Submitted:
04 September 2024
Posted:
10 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. METHODOLOGY
2.1. ReaxFF-MD Reactive Force Field
2.2. Simulation Details.
3. Results and Discussion
3.1. Detailed Analysis of the Kinetic Mechanisms of the Oxidation Reaction of Naphthalene
3.2. Intermediate Reactions
3.3. Detailed Reaction Map for Naphthalene Oxidation
4. Conclusions
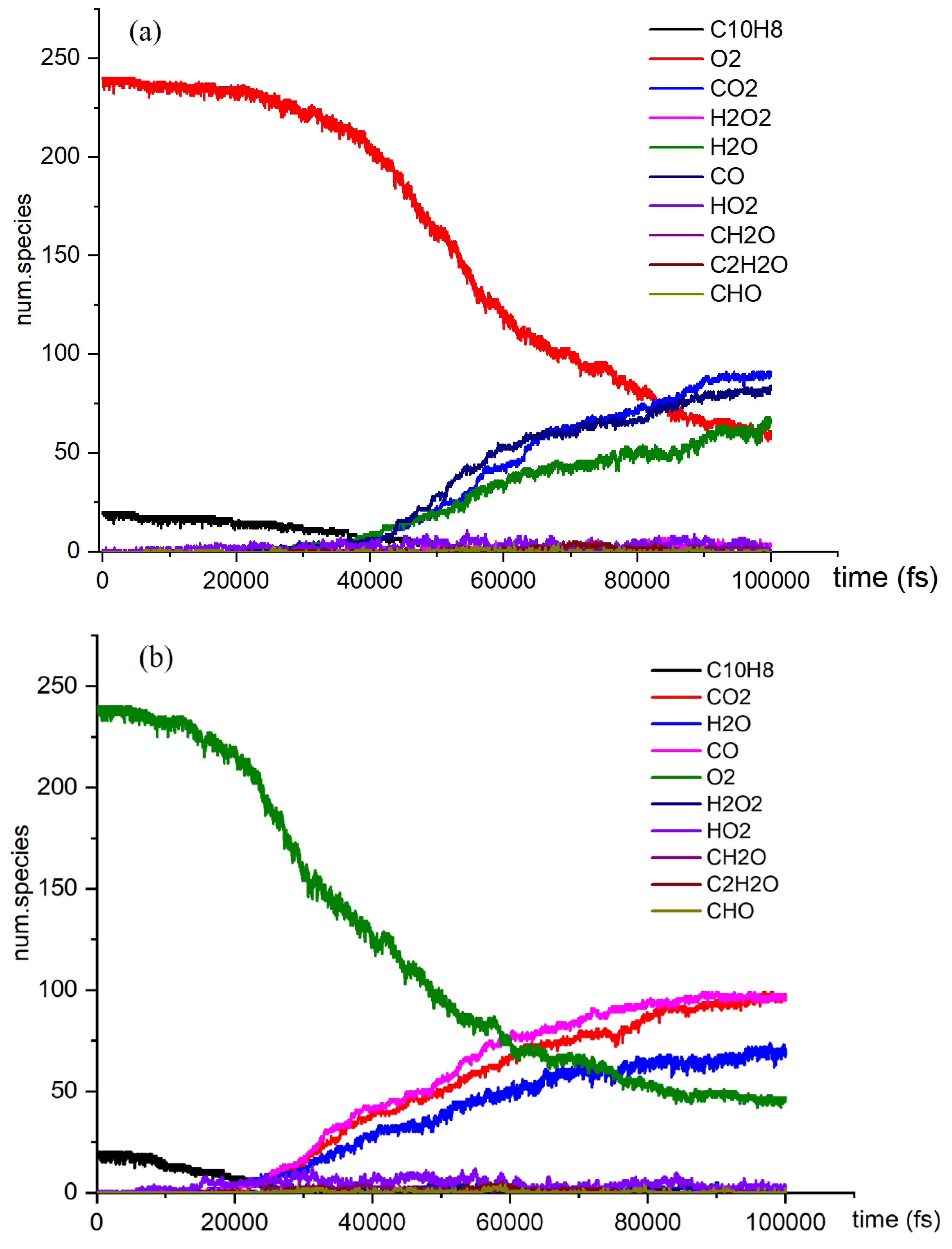
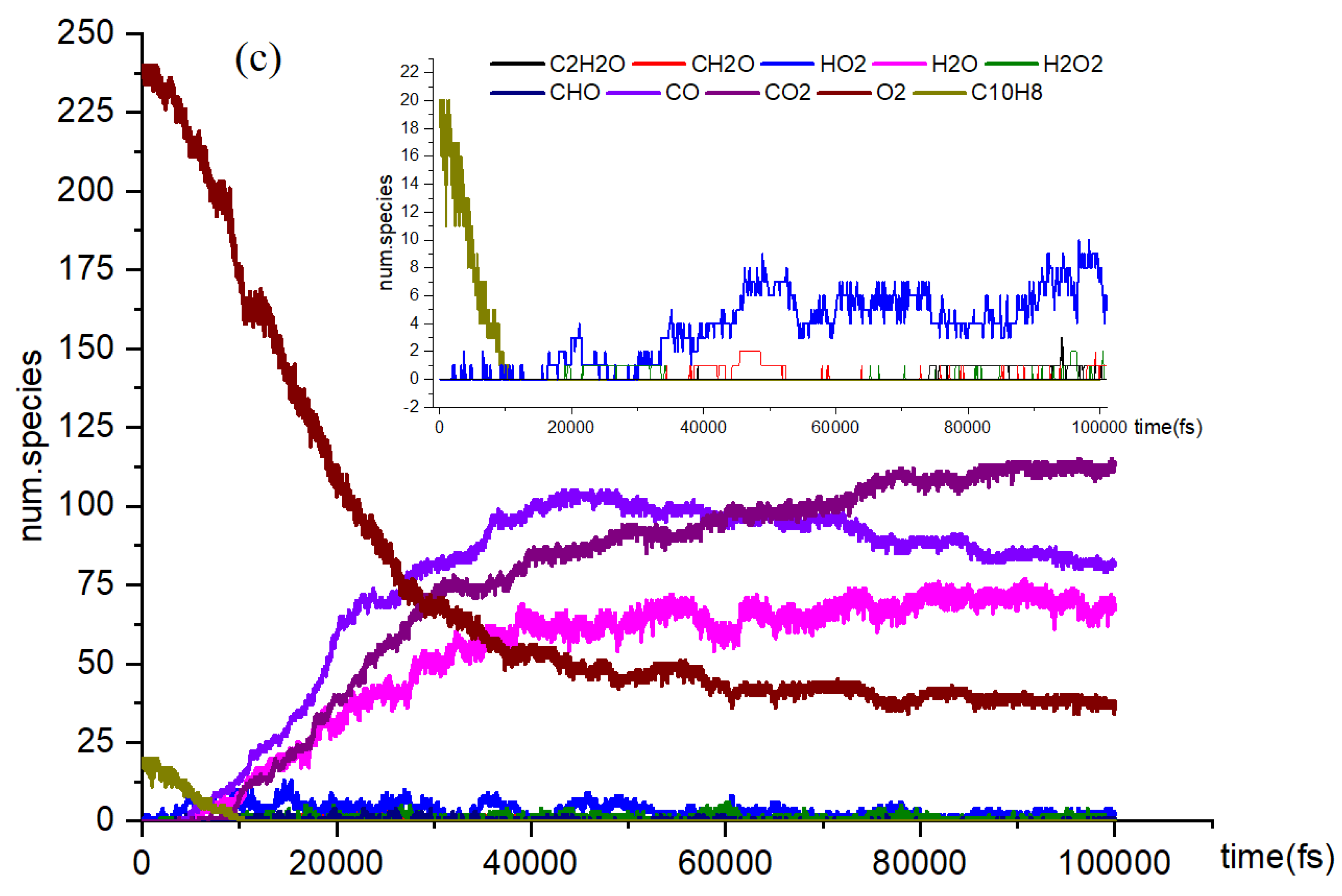
- Naphthalene undergoes oxidation at elevated temperatures using NVE-MD (microcanonical ensemble molecular dynamics) with a reactive force field. To obtain a comprehensive understanding of the oxidation process, simulations are conducted on a model system consisting of 20 naphthalene molecules. The primary species identified in the current simulations include CO2, CO, H2O, HO2, and others.
- The reaction rates of products in oxidation and pyrolysis reactions vary at different time stages. After the intersection of CO2 and CO, the yield of CO2 surpasses that of CO. This phenomenon can be attributed to insufficient collision contact in the early stages and more favorable reaction conditions in the later stages. In the reaction network from C10H8 to C7H6O, multiple types of reactions were identified across four reaction pathways, including dehydrogenation, hydrogenation, and small molecule activation reactions.
