Submitted:
11 September 2024
Posted:
11 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell culture, treatments, and transfections
2.2. DNA Constructs
2.3. Recombinant Protein Production in Escherichia Coli
2.4. Western Blotting
2.5. In Vitro Kinase Assays
2.6. Luciferase Assays
2.7. Quantitative PCR
2.8. Viability Assays
2.9. Statistical Analysis and Figure Preparation
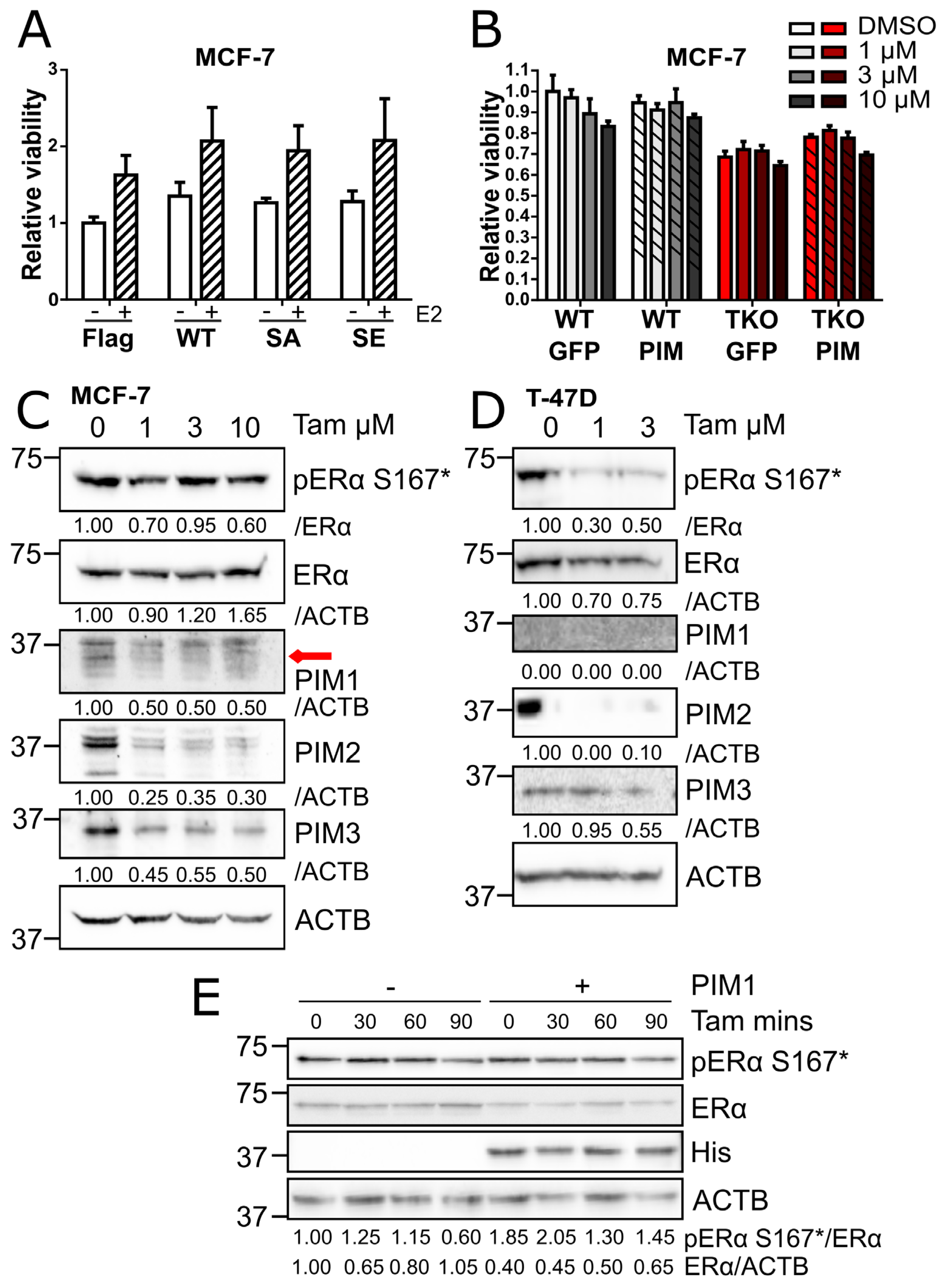
3. Results
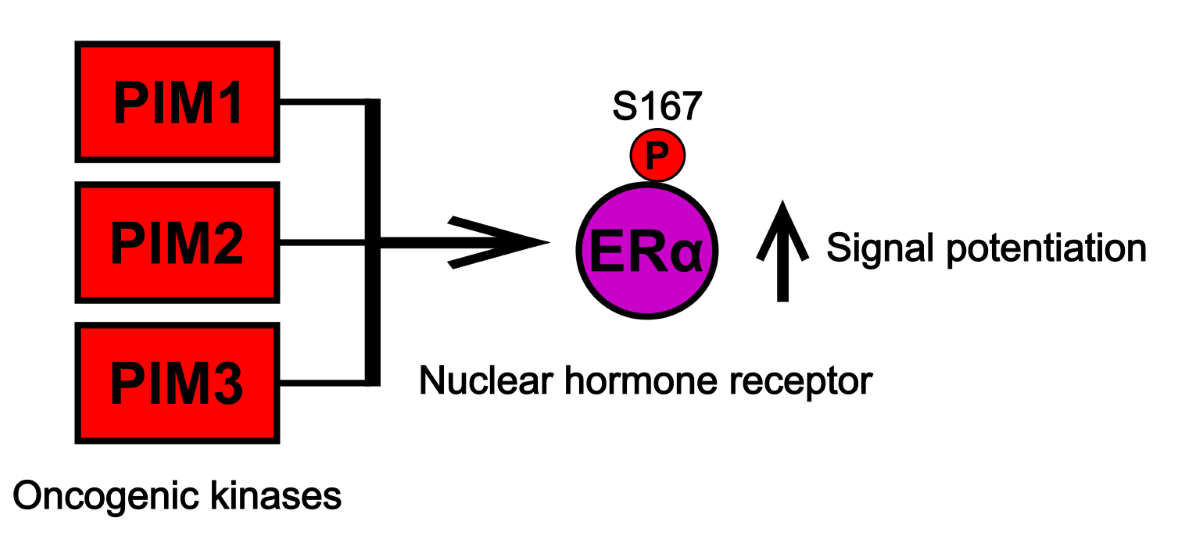
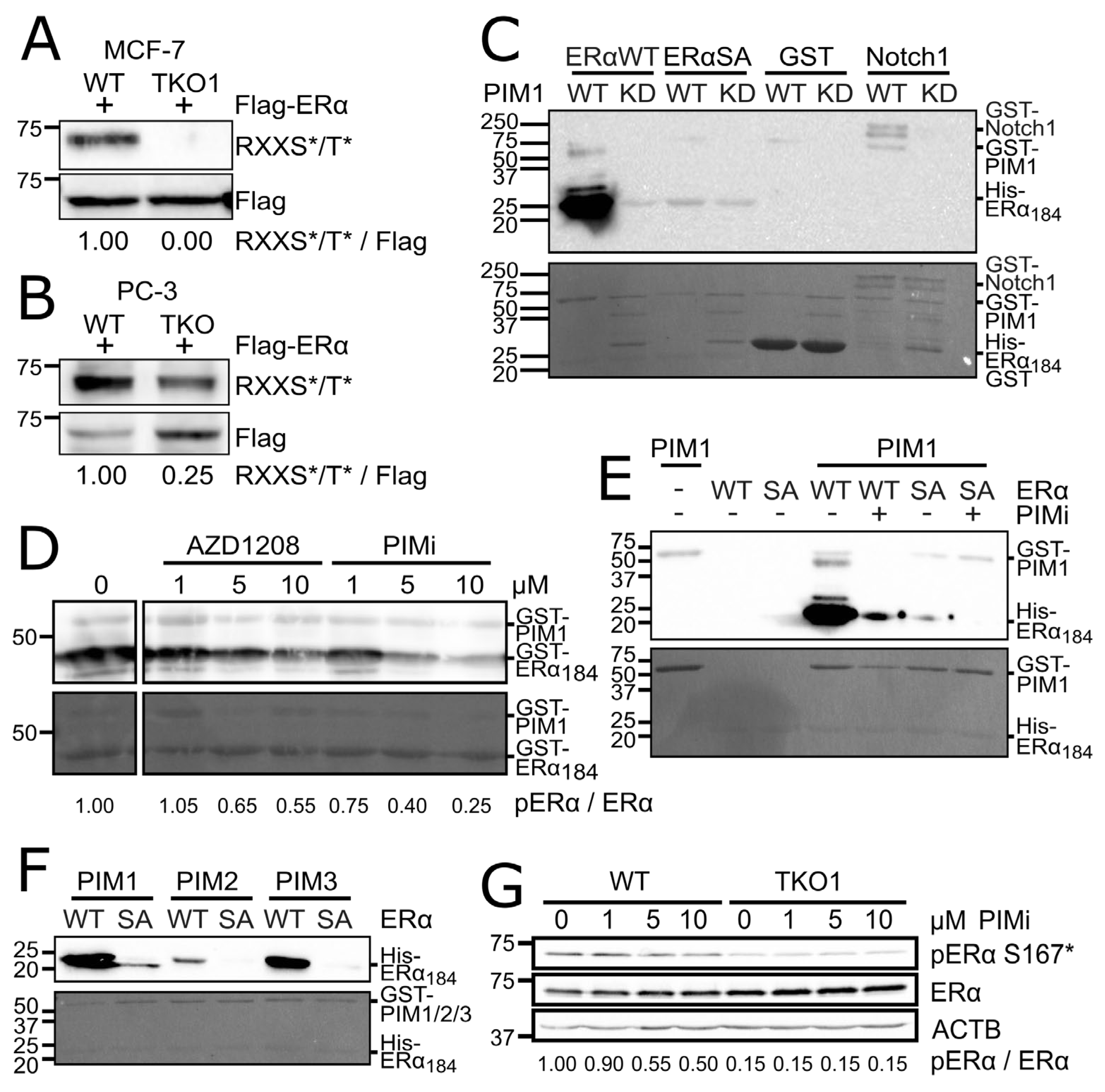
3.1. Estrogen Receptor α is a Novel PIM Substrate
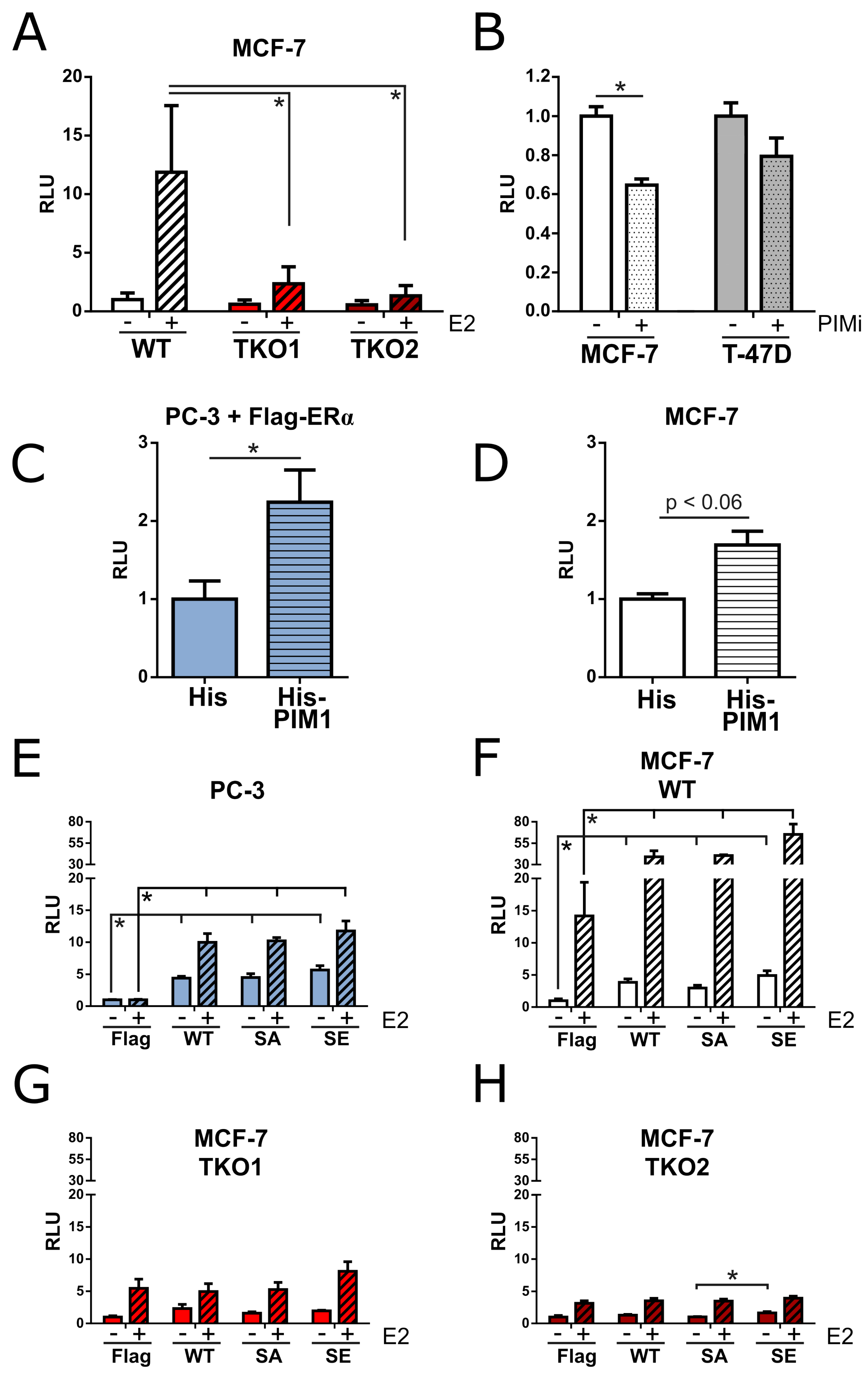
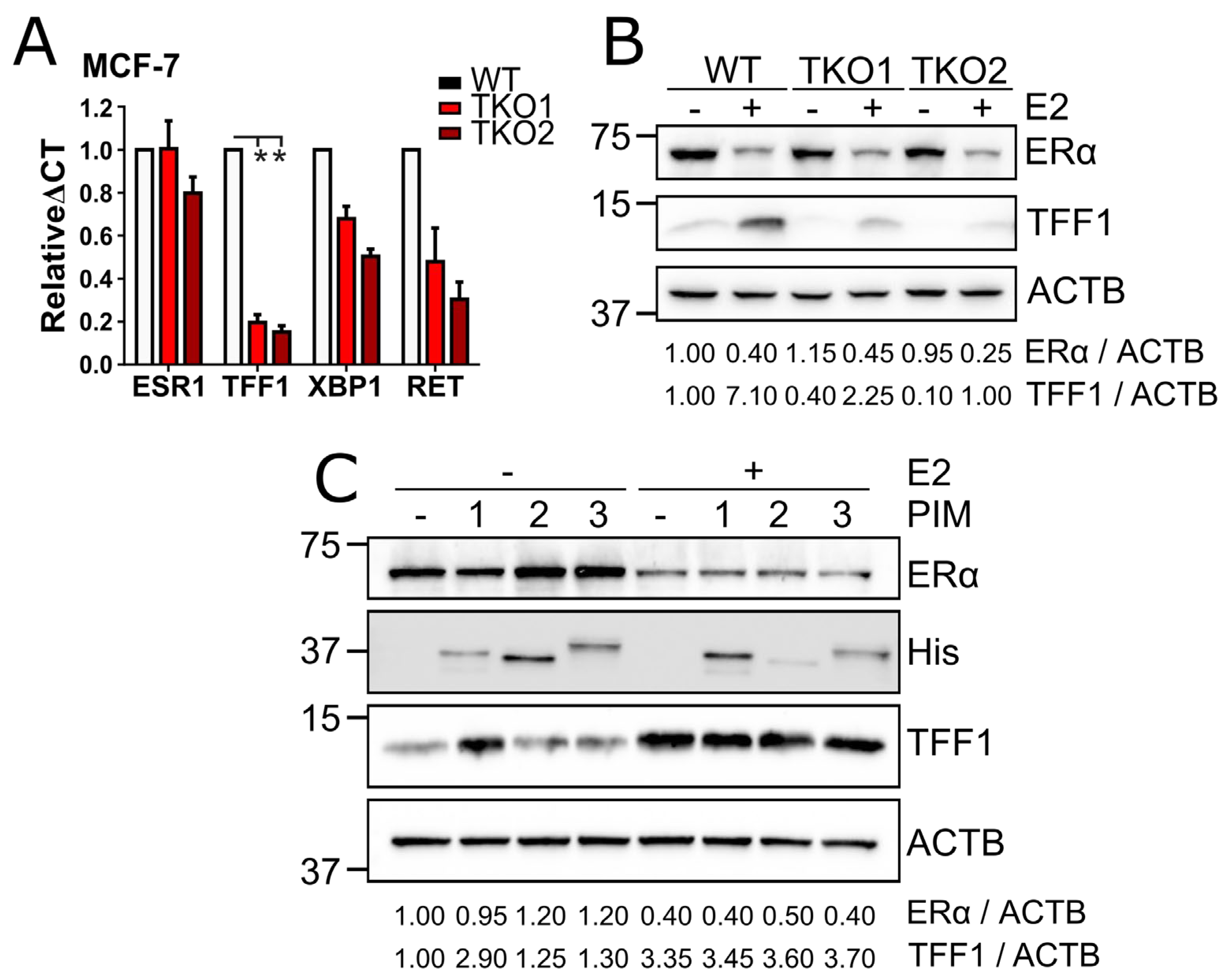
3.2. PIM Kinases Enhance ERα Signaling
3.3. PIM Overexpression Does Not Protect Cells from Tamoxifen
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aho, T.L.T.; Sandholm, J.; Peltola, K.J.; Ito, Y.; Koskinen, P.J. Pim-1 kinase phosphorylates RUNX family transcription factors and enhances their activity. BMC Cell Biol 2006, 7, 21. [CrossRef]
