Submitted:
02 September 2024
Posted:
03 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Literature Search Strategy
2.2. Bibliometric Analysis
3. Results
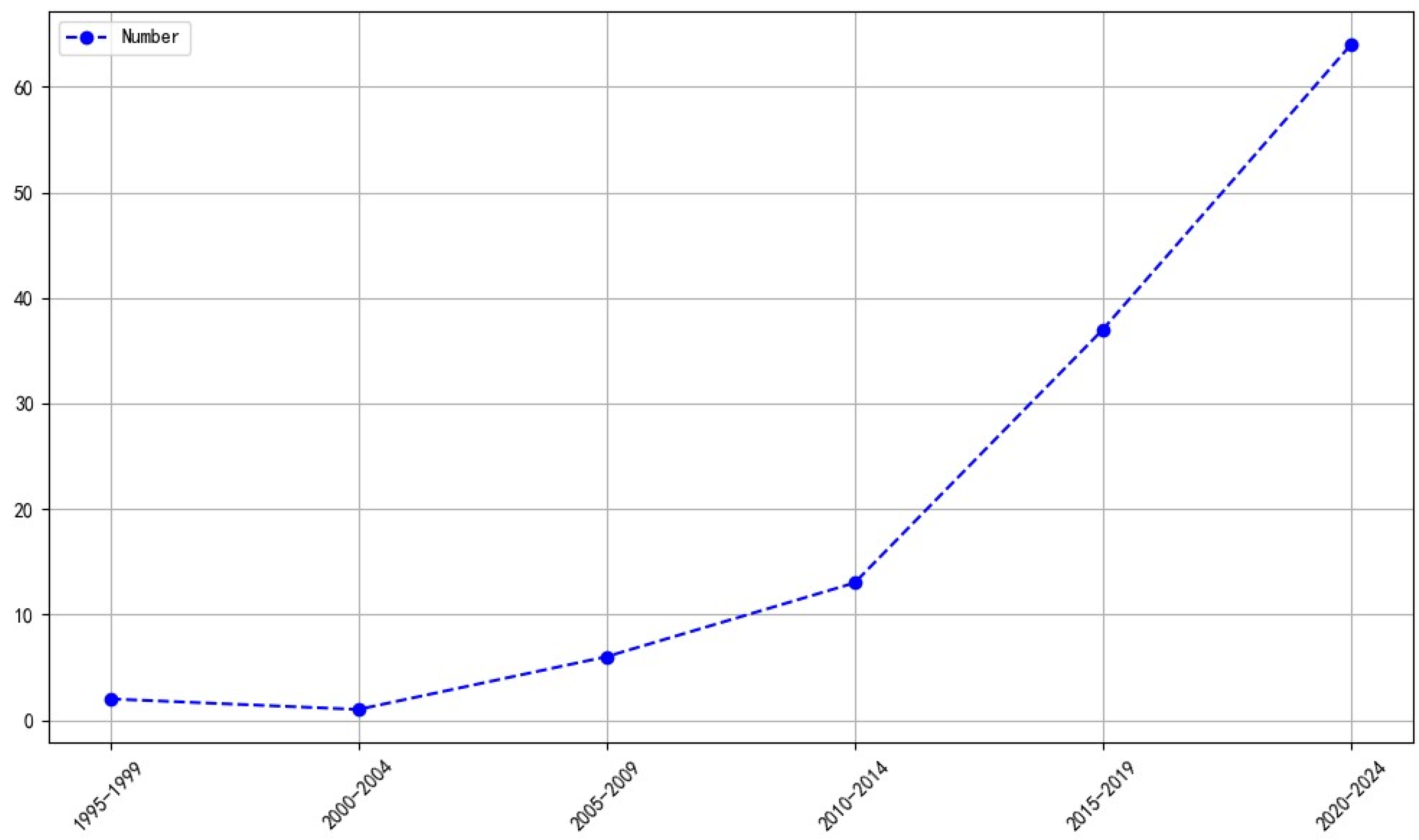
3.1. Global Trends in Publications
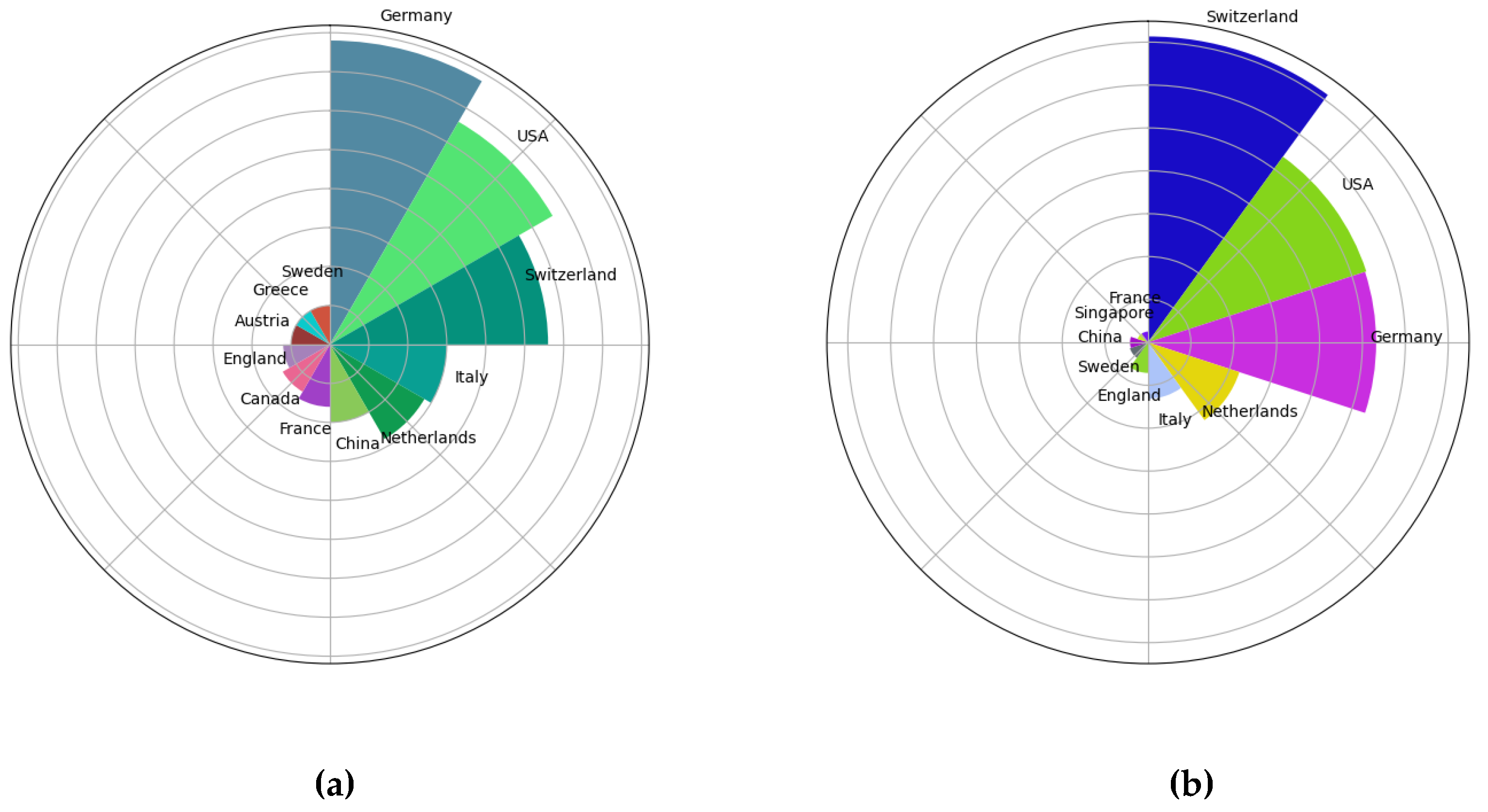
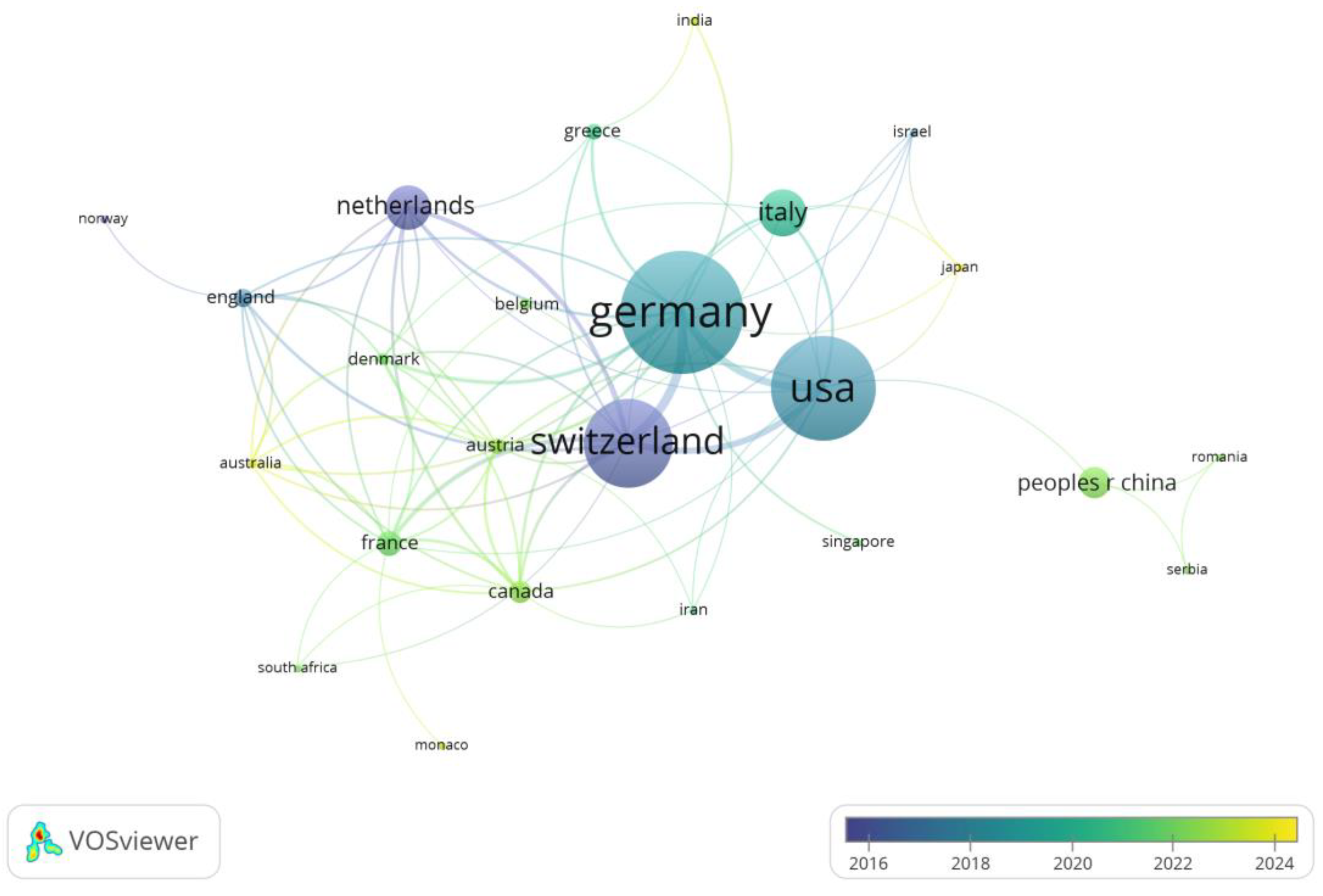
3.2. Analysis of Country Contributions
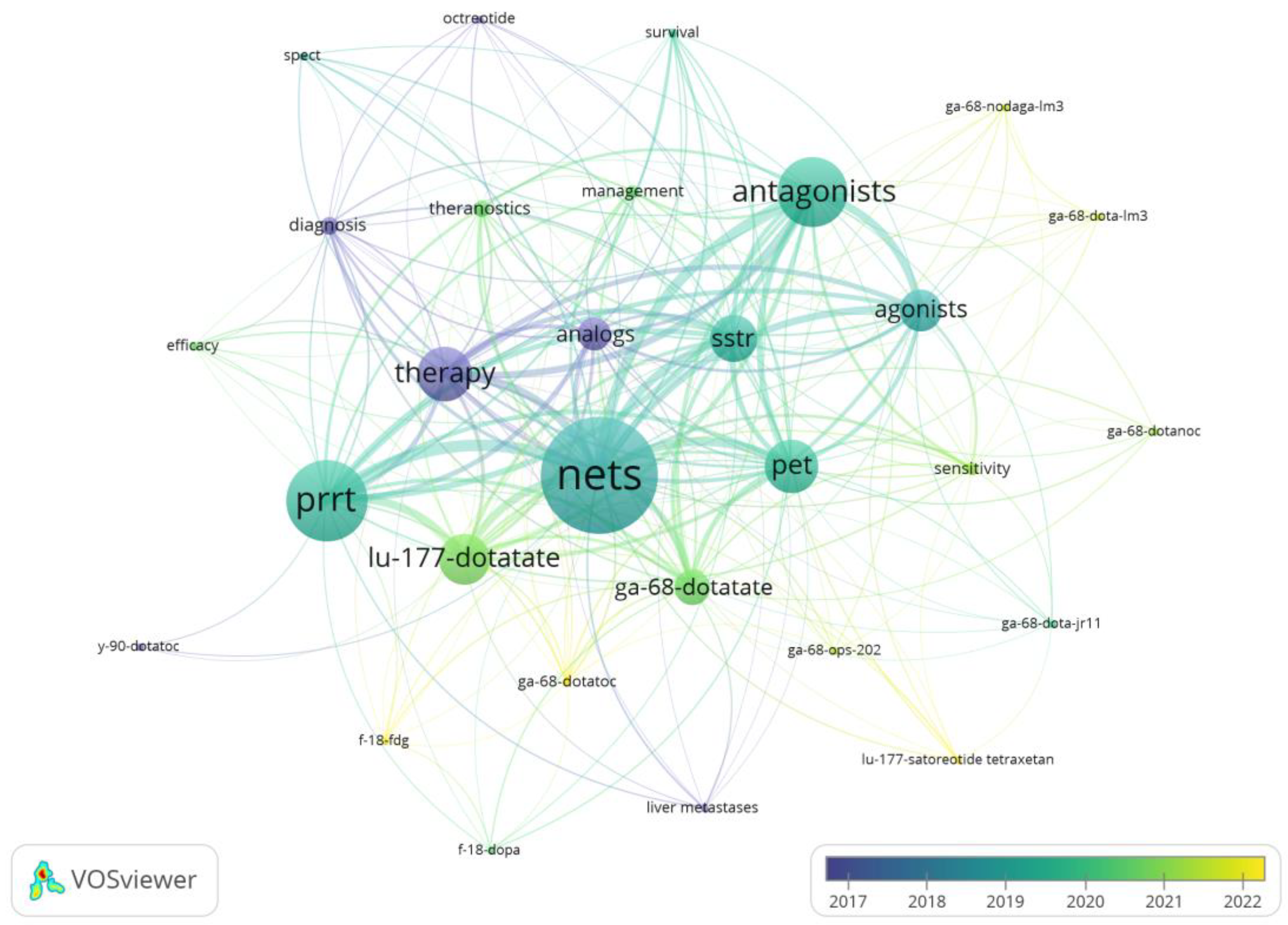
3.3. Analysis of Keywords
3.4. Analyses of Clinically Comparative Articles
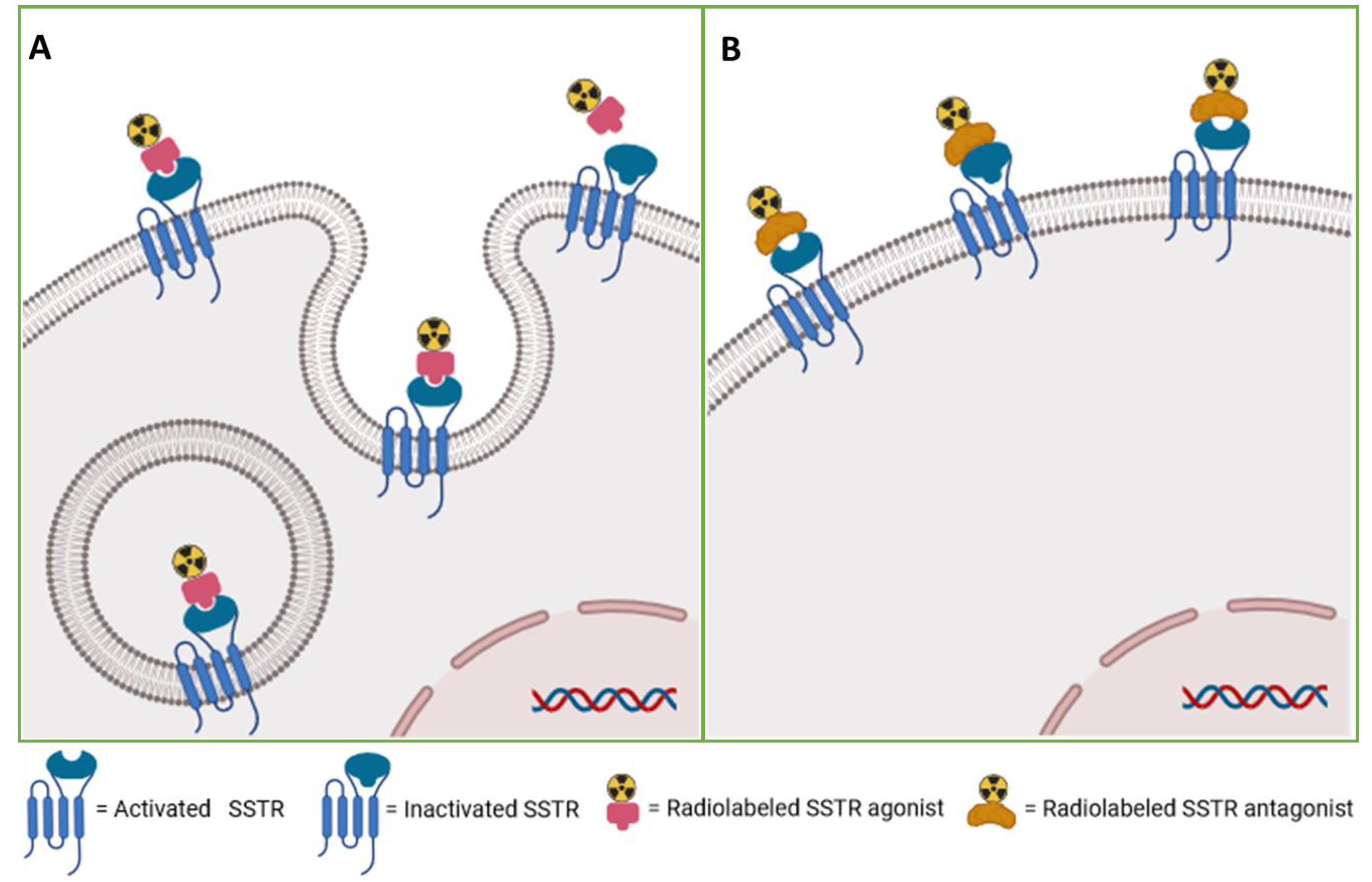
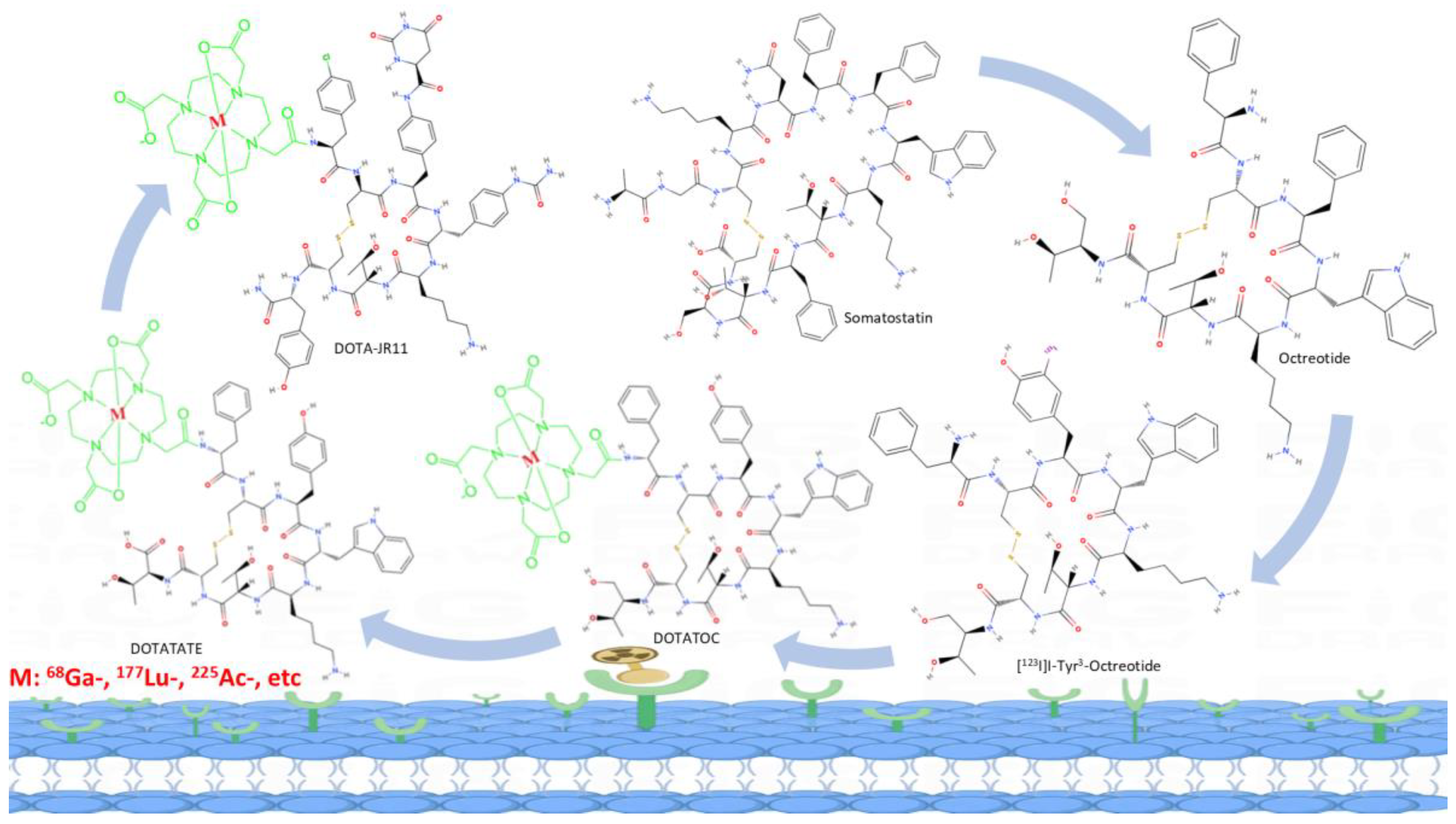
3.4.1. Diagnosis of Neuroendocrine Tumors
3.4.2. Radiotherapeutics for Neuroendocrine Tumors
3.4.3. Paired Theranostic
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fortunati, E.; Bonazzi, N.; Zanoni, L.; Fanti, S.; Ambrosini, V. Molecular imaging Theranostics of Neuroendocrine Tumors. Seminars in nuclear medicine 2023, 53, 539–554. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.; Barnett, E.; Rodger, E.J.; Chatterjee, A. Subramaniam RM: Neuroendocrine Neoplasms: Genetics and Epigenetics. PET clinics 2023, 18, 169–187. [Google Scholar] [CrossRef]
- Yao, J.C.; Hassan, M.; Phan, A.; Dagohoy, C.; Leary, C.; Mares, J.E.; Abdalla, E.K.; Fleming, J.B.; Vauthey, J.N.; Rashid, A.; et al. One hundred years after "carcinoid": epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2008, 26, 3063–3072. [Google Scholar] [CrossRef] [PubMed]
- Modlin, I.M.; Oberg, K.; Chung, D.C.; Jensen, R.T.; de Herder, W.W.; Thakker, R.V.; Caplin, M.; Delle Fave, G.; Kaltsas, G.A.; Krenning, E.P.; et al. Gastroenteropancreatic neuroendocrine tumours. The Lancet Oncology 2008, 9, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Hope, T.A.; Pavel, M.; Bergsland, E.K. Neuroendocrine Tumors and Peptide Receptor Radionuclide Therapy: When Is the Right Time? Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2022, 40, 2818–2829. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, V.; Kunikowska, J.; Baudin, E.; Bodei, L.; Bouvier, C.; Capdevila, J.; Cremonesi, M.; de Herder, W.W.; Dromain, C.; Falconi, M.; et al. Consensus on molecular imaging and theranostics in neuroendocrine neoplasms. European journal of cancer (Oxford, England : 1990) 2021, 146, 56–73. [Google Scholar] [CrossRef]
