Submitted:
09 September 2024
Posted:
10 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
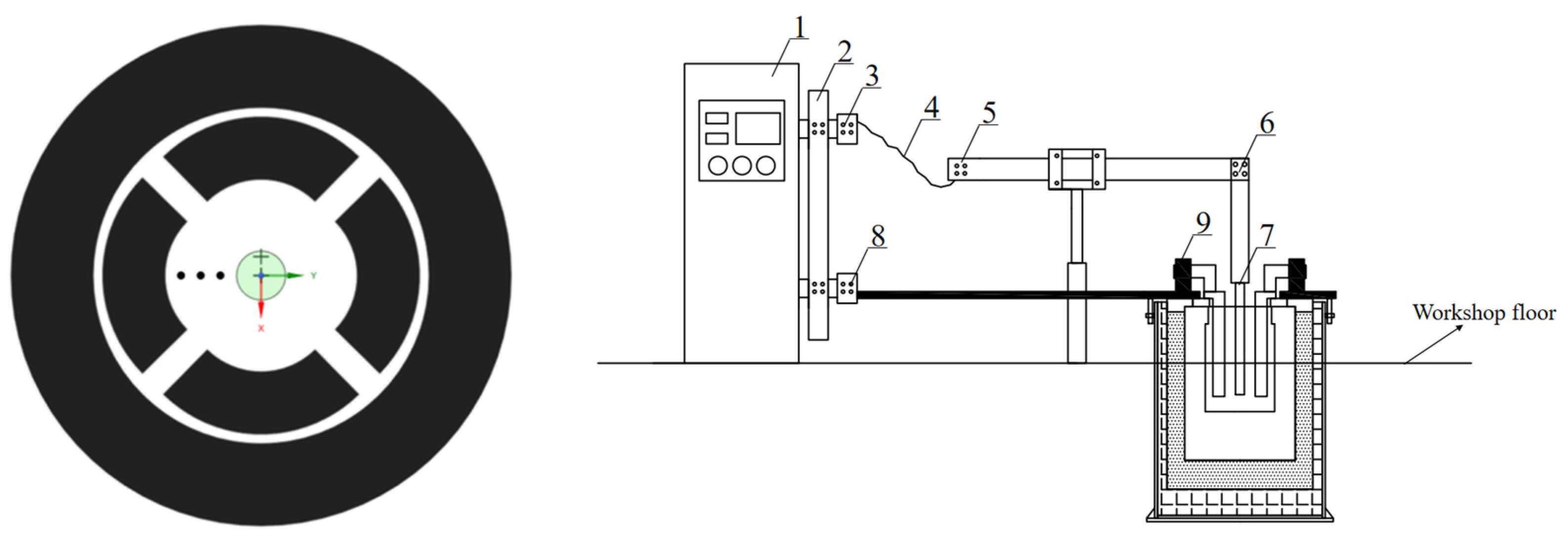
2. Experimental
2.2. Analysis of the Structure