Submitted:
02 September 2024
Posted:
04 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Handelsman, J. Metagenomics: Application of Genomics to Uncultured Microorganisms. Microbiol. Mol. Biol. Rev. 2004, 68, 669–685. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Silva B, Vilo-Muñoz C, Galetovic A, Dong Q, Castelan-Sanchez H et al. Metagenomics of Atacama lithobiontic extremophile life unveils highlights on fungal communities, biogeochemical cycles, and carbohydrate-active enzymes. Microorganisms 2019, 7, 619. [CrossRef]
- Gómez-Silva, B.; Rainey, F.A.; Warren-Rhodes, K.A.; McKay, C.P. Navarro-González, R. Atacama Desert Soil Microbiology. In Microbiology of Extreme Soils. Soil Biology Series; P Dion and CS Nautiyal, Ed.; Springer-Verlag: Berlin, Heidelberg, Germany, 2008; Chapter 6, Vol. 13, pp 117-132; ISBN 978-3-540-74230-2. [Google Scholar]
- Gómez-Silva, B. On the Limits Imposed to Life by the Hyperarid Atacama Desert in Northern Chile. In: Astrobiology: Emergence, Search and Detection of Life. Basiuk, V.A, Ed., American Scientific Publishers: Los Angeles, CA, USA, 2010; Chapter 9, pp. 199–213.
- Azua-Bustos, A., Urrejola, C., Vicuña, R. Life at the dry edge: Microorganisms of the Atacama Desert. FEBS Lett. 2012, 586, 2939-2945. Albarracín, V.; Kurth, D.; Ordoñez, O.F.; Belfiore, C.; Luccini, E.; et al. High-up: A remote reservoir of microbial extremophiles in central Andean wetlands. Front. Microbiol. 2015, 6, 1404. [CrossRef]
- Wierzchos, J., Casero, M.C., Artieda, O., Ascaso, C. Endolithic microbial habitats as refuges for life in polyextreme environment of the Atacama Desert. Curr. Opin. Microbiol. 2018, 43, 124–131. [CrossRef]
- Gómez-Silva, B. Lithobiontic life: Atacama rocks are well and alive. Antonie van Leeuwenhoek. 2018, 111, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Silva, B.; Batista-García, R.A. The Atacama Desert: A Biodiversity Hotspot and Not Just a Mineral-Rich Region. Front. Microbiol. 2022, 13, 812842. [Google Scholar] [CrossRef] [PubMed]
- Houston, J. Variability of precipitation in the Atacama Desert: Its causes and hydrological impact. Int. J. Climatol. 2006, 26, 2181–2198. [Google Scholar] [CrossRef]
- Albarracín, V.; Kurth, D.; Ordoñez, O.F.; Belfiore, C.; Luccini, E.; et al. High-up: A remote reservoir of microbial extremophiles in central Andean wetlands. Front. Microbiol. 2015, 6, 1404. [Google Scholar] [CrossRef]
- Armienta, M.A. , Vilaclara, G., De la Cruz-Reyna, S., Ramos, S., Ceniceros, N., Cruz, O., Aguayo, A., Arcega-Cabrera, F. Water chemistry of lakes related to active and inactive Mexican volcanoes. J. Volcanol. Geotherm. Res. 2008, 178, 249–258. [Google Scholar] [CrossRef]
- SERNAGEOMIN Chile: Territorio volcánico. https://www.sernageomin.cl/chile-territorio-volcanico/. accessed July 18th 2018, 2022.