- Benali, N.; Ferjoux, G.; Puente, E.; Buscail, L.; Susini, C. Somatostatin receptors. Digestion 2000, 62 Suppl 1, 27–32. [Google Scholar] [CrossRef]
- Reubi, J.C. Peptide receptors as molecular targets for cancer diagnosis and therapy. Endocrine reviews 2003, 24, 389–427. [Google Scholar] [CrossRef]
- Maqsood, M.H.; Tameez Ud Din, A.; Khan, A.H. Neuroendocrine Tumor Therapy with Lutetium-177: A Literature Review. Cureus 2019, 11, e3986. [Google Scholar] [CrossRef]
- Maccauro, M.; Follacchio, G.A.; Spreafico, C.; Coppa, J.; Seregni, E. Safety and Efficacy of Combined Peptide Receptor Radionuclide Therapy and Liver Selective Internal Radiation Therapy in a Patient With Metastatic Neuroendocrine Tumor. Clinical nuclear medicine 2019, 44, e286–e288. [Google Scholar] [CrossRef]
- Marinova, M.; Mücke, M.; Fischer, F.; Essler, M.; Cuhls, H.; Radbruch, L.; Ghaei, S.; Conrad, R.; Ahmadzadehfar, H. Quality of life in patients with midgut NET following peptide receptor radionuclide therapy. European journal of nuclear medicine and molecular imaging 2019, 46, 2252–2259. [Google Scholar] [CrossRef] [PubMed]
- Martini, C.; Buxbaum, S.; Rodrigues, M.; Nilica, B.; Scarpa, L.; Holzner, B.; Virgolini, I.; Gamper, E.M. Quality of Life in Patients with Metastatic Gastroenteropancreatic Neuroendocrine Tumors Receiving Peptide Receptor Radionuclide Therapy: Information from a Monitoring Program in Clinical Routine. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2018, 59, 1566–1573. [Google Scholar] [CrossRef] [PubMed]
- Maecke, H.R.; Hofmann, M.; Haberkorn, U. (68)Ga-labeled peptides in tumor imaging. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2005, 46 Suppl 1, 172s–178s. [Google Scholar] [PubMed]
- Ambrosini, V.; Fani, M.; Fanti, S.; Forrer, F.; Maecke, H.R. Radiopeptide imaging and therapy in Europe. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2011, 52 Suppl 2, 42s–55s. [Google Scholar] [CrossRef]
- Sakellis, C.; Jacene, H.A. Neuroendocrine Tumors: Diagnostics. PET clinics 2024, 19, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Marincek, N.; Jörg, A.C.; Brunner, P.; Schindler, C.; Koller, M.T.; Rochlitz, C.; Müller-Brand, J.; Maecke, H.R.; Briel, M.; Walter, M.A. Somatostatin-based radiotherapy with [90Y-DOTA]-TOC in neuroendocrine tumors: long-term outcome of a phase I dose escalation study. Journal of translational medicine 2013, 11, 17. [Google Scholar] [CrossRef]