- The reaction network from C10H8 to C7H6O was examined. The findings of this investigation will enhance our understanding of the process. Among the reactions in the network, the hydrogenation reaction exhibits the highest reaction rate. Identifying the reaction network through conventional experiments is challenging due to the complexity of the reactions involved. In the study of oxidative pyrolysis reactions, computational simulations offer a novel research approach.
Author Contributions
Funding
Conflicts of Interest
References
- Alade, O.S.; Hamdy, M.; Mahmoud, M.; Al Shehri, D.A.; Mokheimer, E.; Patil, S. Ayman Al-Nakhli. A preliminary assessment of thermochemical fluid for heavy oil recovery. J. Pet. Sci. Eng. 2020, 186, 106702. [Google Scholar] [CrossRef]
- Zhao, S.; Sun, Y.; Lü, X.; Li, Q. Energy consumption and product release characteristics evaluation of oil shale non-isothermal pyrolysis based on TG-DSC. J. Pet. Sci. Eng. 2020, 187, 106812. [Google Scholar] [CrossRef]
- Jia, Y.; Cao, Y.; Wang, H.; Ma, B. Influence of multiphase carbonate cementations on the Eocene delta sandstones of the Bohai Bay Basin, China. J. Pet. Sci. Eng. 2021, 205, 108866. [Google Scholar] [CrossRef]
- Zhang, X.; Guo, W.; Pan, J.; Zhu, C.; Deng, S. In-situ pyrolysis of oil shale in pressured semi-closed system: Insights into products characteristics and pyrolysis mechanism. Energy 2024, 168, 129608. [Google Scholar] [CrossRef]
- Tao, L.; Hu, Z.; Xu, Z.; Zhang, X.; Ding, Y.; Wang, C.; Chen, D.; Li, S. Experimental investigation of in situ combustion (ISC) in heavy oil thermal recovery. Geoenergy Sci. Eng. 2024, 233, 212488. [Google Scholar] [CrossRef]
- Yang, M.; Liu, Y.; Lu, N.; Chai, M.; Wang, S.; Feng, Q.; Chen, Z. Integration of ramped temperature oxidation and combustion tube tests for kinetic modeling of heavy oil in-situ combustion. Energy 2023, 274, 127435. [Google Scholar] [CrossRef]
- Jayaraman, K.; Kök, M.V.; Gökalp, I. Combustion mechanism and model-free kinetics of different origin coal samples: Thermal analysis approach. Energy 2020, 204, 117905. [Google Scholar] [CrossRef]
- Mahvelati, E.; Forcinito, M.; Fitschy, L.; Maesen, A. Three-dimensional CFD Model Development and Validation for Once Through Steam Generator (OTSG): Coupling Combustion Heat Transfer and Steam Generation. ChemEngineering 2022, 6, 23. [Google Scholar] [CrossRef]
- Askarova, A.; Popov, E.; Ursenbach, M.; Moore, G., Sudarshan Mehta and Alexey Cheremisin. Experimental Investigations of Forward and Reverse Combustion for Increasing Oil Recovery of a Real Oil Field. Energies 2020, 13, 4581.
- Huang, S.; Sheng, J.J.; Jiang, Q.; Liu, J. Screening of Spontaneous Ignition Feasibility During Air Injection EOR Process Based on Thermal Experiments. Screening of Spontaneous Ignition Feasibility During Air Injection EOR Process Based on Thermal Experiments. Energies 2019, 12, 3687. [Google Scholar] [CrossRef]
- Zhao RB.; Xia XT.; Luo WW.; et al. Alteration of heavy oil properties under in-situ combustion: a field study. Energy Fuels 2015, 29, 6839. [CrossRef]
- RB, Z.; CH, Z.; FX, Y.; MH, H.; PT, S.; Wang, Y.J. Influence of temperature field on rock and heavy components variation during in-situ combustion process. Fuel 2018, 230, 244. [Google Scholar]
- Sun, Z., Jincheng Ji and Weihua Zhu. Effects of Nanoparticle Size on the Thermal Decomposition Mechanisms of 3, 5-Diamino-6-hydroxy-2-oxide-4-nitropyrimidone through ReaxFF Large-Scale Molecular Dynamics Simulations. Molecules 2024, 29, 56.
