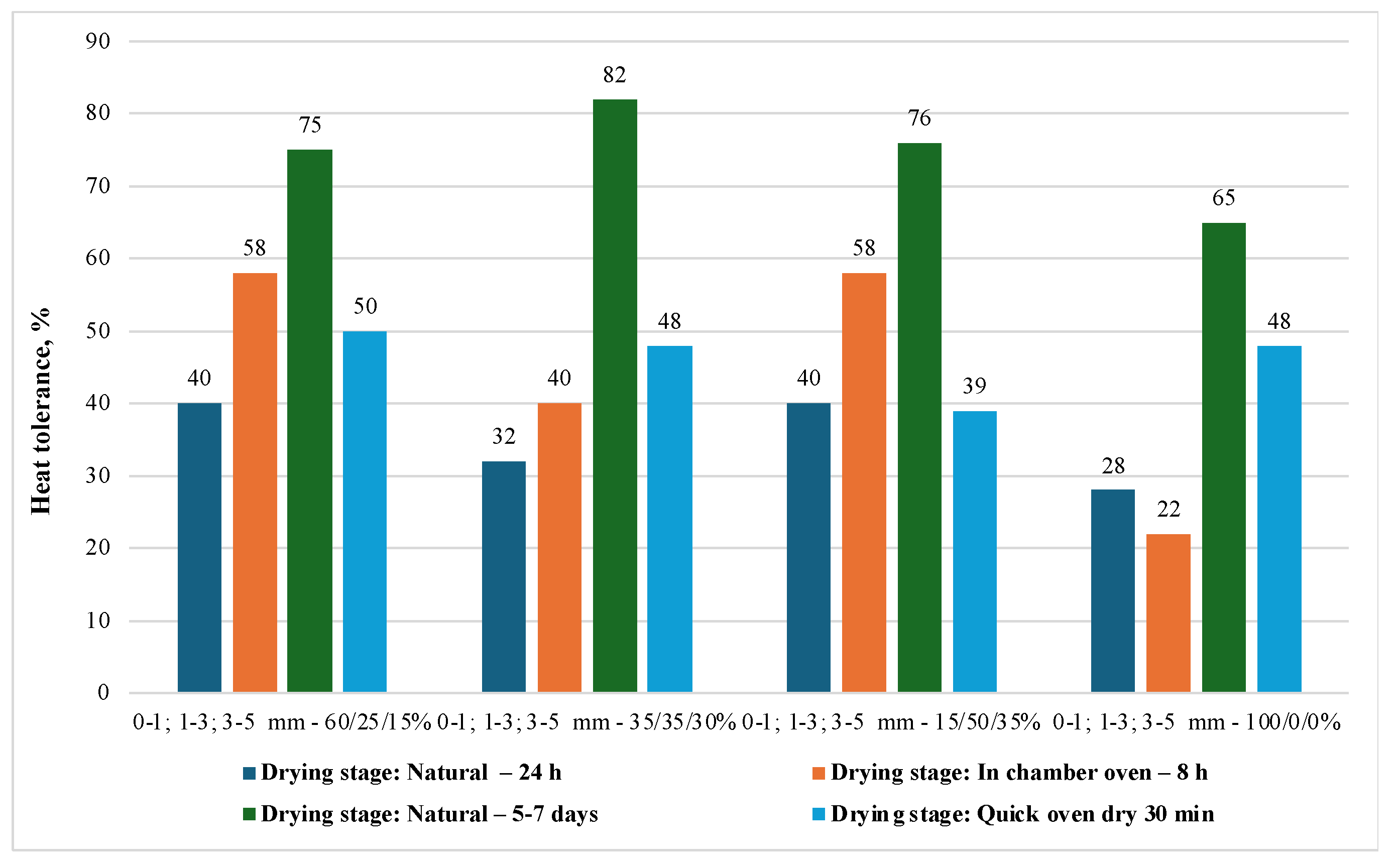3. Results and Discussion
The results of measurements of the specific surface area and average particle size of microsilica are presented in
Table 1.
The obtained specific surface area (11730.67 cm²/g) is quite high for bulk materials used in metallurgical processing. At the same time, the high specific surface area indicates its fine dispersion, high absorptive, and reactive capacity. Based on measurements from the dispersion analysis instrument, it can be concluded that the specific surface area of microsilica is the sum of the surface areas of all its particles, and the smaller the size of these particles, the greater the total specific surface area.
Next, experiments of briquettes heat tolerance evaluation were conducted.
Figure 5 shows a photo of the briquette before the thermal shock (a) and immediately after it (b).