- Cescato, R.; Schulz, S.; Waser, B.; Eltschinger, V.; Rivier, J.E.; Wester, H.J.; Culler, M.; Ginj, M.; Liu, Q.; Schonbrunn, A.; et al. Internalization of sst2, sst3, and sst5 receptors: effects of somatostatin agonists and antagonists. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2006, 47, 502–511. [Google Scholar]
- Weckbecker, G.; Lewis, I.; Albert, R.; Schmid, H.A.; Hoyer, D.; Bruns, C. Opportunities in somatostatin research: biological, chemical and therapeutic aspects. Nature reviews Drug discovery 2003, 2, 999–1017. [Google Scholar] [CrossRef]
- Leung, K. (68)Ga-1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraacetic acid-Cpa-cyclo(d-Cys-amino-Phe-hydroorotic acid-D-4-amino-Phe(carbamoyl)-Lys-Thr-Cys)-D-Tyr-NH(2) (JR11). In Molecular Imaging and Contrast Agent Database (MICAD); edn.; National Center for Biotechnology Information (US): Bethesda (MD), 2004. [Google Scholar]
- Ginj, M.; Zhang, H.; Waser, B.; Cescato, R.; Wild, D.; Wang, X.; Erchegyi, J.; Rivier, J.; Mäcke, H.R.; Reubi, J.C. Radiolabeled somatostatin receptor antagonists are preferable to agonists for in vivo peptide receptor targeting of tumors. Proceedings of the National Academy of Sciences of the United States of America 2006, 103, 16436–16441. [Google Scholar] [CrossRef]
- Wild, D.; Fani, M.; Behe, M.; Brink, I.; Rivier, J.E.; Reubi, J.C.; Maecke, H.R.; Weber, W.A. First clinical evidence that imaging with somatostatin receptor antagonists is feasible. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2011, 52, 1412–1417. [Google Scholar] [CrossRef]
- Wild, D.; Fani, M.; Fischer, R.; Del Pozzo, L.; Kaul, F.; Krebs, S.; Fischer, R.; Rivier, J.E.; Reubi, J.C.; Maecke, H.R.; et al. Comparison of somatostatin receptor agonist and antagonist for peptide receptor radionuclide therapy: a pilot study. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2014, 55, 1248–1252. [Google Scholar] [CrossRef] [PubMed]
- Imperiale, A.; Jha, A.; Meuter, L.; Nicolas, G.P.; Taïeb, D.; Pacak, K. The Emergence of Somatostatin Antagonist-Based Theranostics: Paving the Road Toward Another Success? Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2023, 64, 682–684. [Google Scholar] [CrossRef] [PubMed]