- Sang.; Unocic, R.R.; Iacovella, C.R.; Gogotsi, Y., Adri, C. T. Van Duin and Peter, T. Cummings. An Atomistic Carbide-Derived Carbon Model Generated Using ReaxFF-Based Quenched Molecular Dynamics. J. Carbon Res. 2017, 3, 32.
- Thompson, M.W.; Dyatkin, B.; Wang, H.-W.; Turner, C.H.; Xiahan., *!!! REPLACE !!!*; Li, C.; Yang, Z.; Wu, X.; Shao, S. Xiangying Meng and Gaowu Qin. Reactive Molecular Dynamics Simulations of Polystyrene Pyrolysis. Int. J. Mol. Sci. 2023, 24, 16403. [Google Scholar]
- Fan, Y.; Li, Y.; Zhang, Y.; Shi, K. Mechanism Analysis of Ethanol Production from Cellulosic Insulating Paper Based on Reaction Molecular Dynamics. Polymers 2022, 14, 4918. [Google Scholar] [CrossRef]
- Deng, S.; Zhuo, H.; Wang, Y.; Leng, S.; Zhuang, G.; Zhong, X.; Wei, Z.; Yao, Z.; Wang, J. Multiscale Simulation on Product Distribution from Pyrolysis of Styrene-Butadiene Rubber. Polymers 2019, 11, 1967. [Google Scholar] [CrossRef]
- Liu, H.; Liang, J.; He, R.; Li, X.; Zheng, M.; Ren, C.; An, G.; Xu, X.; Zheng, Z. Overall mechanism of JP-10 pyrolysis unraveled by large-scale reactive molecular dynamics simulation. Combust. Flame 2022, 237, 111865. [Google Scholar] [CrossRef]
- Wang, Y.; Mao, Q.; Wang, Z.; Luo, K.H.; Zhou, L.; Wei, H. A ReaxFF molecular dynamics study of polycyclic aromatic hydrocarbon oxidation assisted by nitrogen oxides. Combust. Flame 2023, 248, 112571. [Google Scholar] [CrossRef]
- Cheng, T.; Jaramillo-Botero, A.; Goddard, W.A.; Sun, S. Adaptive accelerated ReaxFF reactive dynamics with validation from simulating hydrogen combustion. J. Am. Chem. Soc. 2014, 136, 9434. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Li, X.; Li, Z.; Xu, H.; Qing, M.-X.; Jiang, X.; Liu, L. The molecular evolution mechanism of direct pyrolysis and hydropyrolysis of Changqing petroleum coke was compared based on ReaxFF method. J. Mol. Struct. 2023, 1290, 135882. [Google Scholar] [CrossRef]
- T P, S.; van Duin, A.C.T.; Janik, M.J. Methane Activation at the Pd/CeO2 Interface. ACS Catal. 2017, 7, 327. [Google Scholar]
- Ostadhossein, A.; Rahnamoun, A.; Wang, Y.; Zhao, P.; Zhang, S.; V H, C.; van Duin, A.C.T. ReaxFF Reactive Force-Field Study of Molybdenum Disulfide (MoS2). J. Phys. Chem. Lett. 2017, 8, 631. [Google Scholar] [CrossRef]
- Yeon, J.; van Duin, A.C.T.; Kim, S. H. Effects of Water on Tribochemical Wear of Silicon Oxide Interface: Molecular Dynamics (MD) Study with Reactive Force Field (ReaxFF). Langmuir 2016, 32, 1018. [Google Scholar] [CrossRef]
- Yeon, J.; van Duin, A.C.T. ReaxFF Molecular Dynamics Simulations of Hydroxylation Kinetics for Amorphous and Nano-Silica Structure, and Its Relations with Atomic Strain Energy. J. Phys. Chem. C 2016, 120, 305. [Google Scholar] [CrossRef]
- Y K, S.; Gai, L.; Raman, S.; van Duin, A.C.T. Development of a ReaxFF Reactive Force Field for the Pt-Ni Alloy Catalyst. J. Phys. Chem. A. 2016, 120, 8044. [Google Scholar]
- Zhong, Q.; Mao, Q.; Xiao, J.; van Duin, A.C.T.; Jonathan, P. Mathews. ReaxFF simulations of petroleum coke sulfur removal mechanisms during pyrolysis and combustion. Combust. Flame 2018, 198, 146. [Google Scholar] [CrossRef]
- Liu, L.; Xu, H.; Zhu, Q.; Ren, H.; Li, X. Soot formation of n-decane pyrolysis: A mechanistic view from ReaxFF molecular dynamics simulation. Chem. Phys. Lett. 2020, 760, 137983. [Google Scholar] [CrossRef]
- Ashraf, C.; Shabnam, S.; Jain, A.; Xuan, Y.; van Duin, A.C.T. Pyrolysis of binary fuel mixtures at supercritical conditions: A ReaxFF molecular dynamics study. Fuel 2019, 235, 194. [Google Scholar] [CrossRef]
- Senftle; T.P., et al. The ReaxFF reactive force-field: development, applications and future directions. Npj Comput. Mater 2016, 2, 1.
