3. Results
In our previous study [
36] the intrinsic self-healing at 20 °C of one PU made with polycarbonate polyol of molecular weight 1000 Da was ascribed to moderate micro-phase separation, reduced ability of SS to crystallize, the existence of a mixed phase of HS and SS, and the interactions between the polycarbonate soft segments. A mechanism of intrinsic self-healing based on the existence of dynamic non-covalent exchange interactions between polycarbonate soft segments was proposed. Thus, upon damage of the PU and, due to the mobility of the soft segments, the interactions among initially bonded carbonate groups and the free carbonate groups in the soft segments are produced causing a fast intrinsic self-healing.
Considering that the mobility of the soft segments and the number of carbonate groups differ depending on the molecular weight of the polycarbonate polyol, in this study the self-healing at 20 °C of PUs made with polycarbonates of different molecular weights has been assessed.
The increase of the molecular weight of the polycarbonate polyol decreases the hard segments (HS) content of the PUs from 37 wt.% to 13 wt.% (
Table 1) [
32]. This trend can be expected by considering that the number of carbonate of 1,6 hexanediol units in the soft segments of the PUs increases from 3 to 13 by increasing the molecular weight of the polyol from 500 Da to 2000 Da respectively (
Table 1). As a consequence, the mobility of the PU chains would be reduced by increasing the length of the polycarbonate soft segments and their self-healing ability too [
35]. At the same time, the number of carbonate groups in the soft segments increases from 5 to 30 by increasing the molecular weight of the polycarbonate from 500 Da to 2000 Da respectively (
Table 1). Thus, the number of free carbonate groups and carbonate-carbonate interactions between the soft segments will change depending on their length, so different self-healing abilities of the PUs can be expected.
The segmental motion of the PUs is tightly related to their molecular weights and HS contents [
32,
35]. The average in number molecular weights (M
n) of the PUs increase by increasing the molecular weight of the polycarbonate (
Table 2) and they are within the range of other self-healing PUs [
35], so they should exhibit an adequate segmental mobility. However, the average in weight molecular weights of all PUs are within the same range. On the other hand, the M
z values and polydispersity indexes of the PUs decrease by increasing the molecular weight of the polycarbonate (
Table 2), so YC2000 has the narrowest molecular weight distribution and YC500 the widest one.
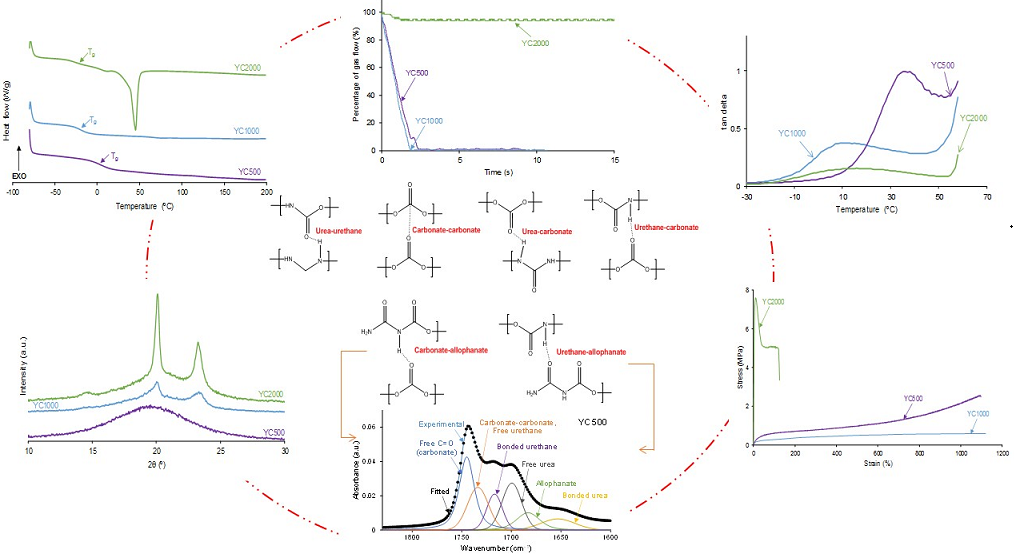
The self-healing of the PUs made with polycarbonates can be affected by their different HS content, distinct molecular weights and length of the polycarbonate soft segments, and the number of free carbonate groups and carbonate-carbonate interactions between the soft segments. The kinetics of self-healing at 20 °C of the PUs are shown in
Figure 1 in which is evidenced that YC500 and YC1000 show fast self-healing (2.5 and 1.4 s, respectively) and that YC2000 does not exhibit self-healing at 20 °C. Therefore, the low hard segment content, the increase of the molecular weight and the increase of the number of carbonate groups/carbonate of 1,6 hexanediol units do not favor PU self-healing. On the other hand,
Figure 1 shows that the self-healing time of YC500 is lower (2.5 s) than the one of YC1000 (1.4 s). Considering the differences in HS content, the length of the polycarbonate soft segments and the molecular weights, faster kinetics and short self-healing time can be expected in YC500 than in YC1000.
Figure 1.
Kinetics of self-healing at 20 °C of PUs made with polycarbonates of different molecular weights.
Figure 1.
Kinetics of self-healing at 20 °C of PUs made with polycarbonates of different molecular weights.
According to
Figure 1, the kinetics of self-healing of YC500 and YC1000 are fast. The curves of
Figure 1 were adjusted to first order kinetics - Eq. 1:
The plot of the gas flow vs. time allows the calculation of the rate constants of the kinetics of self-healing at 20 °C of YC500 and YC1000 – Eq. 2:
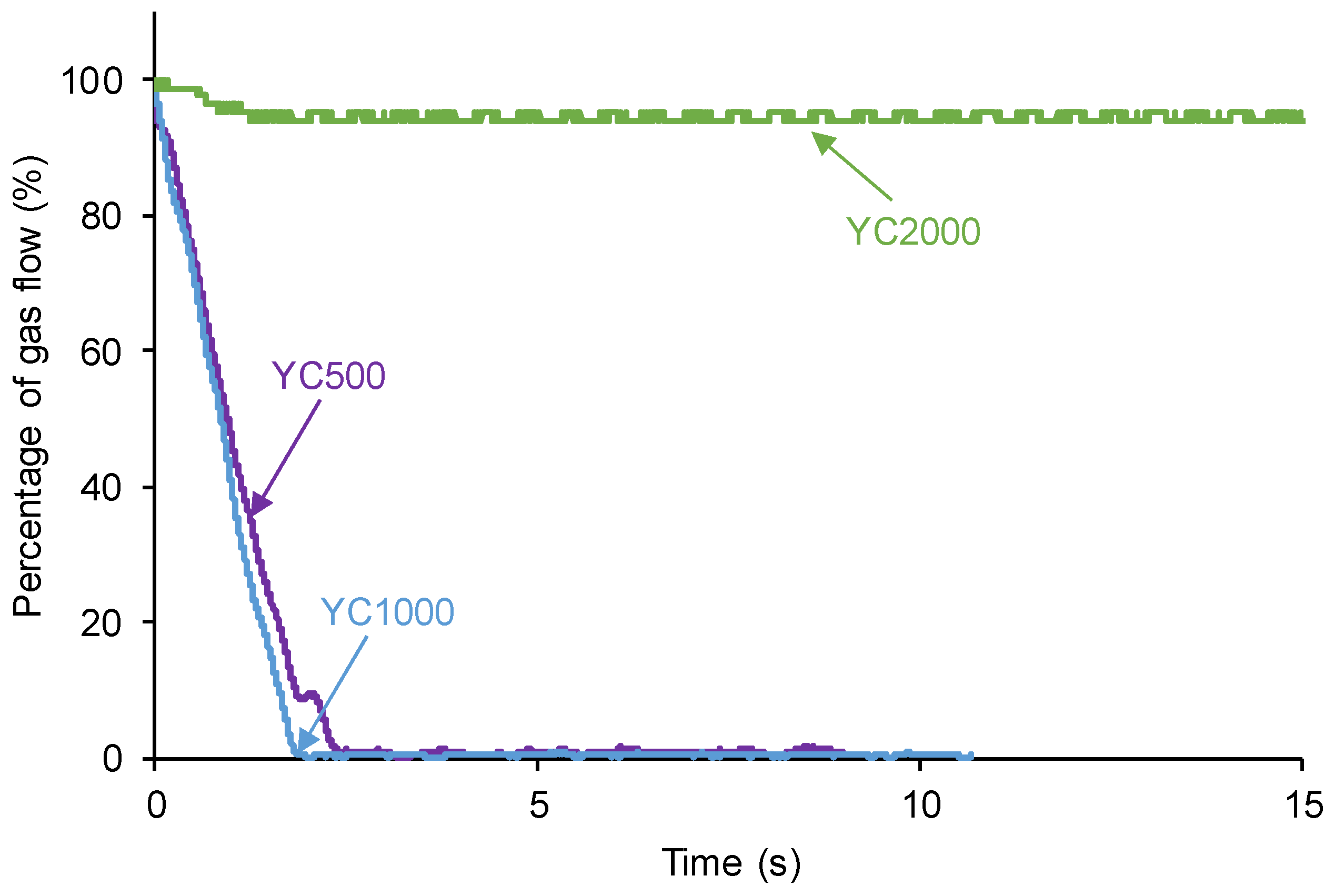
Figure 2 shows that the plots of ln[gas flow] vs time adjust reasonably the kinetics of self-healing of YC500 and YC1000 to a first order (R
2 = 0.97-0.98). The rate constant of YC500 is 0.62 and the one of YC1000 is 0.67. Therefore, the kinetics of self-healing are somewhat similar but slightly higher in YC1000.