of Measured Electric Fields
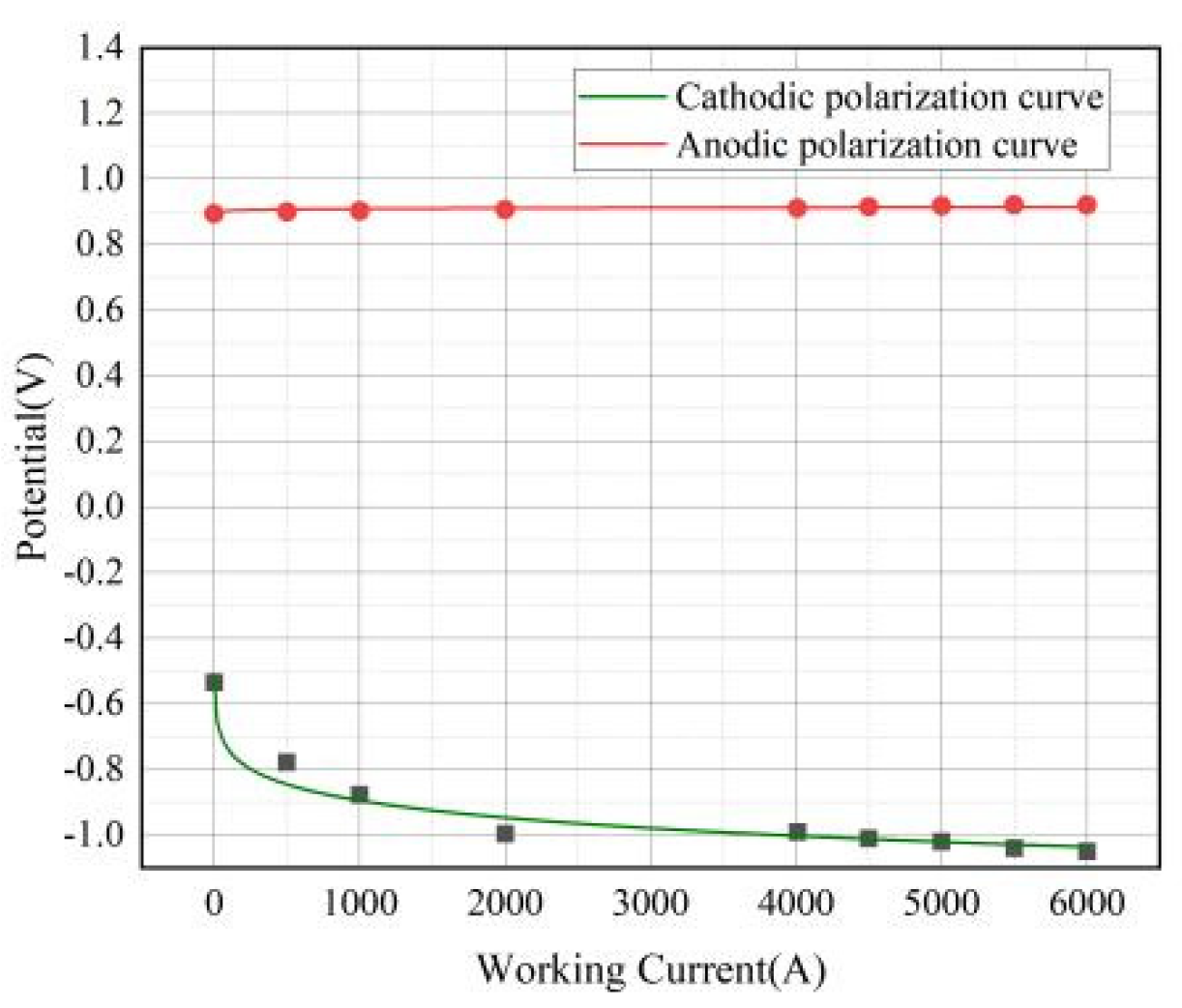
2.3. Analysis of Actual Decomposition Voltage Measurement
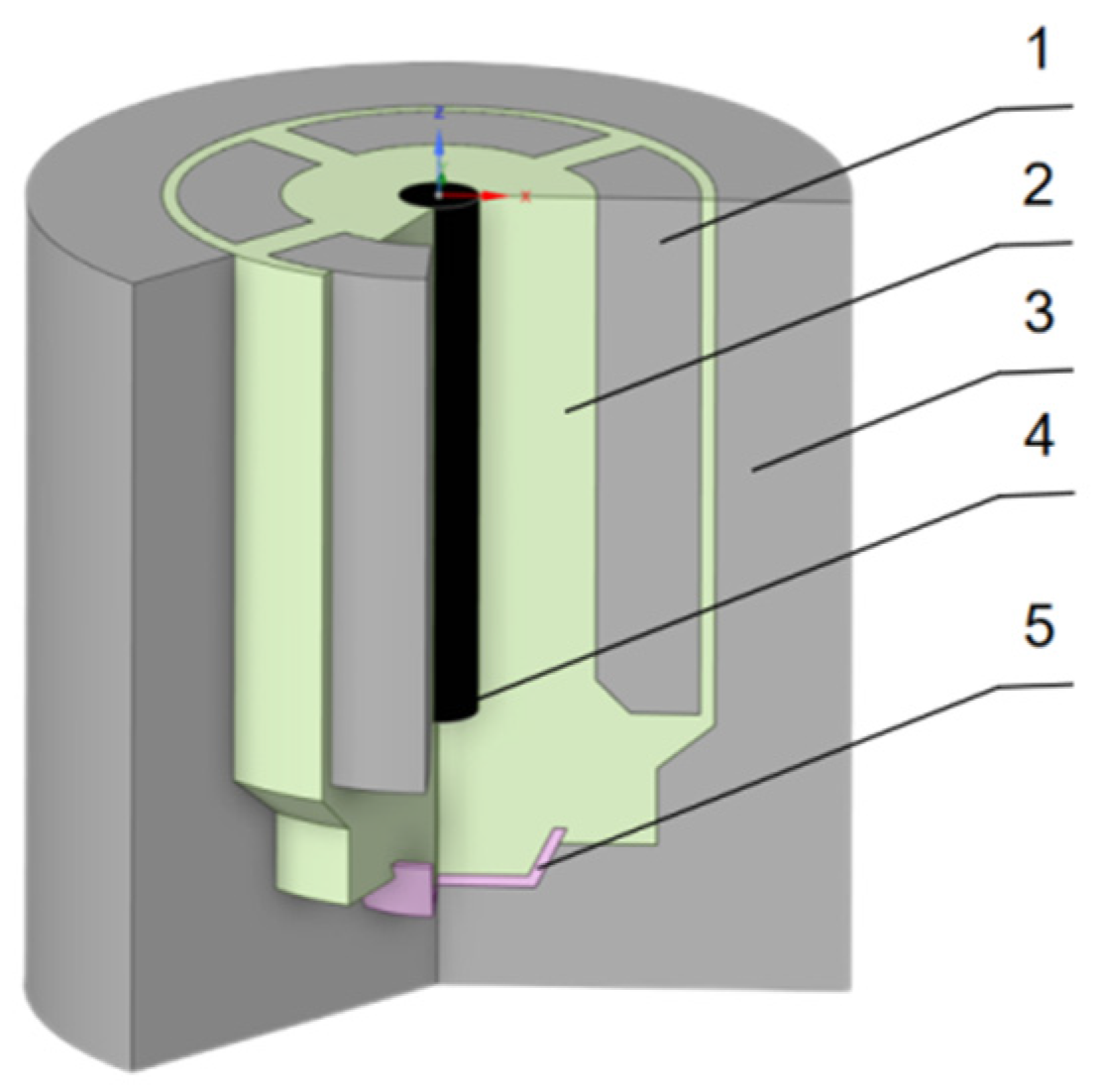
3. Model Description
