Submitted:
09 September 2024
Posted:
11 September 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Research Material
2.2. Determination of Basic Physicochemical Properties of Fruit
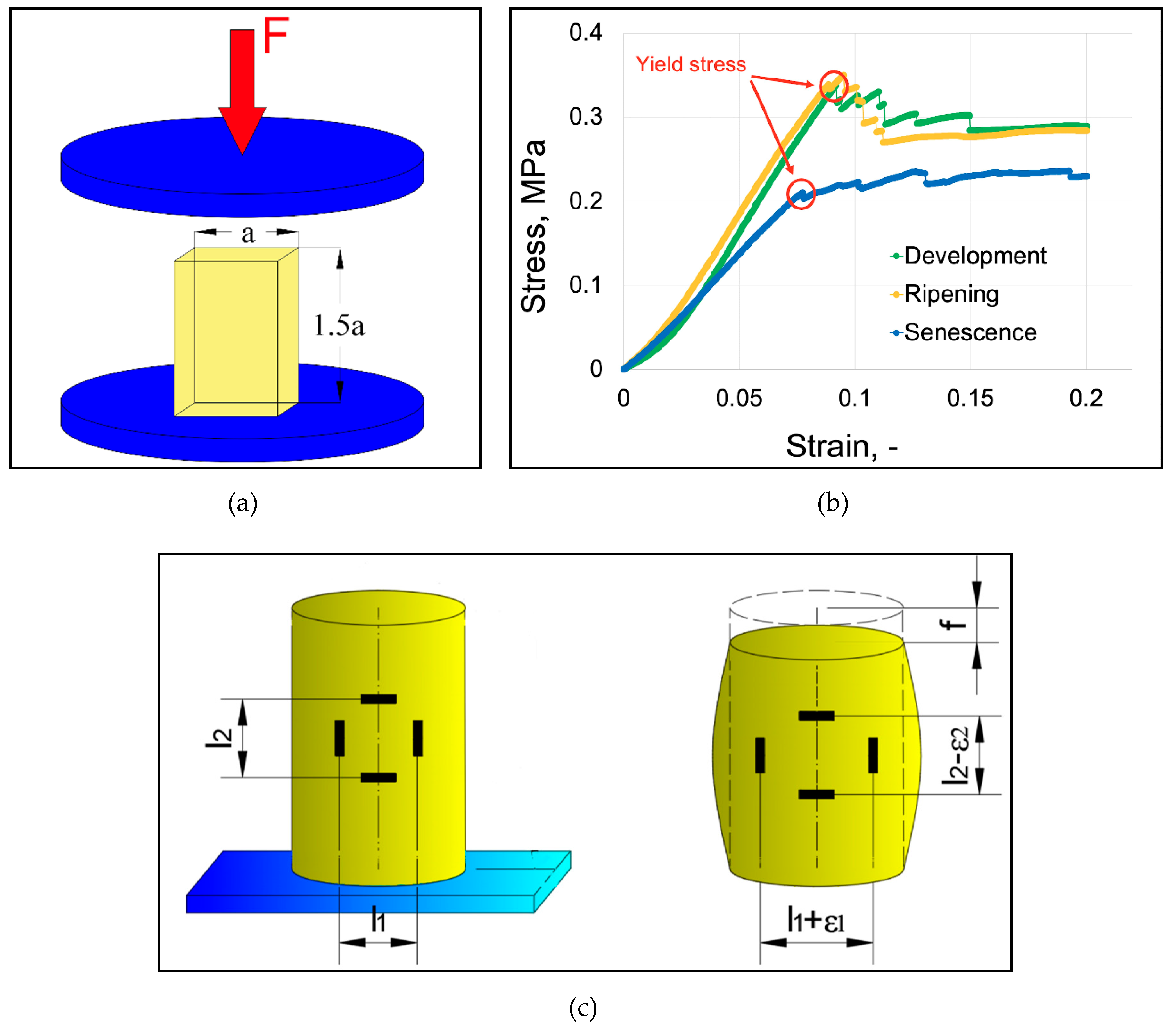
2.3. Determination of Strength Properties of Apple Flesh and Skin
2.3.1. Apple Flesh
