Submitted:
15 August 2024
Posted:
15 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.2. Modification of ACs
2.3. SEM-EDX
2.4. Fourier Transform Infrared Spectroscopy (FTIR)
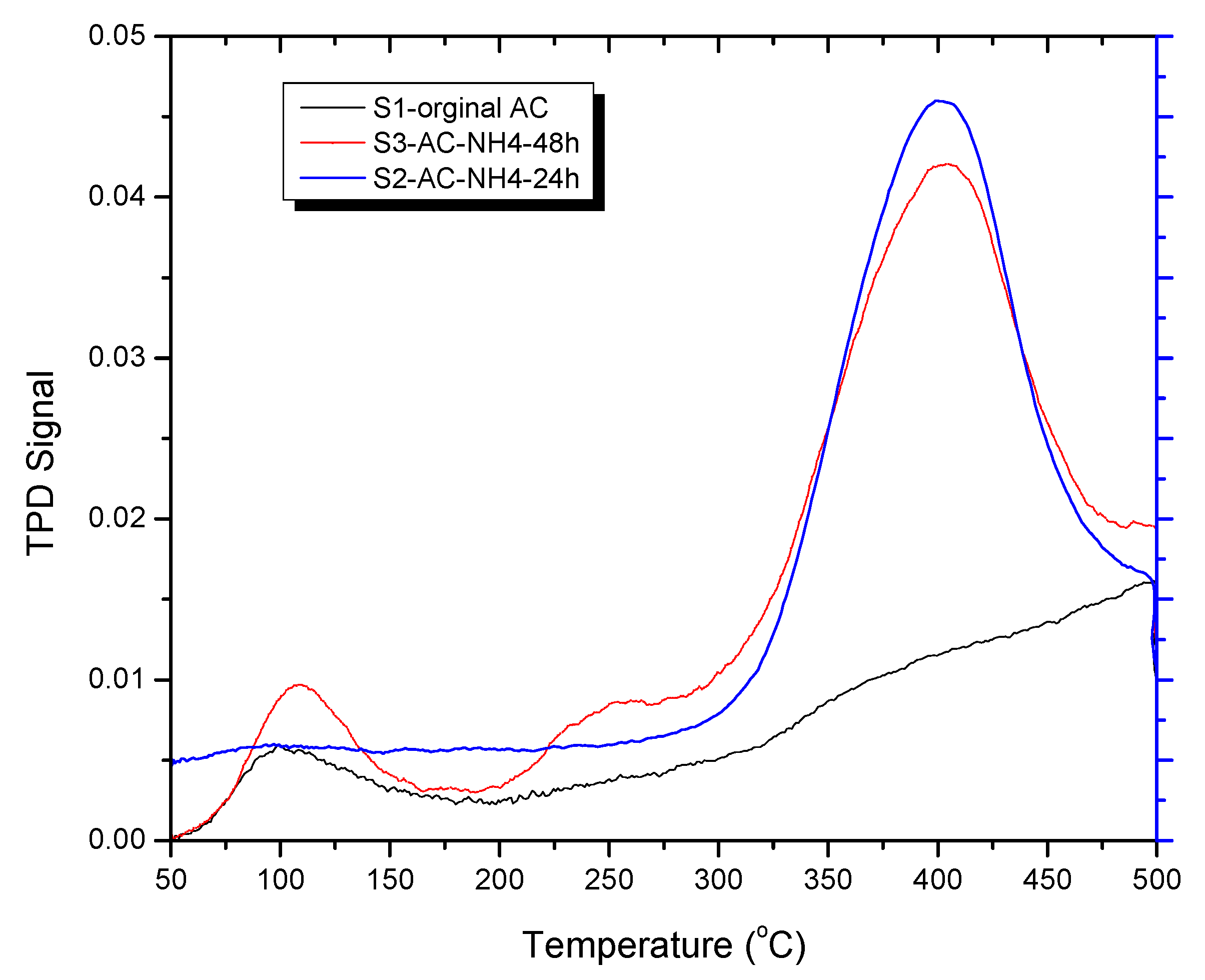
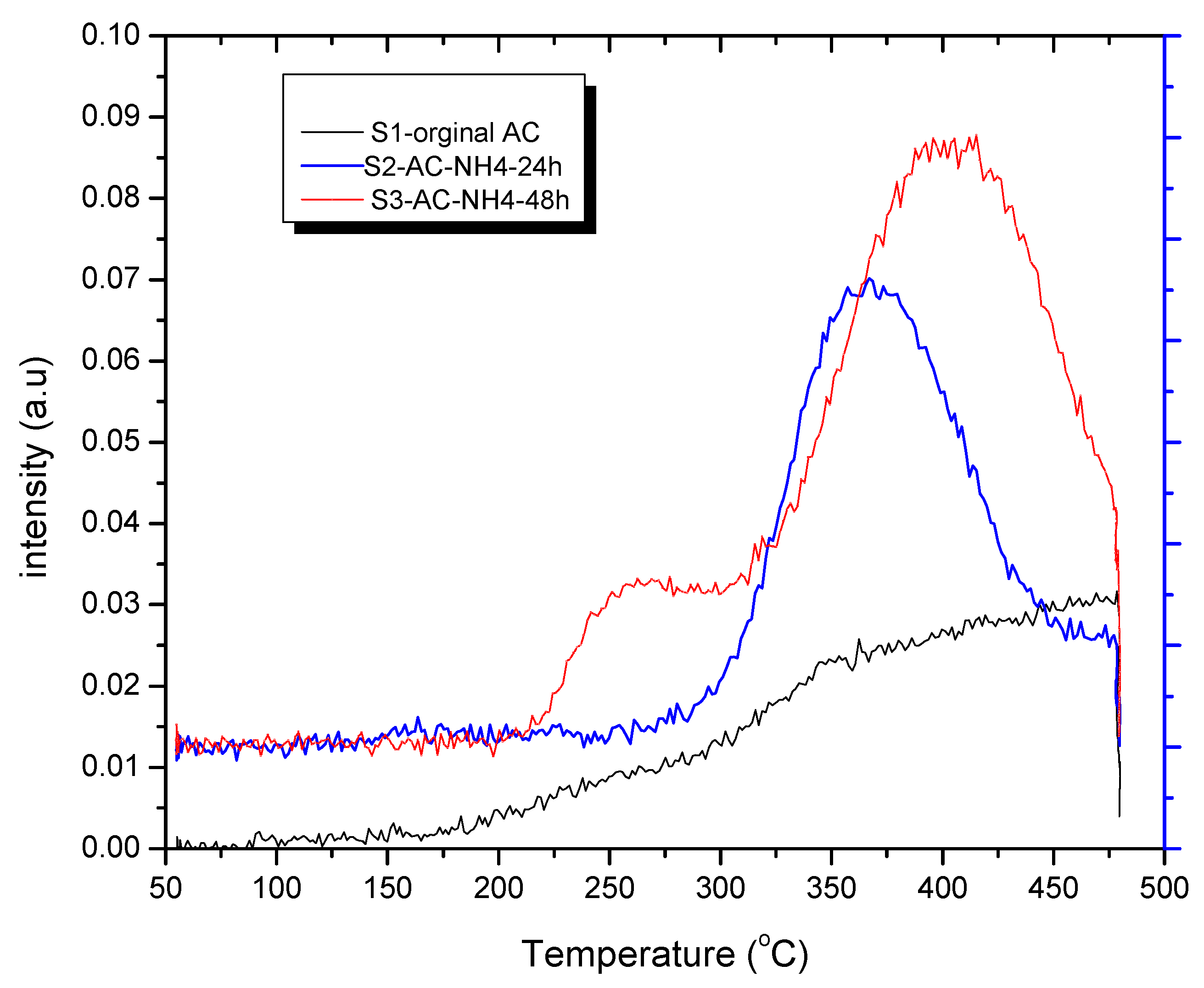
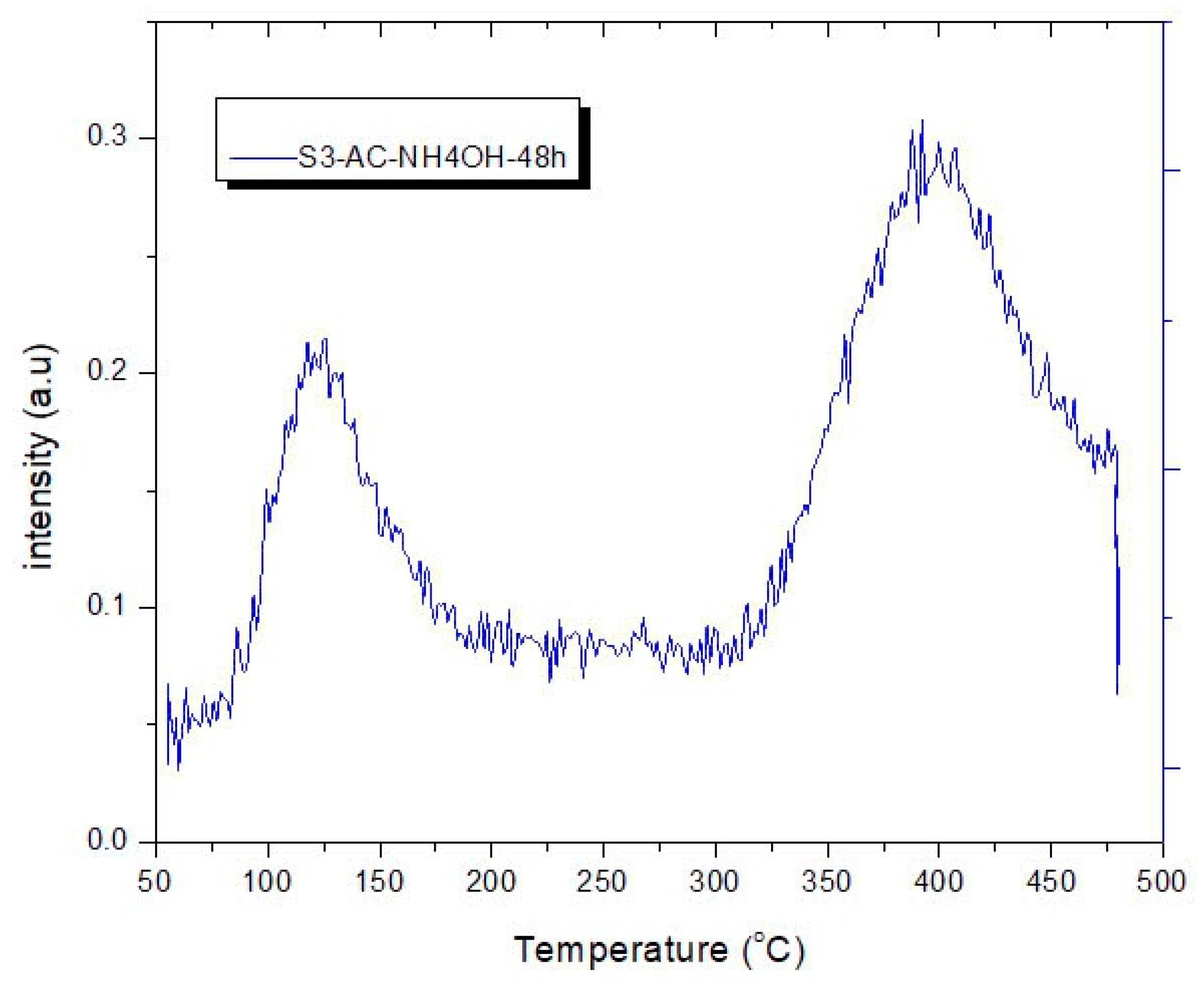
2.5. Temperature Programmed Desorption of CO2 on ACs
3. Results and Discussions
3.1. Impact on Morphology and Composition of Activated Carbons
3.2. Impact on Functional Group of Activated Carbons
3.3. Impact on CO2 Adsorption Ability
3.4. CO2 Temperature-Programmed Desorption (CO2-TPD) Experimental Results
4. Conclusions and Outlooks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mac Dowell, N.; Fennell, P.S.; Shah, N.; Maitland, G.C. The role of CO2 capture and utilization in mitigating climate change. Nat. Clim. Change 2017, 7, 243–249. [Google Scholar] [CrossRef]
- Bandh, S.A; Malla, F.A.; Hoang, T.D.; Qayoom, I.; Mohi-Ud-Din, H.; Bashir, S.; Betts; R.; Le, T.T.; Nguyen Le, D.T.; Linh Le, N.V.; Le, H.C. Track to reach net-zero: Progress and pitfalls. Energy Environ 2024, 0958305X241260793. [Google Scholar] [CrossRef]
- Hoang, T.D.; Nghiem, N. Recent developments and current status of commercial production of fuel ethanol. Fermentation 2021, 7, 314. [Google Scholar] [CrossRef]
- Sezali, N.A.A.; Ong, H.L.; Villagracia, A.R.; Hoang, T.D. Bio-based nanomaterials for energy application: A review. Vietnam J. Chem. 2024, 62, 1–12. [Google Scholar] [CrossRef]
- Malla, F.A.; Dung, T.; Bandh, S.A.; Malla, A.A.; Wani, S.A. Circular Economy to Decarbonize Electricity. In Renewable Energy in Circular Economy, 1st ed.; Bandh, S.A., Malla, F.A., Hoang, A.T. Eds, Eds.; Springer International Publishin: Cham, Switzerland, 2023; pp. 71–87. [Google Scholar]