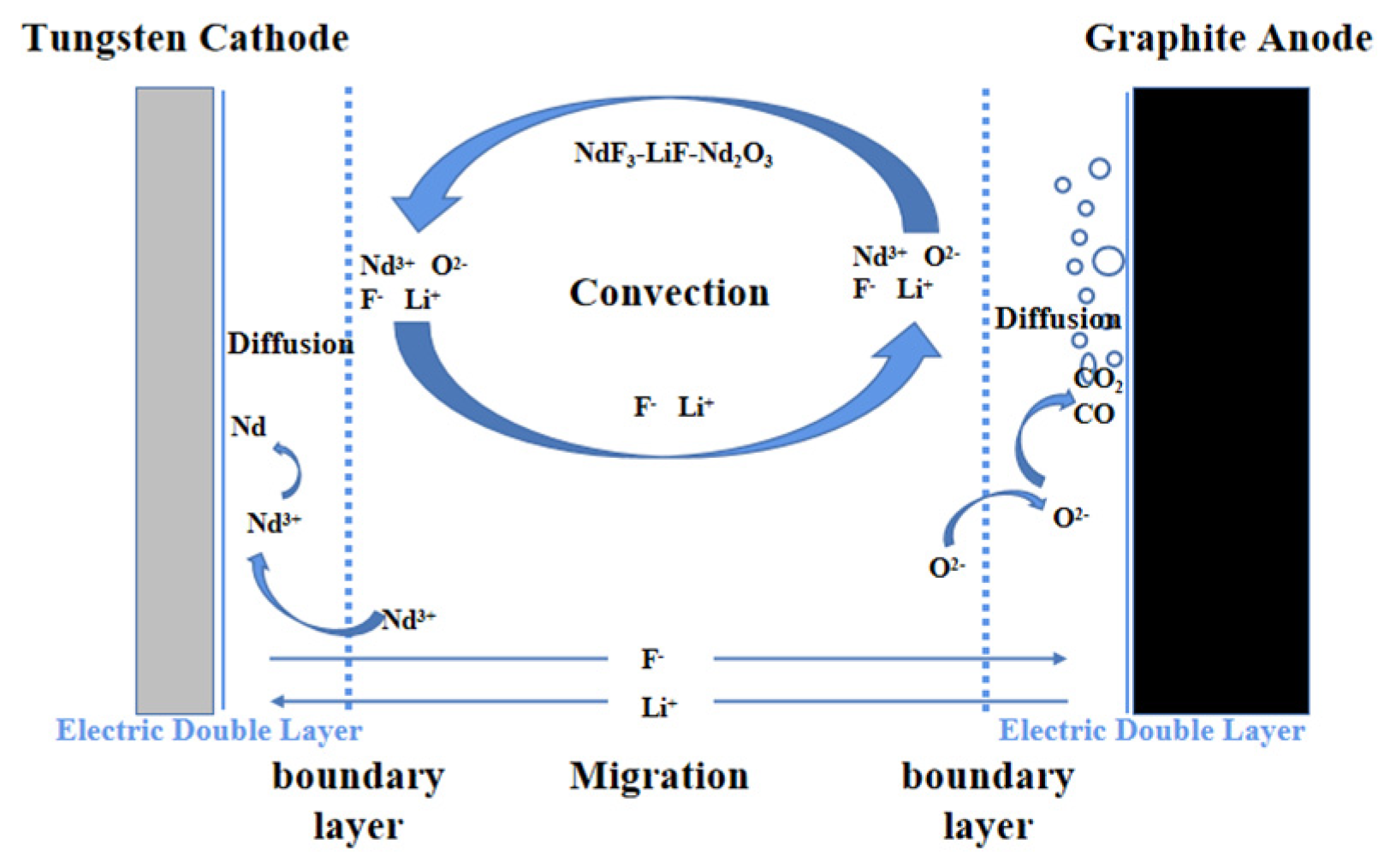
3.1. Physical Model
3.2. Governing Equation
3.3. Boundary Conditions
4. Simulation Results and Discussion
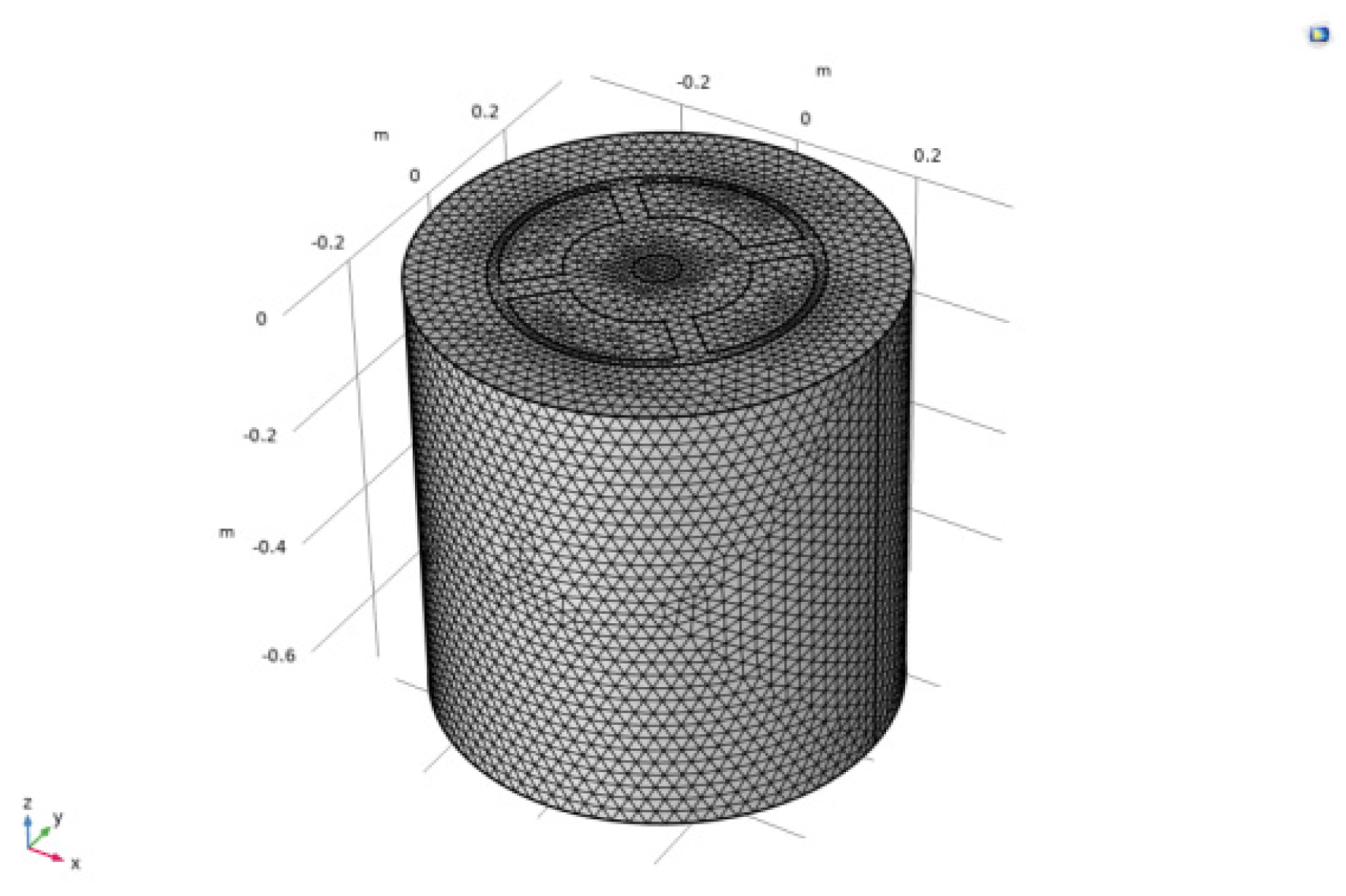