Figure 2.
Adjustment of the kinetics of self-healing at 20 °C of YC500 and YC1000 to fist order kinetics.
Figure 2.
Adjustment of the kinetics of self-healing at 20 °C of YC500 and YC1000 to fist order kinetics.
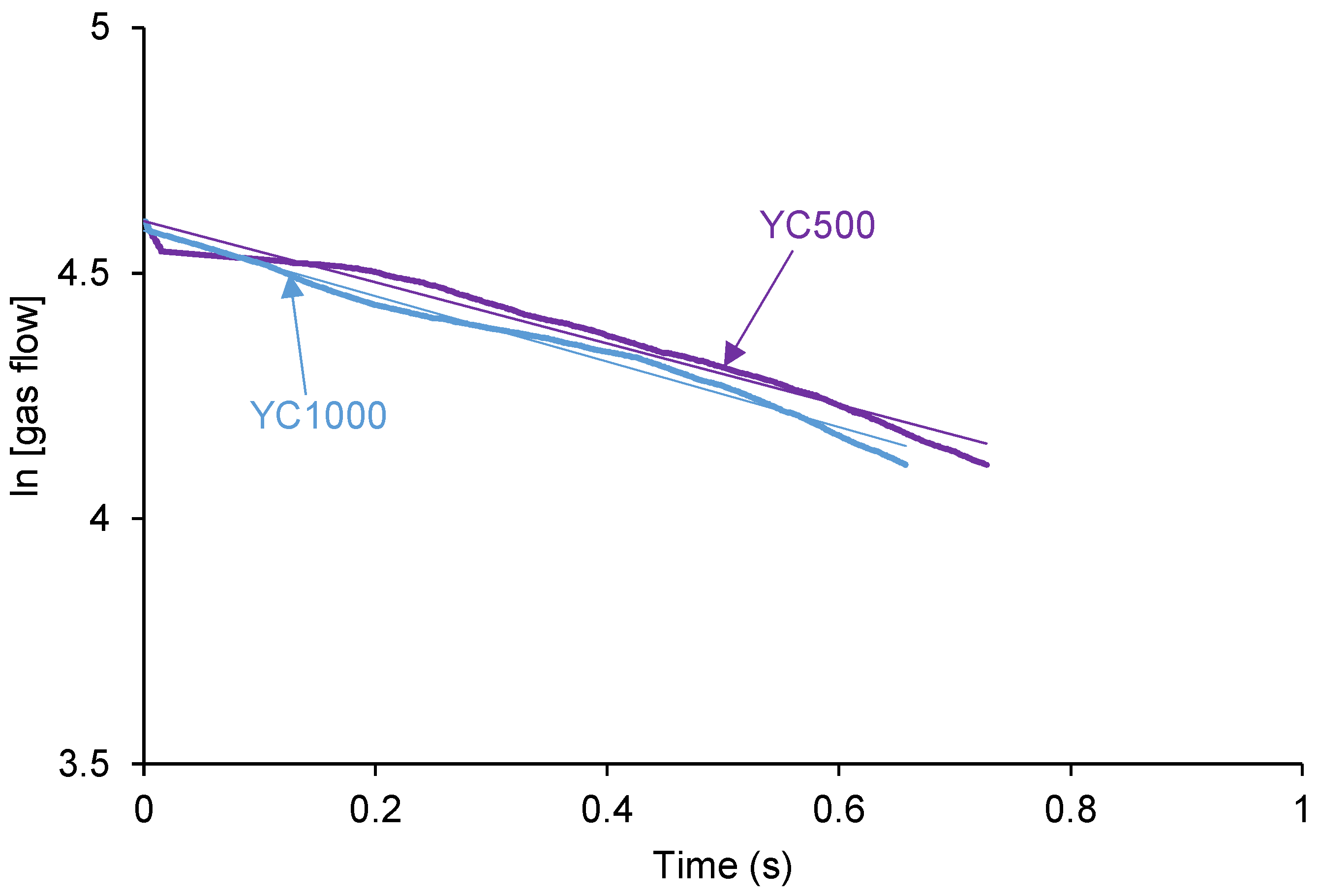
The self-healing of the PUs can be affected by the existence of tack - i.e., a stitch that holds two pieces of material together temporarily. The probe tack is the most common procedure to determine the tack of the polymers.
Figure 3 shows that YC500 is the only exhibiting tack (100 kPa) which is located at the maximum in the stress-strain curve. Tack is caused by the mobility of the short chains in YC500 that will contribute to delay the self-healing because the polymeric chains will tend to displace opposing to the interactions among them. Thus, the self-healing time is longer in YC500 than in YC1000. However, some additional factors should influence the self-healing at 20 °C of YC500 and YC1000.
Figure 3.
Variation of the stress at 20 °C as a function of the strain for PUs made with polycarbonates of different molecular weights. Probe tack test.
Figure 3.
Variation of the stress at 20 °C as a function of the strain for PUs made with polycarbonates of different molecular weights. Probe tack test.
