Submitted:
21 August 2024
Posted:
22 August 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Methodology
Analysis and Results
Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Andraszek, K.; Banaszewska, D.; Szeleszczuk, O.; Kuchta-Gładysz, M.; Grzesiakowska, A. Morphometric Characteristics of the Spermatozoa of Blue Fox (Alopex lagopus) and Silver Fox (Vulpes vulpes). Animals 2020, 10, 1927. [Google Scholar] [CrossRef] [PubMed]
- Awad, M. Effect of some permeating cryoprotectants on CASA motility results in cryopreserved bull spermatozoa. Anim. Reprod. Sci. 2011, 123, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Banaszewska, D.; Andraszek, K.; Czubaszek, M.; Biesiada–Drzazga, B. The effect of selected staining techniques on bull sperm morphometry. Anim. Reprod. Sci. 2015, 159, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Barquero, V.; Roldan, E.R.S.; Soler, C.; Yániz, J.L.; Camacho, M.; Valverde, A. Predictive Capacity of Boar Sperm Morphometry and Morphometric Sub-Populations on Reproductive Success after Artificial Insemination. Animals 2021, 11, 920. [Google Scholar] [CrossRef]
- Barth, A.D. Review: The use of bull breeding soundness evaluation to identify subfertile and infertile bulls. Animal 2018, 12, s158–s164. [Google Scholar] [CrossRef]
- Beletti, M.E.; Costa, L.d.F.; Viana, M.P. A comparison of morphometric characteristics of sperm from fertile Bos taurus and Bos indicus bulls in Brazil. Anim. Reprod. Sci. 2005, 85, 105–116. [Google Scholar] [CrossRef]
- Boersma, A.; Braun, J.; Stolla, R. Influence of Random Factors and Two Different Staining Procedures on Computer-assisted Sperm Head Morphometry in Bulls. Reprod. Domest. Anim. 1999, 34, 77–82. [Google Scholar] [CrossRef]
- Imbachí, F.B.; Zalazar, L.; Pastore, J.I.; Nicolli, A.; Ledesma, A.; Hozbor, F.A.; Cesari, A.; Ballarin, V. Clustering and classification software for sperm subpopulation analysis. Comput. Methods Biomech. Biomed. Eng. Imaging Vis. 2022, 10, 585–598. [Google Scholar] [CrossRef]
- Caldeira, C.; Hernández-Ibánez, S.; Vendrell, A.; Valverde, A.; García-Molina, A.; Gallego, V.; Asturiano, J.F.; Soler, C. Characterisation of European eel (Anguilla anguilla) spermatozoa morphometry using Trumorph tool in fixed and non-fixed samples. Aquaculture 2022, 553. [Google Scholar] [CrossRef]
- Castellini, C.; Bosco, A.D.; Ruggeri, S.; Collodel, G. What is the best frame rate for evaluation of sperm motility in different species by computer-assisted sperm analysis? Fertil. Steril. 2011, 96, 24–27. [Google Scholar] [CrossRef]