4.1. Mesh Independency Study
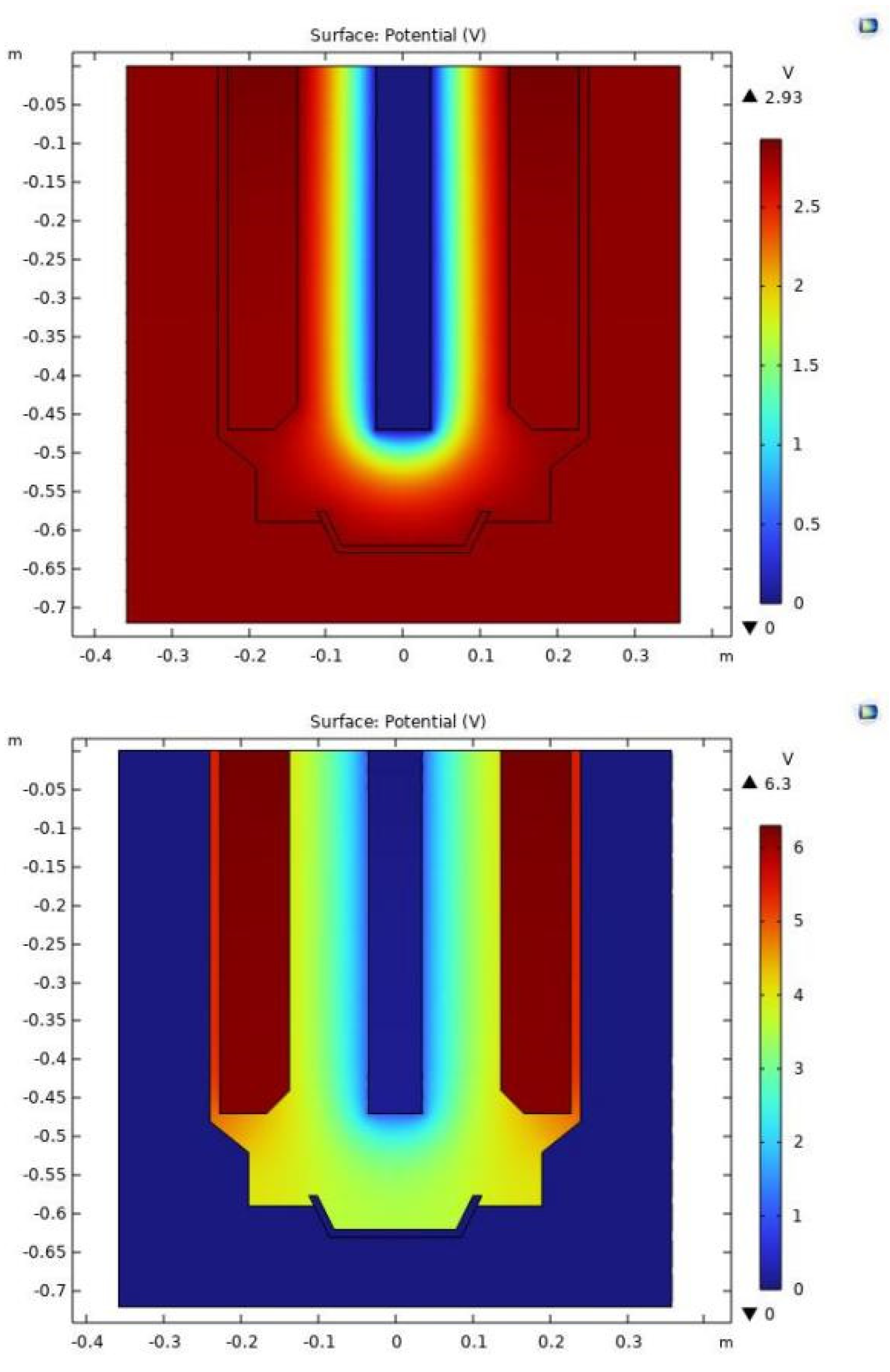
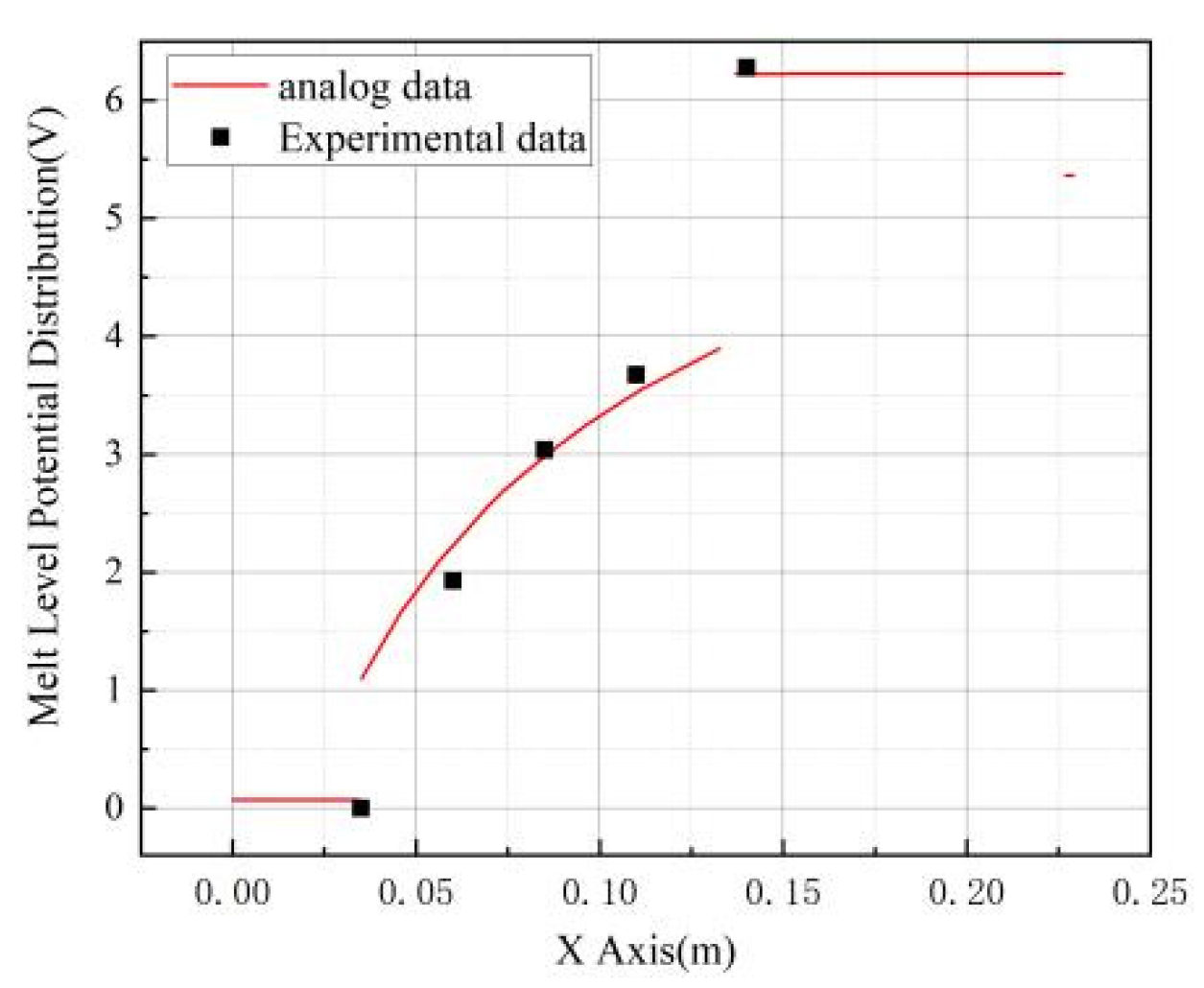
4.2. Model Validation
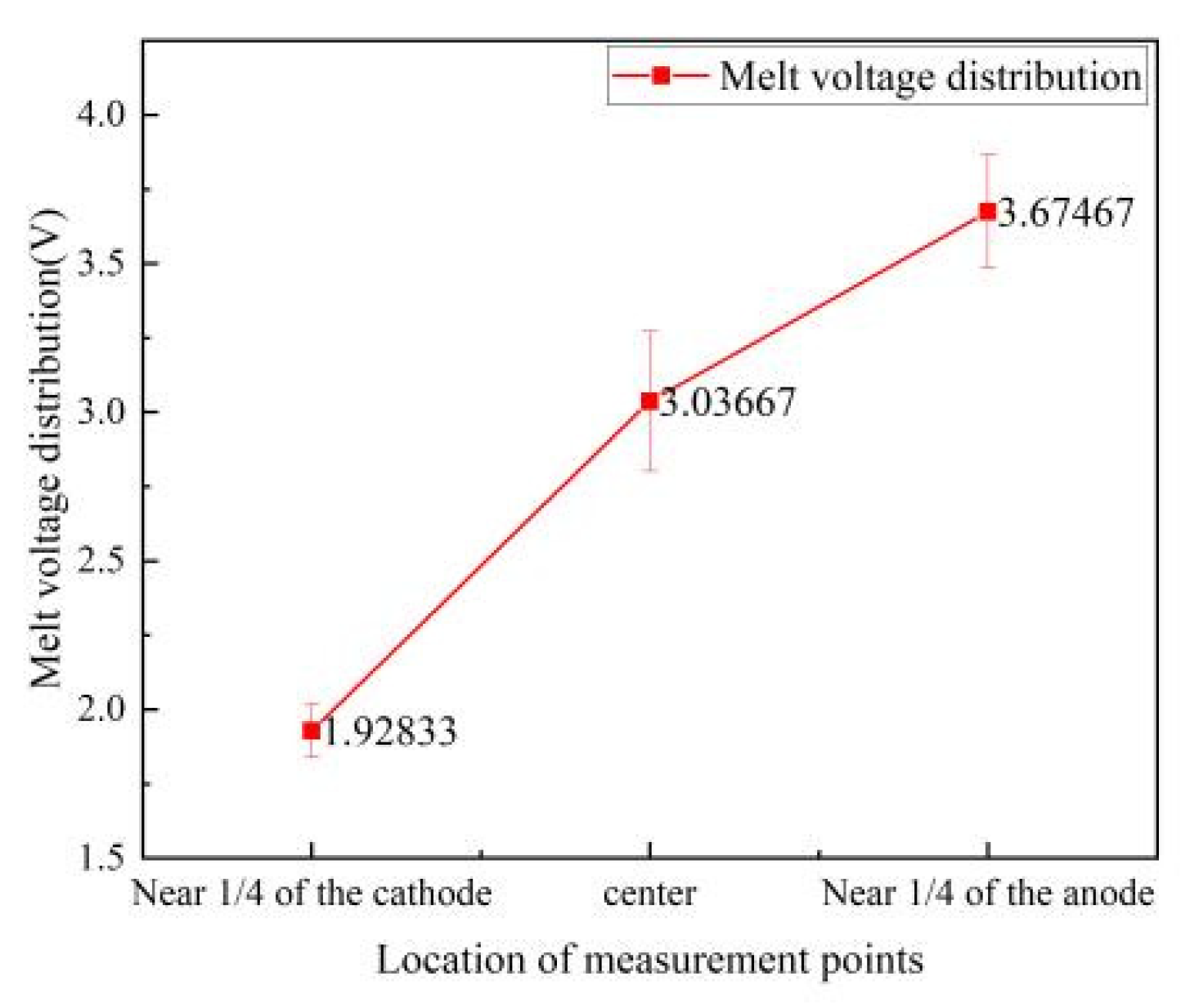
