Submitted:
02 September 2024
Posted:
02 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
2.1. Effect of Diverse Agricultural Materials on the Growth of Rice
2.2. Effect of Selected Extracts by Different Extraction and Planting Methods on Rice Growth
2.3. Effect of Persistence of Four Selected Extracts on Rice Growth
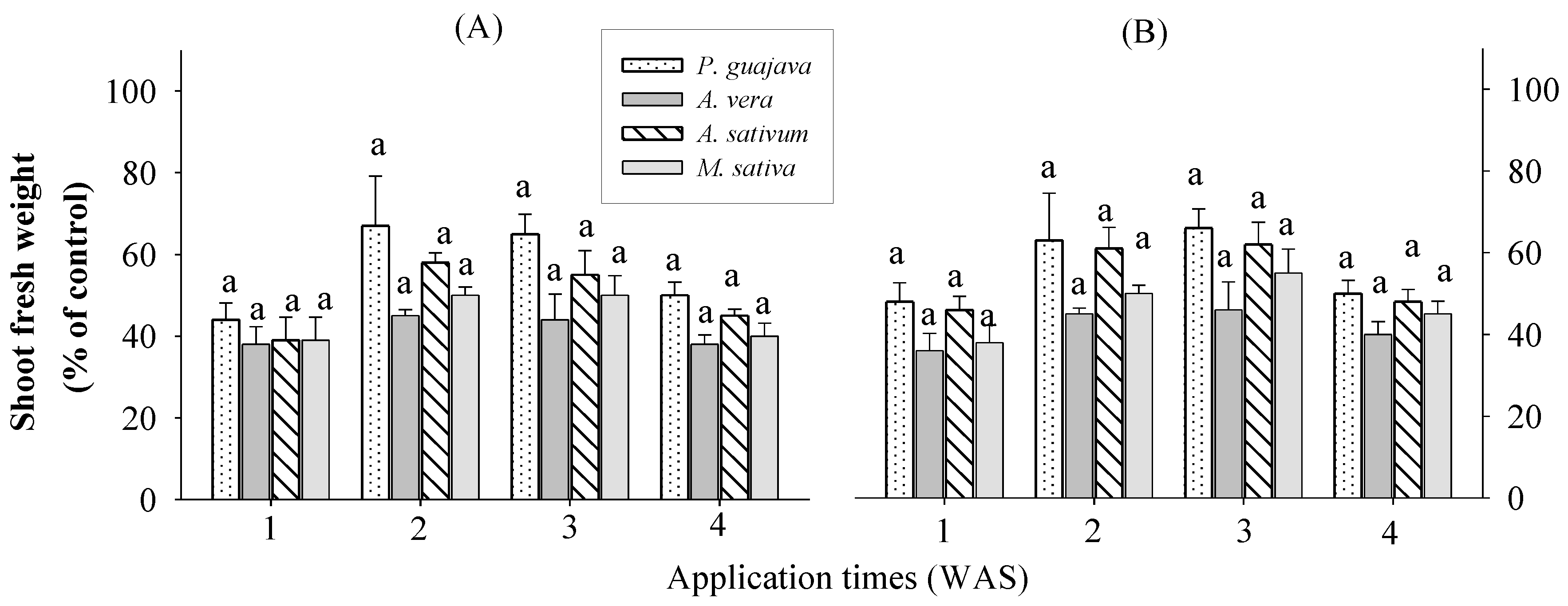
2.4. Effect of Selected Water Extracts on Application Times in Various Growth Stages of Rice
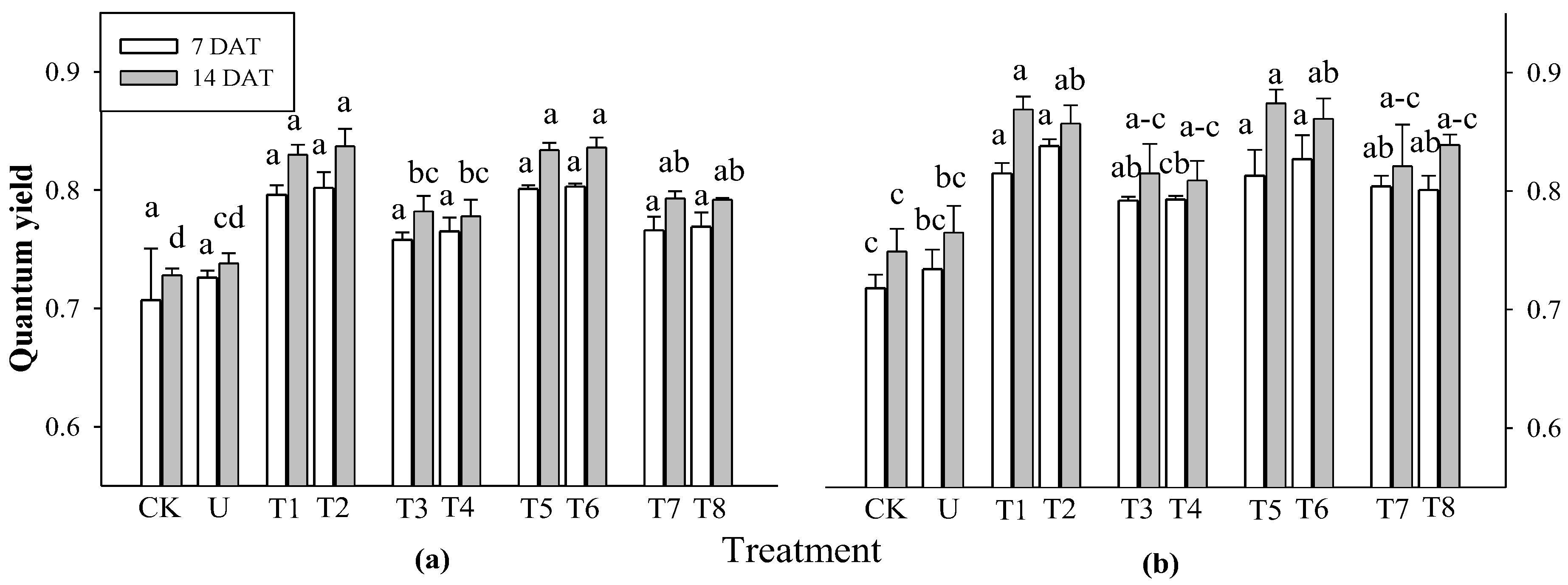
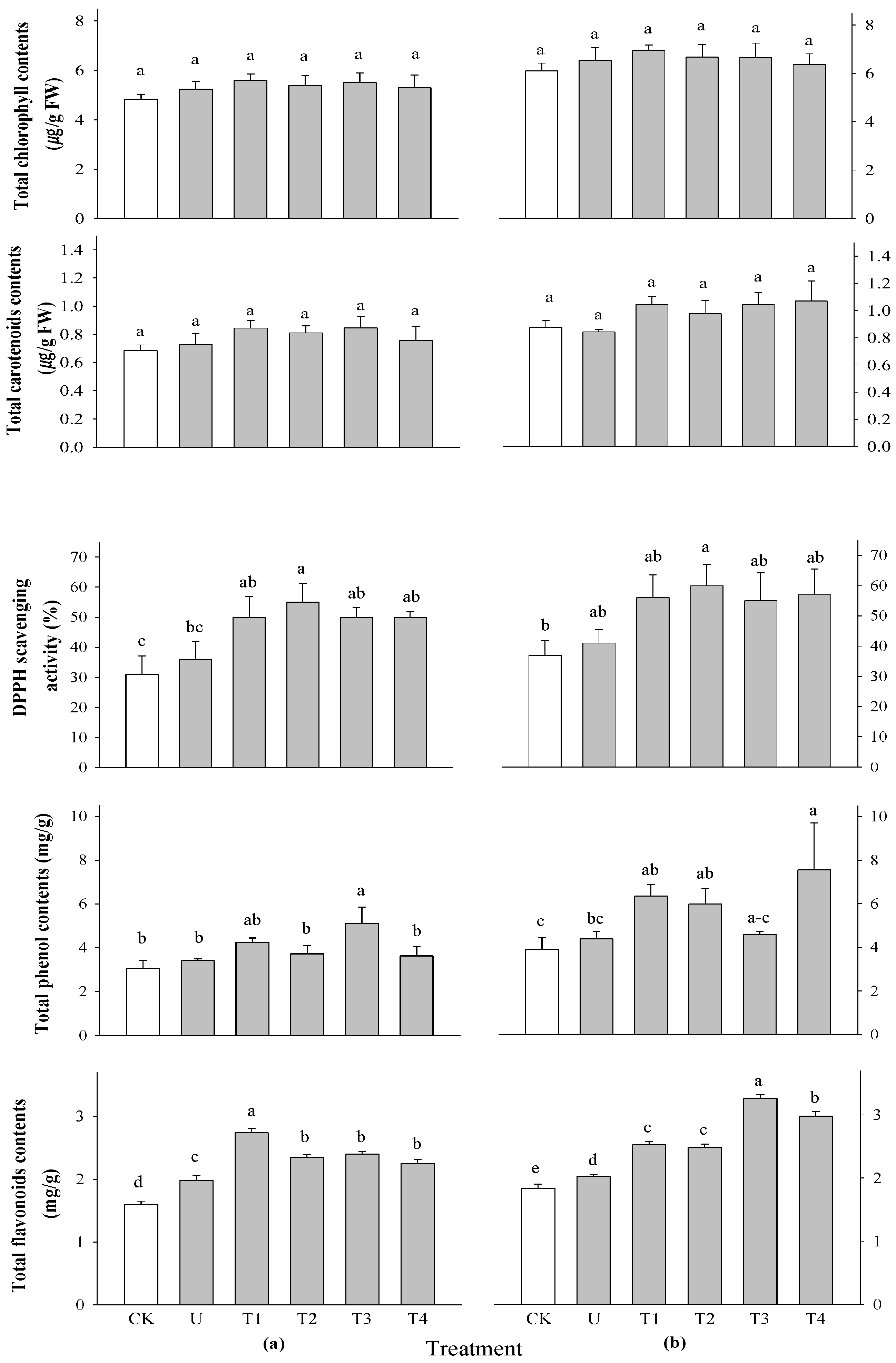
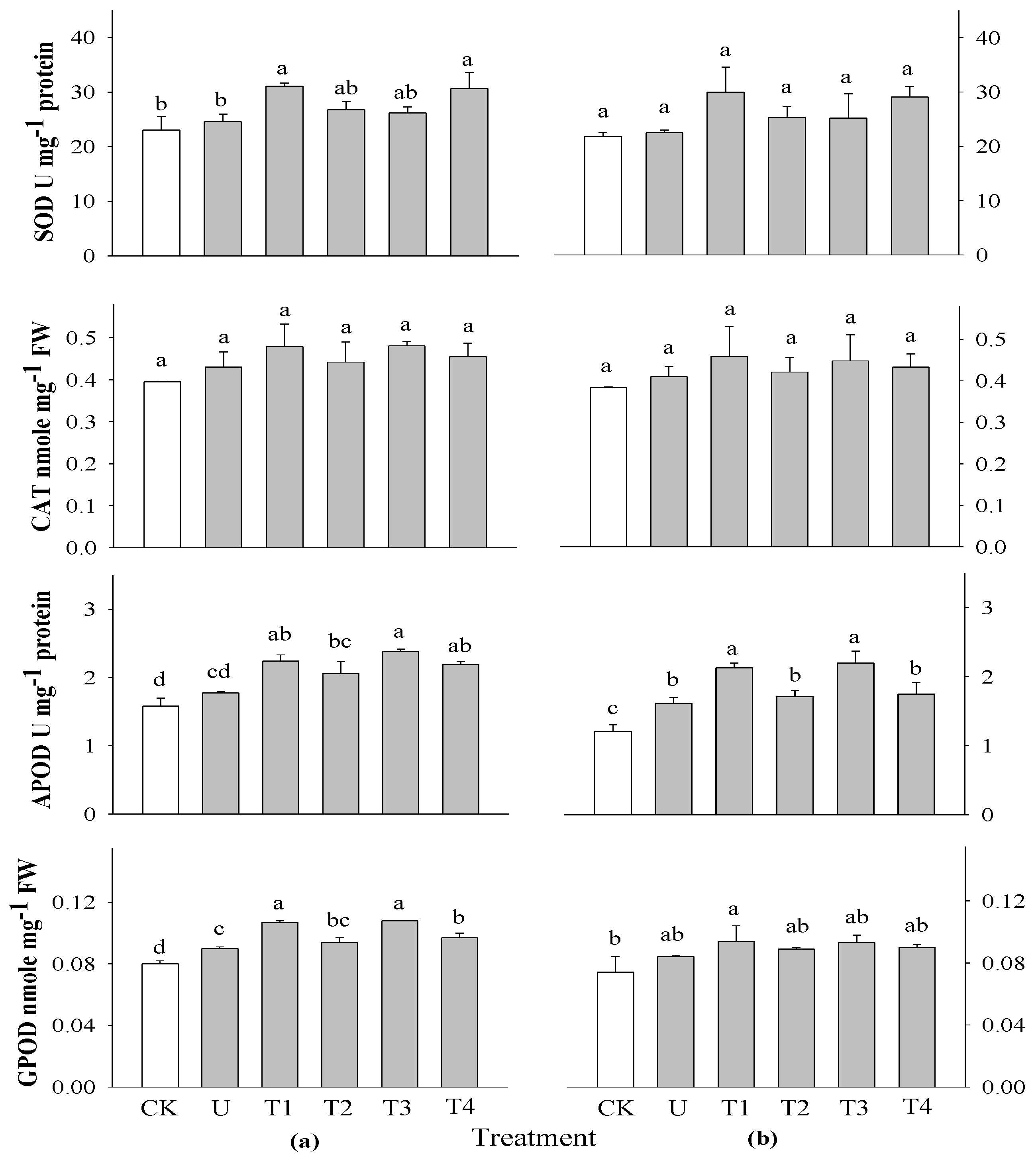
2.5. Effect of Selected Water Extracts on Secondary Metabolites in Rice at Different Application Times
3. Materials and Methods
3.1. Preparation of Plant Materials and Extraction Methods
3.2. Effect of Extracts from Diverse Agricultural Materials on the Growth of Rice
