Submitted:
09 September 2024
Posted:
10 September 2024
You are already at the latest version
Abstract
Keywords:
Introduction
- The type of microorganism (e.g., with high, medium, or low urease activity).
- The condition of the microorganism (e.g., whether it is a live cell, cell fraction, or isolated enzyme).
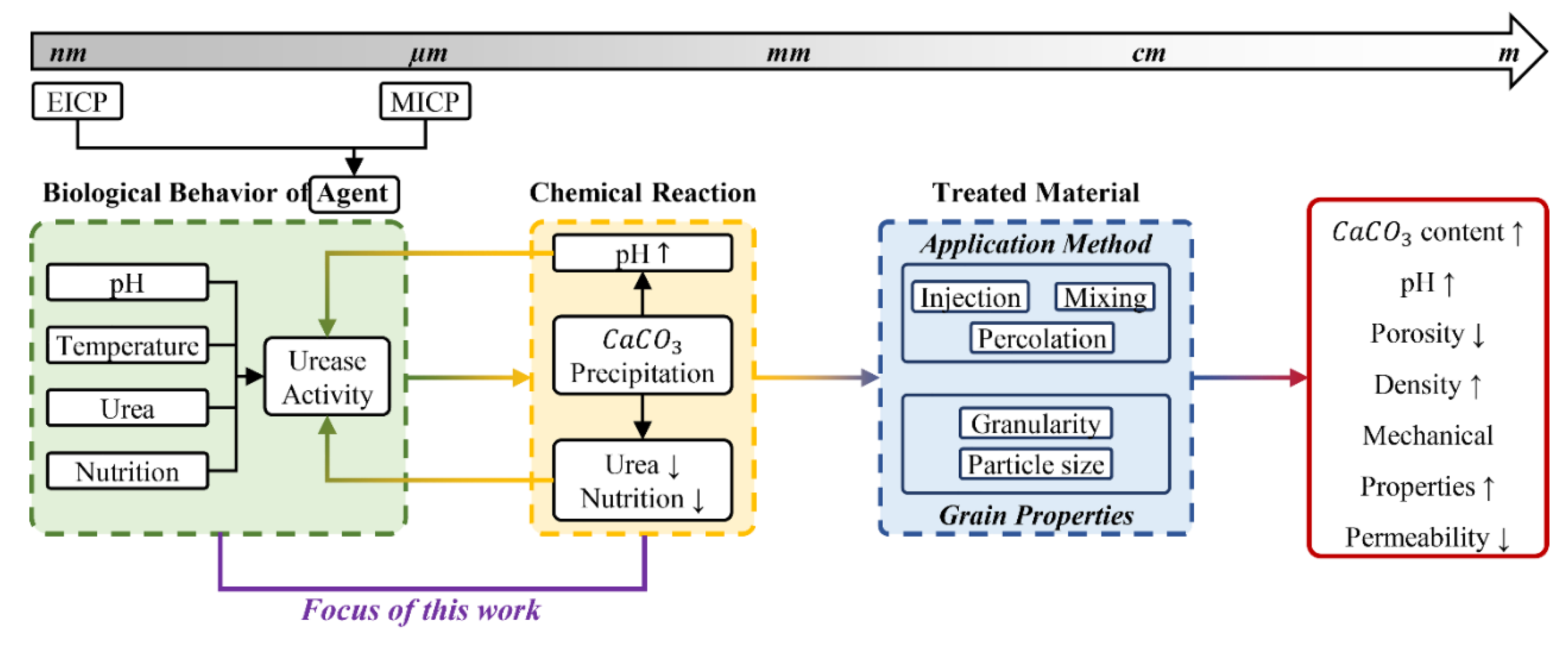
- The method of application (e.g., mixing, spraying, injection, or percolation).
- The environmental conditions, including pH, temperature, and nutrition media, as well as the presence of nucleation sites for bacterial growth (grain size).