- Nobre, L.C.; Teixeira, P.; Pinheiro, C.I.; Palavra, A.M.; Calvete, M.J.; Nieto de Castro, C.A.; Nobre, B.P. The Treatment of Natural Calcium Materials Using the Supercritical Antisolvent Method for CO2 Capture Applications. Processes 2024, 12, 425. [Google Scholar] [CrossRef]
- Hoang, T.D.; Bandh, S.A.; Malla, F.A.; Qayoom, I.; Bashir, S.; Peer, S.B.; Halog, A. Carbon-Based Synthesized Materials for CO2 Adsorption and Conversion: Its Potential for Carbon Recycling. Recycling 2023, 8, 53. [Google Scholar] [CrossRef]
- Azmi, N.Z.M.; Buthiyappan, A.; Raman, A.A.A.; Patah, M.F.A.; Sufian, S. Recent advances in biomass based activated carbon for carbon dioxide capture–A review. J. Ind. Eng. Chem. 2022, 116, 1–20. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, M.; Wang, H.; Li, Z. Enhancement of CO2 adsorption on high surface area activated carbon modified by N2, H2 and ammonia. J. Chem. Eng. 2010, 160, 571–577. [Google Scholar] [CrossRef]
- Pevida, C.; Plaza, M.G.; Arias, B.; Fermoso, J.; Rubiera, F.; Pis, J.J. Surface modification of activated carbons for CO2 capture. Appl. Surf. Sci. 2008, 254, 7165–7172. [Google Scholar] [CrossRef]
- Qasem, N.A.; Ben-Mansour, R. Adsorption breakthrough and cycling stability of carbon dioxide separation from CO2/N2/H2O mixture under ambient conditions using 13X and Mg-MOF-74. Appl. Energy. 2018, 230, 1093–1107. [Google Scholar] [CrossRef]
- Abuelnoor, N.; AlHajaj, A.; Khaleel, M.; Vega, L.F.; Abu-Zahra, M.R. Activated carbons from biomass-based sources for CO2 capture applications. Chemosphere 2021, 282, 131111. [Google Scholar] [CrossRef]