- Ninkov, A.; Frank, J.R.; Maggio, L.A. Bibliometrics: Methods for studying academic publishing. Perspectives on medical education 2022, 11, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Foy, J.P.; Serresse, L.; Decavèle, M.; Allaire, M.; Nathan, N.; Renaud, M.C.; Sabourdin, N.; Souala-Chalet, Y.; Tamzali, Y.; Taytard, J.; et al. Clues for improvement of research in objective structured clinical examination. Medical education online 2024, 29, 2370617. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Cheng, Y.; Wang, X.; Yao, S.; Bai, C.; Zhao, H.; Jia, R.; Xu, J.; Huo, L. Head-to-Head Comparison of (68)Ga-DOTA-JR11 and (68)Ga-DOTATATE PET/CT in Patients with Metastatic, Well-Differentiated Neuroendocrine Tumors: A Prospective Study. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2020, 61, 897–903. [Google Scholar] [CrossRef]
- Liu, M.; Ren, C.; Zhang, H.; Zhang, Y.; Huang, Z.; Jia, R.; Cheng, Y.; Bai, C.; Xu, Q.; Zhu, W.; et al. Evaluation of the safety, biodistribution, dosimetry of [(18)F]AlF-NOTA-LM3 and head-to-head comparison with [(68)Ga]Ga-DOTATATE in patients with well-differentiated neuroendocrine tumors: an interim analysis of a prospective trial. European journal of nuclear medicine and molecular imaging 2024. [Google Scholar]
- Viswanathan, R.; Ballal, S.; Yadav, M.P.; Roesch, F.; Sheokand, P.; Satapathy, S.; Tripathi, M.; Agarwal, S.; Moon, E.S.; Bal, C. Head-to-Head Comparison of SSTR Antagonist [(68)Ga]Ga-DATA(5m)-LM4 with SSTR Agonist [(68)Ga]Ga-DOTANOC PET/CT in Patients with Well Differentiated Gastroenteropancreatic Neuroendocrine Tumors: A Prospective Imaging Study. Pharmaceuticals (Basel, Switzerland) 2024, 17. [Google Scholar]
- Nicolas, G.P.; Schreiter, N.; Kaul, F.; Uiters, J.; Bouterfa, H.; Kaufmann, J.; Erlanger, T.E.; Cathomas, R.; Christ, E.; Fani, M.; et al. Sensitivity Comparison of (68)Ga-OPS202 and (68)Ga-DOTATOC PET/CT in Patients with Gastroenteropancreatic Neuroendocrine Tumors: A Prospective Phase II Imaging Study. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2018, 59, 915–921. [Google Scholar] [CrossRef]