As can be seen from
Figure 5, the briquettes after thermal shock were covered with a white layer of silicon dioxide. In this layer, the carbon component burned out. As a result, the packing density of the particles was disrupted. Therefore, the oxidized layer is not mechanically strong and falls off from the surface of the briquette during the subsequent abrasion test.
Figure 6 presents the results of the briquettes heat tolerance of various compositions at various stages of the drying process.
The graph shows the heat tolerance of briquettes of different fractional compositions at various stages of drying. The highest heat tolerance (82%) was achieved with natural drying for 5–7 days for briquettes with a fractional composition of 35/35/30% (0-1; 1-3; 3-5 mm). Quick drying for 30 minutes in an oven gave the lowest results in terms of heat tolerance for all fractional compositions. In general, longer natural drying times result in better heat tolerance.
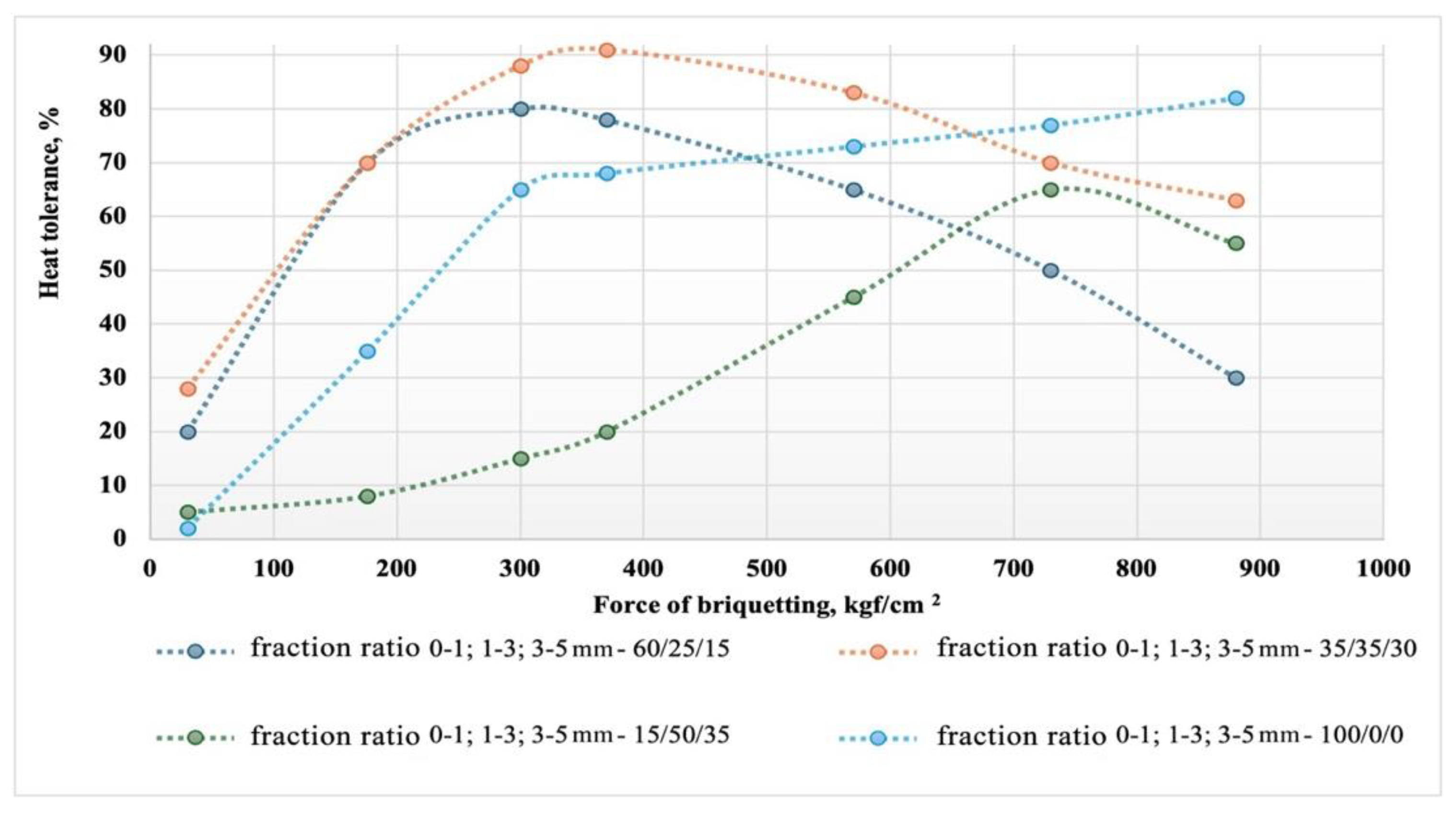
Figure 7 shows graphs of the dependence of the heat tolerance of briquettes for different types of granulometric composition of the composite briquetting charge.
