Submitted:
01 September 2024
Posted:
03 September 2024
You are already at the latest version
Abstract
Keywords:
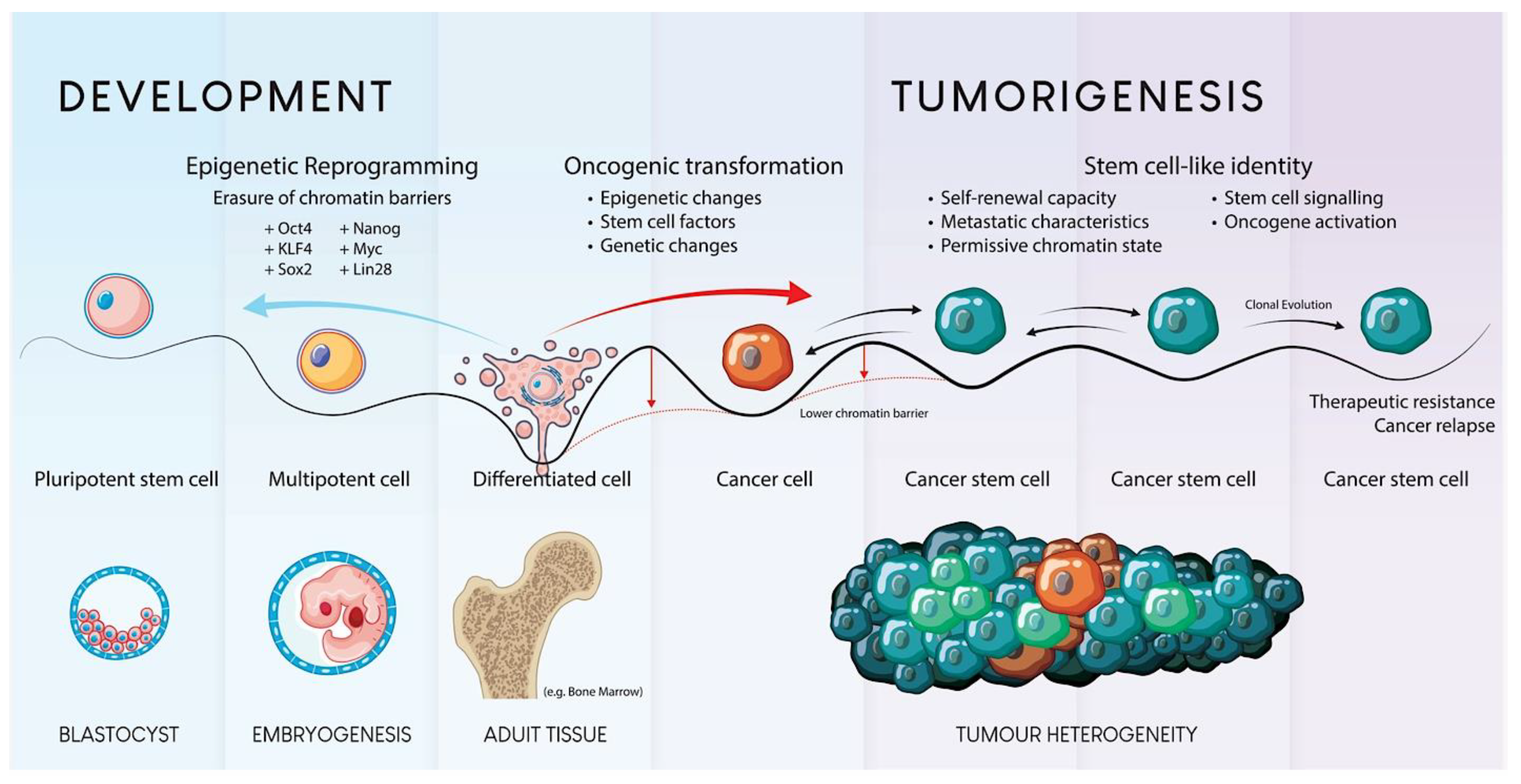
1. An Overview
2. Introduction
2.1. Background on Stem Cell-Derived Therapeutic Products
2.1.1. Definition and Types of Stem Cells
2.1.2. Therapeutic Applications and Clinical Potential
3. Tumorigenic Potential
3.1. Explanation of Tumorigenicity
3.2. Historical Context and Examples of Tumorigenic Outcomes
4. Importance of Addressing Tumorigenic Potential
4.1. Clinical Implications
4.1. Regulatory Concerns and Patient Safety
5. Mechanisms of Tumorigenicity
5.1. Intrinsic Factors
5.1.1. Genetic Mutations and Instability
5.1.2. Epigenetic Changes
5.1.3. Telomerase Activity and Cellular Immortality
5.2. Extrinsic Factors
5.2.1. Microenvironment Influences
5.2.2. Immune System Interactions
5.2.3. Inflammation and Cytokines Milieu
5.2.4. Comparative Analysis with Related Studies
6. Strategies to Mitigate Tumorigenic Risks in Stem Cell Therapies
6.1. Quality Control Measures
6.1.1. Stringent Culture and Expansion Protocols
6.1.2. Monitoring Genetic Stability
6.2. Refinement of Differentiation Protocols
6.2.1. Improved Differentiation Techniques
6.2.2. Minimizing Residual Undifferentiated Cells
6.3. Genomic and Epigenomic Screening Techniques
6.3.1. Advanced Screening Methodologies
6.3.2. Detection and Elimination of Tumorigenic Cells
7. Innovative Approaches to Enhance Safety
7.1. Gene Editing Technologies
7.3.1. Application of CRISPR/Cas9
7.3.2. Other Gene Editing Tools
7.2. Modulation with Small Molecules
7.2.1. Examples of Small Molecules Used in Stem Cell Research
7.2.2. Other Small Molecules
7.3. Studies and Applications
References
- Carvalho, T. Stem Cell-Derived Heart Cells Injected into First Patient. Nat Med 2023, 29. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Zickler, A.M.; El Andaloussi, S. Dosing Extracellular Vesicles. Adv Drug Deliv Rev 2021, 178. [Google Scholar] [CrossRef]
- Guo, Y.; Yu, Y.; Hu, S.; Chen, Y.; Shen, Z. The Therapeutic Potential of Mesenchymal Stem Cells for Cardiovascular Diseases. Cell Death Dis 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Schweitzer, J.S.; Song, B.; Herrington, T.M.; Park, T.-Y.; Lee, N.; Ko, S.; Jeon, J.; Cha, Y.; Kim, K.; Li, Q.; et al. Personalized IPSC-Derived Dopamine Progenitor Cells for Parkinson’s Disease. New England Journal of Medicine 2020, 382. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, S. Pluripotent Stem Cell-Based Cell Therapy—Promise and Challenges. Cell Stem Cell 2020, 27. [Google Scholar] [CrossRef]
- Sierra-Sánchez, Á.; Kim, K.H.; Blasco-Morente, G.; Arias-Santiago, S. Cellular Human Tissue-Engineered Skin Substitutes Investigated for Deep and Difficult to Heal Injuries. NPJ Regen Med 2021, 6. [Google Scholar] [CrossRef]