- Huo, E.; Zhang, S.; Xin, L.; Wang, S.; Cai, S.; Zhang, L.; Bai, M. Pyrolysis mechanism study of n-heptane as an endothermic hydrocarbon fuel: A reactive molecular dynamic simulation and density functional theory calculation study. Comput. Theor. Chem. 2022, 1211, 113696. [Google Scholar] [CrossRef]
- Hirotoshi Hirai. Molecular dynamics simulations for initial formation process of polycyclic aromatic hydrocarbons in n-hexane and cyclohexane combustion. Chem. Phys. 2021, 548, 111225. [Google Scholar] [CrossRef]
- Density functional theory study of the influence of activating and deactivating groups on Naphthalene. Results Chem. 2022, 4, 10669.
- Alamfard, T.; Lorenz, T.; Breitkopf, C. Tommy Lorenz and Cornelia Breitkopf. Thermal Conductivities of Uniform and Random Sulfur Crosslinking in Polybutadiene by Molecular Dynamic Simulation. Polymers 2023, 15, 2058. [Google Scholar] [CrossRef]
- Orekhov, N.; Ostroumova, G.; Stegailov, V. High temperature pure carbon nanoparticle formation: Validation of AIREBO and ReaxFF reactive molecular dynamics. Carbon 2020, 270, 606. [Google Scholar] [CrossRef]
- Döntgen, M.; M D, P.-F.; Kröger L, C.; W A, K.; A E, I.; Leonhard, K. Automated discovery of reaction pathways, rate constants, and transition states using reactive molecular dynamics simulations. J. Chem. Theory Comput 2015, 11, 2517. [Google Scholar] [CrossRef] [PubMed]
- Döntgen, M.; Schmalz, F.; Kopp, W. A.; Kröger, L.C.; Leonhard, K. Automated Chemical Kinetic Modeling via Hybrid Reactive Molecular Dynamics and Quantum Chemistry Simulations. J. Chem. Inf. Model. 2018, 58, 1343. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Yuan, W.; Gaïl, S.; Li, W.; Zhao, L.; Yang, J.; Qi, F.; Li, Y.; Dagaut, P. Exploration on the combustion chemistry of p-xylene: A comprehensive study over wide conditions and comparison among C8H10 isomers. Combust. Flame 2024, 262, 113377. [Google Scholar] [CrossRef]

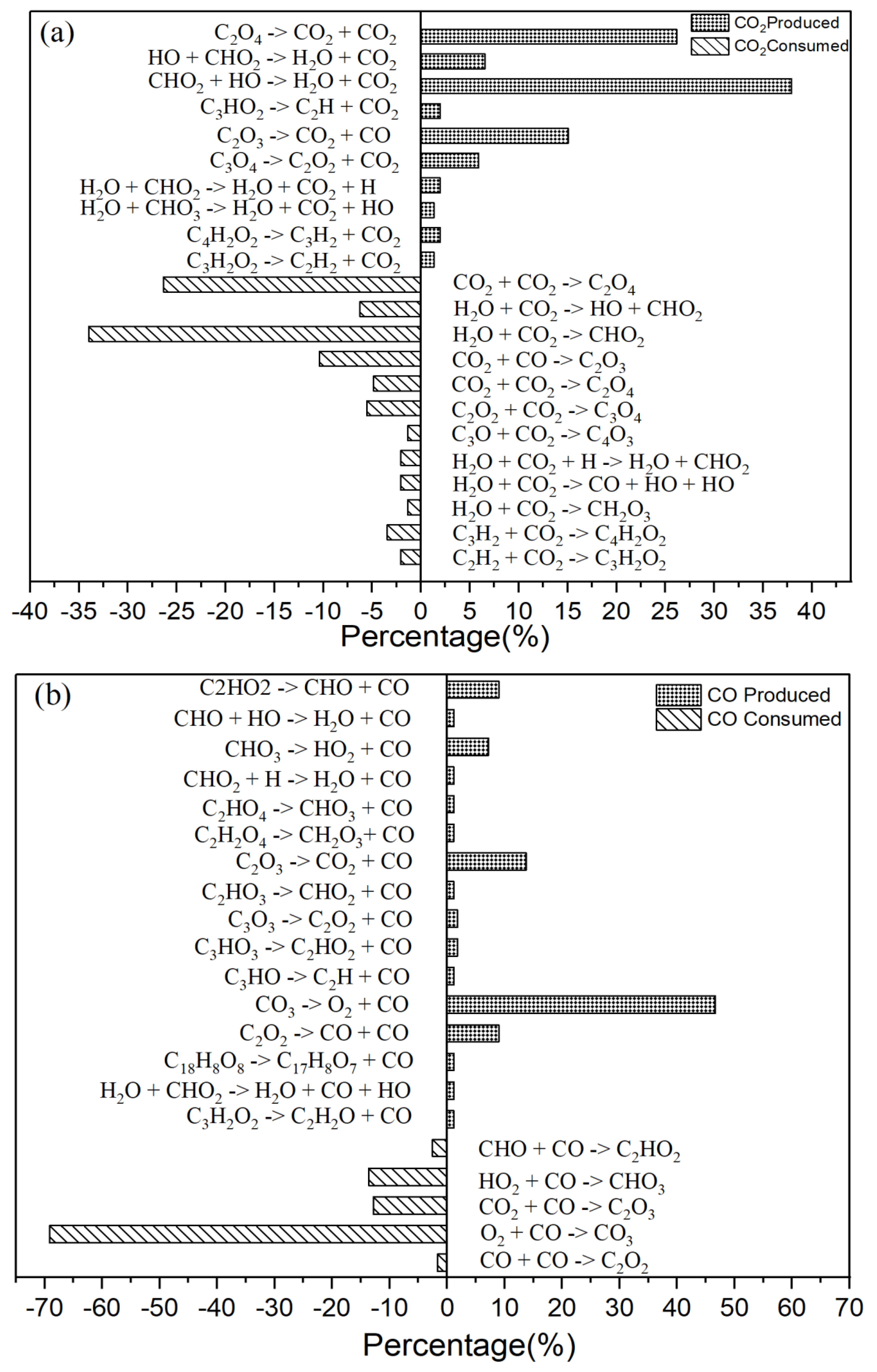
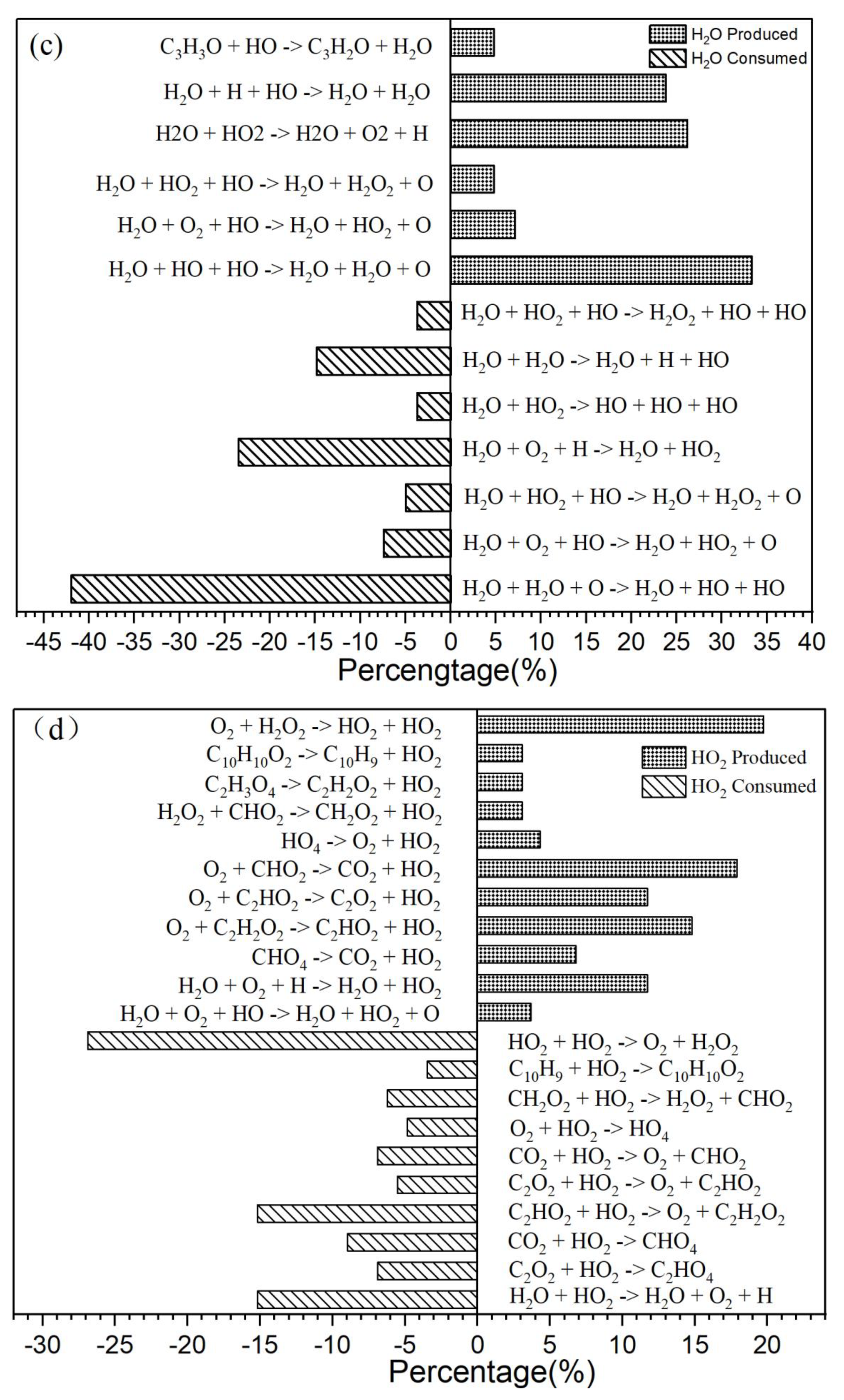
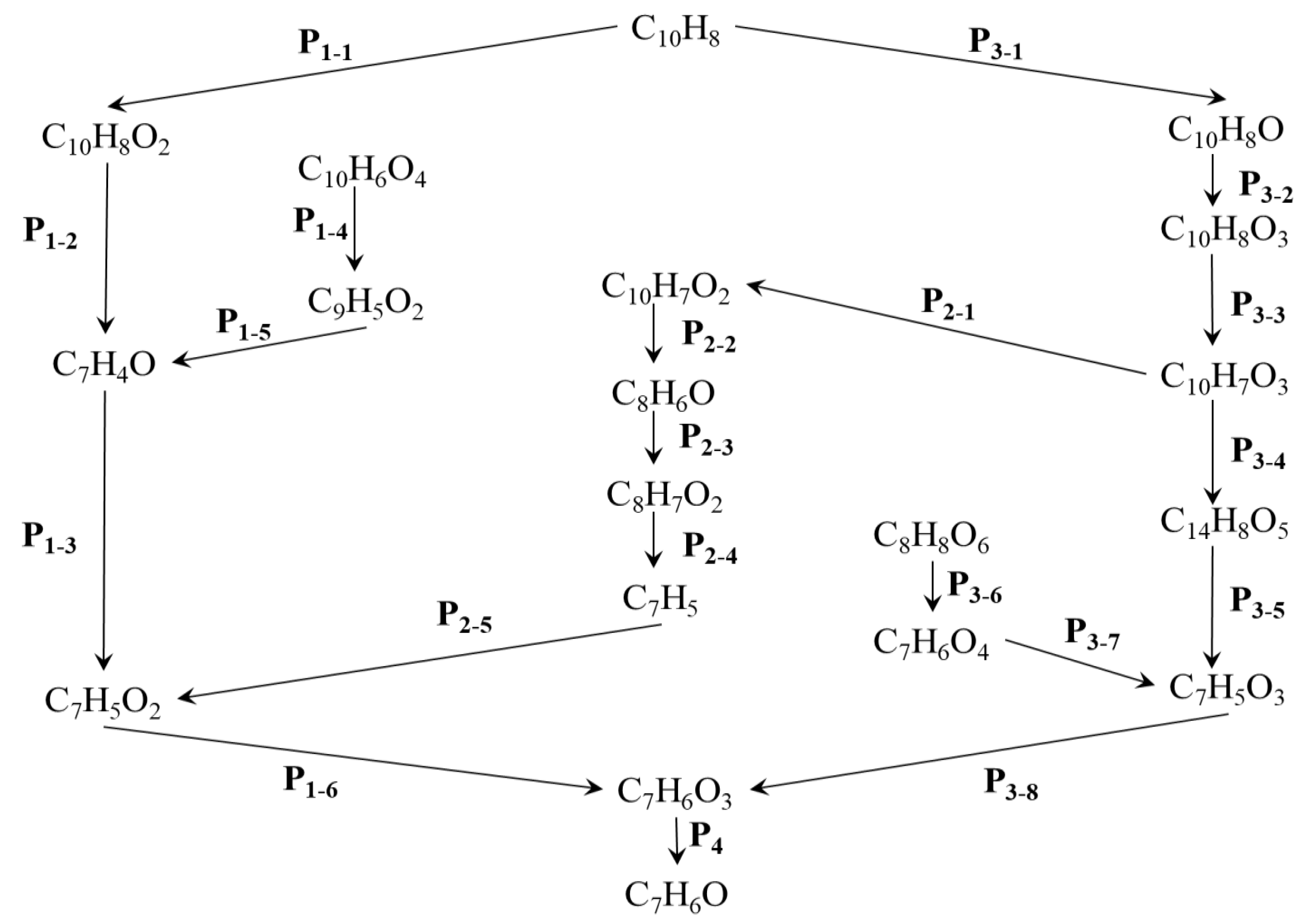
| Reactions | Rate Constants(s-1) | |
|---|---|---|
| 1 | CHO2 + HO → H2O + CO2 | 7.20×1010 |
| 2 | CO3 → O2 + CO | 4.04×1013 |
| 3 | H2O + HO2 → H2O + O2 + H | 7.39×108 |
| 4 | O2 + H2O2 → HO2 + HO2 | 5.41×109 |
| Path | Reactions | Rate Constants K (s-1) |
|---|---|---|
| P1-1 | O2 + C10H8 → C10H8O2 | 1.34×109 |
| P1-2 | C10H8O2 → C7H4O + C3H4O | 3.85×1011 |
| P1-3 | C7H4O + C2H2O2 → C2HO + C7H5O2 | 3.01×1013 |
| P1-4 | C10H6O4 → CO2 + C9H5O2 + H | 5.00×1013 |
| P1-5 | C9H5O2 → C2HO + C7H4O | 2.50×1013 |