The graph illustrates the impact of briquetting force and particle size distribution on the heat tolerance of briquettes made from a composite mixture of microsilica and carbon reducing agents. It was found that heat tolerance increases with the briquetting force up to a certain optimum, which varies depending on the particle size distribution. The optimal briquetting force for achieving maximum heat tolerance is in the range of 400 to 500 kgf/cm², with compositions that have a more balanced particle size distribution showing the most consistent results. Reducing the fine fraction content to 15% results in poorer briquetting conditions and necessitates an increase in pressure to 700-800 kgf/cm² to achieve optimal heat resistance, which is reduced to 60-65%. Further increasing the force (up to 900 kgf/cm²) leads to a decrease in heat tolerance for most compositions, indicating potential structural changes in the briquettes. To ensure optimal heat tolerance, it is recommended to use a briquetting pressure of approximately 370-400 kgf/cm² and a particle size distribution ratio of 0-1; 1-3; 3-5 mm in the proportion 35/35/30. However, briquettes with a high fine fraction content (0-1 mm) maintain or even improve their heat tolerance at maximum force, making them promising for use under conditions of high thermal load. These findings underscore the importance of selecting both the particle size distribution and briquetting force to optimize briquette properties in metallurgical silicon production.
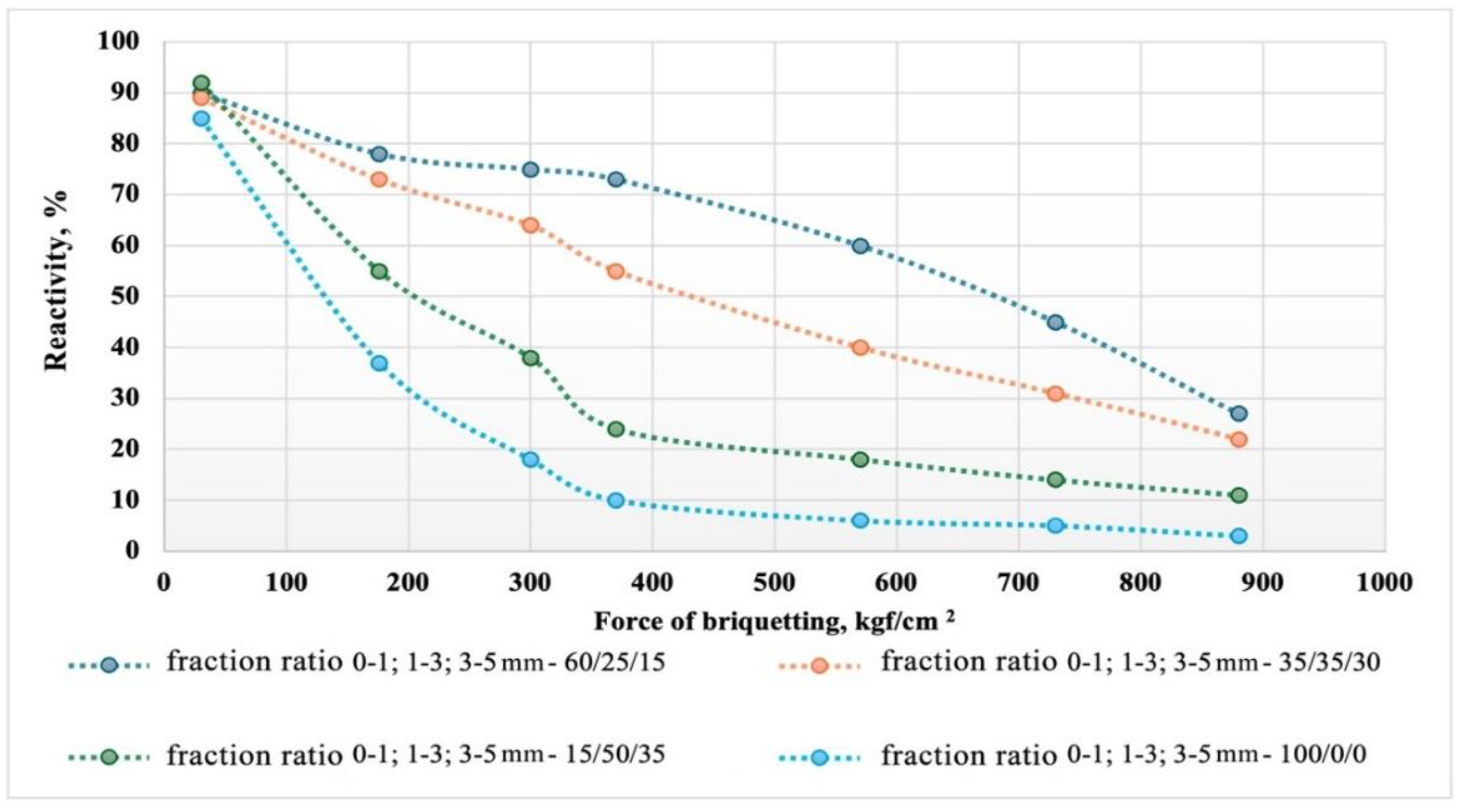
Figure 8 shows graphs of the dependence of the reactive activity of the briquettes on the briquetting pressure and the granulometric composition of the briquetting mix.