- Heidari, A.; Younesi, H.; Rashidi, A.; Ghoreyshi, A.A. Evaluation of CO2 adsorption with eucalyptus wood based activated carbon modified by ammonia solution through heat treatment. J. Chem. Eng. 2014, 254, 503–513. [Google Scholar] [CrossRef]
- Gholidoust, A.; Atkinson, J.D.; Hashisho, Z. Enhancing CO2 adsorption via amine-impregnated activated carbon from oil sands coke. Energy & Fuels. 2017, 31, 1756–1763. [Google Scholar]
- Abd, A.A.; Othman, M.R.; Kim, J. A review on application of activated carbons for carbon dioxide capture: present performance, preparation, and surface modification for further improvement. Environ Sci Pollut Res. 2021, 28, 43329–43364. [Google Scholar] [CrossRef] [PubMed]
- Przepiórski, J.; Skrodzewicz, M.; Morawski, A.W. High temperature ammonia treatment of activated carbon for enhancement of CO2 adsorption. Appl. Surf. Sci. 2004, 225, 235–242. [Google Scholar] [CrossRef]
- Feng, J.Y.; Wang, R.; Thakur, K.; Ni, Z.J.; Zhu, Y.Y.; Hu, F.; Zhang, J.G.; Wei, Z.J. Evolution of okara from waste to value added food ingredient: An account of its bio-valorization for improved nutritional and functional effects. Trends Food Sci Technol. 2021, 116, 669–680. [Google Scholar] [CrossRef]
- Hoang, T.D.; Liu, Y.; Le, M.T. Synthesis and Characterization of Biochars and Activated Carbons Derived from Various Biomasses. Sustainability 2024, 16, 5495. [Google Scholar] [CrossRef]
- Abi Bianasari, A.; Khaled, M.S.; Hoang, T.D.; Reza, M.S.; Bakar, M.S.A.; Azad, A.K. Influence of combined catalysts on the catalytic pyrolysis process of biomass: A systematic literature review. Energy Convers. Manag 2024, 309, 118437. [Google Scholar] [CrossRef]