- Rainio, E.-M.; Sandholm, J.; Koskinen, P.J. Cutting Edge: Transcriptional activity of NFATc1 is enhanced by the Pim-1 kinase. J Immunol 2002, 168, 1524–1527. [CrossRef]
- Santio, N.M.; Salmela, M.; Arola, H.; Eerola, S.K.; Heino, J.; Rainio, E.M.; Koskinen, P.J. The PIM1 kinase promotes prostate cancer cell migration and adhesion via multiple signaling pathways. Exp Cell Res 2016, 342, 113-124. [CrossRef]
- Santio, N.M.; Landor, S.K.-J.; Vahtera, L.; Ylä-Pelto, J.; Paloniemi, E.; Imanishi, S.Y.; Corthals, G.; Varjosalo, M.; Manoharan, G.B.; Uri, A.; et al. Phosphorylation of Notch1 by Pim kinases promotes oncogenic signaling in breast and prostate cancer cells. Oncotarget 2016, 7, 43220–43238. [CrossRef]
- Landor, S.K.J.; Santio, N.M.; Eccleshall, W.B.; Paramonov, V.M.; Gagliani, E.K.; Hall, D.; Jin, S.B.; Dahlström, K.M.; Salminen, T.A.; Rivero-Müller, A.; et al. PIM-induced phosphorylation of Notch3 promotes breast cancer tumorigenicity in a CSL-independent fashion. J Biol Chem 2021, 296:100593. [CrossRef]
- Mung, K.L.; Eccleshall, W.B.; Santio, N.M.; Rivero-Müller, A.; Koskinen, P.J. PIM kinases inhibit AMPK activation and promote tumorigenicity by phosphorylating LKB1. Cell Commun Signal 2021, 19(1):68. [CrossRef]
- Mung, K.L.; Meinander, A.; Koskinen, P.J. PIM kinases phosphorylate lactate dehydrogenase A at serine 161 and suppress its nuclear ubiquitination. FEBS J 2023, 290(9):2489-2502. [CrossRef]
- Zhang, F.; Beharry, Z.M.; Harris, T.E.; Lilly, M.B.; Smith, C.D.; Mahajan, S.; Kraft, A.S. PIM1 protein kinase regulates PRAS40 phosphorylation and mTOR activity in FDCP1 cells. Cancer Biol Ther 2009, 8(9):846-53. [CrossRef]
