Submitted:
29 August 2024
Posted:
30 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
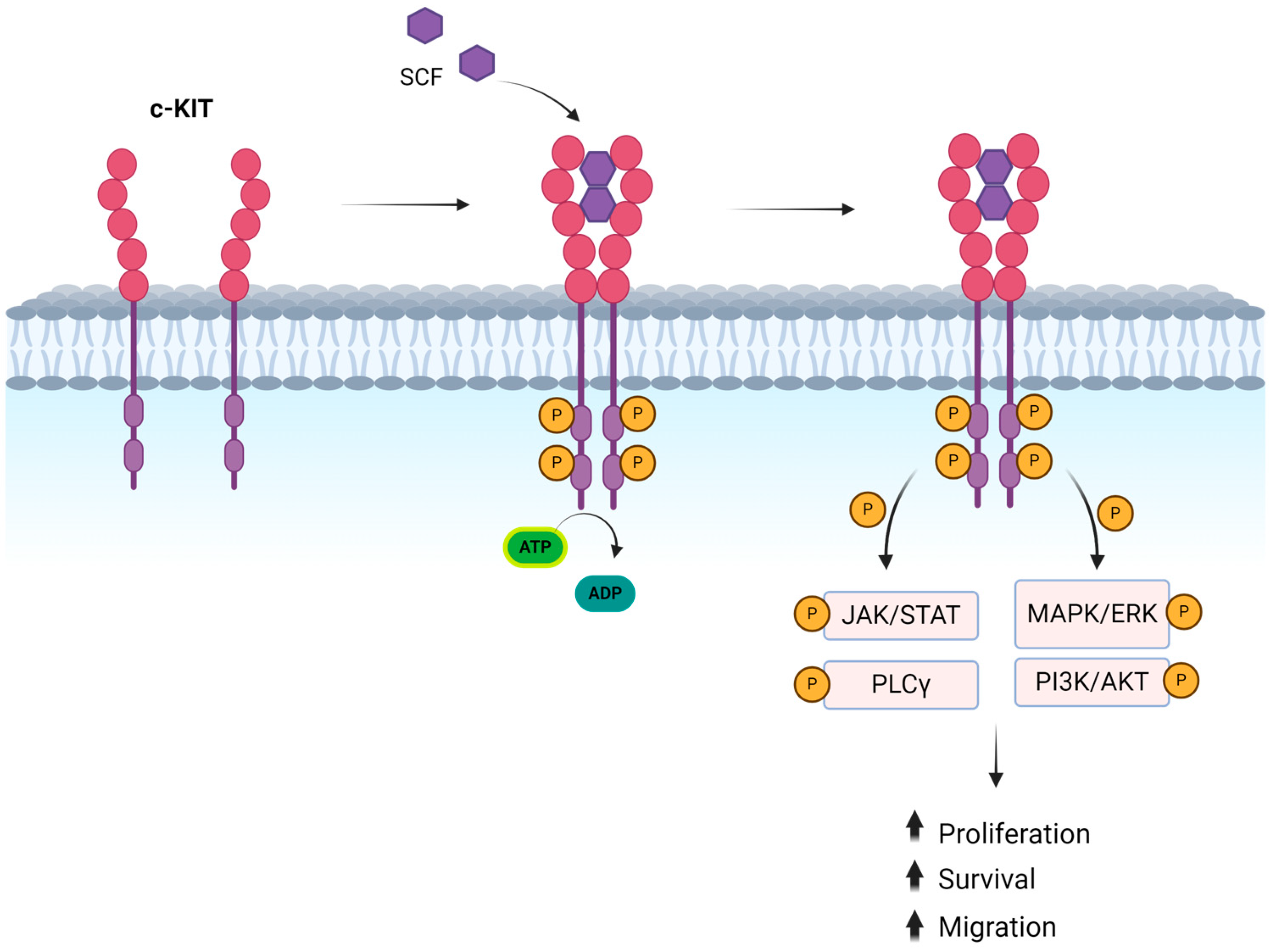
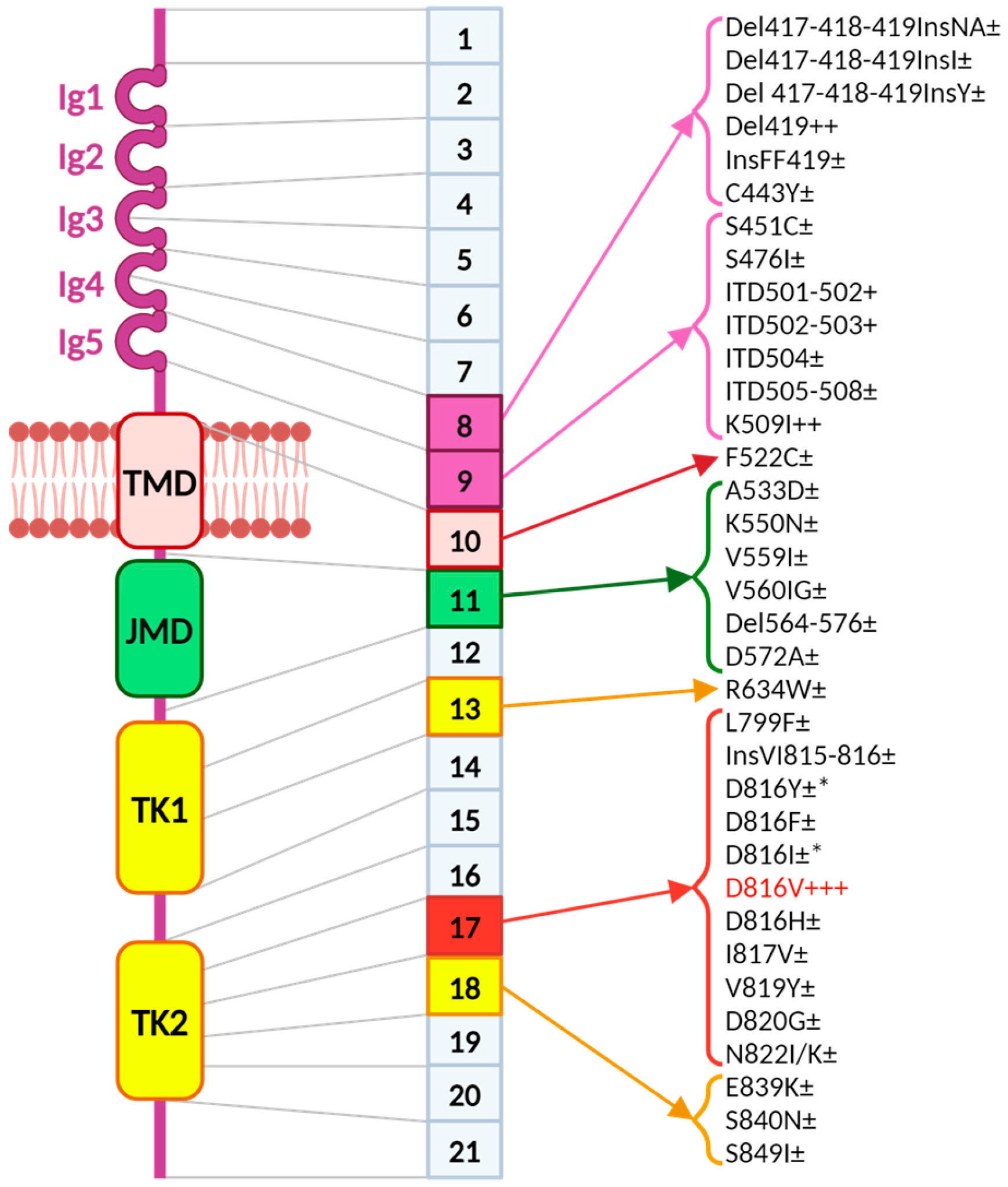
2. KIT/CD117
3. Downstream Signaling of KIT
4. KIT Is Activated in Many Tumors
5. Methods for the Detection of c-KIT Mutations
6. Genomic DNA (gDNA) and mRNA: Which Is Better?
7. What to Do when the Mutational Test Is Negative but the Clinical Suspicion Is Strong
8. What Material Should Be Used for the Mutational Analysis of c-KIT? Differences between BM or PB
9. How Frequent c-KIT p.D816V Allelic Burden Should Be Monitored?
10. Significance of c-KIT p.D816V Allelic Burden
12. Additional Molecular and Cytogenetic Lesions
13. Summary and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heybeli, C. Mast Cells, Mastocytosis, and Related Disorders. N Engl J Med 2015, 373, 1885. [CrossRef]
- Tzankov, A.; Duncavage, E.; Craig, F.E.; Kelemen, K.; King, R.L.; Orazi, A.; Quintanilla-Martinez, L.; Reichard, K.K.; Rimsza, L.M.; Wang, S.A.; et al. Mastocytosis. American Journal of Clinical Pathology 2021, 155, 239–266. [CrossRef]
- Valent, P.; Akin, C.; Hartmann, K.; Nilsson, G.; Reiter, A.; Hermine, O.; Sotlar, K.; Sperr, W.R.; Escribano, L.; George, T.I.; et al. Mast Cells as a Unique Hematopoietic Lineage and Cell System: From Paul Ehrlich’s Visions to Precision Medicine Concepts. Theranostics 2020, 10, 10743–10768. [CrossRef]
- Kanakura, Y.; Furitsu, T.; Tsujimura, T.; Butterfield, J.H.; Ashman, L.K.; Ikeda, H.; Kitayama, H.; Kanayama, Y.; Matsuzawa, Y.; Kitamura, Y. Activating Mutations of the C-Kit Proto-Oncogene in a Human Mast Cell Leukemia Cell Line. Leukemia 1994, 8 Suppl 1, S18-22.
- Metcalfe, D.D.; Mekori, Y.A. Pathogenesis and Pathology of Mastocytosis. Annu Rev Pathol 2017, 12, 487–514. [CrossRef]