- Adame-Pereira, M.; Durán-Valle, C.J.; Fernández-González, C. Hydrothermal Carbon Coating of an Activated Carbon—A New Adsorbent. Molecules 2023, 28, 4769. [Google Scholar] [CrossRef]
- Gupta, V.K.; Singh, L.P.; Chaudhary, M.; Kushwaha, S. A novel approach to develop activated carbon by an ingenious hydrothermal treatment methodology using Phyllanthus emblica fruit stone. J. Clean. Prod 2021, 288, 125643. [Google Scholar]
- Hoang, T.D.; Van Anh, N.; Yusuf, M.; Ali S. A, M.; Subramanian, Y.; Hoang Nam, N.; Minh Ky, N.; Le, V.G.; Thi Thanh Huyen, N.; Abi Bianasari, A. and K Azad, A. Valorization of Agriculture Residues into Value-Added Products: A Comprehensive Review of Recent Studies. Chem. Rec. 2024, p.e202300333.
- Kourtelesis, M.; Kousi, K.; Kondarides, D.I. CO2 hydrogenation to methanol over La2O3-promoted CuO/ZnO/Al2O3 catalysts: a kinetic and mechanistic study. Catalysts 2020, 10, 183. [Google Scholar] [CrossRef]
- Ghimbeu, C.M.; Gadiou, R., Dentzer, J.; Schwartz, D. and Vix-Guterl, C. Influence of surface chemistry on the adsorption of oxygenated hydrocarbons on activated carbons. Langmuir 2010, 26, 18824–18833. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.H. and Ruckenstein, E. Temperature-programmed desorption of CO adsorbed on NiO/MgO. Journal of Catalysis 1996, 163, 306–311. [Google Scholar] [CrossRef]
- Liu, C.J.; Lobban, L.L. and Mallinson, R.G. Experimental investigations on the interaction between plasmas and catalyst for plasma catalytic methane conversion (PCMC) over zeolites. In Studies in surface science and catalysis 2nd ed,; Liu, C.J., Lobban, L.L., Eds.; Mallinson, R.G. Elsevier: Louvain-la-Neuve, Belgium, 1998. [Google Scholar]
