Submitted:
20 August 2024
Posted:
21 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Evans, G. Vaccine injury compensation programs worldwide. Vaccine 1999, 17 (Suppl 3), S25–S35. [Google Scholar] [PubMed]
- Meissner HC, Nair N, Plotkin SA. The National Vaccine Injury Compensation Program: Striking a balance between individual rights and community benefit. JAMA.
- Cook KM, Evans G. The National Vaccine Injury Compensation Program. Pediatrics, 1.
- Wang, PC. Updates on Vaccine Injury Compensation Program in Taiwan and program evaluation. Taiwan Epidemiology Bulletin, 2015, 31, 149–158. [Google Scholar]
- Halabi SF, Omer SB. A global vaccine injury compensation system. JAMA, 2017, 317, 471–2. [Google Scholar]
- Looker C, Kelly H. No-fault compensation following adverse events attributed to vaccination: a review of international programmes. Bull World Health Organ, 2011, 89, 371–8. [Google Scholar]
- Ridgway, D. No-fault vaccine insurance: lessons from the National Vaccine Injury Compensation Program. J Health Polit Policy Law 1999, 24, 59–90. [Google Scholar] [PubMed]
- The manual of the vaccine injury compensation program by Taiwan CDC (in Traditional Chinese) Available online:. Available online: https://www.cdc.gov.tw/vicp (accessed on 16 August 2024).
- Regulations governing collection and review of relief fund for victims of immunization, 2004 Available online:. Available online: https://law.moj.gov.tw/ENG/LawClass/LawAll.aspx?pcode=L0050021 (accessed on 16 August 2024).
- Taiwan Center of Disease Control. Available online: https://www.cdc.gov.tw/Category/Page/OM4QgMRlmzYg5UDLHlnXRw (accessed on 16 August 2024).
- Colditz GA, Berkey CS, Mosteller F, Brewer TF, Wilson ME, Burdick E, et al. The efficacy of bacillus Calmette-Guérin vaccination of newborns and infants in the prevention of tuberculosis: meta-analyses of the published literature. Pediatrics, 1995, 96, 29–35. [Google Scholar]
- Chan, P. , Huang, W., Wang, K., Ma, C., Lu, B., Lin, F., et al. The Active surveillance of BCG-related adverse events. Taiwan Epidemiology Bulletin 2012, 28, 13–22. [Google Scholar]
- Chiu N, Lin M, Lin W, et al. Mycobacterium bovis BCG–associated osteomyelitis/osteitis, Taiwan. Emerg Infect Dis 2015, 21, 539–40. [Google Scholar]
- Yang TL, Lee CM, Lee KL, et al. Clinical features of tuberculosis and Bacillus Calmette-Guérin (BCG) -associated adverse effects in children: A 12-year study. J Formos Med Assoc, 2021, 120, 443–51. [Google Scholar]
- Huang CY, Chiu NC, Chi H, Huang FY, Chang PH. Clinical manifestations, management, and outcomes of osteitis/osteomyelitis caused by Mycobacterium bovis Bacillus Calmette-Guérin in children: comparison by site(s) of affected bones. J Pediatr, 2019, 207, 97–102. [Google Scholar]
- Tsai YL, Chen YJ, Lai YC. Long-term follow-up of osteomyelitis caused by Bacille Calmette-Guérin vaccination in immuno-competent children. Sci Rep, 2020, 10, 4072. [Google Scholar]
- Wei Huang, Chiu N-C, Chi H, Huang FY, Huang CY. Inoculation Age of Bacillus Calmette-Guérin Tokyo-172 Strain and Vaccine-related Adverse Reactions in Taiwan Birth Cohort of 2012–2017. Clin Infect Dis, 2021, 73, e1554–9. [Google Scholar] [CrossRef]
- Taiwan Centers for Disease Control. Recommendations for BCG injection site (in traditional Chinese). Available online: https://www.cdc.gov.tw/Category/ListContent/4T8vDeSxOpmz0ILvF_D8Yw?uaid=QsqXMEzXYCw6Gp1n7YLSDA (accessed on 16 August 2024).