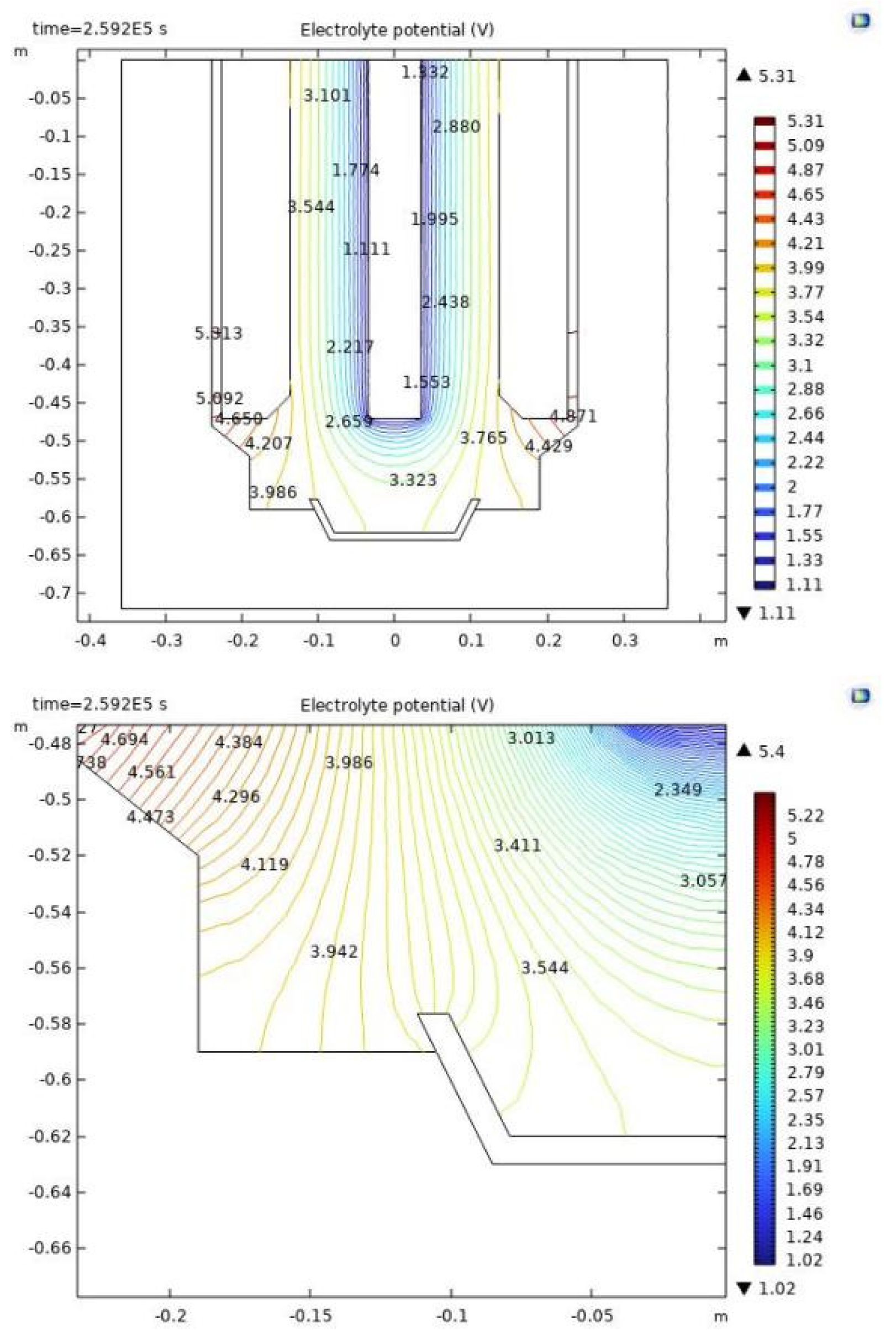
4.3. Electrolyte Voltage Drop
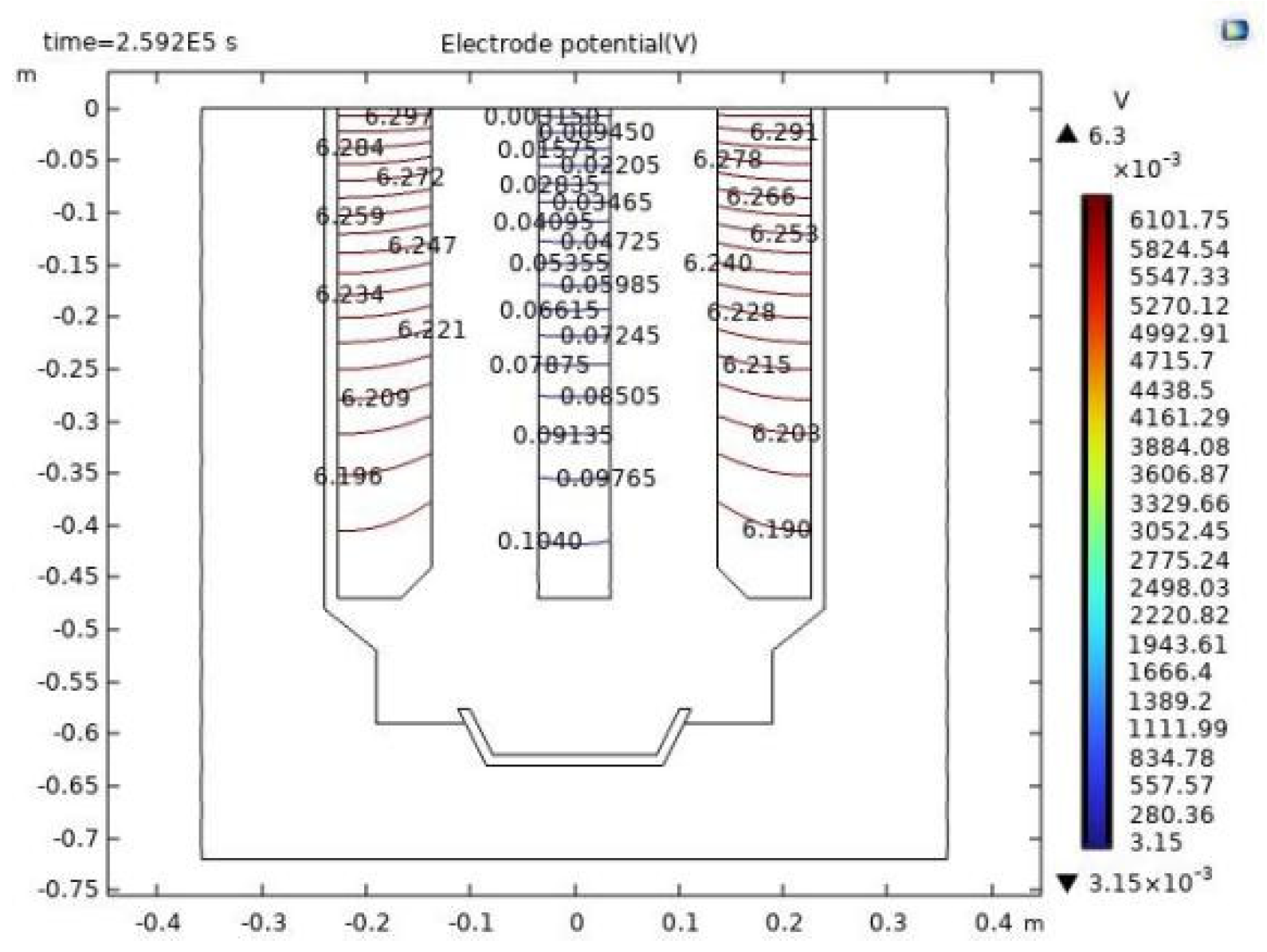
4.4. Voltage Drop between Cathode and Anode