Theoretical Background
Governing Parameters
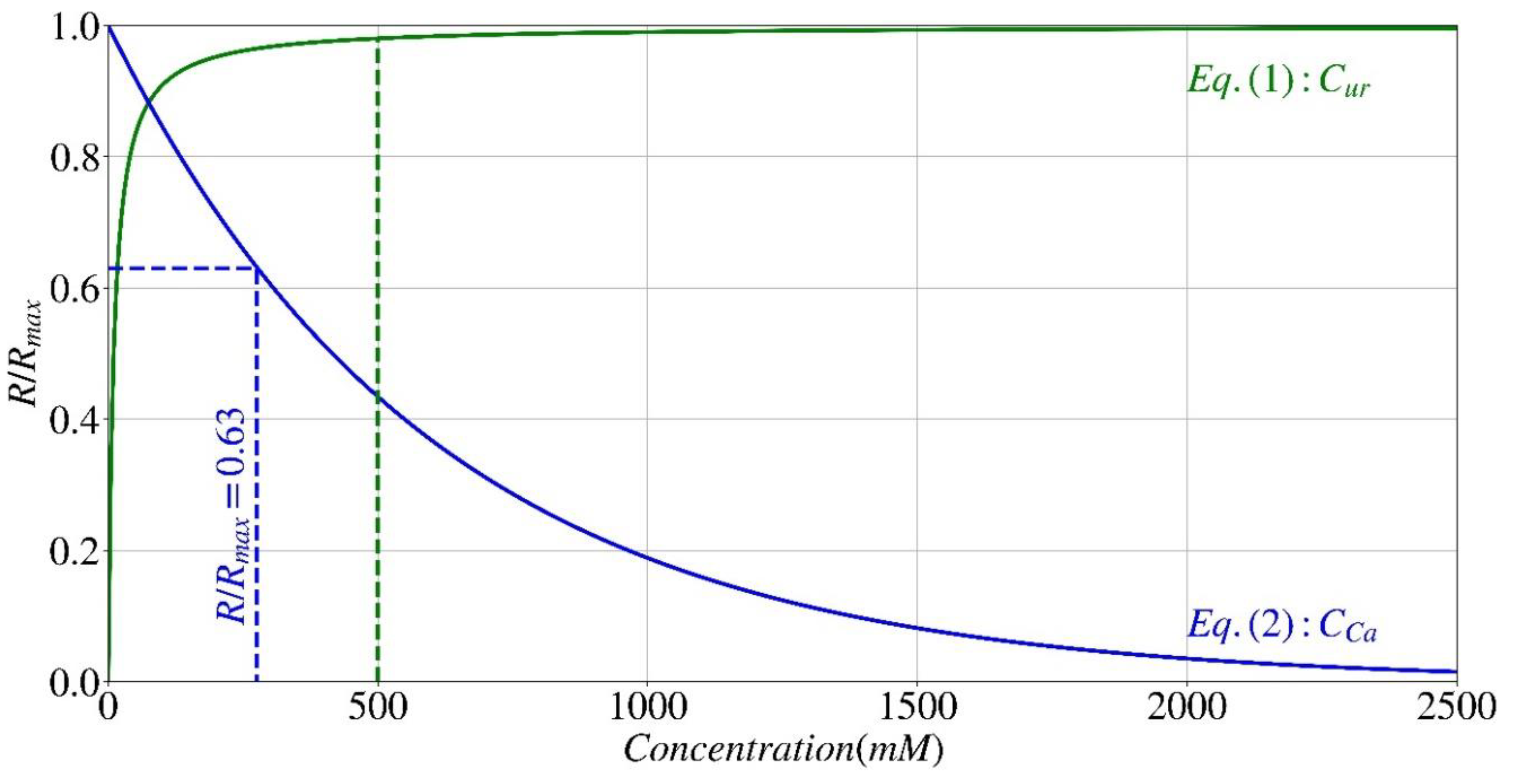
Effect of Urea and Calcium
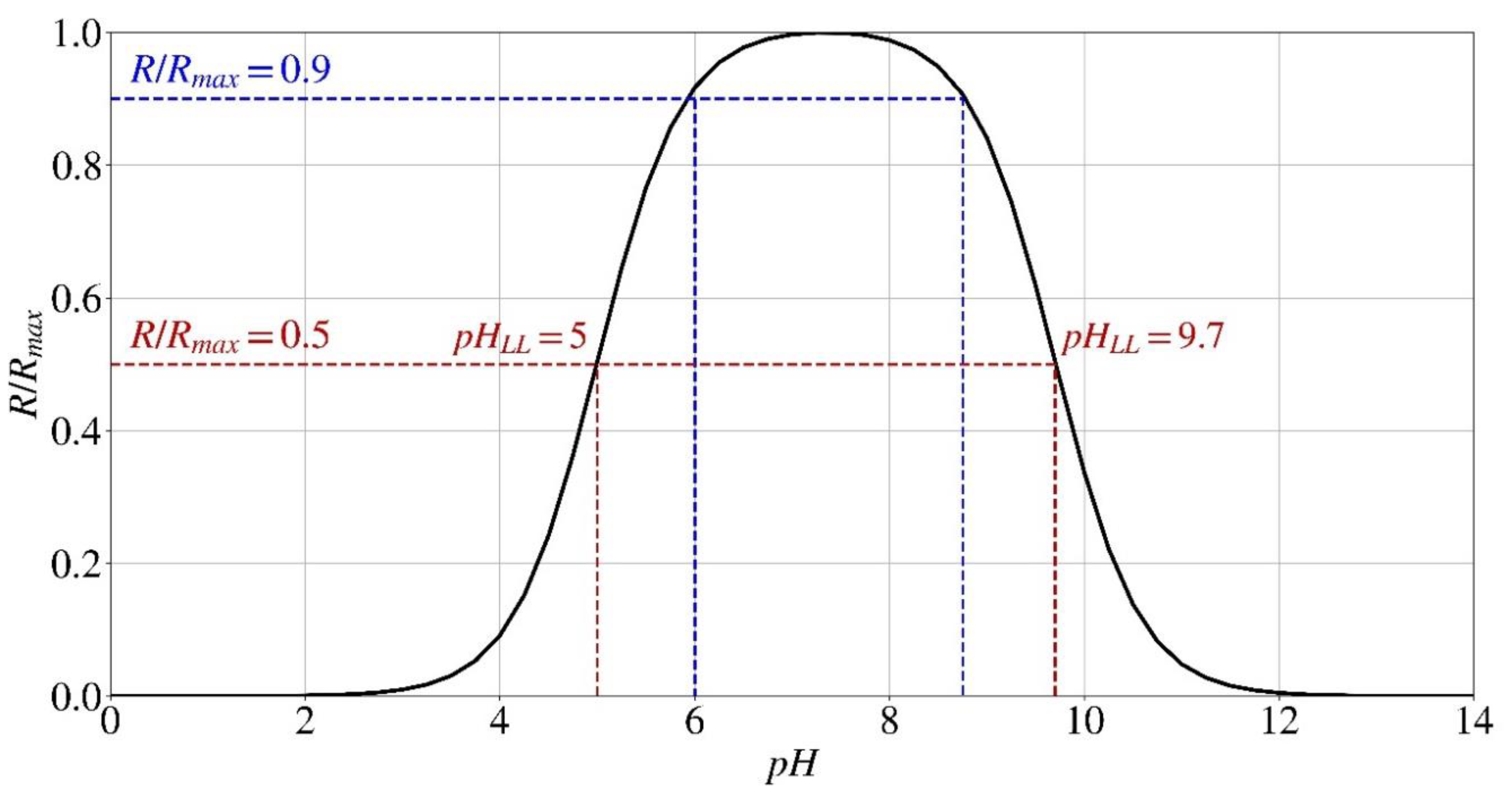
Effect of pH
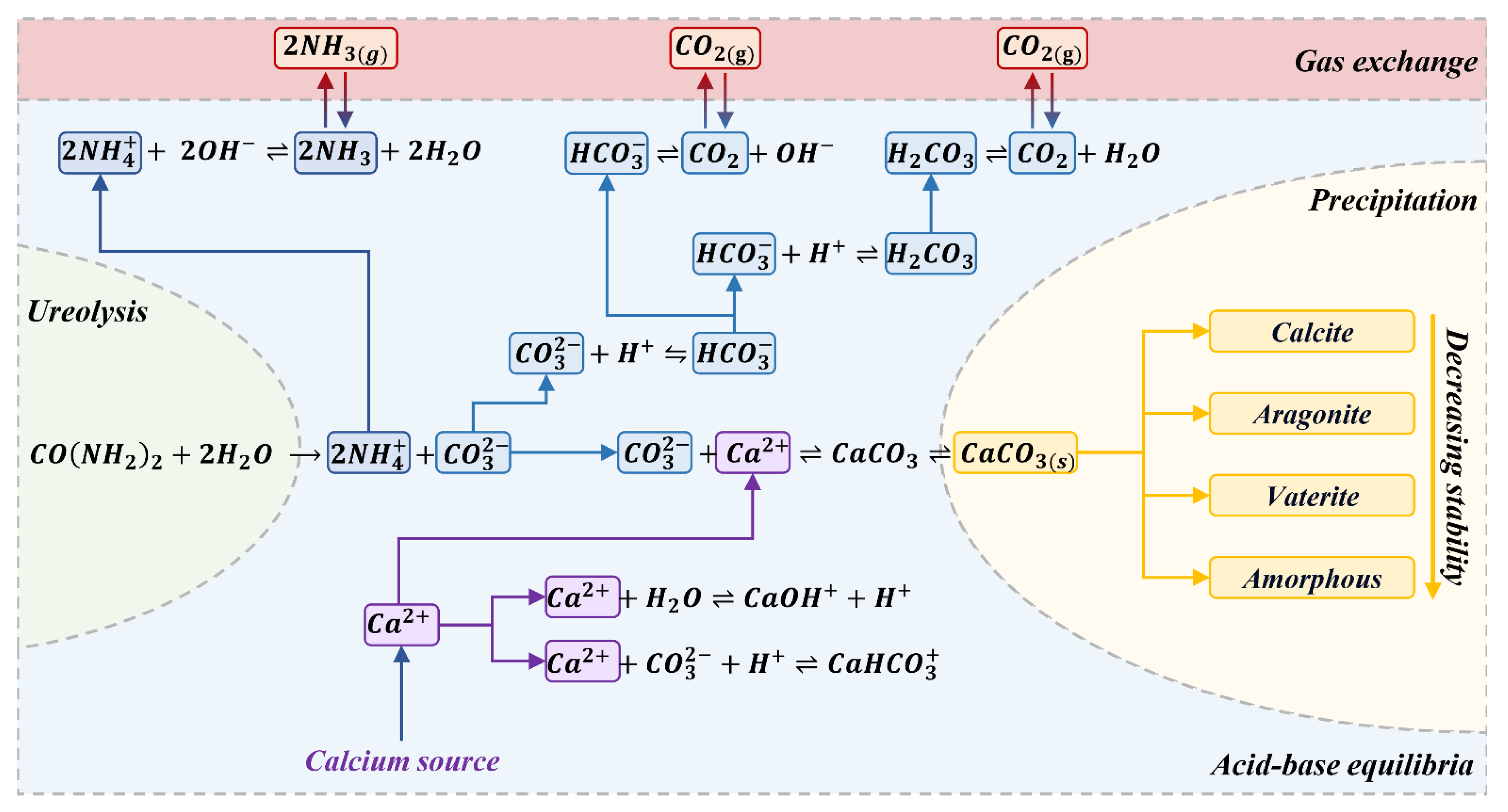
Chemical Reactions
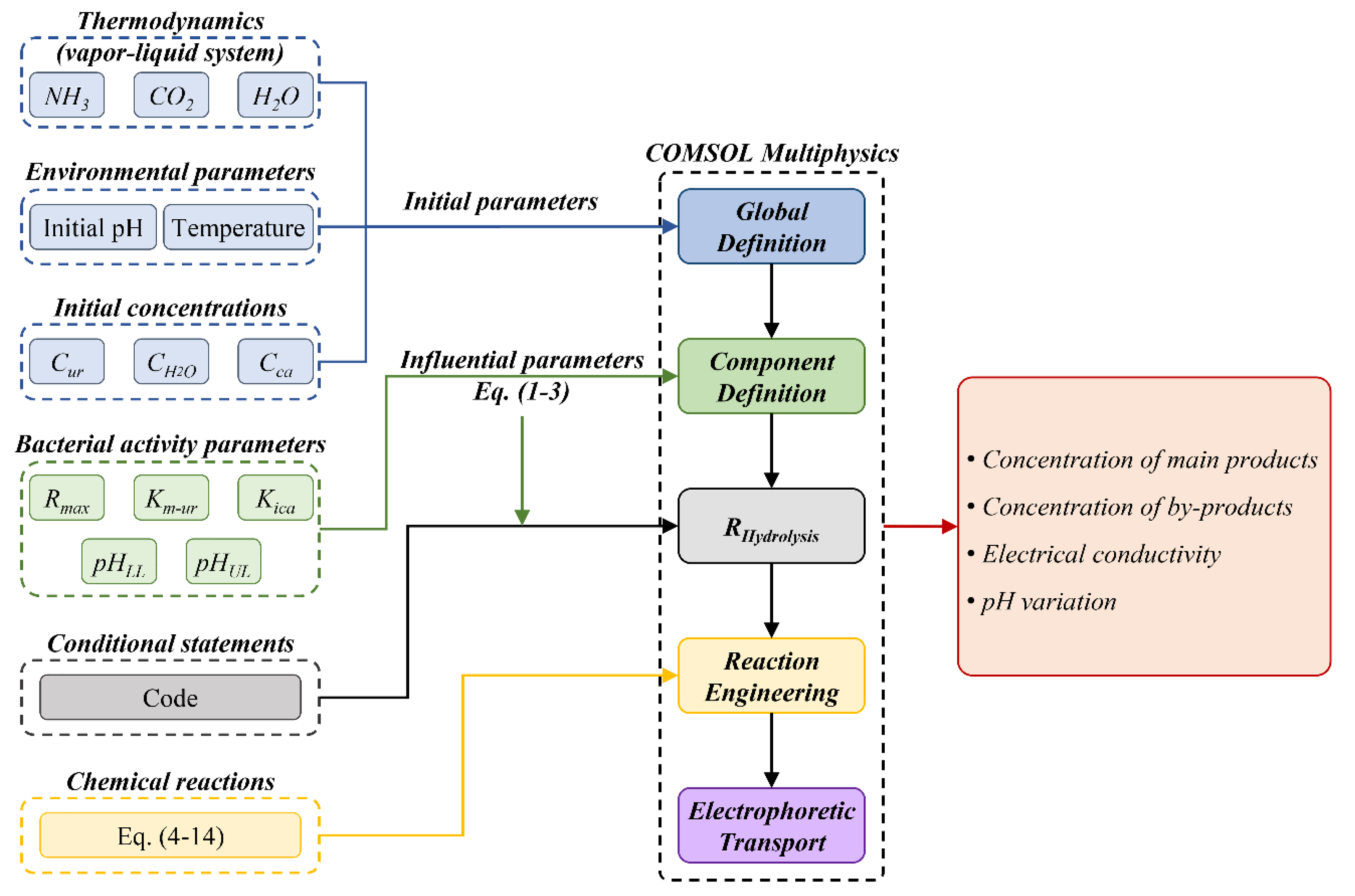
Computational Model's Structure
- If the enzyme is denaturated by the environment's pH (pH < 3.5 or > 12), no precipitation occurs, and the hydrolysis rate is zero.
- If the pH value is within the suitable range of 3.5 to 12 and the normalized hydrolysis rate (R/Rmax) from the effect of pH (Eq. (3)) is above 0.5 and the one from the effect of calcium concentration is above 0.67 (Eq. (2)), the hydrolysis rate is mainly affected by the urea concentration (Eq. (1)), which triggers the reactions.
- If any of these requirements is not met, the average value of the normalized hydrolysis rate (R/Rmax) will be considered among the three functions.
- The process concludes when the system achieves charge balance (Eq. (15)) for all species and the concentration of urea is too low to initiate ureolysis.