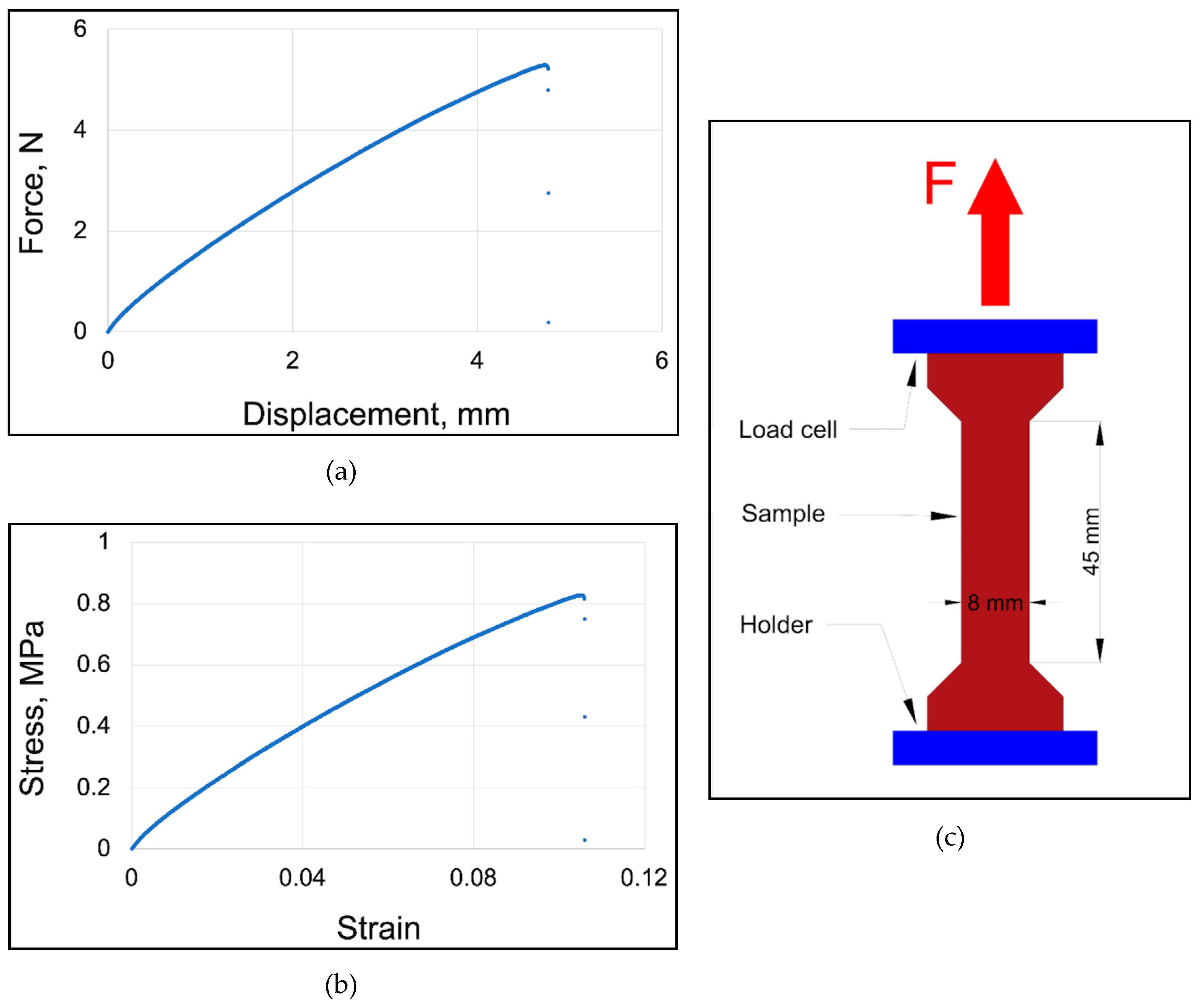
2.3.2. Apple Skin
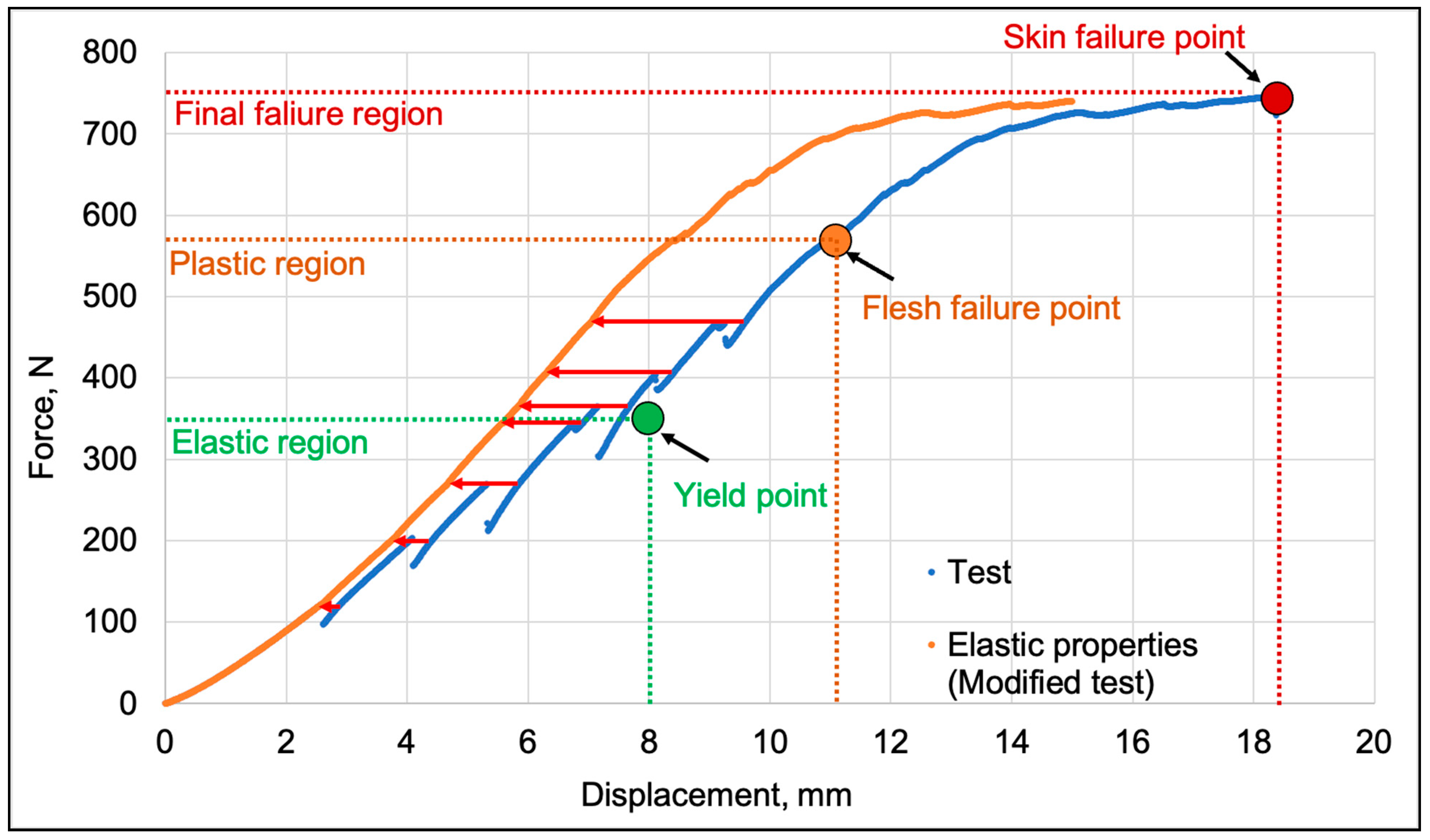
2.4. Determination of Strength Properties of Apple Fruit
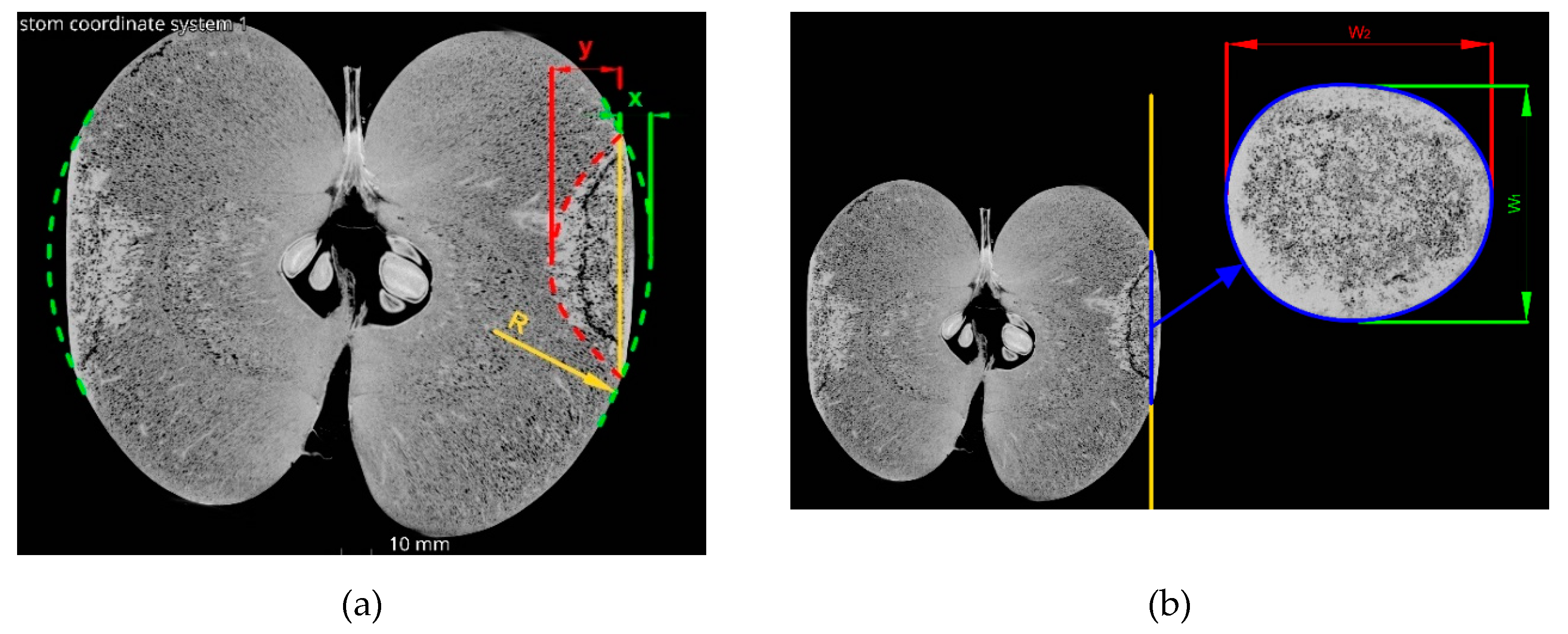
2.5. Determination of the Bruise Geometry and Volume Using Micro-Computed Tomography
3. Results