3.3. Effect of Selected Extracts by Different Extraction and Planting Methods on Rice Growth
3.4. Effect of Persistence of Four Selected Extracts on Rice Growth
3.5. Effect of Selected Water Extracts on Application Times in Rice Crop
3.6. Effect of Selected Water Extracts on Secondary Metabolites in Rice at Different Application Times
3.6.1. Determination of Quantum Yield, Total Chlorophyll and Carotenoid Contents
3.6.2. Determination of DPPH Radical Scavenging Activity, Total Phenol, and Flavonoid Contents
3.6.3. Determination of Antioxidant Activities
3.7. Experimental Design and Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- IRRI (International Rice Research Institute). Annual Report 2010; Los Baños, Philippines, International Rice Research Institute, 2011; pp. 1–44. [Google Scholar]
- World Agricultural Production. Global Market Analysis. Foreign agricultural service, USDA, Circular Series WAP; 2023; pp. 8–23. [Google Scholar]
- Godlewska, K.; Ronga, D.; Michalak, I. Plant extracts – importance in sustainable agriculture. Italian Journal of Agronomy 2021, 16, 1851. [Google Scholar] [CrossRef]
- De Pascale, S.; Rouphael, Y.; Colla, G. Plant biostimulants: Innovative tool for enhancing plant nutrition in organic farming. European Journal of Horticultural Science 2017, 82, 277–85. [Google Scholar] [CrossRef]
- Dipak Kumar, H.; Aloke, P. Role of biostimulant formulations in crop production: an overview. International Journal of Agricultural Science and Veterinary Medicine 2020, 8, 8–46. [Google Scholar]
- Ertani, A.; Sambo, P.; Nicoletto, C.; Santagata, S.; Schiavon, M.; Nardi, S. The use of organic biostimulants in hot pepper plants to help low-input sustainable agriculture. Chemical and Biological Technologies in Agriculture 2015, 2, 1–10. [Google Scholar] [CrossRef]
- Parađiković, N.; Teklić, T.; Zeljković, S.; Lisjak, M.; Špoljarević, M. Biostimulants research in some horticultural plant species - a review. Food and Energy Security 2018, 8, 1–17. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G. Synergistic biostimulatory action: designing the next generation of plant biostimulants for sustainable agriculture. Frontiers Plant Science 2018, 871, 1–7. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G. Toward a sustainable agriculture through plant biostimulants: from experimental data to practical applications. Agronomy 2020, 10, 1–10. [Google Scholar] [CrossRef]
- RDA National Academy of Agricultural Science. 2016; p. 293.