- Almoneef, M.M.; Jedli, H.; Mbarek, M. Experimental study of CO2 adsorption using activated carbon. Mater. Res. Express 2021, 8, 065602. [Google Scholar] [CrossRef]
- Ghouma, I.; Jeguirim, M.; Sager, U.; Limousy, L.; Bennici, S.; Däuber, E.; Asbach, C.; Ligotski, R.; Schmidt, F.; Ouederni, A. The potential of activated carbon made of agro-industrial residues in NOx immissions abatement. Energies 2017, 10, 1508. [Google Scholar] [CrossRef]
- Amalina, F.; Abd Razak, A.S.; Krishnan, S.; Sulaiman, H.; Zularisam, A.W.; Nasrullah, M. Biochar production techniques utilizing biomass waste-derived materials and environmental applications–A review. Hazard. Mater. 2022, 7, 100134. [Google Scholar] [CrossRef]
- Abdullah, A.H.D.; Chalimah, S.; Primadona, I. and Hanantyo, M.H.G. Physical and chemical properties of corn, cassava, and potato starchs. In IOP Conference Series: Earth and Environmental Science Abdullah, A.H.D.; Chalimah, S., Ed.; Primadona, I. and Hanantyo, M.H.G. 2nd ed,; IOP Publishing: Briston, UK, 2018. [Google Scholar]
- Li, J.; Wang, Z.; Yang, X.; Hu, L.; Liu, Y. and Wang, C. Evaluate the pyrolysis pathway of glycine and glycylglycine by TG–FTIR. J Anal Appl Pyrol. 2007, 80(1), pp.247-253.
- Jiménez-García, E.A.; Pérez-Huertas, S.; Pérez, A.; Calero, M.; Blázquez, G. Recycling PVC Waste into CO2 Adsorbents: Optimizing Pyrolysis Valorization with Neuro-Fuzzy Models. Processes 2024, 12, 431. [Google Scholar] [CrossRef]