3.1. Characteristics of the Material Selected for Testing
3.1. Basic Physicochemical Properties of Fruit
3.1. Strength Properties of Apple Flesh and Skin
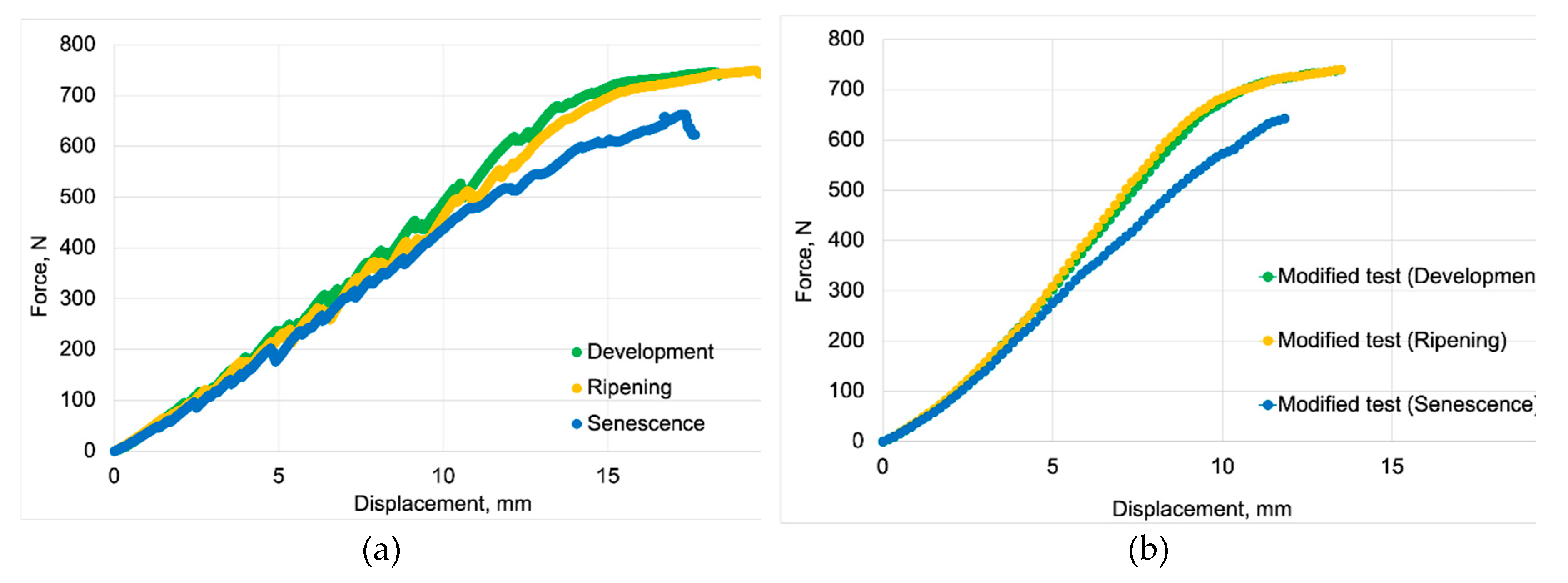
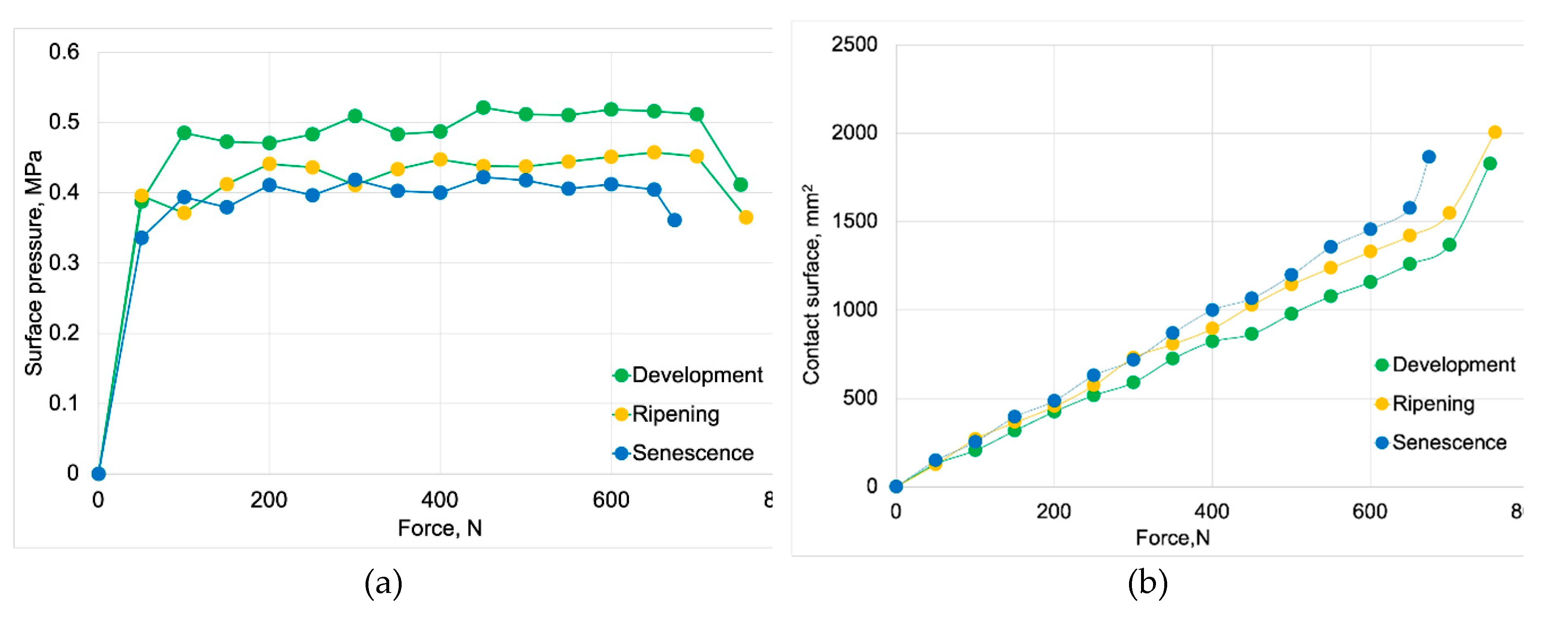
3.1. Strength Properties of Apple Fruit
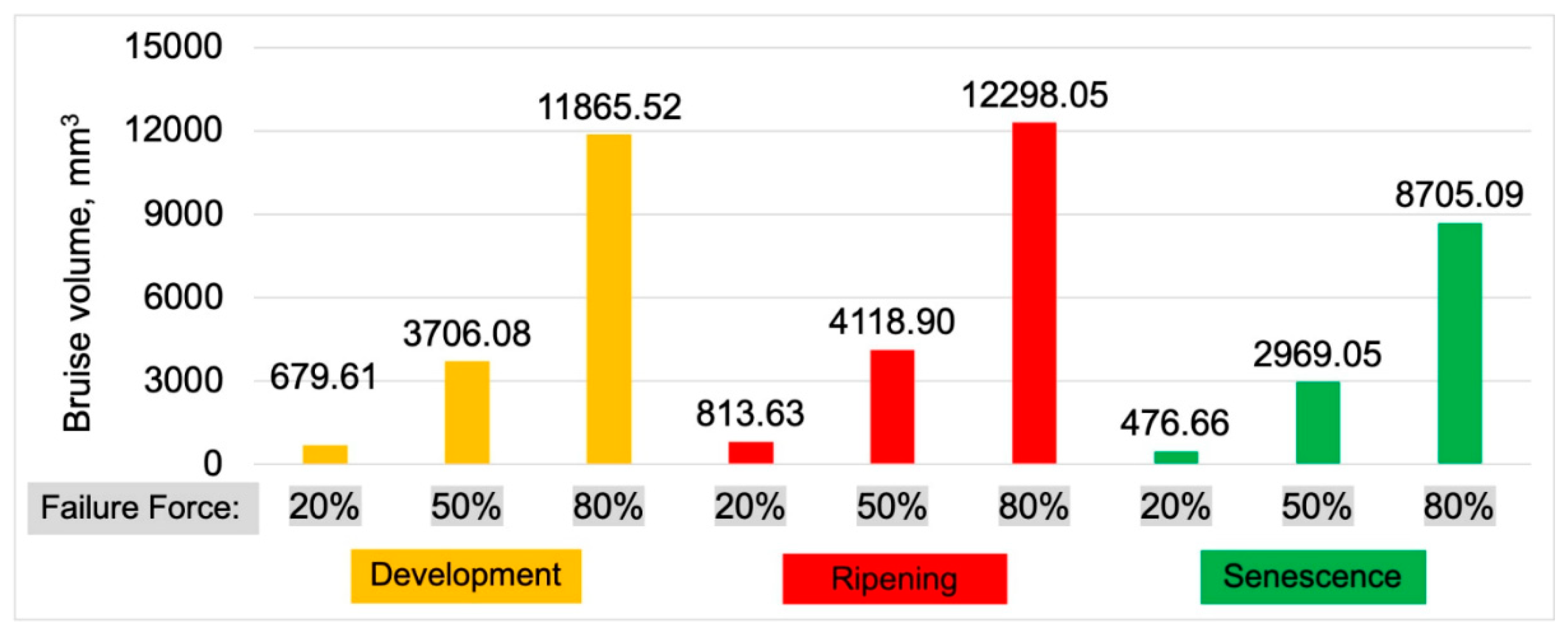
3.1. Bruises Volume
3. Discussion
4. Conclusions
- During the research, a significant difference was found between fruit at the stage of senescence ripeness and fruit at the stage of development and ripening maturity.
- The results of mechanical and Micro-CT tests for specific and verified stages of apple ripeness were determined, providing a complete dataset for building and validating elastic and elastic-plastic FE models. Further conclusions on the superiority of Elastic-Plastic models over Viscoelastic models (for tissue models covering the full range of fruit loading) will be presented in the next article, which will focus exclusively on modeling.