- Guh, J.O.; Kim, M.H.; Kim, Y.J. “Ongoijisin” for modern application of technologies used in old agriculture books; RDA: Korea, 2011; p. 300. (In Korean) [Google Scholar]
- Jang, S.J.; Kuk, Y.I. Growth promotion effects of plant extracts on various leafy vegetable crops. Horticultural Science and Technology 2019, 37, 322–336. [Google Scholar] [CrossRef]
- Calvo, P.; Nelson, L.; Klopper, J.W. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Puga-Freitas, R.; Blouin, M. A review of the effects of soil organisms on plant hormone signaling pathways. Environmental and Experimental Botany 2015, 114, 104–116. [Google Scholar] [CrossRef]
- Lariguet, P.; Ranocha, P.; Meyer, M.D.; Barbier, O.; Penel, C.; Dunand, C. Identification of a hydrogen peroxide signaling pathway in the control of light-dependent germination in Arabidopsis. An International Journal of Plant Biology 2013, 238, 381–395. [Google Scholar]
- Candan, N.; Tarhan, L. Relationship among chlorophyll carotenoid content, antioxidant enzyme activities, and lipid peroxidation levels by Mg2+ deficiency in the Mentha pulegium leaves. Plant Physiology and Biochemistry 2003, 41, 35–40. [Google Scholar] [CrossRef]
- Han, X.; Cheng, Z.; Meng, H.; Yang, X.; Ahmad, I. Allelopathic effect of decomposed garlic (Allium sativum L.) stalk on lettuce (L. Sativa var. crispa L.). Pakistan Journal of Botany 2013, 45, 225–233. [Google Scholar]
- Leelarungrayub, N.; Rattanapanone, V.; Chanarat, N.; Gebicki, J.M. Quantitative evaluation of the antioxidant properties of garlic and shallot preparations. Nutrition 2016, 22, 266–274. [Google Scholar] [CrossRef]
- Tong, F.; Cheng, Z.H.; Jin, R.; Zhou, L.Y. Allelopathy of methanol dissolved ingredient from garlic plant aqueous extracts. Journal of Northwest A&F University (Natural Science Edition) 2007, 35, 119–124. [Google Scholar]
- Li, S.; Zhihui, C. Allium sativum extract as a biopesticide affecting pepper blight. International Journal of Vegetable Science 2009, 15, 13–23. [Google Scholar] [CrossRef]
- Kunicki, E.; Grabowska, A.; Sekara, A.; Wojciechowska, R. The effect of cultivar type, time of cultivation, and biostimulant treatment on the yield of spinach (Spinacia oleracea L.). Folia Horticulturae Ann. 2010, 22, 9–13. [Google Scholar] [CrossRef]
- Leskovar, D.I.; Othman, Y.A. Direct seeding and transplanting influence root dynamics, morpho-physiology, yield, and heat quality of globe artichoke. Plants 2021, 10, 899. [Google Scholar] [CrossRef]
- Hongyan, L.; Weiqin, W.; Aibin, H.; Lixiao, N. Correlation of leaf and root senescence during ripening in dry seeded and transplanted rice. Rice Science 2018, 25, 279–285. [Google Scholar] [CrossRef]
- Fanadzo, M.; Chiduza, C.; Mnkeni, P. Comparative response of direct seeded and transplanted maize (Zea mays L.) to nitrogen fertilization at Zanyokwe irrigation scheme, Eastern Cape, South Africa. African Journal of Agricultural Research 2009, 4, 689–694. [Google Scholar]
- Leskovar, D.; Cantliffe, D.; Stoffella, P. Root growth and root-shoot interaction in transplants and direct seeded pepper plants. Environmental and Experimental Botany 2019, 30, 349–354. [Google Scholar] [CrossRef]
- Jumaa, R.M.A. The Effect of Some Natural Extracts on the Growth and Chemical Components of Schefflera araboricola Plant. MSc. Thesis, College of Agriculture, Cairo University, 2015. [Google Scholar]