- Moyo, M.; Nyamhere, G.; Sebata, E.; Guyo, U. Kinetic and equilibrium modelling of lead sorption from aqueous solution by activated carbon from goat dung. Desalin Water Treat 2016, 57, 765–775. [Google Scholar] [CrossRef]
- Wang, B.; Côté, A.P.; Furukawa, H.; O’Keeffe, M.; Yaghi, O.M. Colossal cages in zeolitic imidazolate frameworks as selective carbon dioxide reservoirs. Nature 2008, 453, 207–211. [Google Scholar] [CrossRef]
- Goetz, V.; Pupier, O.; Guillot, A. Carbon dioxide-methane mixture adsorption on activated carbon. Adsorption 2006, 12, 55–63. [Google Scholar] [CrossRef]
- González Plaza, M.; González García, A.S.; Pevida García, C.; Pis Martínez, J.J.; Rubiera González, F. Valorisation of spent coffee grounds as CO2 adsorbents for postcombustion capture applications. Appl. Energy. 2012, 99, 272–279. [Google Scholar] [CrossRef]
- Li, D.; Ma, T., Zhang, R.; Tian, Y. and Qiao, Y. Preparation of porous carbons with high low-pressure CO2 uptake by KOH activation of rice husk char. Fuel 2015, 139, 68–70. [Google Scholar] [CrossRef]
- Serafin, J.; Narkiewicz, U.; Morawski, A.W.; Wróbel, R.J.; Michalkiewicz, B. Highly microporous activated carbons from biomass for CO2 capture and effective micropores at different conditions. J Co2 Util 2017, 18, 73–79. [Google Scholar] [CrossRef]
- Wang, R.; Wang, P.; Yan, X.; Lang, J.; Peng, C.; Xue, Q. Promising porous carbon derived from celtuce leaves with outstanding supercapacitance and CO2 capture performance. ACS Appl. Mater. Interfaces. 2012, 4, 5800–5806. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Tian, Y.; Li, L.; Li, J.; Zhang, H. Production of highly microporous carbons with large CO 2 uptakes at atmospheric pressure by KOH activation of peanut shell char. J. Porous Mater. 2015, 22, 1581–1588. [Google Scholar] [CrossRef]