Obviously, for all studied variants of briquettes, with increasing briquetting force, the reactivity decreases. At the same time, there is a direct proportional dependence of the briquette's reactivity on the granulometric composition.
With an increase in the proportion of particles with sizes of 1-3 and 3-5 mm, the reactivity of the briquettes increases. This indirectly confirms the mechanism of the silicon reduction process through the gas phase involving silicon monoxide and silicon carbide. Despite the highly developed reactivity of the fine-dispersed component of the briquettes, increasing its share does not lead to an acceleration of the reduction. On the contrary, the over-compaction of the fine-dispersed phase leads to increased diffusion inhibition during the movement of silicon monoxide and carbon within the briquette. The presence of particles of different diameters in the briquette helps to improve the gas permeability of the reaction products layer due to the formation of macropores within the briquette during the gasification of carbon inclusions.
This type of briquette visually differs from briquettes made using other types of reducers, which allows it to be identified by external signs. Based on graph-analytical analysis, it was assumed that to achieve full heat tolerance of the briquette, the briquetting force should be 800-900 kgf/cm² (80-90 MPa), which corresponds to the literature data [
1,
2]. However, this did not happen. Briquetting at a pressure of 570 kgf/cm² led to significant delamination of the briquette and a decrease in heat tolerance to 65%. Delamination refers to cracks developing perpendicular to the force application vector during briquetting, encircling the briquette around its perimeter. The cracks are mainly surface-level, penetrating the briquette body to a depth of 1-3 mm as shown in
Figure 9.
However, there are cracks that penetrate through the entire body of the briquette, leading to transverse rupture either during the briquetting process with clogging of the working surface of the press, or to the rupture of the finished briquette when a slight force is applied in the corresponding direction. Thus, anisotropy of mechanical and thermomechanical properties is observed.
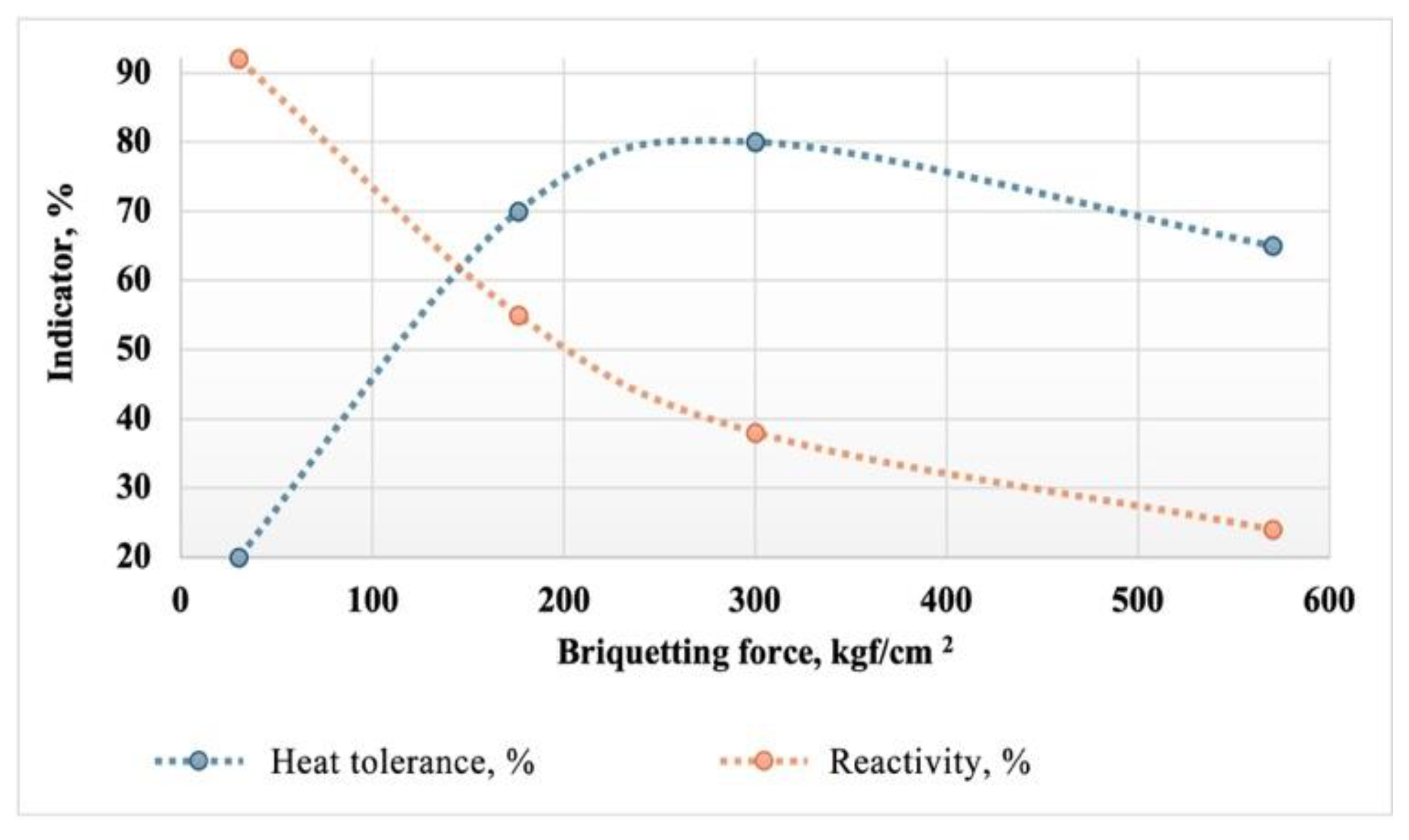
It has been established that heat tolerance depends on the specific pressing force and the granulometric composition of the charge. The dependence of the heat tolerance and reactivity of the briquettes on the briquetting force is shown in
Figure 10.
