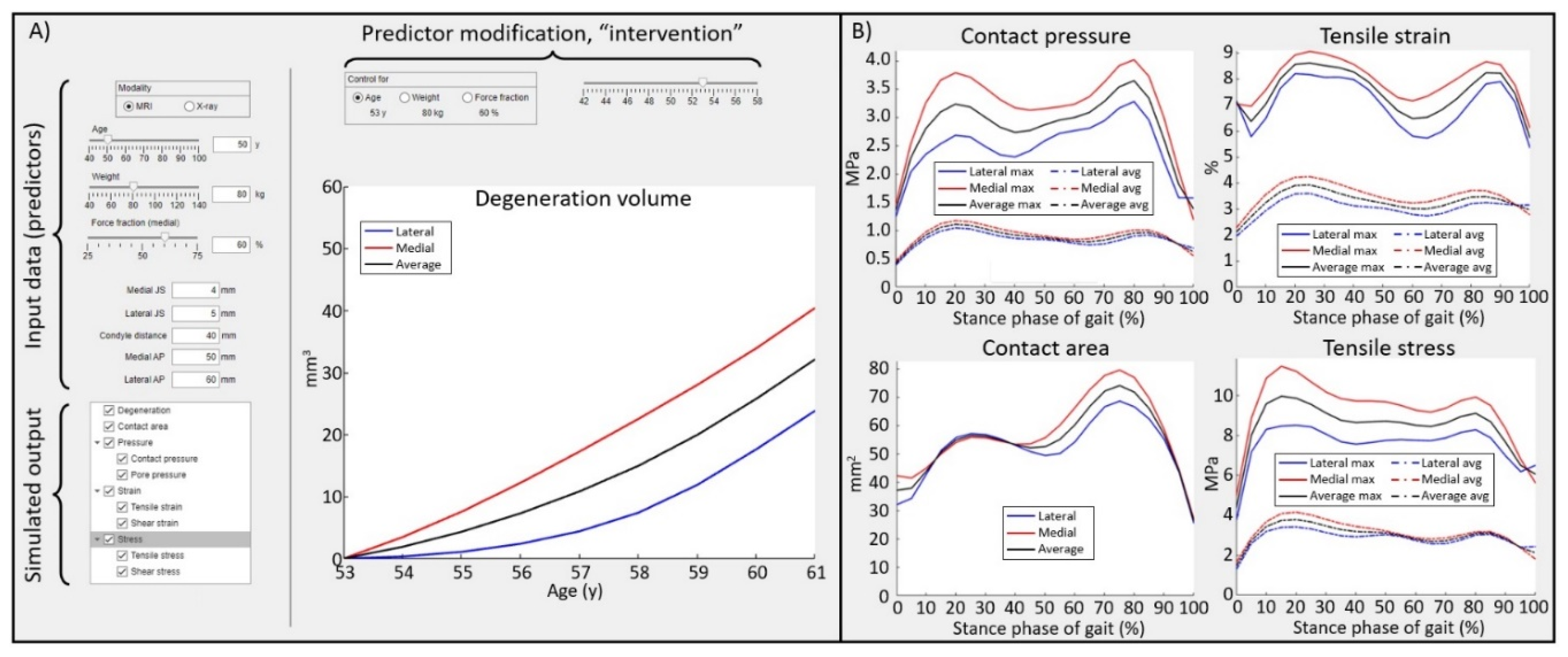
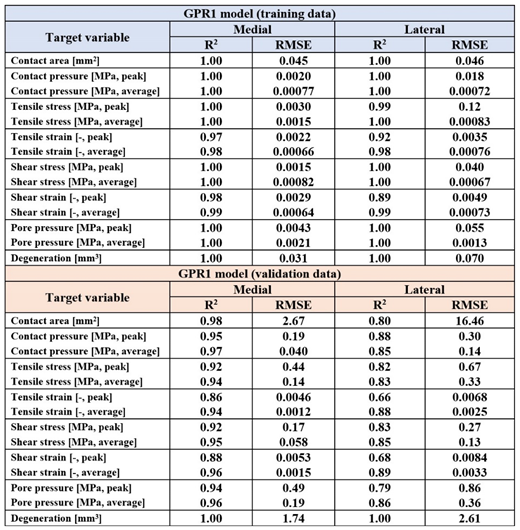
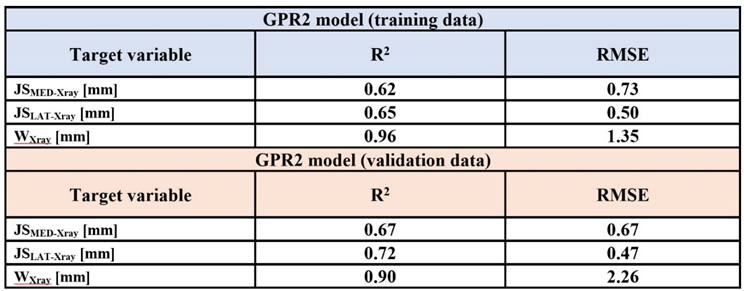
Results
3. Discussion
Supplementary Materials
Funding Sources
Role of the Funding Sources
Data availability
Acknowledgements
Conflict of Interest Statement
Ethical approval
References
- Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States: Part II. Arthritis Rheum. 2008;58(1):26-35. [CrossRef]
- Shane Anderson A, Loeser RF. Why is osteoarthritis an age-related disease? Best Pract Res Clin Rheumatol. 2010;24(1):15-26. [CrossRef]
- Theis KA, Murphy LB, Guglielmo D, et al. Prevalence of Arthritis and Arthritis-Attributable Activity Limitation — United States, 2016–2018. MMWR Morb Mortal Wkly Rep. 2021;70(40):1401-1407. [CrossRef]
- Törmälehto S, Mononen ME, Aarnio E, Arokoski JPA, Korhonen RK, Martikainen J. Health-related quality of life in relation to symptomatic and radiographic definitions of knee osteoarthritis: data from Osteoarthritis Initiative (OAI) 4-year follow-up study. Health Qual Life Outcomes. 2018;16(1):154. [CrossRef]
- Duffell LD, Southgate DFL, Gulati V, McGregor AH. Balance and gait adaptations in patients with early knee osteoarthritis. Gait Posture. 2014;39(4):1057-1061. [CrossRef]
- Felson DT, Hodgson R. Identifying and Treating Preclinical and Early Osteoarthritis. Rheumatic Disease Clinics of North America. 2014;40(4):699-710. [CrossRef]
- Murphy LB, Cisternas MG, Pasta DJ, Helmick CG, Yelin EH. Medical Expenditures and Earnings Losses Among US Adults With Arthritis in 2013. Arthritis Care Res (Hoboken). 2018;70(6):869-876. [CrossRef]
- Musumeci G, Aiello F, Szychlinska M, Di Rosa M, Castrogiovanni P, Mobasheri A. Osteoarthritis in the XXIst Century: Risk Factors and Behaviours that Influence Disease Onset and Progression. Int J Mol Sci. 2015;16(12):6093-6112. [CrossRef]
- Bliddal H, Leeds AR, Christensen R. Osteoarthritis, obesity and weight loss: evidence, hypotheses and horizons – a scoping review. Obesity Reviews. 2014;15(7):578-586. [CrossRef]
- Hirvasniemi J, Runhaar J, van der Heijden RA, et al. The KNee OsteoArthritis Prediction (KNOAP2020) challenge: An image analysis challenge to predict incident symptomatic radiographic knee osteoarthritis from MRI and X-ray images. Osteoarthritis Cartilage. 2023;31(1):115-125. [CrossRef]