- Cheah, Y.; Yang, W. Functions of essential nutrition for high quality spermatogenesis. Adv. Biosci. Biotechnol. 2011, 02, 182–197. [Google Scholar] [CrossRef]
- Franken, D.R. How accurate is sperm morphology as an indicator of sperm function? Andrologia 2015, 47, 720–723. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, M.T.; Smith, D.J.; Kirkman-Brown, J.C. CASA: tracking the past and plotting the future. Reprod. Fertil. Dev. 2018, 30, 867–874. [Google Scholar] [CrossRef]
- García-Herreros, M.; Aparicio, I.M.; Barón, F.J.; García-Marín, L.J.; Gil, M.C. Standardization of sample preparation, staining and sampling methods for automated sperm head morphometry analysis of boar spermatozoa. Int. J. Androl. 2006, 29, 553–563. [Google Scholar] [CrossRef]
- García-Molina, A.; Navarro, N.; Cerveró, C.; Sadeghi, S.; Valverde, A.; Roldan, E.R.; Bompart, D.; Garrido, N.; Soler, C. Effect of incubation and analysis temperatures on sperm kinematics and morphometrics during human semen analysis. Rev. Int. De Androl. 2022, 21, 100350. [Google Scholar] [CrossRef]
- García-Molina, A.; Navarro, N.; Cerveró, C.; Sadeghi, S.; Valverde, A.; Roldan, E.R.; Bompart, D.; Garrido, N.; Soler, C. Effect of incubation and analysis temperatures on sperm kinematics and morphometrics during human semen analysis. Rev. Int. De Androl. 2023, 21, 100350. [Google Scholar] [CrossRef] [PubMed]
- Giaretta, E.; Munerato, M.; Yeste, M.; Galeati, G.; Spinaci, M.; Tamanini, C.; Mari, G.; Bucci, D. Implementing an open-access CASA software for the assessment of stallion sperm motility: Relationship with other sperm quality parameters. Anim. Reprod. Sci. 2017, 176, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Gravance, C.; Casey, M.; Casey, P. Pre-freeze bull sperm head morphometry related to post-thaw fertility. Anim. Reprod. Sci. 2009, 114, 81–88. [Google Scholar] [CrossRef]
- Holt, W.; Van Look, K. Concepts in sperm heterogeneity, sperm selection and sperm competition as biological foundations for laboratory tests of semen quality. Reproduction 2004, 127, 527–535. [Google Scholar] [CrossRef]
- Hook, K.A.; Fisher, H.S. Methodological considerations for examining the relationship between sperm morphology and motility. Mol. Reprod. Dev. 2020, 87, 633–649. [Google Scholar] [CrossRef]
- Kaiser, H.F. The varimax criterion for analytic rotation in factor analysis. Psychometrika 1958, 23, 187–200. [Google Scholar] [CrossRef]
- Gentle, J.E. Finding Groups in Data: An Introduction to Cluster Analysis. Biometrics 1991, 47, 788. [Google Scholar] [CrossRef]
- Kramer, E.M.; Enelamah, J.; Fang, H.; Tayjasanant, P.A. Karyotype depends on sperm head morphology in some amniote groups. Front. Genet. 2024, 15, 1396530. [Google Scholar] [CrossRef]