- Results of Micro-Ct and the results of modified and real compression tests of whole fruit have allowed us to formulate the research hypothesis regarding the influence of flesh cracking (characterized by local drops of force), influencing the bruise visibility and detection.
- The research confirmed the need to carefully take into account the maturity stage in mechanical research, and in particular in the construction of MES models for food industry.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Komarnicki, P.; Stopa, R.; Szyjewicz, D.; Młotek, M. Evaluation of Bruise Resistance of Pears to Impact Load. Postharvest Biol Technol 2016, 114, 36–44. [Google Scholar] [CrossRef]
- Kistechok, A.; Wrona, D.; Krupa, T. Effect of Storage Conditions on the Storability and Nutritional Value of New Polish Apples Grown in Central Poland. Agriculture (Switzerland) 2024, 14, 59. [Google Scholar] [CrossRef]
- Ponder, A.; Jariené, E.; Hallmann, E. The Effect of Storage Conditions on the Content of Molecules in Malus Domestica ‘Chopin’ Cv. and Their In Vitro Antioxidant Activity. Molecules 2022, 27, 6979. [Google Scholar] [CrossRef] [PubMed]
- Kistechok, A.; Wrona, D.; Krupa, T. Quality and Nutritional Value of ‘Chopin’ and Clone ‘JB’ in Relation to Popular Apples Growing in Poland. Agriculture (Switzerland) 2022, 12, 1876. [Google Scholar] [CrossRef]
- Noiton, D.A.M.; Alspach, P.A. Founding Clones, Inbreeding, Coancestry, and Status Number of Modern Apple Cultivars; 1996; Vol. 121.
- Zygmunt Hejnowicz Anatomia i Histogeneza Roślin Naczyniowych; Wydawnictwo Naukowe PWN: Warszawa, 2002; Vol. 1.
- Pratt, C. Apple Flower and Fruit: Morphology and Anatomy. 1988.
- Mccann1, M.C.; Roberts, K. Changes in Cell Wall Architecture during Cell Elongation; 1994; Vol. 45.
- Zdunek, A.; Kozioł, A.; Cybulska, J.; Lekka, M.; Pieczywek, P.M. The Stiffening of the Cell Walls Observed during Physiological Softening of Pears. Planta 2016, 243, 519–529. [Google Scholar] [CrossRef]
- Billy, L.; Mehinagic, E.; Royer, G.; Renard, C.M.G.C.; Arvisenet, G.; Prost, C.; Jourjon, F. Relationship between Texture and Pectin Composition of Two Apple Cultivars during Storage. Postharvest Biol Technol 2008. [CrossRef]
- Willats, W.G.T.; Mccartney, L.; Mackie, W.; Knox, J.P. Pectin: Cell Biology and Prospects for Functional Analysis; 2001; Vol. 47;
- Videcoq, P.; Barbacci, A.; Assor, C.; Magnenet, V.; Arnould, O.; Le Gall, S.; Lahaye, M. Examining the Contribution of Cell Wall Polysaccharides to the Mechanical Properties of Apple Parenchyma Tissue Using Exogenous Enzymes. J Exp Bot 2017, 68, 5137–5146. [Google Scholar] [CrossRef]
- Bich, L.; Pradeu, T.; Moreau, J.F. Understanding Multicellularity: The Functional Organization of the Intercellular Space. Front Physiol 2019, 10, 1170. [Google Scholar] [CrossRef]
- Wei, J.; Ma, F.; Shi, S.; Qi, X.; Zhu, X.; Yuan, J. Changes and Postharvest Regulation of Activity and Gene Expression of Enzymes Related to Cell Wall Degradation in Ripening Apple Fruit. Postharvest Biol Technol 2010, 56, 147–154. [Google Scholar] [CrossRef]
- Abbott, J.A. Quality Measurement of Fruits and Vegetables; 1999; Vol. 15;
- Oey, M.; Vanstreels, E.; Debaerdemaeker, J.; Tijskens, B.; Ramon, H.; Nicolaï, B.M. Influence of Turgor on Micromechanical and Structural Properties of Apple Tissue. In Proceedings of the 13th World Congress of Food Science & Technology; EDP Sciences: Les Ulis, France, 2006. [Google Scholar]
- Juxia, W.; Qingliang, C.; Hongbo, L.; Yaping, L. Experimental Research on Mechanical Properties of Apple Peels. Journal of Engineering and Technological Sciences 2015, 47, 688–705. [Google Scholar] [CrossRef]
- Grotte, M.; Duprat, F.; Loonis, D.; Piétri, E. Mechanical Properties of the Skin and the Flesh of Apples. Int J Food Prop 2001, 4, 149–161. [Google Scholar] [CrossRef]
- Ruiz-Altisent, M.; Moreda, G.P. Fruits, Mechanical Properties and Bruise Susceptibility. In Encyclopedia of Earth Sciences Series; 2011; Vol. Part 4.