Results and Discussion
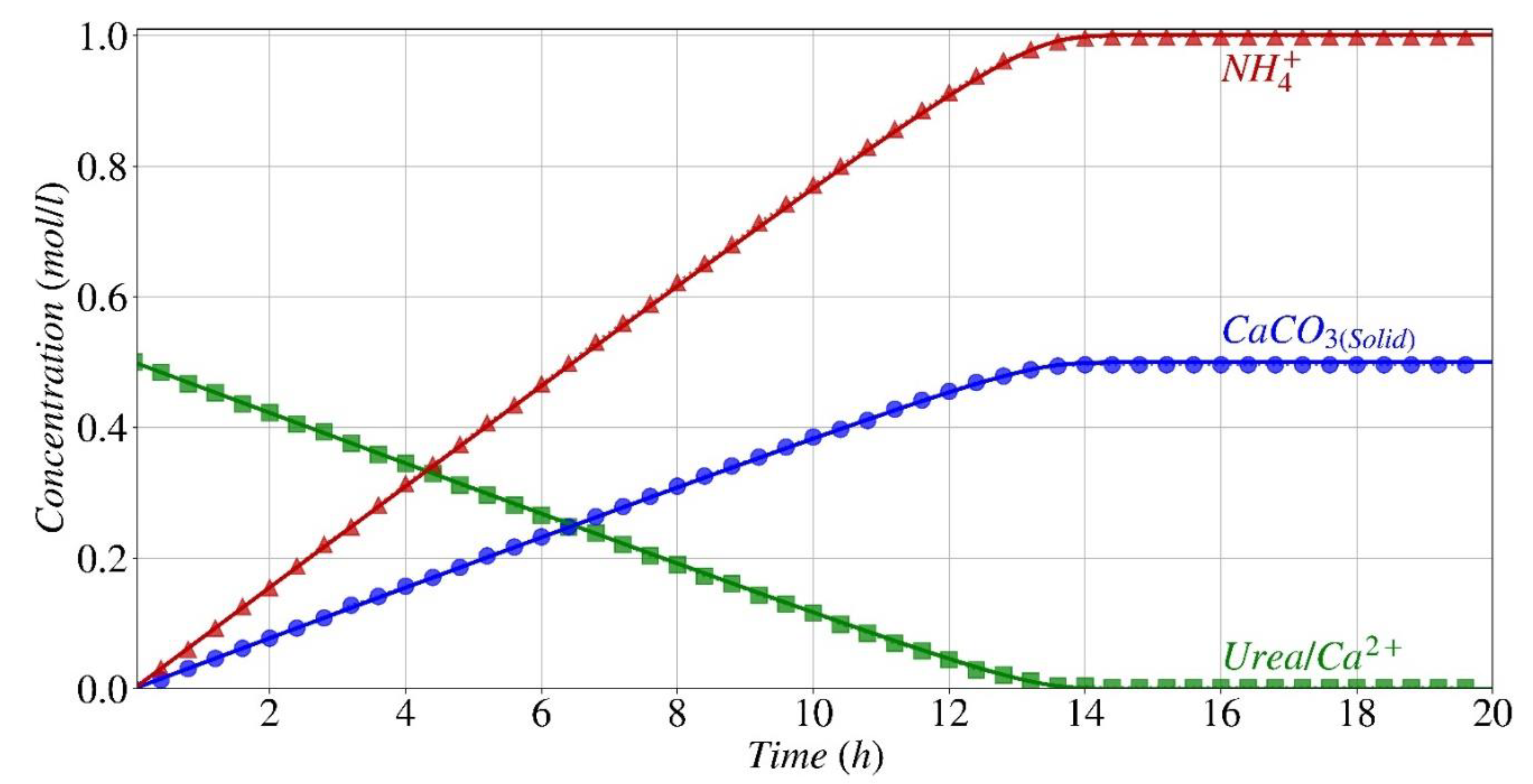
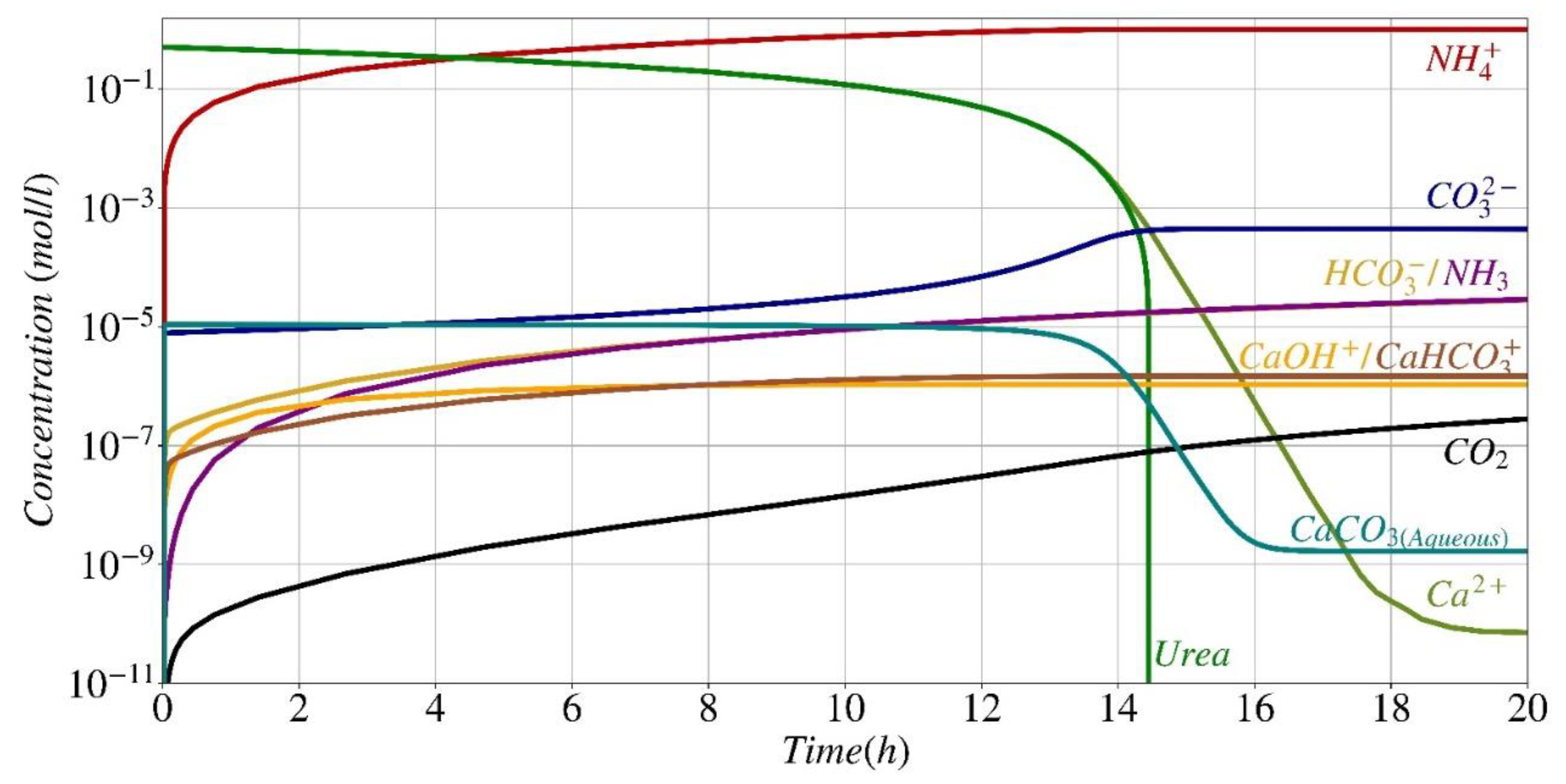
Reactants, Main Products, and By-Products Concentration
Electrical Conductivity
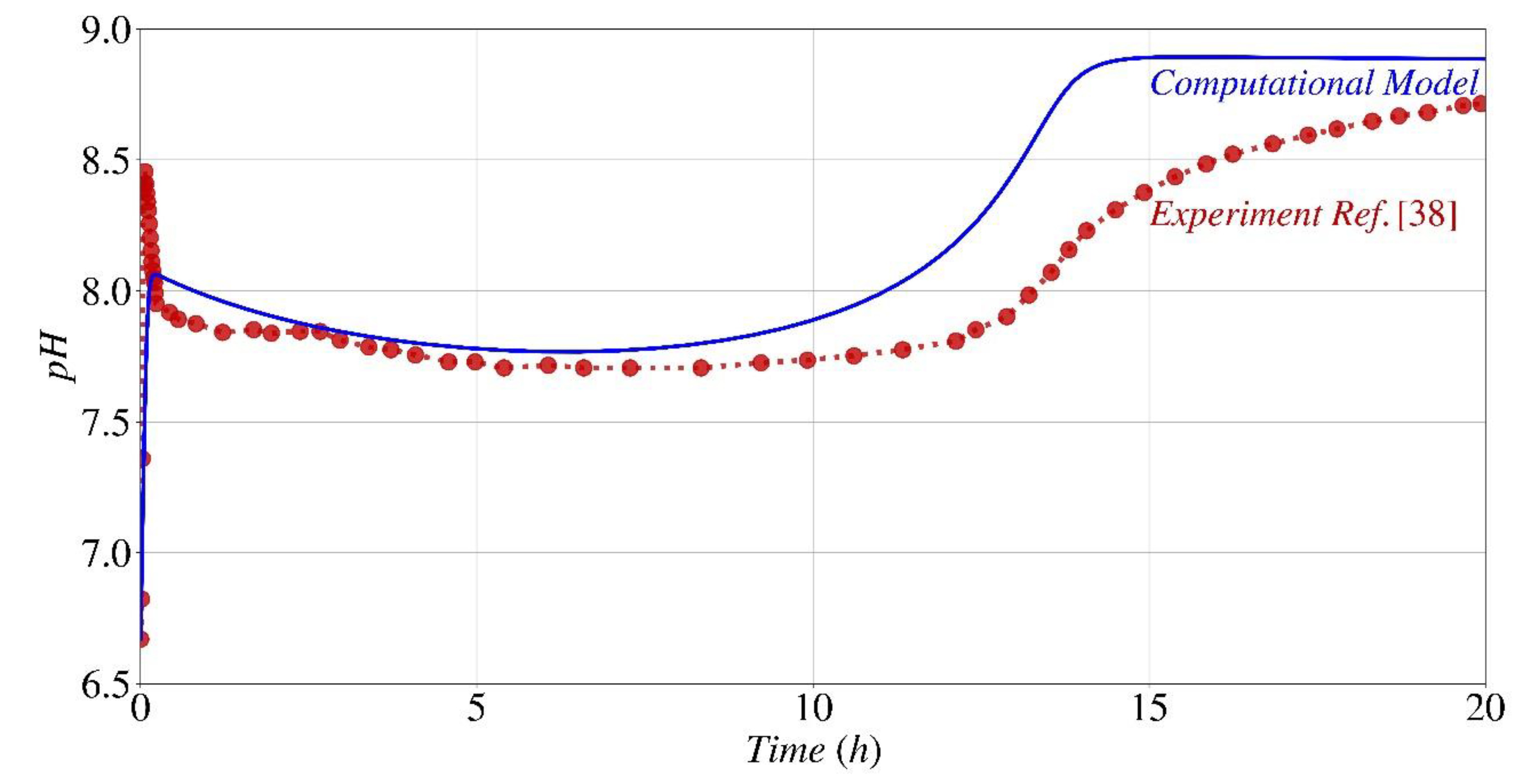
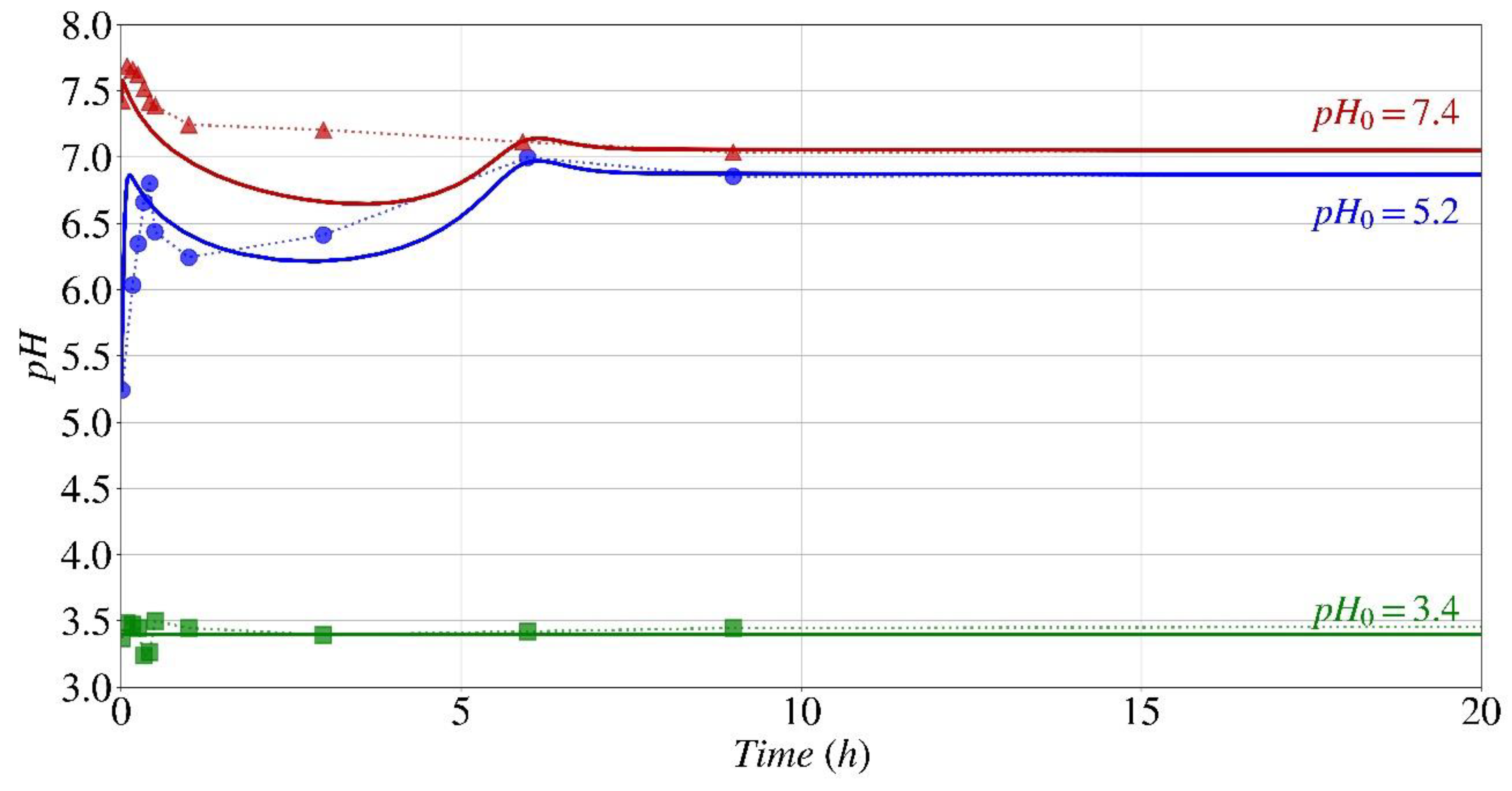
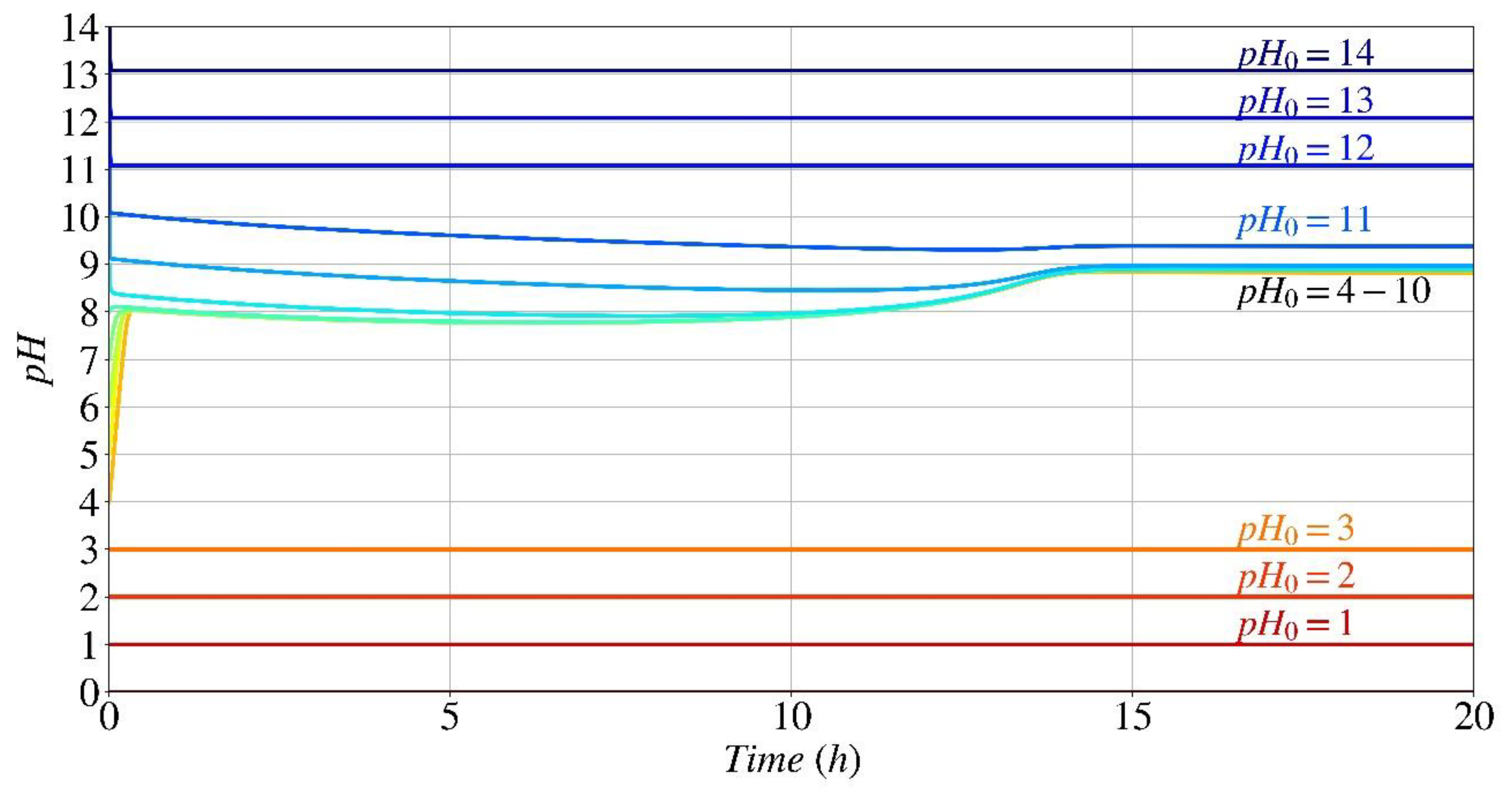
pH Fluctuation
Conclusions