- Burnham, A.J.; Daley-Bauer, L.P.; Horwitz, E.M. Mesenchymal Stromal Cells in Hematopoietic Cell Transplantation. Blood Adv 2020, 4. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y.G. Intestinal Epithelial Plasticity and Regeneration via Cell Dedifferentiation. Cell Regeneration 2020, 9. [Google Scholar] [CrossRef]
- Wu, H.H.; Symersky, J.; Lu, M. Structure and Mechanism of a Redesigned Multidrug Transporter from the Major Facilitator Superfamily. Sci Rep 2020, 10. [Google Scholar] [CrossRef]
- Li, P.; Ou, Q.; Shi, S.; Shao, C. Immunomodulatory Properties of Mesenchymal Stem Cells/Dental Stem Cells and Their Therapeutic Applications. Cell Mol Immunol 2023, 20. [Google Scholar] [CrossRef]
- Müller, L.; Tunger, A.; Wobus, M.; von Bonin, M.; Towers, R.; Bornhäuser, M.; Dazzi, F.; Wehner, R.; Schmitz, M. Immunomodulatory Properties of Mesenchymal Stromal Cells: An Update. Front Cell Dev Biol 2021, 9. [Google Scholar] [CrossRef]
- Shi, M.; Liu, Z.W.; Wang, F.S. Immunomodulatory Properties and Therapeutic Application of Mesenchymal Stem Cells. Clin Exp Immunol 2011, 164. [Google Scholar] [CrossRef] [PubMed]
- Yi, T.; Song, S.U. Immunomodulatory Properties of Mesenchymal Stem Cells and Their Therapeutic Applications. Arch Pharm Res 2012, 35, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.; Qiu, L.; Veeraraghavan, V.P.; Sheu, C.-L.; Mony, U. Advances in IPSC Technology in Neural Disease Modeling, Drug Screening, and Therapy. Curr Stem Cell Res Ther 2024, 19, 809–819. [Google Scholar] [CrossRef]
- Elitt, M.S.; Barbar, L.; Tesar, P.J. Drug Screening for Human Genetic Diseases Using IPSC Models. Hum Mol Genet 2018, 27. [Google Scholar] [CrossRef]
- Rowe, R.G.; Daley, G.Q. Induced Pluripotent Stem Cells in Disease Modelling and Drug Discovery. Nat Rev Genet 2019, 20, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Gao, J.; Liang, Z.; Gao, C.; Niu, Q.; Wu, F.; Zhang, L. Mesenchymal Stem Cells and Their Microenvironment. Stem Cell Res Ther 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Jiang, J.; Gu, Z.; Zhang, J.; Chen, Y.; Liu, X. Mesenchymal Stromal Cell Therapies: Immunomodulatory Properties and Clinical Progress. Stem Cell Res Ther 2020, 11. [Google Scholar] [CrossRef]
- Lee, A.S.; Tang, C.; Rao, M.S.; Weissman, I.L.; Wu, J.C. Tumorigenicity as a Clinical Hurdle for Pluripotent Stem Cell Therapies. Nat Med 2013, 19. [Google Scholar] [CrossRef]
- Sato, Y.; Bando, H.; Di Piazza, M.; Gowing, G.; Herberts, C.; Jackman, S.; Leoni, G.; Libertini, S.; MacLachlan, T.; McBlane, J.W.; et al. Tumorigenicity Assessment of Cell Therapy Products: The Need for Global Consensus and Points to Consider. Cytotherapy 2019, 21, 1095–1111. [Google Scholar] [CrossRef]
- Keyvani-Ghamsari, S.; Khorsandi, K.; Rasul, A.; Zaman, M.K. Current Understanding of Epigenetics Mechanism as a Novel Target in Reducing Cancer Stem Cells Resistance. Clin Epigenetics 2021, 13, 120. [Google Scholar] [CrossRef]
- Günes, C.; Rudolph, K.L. The Role of Telomeres in Stem Cells and Cancer. Cell 2013, 152. [Google Scholar] [CrossRef] [PubMed]