- De Matteis, G.; Zanotti, R.; Colarossi, S.; De Benedittis, C.; Garcia-Montero, A.; Bonifacio, M.; Sartori, M.; Aprili, F.; Caruso, B.; Paviati, E.; et al. The Impact of Sensitive KIT D816V Detection on Recognition of Indolent Systemic Mastocytosis. Leukemia Research 2015, 39, 273–278. [CrossRef]
- Valent, P.; Akin, C.; Hartmann, K.; Alvarez-Twose, I.; Brockow, K.; Hermine, O.; Niedoszytko, M.; Schwaab, J.; Lyons, J.J.; Carter, M.C.; et al. Updated Diagnostic Criteria and Classification of Mast Cell Disorders: A Consensus Proposal. HemaSphere 2021, 5, e646. [CrossRef]
- Khoury, J.D.; Solary, E.; Abla, O.; Akkari, Y.; Alaggio, R.; Apperley, J.F.; Bejar, R.; Berti, E.; Busque, L.; Chan, J.K.C.; et al. The 5th Edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 2022, 36, 1703–1719. [CrossRef]
- Arber, D.A.; Orazi, A.; Hasserjian, R.P.; Borowitz, M.J.; Calvo, K.R.; Kvasnicka, H.-M.; Wang, S.A.; Bagg, A.; Barbui, T.; Branford, S.; et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: Integrating Morphologic, Clinical, and Genomic Data. Blood 2022, 140, 1200–1228. [CrossRef]
- Arock, M.; Hoermann, G.; Sotlar, K.; Hermine, O.; Sperr, W.R.; Hartmann, K.; Brockow, K.; Akin, C.; Triggiani, M.; Broesby-Olsen, S.; et al. Clinical Impact and Proposed Application of Molecular Markers, Genetic Variants, and Cytogenetic Analysis in Mast Cell Neoplasms: Status 2022. Journal of Allergy and Clinical Immunology 2022, 149, 1855–1865. [CrossRef]
- Valent, P.; Hoermann, G.; Bonadonna, P.; Hartmann, K.; Sperr, W.R.; Broesby-Olsen, S.; Brockow, K.; Niedoszytko, M.; Hermine, O.; Chantran, Y.; et al. The Normal Range of Baseline Tryptase Should Be 1 to 15 Ng/mL and Covers Healthy Individuals With HαT. The Journal of Allergy and Clinical Immunology: In Practice 2023, 11, 3010–3020. [CrossRef]
- Alvarez-Twose, I.; González-de-Olano, D.; Sánchez-Muñoz, L.; Matito, A.; Jara-Acevedo, M.; Teodosio, C.; García-Montero, A.; Morgado, J.M.; Orfao, A.; Escribano, L. Validation of the REMA Score for Predicting Mast Cell Clonality and Systemic Mastocytosis in Patients with Systemic Mast Cell Activation Symptoms. Int Arch Allergy Immunol 2012, 157, 275–280. [CrossRef]