- The established model precisely forecasts the pattern and quantity of consumption of the main reactants (urea and calcium), as well as the production of the resulting products (precipitated calcium carbonate) and by-products (ammonium).
- Additionally, it has the capability to predict the amount of all other secondary by-products Eqs. (6)-(13).
- The model has the capability to forecast the progression of electrical conductivity in the system during the biocementation process.
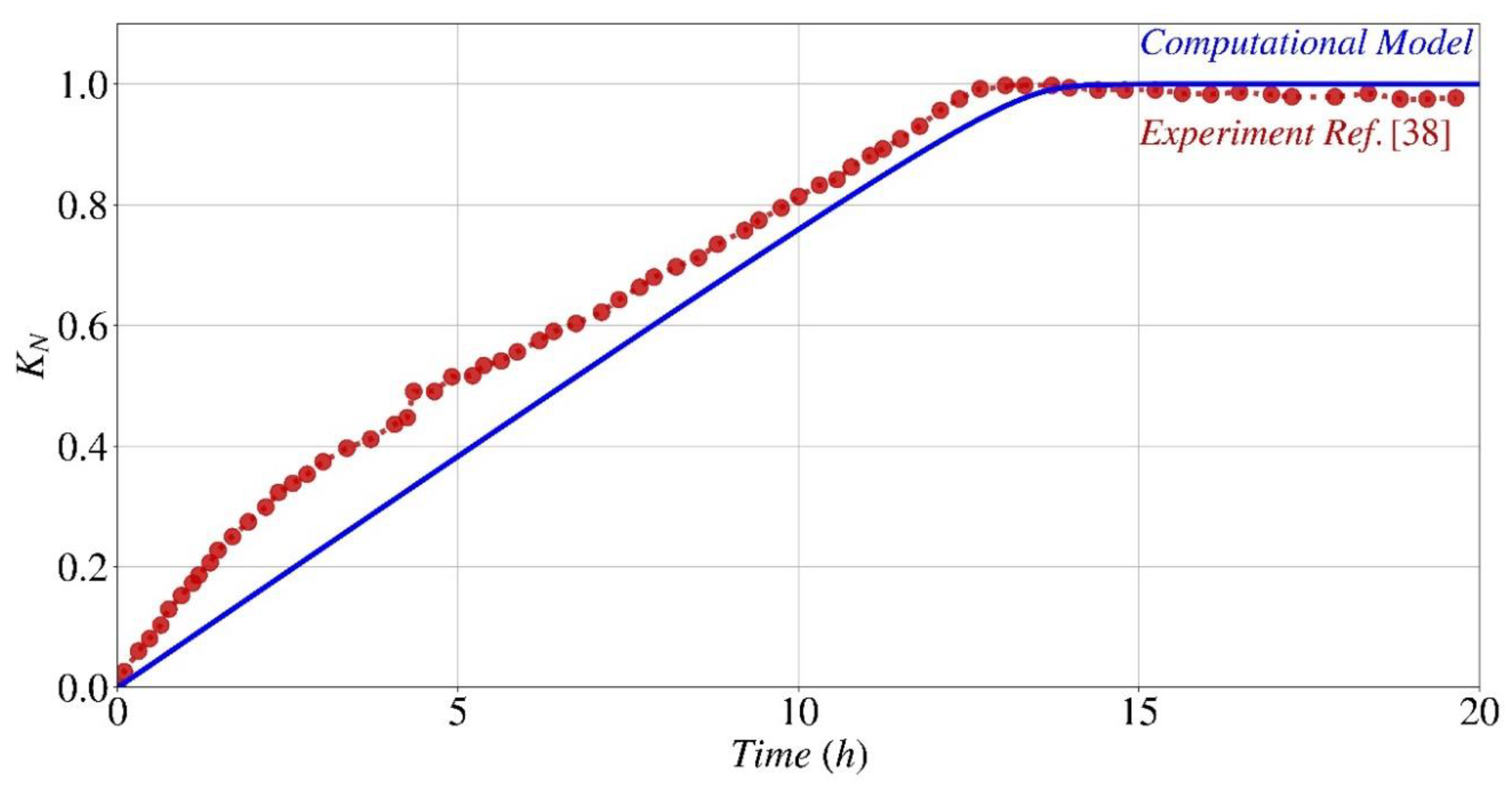
- The parametric analysis conducted using the computational model and experimental findings shown that the provided model and generated code had the capability to accurately predict the kinetics of the biocementation process under various starting pH conditions.