- Liu, M.; Cheng, Y.; Bai, C.; Zhao, H.; Jia, R.; Chen, J.; Zhu, W.; Huo, L. Gallium-68 labeled somatostatin receptor antagonist PET/CT in over 500 patients with neuroendocrine neoplasms: experience from a single center in China. European journal of nuclear medicine and molecular imaging 2024, 51, 2002–2011. [Google Scholar] [CrossRef]
- Baum, R.P.; Zhang, J.; Schuchardt, C.; Müller, D.; Mäcke, H. First-in-Humans Study of the SSTR Antagonist (177)Lu-DOTA-LM3 for Peptide Receptor Radionuclide Therapy in Patients with Metastatic Neuroendocrine Neoplasms: Dosimetry, Safety, and Efficacy. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2021, 62, 1571–1581. [Google Scholar] [CrossRef]
- Krebs, S.; O'Donoghue, J.A.; Biegel, E.; Beattie, B.J.; Reidy, D.; Lyashchenko, S.K.; Lewis, J.S.; Bodei, L.; Weber, W.A.; Pandit-Taskar, N. Comparison of (68)Ga-DOTA-JR11 PET/CT with dosimetric (177)Lu-satoreotide tetraxetan ((177)Lu-DOTA-JR11) SPECT/CT in patients with metastatic neuroendocrine tumors undergoing peptide receptor radionuclide therapy. European journal of nuclear medicine and molecular imaging 2020, 47, 3047–3057. [Google Scholar] [CrossRef]
- Hou, L.; Pan, Y.; Zhu, J.J.H. Impact of scientific, economic, geopolitical, and cultural factors on international research collaboration. Journal of Informetrics 2021, 15, 101194. [Google Scholar] [CrossRef]
- Brower, H.H.; Nicklas, B.J.; Nader, M.A.; Trost, L.M.; Miller, D.P. Creating effective academic research teams: Two tools borrowed from business practice. Journal of clinical and translational science 2020, 5, e74. [Google Scholar] [CrossRef] [PubMed]
- Bennett, L.M.; Gadlin, H. Collaboration and team science: from theory to practice. Journal of investigative medicine : the official publication of the American Federation for Clinical Research 2012, 60, 768–775. [Google Scholar] [CrossRef]
- Otte, A.; Herrmann, R.; Heppeler, A.; Behe, M.; Jermann, E.; Powell, P.; Maecke, H.R.; Muller, J. Yttrium-90 DOTATOC: first clinical results. European journal of nuclear medicine 1999, 26, 1439–1447. [Google Scholar] [CrossRef]
- Forrer, F.; Uusijärvi, H.; Waldherr, C.; Cremonesi, M.; Bernhardt, P.; Mueller-Brand, J.; Maecke, H.R. A comparison of (111)In-DOTATOC and (111)In-DOTATATE: biodistribution and dosimetry in the same patients with metastatic neuroendocrine tumours. European journal of nuclear medicine and molecular imaging 2004, 31, 1257–1262. [Google Scholar] [CrossRef] [PubMed]