- Golden, D.B.K.; Wang, J.; Waserman, S.; Akin, C.; Campbell, R.L.; Ellis, A.K.; Greenhawt, M.; Lang, D.M.; Ledford, D.K.; Lieberman, J.; et al. Anaphylaxis: A 2023 Practice Parameter Update. Annals of Allergy, Asthma & Immunology 2024, 132, 124–176. [CrossRef]
- Gulen, T. Management of Mediator Symptoms, Allergy, and Anaphylaxis in Mastocytosis. Immunology and Allergy Clinics of North America 2023, 43, 681–698. [CrossRef]
- Giannetti, M.P.; Nicoloro-SantaBarbara, J.; Godwin, G.; Middlesworth, J.; Espeland, A.; Douvas, J.L.; Castells, M.C. Challenges in Drug and Hymenoptera Venom Hypersensitivity Diagnosis and Management in Mastocytosis. Diagnostics 2024, 14, 123. [CrossRef]
- Valent, P. Risk Factors and Management of Severe Life-threatening Anaphylaxis in Patients with Clonal Mast Cell Disorders. Clin Experimental Allergy 2014, 44, 914–920. [CrossRef]
- Roskoski, R. Structure and Regulation of Kit Protein-Tyrosine Kinase—The Stem Cell Factor Receptor. Biochemical and Biophysical Research Communications 2005, 338, 1307–1315. [CrossRef]
- Blechman, J.M.; Lev, S.; Givol, D.; Yarden, Y. Structure-Function Analyses of the Kit Receptor for the Steel Factor. STEM CELLS 1996, 11, 12–21. [CrossRef]
- Giebel, L.B.; Strunk, K.M.; Holmes, S.A.; Spritz, R.A. Organization and Nucleotide Sequence of the Human KIT (Mast/Stem Cell Growth Factor Receptor) Proto-Oncogene. Oncogene 1992, 7, 2207–2217.
- Chan, E.C.; Bai, Y.; Bandara, G.; Simakova, O.; Brittain, E.; Scott, L.; Dyer, K.D.; Klion, A.D.; Maric, I.; Gilfillan, A.M.; et al. KIT GNNK Splice Variants: Expression in Systemic Mastocytosis and Influence on the Activating Potential of the D816V Mutation in Mast Cells. Experimental Hematology 2013, 41, 870-881.e2. [CrossRef]
- Lennartsson, J.; Jelacic, T.; Linnekin, D.; Shivakrupa, R. Normal and Oncogenic Forms of the Receptor Tyrosine Kinase Kit. STEM CELLS 2005, 23, 16–43. [CrossRef]