- The parametric study indicates that the optimal initial pH range for the formation of calcium carbonate and the subsequent pH changes over time, when utilizing Sporosarcina pasteurii for bio-cementation, is between 4 and 10.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- R. Devrani, A.A. Dubey, K. Ravi, L. Sahoo. Applications of bio-cementation and bio-polymerization for aeolian erosion control. J. Arid Environ. 2021, 187, 104433. [Google Scholar] [CrossRef]
- S.M. Fattahi, A. Soroush, N. Huang. Biocementation Control of Sand against Wind Erosion. J. Geotech. Geoenvironmental Eng. 2020, 146, 04020045. [Google Scholar] [CrossRef]
- A.A. Dubey, R. Devrani, K. Ravi, N.K. Dhami, A. Mukherjee, L. Sahoo. Experimental investigation to mitigate aeolian erosion via biocementation employed with a novel ureolytic soil isolate. Aeolian Res. 2021, 52, 100727. [Google Scholar] [CrossRef]
- S.M.A. Zomorodian, H. Ghaffari, B.C. O’Kelly. Stabilisation of crustal sand layer using biocementation technique for wind erosion control. Aeolian Res. 2019, 40, 34–41. [Google Scholar] [CrossRef]
- A.I. Omoregie, E.A. Palombo, D.E.L. Ong, P.M. Nissom. A feasible scale-up production of Sporosarcina pasteurii using custom-built stirred tank reactor for in-situ soil biocementation. Biocatal. Agric. Biotechnol. 2020, 24, 101544. [Google Scholar] [CrossRef]
- K.M.N.S. Wani, B.A. Mir. An Experimental Study on the Bio-cementation and Bio-clogging Effect of Bacteria in Improving Weak Dredged Soils. Geotech. Geol. Eng. 2021, 39, 317–334. [Google Scholar] [CrossRef]
- A.M. Sharaky, N.S. Mohamed, M.E. Elmashad, N.M. Shredah. Application of microbial biocementation to improve the physico-mechanical properties of sandy soil. Constr. Build. Mater. 2018, 190, 861–869. [Google Scholar] [CrossRef]
- K. Xu, M. Huang, C. Xu, J. Zhen, G. Jin, H. Gong. Assessment of the bio-cementation effect on shale soil using ultrasound measurement. Soils Found. 2023, 63, 101249. [Google Scholar] [CrossRef]
- A.A. Dubey, K. Ravi, A. Mukherjee, L. Sahoo, M.A. Abiala, N.K. Dhami. Biocementation mediated by native microbes from Brahmaputra riverbank for mitigation of soil erodibility, Sci. Rep. 2021, 11, 15250. [Google Scholar] [CrossRef]
- L. Cheng, M.A. Shahin, R. Cord-Ruwisch. Bio-cementation of sandy soil using microbially induced carbonate precipitation for marine environments. Géotechnique 2014, 64, 1010–1013. [Google Scholar] [CrossRef]
- H. Abdel-Aleem, T. Dishisha, A. Saafan, A.A. AbouKhadra, Y. Gaber. Biocementation of soil by calcite/aragonite precipitation using Pseudomonas azotoformans and Citrobacter freundii derived enzymes. RSC Adv. 2019, 9, 17601–17611. [Google Scholar] [CrossRef] [PubMed]
- J. Yin, J.-X. Wu, K. Zhang, M.A. Shahin, L. Cheng. Comparison between MICP-Based Bio-Cementation Versus Traditional Portland Cementation for Oil-Contaminated Soil Stabilisation. Sustainability 2022, 15, 434. [Google Scholar] [CrossRef]
- P.J. Venda Oliveira, J.P.G. Neves. Effect of Organic Matter Content on Enzymatic Biocementation Process Applied to Coarse-Grained Soils. J. Mater. Civ. Eng. 2019, 31, 04019121. [Google Scholar] [CrossRef]
- V.S. Whiffin, L.A. van Paassen, M.P. Harkes. Microbial carbonate precipitation as a soil improvement technique. Geomicrobiol. J. 2007, 24, 417–423. [Google Scholar] [CrossRef]
- D. Mujah, M.A. Shahin, L. Cheng. State-of-the-Art Review of Biocementation by Microbially Induced Calcite Precipitation (MICP) for Soil Stabilization. Geomicrobiol. J. 2017, 34, 524–537. [Google Scholar] [CrossRef]
- S. Khoshtinat. Advancements in Exploiting Sporosarcina pasteurii as Sustainable Construction Material: A Review. Sustainability 2023, 15, 13869. [Google Scholar] [CrossRef]
- A. I. Omoregie, E.A. Palombo, P.M. Nissom. Bioprecipitation of calcium carbonate mediated by ureolysis: A review. Environ. Eng. Res. 2021, 26. [Google Scholar] [CrossRef]
- D. M. Iqbal, L.S. Wong, S.Y. Kong. Bio-Cementation in Construction Materials: A Review. Materials 2021, 14, 2175. [Google Scholar] [CrossRef]
- N.N.T. Huynh, K. Imamoto, C. Kiyohara. A Study on Biomineralization using Bacillus Subtilis Natto for Repeatability of Self-Healing Concrete and Strength Improvement. J. Adv. Concr. Technol. 2019, 17, 700–714. [Google Scholar] [CrossRef]
- M. Bagga, C. Hamley-Bennett, A. Alex, B.L. Freeman, I. Justo-Reinoso, I.C. Mihai, S. Gebhard, K. Paine, A.D. Jefferson, E. Masoero, I.D. Ofiţeru. Advancements in bacteria based self-healing concrete and the promise of modelling. Constr. Build. Mater. 2022, 358, 129412. [Google Scholar] [CrossRef]
- N.N.T. Huynh, N.M. Phuong, N.P.A. Toan, N.K. Son. Bacillus Subtilis HU58 Immobilized in Micropores of Diatomite for Using in Self-healing Concrete. Procedia Eng. 2017, 171, 598–605. [Google Scholar] [CrossRef]
- N.N.T. Huynh, K. Imamoto, C. Kiyohara. Biomineralization Analysis and Hydration Acceleration Effect in Self-healing Concrete using Bacillus subtilis natto, J. Adv. Concr. Technol. 2022, 20, 609–623. [Google Scholar] [CrossRef]
- N. Huynh, K. Imamoto, C. Kiyohara, Compressive Strength Improvement and Water Permeability of Self-Healing Concrete Using Bacillus Subtilis Natto, in: XV Int. Conf. Durab. Build. Mater. Compon. EBook Proc., CIMNE, 2020. [CrossRef]
- S. Joshi, S. Goyal, M.S. Reddy. Corn steep liquor as a nutritional source for biocementation and its impact on concrete structural properties. J. Ind. Microbiol. Biotechnol. 2018, 45, 657–667. [Google Scholar] [CrossRef] [PubMed]