- Lateef, H.B.; Suresh, P.M.; Bharathi, P.; Pathak, S.; Banerjee, A. A Brief Overview of Telomeres and Telomerase in Aging and Cancer. Curr Appl Sci Technol 2023, 23. [Google Scholar]
- Lansdorp, P.M. Telomeres, Aging, and Cancer: The Big Picture. Blood 2022, 139. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Chen, X.; Zhang, S.; Fang, J.; Chen, M.; Xu, Y.; Chen, X. Mesenchymal Stem Cells as a Double-Edged Sword in Tumor Growth: Focusing on MSC-Derived Cytokines. Cell Mol Biol Lett 2021, 26. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic Stem Cell Lines Derived from Human Blastocysts. Science (1979) 1998, 282, 1145–1147. [Google Scholar] [CrossRef] [PubMed]
- Golchin, A.; Chatziparasidou, A.; Ranjbarvan, P.; Niknam, Z.; Ardeshirylajimi, A. Embryonic Stem Cells in Clinical Trials: Current Overview of Developments and Challenges. In Advances in Experimental Medicine and Biology; Springer, 2021; Vol. 1312, pp. 19–37.
- Miura, K.; Okada, Y.; Aoi, T.; Okada, A.; Takahashi, K.; Okita, K.; Nakagawa, M.; Koyanagi, M.; Tanabe, K.; Ohnuki, M.; et al. Variation in the Safety of Induced Pluripotent Stem Cell Lines. Nat Biotechnol 2009, 27. [Google Scholar] [CrossRef]
- Okita, K.; Ichisaka, T.; Yamanaka, S. Generation of Germline-Competent Induced Pluripotent Stem Cells. Nature 2007, 448. [Google Scholar] [CrossRef]
- Yamanaka, S. The Winding Road to Pluripotency (Nobel Lecture). Angewandte Chemie International Edition 2013, 52, 13900–13909. [Google Scholar] [CrossRef]
- Tolar, J.; Nauta, A.J.; Osborn, M.J.; Panoskaltsis Mortari, A.; McElmurry, R.T.; Bell, S.; Xia, L.; Zhou, N.; Riddle, M.; Schroeder, T.M.; et al. Sarcoma Derived from Cultured Mesenchymal Stem Cells. Stem Cells 2007, 25. [Google Scholar] [CrossRef]
- Foudah, D.; Redaelli, S.; Donzelli, E.; Bentivegna, A.; Miloso, M.; Dalprà, L.; Tredici, G. Monitoring the Genomic Stability of in Vitro Cultured Rat Bone-Marrow-Derived Mesenchymal Stem Cells. Chromosome Research 2009, 17. [Google Scholar] [CrossRef] [PubMed]
- Caplan, A.I. Mesenchymal Stem Cells: Time to Change the Name! Stem Cells Transl Med 2017, 6, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Neri, S. Genetic Stability of Mesenchymal Stromal Cells for Regenerative Medicine Applications: A Fundamental Biosafety Aspect. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Amariglio, N.; Hirshberg, A.; Scheithauer, B.W.; Cohen, Y.; Loewenthal, R.; Trakhtenbrot, L.; Paz, N.; Koren-Michowitz, M.; Waldman, D.; Leider-Trejo, L.; et al. Donor-Derived Brain Tumor Following Neural Stem Cell Transplantation in an Ataxia Telangiectasia Patient. PLoS Med 2009, 6. [Google Scholar] [CrossRef] [PubMed]
- Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products: Minimal Manipulation and Homologous Use | FDA Available online:. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/regulatory-considerations-human-cells-tissues-and-cellular-and-tissue-based-products-minimal (accessed on 2 July 2024).