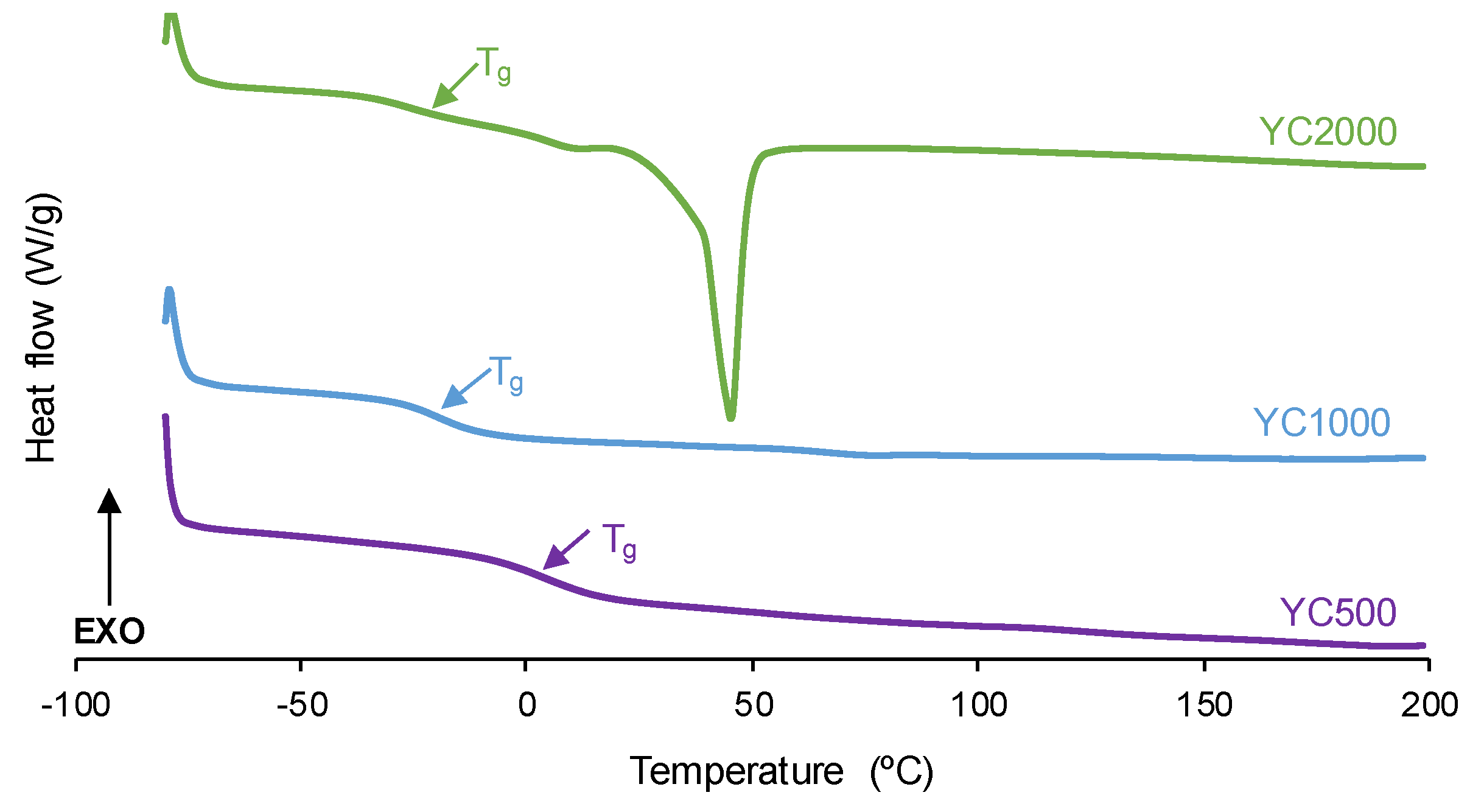
The degree of micro-phase separation determines the self-healing of the PUs and it was assessed by DSC. The DSC curves of the first heating run of the PUs show the glass transition temperature (T
g) of the soft segments which decreases by increasing the polycarbonate molecular weight (
Figure 4,
Table 3). The decrease in T
g is expected by decreasing the hard segment content in the PUs [
32]. The interactions between the soft segments determine the heat capacity at constant pressure (∆c
p) values which decrease by increasing the polycarbonate molecular weight. Thus, the interactions between the polycarbonate soft segments are stronger in YC500 and less important in YC2000. On the other hand, only YC2000 shows a small cold crystallization at 20 °C and the melting of the soft segments at 45 °C (melting enthalpy: 23 J/g), so the movement of the polymeric chains is restricted. Consequently, it cannot be expected that YC2000 exhibits self-healing at 20 °C.
Figure 4.
DSC curves of PUs made with polycarbonates of different molecular weights. First heating run.
Figure 4.
DSC curves of PUs made with polycarbonates of different molecular weights. First heating run.
Once the thermal history of the PUs is removed by cooling down to -80 °C, a second DSC heating run was carried out (
Figure S1 of Supplementary Materials File). The DSC curves show the glass transitions of the soft segments (T
ss) and the hard segments (T
hs). Whereas T
hs values are somewhat similar in all PUs (236-240 °C), the T
ss values decrease by increasing the polycarbonate molecular weight (
Table 4). Thus, considering the differences between T
ss and T
hs values, YC2000 shows the greatest micro-phase separation and YC500 the lowest. The lower micro-phase separation favors the self-healing ability at 20 °C of the PUs [
36], so the self-healing will be favored in the PUs made with polycarbonates of lower molecular weights (YC500 and YC1000).
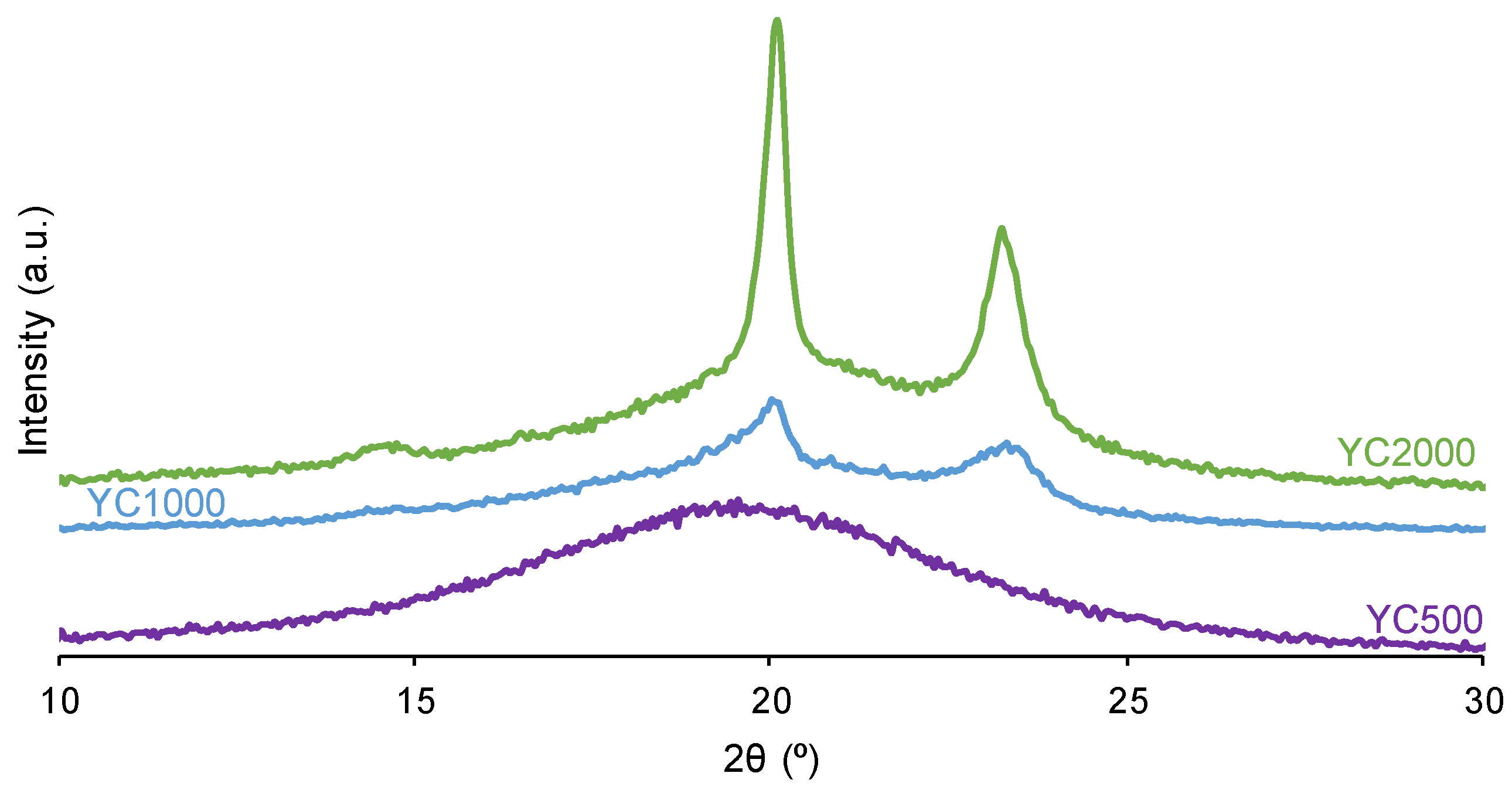
The crystallinity of the soft segments and the micro-phase separation affect the PU self-healing ability.