- Parameshwarappa, M.A.; Veerasamy, S.; Pandarathil, R.V.; Tulasiramu, S.S.; Madiajagan, B.; Govindan, K.; Raghavendra, B. Resilient capacity of cattle to environmental challenges – An updated review. J. Anim. Behav. Biometeorol. 2019, 7, 104–118. [Google Scholar] [CrossRef]
- Garde, J.; Maroto-Morales, A.; García-Álvarez, O.; Ramón, M.; Martínez-Pastor, F.; Fernández-Santos, M.; Soler, A. Current status and potential of morphometric sperm analysis. Asian J. Androl. 2016, 18, 863–870. [Google Scholar] [CrossRef]
- Martínez-Fresneda, L.; O'Brien, E.; Velázquez, R.; Toledano-Díaz, A.; Martínez-Cáceres, C.M.; Tesfaye, D.; Schellander, K.; García-Vázquez, F.A.; Santiago-Moreno, J. Seasonal variation in sperm freezability associated with changes in testicular germinal epithelium in domestic (Ovis aries) and wild (Ovis musimon) sheep. Reprod. Fertil. Dev. 2019, 31, 1545–1557. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Pastor, F. What is the importance of sperm subpopulations? Anim. Reprod. Sci. 2021, 246, 106844. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, S. T. , & De Jonge, C. J. (2018). CASA—Computer-Aided Sperm Analysis. Encyclopedia of Reproduction, 59-63. [CrossRef]
- Yániz, J.; Palacin, I.; Santolaria, P.; Alquezar-Baeta, C.; Soler, C.; Silvestre, M. Relationship of sperm plasma membrane and acrosomal integrities with sperm morphometry in Bos taurus. Asian J. Androl. 2020, 22, 578–582. [Google Scholar] [CrossRef]
- Petrunkina, A.M.; Waberski, D.; Günzel-Apel, A.R.; Töpfer-Petersen, E. Determinants of sperm quality and fertility in domestic species. Reproduction 2007, 134, 3–17. [Google Scholar] [CrossRef]
- Ramón, M.; Jiménez-Rabadán, P.; García-Álvarez, O.; Maroto-Morales, A.; Soler, A.; Santos, F.; Guzmán, P.; Garde, J. Understanding Sperm Heterogeneity: Biological and Practical Implications. Reprod. Domest. Anim. 2014, 49, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Guillén, J.; González, D.; Garde, J.; Esteso, M.; Santos, F.; Rodríguez-Gíl, J.; Madrid-Bury, N.; Quintero-Moreno, A. Effects of Cryopreservation on Bull Spermatozoa Distribution in Morphometrically Distinct Subpopulations. Reprod. Domest. Anim. 2007, 42, 354–357. [Google Scholar] [CrossRef]