- Stem Cell-Based Medicinal Products - Scientific Guideline | European Medicines Agency Available online:. Available online: https://www.ema.europa.eu/en/stem-cell-based-medicinal-products-scientific-guideline (accessed on 2 July 2024).
- Hyun, I. The Bioethics of Stem Cell Research and Therapy. Journal of Clinical Investigation 2010, 120. [Google Scholar] [CrossRef]
- Daley, G.Q.; Hyun, I.; Apperley, J.F.; Barker, R.A.; Benvenisty, N.; Bredenoord, A.L.; Breuer, C.K.; Caulfield, T.; Cedars, M.I.; Frey-Vasconcells, J.; et al. Setting Global Standards for Stem Cell Research and Clinical Translation: The 2016 ISSCR Guidelines. Stem Cell Reports 2016, 6. [Google Scholar] [CrossRef]
- Turner, L. ISSCR’s Guidelines for Stem Cell Research and Clinical Translation: Supporting Development of Safe and Efficacious Stem Cell-Based Interventions. Stem Cell Reports 2021, 16. [Google Scholar] [CrossRef]
- Zhang, J.; Suo, M.; Wang, J.; Liu, X.; Huang, H.; Wang, K.; Liu, X.; Sun, T.; Li, Z.; Liu, J. Standardisation Is the Key to the Sustained, Rapid and Healthy Development of Stem Cell-based Therapy. Clin Transl Med 2024, 14. [Google Scholar] [CrossRef]
- Nowak, M.A.; Komarova, N.L.; Sengupta, A.; Jallepalli, P.V.; Shih, I.-M.; Vogelstein, B.; Lengauer, C. The Role of Chromosomal Instability in Tumor Initiation; 2002. [Google Scholar]
- Carloni, V.; Morganti, E.; Galli, A.; Mazzocca, A. The Adaptability of Chromosomal Instability in Cancer Therapy and Resistance. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Giam, M.; Rancati, G. Aneuploidy and Chromosomal Instability in Cancer: A Jackpot to Chaos. Cell Div 2015, 10. [Google Scholar] [CrossRef]
- Nguyen, H.T.; Geens, M.; Spits, C. Genetic and Epigenetic Instability in Human Pluripotent Stem Cells. Hum Reprod Update 2013, 19, 187–205. [Google Scholar] [CrossRef] [PubMed]
- Raptis, S.; Bapat, B. Genetic Instability in Human Tumors. EXS 2006.
- Michor, F. Chromosomal Instability and Human Cancer. In Proceedings of the Philosophical Transactions of the Royal Society B: Biological Sciences; Royal Society, March 29 2005; Vol. 360; pp. 631–635. [Google Scholar]
- Reynolds, T.Y.; Rockwell, S.; Glazer, P.M. Genetic Instability Induced by the Tumor Microenvironment. Cancer Res 1996, 56. [Google Scholar] [CrossRef]
- Gronroos, E.; Lopez-García, C. Tolerance of Chromosomal Instability in Cancer: Mechanisms and Therapeutic Opportunities. Cancer Res 2018, 78. [Google Scholar] [CrossRef]
- He, Q.; Au, B.; Kulkarni, M.; Shen, Y.; Lim, K.J.; Maimaiti, J.; Wong, C.K.; Luijten, M.N.H.; Chong, H.C.; Lim, E.H.; et al. Chromosomal Instability-Induced Senescence Potentiates Cell Non-Autonomous Tumourigenic Effects. Oncogenesis 2018, 7. [Google Scholar] [CrossRef]
- Nam, H.; Lee, I.; Sa, J.K.; Kim, S.S.; Pyeon, H.; Lee, K.H.; Lee, K.; Lee, S.; Joo, K.M. Correction to: Effects of Long-Term In Vitro Expansion on Genetic Stability and Tumor Formation Capacity of Stem Cells. Stem Cell Rev Rep 2022, 18. [Google Scholar] [CrossRef]
- Ferguson, L.P.; Diaz, E.; Reya, T. The Role of the Microenvironment and Immune System in Regulating Stem Cell Fate in Cancer. Trends Cancer 2021, 7. [Google Scholar] [CrossRef]
- Seet, W.T.; Afandi, M.A.M.; Shamsuddin, S.A.; Lokanathan, Y.; Ng, M.H.; Maarof, M. Current Good Manufacturing Practice (CGMP) Facility and Production of Stem Cell. In Stem Cell Production: Processes, Practices and Regulations; 2022.