Figure 5 shows that the X-ray diffractograms of YC1000 and YC2000 exhibit the characteristic diffraction peaks at 2θ = 20.0-20.1° and 2θ = 23.3° due to interactions between polycarbonate soft segments [
35,
37]. The intensities of the two diffraction peaks are significantly higher in YC2000 than in YC1000 (
Figure 5,
Table S1 of Supplementary Materials File), i.e., YC2000 exhibits higher crystallinity. On the other hand, the X-ray diffractogram of YC500 shows a wide shallow peak typical of an amorphous structure and the intensity of the peak is higher than the corresponding one in YC1000. The absence of crystallinity in YC500 contributes to its self-healing and the important crystallinity of YC2000 favors poor self-healing. Because YC1000 shows the shorter self-healing time among the three PUs and exhibits some crystallinity, the interactions between the polycarbonate soft segments could play an important role in its self-healing ability. In fact, the soft segments of YC1000 have 13 carbonate groups and 6 carbonate of 1,6 hexanediol units, so the creation of net carbonate-carbonate interactions is feasible. However, the soft segments of YC500 are short as they have 3 carbonate of 1,6 hexanediol units and 5 carbonate groups only, so they should have less number of carbonate-carbonate interactions that are needed for presenting dynamic non-covalent interactions between the polycarbonate soft segments (proposed mechanism of self-healing). Therefore, the higher number of carbonate-carbonate interactions in YC1000 together with its reduced crystallinity may contribute to its faster self-healing as compared to the one of YC500.
Figure 5.
X-ray diffractograms of PUs made with polycarbonates of different molecular weights.
Figure 5.
X-ray diffractograms of PUs made with polycarbonates of different molecular weights.
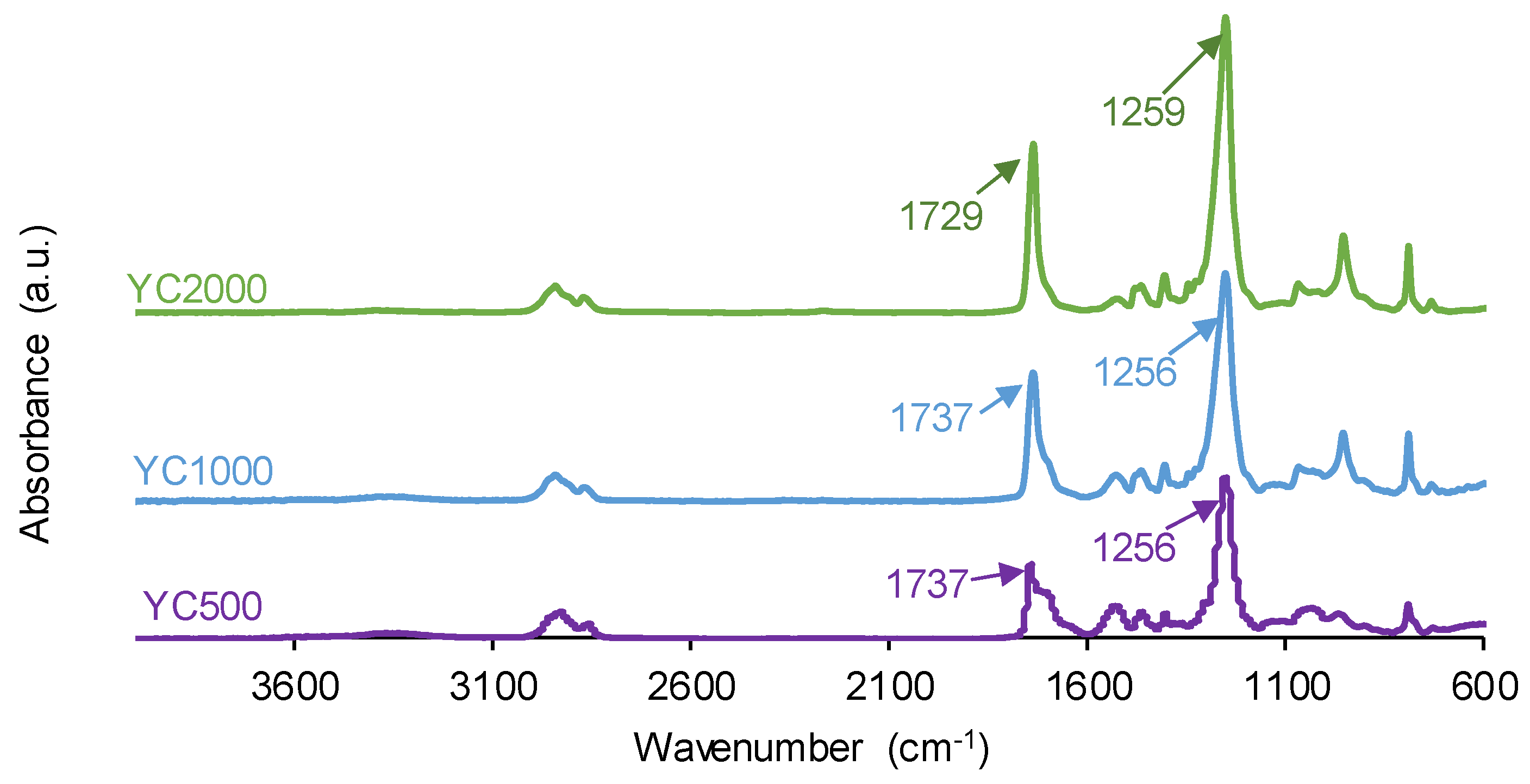
The chemistries of the PUs synthesized with polycarbonates of different molecular weights were assessed by ATR-IR spectroscopy. The ATR-IR spectra of all PUs show the same absorption bands (
Figure 6), mainly N-H stretching band at 3351-3384 cm
−1, C=O stretching band of urethane and urea groups at 1729–1737 cm
−1, C-H stretching bands at 2930-2384 and 2850-2866 cm
−1, and C-O stretching of carbonate groups at 1256-1259 cm
−1. The increase of the molecular weight of the polycarbonate displaces the carbonyl band in the ATR-IR spectra of the PUs to lower wavenumber; furthermore, the shape of the carbonyl band varies depending on the molecular weight of the polycarbonate. These changes can be ascribed to the existence of different free and bonded (hydrogen bond, dipole carbonate-carbonate bond) C=O species in the PUs synthesized with polycarbonates of different molecular weights.
Figure 6.
ATR-IR spectra of PUs made with polycarbonates of different molecular weights.
Figure 6.
ATR-IR spectra of PUs made with polycarbonates of different molecular weights.