- Kim MK, Lee YK, Kim TE, Kong I, Yang HJ, Suh ES. Surveillance and compensation claims for adverse events following immunization from 2011 to 2016 in the Republic of Korea. Clin Exp Vaccine Res, 2017, 6, 146–55. [Google Scholar]
- Patel PK, Al-Rawahi B, Al-Jawari A, Al-Abaidani I, Al-Abri S. Surveillance of adverse events following immunization in Oman, 2006-2015. East Mediterr Health J, 2018, 24, 119–26. [Google Scholar]
- Guo B, Page A, Wang H, Taylor R, McIntyre P. Systematic review of reporting rates of adverse events following immunization: an international comparison of post-marketing surveillance programs with reference to China. Vaccine, 2013, 31, 603–17. [Google Scholar]
- Clothier HJ, Hosking L, Crawford NW, Russell M, Easton ML, Quinn JA, et al. Bacillus Calmette-Guérin (BCG) vaccine adverse events in Victoria, Australia: analysis of reports to an enhanced passive surveillance system. Drug Saf, 2015, 38, 79–86. [Google Scholar]
- Krysztopa-Grzybowska K, Paradowska-Stankiewicz I, Lutyńska A. The rate of adverse events following BCG vaccination in Poland. Przegl Epidemiol, 2012, 66, 465–9. [Google Scholar]
- Roh EJ, Lee YK, Lee MH, Kim MK, Kim TE, Lee SG, et al. Investigation of adverse events following bacille Calmette-Guérin immunization using immunization safety surveillance system in Korea Centers for Disease Control and Prevention. Clin Exp Vaccine Res, 2020, 9, 133–145. [Google Scholar]
- Thompson KM, Orenstein WA, Hinman AR. Performance of the United States Vaccine Injury Compensation Program (VICP): 1988-2019. Vaccine, 2020, 38, 2136–43. [Google Scholar]
- Ehrengut W, Allerdist H. Uber neurologische Komplikationen nach der Influenza-schutzimpfung [Neurological complications after influenza vaccination]. MMW Munch Med Wochenschr, 1977, 119, 705–10. [Google Scholar]
- Ehrengut, W. Guillain-Barré syndrome and influenza vaccine. Br Med J, 1977, 1, 1662–3. [Google Scholar]
- Schonberger LB, Bregman DJ, Sullivan-Bolyai JZ, Keenlyside RA, Ziegler DW, Retailliau HF, et al. Guillain-Barre syndrome following vaccination in the National Influenza Immunization Program, United States, 1976-1977. Am J Epidemiol.
- Langmuir AD, Bregman DJ, Kurland LT, Nathanson N, Victor M. An epidemiologic and clinical evaluation of Guillain-Barré syndrome reported in association with the administration of swine influenza vaccines. Am J Epidemiol, 1984, 119, 841–79. [Google Scholar]
- Marks JS, Halpin TJ. Guillain-Barré syndrome in recipients of A/New Jersey influenza vaccine. JAMA, 2490.