- Aho, T.L.T.; Sandholm, J.; Peltola, K.J.; Mankonen, H.; Lilly, M.; Koskinen, P.J. Pim-1 kinase promotes inactivation of the pro-apoptotic Bad protein by phosphorylating it on the Ser112 gatekeeper site. FEBS Lett 2004, 571, 43-49,. [CrossRef]
- Grundler, R.; Brault, L.; Gasser, C.; Bullock, A.N.; Dechow, T.; Woetzel, S.; Pogacic, V.; Villa, A.; Ehret, S.; Berridge, G.; et al. Dissection of PIM serine/threonine kinases in FLT3-ITD-induced leukemogenesis reveals PIM1 as regulator of CXCL12-CXCR4-mediated homing and migration. J Exp Med 2009, 206(9):1957-70. [CrossRef]
- Santio, N.M.; Vainio, V.; Hoikkala, T.; Mung, K.L.; Lång, M.; Vahakoski, R.; Zdrojewska, J.; Coffey, E.T.; Kremneva, E.; Rainio, E.M.; et al. PIM1 accelerates prostate cancer cell motility by phosphorylating actin capping proteins. Cell Commun Signal 2020, 18(1):121. [CrossRef]
- Santio, N.M.; Koskinen, P.J. PIM Kinases: From survival factors to regulators of cell motility. Int J Biochem Cell Biol 2017, 93, 74–85.