- Ekrami-Rad, N.; Khazaei, J.; Khoshtaghaza, M.H. Selected Mechanical Properties of Pomegranate Peel and Fruit. Int J Food Prop 2011, 14, 570–582. [Google Scholar] [CrossRef]
- Li, Z.; Thomas, C. Quantitative Evaluation of Mechanical Damage to Fresh Fruits. Trends Food Sci Technol 2014, 35, 138–150. [Google Scholar] [CrossRef]
- Farkas, C.; Petróczki, K.; Fenyvesi, L. Method for Measuring Fruit Failure Caused by Different Mechanical Loads. Hungarian Agricultural Engineering 2016, 51–54. [Google Scholar] [CrossRef]
- Thielen, M.; Speck, T.; Seidel, R. Viscoelasticity and Compaction Behaviour of the Foam-like Pomelo (Citrus Maxima) Peel. J Mater Sci 2013, 48, 3469–3478. [Google Scholar] [CrossRef]
- Fathizadeh, Z.; Aboonajmi, M.; Beygi, S.R.H. Nondestructive Firmness Prediction of Apple Fruit Using Acoustic Vibration Response. Sci Hortic 2020, 262, 109073. [Google Scholar] [CrossRef]
- Opara, U.L.; Pathare, P.B. Bruise Damage Measurement and Analysis of Fresh Horticultural Produce-A Review. Postharvest Biol Technol 2014, 91, 9–24. [Google Scholar] [CrossRef]
- Fadiji, T.; Coetzee, C.; Chen, L.; Chukwu, O.; Opara, U.L. Susceptibility of Apples to Bruising inside Ventilated Corrugated Paperboard Packages during Simulated Transport Damage. Postharvest Biol Technol 2016, 118, 111–119. [Google Scholar] [CrossRef]
- Fadiji, T.; Coetzee, C.; Pathare, P.; Opara, U.L. Susceptibility to Impact Damage of Apples inside Ventilated Corrugated Paperboard Packages: Effects of Package Design. Postharvest Biol Technol 2016, 111, 286–296. [Google Scholar] [CrossRef]
- Stropek, Z.; Gołacki, K. A New Method for Measuring Impact Related Bruises in Fruits. Postharvest Biol Technol 2015, 110, 131–139. [Google Scholar] [CrossRef]
- Diels, E.; van Dael, M.; Keresztes, J.; Vanmaercke, S.; Verboven, P.; Nicolai, B.; Saeys, W.; Ramon, H.; Smeets, B. Assessment of Bruise Volumes in Apples Using X-Ray Computed Tomography. Postharvest Biol Technol 2017, 128, 24–32. [Google Scholar] [CrossRef]
- Van De Looverbosch, T.; Vandenbussche, B.; Verboven, P.; Nicolaï, B. Nondestructive High-Throughput Sugar Beet Fruit Analysis Using X-Ray CT and Deep Learning. Comput Electron Agric 2022, 200, 107228. [Google Scholar] [CrossRef]
- Alamar, M.C.; Vanstreels, E.; Oey, M.L.; Moltó, E.; Nicolaï, B.M. Micromechanical Behaviour of Apple Tissue in Tensile and Compression Tests: Storage Conditions and Cultivar Effect. J Food Eng 2008, 86, 324–333. [Google Scholar] [CrossRef]
- Nicolaï, B.M.; Beullens, K.; Bobelyn, E.; Peirs, A.; Saeys, W.; Theron, K.I.; Lammertyn, J. Nondestructive Measurement of Fruit and Vegetable Quality by Means of NIR Spectroscopy: A Review. Postharvest Biol Technol 2007, 46, 99–118. [Google Scholar] [CrossRef]
- Herremans, E.; Melado-Herreros, A.; Defraeye, T.; Verlinden, B.; Hertog, M.; Verboven, P.; Val, J.; Fernández-Valle, M.E.; Bongaers, E.; Estrade, P.; et al. Comparison of X-Ray CT and MRI of Watercore Disorder of Different Apple Cultivars. Postharvest Biol Technol 2014, 87, 42–50. [Google Scholar] [CrossRef]
- Wang, L.J.; Zhang, Q.; Song, H.; Wang, Z.W. Mechanical Damage of ‘Huangguan’ Pear Using Different Packaging under Random Vibration. Postharvest Biol Technol 2022, 187, 111847. [Google Scholar] [CrossRef]
- Bejaei, M.; Stanich, K.; Cliff, M.A. Modelling and Classification of Apple Textural Attributes Using Sensory, Instrumental and Compositional Analyses. Foods 2021, 10, 384. [Google Scholar] [CrossRef]
- Stopa, R.; Szyjewicz, D.; Komarnicki, P.; Kuta, Ł. Determining the Resistance to Mechanical Damage of Apples under Impact Loads. Postharvest Biol Technol 2018, 146, 79–89. [Google Scholar] [CrossRef]