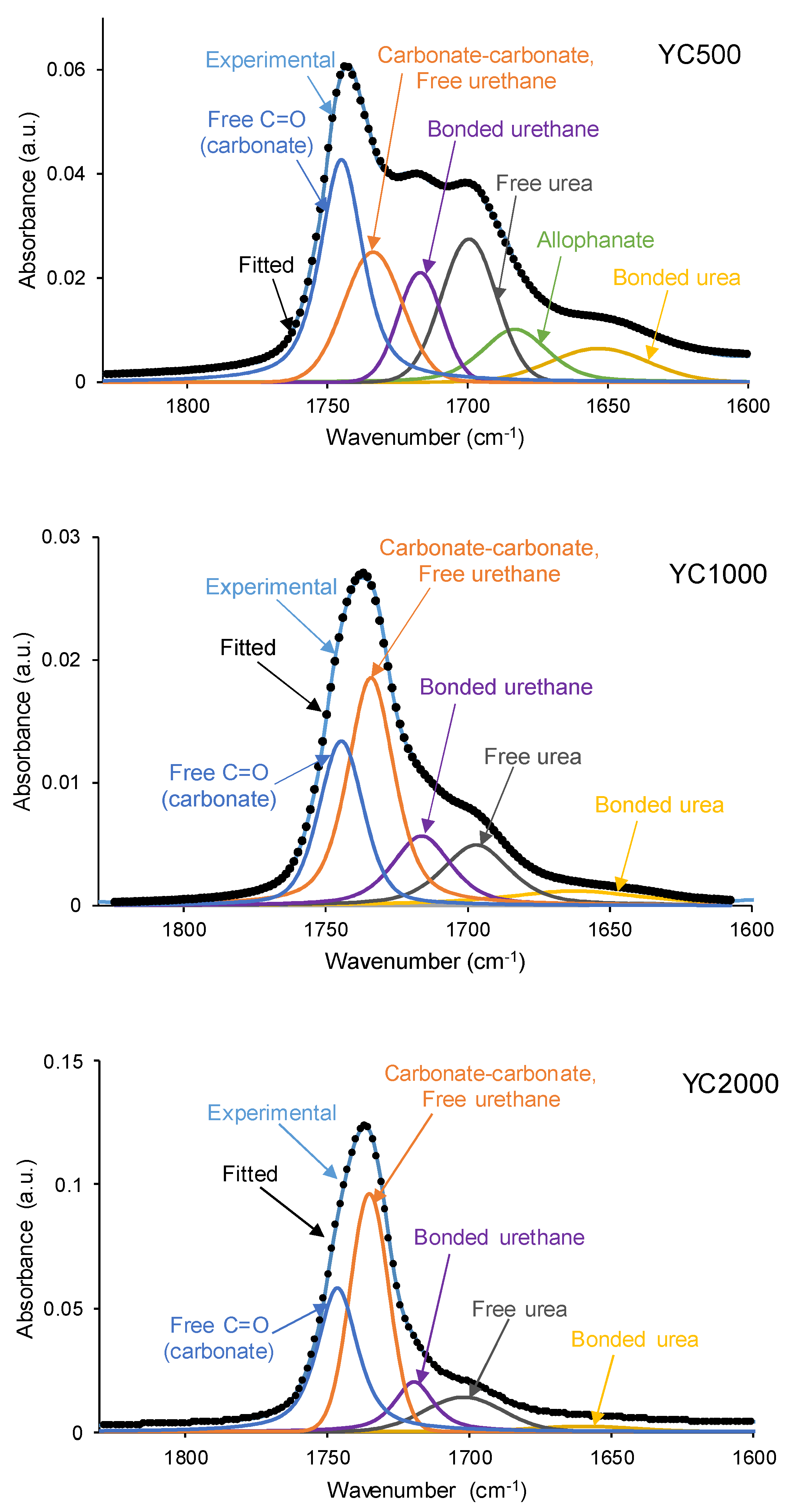
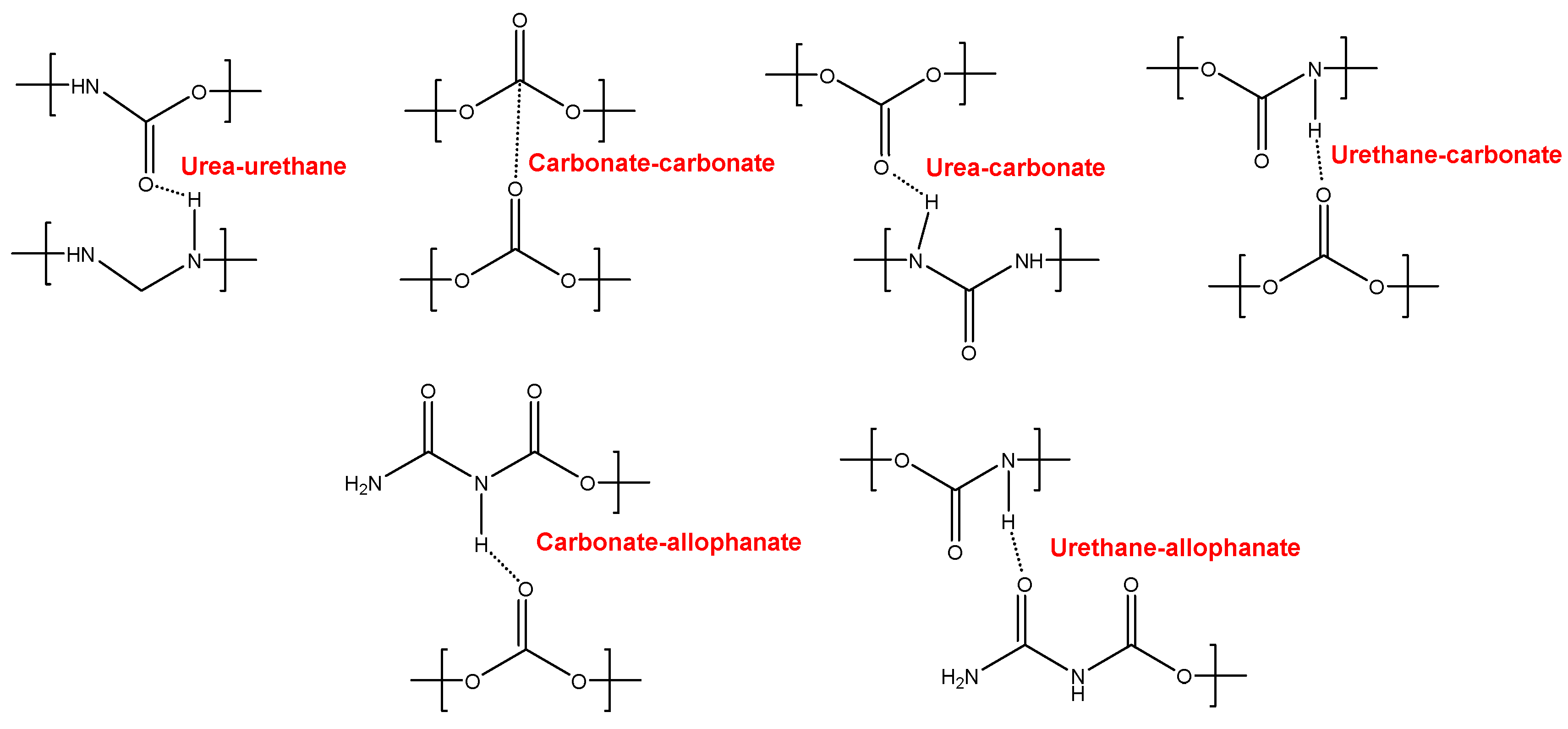
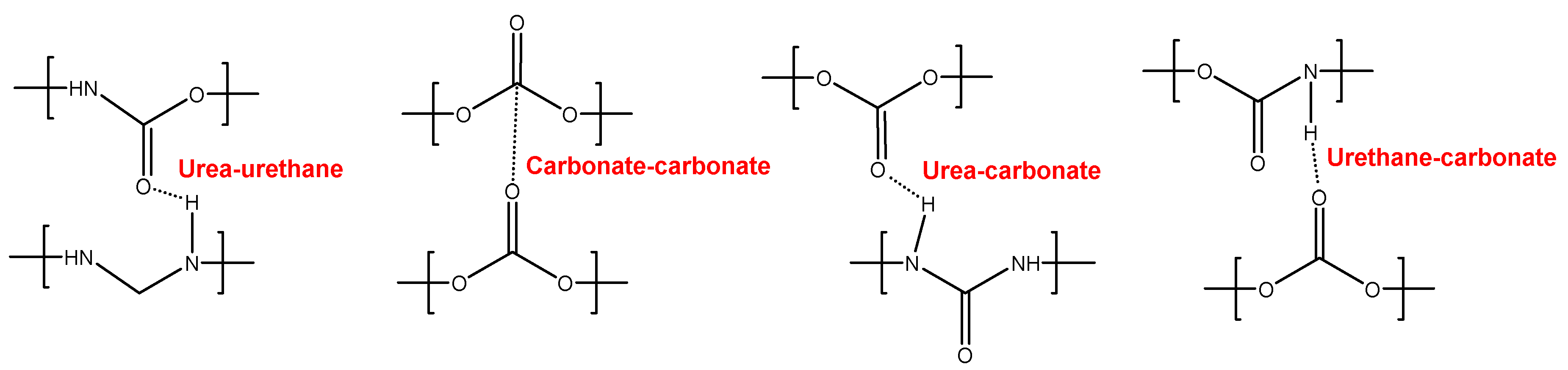
The different C=O species in the PUs were assessed by curve fitting (a Gaussian function was used) of the carbonyl stretching bands in the ATR-IR spectra (
Figure 7). All PUs show the existence of five different C=O species (
Table 5) and, additionally, only YC500 exhibits allophanate groups at 1685 cm
-1 [
39]. The allophanate groups are produced by reaction of urethane and isocyanate and only appear in YC500 because of the short polyol chain. The existence of allophanate groups inhibits the segmental motion of the polymer chains and this can be associated to longer self-healing time in YC500. All PUs have 24-34 % free carbonate groups at 1742-1746 cm
-1 [
40] and YC1000 has the lowest percentage. Furthermore, the curve fitting of all PUs shows free urethane and dipole carbonate-carbonate interactions at 1732-1733 cm
-1 [
40,
41], and its percentage increases from YC500 to YC1000. The hydrogen-bonded urethane species appear at 1713-1722 cm
-1 and its percentage is somewhat similar in all PUs. Additionally, the free and hydrogen bonded urea groups appear at 1695-1697 and 1650-1654 cm
-1 respectively [
42], and their percentages decrease by increasing the molecular weight of the polycarbonate. Therefore, YC500 shows a significant percentage of free carbonate and free and hydrogen bonded urea groups, a low percentage of urethane groups and is the only having allophanate species. (