- Forrer, F.; Uusijärvi, H.; Storch, D.; Maecke, H.R.; Mueller-Brand, J. Treatment with 177Lu-DOTATOC of patients with relapse of neuroendocrine tumors after treatment with 90Y-DOTATOC. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2005, 46, 1310–1316. [Google Scholar] [PubMed]
- Otte, A.; Jermann, E.; Behe, M.; Goetze, M.; Bucher, H.C.; Roser, H.W.; Heppeler, A.; Mueller-Brand, J.; Maecke, H.R. DOTATOC: a powerful new tool for receptor-mediated radionuclide therapy. European journal of nuclear medicine 1997, 24, 792–795. [Google Scholar] [CrossRef] [PubMed]
- Hennrich, U.; Benešová, M. [(68)Ga]Ga-DOTA-TOC: The First FDA-Approved (68)Ga-Radiopharmaceutical for PET Imaging. Pharmaceuticals (Basel, Switzerland) 2020, 13. [Google Scholar]
- Öberg, K.; Sundin, A. Imaging of Neuroendocrine Tumors. Frontiers of hormone research 2016, 45, 142–151. [Google Scholar]
- Pouget, J.P.; Navarro-Teulon, I.; Bardiès, M.; Chouin, N.; Cartron, G.; Pèlegrin, A.; Azria, D. Clinical radioimmunotherapy--the role of radiobiology. Nature reviews Clinical oncology 2011, 8, 720–734. [Google Scholar] [CrossRef]
- Theodoropoulou, M.; Stalla, G.K. Somatostatin receptors: from signaling to clinical practice. Frontiers in neuroendocrinology 2013, 34, 228–252. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Parihar, A.S.; Bodei, L.; Hope, T.A.; Mallak, N.; Millo, C.; Prasad, K.; Wilson, D.; Zukotynski, K.; Mittra, E. Somatostatin Receptor Imaging and Theranostics: Current Practice and Future Prospects. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2021, 62, 1323–1329. [Google Scholar] [CrossRef] [PubMed]
- Sheikhbahaei, S.; Sadaghiani, M.S.; Rowe, S.P.; Solnes, L.B. Neuroendocrine Tumor Theranostics: An Update and Emerging Applications in Clinical Practice. AJR American journal of roentgenology 2021, 217, 495–506. [Google Scholar] [CrossRef] [PubMed]
- Salih, S.; Alkatheeri, A.; Alomaim, W.; Elliyanti, A. Radiopharmaceutical Treatments for Cancer Therapy, Radionuclides Characteristics, Applications, and Challenges. Molecules (Basel, Switzerland) 2022, 27. [Google Scholar] [CrossRef]