- Martín Arias LH, Sanz R, Sáinz M, Treceño C, Carvajal A. Guillain-Barré syndrome and influenza vaccines: A meta-analysis. Vaccine, 2015, 33, 3773–78. [Google Scholar]
- Principi N, Esposito S. Vaccine-preventable diseases, vaccines, and Guillain-Barre syndrome. Vaccine, 2019, 37, 5544–50. [Google Scholar]
- Dieleman J, Romio S, Johansen K, Weibel D, Bonhoeffer J, Sturkenboom M; VAESCO-GBS case-control study group. Guillain-Barre syndrome and adjuvanted pandemic influenza A (H1N1) 2009 vaccine: multinational case-control study in Europe. BMJ, 2011, 343, d3908. [Google Scholar]
- Salmon DA, Proschan M, Forshee R, Gargiullo P, Bleser W, Burwen DR, et al. ; H1N1 GBS meta-analysis working group. Association between Guillain-Barré syndrome and influenza A (H1N1) 2009 monovalent inactivated vaccines in the USA: a meta-analysis. Lancet, 2013, 381, 1461–8. [Google Scholar]
- Petráš M, Lesná IK, Dáňová J, Čelko AM. Is an increased risk of developing Guillain-Barré syndrome associated with seasonal influenza vaccination? A systematic review and meta-analysis. Vaccines, 2020, 8, 150. [Google Scholar]
- Vellozzi C, Iqbal S, Broder K. Guillain-Barre syndrome, influenza, and influenza vaccination: the epidemiologic evidence. Clin Infect Dis, 2014, 58, 1149–55. [Google Scholar]
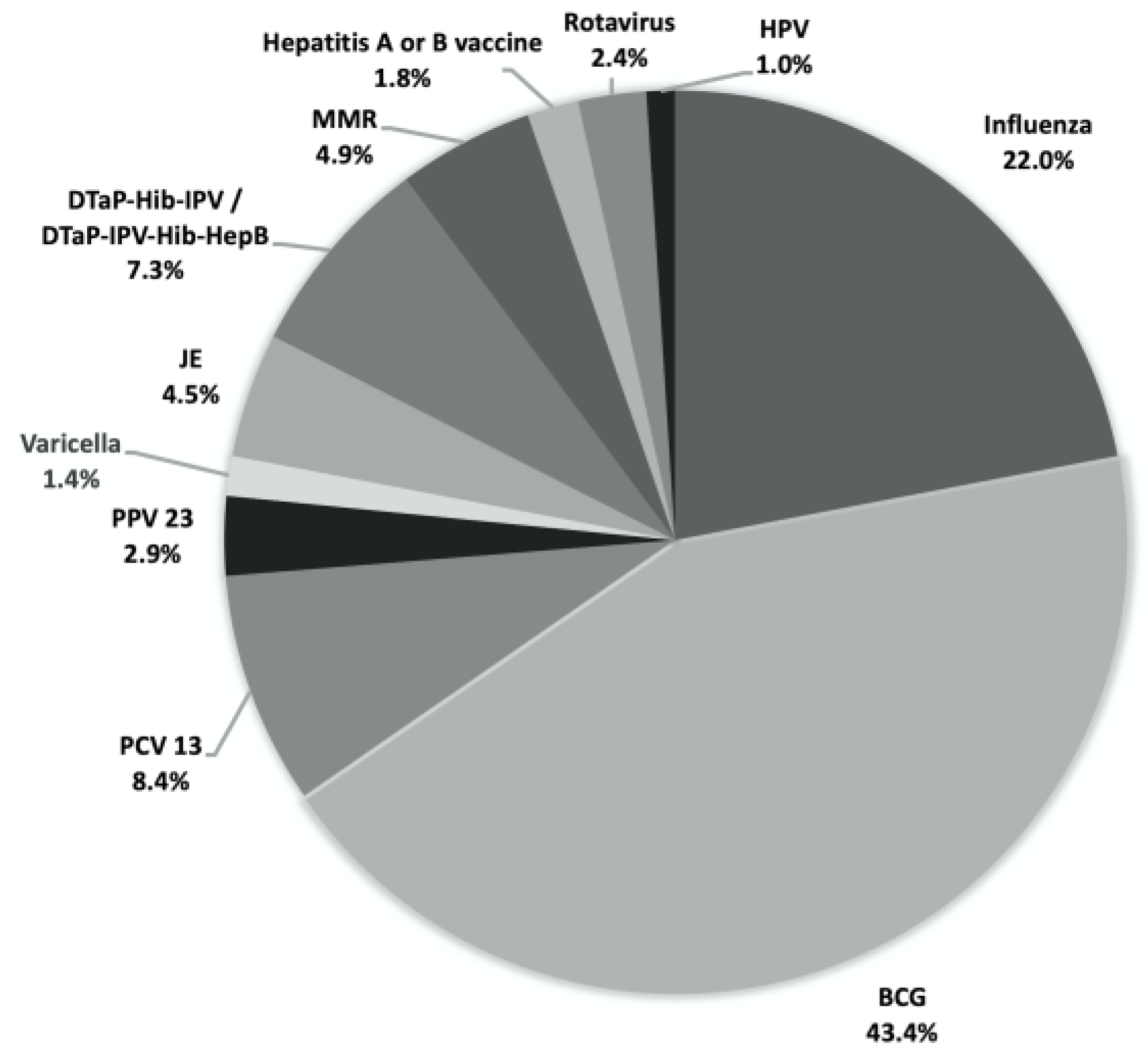
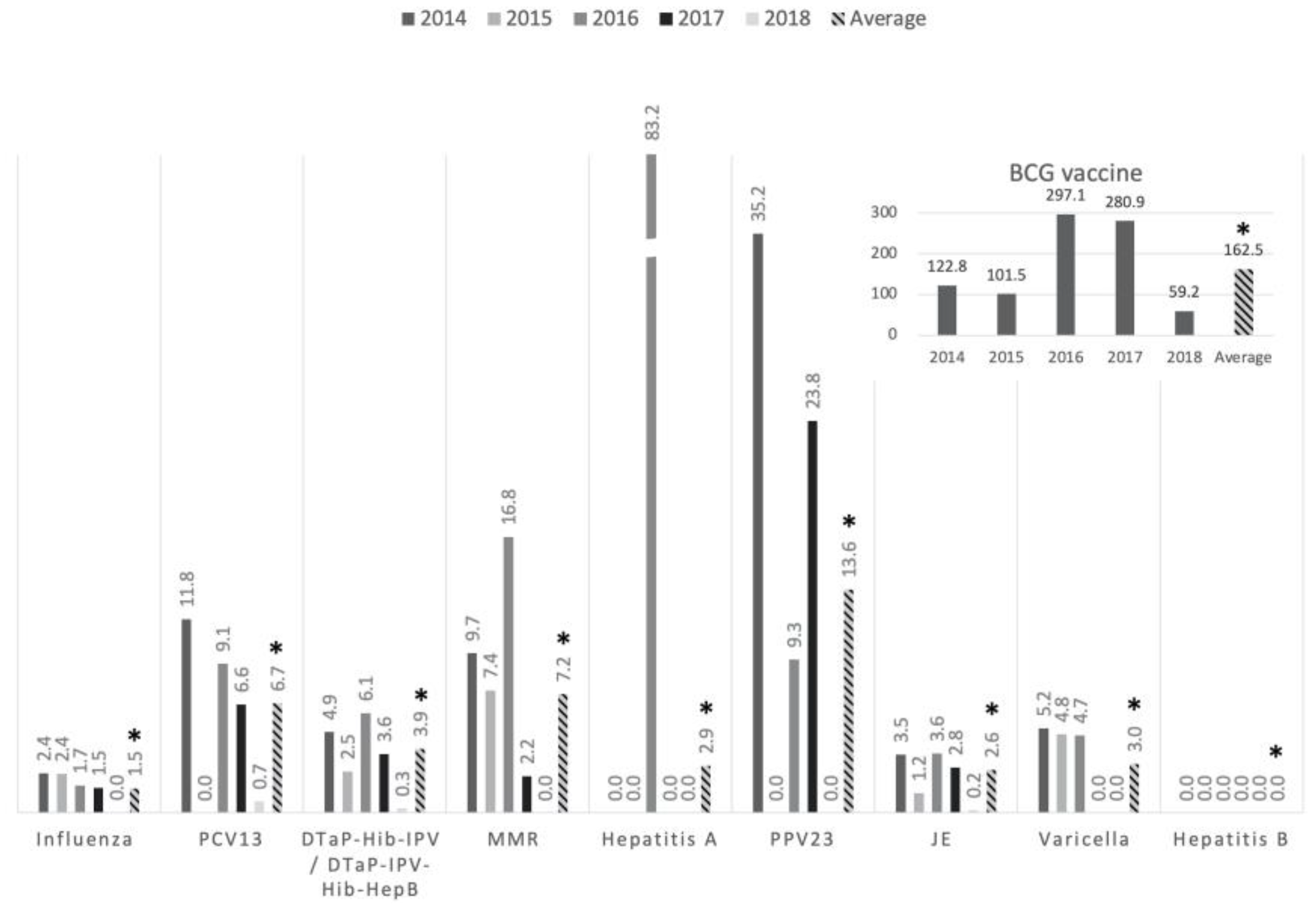
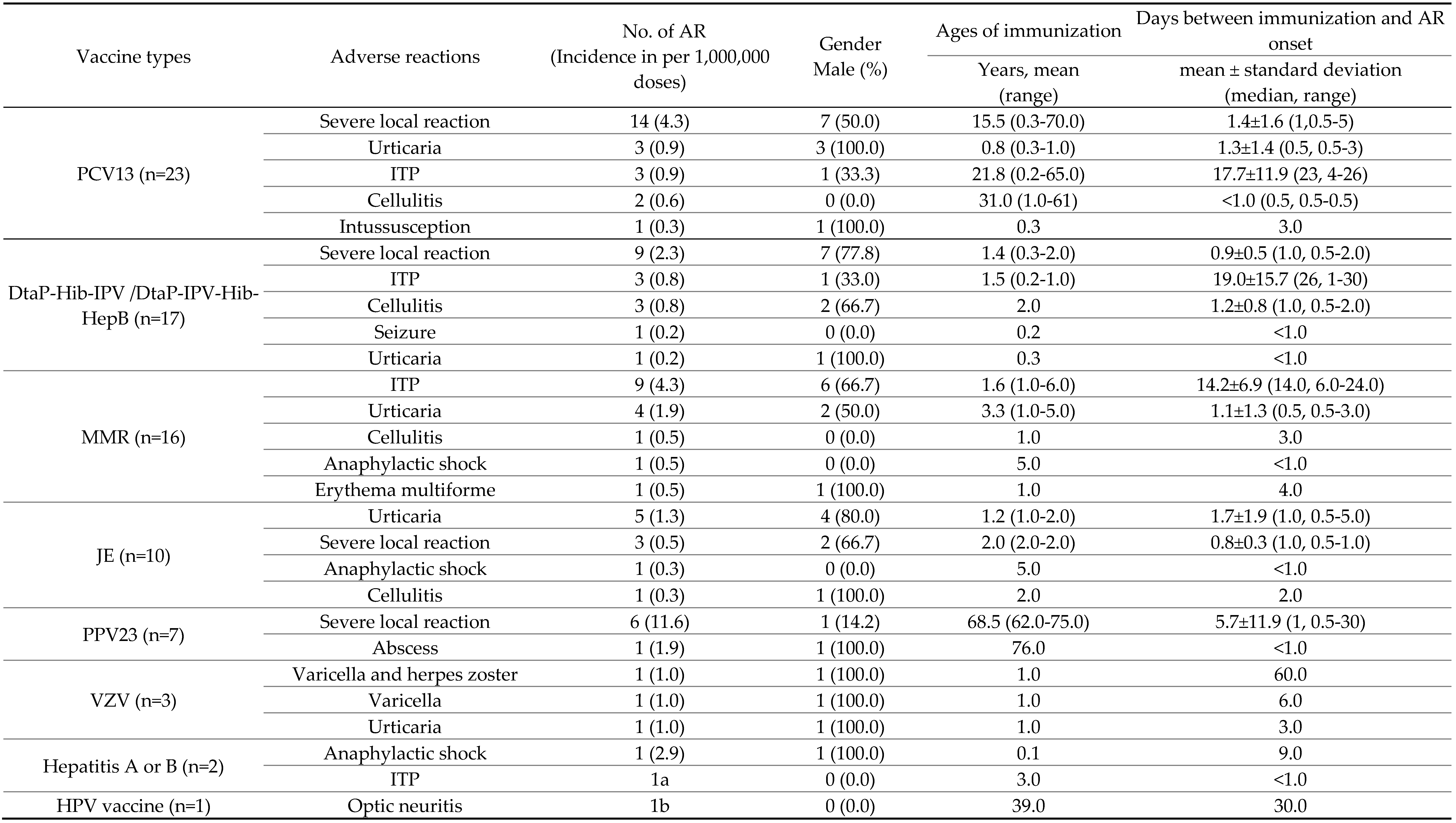
| Vaccine type | No. of events (%) | |||
| Adverse reactions | Unrelated events | |||
| Related | Indeterminate | Total | ||