Table 5). On the other hand, YC1000 shows a low percentage of free carbonate and significant percentages of free urethane, dipole carbonate-carbonate interactions, and bonded urea species (
Table 5). Considering the different C=O species, YC500 shows allophanate species that increase the interactions between its polymeric chains whereas YC1000 has a lower percentage of free carbonate and a higher percentage of dipole carbonate-carbonate interactions, and these different chemical species may contribute to the longer self-healing time of YC500.
Figure 7.
Curve fitting of the carbonyl stretching region of the ATR-IR spectra of PUs made with polycarbonates of different molecular weights.
Figure 7.
Curve fitting of the carbonyl stretching region of the ATR-IR spectra of PUs made with polycarbonates of different molecular weights.
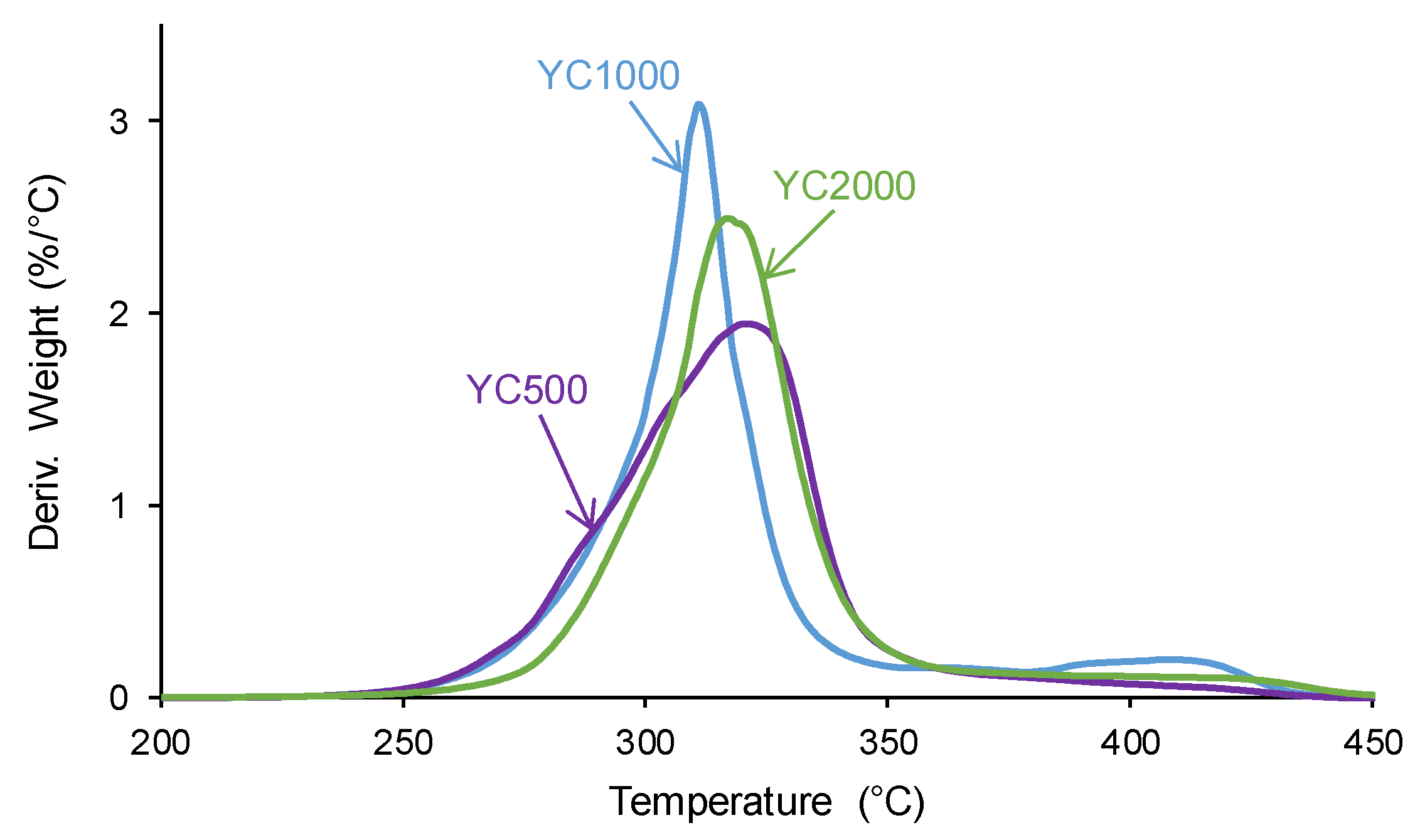
The structural differences between the PUs were also evidenced by TGA. The TGA curves of the PUs (
Figure S2 of Supplementary Material File) show a different trend in YC1000 with respect to the other PUs. In fact, below 300 °C, similar TGA curves are obtained in YC500 and YC1000, and, above 340 °C, the TGA curve of YC1000 is above the others. The different thermal events can be better distinguished in the derivatives of the TGA curves.