- Bodei, L.; Weber, W.A. Somatostatin Receptor Imaging of Neuroendocrine Tumors: From Agonists to Antagonists. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2018, 59, 907–908. [Google Scholar] [CrossRef]
- Fani, M.; Braun, F.; Waser, B.; Beetschen, K.; Cescato, R.; Erchegyi, J.; Rivier, J.E.; Weber, W.A.; Maecke, H.R.; Reubi, J.C. Unexpected sensitivity of sst2 antagonists to N-terminal radiometal modifications. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2012, 53, 1481–1489. [Google Scholar] [CrossRef]
| Author | Zhu, W. | Liu, M. | Viswanathan, R. | |||
| Year | 2020 | 2024 | 2024 | |||
| Radiopharmaceuticals | [68Ga]Ga-DOTA-JR11 | [68Ga]Ga-DOTATATE | [18F]AlF-NOTA-LM3 | [68Ga]Ga-DOTATATE | [68Ga]Ga-DATA5m-LM4 | [68Ga]Ga-DOTANOC |
| Administration activity(MBq) | 155 ± 52 | 148 ± 52 | 288 ± 28 | 148 ± 18 | 150 | 92.5 |
| Spleen | 3.2 ± 1.3 | 22.5 ± 8.0 | 6.9 (2.8-12.1) | 20.2 (9.7-29.7) | 6.6 (4.0-9.7) | 11.3 (7.2-18.3) |
| p < 0.001 | p < 0.01 | p < 0.0001 | ||||
| Renal cortex/kidney | 6.7 ± 1.6 | 14.6 ± 3.8 | 10.3 (7.7-12.8) | 12.2 (9.5-14.8) | 9.8 (6.6-13.8) | 6.1 (4.1-9.6) |
| p < 0.001 | p = 0.04 | p = 0.0004 | ||||
| Adrenal glands | 2.1 ± 0.8 | 11.3 ± 4.4 | 11.6 (5.9-17.0) | 16.3 (9.7-25.1) | 4.1 (2.4-6.1) | 5.4 (1.6-6.6) |
| p < 0.001 | p < 0.01 | p = 0.695 | ||||
| Pituitary gland | 2.1 ± 1.6 | 7.7 ± 3.2 | 8.8 (3.4-10.9) | 11.1 (7.6-26.3) | 2.1 (1.2-3.5) | 1.7 (0.8-2.5) |
| p < 0.001 | p < 0.01 | p = 0.025 | ||||
| Stomach | 1.9 ± 0.6 | 7.1 ± 4.2 | 2.7 (2.0-3.4) | 6.2 (3.2-8.8) | - | - |
| p < 0.001 | p = 0.02 | - | ||||
| Liver | 2.8 ± 0.9 | 9.7 ± 3.0 | 2.9 (2.3-3.8) | 6.2 (3.6-9.9) | 2.5 (1.7-3.8) | 3.5 (2.4-5.3) |
| p < 0.001 | p < 0.01 | p = 0.01 | ||||
| Small intestine | 1.9 ± 0.5 | 6.1 ± 1.8 | 2.6 (2.0-3.6) | 5.8 (3.6-10.9) | - | - |
| p < 0.001 | p < 0.01 | - | ||||
| Pancreas | 1.7 ± 0.6 | 4.3 ± 1.9 | 1.8 (1.4-2.3) | 2.8 (1.8-4.3) | - | - |
| p < 0.001 | p < 0.01 | - | ||||
| Value | SUVmax | SUVmax | SULpeak | |||
| Author | Year | Radiopharmaceuticals | Administration activity(MBq) | All lesions | Liver lesions | Tumor-to-liver ratio | value | |||
| Nicolas, G. P. | 2018 | [68Ga]Ga-OPS202 (15 μg peptide) | 161 ± 21 | 12.3 (9.9-16.9) | p = 0.880 | 10.9 (9.5-20.7) | p = 0.547 | 5.3 (2.9-5.7) | p = 0.004 | SUVmax |
| [68Ga]Ga-DOTATOC (15 μg peptide) | 161 ± 11 | 12.4 (12.0-14.5) | 11.8 (10.6-14.0) | 1.9 (1.4-2.9) | ||||||