- Fakeeha, A.H.; Kasim, S.O.; Ibrahim, A.A.; Abasaeed, A.E.; Al-Fatesh, A.S. Influence of nature support on methane and CO2 conversion in a dry reforming reaction over nickel-supported catalysts. Materials 2019, 12, 1777. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Wang, J.; Zhu, D.; Song, G.; Yang, H.; Chen, L.; Sun, L.; Yang, S.; Guan, H.; Xie, X. Adsorption characteristics of gas molecules (H2O, CO2, CO, CH4, and H2) on CaO-based catalysts during biomass thermal conversion with in situ CO2 capture. Catalysts 2019, 9, 757. [Google Scholar] [CrossRef]
- Li, N.; Ma, X.; Zha, Q.; Kim, K.; Chen, Y.; Song, C. Maximizing the number of oxygen-containing functional groups on activated carbon by using ammonium persulfate and improving the temperature-programmed desorption characterization of carbon surface chemistry. Carbon 2011, 49, 5002–5013. [Google Scholar] [CrossRef]
- Thommes, M.; Morlay, C.; Ahmad, R.; Joly, J.P. Assessing surface chemistry and pore structure of active carbons by a combination of physisorption (H2O, Ar, N2, CO2), XPS and TPD-MS. Adsorption 2011, 17, 653–661. [Google Scholar] [CrossRef]
- Ghimbeu, C.M.; Gadiou, R.; Dentzer, J.; Vidal, L.; Vix-Guterl, C. A TPD-MS study of the adsorption of ethanol/cyclohexane mixture on activated carbons. Adsorption 2011, 17, 227–233. [Google Scholar] [CrossRef]

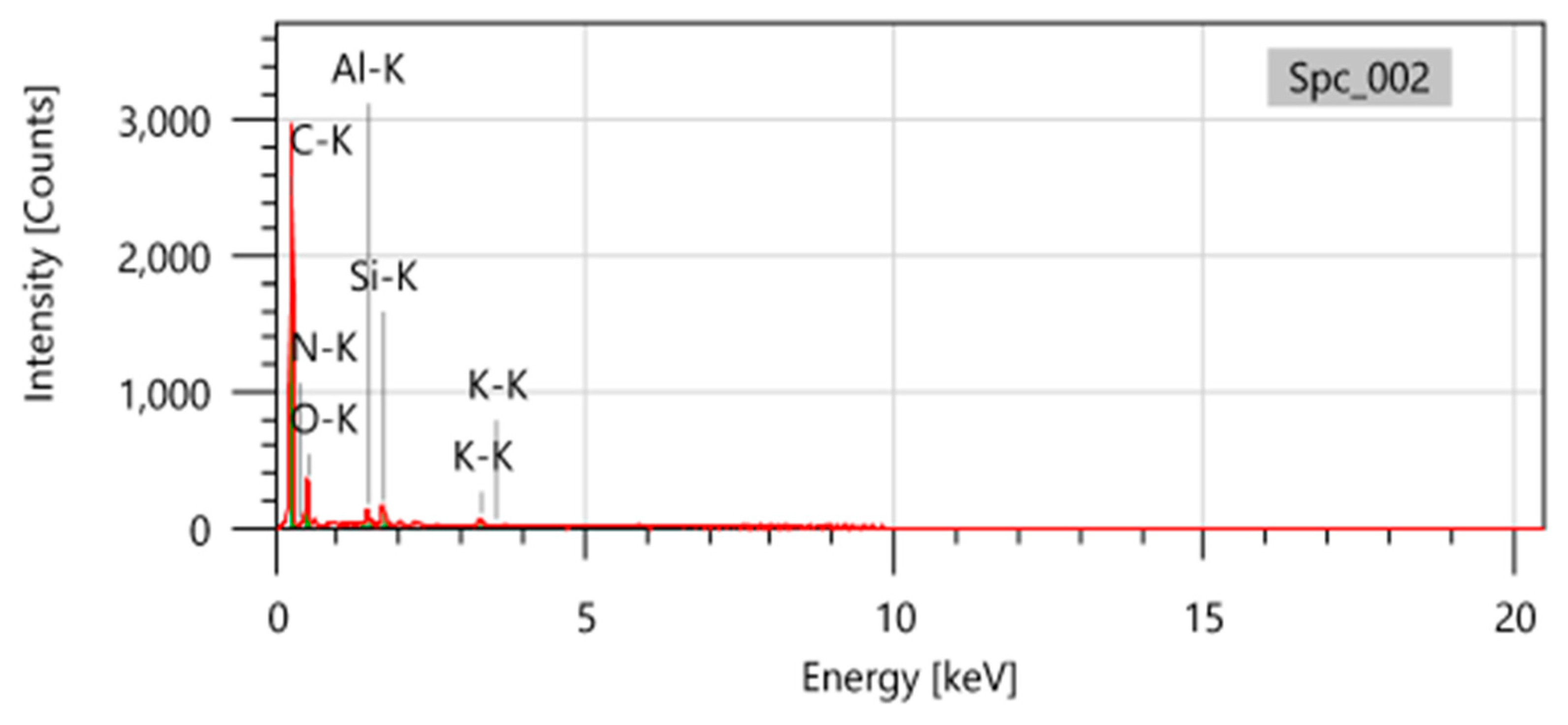

| Elements | S1 (Original AC) |
S2 (AC-NH4OH-24h) |
S3 (AC-NH4OH-48h) |
S4 (AC-NH4OH-72h) |
||||
|---|---|---|---|---|---|---|---|---|
| Mass% | Atom% | Mass% | Atom % | Mass% | Atom% | Mass% | Atom% | |