Figure 8 and
Table 6 show that the lowest temperature of maximum thermal decomposition corresponds to YC1000. All PUs shows three thermal decompositions at 285-292 °C (carbonate-carbonate interactions [
36]), 311-321 °C (mixed phase due to carbonyl groups of carbonate-urethane interactions [
37,
43]), and 412-421 °C (hard segments). The DTGA curve of YC500 shows the lowest temperature of thermal degradation of the carbonate-carbonate interactions and the highest temperature and weigth loss of the mixed phase, in agreement with the DSC results. The DTGA curve of YC1000 shows the lowest degradation temperatures of the mixed phase and hard segments, so the mixed phase due to carbonate-urethane interactions decomposes at lower temperature than in YC500, i.e., the movement of the polymeric chains in YC1000 is more favored than in YC500. This can be associated to the higher time of self-healing in YC500. On the other hand, the DTGA curve of YC2000 show the highest thermal degradation temperatures.
Figure 8.
Derivatives of the TGA curves of PUs made with polycarbonates of different molecular weights.
Figure 8.
Derivatives of the TGA curves of PUs made with polycarbonates of different molecular weights.
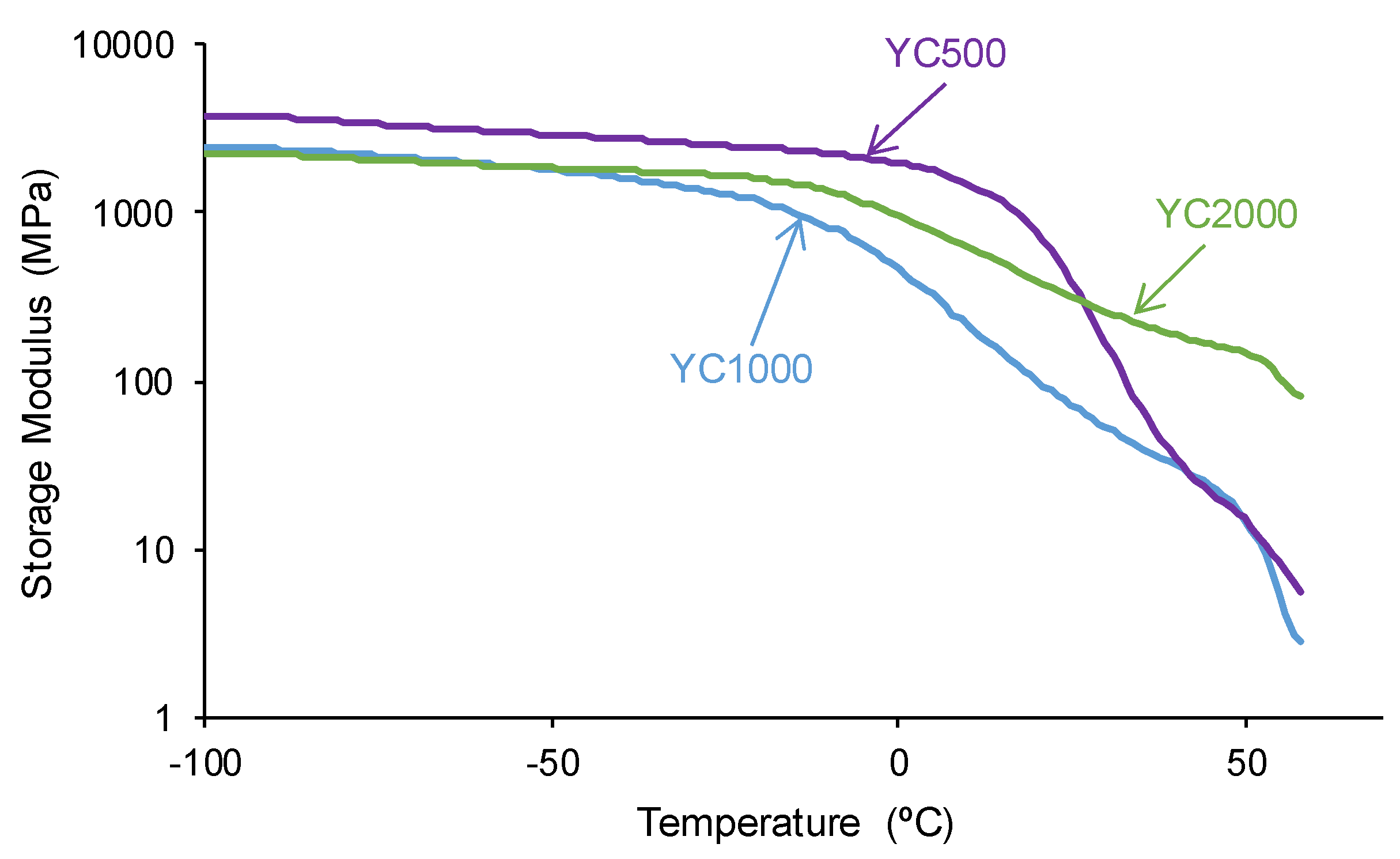
The structural differences between the PUs made with polycarbonates of different molecular weight affect their viscoelastic properties. The variation of the storage modulus as a function of the temperature (
Figure 9) shows higher storage moduli in the glassy region in YC500 than in YC1000 and YC2000. Once the glass transition is reached, a decline of the storage moduli is produced and the decrease of the storage moduli is more marked in YC500 and YC1000 and less pronounced in YC2000. Thus, the movement of the polymeric chains is more favored in YC500 and YC1000 than in YC2000, and this may contribute to the existence of self-healing. This in in agreement with the results provided by DSC and TGA.
Figure 9.
Variation of the storage (E´) moduli of PUs made with polycarbonates of different molecular weights as a function of the temperature. DMA experiments.
Figure 9.
Variation of the storage (E´) moduli of PUs made with polycarbonates of different molecular weights as a function of the temperature. DMA experiments.