- Zemskova, M.; Sahakian, E.; Bashkirova, S.; Lilly, M. The PIM1 kinase is a critical component of a survival pathway activated by docetaxel and promotes survival of docetaxel-treated prostate cancer cells. J Biol Chem 2008, 283(30): 20635–20644. [CrossRef]
- Peltola, K.; Hollmen, M.; Maula, S.M.; Rainio, E.; Ristamäki, R.; Luukkaa, M.; Sandholm, J.; Sundvall, M.; Elenius, K.; Koskinen, P.J.; et al. Pim-1 kinase expression predicts radiation response in squamocellular carcinoma of head and neck and is under the control of Epidermal Growth Factor Receptor. Neoplasia 2009, 11(7): 629–636. [CrossRef]
- Marayati, R.; Stafman, L.L.; Williams, A.P.; Bownes, L. V.; Quinn, C.H.; Aye, J.M.; Stewart, J.E.; Yoon, K.J.; Anderson, J.C.; Willey, C.D.; et al. PIM kinases mediate resistance to cisplatin chemotherapy in hepatoblastoma. Sci Rep 2021, 11(1):5984. [CrossRef]
- Mikkers, H.; Nawijn, M.; Allen, J.; Brouwers, C.; Verhoeven, E.; Jonkers, J.; Berns, A. Mice deficient for all PIM Kinases display reduced body size and impaired responses to hematopoietic growth factors. Mol Cell Biol 2004, 24, 6104–6115. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.; Tamura, K.; DeAngelo, D.J.; de Bono, J.; Lorente, D.; Minden, M.; Uy, G.L.; Kantarjian, H.; Chen, L.S.; Gandhi, V.; et al. Phase I Studies of AZD1208, a proviral integration Moloney virus kinase inhibitor in solid and haematological cancers. Br J Cancer 2018, 118, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Luszczak, S.; Kumar, C.; Sathyadevan, V.K.; Simpson, B.S.; Gately, K.A.; Whitaker, H.C.; Heavey, S. PIM kinase inhibition: Co-targeted therapeutic approaches in prostate cancer. Signal Transduct Target Ther 2020, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Clusan, L.; Ferrière, F.; Flouriot, G.; Pakdel, F. A basic review on estrogen receptor signaling pathways in breast cancer. Int J Mol Sci 2023, 24(7): 6834. [CrossRef]
- American Cancer Society. Breast Cancer Facts and Figures 2022–2024. Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/2022-2024-breast-cancer-fact-figures-acs.pdf (accessed on 23 August 2024).
- Pejerrey, S.M.; Dustin, D.; Kim, J.A.; Gu, G.; Rechoum, Y.; Fuqua, S.A.W. The impact of ESR1 mutations on the treatment of metastatic breast cancer. Horm Cancer 2018, 9(4): 215–228.
- Jeselsohn, R.; Bergholz, J.S.; Pun, M.; Cornwell, M.; Liu, W.; Nardone, A.; Xiao, T.; Li, W.; Qiu, X.; Buchwalter, G.; et al. Allele-specific chromatin recruitment and therapeutic vulnerabilities of ESR1 activating mutations. Cancer Cell 2018, 33(2):173-186. [CrossRef]
- Lei, J.T.; Shao, J.; Zhang, J.; Iglesia, M.; Chan, D.W.; Cao, J.; Anurag, M.; Singh, P.; He, X.; Kosaka, Y.; et al. Functional annotation of ESR1 gene fusions in estrogen receptor-positive breast cancer. Cell Rep 2018, 7;24(6):1434-1444. [CrossRef]
- Zhou, Y.; Gerrard, D.L.; Wang, J.; Li, T.; Yang, Y.; Fritz, A.J.; Rajendran, M.; Fu, X.; Schiff, R.; Lin, S.; et al. Temporal dynamic reorganization of 3D chromatin architecture in hormone-induced breast cancer and endocrine resistance. Nat Commun 2019, 10(1):1522. [CrossRef]
- Achinger-Kawecka, J.; Valdes-Mora, F.; Luu, P.L.; Giles, K.A.; Caldon, C.E.; Qu, W.; Nair, S.; Soto, S.; Locke, W.J.; Yeo-Teh, N.S.; et al. Epigenetic reprogramming at estrogen-receptor binding sites alters 3D chromatin landscape in endocrine-resistant breast cancer. Nat Commun 2020, 11(1):320. [CrossRef]
- Xu, G.; Chhangawala, S.; Cocco, E.; Razavi, P.; Cai, Y.; Otto, J.E.; Ferrando, L.; Selenica, P.; Ladewig, E.; Chan, C.; et al. ARID1A determines luminal identity and therapeutic response in estrogen-receptor-positive breast cancer. Nat Genet 2020, 52(2):198-207. [CrossRef]
- Nagarajan, S.; Rao, S. V.; Sutton, J.; Cheeseman, D.; Dunn, S.; Papachristou, E.K.; Prada, J.E.G.; Couturier, D.L.; Kumar, S.; Kishore, K.; et al. ARID1A influences HDAC1/BRD4 activity, intrinsic proliferative capacity and breast cancer treatment response. Nat Genet 2020, 52(2):187-197. [CrossRef]
- Hurtado, A.; Holmes, K.A.; Ross-Innes, C.S.; Schmidt, D.; Carroll, J.S. FOXA1 is a key determinant of estrogen receptor function and endocrine response. Nat Genet 2011, 43(1): 27–33. [CrossRef]