- Sampaio, W.V.; Oliveira, K.G.; Leão, D.L.; Caldas-Bussiere, M.C.; Queiroz, H.L.; Paim, F.P.; Santos, R.R.; Domingues, S.F. Morphologic analysis of sperm from two neotropical primate species: comparisons between the squirrel monkeysSaimiri collinsiandSaimiri vanzolinii. Zygote 2017, 25, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Santolaria, P.; Vicente-Fiel, S.; Palacín, I.; Fantova, E.; Blasco, M.; Silvestre, M.; Yániz, J. Predictive capacity of sperm quality parameters and sperm subpopulations on field fertility after artificial insemination in sheep. Anim. Reprod. Sci. 2015, 163, 82–88. [Google Scholar] [CrossRef]
- Saranholi, D.A.C.; de Paula, R.R.; Pytilak, E.; Afonso, F.; Canela, L.F.; de Almeida, A.B.M.; Hidalgo, M.M.T.; Martins, M.I.M.; Blaschi, W.; Barreiros, T.R.R. Comparison of seminal characteristics of Aberdeen Angus, Holstein and Nelore bulls before and after cryopreservation. Res. Soc. Dev. 2021, 10. [Google Scholar] [CrossRef]
- Soler, C.; Alambiaga, A.; Martí, M.; García-Molina, A.; Valverde, A.; Contell, J.; Campos, M. Dog sperm head morphometry: its diversity and evolution. Asian J. Androl. 2017, 19, 149–153. [Google Scholar] [CrossRef]
- Soler, C.; Contell, J.; Bori, L.; Sancho, M.; García-Molina, A.; Valverde, A.; Segarvall, J. Sperm kinematic, head morphometric and kinetic-morphometric subpopulations in the blue fox (Alopex lagopus). Asian J. Androl. 2017, 19, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Soler, C.; Cooper, T.; Valverde, A.; Yániz, J. Afterword to Sperm morphometrics today and tomorrow special issue in Asian Journal of Andrology. Asian J. Androl. 2016, 18, 895–897. [Google Scholar] [CrossRef]
- Soler, C.; Gadea, B.; Soler, A.; Fernández-Santos, M.; Esteso, M.; Núñez, J.; Moreira, P.; Núñez, M.; Gutiérrez, R.; Sancho, M.; et al. Comparison of three different staining methods for the assessment of epididymal red deer sperm morphometry by computerized analysis with ISAS®. Theriogenology 2005, 64, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Soler, C.; García-Molina, A.; Contell, J.; Silvestre, M.; Sancho, M. The Trumorph℗® system: The new univ the morphology of living sperm. Anim. Reprod. Sci. 2015, 158, 1–10. [Google Scholar] [CrossRef]
- Soler, C. , Valverde, A., Bompart, D., Fereidounfar, S., Sancho, M., Yániz, J., Garcia-Molina, A., & Korneenko- Zhilyaev, Yu. A. New methods of semen analysis by casa. Sel'skokhozyaistvennaya Biol. 2017, 52, 232–241. [Google Scholar] [CrossRef]
- Spencer, N.H. Essentials of Multivariate Data Analysis; Taylor & Francis: London, United Kingdom, 2013. [Google Scholar]
- Taylor, J.F.; Schnabel, R.D.; Sutovsky, P. Review: Genomics of bull fertility. Animal 2018, 12, S172–S183. [Google Scholar] [CrossRef] [PubMed]
- Terán, E.; Azcona, F.; Ramón, M.; Molina, A.; Dorado, J.; Hidalgo, M.; Ross, P.; Goszczynski, D.; Demyda-Peyrás, S. Sperm morphometry is affected by increased inbreeding in the Retinta cattle breed: A molecular approach. Mol. Reprod. Dev. 2021, 88, 416–426. [Google Scholar] [CrossRef]