- Nardi, S.; Carletti, P.; Pizzeghello, D.; Muscolo, A. Biological activities of humic substances, in biophysico-chemical processes involving natural nonliving organic matter in environmental systems. In Part I. Fundamentals and impact of mineral-organic biota interactions on the formation, transformation, turnover, and storage of natural nonliving organic matter (NOM); Senesi, N., Xing, B., Huang, P.M., Eds.; John Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Muhammad, A.; Cheng, Z.; Sikandar, H.; Husain, A.; Muhammad, I.G.; Liu, T. Foliar spraying of aqueous garlic bulb extract stimulates growth and antioxidant enzyme activity in eggplant (Solanum melongena L.). Journal of Integrative Agriculture 2019, 18, 1001–1013. [Google Scholar]
- Bhandari, S.R.; Yoon, M.K.; Kwak, J.H. Contents of phytochemical constituents and antioxidant activity of 19 garlic (Allium sativum L.) parental lines and cultivars. Horticulture, Environment, and Biotechnology 2019, 55, 138–147. [Google Scholar] [CrossRef]
- El-Hamied, S.A.A.; El-Amary, E.I. Improving growth and productivity of “pear” trees using some natural plant extracts under North Sinai conditions. IOSR Journal of Agriculture and Veterinary Sciences 2015, 8, 01–09. [Google Scholar]
- Nautiyal, P.; Lal, S.; Singh, C.P. Effect of shoot pruning severity and plant spacing on leaf nutrient status and yield of guava cv. Pant Prabhat. International Journal of Basic and Applied Agricultural Research 2016, 14, 288–294. [Google Scholar]
- Zhou, Y.L.; Zhihui, C.; Wen, M.H.; Chun, G.H. Allelopathy of garlic root aqueous extracts and root exudates. Journal of Northwest A&F University (Natural Science Edition) 2007, 35, 87–92. (In Chinese) [Google Scholar]
- Baliyan, S.; Mukherjee, R.; Priyadarshini, A.; Vibhuti, A.; Gupta, A.; Pandey, R.P.; Chang, C.M. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiose. Journal of Molecules 2022, 27, 1326. [Google Scholar] [CrossRef] [PubMed]
- Akullo, J.O.; Kiage-Mokua, B.N.; Nakimbugwe, D.; Ng’ang’a, J.; Kinyuru, J. Phytochemical profile and antioxidant activity of various solvent extracts of two varieties of ginger and garlic. Heliyon 2023, 9, e18806. [Google Scholar] [CrossRef]
- Aryal, S.; Baniya, M.K.; Danekhu, K.; Kunwar, P.; Gurung, R.; Koirala, N. Total phenolic content, flavonoid content, and antioxidant potential of wild vegetables from Western Nepal. Journal of Plants 2019, 8, 96. [Google Scholar] [CrossRef]
- Wang, H.; Li, X.; Shen, D.; Oiu, Y.; Song, J. Diversity evaluation of morphological traits and allicin content in garlic (Allium sativum L.) from China. Euphytica 2014, 198, 243–254. [Google Scholar] [CrossRef]
- Park, H.H. Increasement of Growth and Secondary Metabolites in Chicory (Cichorium intybus L.) Plants by Extraction and Application Methods of Agricultural By-products. MSc. Dissertation, Sunchon National University, South Korea, 2021; p. 56. (In Korean). [Google Scholar]