- Ramazanian T, Fu S, Sohn S, Taunton MJ, Kremers HM. Prediction Models for Knee Osteoarthritis: Review of Current Models and Future Directions. Arch Bone Jt Surg. 2023;11(1):1-11. [CrossRef]
- Appleyard T, Thomas MJ, Antcliff D, Peat G. Prediction Models to Estimate the Future Risk of Osteoarthritis in the General Population: A Systematic Review. Arthritis Care Res (Hoboken). 2023;75(7):1481-1493. [CrossRef]
- Cooper RJ, Wilcox RK, Jones AC. Finite element models of the tibiofemoral joint: A review of validation approaches and modelling challenges. Med Eng Phys. 2019;74:1-12. [CrossRef]
- Paz A, Orozco GA, Korhonen RK, García JJ, Mononen ME. Expediting Finite Element Analyses for Subject-Specific Studies of Knee Osteoarthritis: A Literature Review. Applied Sciences. 2021;11(23):11440. [CrossRef]
- Halonen KS, Dzialo CM, Mannisi M, Venäläinen MS, de Zee M, Andersen MS. Workflow assessing the effect of gait alterations on stresses in the medial tibial cartilage - combined musculoskeletal modelling and finite element analysis. Sci Rep. 2017;7(1):17396. [CrossRef]
- Liukkonen MK, Mononen ME, Vartiainen P, et al. Evaluation of the Effect of Bariatric Surgery-Induced Weight Loss on Knee Gait and Cartilage Degeneration. J Biomech Eng. 2018;140(4). [CrossRef]
- Mononen ME, Liukkonen MK, Turunen MJ. X-ray with finite element analysis is a viable alternative for MRI to predict knee osteoarthritis: Data from the Osteoarthritis Initiative. Journal of Orthopaedic Research. Published online April 22, 2024. [CrossRef]
- Paz A, García J, Korhonen R, Mononen M. Towards a Transferable Modeling Method of the Knee to Distinguish Between Future Healthy Joints from Osteoarthritic Joints: Data from the Osteoarthritis Initiative. Ann Biomed Eng. 2023; [CrossRef]
- Mononen ME, Liukkonen MK, Korhonen RK. Utilizing Atlas-Based Modeling to Predict Knee Joint Cartilage Degeneration: Data from the Osteoarthritis Initiative. Ann Biomed Eng. 2019;47(3). [CrossRef]
- Widén E, Junna N, Ruotsalainen S, et al. How Communicating Polygenic and Clinical Risk for Atherosclerotic Cardiovascular Disease Impacts Health Behavior: an Observational Follow-up Study. Circ Genom Precis Med. 2022;15(2). [CrossRef]