- Thurston, L.M.; Siggins, K.; Mileham, A.J.; Watson, P.F.; Holt, W.V. Identification of Amplified Restriction Fragment Length Polymorphism Markers Linked to Genes Controlling Boar Sperm Viability Following Cryopreservation1. Biol. Reprod. 2002, 66, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Toner, J.P.; Mossad, H.; Grow, D.R.; Morshedi, M.; Swanson, R.; Oehninger, S. Value of sperm morphology assessed by strict criteria for prediction of the outcome of artificial (intrauterine) insemination. Andrologia 1995, 27, 143–148. [Google Scholar] [CrossRef]
- Soler, C.; Valverde, A.; Arenán, H.; Sancho, M.; Contell, J.; Yániz, J.; Fernández, A. Morphometry and subpopulation structure of Holstein bull spermatozoa: variations in ejaculates and cryopreservation straws. Asian J. Androl. 2016, 18, 851–857. [Google Scholar] [CrossRef]
- Valverde, A.; Madrigal-Valverde, M.; Castro-Morales, O.; Gadea-Rivas, A.; Johnston, S.; Soler, C. Kinematic and head morphometric characterisation of spermatozoa from the Brown Caiman (Caiman crocodilus fuscus). Anim. Reprod. Sci. 2019, 207, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Abarca, A.V.; Castro-Morales, O.; Mdrigal-Valverde, M. Sperm kinematics and morphometric subpopulations analysis with CASA systems: a review. Rev. De Biol. Trop. 2019, 67. [Google Scholar] [CrossRef]
- Vicente-Fiel, S.; Palacín, I.; Santolaria, P.; Hidalgo, C.; Silvestre, M.; Arrebola, F.; Yániz, J. A comparative study of the sperm nuclear morphometry in cattle, goat, sheep, and pigs using a new computer-assisted method (CASMA-F). Theriogenology 2013, 79, 436–442. [Google Scholar] [CrossRef]
- Villaverde-Morcillo, S.; Soler, A.; Esteso, M.; Castaño, C.; Miñano-Berna, A.; Gonzalez, F.; Santiago-Moreno, J. Immature and mature sperm morphometry in fresh and frozen-thawed falcon ejaculates. Theriogenology 2017, 98, 94–100. [Google Scholar] [CrossRef]
- Víquez, L.; Barquero, V.; Soler, C.; Roldan, E.R.; Valverde, A. Kinematic Sub-Populations in Bull Spermatozoa: A Comparison of Classical and Bayesian Approaches. Biology 2020, 9, 138. [Google Scholar] [CrossRef]
- Waberski, D.; Suarez, S.S.; Henning, H. Assessment of sperm motility in livestock: Perspectives based on sperm swimming conditions in vivo. Anim. Reprod. Sci. 2021, 246, 106849. [Google Scholar] [CrossRef] [PubMed]
- Yániz, J.L.; Palacín, I.; Caycho, K.S.; Soler, C.; Silvestre, M.A.; Santolaria, P. Determining the relationship between bull sperm kinematic subpopulations and fluorescence groups using an integrated sperm quality analysis technique. Reprod. Fertil. Dev. 2018, 30, 919–923. [Google Scholar] [CrossRef]