| P1-6 | C7H5O2 + HO → C7H6O3 | 4.40×1012 |
| Path | Reactions | Rate Constants K (s-1) |
|---|---|---|
| P2-1 | C10H7O3 → C10H7O2 + O | 8.13×1011 |
| P2-2 | C10H7O2 → C8H6O + C2HO | 4.54×1012 |
| P2-3 | HO + C8H6O → C8H7O2 | 1.00×1013 |
| P2-4 | C8H7O2 → C7H5 + CH2O2 | 5.00×1013 |
| P2-5 | C7H5 + O2 → C7H5O2 | 5.51×1010 |
| Path | Reactions | Rate Constants K (s-1) |
|---|---|---|
| P3-1 | C10H7O + C10H8 → C10H8O + C10H7 | 1.44×1010 |
| P3-2 | O2 + C10H8O → C10H8O3 | 2.01×1011 |
| P3-3 | C10H8O3 → H + C10H7O3 | 7.69×1012 |
| P3-4 | C10H7O3 + C4HO2 → C14H8O5 | 1.51×1013 |
| P3-5 | C14H8O5 → C7H3O2 + C7H5O3 | 1.67×1013 |
| P3-6 | C8H8O6→ C7H6O4 + CHO + HO | 5.00×1012 |
| P3-7 | C7H6O4 → C7H5O3 + HO | 3.33×1013 |
| P3-8 | C7H5O3 + H → C7H6O3 | 6.02×1013 |
| P4 | C7H6O3 → C7H6O + O2 | 5.00×1013 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