- Crosier, P.S.; Ricciardi, S.T.; Hall, L.R.; Vitas, M.R.; Clark, S.C.; Crosier, K.E. Expression of Isoforms of the Human Receptor Tyrosine Kinase C-Kit in Leukemic Cell Lines and Acute Myeloid Leukemia. Blood 1993, 82, 1151–1158.
- Lennartsson, J.; Rönnstrand, L. Stem Cell Factor Receptor/c-Kit: From Basic Science to Clinical Implications. Physiological Reviews 2012, 92, 1619–1649. [CrossRef]
- Reber, L.; Da Silva, C.A.; Frossard, N. Stem Cell Factor and Its Receptor C-Kit as Targets for Inflammatory Diseases. European Journal of Pharmacology 2006, 533, 327–340. [CrossRef]
- Ray, P.; Krishnamoorthy, N.; Oriss, T.B.; Ray, A. Signaling of C-kit in Dendritic Cells Influences Adaptive Immunity. Annals of the New York Academy of Sciences 2010, 1183, 104–122. [CrossRef]
- Waskow, C.; Terszowski, G.; Costa, C.; Gassmann, M.; Rodewald, H.-R. Rescue of Lethal C-KitW/W Mice by Erythropoietin. Blood 2004, 104, 1688–1695. [CrossRef]
- Leong, K.G.; Wang, B.-E.; Johnson, L.; Gao, W.-Q. Generation of a Prostate from a Single Adult Stem Cell. Nature 2008, 456, 804–808. [CrossRef]
- Laurson, J.; Selden, C.; Clements, M.; Mavri-Damelin, D.; Coward, S.; Lowdell, M.; Hodgson, H.J.F. Putative Human Liver Progenitor Cells in Explanted Liver. Cells Tissues Organs 2007, 186, 180–191. [CrossRef]
- Zaruba, M.-M.; Soonpaa, M.; Reuter, S.; Field, L.J. Cardiomyogenic Potential of C-Kit + –Expressing Cells Derived From Neonatal and Adult Mouse Hearts. Circulation 2010, 121, 1992–2000. [CrossRef]
- Liang, J.; Wu, Y.-L.; Chen, B.-J.; Zhang, W.; Tanaka, Y.; Sugiyama, H. The C-Kit Receptor-Mediated Signal Transduction and Tumor-Related Diseases. Int. J. Biol. Sci. 2013, 9, 435–443. [CrossRef]
- Thömmes, K.; Lennartsson, J.; Carlberg, M.; Rönnstrand, L. Identification of Tyr-703 and Tyr-936 as the Primary Association Sites for Grb2 and Grb7 in the c-Kit/Stem Cell Factor Receptor. Biochem J 1999, 341 ( Pt 1), 211–216.