- Ross-Innes, C.S.; Stark, R.; Teschendorff, A.E.; Holmes, K.A.; Ali, H.R.; Dunning, M.J.; Brown, G.D.; Gojis, O.; Ellis, I.O.; Green, A.R.; et al. Differential oestrogen receptor binding is associated with clinical outcome in breast cancer. Nature 2012, 481(7381): 389–393. [CrossRef]
- Fu, X.; Jeselsohn, R.; Pereira, R.; Hollingsworth, E.F.; Creighton, C.J.; Li, F.; Shea, M.; Nardone, A.; De Angelis, C.; Heiser, L.M.; et al. FOXA1 overexpression mediates endocrine resistance by altering the ER transcriptome and IL-8 expression in ER-positive breast cancer. Proc Natl Acad Sci USA 2016, 113(43):E6600-E6609. [CrossRef]
- Fu, X.; Pereira, R.; De Angelis, C.; Veeraraghavan, J.; Nanda, S.; Qin, L.; Cataldo, M.L.; Sethunath, V.; Mehravaran, S.; Gutierrez, C.; et al. FOXA1 upregulation promotes enhancer and transcriptional reprogramming in endocrine-resistant breast cancer. Proc Natl Acad Sci USA 2019, 116(52):26823-26834. [CrossRef]
- Haricharan, S.; Punturi, N.; Singh, P.; Holloway, K.R.; Anurag, M.; Schmelz, J.; Schmidt, C.; Lei, J.T.; Suman, V.; Hunt, K.; et al. Loss of Mutl disrupts CHK2-dependent cell-cycle control through CDK4/6 to promote intrinsic endocrine therapy resistance in primary breast cancer. Cancer Discov 2017, 7(10):1168-1183. [CrossRef]
- Frogne, T.; Benjaminsen, R. V.; Sonne-Hansen, K.; Sorensen, B.S.; Nexo, E.; Laenkholm, A.V.; Rasmussen, L.M.; Riese, D.J.; De Cremoux, P.; Stenvang, J.; et al. Activation of ErbB3, EGFR and Erk is essential for growth of human breast cancer cell lines with acquired resistance to fulvestrant. Breast Cancer Res Treat 2009, 114(2):263-75. [CrossRef]
- McClaine, R.J.; Marshall, A.M.; Wagh, P.K.; Waltz, S.E. Ron receptor tyrosine kinase activation confers resistance to tamoxifen in breast cancer cell lines. Neoplasia 2010, 12(8): 650–658. [CrossRef]
- Turner, N.; Pearson, A.; Sharpe, R.; Lambros, M.; Geyer, F.; Lopez-Garcia, M.A.; Natrajan, R.; Marchio, C.; Iorns, E.; Mackay, A.; et al. FGFR1 amplification drives endocrine therapy resistance and is a therapeutic target in breast cancer. Cancer Res 2010, 70(5):2085-94. [CrossRef]
- Fox, E.M.; Miller, T.W.; Balko, J.M.; Kuba, M.G.; Sánchez, V.; Smith, R.A.; Liu, S.; González-Angulo, A.M.; Mills, G.B.; Ye, F.; et al. A kinome-wide screen identifies the insulin/IGF-I receptor pathway as a mechanism of escape from hormone dependence in breast cancer. Cancer Res 2011, 71(21):6773-84. [CrossRef]
- Kirkegaard, T.; Witton, C.J.; McGlynn, L.M.; Tovey, S.M.; Dunne, B.; Lyon, A.; Bartlett, J.M.S. AKT activation predicts outcome in breast cancer patients treated with tamoxifen. J Pathol 2005, 207(2):139-46. [CrossRef]
- Miller, T.W.; Hennessy, B.T.; González-Angulo, A.M.; Fox, E.M.; Mills, G.B.; Chen, H.; Higham, C.; García-Echeverría, C.; Shyr, Y.; Arteaga, C.L. Hyperactivation of Phosphatidylinositol-3 Kinase promotes escape from hormone dependence in estrogen receptor-positive human breast cancer. J Clin Invest 2010, 120(7):2406-13. [CrossRef]
- Staka, C.M.; Nicholson, R.I.; Gee, J.M.W. Acquired resistance to oestrogen deprivation: role for growth factor signalling kinases/oestrogen receptor cross-talk revealed in new MCF-7X model. Endocr Relat Cancer 2005, 12 Suppl 1:S85-97. [CrossRef]
- Likhite, V.S.; Stossi, F.; Kim, K.; Katzenellenbogen, B.S.; Katzenellenbogen, J.A. Kinase-specific phosphorylation of the estrogen receptor changes receptor interactions with ligand, deoxyribonucleic acid, and coregulators associated with alterations in estrogen and tamoxifen activity. Mol Endocrinol 2006, 20(12):3120-32. [CrossRef]
- Chen, M.; Cui, Y.K.; Huang, W.H.; Man, K.; Zhang, G.J. Chen, M.; Cui, Y.K.; Huang, W.H.; Man, K.; Zhang, G.J. Phosphorylation of estrogen receptor α at serine 118 is correlated with breast cancer resistance to tamoxifen. Oncol Lett 2013, 6(1): 118–124. [CrossRef]
- Yamnik, R.L.; Digilova, A.; Davis, D.C.; Brodt, Z.N.; Murphy, C.J.; Holz, M.K. S6 kinase 1 regulates estrogen receptor α in control of breast cancer cell proliferation. J Biol Chem 2009, 284(10): 6361-9. [CrossRef]
- Guo, J.-P.; Shu, S.-K.; Esposito, N.N.; Coppola, D.; Koomen, J.M.; Cheng, J.Q. IKKϵ Phosphorylation of estrogen receptor α Ser-167 and contribution to tamoxifen resistance in breast cancer. J Biol Chem 2016, 285(6): 3676–3684. [CrossRef]