| [68Ga]Ga-OPS202 (50 μg peptide) | 172 ± 14 | 14.4 (9.6-16.3) | p = 0.966 | 12.6 ( 9.6-19.9) | p = 0.652 | 4.3 (3.4-6.3) | p = 0.008 | |||
| [68Ga]Ga-DOTATOC (15 μg peptide) | 161 ± 11 | 12.4 (12.0-14.5) | 11.8 (10.6-14.0) | 1.9 (1.4-2.9) | ||||||
| Zhu, W. | 2020 | [68Ga]Ga-DOTA-JR11 | 148 ± 52 | - | - | 18.6 ± 12.5 | p < 0.001 | 7.7 ± 5.4 | p < 0.001 | SUVmax |
| [68Ga]Ga-DOTATATE | 155 ± 52 | - | 27.3 ± 15.4 | 3.4 ± 2.0 | ||||||
| Liu, M. | 2024 | [68Ga]Ga-NODAGA-LM3 | 200 ± 11 | 57.4 ±38.5 | p < 0.001 | - | - | 10.4 ± 8.2 | p < 0.001 | SUVmax |
| [68Ga]Ga-DOTATATE | - | 40.0 ±22.8 | - | 4.4 ± 3.6 | ||||||
| [68Ga]Ga-NODAGA-JR11 | - | 39.7 ± 26.5 | p = 0.108 | - | - | 10.3 ± 9.9 | p < 0.001 | |||
| [68Ga]Ga-DOTATATE | - | 34.3 ± 23.9 | - | 5.3 ± 5.3 | ||||||
| [68Ga]Ga-DOTA-LM3 | 172±21 | 38.9 ± 32.1 | p = 0.858 | - | - | 14.1 ± 13.2 | p < 0.001 | |||
| [68Ga]Ga-DOTATATE | 155±52 | 37.2 ± 22.1 | - | 5.3 ± 5.2 | ||||||
| [68Ga]Ga-DOTA-JR11 | 148±52 | 28.9 ± 26.1 | p = 0.001 | - | - | 12.0 ± 10.6 | p < 0.001 | |||
| [68Ga]Ga-DOTATATE | 155±52 | 44.0 ± 25.7 | - | 5.5 ± 3.8 | ||||||
| All antagonists | - | 40.1 ± 32.5 | p = 0.772 | - | - | 12.1 ± 10.8 | p < 0.001 | |||
| [68Ga]Ga-DOTATATE | - | 39.4 ± 23.8 | - | 5.2 ± 4.5 | ||||||
| Liu, M. | 2024 | [18F]AlF-NOTA-LM3 | 288±28 | 22.1 (10.2-49.6) | p = 0.616 | 32.7 (12.7-50.9) | p = 0.713 | 12.1(4.8-24.3) | p < 0.001 | SUVmax |
| [68Ga]Ga-DOTATATE | 148±18 | 17.1 (10.0-48.1) | 31.4 (15.6-60.0) | 4.9 (1.6-9.8) | ||||||
| Viswanathan, R. | 2024 | [68Ga]Ga-DATA5m-LM4 | - | - | - | 15.7 (8.3-25.7) | p = 0.484 | 7.71 (3.3-14.4) | p = 0.008 | SULpeak |
| [68Ga]Ga-DOTANOC | - | - | 14.7 (7.3-20.9) | 4.1 (1.9-6.4) | ||||||
| Author | year | Radiopharmaceuticals | Absorbed dose by organs and tumors(Gy/GBq) | ||||
| whole body | kidney | spleen | tumor | liver metastases | |||
| Wild, D. | 2014 | [177Lu]Lu-DOTA-JR11 | 0.13 ± 0.031 | 1.8 ± 0.44 | 3.0 ± 2.3 | 7.0 (5.7–16) | - |
| [177Lu]Lu-DOTATATE | 0.083 ± 0.021 | 1.2 ± 0.35 | 2.5 ± 1.5 | 2.0 (1.2–4.6) | - | ||
| Baum, R. P. | 2021 | [177Lu]Lu-DOTA-LM3 | 0.121 | 2.3 | 3.4 | 51 | 93 |
| [177Lu]Lu-DOTATOC | 0.04 | 0.6 | 0.8 | 10 | 12 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