- Chian, R.; Young, S.; Danilkovitch-Miagkova, A.; Rönnstrand, L.; Leonard, E.; Ferrao, P.; Ashman, L.; Linnekin, D. Phosphatidylinositol 3 Kinase Contributes to the Transformation of Hematopoietic Cells by the D816V C-Kit Mutant. Blood 2001, 98, 1365–1373. [CrossRef]
- Hashimoto, K.; Matsumura, I.; Tsujimura, T.; Kim, D.-K.; Ogihara, H.; Ikeda, H.; Ueda, S.; Mizuki, M.; Sugahara, H.; Shibayama, H.; et al. Necessity of Tyrosine 719 and Phosphatidylinositol 3′-Kinase–Mediated Signal Pathway in Constitutive Activation and Oncogenic Potential of c-Kit Receptor Tyrosine Kinase with the Asp814Val Mutation. Blood 2003, 101, 1094–1102. [CrossRef]
- Sun, J.; Pedersen, M.; Rönnstrand, L. Gab2 Is Involved in Differential Phosphoinositide 3-Kinase Signaling by Two Splice Forms of c-Kit. Journal of Biological Chemistry 2008, 283, 27444–27451. [CrossRef]
- Masson, K.; Heiss, E.; Band, H.; Rönnstrand, L. Direct Binding of Cbl to Tyr568 and Tyr936 of the Stem Cell Factor Receptor/c-Kit Is Required for Ligand-Induced Ubiquitination, Internalization and Degradation. Biochemical Journal 2006, 399, 59–67. [CrossRef]
- Hirota, S.; Isozaki, K.; Moriyama, Y.; Hashimoto, K.; Nishida, T.; Ishiguro, S.; Kawano, K.; Hanada, M.; Kurata, A.; Takeda, M.; et al. Gain-of-Function Mutations of c- Kit in Human Gastrointestinal Stromal Tumors. Science 1998, 279, 577–580. [CrossRef]
- Duensing, A.; Heinrich, M.C.; Fletcher, C.D.M.; Fletcher, J.A. Biology of Gastrointestinal Stromal Tumors: KIT Mutations and Beyond. Cancer Investigation 2004, 22, 106–116. [CrossRef]
- Curtin, J.A.; Busam, K.; Pinkel, D.; Bastian, B.C. Somatic Activation of KIT in Distinct Subtypes of Melanoma. JCO 2006, 24, 4340–4346. [CrossRef]
- Sakuma, Y.; Sakurai, S.; Oguni, S.; Hironaka, M.; Salto, K. Alterations of the C-kit Gene in Testicular Germ Cell Tumors. Cancer Science 2003, 94, 486–491. [CrossRef]
- Longley, B.J.; Metcalfe, D.D.; Tharp, M.; Wang, X.; Tyrrell, L.; Lu, S.; Heitjan, D.; Ma, Y. Activating and Dominant Inactivating C- KIT Catalytic Domain Mutations in Distinct Clinical Forms of Human Mastocytosis. Proc. Natl. Acad. Sci. U.S.A. 1999, 96, 1609–1614. [CrossRef]
- Beghini, A.; Ripamonti, C.B.; Cairoli, R.; Cazzaniga, G.; Colapietro, P.; Elice, F.; Nadali, G.; Grillo, G.; Haas, O.A.; Biondi, A.; et al. KIT Activating Mutations: Incidence in Adult and Pediatric Acute Myeloid Leukemia, and Identification of an Internal Tandem Duplication. Haematologica 2004, 89, 920–925.