- Mahmood, S.K.; Abdulla, S.M.; Hassan, K.I.; Mahmood, A.B. Effect of some processing methods on the physiochemical properties of black mulberry. Euphrates Journal of Agricultural Science 2024, 16, 440–451. [Google Scholar]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutase. 1. Occurrence in higher plants. Plant Physiology 1977, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.R.; Choudhuri, M.A. Hydrogen peroxide metabolism as an index of water stress tolerance in jute. Physiologia Plantarum 1985, 65, 476–489. [Google Scholar] [CrossRef]
- Zhang, J.; Cui, S.; Li, J.; Kirkham, M.B. Protoplasmic factors, antioxidant responses, and chilling resistance in maize. Plant Physiology and Biochemistry 1995, 33, 567–575. [Google Scholar]
- Chance, B.; Maehly, A.C. Assay of catalases and peroxidases. Methods in Enzymology 1995, 2, 764–775. [Google Scholar]
- Fu, J.; Huang, B. Involvement of antioxidants and lipid peroxidation in the adaptation of two cool-season grasses to localized drought stress. Environmental and Experimental Botany 2001, 45, 105–114. [Google Scholar] [CrossRef]
- Chen, G.; Asada, K. Ascorbate peroxidase in tea leaves: occurrence of two isozymes and the differences in their enzymatic and molecular properties. Plant and Cell Physiology 1989, 30, 987–998. [Google Scholar]
| Growth promotion rates (% of control) | Shoot length | Root length | Seedling length (Shoot + Root) |
|---|---|---|---|
| 10-20 |
Mentha arvensis Rheum undulatum Cyperus rountus Perilla frutescens Oryza sativa (hull) Vigna unguiculata |
Vigna unguiculata | |
| 21-30 |
Centella asiatica Allium sativum Medicago sativa Moringa oleifera Vigna radiata |
Mentha arvensis Cyperus rountus Perilla frutescens Oryza sativa (straw) Vigna radiata Vigna unguiculata |
Mentha arvensis Cyperus rountus Centella asiatica Perilla frutescens Oryza sativa (straw) Oryza sativa (hull) Vigna radiata |
| 31-40 |
Allium tuberosum Oryza sativa (straw) Glycine max (leaves) |
Rheum undulatum Centella asiatica Aloe vera Oryza sativa (hull) Moringa oleifera |
Rheum undulatum Aloe vera Medicago sativa Moringa oleifera Allium tuberosum |
| 41-50 |
Psidium guajava Aloe vera Glycine max (stem) |
Medicago sativa Allium tuberosum Glycine max (leaves) Glycine max (stem) |
Psidium guajava Allium sativum Glycine max (stems) Glycine max (leaves) |
| 51-60 | Psidium guajava | ||
| 61-70 | Allium sativum |
| Scientific name | Common name | Family name | Parts used |
|---|---|---|---|
| Allium sativum | Garlic | Amaryllidaceae | Tubers |
| Allium tuberosum | Chinese (garlic) chives or Chinese leeks | Amaryllidaceae | Leaves |
| Centella asiatica | Pennywort (Gotu Kola) | Apiaceae | Leaves |
| Aloe vera | Aloe | Asphodelaceae | Leaves |
| Cyperus rountus | Nutgrass | Cyperaceae | Aboveground plant parts |
| Mentha arvensis | Wild Mint | Lamiaceae | Leaves |
| Perilla frutescens | Chinese basil, wild basil, perilla mint | Lamiaceae | Aboveground plant parts |
| Medicago sativa | Alfalfa | Leguminosae | Aboveground plant parts |
| Vigna radiata | Mung bean | Leguminosae | Leaves |
| Vigna unguiculata | Cowpea | Leguminosae | Leaves |
| Glycine max | Soybean | Leguminosae | Leaves |
| Glycine max | Soybean | Leguminosae | Stems |
| Oryza sativa | Rice straw | Gramineae | Straws |
| Oryza sativa | Rice hull | Gramineae | Hulls |
| Moringa oleifera | Drumstick | Moringaceae | Leaves |
| Psidium guajava | Guava | Myrtaceae | Leaves |
| Rheum undulatum | - | Polygonaceae | Roots |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