- López, D.L.; Bundschuh, J.; Birkle, P.; Armienta, M.A.; Cumbal, L.; Sracek, O.; Cornejo, L. Ormacheam M. Arsenic in volanic geothermal fluids of Latin America. Sci. Total Environ. 2012, 429, 57–75. [Google Scholar] [CrossRef]
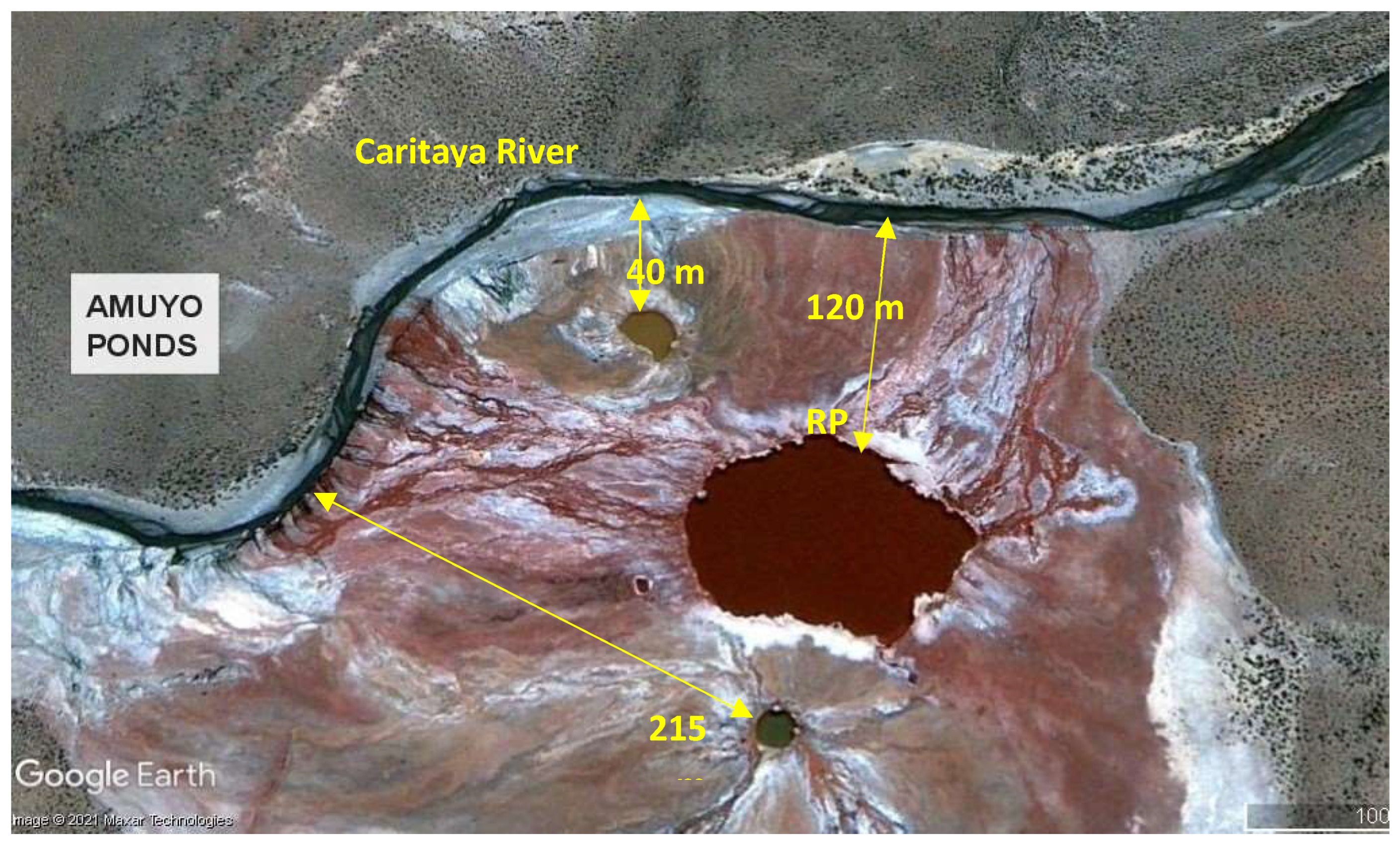
- Cornejo, L.; Lienqueo, H.; Vilca, P. Hydro-chemical characteristics, water quality assessment and water relationship (HCA) of the Amuyo Lagoons, Andean Altiplano, Chile. Desalination Water Treat. 2019, 153, 36–45. [Google Scholar] [CrossRef]
- Tapia, J. , Schneider, B., Inostroza, M., Álvarez-Amado, F., Luque, J.A., Aguilera, F., Parra, S., Bravo, M. Naturally elevated arsenic in the Altiplano-Puna and the link to recent (Mio-Pliocene to Quaternary) volcanic activity, high crustal thickness, and geological structures. J. South Am. Earth Sci. 2021, 105, 102905. [Google Scholar] [CrossRef]
- Pincetti-Zúniga, G.P.; Richards, L.A.; Daniele, L.; Boyce, A.J. Hydrochemical characterization, spatial distribution, and geochemical controls on arsenic and boron in waters from arid Arica and Parinacota, northern Chile. Sci. Total Environ. 2022, 806, 150206. [Google Scholar] [CrossRef] [PubMed]
- MINAGRI, Ministerio de Agricultura, CNR. Diagnóstico de la subcuenca aportante al Embalse Caritaya Región de Arica y Parinacota. 2014. https://bibliotecadigital.ciren.cl/items/6eb72957-ffe2-4fed-89be-fadd7e8162d9 [Accessed october 17, 2021].