- M. Wu, X. Hu, Q. Zhang, D. Xue, Y. Zhao. Growth environment optimization for inducing bacterial mineralization and its application in concrete healing, Constr. Build. Mater. 2019, 209, 631–643. [Google Scholar] [CrossRef]
- S. Krishnapriya, D.L. Venkatesh Babu, P.A. G. Isolation and identification of bacteria to improve the strength of concrete. Microbiol. Res. 2015, 174, 48–55. [Google Scholar] [CrossRef]
- M.J. Castro-Alonso, L.E. Montañez-Hernandez, M.A. Sanchez-Muñoz, M.R. Macias Franco, R. Narayanasamy, N. Balagurusamy. Microbially Induced Calcium Carbonate Precipitation (MICP) and Its Potential in Bioconcrete: Microbiological and Molecular Concepts, Front. Mater. 2019, 6, 126. [Google Scholar] [CrossRef]
- H. Porter, A. Mukherjee, R. Tuladhar, N.K. Dhami. Life Cycle Assessment of Biocement: An Emerging Sustainable Solution?, Sustainability 2021, 13, 13878. [Google Scholar] [CrossRef]
- A. Bandyopadhyay, A. Saha, D. Ghosh, B. Dam, A.K. Samanta, S. Dutta. Microbial repairing of concrete & its role in CO2 sequestration: a critical review. Beni-Suef Univ. J. Basic Appl. Sci. 2023, 12, 7. [Google Scholar] [CrossRef]
- C. Wu, J. Chu, S. Wu, Y. Hong. 3D characterization of microbially induced carbonate precipitation in rock fracture and the resulted permeability reduction. Eng. Geol. 2019, 249, 23–30. [Google Scholar] [CrossRef]
- Y. Wang, K. Soga, J.T. Dejong, A.J. Kabla. Microscale Visualization of Microbial-Induced Calcium Carbonate Precipitation Processes. J. Geotech. Geoenvironmental Eng. 2019, 145. [Google Scholar] [CrossRef]
- X. Xu, H. Guo, M. Li, X. Deng. Bio-cementation improvement via CaCO3 cementation pattern and crystal polymorph: A review. Constr. Build. Mater. 2021, 297, 123478. [Google Scholar] [CrossRef]
- K.D. Mutitu, M.O. Munyao, M.J. Wachira, R. Mwirichia, K.J. Thiong’o, M.J. Marangu. Effects of biocementation on some properties of cement-based materials incorporating Bacillus Species bacteria – a review. J. Sustain. Cem.-Based Mater. 2019, 8, 309–325. [Google Scholar] [CrossRef]
- N. Erdmann, D. Strieth. Influencing factors on ureolytic microbiologically induced calcium carbonate precipitation for biocementation. World J. Microbiol. Biotechnol. 2023, 39, 61. [Google Scholar] [CrossRef]
- F. Hammes, W. Verstraete*. Key roles of pH and calcium metabolism in microbial carbonate precipitation. Rev. Environ. Sci. Biotechnol. 2002, 1, 3–7. [Google Scholar] [CrossRef]
- C.-Z. Qin, S.M. Hassanizadeh, A. Ebigbo. Pore-scale network modeling of microbially induced calcium carbonate precipitation: Insight into scale dependence of biogeochemical reaction rates. Water Resour. Res. 2016, 52, 8794–8810. [Google Scholar] [CrossRef]
- M. Sharma, N. Satyam, N. Tiwari, S. Sahu, K.R. Reddy. Simplified biogeochemical numerical model to predict pore fluid chemistry and calcite precipitation during biocementation of soil, Arab. J. Geosci. 2021, 14, 807. [Google Scholar] [CrossRef]
- L.A. Van Paassen, Biogrout, ground improvement by microbial induced carbonate precipitation, (2009). http://resolver.tudelft.nl/uuid:5f3384c4-33bd-4f2a-8641-7c665433b57b (accessed August 13, 2023).
- H.-J. Lai, M.-J. Cui, J. Chu, Effect of pH on soil improvement using one-phase-low-pH MICP or EICP biocementation method, Acta Geotech. (2022). [CrossRef]
- V.S. Whiffin, Microbial CaCO3 precipitation for the production of biocement - Murdoch University, Murdoch University, 2004. https://researchportal.murdoch.edu.au/esploro/outputs/doctoral/Microbial-CaCO3-precipitation-for-the-production/991005540291407891 (accessed January 9, 2024).
- IWA Task Group for Mathematical Modelling of Anaerobic Digestion Processes, Anaerobic Digestion Model No.1 (ADM1), IWA Publishing, 2005. [CrossRef]
- S. Stocks-Fischer, J.K. Galinat, S.S. Bang. Microbiological precipitation of CaCO3, Soil Biol. Biochem. 1999, 31, 1563–1571. [Google Scholar] [CrossRef]
- Equilibrium Constants - Chemistry LibreTexts, Chem. Libr. (2014). https://chem.libretexts.org/Ancillary_Materials/Reference/Reference_Tables/Equilibrium_Constants (accessed February 10, 2024).
- W.M. Haynes, ed., CRC Handbook of Chemistry and Physics, 95th ed., CRC Press, Boca Raton, 2014. [CrossRef]
- 7.5.1: About Carbonic Anhydrase, Chem. Libr. (2021). https://chem.libretexts.org/Courses/Earlham_College/CHEM_361%3A_Inorganic_Chemistry_(Watson)/07%3A_Bioinorganic_Chemistry/7.05%3A_The_Reaction_Pathways_of_Zinc_Enzymes_and_Related_Biological_Catalysts/7.5.01%3A_About_Carbonic_Anhydrase (accessed March 24, 2024).
- E1: Acid Dissociation Constants at 25°C, Chem. Libr. (2014). https://chem.libretexts.org/Ancillary_Materials/Reference/Reference_Tables/Equilibrium_Constants/E1%3A_Acid_Dissociation_Constants_at_25C (accessed March 24, 2024).
- E5: Acid Dissociation Constants of Organics, Chem. Libr. (2013). https://chem.libretexts.org/Ancillary_Materials/Reference/Reference_Tables/Equilibrium_Constants/E5%3A_Acid_Dissociation_Constants_of_Organics (accessed March 24, 2024).
- E3: Solubility Constants for Compounds at 25°C, Chem. Libr. (2014). https://chem.libretexts.org/Ancillary_Materials/Reference/Reference_Tables/Equilibrium_Constants/E3._Solubility_Constants_for_Compounds_at_25C (accessed March 24, 2024).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