- Zheng, X.Q.; Guo, J.P.; Yang, H.; Kanai, M.; He, L.L.; Li, Y.Y.; Koomen, J.M.; Minton, S.; Gao, M.; Ren, X.B.; et al. Aurora-A Is a determinant of tamoxifen sensitivity through phosphorylation of ERα in breast cancer. Oncogene 2014, 33(42):4985-96. [CrossRef]
- Malinen, M.; Jääskeläinen, T.; Pelkonen, M.; Heikkinen, S.; Väisänen, S.; Kosma, V.M.; Nieminen, K.; Mannermaa, A.; Palvimo, J.J. Proto-oncogene PIM-1 is a novel estrogen receptor target associating with high grade breast tumors. Mol Cell Endocrinol 2013, 365(2), 270–276. [CrossRef]
- Joel, P.B.; Smith, J.; Sturgill, T.W.; Fisher, T.L.; Blenis, J.; Lannigan, D.A. Pp90 Rsk1 regulates estrogen receptor-mediated transcription through phosphorylation of Ser-167. Mol Cell Biol 1998, 18(4): 1978–1984. [CrossRef]
- Clark, D.E.; Poteet-Smith, C.E.; Smith, J.A.; Lannigan, D.A. Rsk2 allosterically activates estrogen receptor α by docking to the hormone-binding domain. EMBO J 2001, 20(13):3484-94. [CrossRef]
- Campbell, R.A.; Bhat-Nakshatri, P.; Patel, N.M.; Constantinidou, D.; Ali, S.; Nakshatri, H. Phosphatidylinositol 3-Kinase/AKT-mediated activation of estrogen receptor α: A new model for anti-estrogen resistance. J Biol Chem 2001, 276(13):9817-24. [CrossRef]
- Arnold, S.F.; Obourn, J.D.; Jaffe, H.; Notides, A.C. Serine 167 is the major estradiol-induced phosphorylation site on the human estrogen receptor. Mol Endocrinol 1994, 8(9):1208-14. [CrossRef]
- Stenoien, D.L.; Mancini, M.G.; Patel, K.; Allegretto*, E.A.; Smith, C.L.; Mancini, M.A. Subnuclear trafficking of estrogen receptor-α and steroid receptor coactivator-1. Mol Endocrinol 2000, 14(4):518-34. [CrossRef]
- Hall, J.M.; McDonnel, D.P. The estrogen receptor β-isoform (ERβ) of the human estrogen receptor modulates ERα transcriptional activity and is a key regulator of the cellular response to estrogens and antiestrogens. Endocrinology 1999, 140(12):5566-78. [CrossRef]
- Kiriazis, A.; Vahakoski, R.L.; Santio, N.M.; Arnaudova, R.; Eerola, S.K.; Rainio, E.M.; Aumüller, I.B.; Yli-Kauhaluoma, J.; Koskinen, P.J. Tricyclic benzo[Cd]azulenes selectively inhibit activities of Pim kinases and restrict growth of Epstein-Barr virus-transformed cells. PLoS One 2013, 8(2):e55409. [CrossRef]
- Santio, N.M.; Vahakoski, R.L.; Rainio, E.M.; Sandholm, J.A.; Virtanen, S.S.; Prudhomme, M.; Anizon, F.; Moreau, P.; Koskinen, P.J. Pim-selective inhibitor DHPCC-9 reveals Pim kinases as potent stimulators of cancer cell migration and invasion. Mol Cancer 2010, 9:279. [CrossRef]
- Palaty, C.K.; Clark-lewis, I.; Leung, D.; Pelech, S.L. Phosphorylation site substrate specificity determinants for the Pim-1 proto-oncogene-encoded protein kinase. Biochem Cell Biol 1997, 75(2):153–162. [CrossRef]
- Peng, C.; Knebel, A.; Morrice, N.A.; Li, X.; Barringer, K.; Li, J.; Jakes, S.; Werneburg, B.; Wang, L. Pim kinase substrate identification and specificity. J Biol Chem 2007, 141, 353–362. [CrossRef]
- Masiakowski, P.; Breathnach, R.; Bloch, J.; Gannon, F.; Krust, A.; Chambon, P. Cloning of cDNA sequences of hormone-regulated genes from the MCF-7 human breast cancer cell line. Nucleic Acids Res 1982, 10(24):7895-903. [CrossRef]
- Boulay, A.; Breuleux, M.; Stephan, C.; Fux, C.; Brisken, C.; Fiche, M.; Wartmann, M.; Stumm, M.; Lane, H.A.; Hynes, N.E. The Ret receptor tyrosine kinase pathway functionally interacts with the ERα pathway in breast cancer. Cancer Res 2008, 68(10):3743-51. [CrossRef]
- Bertucci, F.; Houlgatte, R.; Benziane, A.; Granjeaud, S.; Adélaïde, J.; Tagett, R.; Loriod, B.; Jacquemier, J.; Viens, P.; Jordan, B.; et al. Gene expression profiling of primary breast carcinomas using arrays of candidate genes. Hum Mol Genet 2000, 9(20):2981-91. [CrossRef]
- Creighton, C.J.; Cordero, K.E.; Larios, J.M.; Miller, R.S.; Johnson, M.D.; Chinnaiyan, A.M.; Lippman, M.E.; Rae, J.M. Genes regulated by estrogen in breast tumor cells in vitro are similarly regulated in vivo in tumor xenografts and human breast tumors. Genome Biol 2006, 7(4):R28. [CrossRef]
- Hornbeck, P. V.; Zhang, B.; Murray, B.; Kornhauser, J.M.; Latham, V.; Skrzypek, E. PhosphoSitePlus, 2014: mutations, PTMs and recalibrations. Nucleic Acids Res 2015, 43(Database issue):D512-20. [CrossRef]
- Anbalagan, M.; Rowan, B.G. Estrogen receptor alpha phosphorylation and its functional impact in human breast cancer. Mol Cell Endocrinol 2015, 418(3):264-72.