| C | 88.39 | 95.63 | 66.64 | 74.57 | 71.25 | 78.29 | 71.30 | 87.74 |
| Al | 1.16 | 0.56 | 2.08 | 1.04 | 0.97 | 0.47 | 2.06 | 1.13 |
| Si | 1.81 | 0.84 | 3.23 | 1.55 | ||||
| Cl | 3.05 | 1.12 | ||||||
| K | 5.59 | 1.86 | 2.48 | 0.85 | 4.90 | 1.65 | 25.07 | 9.48 |
| N | 4.34 | 4.17 | 6.05 | 5.70 | 1.58 | 1.16 | ||
| O | 21.22 | 17.83 | 16.82 | 13.88 | ||||
| Total | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| C/N atom ratio | 0 | 17.88 | 13.74 | 75.63 | ||||
| Adsorbents | Sample code | Temperature (K) | Impregnation method and agent | CO2 uptake (mmol/g) | References |
|---|---|---|---|---|---|
| Okara | S1-Original/raw or unmodified AC | 298 | Chemical, wet, NH4OH | 0.24 | This work |
| Crystallized materials | ZIF-100 | 298 | - | 1.05 | [34] |
| Okara | S4-AC-NH4OH-72h | 298 | Chemical, wet, NH4OH | 1.26 | This work |
| Commercial AC | Norit RB2 | 298 | - | 1.5 | [35] |
| Okara | S2-AC-NH4OH-24h | 298 | Chemical, wet, NH4OH | 1.78 | This work |
| Okara | S3-AC-NH4OH-48h | 298 | Chemical, wet, NH4OH | 2.24 | This work |
| Zeolite-based adsorbents | 13X | 298 | - | 2.27 | [11] |
| Commercial ACs | Commercial ACs | 298 | Chemical, wet, NH4OH | 2.92 | [9] |
| Coffee Grounds | - | 298 | Chemical, wet, KOH | 3.00 | [36] |
| Eucalyptus wood | - | 298 | Chemical, H3PO4 | 3.22 | [13] |
| Rice Husk Char | - | 298 | Chemical, wet, KOH | 3.71 | [37] |
| Carrot Peels | - | 298 | Chemical, wet, KOH | 4.18 | [38] |
| Celtuce Leaves | - | 298 | Chemical, wet, KOH | 4.36 | [39] |
| Peanut Shell Char | - | 298 | Chemical, wet, KOH | 4.41 | [40] |
| MOF | Mg-MOF-74 | 298 | - | 5.77 | [11] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