- Muñoz-Torres, P.; Márquez, S.L.; Sepúlveda-Chavera, G.; Cárdenas-Ninasivincha, S.; et al. Isolation and Identification of Bacteria from Three Geothermal Sites of the Atacama Desert and Their Plant-Beneficial Characteristics. Microorganisms 2023, 11, 2635. [Google Scholar] [CrossRef] [PubMed]
- Keegan KP, Glass EM, Meyer F. MG-RAST, a Metagenomics Service for Analysis of Microbial Community Structure and Function. Methods Mol Biol. 2016, 1399, 207–233. [CrossRef]
- Li D, Luo R, Liu CM, Leung CM, Ting HF, Sadakane K; et al. (2016). MEGAHIT v1. 0: A fast and scalable metagenome assembler driven by advanced methodologies and community practices. Methods. 2016, 102, 3–11. [CrossRef]
- Wattam AR, Abraham D, Dalay O, Disz TL, Driscoll T; et al. PATRIC, the bacterial bioinformatics database and analysis resource. Nucleic Acids Res. 2014, 42, D581–D591. [CrossRef]
- Rothschild, L.; Mancinelli, R. Life in extreme environments. Nature 2001, 409, 1092–1101. [Google Scholar] [CrossRef]
- Vilo, C.; Galetovic, A.; Dong, Q.; Gómez-Silva, B. A Metagenomics Insight in the Cyanosphere of Edible Andean Macrocolonies (Llayta). Austin J Proteomics Bioinform. & Genomics. 2023, 8, 1034. [Google Scholar]
- McKay, C.P.; Friedmann, E.I.; Gómez-Silva, B.; Cáceres, L.; Andersen, D.T.; Landheim, R. Temperature and moisture conditions for life in the extreme arid region of the Atacama Desert: Four years of observations including the El Niño of 1997-98. Astrobiology 2003, 3, 393–406. [Google Scholar] [CrossRef]
- Bull, A.T.; Asenjo, J.A.; Goodfellow, M; Gómez-Silva, B. The Atacama Desert: Technical Resources and the Growing Importance of Novel Microbial Diversity. Ann. Rev. Microbiol. 2016, 70, 215–234. [CrossRef]
- Uritskiy, G.; Tisza, M.J.; Gelsinger, D.R.; Munn, A.; Taylor, J.; DiRuggiero, J. Cellular life from the three domains and viruses are transcriptionally active in a hypersaline desert community. Environ. Microbiol. 2021, 23, 3401–3417. [Google Scholar] [CrossRef] [PubMed]