- Warfel, N.A.; Kraft, A.S. PIM kinase (and Akt) biology and signaling in tumors. Pharmacol Ther 2015, 151: 41-49.
- Shah, Y.M.; Rowan, B.G. The Src kinase pathway promotes tamoxifen agonist action in Ishikawa endometrial cells through phosphorylation-dependent stabilization of estrogen receptor (Alpha) promoter interaction and elevated steroid receptor coactivator 1 activity. Mol Endocrinol 2005, 19: 732–748. [CrossRef]
- Pardeshi, J.; McCormack, N.; Gu, L.; Ryan, C.S.; Schröder, M. DDX3X Functionally and physically interacts with estrogen receptor-alpha. Biochim Biophys Acta Gene Regul Mech 2022, 1865(2):194787. [CrossRef]
- Huderson, B.P.; Duplessis, T.T.; Williams, C.C.; Seger, H.C.; Marsden, C.G.; Pouey, K.J.; Hill, S.M.; Rowan, B.G. Stable Inhibition of Specific Estrogen Receptor α(ERα) Phosphorylation confers increased growth, migration/invasion, and disruption of estradiol signaling in MCF-7 breast cancer cells. Endocrinology 2012, 153(9):4144-59. [CrossRef]
- Motomura, K.; Ishitobi, M.; Komoike, Y.; Koyama, H.; Nagase, H.; Inaji, H.; Noguchi, S. Expression of estrogen receptor beta and phosphorylation of estrogen receptor alpha serine 167 correlate with progression-free survival in patients with metastatic breast cancer treated with aromatase inhibitors. Oncology 2010, 79(1-2):55-61. [CrossRef]
- Ishida, N.; Baba, M.; Hatanaka, Y.; Hagio, K.; Okada, H.; Hatanaka, K.C.; Togashi, K.; Matsuno, Y.; Yamashita, H. PIK3CA mutation, reduced AKT serine 473 phosphorylation, and increased ERα serine 167 phosphorylation are positive prognostic indicators in postmenopausal estrogen receptor-positive early breast cancer. Oncotarget 2018, 9(25):17711-17724. [CrossRef]
- amashita, H.; Nishio, M.; Kobayashi, S.; Ando, Y.; Sugiura, H.; Zhang, Z.; Hamaguchi, M.; Mita, K.; Fujii, Y.; Iwase, H. Phosphorylation of estrogen receptor α serine 167 is predictive of response to endocrine therapy and increases postrelapse survival in metastatic breast cancer. Breast Cancer Res 2005, 7(5):R753-64. [CrossRef]
- Jiang, J.; Sarwar, N.; Peston, D.; Kulinskaya, E.; Shousha, S.; Coombes, R.C.; Ali, S. Phosphorylation of estrogen receptor-α at Ser167 is indicative of longer disease-free and overall survival in breast cancer patients. Clin Cancer Res 2007, 13(19):5769-76. [CrossRef]
- Smyth, L.M.; Batist, G.; Meric-Bernstam, F.; Kabos, P.; Spanggaard, I.; Lluch, A.; Jhaveri, K.; Varga, A.; Wong, A.; Schram, A.M.; et al. Selective AKT Kinase inhibitor capivasertib in combination with fulvestrant in PTEN-mutant ER-positive metastatic breast cancer. NPJ Breast Cancer 2021, 7(1):44. [CrossRef]
- Hortobagyi, G.N.; Stemmer, S.M.; Burris, H.A.; Yap, Y.-S.; Sonke, G.S.; Hart, L.; Campone, M.; Petrakova, K.; Winer, E.P.; Janni, W.; et al. Overall survival with ribociclib plus letrozole in advanced breast cancer. New Engl J Med 2022, 386(10):942-950. [CrossRef]
- Johnston, S.R.D.; Toi, M.; O’Shaughnessy, J.; Rastogi, P.; Campone, M.; Neven, P.; Huang, C.S.; Huober, J.; Jaliffe, G.G.; Cicin, I.; et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (MonarchE): Results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol 2023, 24(1):77-90. [CrossRef]
- Watanabe, K.; Niikura, N.; Kikawa, Y.; Oba, M.; Kobayashi, K.; Tada, H.; Ozaki, S.; Toh, U.; Yamamoto, Y.; Tsuneizumi, M.; et al. Fulvestrant plus palbociclib in advanced or metastatic hormone receptor-positive/human epidermal growth factor receptor 2-negative breast cancer after fulvestrant monotherapy: Japan Breast Cancer Research Group-M07 (FUTURE Trial). Breast Cancer Res Treat 2023, 199(2):253-263. [CrossRef]
- Riemsma, R.; Forbes, C.A.; Amonkar, M.M.; Lykopoulos, K.; Diaz, J.R.; Kleijnen, J.; Rea, D.W. Systematic review of lapatinib in combination with letrozole compared with other first-line treatments for hormone receptor positive(HR+) and HER2+ advanced or metastatic breast cancer(MBC). Curr Med Res Opin 2012, 28(8):1263-79.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