- Aghayan, H.R.; Payab, M.; Mohamadi-Jahani, F.; Aghayan, S.S.; Larijani, B.; Arjmand, B. GMP-Compliant Production of Human Placenta-Derived Mesenchymal Stem Cells. In Methods in Molecular Biology; 2021; Vol. 2286.
- Ozkan, E.; Lacerda, M.P. Genetics, Cytogenetic Testing And Conventional Karyotype; 2021. [Google Scholar]
- Jackson-Holmes, E.L.; McDevitt, T.C.; Lu, H. A Microfluidic Trap Array for Longitudinal Monitoring and Multi-Modal Phenotypic Analysis of Individual Stem Cell Aggregates. Lab Chip 2017, 17. [Google Scholar] [CrossRef]
- Vitillo, L.; Durance, C.; Hewitt, Z.; Moore, H.; Smith, A.; Vallier, L. GMP-Grade Neural Progenitor Derivation and Differentiation from Clinical-Grade Human Embryonic Stem Cells. Stem Cell Res Ther 2020, 11. [Google Scholar] [CrossRef]
- Miura, T.; Yasuda, S.; Sato, Y. A Simple Method to Estimate the In-House Limit of Detection for Genetic Mutations with Low Allele Frequencies in Whole-Exome Sequencing Analysis by next-Generation Sequencing. BMC Genom Data 2021, 22. [Google Scholar] [CrossRef]
- Mulligan, S.P. Karyotype and Outcome in CLL. Blood 2023, 142. [Google Scholar] [CrossRef]
- Laing, O.; Halliwell, J.; Barbaric, I. Rapid PCR Assay for Detecting Common Genetic Variants Arising in Human Pluripotent Stem Cell Cultures. Curr Protoc Stem Cell Biol 2019, 49. [Google Scholar] [CrossRef] [PubMed]
- Bielski, C.M.; Taylor, B.S. Homing in on Genomic Instability as a Therapeutic Target in Cancer. Nat Commun 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.H.K.; Ogando, C.R.; Wang See, C.; Chang, T.Y.; Barabino, G.A. Changes in Phenotype and Differentiation Potential of Human Mesenchymal Stem Cells Aging in Vitro. Stem Cell Res Ther 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Chen, H.; Huang, D.; Chen, H.; Fei, L.; Cheng, C.; Huang, H.; Yuan, G.C.; Guo, G. Mapping Human Pluripotent Stem Cell Differentiation Pathways Using High Throughput Single-Cell RNA-Sequencing. Genome Biol 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Naujok, O.; Francini, F.; Picton, S.; Jörns, A.; Bailey, C.J.; Lenzen, S. A New Experimental Protocol for Preferential Differentiation of Mouse Embryonic Stem Cells into Insulin-Producing Cells. Cell Transplant 2008, 17. [Google Scholar] [CrossRef]
- Mitsui, K.; Ide, K.; Takahashi, T.; Kosai, K. ichiro Viral Vector-Based Innovative Approaches to Directly Abolishing Tumorigenic Pluripotent Stem Cells for Safer Regenerative Medicine. Mol Ther Methods Clin Dev 2017, 5. [Google Scholar]
- Afjeh-Dana, E.; Naserzadeh, P.; Moradi, E.; Hosseini, N.; Seifalian, A.M.; Ashtari, B. Stem Cell Differentiation into Cardiomyocytes: Current Methods and Emerging Approaches. Stem Cell Rev Rep 2022, 18. [Google Scholar]
- Naujok, O.; Diekmann, U.; Lenzen, S. The Generation of Definitive Endoderm from Human Embryonic Stem Cells Is Initially Independent from Activin A but Requires Canonical Wnt-Signaling. Stem Cell Rev Rep 2014, 10. [Google Scholar] [CrossRef]
- Fong, C.Y.; Peh, G.S.L.; Gauthaman, K.; Bongso, A. Separation of SSEA-4 and TRA-1-60 Labelled Undifferentiated Human Embryonic Stem Cells from a Heterogeneous Cell Population Using Magnetic-Activated Cell Sorting (MACS) and Fluorescence-Activated Cell Sorting (FACS). Stem Cell Rev Rep 2009, 5. [Google Scholar] [CrossRef]
- Li, Y.; Shen, Z.; Chai, Z.; Zhan, Y.; Zhang, Y.; Liu, Z.; Liu, Y.; Li, Z.; Lin, M.; Zhang, Z.; et al. Targeting MS4A4A on Tumour-Associated Macrophages Restores CD8+ T-Cell-Mediated Antitumour Immunity. Gut 2023, 72. [Google Scholar] [CrossRef] [PubMed]
- Buccarelli, M.; Beninati, S.; Tabolacci, C. Editorial: Cancer Stem Cell Differentiation: A Realistic Potential Therapeutic Option? Front Oncol 2023, 13. [Google Scholar] [CrossRef]
- Sutermaster, B.A.; Darling, E.M. Considerations for High-Yield, High-Throughput Cell Enrichment: Fluorescence versus Magnetic Sorting. Sci Rep 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Ye, G.; Deng, H.; Woodworth, C.D.; Kaya, M. An Unsupervised Machine Learning Algorithm to Detect Undifferentiated Cell Clusters of Immortalized Human Cervical Epithelial Cell. In Proceedings of the BioSMART 2021 - Proceedings: 4th International Conference on Bio-Engineering for Smart Technologies; 2021. [Google Scholar]
- Marei, H.E.; Althani, A.; Afifi, N.; Hasan, A.; Caceci, T.; Pozzoli, G.; Morrione, A.; Giordano, A.; Cenciarelli, C. P53 Signaling in Cancer Progression and Therapy. Cancer Cell Int 2021, 21. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wood, B.L. Minimal Residual Disease in Acute Lymphoblastic Leukemia: Techniques and Application. In Clinical Management of Acute Lymphoblastic Leukemia: From Bench to Bedside; 2022.