- Pérez, V.; Cortés, J.; Marchant, F.; Dorador, C.; Molina, V.; et al. Aquatic Thermal Reservoirs of Microbial Life in a Remote and Extreme High Andean Hydrothermal System. Microorganisms 2020, 8, 208. [Google Scholar] [CrossRef] [PubMed]
- Borsodi, A.K.; Aszalós, J.M.; Megyes, M.; Nagy, B. Benthic Bacterial Diversity of High-Altitude Athalassohaline Lakes of the Puna de Atacama (Central Andes). Geomicrobiol. J. 2021, 39, 28–38. [Google Scholar] [CrossRef]
- Podar, P.T.; Yang, Z.; Björnsdóttir, S.H.; Podar, M. Comparative Analysis of Microbial Diversity Across Temperature Gradients in Hot Springs from Yellowstone and Iceland. Front. Microbiol. 2020, 11, 1625. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Garcia, L.; Fernandez-Martinez, M.A.; García-Villadangos, M.; Blanco, Y.; et al. Microbial Biomarker Transition in High-Altitude Sinter Mounds From El Tatio (Chile) Through Different Stages of Hydrothermal Activity. Front. Microbiol. 2019, 9, 3350. [Google Scholar] [CrossRef]
- Valenzuela, B.; Solís-Cornejo, F.; Araya, R.; Zamorano, P. Isolation and Characterization of Thermus thermophilus Strain ET-1: An Extremely Thermophilic Bacterium with Extracellular Thermostable Proteolytic Activity Isolated from El Tatio Geothermal Field, Antofagasta, Chile. Int. J. Mol. Sci. 2023, 24, 14512. [Google Scholar] [CrossRef]
- Santos, F; Yarza, P.; Parro, V.; Meseguer, I.; Rosselló-Móra, R.; Antón, J. Culture-Independent Approaches for Studying Viruses from Hypersaline Environments. Appl. Environ. Microbiol. 2012, 78. [CrossRef]
- Nazaries, L.; Murrell, J.C.; Millard, P.; Baggs, L.; Singh, B.K. Methane, microbes and models: Fundamental understanding of the soil methane cycle for future predictions. Environ. Microbiol. 2013, 15, 2395–2417. [Google Scholar] [CrossRef]
- Schulze-Makuch, D.; Wagner, D.; Kounaves, S.P.; Mangelsdorf, K.; et al. Transitory microbial habitat in the hyperarid Atacama Desert. PNAS 2018, 115, 2670–2675. [Google Scholar] [CrossRef]
- Hwang, Y.; Rahlff, J.; Schulze-Makuch, D; Schloter, M.; Probst, A.J. Diverse viruses carrying genes for microbial extremotolerance in the Atacama Desert hyperarid soil. mSystems 2021, 6, e00385–21. [CrossRef]