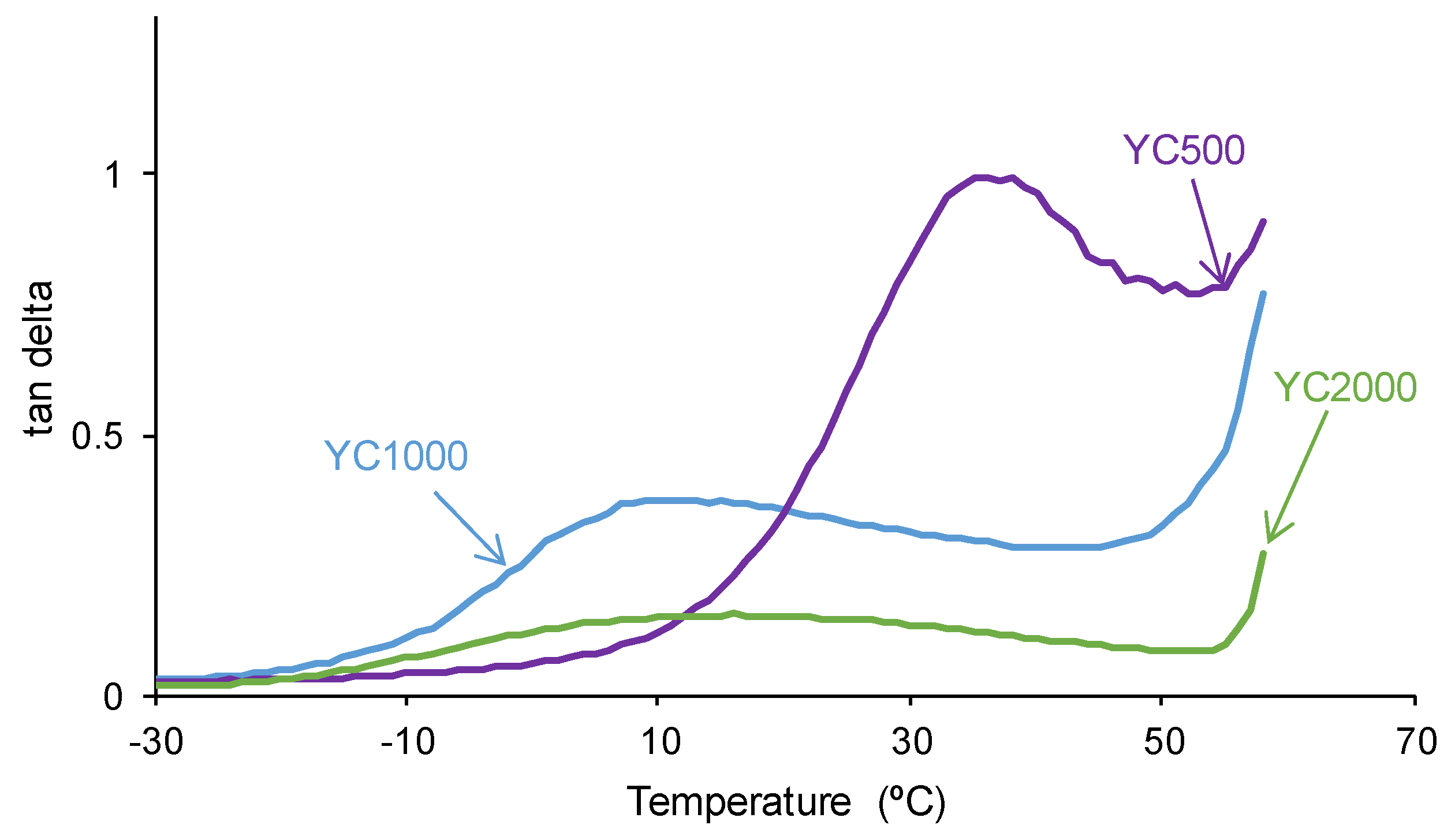
The tan delta vs temperature plots of the PUs (
Figure 10) show one structural relaxation only. YC500 shows the highest tan delta value and temperature of the structural relaxation, indicating a marked rheological viscous regime and, therefore, good chains mobility (
Table 7). YC1000 shows an intermediate tan delta value and the lowest temperature of the structural relaxation, and the viscous rheological contribution is important. However, YC2000 shows a low tan delta value and dominant elastic rheological regime that supports the absence of self-healing.
Figure 10.
Variation of tan delta as a function of the temperature for PUs made with polycarbonates of different molecular weights as a function of the temperature. DMA experiments.
Figure 10.
Variation of tan delta as a function of the temperature for PUs made with polycarbonates of different molecular weights as a function of the temperature. DMA experiments.
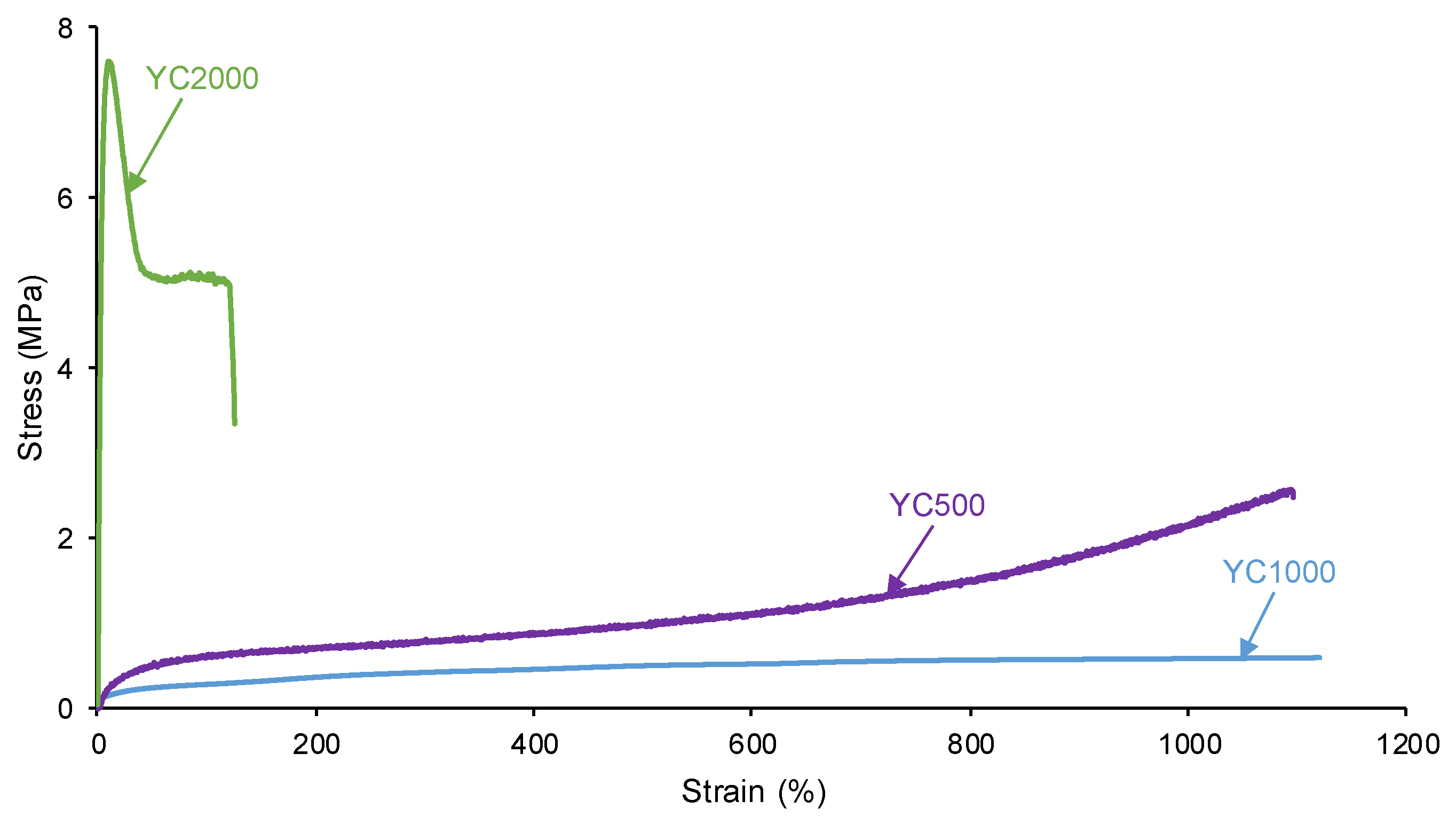
The self-healing PUs should exhibit adequate mechanical properties. The stress-strain curves of the PUs are different (
Figure 11). YC2000 shows the stress-strain curve of a stiff thermoplastic material, i.e., high tensile strength (5.0 MPa) and low elongation-at-break (119 %) (
Table 8). However, YC500 and YC1000 show typical stress-strain curves of elastomeric materials. Interestingly, the mechanical properties of YC1000 are lower than the ones of YC500 and this can be associated to higher mobility of the polymeric chains.
Figure 11.
Stress-strain curves of PUs made with polycarbonates of different molecular weights.
Figure 11.
Stress-strain curves of PUs made with polycarbonates of different molecular weights.