- Yang, X.; Liu, M.; Li, M.; Zhang, S.; Hiju, H.; Sun, J.; Mao, Z.; Zheng, M.; Feng, B. Epigenetic Modulations of Noncoding RNA: A Novel Dimension of Cancer Biology. Mol Cancer 2020, 19. [Google Scholar] [CrossRef]
- Wang, Q.; Xiong, F.; Wu, G.; Liu, W.; Chen, J.; Wang, B.; Chen, Y. Gene Body Methylation in Cancer: Molecular Mechanisms and Clinical Applications. Clin Epigenetics 2022, 14. [Google Scholar] [CrossRef]
- De Cario, R.; Kura, A.; Suraci, S.; Magi, A.; Volta, A.; Marcucci, R.; Gori, A.M.; Pepe, G.; Giusti, B.; Sticchi, E. Sanger Validation of High-Throughput Sequencing in Genetic Diagnosis: Still the Best Practice? Front Genet 2020, 11. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, K.; Wang, W.L.; Yin, T.T.; Dong, W.Q.; Xu, C.J. A High-Throughput SNP Discovery Strategy for RNA-Seq Data. BMC Genomics 2019, 20. [Google Scholar] [CrossRef]
- Hong, M.; Tao, S.; Zhang, L.; Diao, L.T.; Huang, X.; Huang, S.; Xie, S.J.; Xiao, Z.D.; Zhang, H. RNA Sequencing: New Technologies and Applications in Cancer Research. J Hematol Oncol 2020, 13. [Google Scholar] [CrossRef]
- Roychowdhury, S.; Iyer, M.K.; Robinson, D.R.; Lonigro, R.J.; Wu, Y.M.; Cao, X.; Kalyana-Sundaram, S.; Sam, L.; Balbin, O.A.; Quist, M.J.; et al. Personalized Oncology through Integrative High-Throughput Sequencing: A Pilot Study. Sci Transl Med 2011, 3. [Google Scholar] [CrossRef]
- Khoshandam, M.; Soltaninejad, H.; Mousazadeh, M.; Hamidieh, A.A.; Hosseinkhani, S. Clinical Applications of the CRISPR/Cas9 Genome-Editing System: Delivery Options and Challenges in Precision Medicine. Genes Dis 2024, 11. [Google Scholar] [CrossRef]
- Li, T.; Li, S.; Kang, Y.; Zhou, J.; Yi, M. Harnessing the Evolving CRISPR/Cas9 for Precision Oncology. J Transl Med 2024, 22, 749. [Google Scholar] [CrossRef] [PubMed]
- Chehelgerdi, M.; Chehelgerdi, M.; Khorramian-Ghahfarokhi, M.; Shafieizadeh, M.; Mahmoudi, E.; Eskandari, F.; Rashidi, M.; Arshi, A.; Mokhtari-Farsani, A. Comprehensive Review of CRISPR-Based Gene Editing: Mechanisms, Challenges, and Applications in Cancer Therapy. Mol Cancer 2024, 23. [Google Scholar]
- Bak, R.O.; Dever, D.P.; Porteus, M.H. CRISPR/Cas9 Genome Editing in Human Hematopoietic Stem Cells. Nat Protoc 2018, 13. [Google Scholar] [CrossRef]
- Ugalde, L.; Fañanas, S.; Torres, R.; Quintana-Bustamante, O.; Río, P. CRISPR/Cas9-Mediated Gene Editing. A Promising Strategy in Hematological Disorders. Cytotherapy 2023, 25. [Google Scholar] [CrossRef] [PubMed]
- Chemello, F.; Bassel-Duby, R.; Olson, E.N. Correction of Muscular Dystrophies by CRISPR Gene Editing. Journal of Clinical Investigation 2020, 130. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Shi, H.; Gou, S.; Wang, X.; Li, L.; Jin, Q.; Wu, H.; Zhang, H.; Li, Y.; Wang, L.; et al. In Vivo Genome Editing in Mouse Restores Dystrophin Expression in Duchenne Muscular Dystrophy Patient Muscle Fibers. Genome Med 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- DeWitt, M.A.; Magis, W.; Bray, N.L.; Wang, T.; Berman, J.R.; Urbinati, F.; Muñoz, D.P.; Kohn, D.B.; Walters, M.C.; Carroll, D.; et al. Efficient Correction of the Sickle Mutation in Human Hematopoietic Stem Cells Using a Cas9 Ribonucleoprotein Complex. bioRxiv 2016. [Google Scholar]
- Rheney, J. Using Crisper in the Treatment of Sickle Cell Disease. Journal of Student Research 2023, 12. [Google Scholar] [CrossRef]
- Kits, E. CRISPR-Cas9, TALENs and ZFNs - the Battle in Gene Editing. ProteinTech 2018. [Google Scholar]
- Castro, N.G.; Bjelic, J.; Malhotra, G.; Huang, C.; Alsaffar, S.H. Comparison of the Feasibility, Efficiency, and Safety of Genome Editing Technologies. Int J Mol Sci 2021, 22. [Google Scholar]
- Zentelytė, A.; Žukauskaitė, D.; Jacerytė, I.; Borutinskaitė, V.V.; Navakauskienė, R. Small Molecule Treatments Improve Differentiation Potential of Human Amniotic Fluid Stem Cells. Front Bioeng Biotechnol 2021, 9. [Google Scholar] [CrossRef]