- Crits-Christoph, A.; Gelsinger, D.R.; Ma, B.; Wierzchos, J.; et al. Functional interactions of archaea, bacteria and viruses in a hypersaline endolithic community. Environ. Microbiol. 2016, 18, 2064–2077. [Google Scholar] [CrossRef] [PubMed]
- Busse, L.; Tisza, M.; DiRuggiero, J. Viruses Ubiquity and Diversity in Atacama Desert Endolithic Communities. Viruses 2022, 14, 1983. [Google Scholar] [CrossRef] [PubMed]

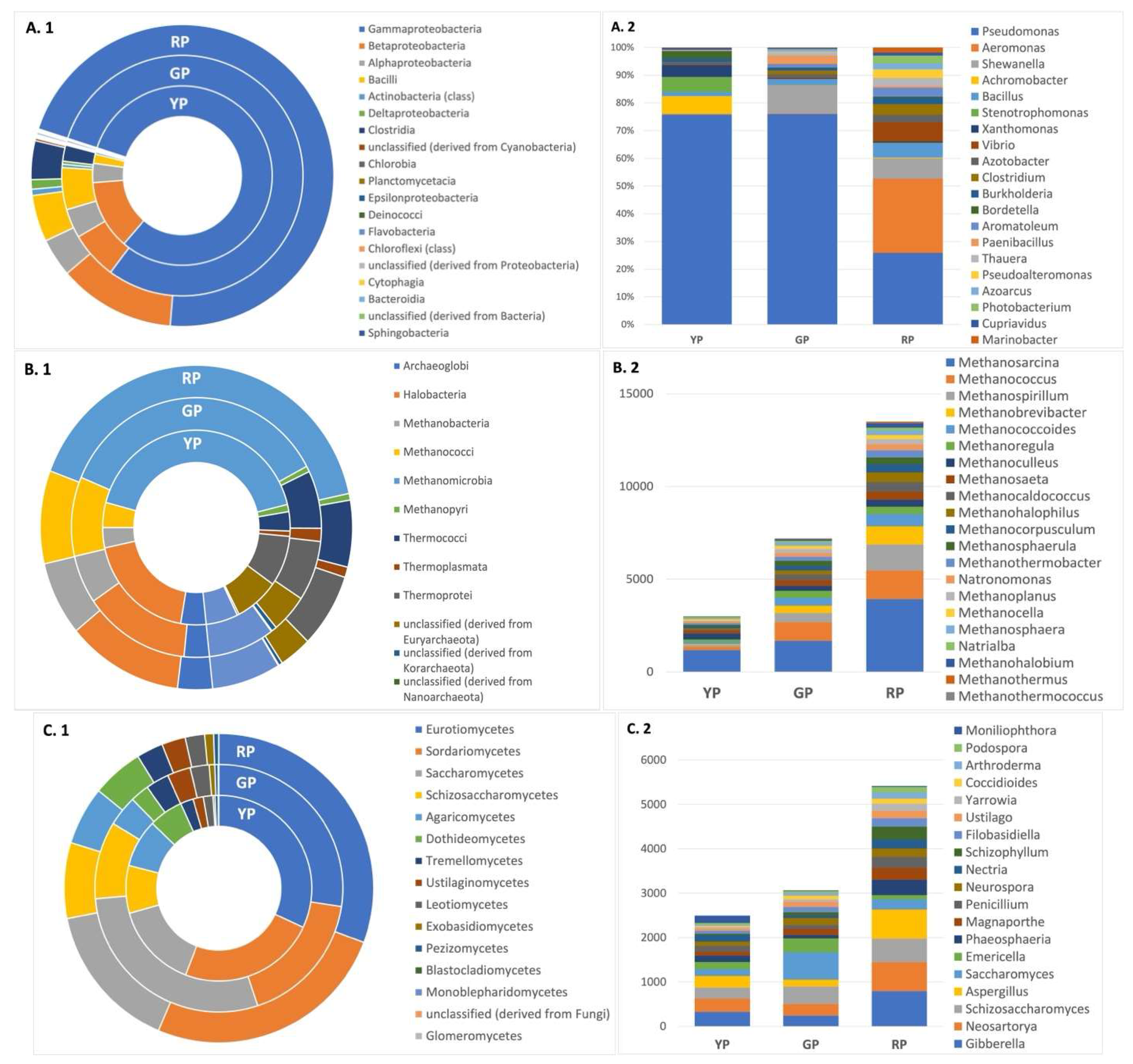

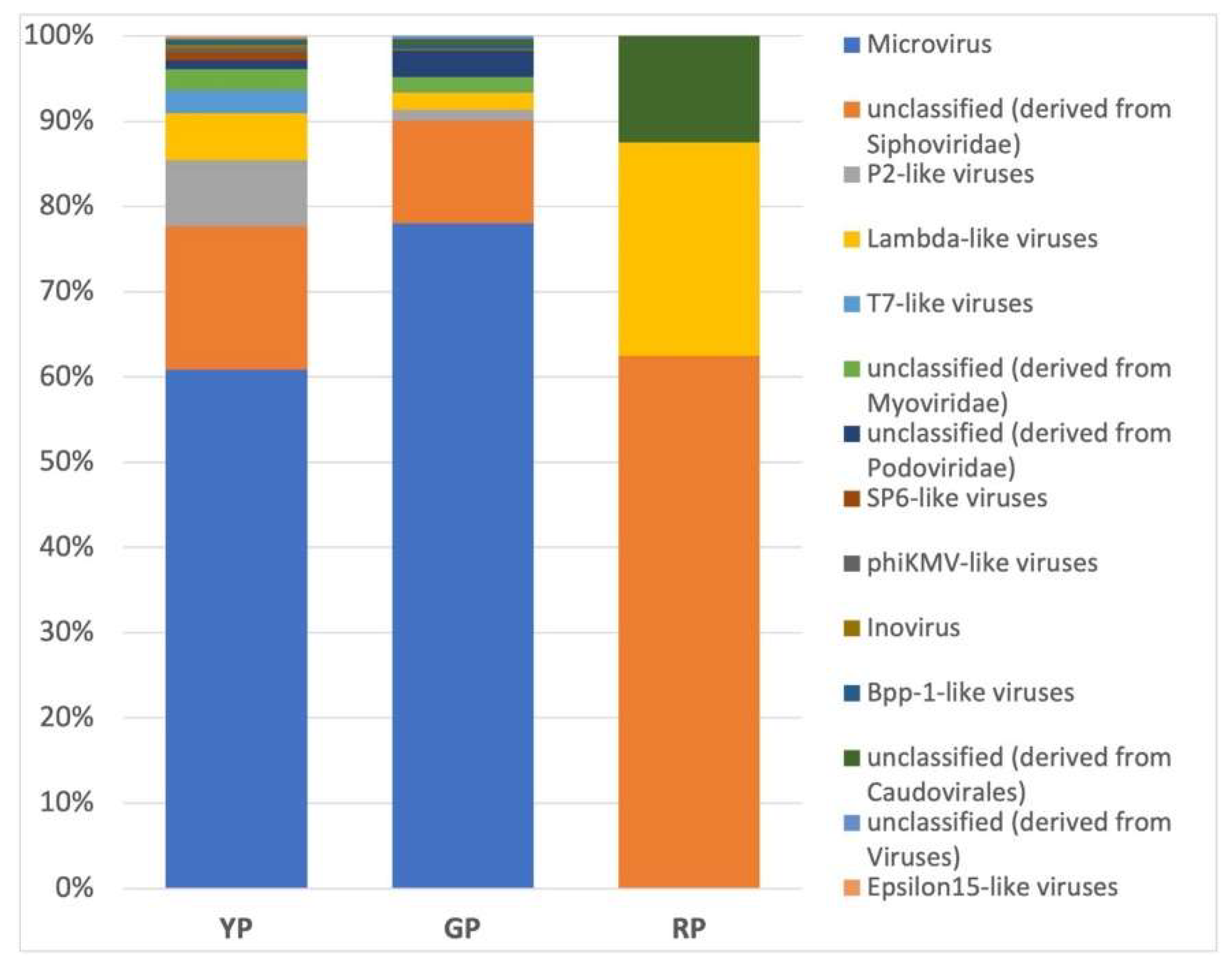
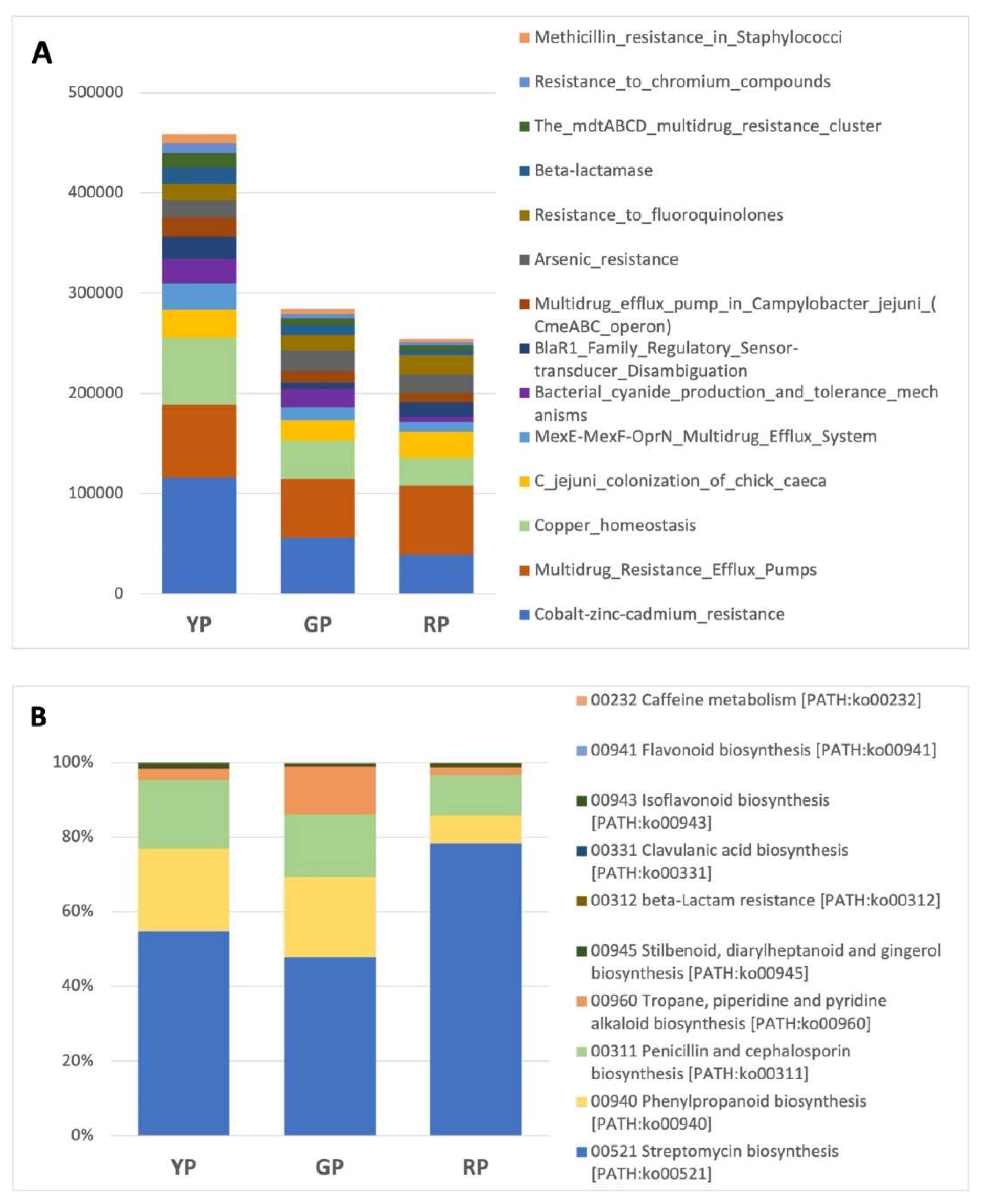
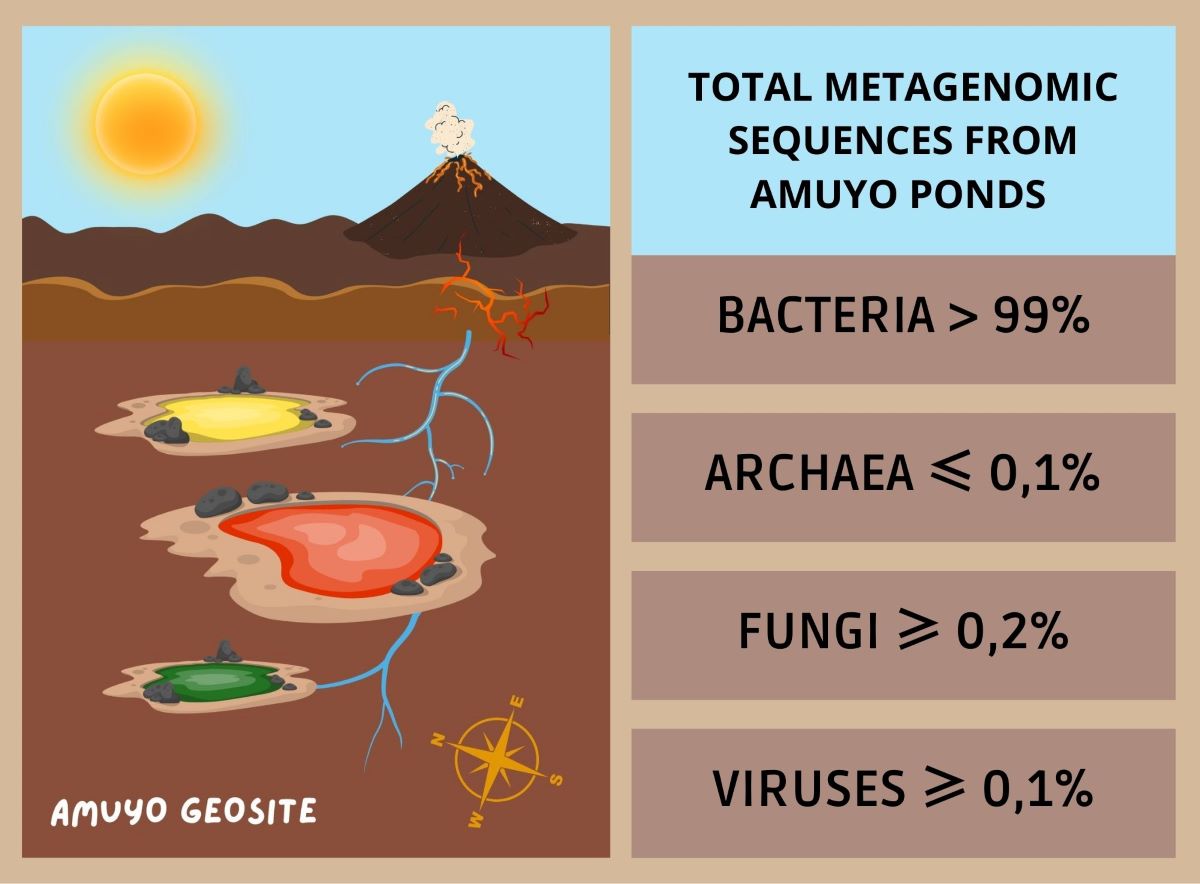
| Pond | Sequences post QC | GC % | Predicted function |
|---|---|---|---|
| GREEN | 23,128,825 | 57 ± 10 | 8,268,056 |
| YELLOW | 25,535,805 | 62 ± 7 | 7,424,754 |
| RED | 27,505,664 | 57 ± 12 | 10,157,651 |
| DOMAIN | GREEN POND | YELLOW POND | RED POND |
|---|---|---|---|
| BACTERIA | 99,52% | 99,71% | 99,38% |
| ARCHAEA | 00,08% | 00,03% | 00,13% |