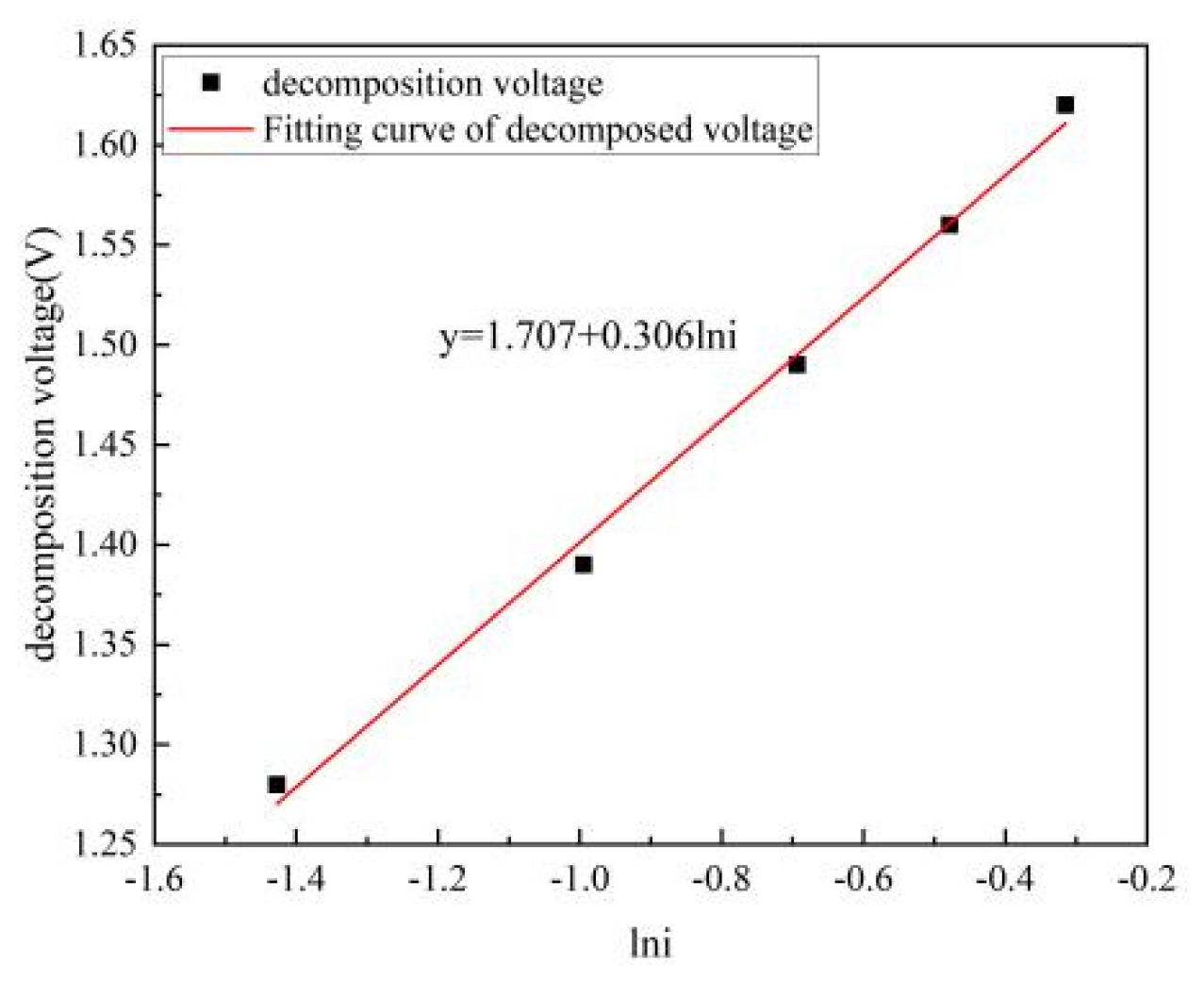
4.5. Actual Decomposition Voltage
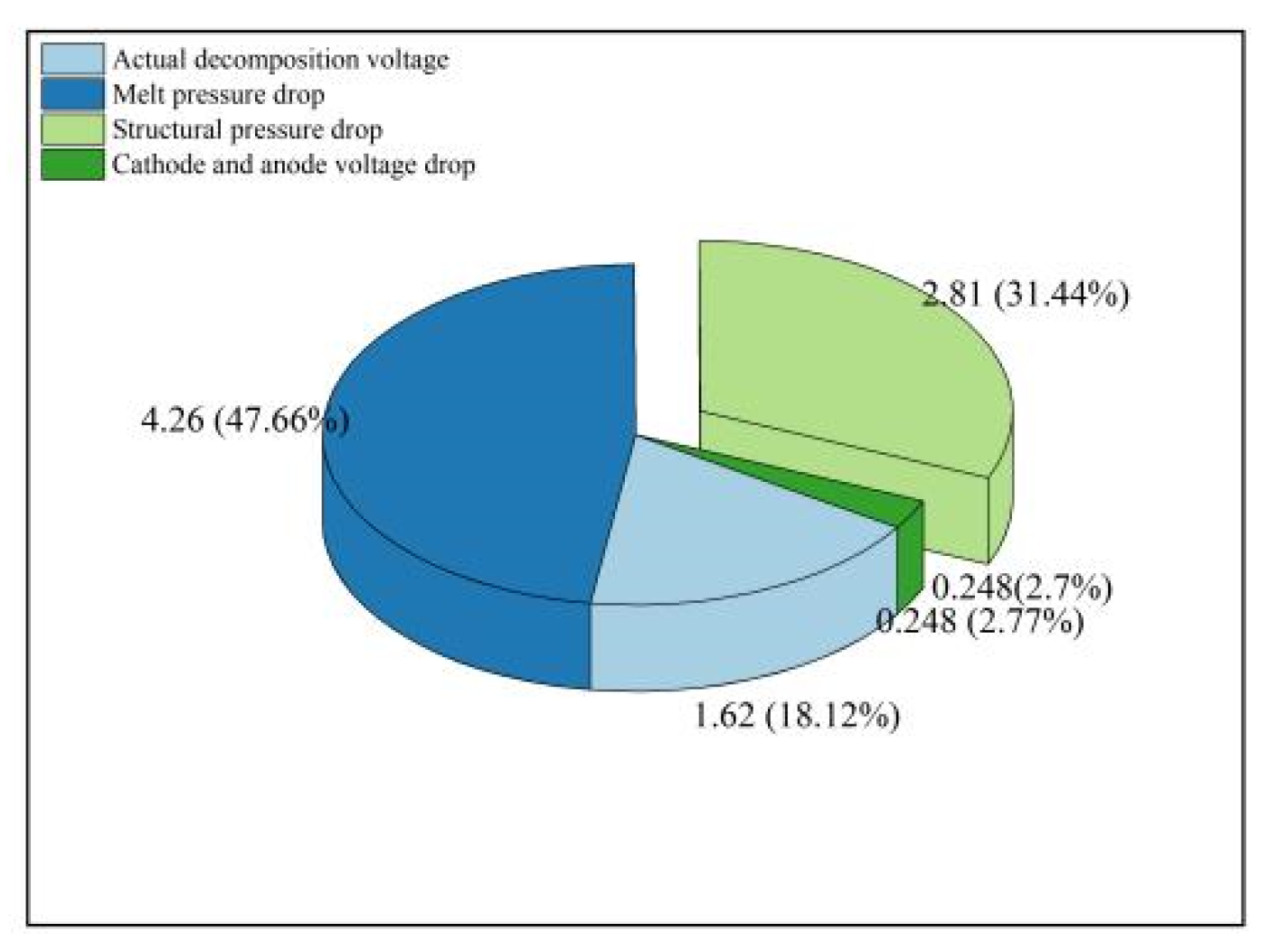
4.6. Voltage Balance of Electrolytic Cells
5. Conclusion
Acknowledgments
References
- The Editorial Board of 'Nonferrous Metal Metallurgy' has authored the 'Chinese Metallurgical Encyclopedia' [M]. Published by Metallurgical Industry Press, Beijing, November 1998.