- Schugar, R.C.; Robbins, P.D.; Deasy, B.M. Small Molecules in Stem Cell Self-Renewal and Differentiation. Gene Ther 2008, 15. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, W.; Liu, S.; Chen, C. Targeting Breast Cancer Stem Cells. Int J Biol Sci 2023, 19. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Bhonde, R. Genetic and Epigenetic Stability of Stem Cells: Epigenetic Modifiers Modulate the Fate of Mesenchymal Stem Cells. Genomics 2020, 112. [Google Scholar] [CrossRef]
- Tran, K.A.; Jackson, S.A.; Olufs, Z.P.G.; Zaidan, N.Z.; Leng, N.; Kendziorski, C.; Roy, S.; Sridharan, R. Collaborative Rewiring of the Pluripotency Network by Chromatin and Signalling Modulating Pathways. Nat Commun 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Huangfu, D.; Osafune, K.; Maehr, R.; Guo, W.; Eijkelenboom, A.; Chen, S.; Muhlestein, W.; Melton, D.A. Induction of Pluripotent Stem Cells from Primary Human Fibroblasts with Only Oct4 and Sox2. Nat Biotechnol 2008, 26. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Chen, X.; Yu, D.; Li, T.; Cui, J.; Wang, G.; Hu, J.F.; Li, W. Histone Deacetylase Inhibitor Valproic Acid Promotes the Induction of Pluripotency in Mouse Fibroblasts by Suppressing Reprogramming-Induced Senescence Stress. Exp Cell Res 2015, 337. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tristan, C.A.; Chen, L.; Jovanovic, V.M.; Malley, C.; Chu, P.H.; Ryu, S.; Deng, T.; Ormanoglu, P.; Tao, D.; et al. A Versatile Polypharmacology Platform Promotes Cytoprotection and Viability of Human Pluripotent and Differentiated Cells. Nat Methods 2021, 18. [Google Scholar] [CrossRef]
- Geng, Y.; Amante, J.J.; Goel, H.L.; Zhang, X.; Walker, M.R.; Luther, D.C.; Mercurio, A.M.; Rotello, V.M. Differentiation of Cancer Stem Cells through Nanoparticle Surface Engineering. ACS Nano 2020, 14. [Google Scholar] [CrossRef]
- Marquardt, J.U.; Raggi, C.; Andersen, J.B.; Seo, D.; Avital, I.; Holczbauer, A.; Factor, V.M.; Thorgeirsson, S.S. Abstract 4252: Mechanistic and Clinical Implications of Epigenetic Modulation in Liver Cancer Stem Cells. Cancer Res 2010, 70. [Google Scholar] [CrossRef]
- KalantarMotamedi, Y.; Peymani, M.; Baharvand, H.; Nasr-Esfahani, M.H.; Bender, A. Systematic Selection of Small Molecules to Promote Differentiation of Embryonic Stem Cells and Experimental Validation for Generating Cardiomyocytes. Cell Death Discov 2016, 2. [Google Scholar] [CrossRef] [PubMed]
- Kevin, W.-H.L.; Jenny, C.-Y.H.; Kong, C.-W.; Lee, Y.-K.; Ng, K.-M.; Au, K.-W.; Chan, Y.-C.; Lau, C.-P.; Tse, H.-F.; Siu, C.-W. P135. Transient Rho Kinase Inhibition Enhance the Generation of Human Induced Pluripotent Stem Cells in Feeder-Independent, Serum-Free Culture System. Differentiation 2010, 80. [Google Scholar] [CrossRef]
| Type of Stem Cell | Definition | Differentiation Capabilities | Sources | Applications |
| Mesenchymal Stem Cells (MSCs) | Multipotent stromal cells that can differentiate into various cell types. | Osteoblasts (bone cells), chondrocytes (cartilage cells), myocytes (muscle cells), adipocytes (fat cells). | Bone marrow, peripheral blood, adipose tissue, placenta, umbilical cord, cord blood. | Bone regeneration, cartilage repair, muscle and adipose tissue regeneration, treatment of osteoarthritis, bone fractures, autoimmune diseases. |
| Embryonic Stem Cells (ESCs) | Derived from the inner cell mass of a blastocyst, capable of developing into any of the 200+ human cell types. | Any human cell type. | Inner cell mass of a blastocyst (early stage pre-implantation embryo). | Research tool for understanding human development and diseases, inspiring further exploration and discovery. |