- Chantran, Y.; Valent, P.; Arock, M. KIT Mutations and Other Genetic Defects in Mastocytosis. Immunology and Allergy Clinics of North America 2023, 43, 651–664. [CrossRef]
- Navarro-Navarro, P.; Álvarez-Twose, I.; Pérez-Pons, A.; Henriques, A.; Mayado, A.; García-Montero, A.C.; Sánchez-Muñoz, L.; González-López, O.; Matito, A.; Caldas, C.; et al. KIT D816V Mutation in Blood for the Diagnostic Screening of Systemic Mastocytosis and Mast Cell Activation Syndromes. Allergy 2023, 78, 1347–1359. [CrossRef]
- Nedoszytko, B.; Arock, M.; Lyons, J.; Bachelot, G.; Schwartz, L.; Reiter, A.; Jawhar, M.; Schwaab, J.; Lange, M.; Greiner, G.; et al. Clinical Impact of Inherited and Acquired Genetic Variants in Mastocytosis. IJMS 2021, 22, 411. [CrossRef]
- Bibi, S.; Langenfeld, F.; Jeanningros, S.; Brenet, F.; Soucie, E.; Hermine, O.; Damaj, G.; Dubreuil, P.; Arock, M. Molecular Defects in Mastocytosis. Immunology and Allergy Clinics of North America 2014, 34, 239–262. [CrossRef]
- Bodemer, C.; Hermine, O.; Palmérini, F.; Yang, Y.; Grandpeix-Guyodo, C.; Leventhal, P.S.; Hadj-Rabia, S.; Nasca, L.; Georgin-Lavialle, S.; Cohen-Akenine, A.; et al. Pediatric Mastocytosis Is a Clonal Disease Associated with D816V and Other Activating C-KIT Mutations. Journal of Investigative Dermatology 2010, 130, 804–815. [CrossRef]
- Nakagomi, N.; Hirota, S. Juxtamembrane-Type c-Kit Gene Mutation Found in Aggressive Systemic Mastocytosis Induces Imatinib-Resistant Constitutive KIT Activation. Laboratory Investigation 2007, 87, 365–371. [CrossRef]
- Georgin-Lavialle, S.; Aguilar, C.; Guieze, R.; Lhermitte, L.; Bruneau, J.; Fraitag, S.; Canioni, D.; Chandesris, M.-O.; Suarez, F.; Grandpeix-Guyodo, C.; et al. Mast Cell Sarcoma: A Rare and Aggressive Entity—Report of Two Cases and Review of the Literature. JCO 2013, 31, e90–e97. [CrossRef]
- Valent, P.; Berger, J.; Cerny-Reiterer, S.; Peter, B.; Eisenwort, G.; Hoermann, G.; Müllauer, L.; Mannhalter, C.; Steurer, M.; Bettelheim, P.; et al. Chronic Mast Cell Leukemia (MCL) with KIT S476I: A Rare Entity Defined by Leukemic Expansion of Mature Mast Cells and Absence of Organ Damage. Ann Hematol 2015, 94, 223–231. [CrossRef]
- Meni, C.; Georgin-Lavialle, S.; Le Saché De Peufeilhoux, L.; Jais, J.P.; Hadj-Rabia, S.; Bruneau, J.; Fraitag, S.; Hanssens, K.; Dubreuil, P.; Hermine, O.; et al. Paediatric Mastocytosis: Long-Term Follow-up of 53 Patients with Whole Sequencing of KIT . A Prospective Study. Br J Dermatol 2018, 179, 925–932. [CrossRef]
- Hoermann, G.; Sotlar, K.; Jawhar, M.; Kristensen, T.; Bachelot, G.; Nedoszytko, B.; Carter, M.C.; Horny, H.-P.; Bonadonna, P.; Sperr, W.R.; et al. Standards of Genetic Testing in the Diagnosis and Prognostication of Systemic Mastocytosis in 2022: Recommendations of the EU-US Cooperative Group. The Journal of Allergy and Clinical Immunology: In Practice 2022, 10, 1953–1963. [CrossRef]
- Akin, C. Multilineage Hematopoietic Involvement in Systemic Mastocytosis. Leukemia Research 2003, 27, 877–878. [CrossRef]
- Garcia-Montero, A.C. KIT Mutation in Mast Cells and Other Bone Marrow Hematopoietic Cell Lineages in Systemic Mast Cell Disorders: A Prospective Study of the Spanish Network on Mastocytosis (REMA) in a Series of 113 Patients. Blood 2006, 108, 2366–2372. [CrossRef]