| EUKARYA | 00,28% | 00,11% | 00,26% |
| VIRUS | 00,10% | 00,11% | 00,17% |
| OTHERS | 00,05% | 00,01% | 00,07% |
| Pond | Genus | Completeness (%) | Error (%) |
| Yellow | Stenotrophomonas | 100 | 0 |
| Achromobacter | 100 | 1 | |
| Pseudomonas | 95 | 2 | |
| Bacillus | 98 | 2 | |
| Thalassospira | 99 | 2 | |
| Green | Azoarcus | 100 | 4 |
| Pseudomonas | 100 | 6.1 | |
| Aeromonas | 100 | 5 | |
| Red | Azoarcus | 100 | 4 |
| Anaerobacillus | 92 | 3 | |
| Pseudomonas | 91 | 4 | |
| Pseudomonas | 99 | 11 | |
| Bacillus | 94 | 16 |
| Pond | Virus | Completeness (%) | Error (%) |
| YP | Bacillus phage | 101.58 | 2.07 |
| NA | 100 | 4 | |
| NA | 100 | 7.78 | |
| NA | 95.82 | 9.69 | |
| NA | 92.68 | 8.4 | |
| GP | NA | 102.08 | 3.78 |
| Bacteriophage Lily | 100 | 5.98 | |
| NA | 100 | 3.7 | |
| NA | 100 | 2.7 | |
| NA | 100 | 5.57 | |
| Rhizobium phage RR1-B | 90.21 | 2.16 | |
| RP | Circular genetic element sp | 100 | 8.48 |
| NA | 98.6 | 9.69 | |
| NA | 90.41 | 1.29 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