| Induced Pluripotent Stem Cells (iPSCs) | Generated directly from somatic cells, closely resembling ESCs in their ability to differentiate into any cell type. | Any cell type in the body. | Adult tissues (reprogrammed somatic cells). | Personalized medicine, disease modeling, drug screening, transplantation therapies, creation of patient-specific stem cell lines. |
| Therapeutic Application | Stem Cell Type | Clinical Potential | Challenges and Limitations | References |
| Cardiac Repair | MSCs, iPSCs | Restoration of cardiac muscle function, targeting damaged proteins to reverse changes caused by heart attack. | Survival, integration, and maturation of transplanted cells; potential immune rejection; ethical considerations. | [1,2,3] |
| Neurodegenerative Diseases | iPSCs | Differentiation into dopaminergic neurons for treating Parkinson’s disease; potential treatment for Alzheimer’s disease. | Survival, integration, and maturation of transplanted cells; potential immune rejection; ethical considerations. | [4,5,6] |
| Immunomodulation | MSCs, HSCs | Migration to sites of inflammation and injury; modulation of pathogenic immune responses; treatment of GVHD. | Interaction with immune cells; secretion of anti-inflammatory cytokines; reducing GVHD severity. | [7,8,9,10,11,12,13] |
| Drug Screening and Disease Modeling | iPSCs | Generation of unlimited disease-relevant cell types; identification of novel molecular targets; large-scale phenotypic screens; modeling diseases in vitro. | Ensuring cell survival and integration; ethical considerations; potential immune rejection. | [14,15,16] |
| Strategy | Description | Advantages | Challenges | References |
| Quality Control Measures | ||||
| Stringent Culture and Expansion Protocols | Adhering to GMP standards to ensure safety, efficacy, and consistency in stem cell production. Continuous genetic and phenotypic monitoring to detect abnormalities. | Ensures low immunogenicity and high safety standards. Reduces heterogeneity in stem cell cultures. | Time-consuming and costly. Requires stringent environmental controls and quality checks. | [53]; [54]; [60]; [56]; |
| Monitoring Genetic Stability | Using karyotyping, NGS, and CGH techniques to detect and eliminate cells with mutations. Regular screening to enable early detection of mutations. | Increases genetic stability, reducing the risk of tumor formation. | Requires frequent monitoring and advanced techniques. | [58,59,60,62] |
| Refinement of Differentiation Protocols | ||||
| Improved Differentiation Techniques | Using specific culture conditions, growth factors, and co-culturing to promote desired differentiation. | Reduces the number of undifferentiated cells, minimizing tumorigenic risks. | Requires extensive research and validation. | [63,64,65,66,67] |
| Minimizing Residual Undifferentiated Cells | Using flow cytometry and MACS to ensure the purity of differentiated cells by removing undifferentiated cells. | Enhances safety and efficacy of stem cell therapies. | Labor-intensive and costly. | [69,70,72] |
| Genomic and Epigenomic Screening Techniques | ||||
| Advanced Screening Methodologies | High-throughput sequencing (HTS) is essential for detecting genetic and epigenetic aberrations in stem cells. HTS enables comprehensive genome-wide analysis and identification of mutations, copy number variations, and other genetic changes. Epigenomic profiling examines DNA methylation, histone modifications, and chromatin accessibility. | Enables sensitive monitoring of minimal residual disease. Identifies gene mutations such as TP53, crucial for genomic stability. | High cost and technical complexity. | [74,75,76,77,78] |
| Detection and Elimination of Tumorigenic Cells | Using targeted sequencing and high-throughput single-cell sequencing to detect and eliminate tumorigenic cells. | Precise identification and removal of potentially dangerous cells. | Requires advanced techniques and frequent monitoring. | [78,79,80] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