- Escribano, L.; Álvarez-Twose, I.; Sánchez-Muñoz, L.; Garcia-Montero, A.; Núñez, R.; Almeida, J.; Jara-Acevedo, M.; Teodósio, C.; García-Cosío, M.; Bellas, C.; et al. Prognosis in Adult Indolent Systemic Mastocytosis: A Long-Term Study of the Spanish Network on Mastocytosis in a Series of 145 Patients. Journal of Allergy and Clinical Immunology 2009, 124, 514–521. [CrossRef]
- Sotlar, K.; Colak, S.; Bache, A.; Berezowska, S.; Krokowski, M.; Bültmann, B.; Valent, P.; Horny, H. Variable Presence of KIT D816V in Clonal Haematological Non-mast Cell Lineage Diseases Associated with Systemic Mastocytosis (SM–AHNMD). The Journal of Pathology 2010, 220, 586–595. [CrossRef]
- Jawhar, M.; Schwaab, J.; Schnittger, S.; Sotlar, K.; Horny, H.-P.; Metzgeroth, G.; Müller, N.; Schneider, S.; Naumann, N.; Walz, C.; et al. Molecular Profiling of Myeloid Progenitor Cells in Multi-Mutated Advanced Systemic Mastocytosis Identifies KIT D816V as a Distinct and Late Event. Leukemia 2015, 29, 1115–1122. [CrossRef]
- Kristensen, T.; Vestergaard, H.; Bindslev-Jensen, C.; Møller, M.B.; Broesby-Olsen, S.; on Behalf of the Mastocytosis Centre Odense University Hospital (MastOUH) Sensitive KIT D816V Mutation Analysis of Blood as a Diagnostic Test in Mastocytosis. American J Hematol 2014, 89, 493–498. [CrossRef]
- Erben, P.; Schwaab, J.; Metzgeroth, G.; Horny, H.-P.; Jawhar, M.; Sotlar, K.; Fabarius, A.; Teichmann, M.; Schneider, S.; Ernst, T.; et al. The KIT D816V Expressed Allele Burden for Diagnosis and Disease Monitoring of Systemic Mastocytosis. Ann Hematol 2014, 93, 81–88. [CrossRef]
- Greiner, G.; Gurbisz, M.; Ratzinger, F.; Witzeneder, N.; Simonitsch-Klupp, I.; Mitterbauer-Hohendanner, G.; Mayerhofer, M.; Müllauer, L.; Sperr, W.R.; Valent, P.; et al. Digital PCR: A Sensitive and Precise Method for KIT D816V Quantification in Mastocytosis. Clinical Chemistry 2018, 64, 547–555. [CrossRef]
- Hindson, B.J.; Ness, K.D.; Masquelier, D.A.; Belgrader, P.; Heredia, N.J.; Makarewicz, A.J.; Bright, I.J.; Lucero, M.Y.; Hiddessen, A.L.; Legler, T.C.; et al. High-Throughput Droplet Digital PCR System for Absolute Quantitation of DNA Copy Number. Anal. Chem. 2011, 83, 8604–8610. [CrossRef]
- Huggett, J.F.; Cowen, S.; Foy, C.A. Considerations for Digital PCR as an Accurate Molecular Diagnostic Tool. Clinical Chemistry 2015, 61, 79–88. [CrossRef]
- Naumann, N.; Lübke, J.; Baumann, S.; Schwaab, J.; Hoffmann, O.; Kreil, S.; Dangelo, V.; Reiter, L.; Bugert, P.; Kristensen, T.; et al. Adverse Prognostic Impact of the KIT D816V Transcriptional Activity in Advanced Systemic Mastocytosis. IJMS 2021, 22, 2562. [CrossRef]
- Kristensen, T.; Broesby-Olsen, S.; Vestergaard, H.; Bindslev-Jensen, C.; Møller, M.B.; the Mastocytosis Centre Odense University Hospital (MastOUH) Comparison of gDNA -based versus mRNA -based KIT D816V Mutation Analysis Reveals Large Differences between Blood and Bone Marrow in Systemic Mastocytosis. Br J Haematol 2017, 178, 330–332. [CrossRef]
- Greiner, G.; Gurbisz, M.; Ratzinger, F.; Witzeneder, N.; Class, S.V.; Eisenwort, G.; Simonitsch-Klupp, I.; Esterbauer, H.; Mayerhofer, M.; Müllauer, L.; et al. Molecular Quantification of Tissue Disease Burden Is a New Biomarker and Independent Predictor of Survival in Mastocytosis. Haematologica 2020, 105, 366–374. [CrossRef]
- Hoermann, G.; Gleixner, K.V.; Dinu, G.E.; Kundi, M.; Greiner, G.; Wimazal, F.; Hadzijusufovic, E.; Mitterbauer, G.; Mannhalter, C.; Valent, P.; et al. The KIT D 816 V Allele Burden Predicts Survival in Patients with Mastocytosis and Correlates with the WHO Type of the Disease. Allergy 2014, 69, 810–813. [CrossRef]