- Liu Z X, Shi H M, He Y D, Ni J Z. Computer simulation of the electric field [J]. Journal of Baotou Iron and Steel Institute,2003(1): 29-31, 35.
- Li R C, Yang X D, Wang K, Ni J Z. Research Progress of Bioinorganic Chemistry of Rare Earths [J]. Journal of the Chinese Society of Rare Earths, 2004, 22(1):1.
- Wang J L, Yang Y Q. Electric field analysis of rare earth molten salt electrolytic cell based on Comsol[J]. Nonferrous Metals Science and Engineering, 2016, 7(6): 30-34.
- Wu Y F, Liu Z X, Li Z T. Numerical simulation of the electric field [J]. Non-ferrous metals (smelting part), 2010(03):27-30.
- Lv X J, Zhang H X, Li S, Zeng X P. Effect of anode consumption of rare earth metal electrolytic tank on electric field [J]. Chinese Journal of Rare Earth Sciences, 2019,37(04):481-490.
- Zhang H X, Lv X J, Zhong S p, Wang J E, Liu S, Zeng X P, Jian Y Z. Electric field simulation and optimization of cathodic structure [J]. Chinese Journal of Rare Earth Sciences, 2020,38(05):667-676.
- Ren Yonghong, Wang Jinbao, He Youduo, Liu Zhongxing. Electric Field Calculation and Channel Voltage Analysis of Rare Earth Electrolytes [J]. Journal of Baotou Iron and Steel College, 2003 (04): 313-317.
- Chen G H, Wang X Q, Zhang Z H, Liu Z X, Wu Y F. Yongfu. Voltage balance calculation of rare earth molten salt electrolytic cell [J]. Rare, 2013,34(01):86-89.
- Werner J M, Zeng W, Free M L, Zhang Z, Cho j.Modeling and Validation of Local Electrowinning Electrode Current Density Using Two Phase Flow and Nernst-Planck Equations[J]. Journal of The Electrochemical Society,2018,165(5):E190-E207.
- Zeng W Z, Free M L, Werner J, Wang S J.Simulation and Validation Studies of Impurity Particle Behavior in Copper Electrorefining[J].Journal of The Electrochemical Society,2015,162(14):E338-E352.
- Zeng W Z, Werner J, Free M L.Experimental studies on impurity particle behavior in electrolyte and the associated distribution on the cathode in the process of copper electrorefining[J].Hydrometallurgy,2015,156:232-238. [CrossRef]
- Hemmati H, Mohebbi A, Soltani A, Daneshpajouh S.CFD modeling of the electrolyte flow in the copper electrorefining cell of Sarcheshmeh copper complex[J]. Hydrometallurgy,2013,139:54-63.
- Volgin V M, Davydow A D.Numerical simulation of natural convection of electrolyte solution with three types of ions in the electrochemical cell with vertical electrodes[J].Russian Journal of Electrochemistry,2010,46(12):1360-1372. [CrossRef]
- Gao P, Zhu Y M, Yu Y C. Electrochemistry 2nd Edition [M]. Chemical Industry Press, 2019.
- LiuK R, Chen J S, Wei X J. Determination of anode overpotential of neodymium electrolysis [J]. Journal of Materials and Metallurgy, 2003(01):38-41.
| Number | Subject | Measurement |
|---|---|---|
| 1 | Power box loss voltage drop | 0.18V |
| 2 | Vertical contact voltage drop | 0.16V |
| 3 | Vertical bar and busbar contact | 0.25V |
| 4 | Electrode busbar | 0.11V |
| 5 | Bus and elevator contact | 0.07V |
| 6 | Horizontal and vertical beam joints | 0.1V |
| 7 | Cathode welding point | 1.48V |
| 8 | Anode and fixture contact | 0.15V |
| 9 | Fixture and anode contact | 0.31V |
| 10 | Cathode and anode voltage drop | 6.28V |
| 11 | Total | 9.09V |
| Subject | Conductivity | Supplement |
|---|---|---|
| graphite anode | 1e+5 s/m | empirical formula |
| tungsten cathode | 2.5e+6 s/m | experiment |
| electrolyte | 570 s/m | experiment |
| Metal receiver | 3.1e+6 s/m | reference[11] |
| Graphite crucible | 1e+5 s/m | reference[11] |
| Subject | Date | Supplement |
|---|---|---|
| Equilibrium potential | 1.36V | Calculate |
| Cathode exchange current Density | 1000A/m2 | Reference[15] |
| Anode Exchange current Density | 2200A/m2 | Reference[16] |
| Cathode transfer coefficient | 0.66 | Reference[16] |
| Anode transfer coefficient | 0.34 | Reference[16] |
| Cathode limit current density | 266040A/m2 | Experiment |
| Anode limit current density | 14620A/m2 | Experiment |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