- Mortensen JF, Mongelard KBG, Radev DI, et al. MRi of the knee compared to specialized radiography for measurements of articular cartilage height in knees with osteoarthritis. J Orthop. 2021;25:191-198. [CrossRef]
- Kellgren JH, Lawrence JS. Radiological Assessment of Osteo-Arthrosis. Ann Rheum Dis. 1957;16(4):494-502. [CrossRef]
- Mononen ME, Paz A, Liukkonen MK, Turunen MJ. Atlas-based finite element analyses with simpler constitutive models predict personalized progression of knee osteoarthritis: data from the osteoarthritis initiative. Sci Rep. 2023;13(1):8888. [CrossRef]
- Martínez-Camblor P, Pérez-Fernández S, Díaz-Coto S. The area under the generalized receiver-operating characteristic curve. Int J Biostat. 2021;18(1):293-306. [CrossRef]
- Chan LC, Li HHT, Chan PK, Wen C. A machine learning-based approach to decipher multi-etiology of knee osteoarthritis onset and deterioration. Osteoarthr Cartil Open. 2021;3(1):100135. [CrossRef]
- Cai G, Jiang M, Cicuttini F, Jones G. Association of age, sex and BMI with the rate of change in tibial cartilage volume: a 10.7-year longitudinal cohort study. Arthritis Res Ther. 2019;21(1):273. [CrossRef]
- Si L, Xuan K, Zhong J, et al. Knee Cartilage Thickness Differs Alongside Ages: A 3-T Magnetic Resonance Research Upon 2,481 Subjects via Deep Learning. Front Med (Lausanne). 2021;7. [CrossRef]
- Karvonen RL, Negendank WG, Teitge RA, Reed AH, Miller PR, Fernandez-Madrid F. Factors affecting articular cartilage thickness in osteoarthritis and aging. J Rheumatol. 1994;21(7):1310-1318.
- Gwynne-Jones JH, Wilson RA, Wong JMY, Abbott JH, Gwynne-Jones DP. The Outcomes of Nonoperative Management of Patients With Hip and Knee Osteoarthritis Triaged to a Physiotherapy-Led Clinic at Minimum 5-Year Follow-Up and Factors Associated With Progression to Surgery. J Arthroplasty. 2020;35(6):1497-1503. [CrossRef]
- Pagnon D, Domalain M, Reveret L. Pose2Sim: An End-to-End Workflow for 3D Markerless Sports Kinematics—Part 1: Robustness. Sensors. 2021;21(19):6530. [CrossRef]
- Di Raimondo G, Willems M, Killen BA, et al. Peak Tibiofemoral Contact Forces Estimated Using IMU-Based Approaches Are Not Significantly Different from Motion Capture-Based Estimations in Patients with Knee Osteoarthritis. Sensors. 2023;23(9):4484. [CrossRef]

 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