- Jawhar, M.; Schwaab, J.; Naumann, N.; Horny, H.-P.; Sotlar, K.; Haferlach, T.; Metzgeroth, G.; Fabarius, A.; Valent, P.; Hofmann, W.-K.; et al. Response and Progression on Midostaurin in Advanced Systemic Mastocytosis: KIT D816V and Other Molecular Markers. Blood 2017, 130, 137–145. [CrossRef]
- Tefferi, A.; Levine, R.L.; Lim, K.-H.; Abdel-Wahab, O.; Lasho, T.L.; Patel, J.; Finke, C.M.; Mullally, A.; Li, C.-Y.; Pardanani, A.; et al. Frequent TET2 Mutations in Systemic Mastocytosis: Clinical, KITD816V and FIP1L1-PDGFRA Correlates. Leukemia 2009, 23, 900–904. [CrossRef]
- Traina, F.; Visconte, V.; Jankowska, A.M.; Makishima, H.; O’Keefe, C.L.; Elson, P.; Han, Y.; Hsieh, F.H.; Sekeres, M.A.; Mali, R.S.; et al. Single Nucleotide Polymorphism Array Lesions, TET2, DNMT3A, ASXL1 and CBL Mutations Are Present in Systemic Mastocytosis. PLoS ONE 2012, 7, e43090. [CrossRef]
- Soucie, E.; Hanssens, K.; Mercher, T.; Georgin-Lavialle, S.; Damaj, G.; Livideanu, C.; Chandesris, M.O.; Acin, Y.; Létard, S.; De Sepulveda, P.; et al. In Aggressive Forms of Mastocytosis, TET2 Loss Cooperates with c-KITD816V to Transform Mast Cells. Blood 2012, 120, 4846–4849. [CrossRef]
- Schwaab, J.; Schnittger, S.; Sotlar, K.; Walz, C.; Fabarius, A.; Pfirrmann, M.; Kohlmann, A.; Grossmann, V.; Meggendorfer, M.; Horny, H.-P.; et al. Comprehensive Mutational Profiling in Advanced Systemic Mastocytosis. Blood 2013, 122, 2460–2466. [CrossRef]
- Jawhar, M.; Schwaab, J.; Schnittger, S.; Meggendorfer, M.; Pfirrmann, M.; Sotlar, K.; Horny, H.-P.; Metzgeroth, G.; Kluger, S.; Naumann, N.; et al. Additional Mutations in SRSF2, ASXL1 and/or RUNX1 Identify a High-Risk Group of Patients with KIT D816V+ Advanced Systemic Mastocytosis. Leukemia 2016, 30, 136–143. [CrossRef]
- Naumann, N.; Jawhar, M.; Schwaab, J.; Kluger, S.; Lübke, J.; Metzgeroth, G.; Popp, H.D.; Khaled, N.; Horny, H.; Sotlar, K.; et al. Incidence and Prognostic Impact of Cytogenetic Aberrations in Patients with Systemic Mastocytosis. Genes Chromosomes & Cancer 2018, 57, 252–259. [CrossRef]
- Kluin-Nelemans, H.C.; Jawhar, M.; Reiter, A.; Van Anrooij, B.; Gotlib, J.; Hartmann, K.; Illerhaus, A.; Oude Elberink, H.N.G.; Gorska, A.; Niedoszytko, M.; et al. Cytogenetic and Molecular Aberrations and Worse Outcome for Male Patients in Systemic Mastocytosis. Theranostics 2021, 11, 292–303. [CrossRef]
| Bone Marrow Mastocytosis (BBM) | |
| Indolent Systemic Mastocytosis (ISM): | Bone marrow infiltration <20% without associated hematologic disease, absence of C findings, and absence or presence of 1 B finding |
| Smoldering Systemic Mastocytosis (SSM): | Bone marrow infiltration <20% without associated hematologic disease, absence of C findings, and presence of 2 or more B findings |
| Aggressive Systemic Mastocytosis (ASM): | Bone marrow infiltration <20% without associated hematologic disease, presence of at least 1 C finding |
| Systemic Mastocytosis with Associated Hematologic Neoplasm (SM-AHN): | Mast cell percentage <20% and bone marrow histology conclusive for hematologic disease (myelodysplastic syndrome or myeloproliferative syndrome). |
| Mast Cell Leukemia (MCL): | Patients with bone marrow infiltration >20% without signs of other hematologic diseases. Typically presents with weight loss, fatigue, anemia, and other C findings, such as hypotension, flushing, diarrhea, and coagulopathy. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