- Tomala, K.; Grzęda, M.; Guzek, D.; Głąbska, D.; Gutkowska, K. The Effects of Preharvest 1-Methylcyclopropene (1-MCP) Treatment on the Fruit Quality Parameters of Cold-Stored ‘Szampion’ Cultivar Apples. Agriculture (Switzerland) 2020, 10, 80. [Google Scholar] [CrossRef]
- Pijanowski, E.; Mrożewski, S.; Horubała, A.; Jarczyk, A. Technologia Produktów Owocowych i Warzywnych; PWRiL: Warszawa, 1973. [Google Scholar]
- Sawicka, M.; Latocha, P.; Łata, B. Peel to Flesh Bioactive Compounds Ratio Affect Apple Antioxidant Potential and Cultivar Functional Properties. Agriculture (Switzerland) 2023, 13, 478. [Google Scholar] [CrossRef]
- Przybyłko, S.; Kowalczyk, W.; Wrona, D. Article the Effect of Mycorrhizal Fungi and Pgpr on Tree Nutritional Status and Growth in Organic Apple Production. Agronomy 2021, 11, 1402. [Google Scholar] [CrossRef]
- Ji, W.; Qian, Z.; Xu, B.; Tang, W.; Li, J.; Zhao, D. Grasping Damage Analysis of Apple by End-Effector in Harvesting Robot. J Food Process Eng 2017, 40, e12589. [Google Scholar] [CrossRef]
- Jia, W.; Zhang, Y.; Lian, J.; Zheng, Y.; Zhao, D.; Li, C. Apple Harvesting Robot under Information Technology: A Review. Int J Adv Robot Syst 2020, 17, 1729881420925310. [Google Scholar] [CrossRef]
- Roman Stopa Modelowanie Deformacji Korzenia Marchwi w Warunkach Obciążeń Skupionych Metodą Elementów Skończonych. Monografia, Wydawnictwo Uniwersytetu Przyrodniczego we Wrocławiu: Wrocław, 2010.
- Murase, H.; Merva, G.E.; Segerlind, L.J.; Member Member Member, A.; Asae, A.; Abstract, A. Variation of Young’s Modulus of Potato as A Function of Water Potential, 1980.
| Stage of ripeness |
Density, kg⋅m3 |
Average Diameter, mm |
Average Height, mm |
Water content, % |
|---|---|---|---|---|
| Development | 995.16±10.96 | 76.95±1.16 | 64.56±2.17 | 85.57±4.20 |
| Ripening | 1037.21±18.56 | 78.48±1.83 | 63.79±3.44 | 84.10±1.80 |
| Senescence | 1047.22±20.24 | 77.88±1.71 | 65.51±3.58 | 83.42±4.30 |
| Stage of ripeness |
Firmness, kG | Soluble solids content, % |
Starch index, - |
Streif index, - |
Ethylene concentration, µLC2H4 ∙ L-1 |
Malic acid content, % |
Pectin content, g∙100g s.m -1 [%] |
|---|---|---|---|---|---|---|---|
| Development | 7.4 ±0.24 | 11.6±0.49 | 5±0.60 | 0.12 | 0.1 | 0.83 | 9.53 |
| Ripening | 6.9±0.16 | 14.00±0.25 | 7±0.40 | 0.07 | 12 | 0.78 | 7.20 |
| Senescence | 5.8±0.80 | 14.6±0.97 | 9±1.10 | 0.04 | 31 | 0.66 | 4.07 |
|
Stage of ripeness |
Skin | Flesh | |
| Modulus of Elasticity, MPa |
Modulus of Elasticity, MPa |
Poisons Ratio, - |
|
| Development | 10.78±2.26 | 5.04±0.75 | 0.25±0.03 |
| Ripening | 7.18±1.29 | 5.12±0.86 | 0.21±0.07 |
| Senescence | 5.23±1.86 | 3.85±0.78 | 0.35±0.10 |
| Elastic region | ||||
| Stage of ripeness | Yield point, N |
Displacement, mm |
Contact Surface, mm2 | Surface Pressure, MPa |
| Development | 344.72±3.45 | 7.31±0.29 | 723.41±76.67 | 0.49±0.039 |
| Ripening | 339.14±5.96 | 7.55±0.28 | 806.24±25.99 | 0.44±0.014 |
| Senescence | 262.41±2.58 | 4.73±0.38 | 630.65±49.82 | 0.40±0.034 |
| Plastic region | ||||
| Stage of ripeness | Flesh failure point, N | Displacement, mm |
Contact Surface, mm2 | Surface Pressure, MPa |
| Development | 646.15±8.47 | 12.98±0.34 | 1258.8.4±79.98 | 0.52±0.031 |
| Ripening | 658.76±10.14 | 13.16±0.57 | 1419.60±116.64 | 0.46±0.037 |
| Senescence | 576.95±67.24 | 12.99±1.27 | 1355.80±213.119 | 0.41±0.065 |
| Final failure | ||||
| Stage of ripeness | Skin failure point, N | Displacement, mm |
Contact Surface, mm2 | Surface Pressure, MPa |
| Development | 751.29±8.47 | 19.02±0.76 | 1826.00±163.24 | 0.41±0.038 |
| Ripening | 757.50±10.14 | 20.42±1.17 | 2005.20±123.45 | 0.38±0.014 |
| Senescence | 674.18±67.24 | 17.95±2.41 | 1865.20±212.65 | 0.36±0.045 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