| BCG vaccine (n=213) | 188 (88.3) | 18 (8.5) | 206 (96.7) | 7 (3.3) |
| Influenza vaccine (n=108) | 4 (3.7) | 35 (32.4) | 39 (36.1) | 69 (63.9) |
| PCV13 (n=41) | 14 (34.1) | 9 (22.0) | 23 (56.1) | 18 (43.9) |
| DTaP-Hib-IPV/ DTaP-IPV-Hib-HepB vaccine (n=36) | 12 (33.3) | 5 (13.9) | 17 (47.2) | 19 (52.8) |
| MMR vaccine (n=24) | 1 (4.2) | 15 (62.5) | 16 (66.7) | 8 (33.3) |
| JE vaccine (n=22) | 5 (22.7) | 5 (22.7) | 10 (45.5) | 12 (54.5) |
| PPV23 (n=14) | 7 (50.0) | 0 (0) | 7 (50.0) | 7 (50.0) |
| Rotavirus vaccine (n=12) | 0 (0) | 3a (25.0) | 3 (25.0) | 9 (75.0) |
| Hepatitis A or B vaccine (n=9) | 0 (0) | 2 (22.2) | 2 (22.2) | 7 (77.8) |
| Varicella vaccine (n=7) | 0 (0) | 3 (42.9) | 3 (42.9) | 4 (57.1) |
| HPV vaccine (n=5) | 0 (0) | 1 (20.0) | 1 (20.0) | 4 (80.0) |
| Total claims (n=491) | 231 (47.0) | 96 (19.6) | 327 (66.6) | 164 (33.4) |
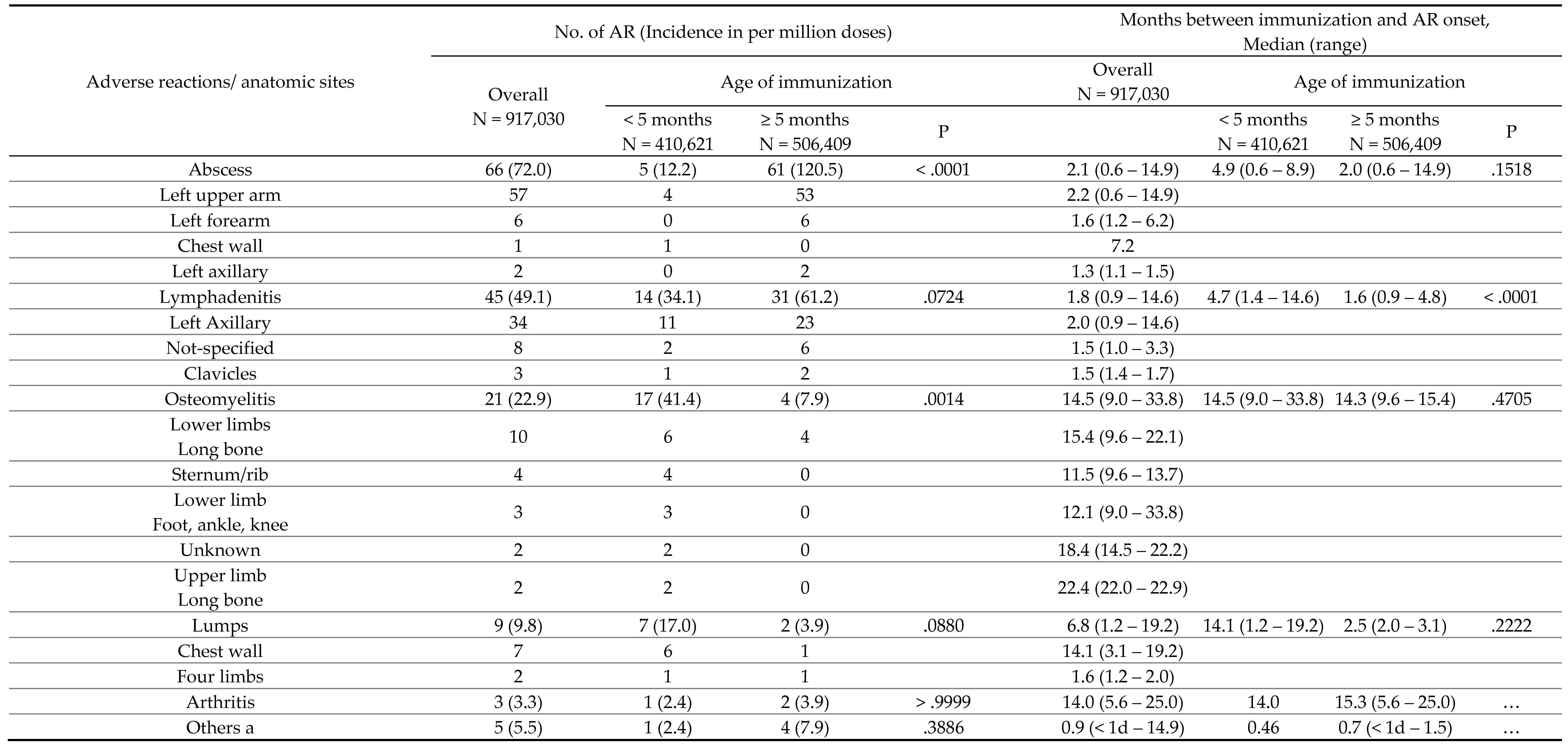 |
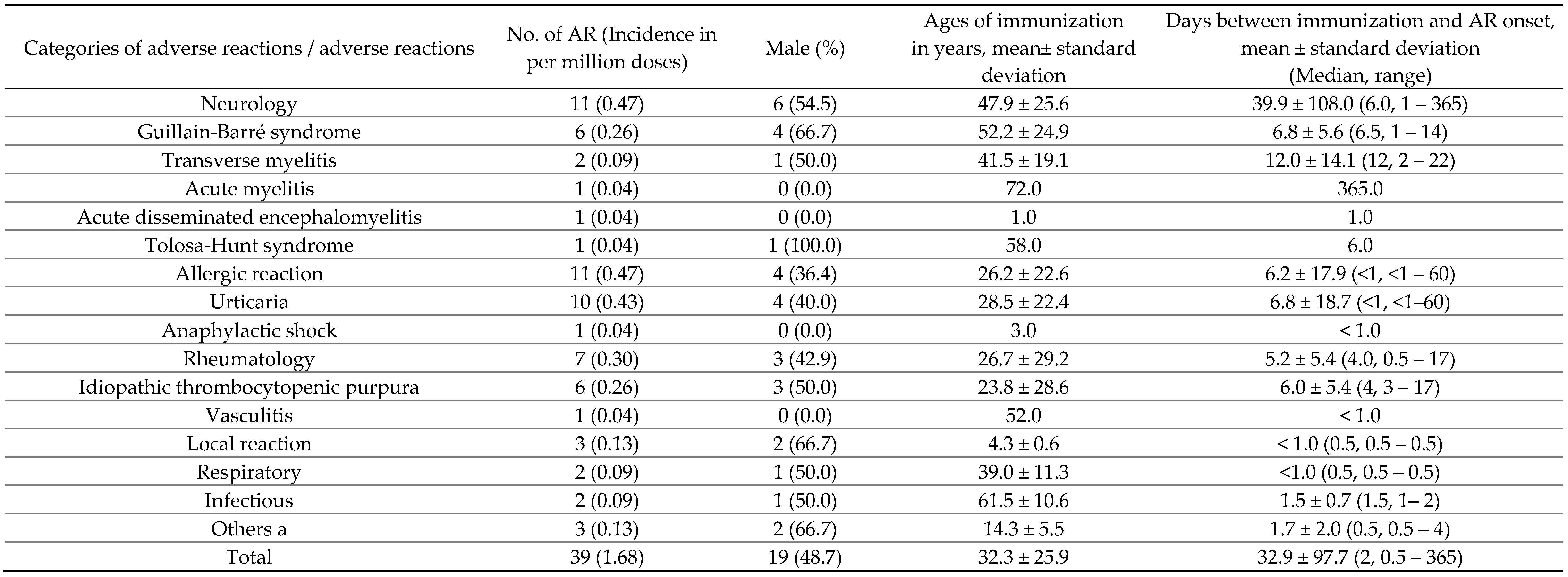 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