The graph shows that with increasing briquetting force, the heat tolerance of the briquettes goes through a maximum in the range of 250-350 kgf/cm². Simultaneously, with the increase in briquetting force, a decrease in the reactivity of the briquettes is observed. At the optimal force in terms of heat tolerance, the reactivity of the briquette is at critical values, around 40 %. To explore the possibility of increasing the reactivity in combination with high heat tolerance, experiments were conducted at lower briquetting forces but with a higher degree of variability in the granulometric composition. This assumption was based on the fact that polydisperse materials typically create a denser packing during briquetting without the presence of macropores within the briquette. Due to the naturally high packing density of the particles, on the one hand, the briquetting force required to ensure high heat tolerance and mechanical strength is reduced. On the other hand, the reduction in briquetting force ensures the presence of micropores, facilitating the removal of gaseous reaction products from the briquette body.
Next, batches of briquettes with the optimal raw material composition were tested as raw material for the production of metallurgical-grade silicon in a submerged arc furnace with a transformer power of 200 kVA (
Figure 11).
Silicon smelting began using a standard traditional charge mixture consisting of quartzite and reducing agents in the form of coal and charcoal, special coke, and the charge also additionally contains wood chips. The ratio of the presented components in the composition of the charge is presented in
Table 2.
Theoretically, the process of smelting metallurgical silicon can be described by the following reaction equation:
Where silica (SiO₂) is reduced by carbon to elemental silicon (Si) and silicon oxide (SiO) in the gaseous state:
The gaseous silicon oxide is then reduced to elemental silicon:
It is worth noting that silicon carbide (SiC) also forms in the high-temperature zone of the furnace:
Silicon carbide interacts with silicon oxide, leading to its decomposition and the formation of additional elemental silicon:
In these tests, a 30% replacement of the traditional charge mixture with briquettes was tested. The negative influence of the amorphous phase composition of microsilica on the process of smelting technical silicon has not been established. There was no shock destruction of briquettes under the influence of thermal and current loads on the fire pit. As a result, a batch of metallurgical silicon was obtained. The fracture of the silicon ingots (
Figure 12) had a steel-gray color with pronounced crystal plates. The structure of the alloy is dense, without shells and foreign inclusions.
The average chemical composition of silicon samples obtained as a result of testing is presented in
Table 3.
At the same time, the extraction of silicon into metal using a standard charge was 71%, and the extraction using a briquetted monocharge was 85%. The results of trial tests have proven the possibility of using briquetted monocharge as a raw material for the smelting of metallurgical grade silicon.