- Yániz, J.L.; Soler, C.; Santolaria, P. Computer assisted sperm morphometry in mammals: A review. Anim. Reprod. Sci. 2015, 156, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yániz, J.; Vicente-Fiel, S.; Soler, C.; Recreo, P.; Carretero, T.; Bono, A.; Berné, J.; Santolaria, P. Comparison of different statistical approaches to evaluate morphometric sperm subpopulations in men. Asian J. Androl. 2016, 18, 819–823. [Google Scholar] [CrossRef] [PubMed]
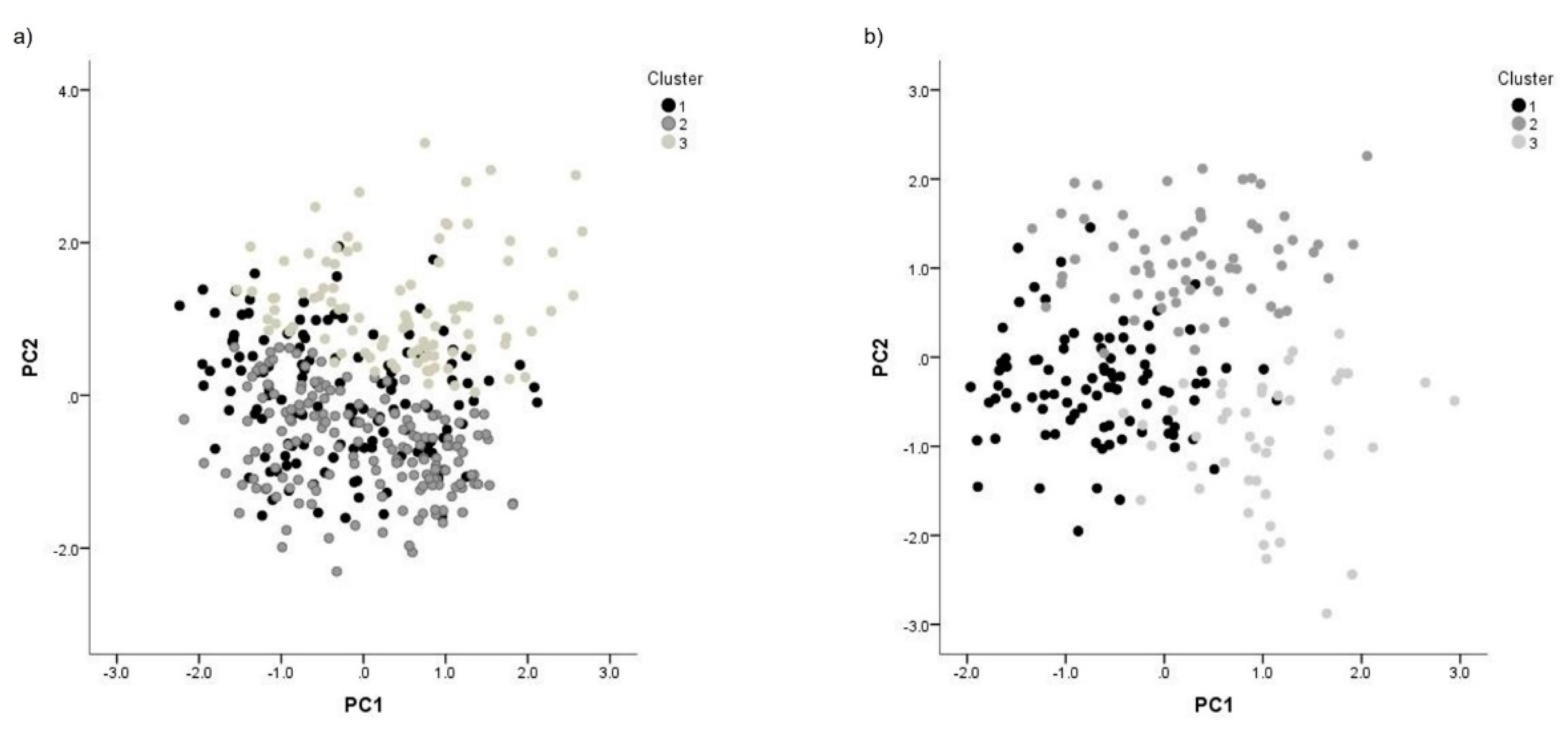
| Variables | Bos indicus | Bos taurus |
|---|---|---|
| Length | 8.969 ± 0.026b | 9.007 ± 0.037a |
| Width | 4.756 ± 0.011b | 4.886 ± 0.016a |
| Area | 37.120 ± 0.124 | 37.792 ± 0.172 |
| Perimeter | 24.767 ± 0.048b | 24.791 ± 0.068a |
| Ellipticity | 1.896 ± 0.006a | 1.846 ± 0.009b |
| Rugosity | 0.761 ± 0.001b | 0.772 ± 0.002a |
| Elongation | 0.307 ± 0.002a | 0.296 ± 0.002b |
| Regularity | 0.901 ± 0.002b | 0.915 ± 0.003a |
| Variable | ||||||
|---|---|---|---|---|---|---|
| Bos indicus | Bos taurus | |||||
| PC1 | PC2 | PC3 | PC1 | PC2 | PC3 | |
| Length | 0.703 | 0.765 | ||||
| Width | 0.898 | |||||
| Area | 0.929 | 0.949 | ||||
| Perimeter | 0.915 | 0.892 | ||||
| Ellipticity | 0.983 | 0.810 | ||||
| Elongation | 0.984 | 0.808 | ||||
| Rugosity | -0.886 | |||||
| Regularity | 0.995 | 0.804 | -0.828 | |||
| Var Exp | 52.772 | 27.114 | 15.503 | 36.317 | 32.628 | 21.853 |
| Variables | Length | Width | Area | Perimeter | Ellipticity | Rugosity | Elongation | Regularity |
|---|---|---|---|---|---|---|---|---|
| B. indicus | ||||||||
| SP1 | 9.110 ±0.031axr | 4.888±0.016ar | 37.272±0.126ar | 24.733±0.050ar | 1.875±0.010br | 0.766±0.002br | 0.301±0.002br | 0.937±0.002ar |
| SP2 | 8.634±0.027bt | 4.678±0.013ct | 35.941±0.109bt | 24.246±0.043bt | 1.856±0.009at | 0.768±0.002ct | 0.297±0.002at | 0.881±0.002bt |
| SP3 | 9.273±0.036cx | 4.821±0.018bx | 39.381±0.147bx | 25.678±0.058cx | 1.934±0.012cx | 0.752±0.002ax | 0.315±0.003cx | 0.889±0.002cx |
| B. taurus | ||||||||
| SP1 | 8.581±0.040as | 4.726±0.023as | 35.232±0.240as | 23.851±0.082as | 1.818±0.009as | 0.778±0.003as | 0.290±0.002as | 0.904±0.003as |
| SP2 | 9.768±0.048bu | 4.829±0.028bu | 39.068±0.288bu | 25.532±0.098bu | 2.025±0.011bu | 0.753±0.003bu | 0.338±0.003bu | 0.949±0.004bu |
| SP3 | 9.009±0.057cy | 5.068±0.034cy | 40.485±0.347cy | 25.815±0.118bx | 1.780±0.013cy | 0.764±0.004cy | 0.280±0.003cy | 0.886±0.005cx |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




